ASTM E918-19
(Practice)Standard Practice for Determining Limits of Flammability of Chemicals at Elevated Temperature and Pressure
Standard Practice for Determining Limits of Flammability of Chemicals at Elevated Temperature and Pressure
SIGNIFICANCE AND USE
5.1 Knowledge of flammable limits at elevated temperatures and pressures is needed for safe and economical operation of some chemical processes. This information may be needed in order to start up a reactor without passing through a flammable range, to operate the reactor safely and economically, or to store or ship the product safely.
5.2 Limits of flammability data obtained in relatively clean vessels must be carefully interpreted and may not always be applicable to industrial conditions. Surface effects due to carbon deposits and other materials can significantly affect limits of flammability, especially in the fuel-rich region. Refer to Bulletin 503 and Bulletin 627.
SCOPE
1.1 This practice covers the determination of the lower and upper concentration limits of flammability of combustible vapor-oxidant mixtures at temperatures up to 200°C and initial pressures up to as much as 1.38 MPa (200 psia). This practice is limited to mixtures which would have explosion pressures less than 13.79 MPa (2000 psia).
1.2 This practice should be used to measure and describe the properties of materials, products, or assemblies in response to heat and flame under controlled laboratory conditions and should not be used to describe or appraise the fire hazard or fire risk of materials, products, or assemblies under actual fire conditions. However, results of this test may be used as elements of a fire risk assessment which takes into account all of the factors which are pertinent to an assessment of the fire hazard of a particular end use.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2019
- Technical Committee
- E27 - Hazard Potential of Chemicals
- Drafting Committee
- E27.04 - Flammability and Ignitability of Chemicals
Relations
- Effective Date
- 01-Dec-2019
- Effective Date
- 01-Jul-2019
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Oct-2009
- Effective Date
- 01-Jan-2007
- Effective Date
- 01-Jun-2004
- Effective Date
- 10-Oct-2001
- Effective Date
- 10-Oct-2001
- Effective Date
- 10-Oct-2001
- Effective Date
- 01-Dec-2019
Overview
ASTM E918-19: Standard Practice for Determining Limits of Flammability of Chemicals at Elevated Temperature and Pressure provides guidelines for determining the lower and upper concentration limits of flammability for combustible vapor-oxidant mixtures. This practice covers temperatures up to 200°C and initial pressures up to 1.38 MPa (200 psia), and it applies to mixtures with explosion pressures below 13.79 MPa (2000 psia). The standard is essential in the chemical industry for identifying safe operating, storage, and transportation conditions for flammable chemicals, especially under elevated temperature and pressure.
Key Topics
- Flammability Limits: Defines the lower flammable limit (LFL) and upper flammable limit (UFL) for different vapor-oxidant mixtures.
- High Temperature and Pressure Testing: Specifies how tests are performed up to the stated temperature and pressure ratings, emphasizing laboratory control to ensure repeatable results.
- Apparatus and Methods: Describes the equipment necessary for testing, including pressure vessels, ignition systems, pressure transducers, and controlled temperature chambers.
- Safety Precautions: Outlines critical safety measures, such as operator protection, explosion shielding, and proper venting, when conducting flammability tests at high pressures.
- Data Interpretation: Advises caution when applying laboratory results to industrial conditions, as factors like vessel cleanliness and surface deposits can impact flammability limits.
- Limitations: The practice is unsuitable for mixtures that react spontaneously or those with deflagration pressures exceeding apparatus ratings.
Applications
Proper knowledge of chemical flammability limits is vital to:
- Chemical Process Design: Ensures reactors and equipment are operated outside hazardous flammable ranges, reducing the risk of fire or explosion during start-up or shutdown.
- Safety Assessments: Supports risk assessments by providing reliable flammability data for safety analyses and accident prevention strategies.
- Storage and Transport: Defines safe conditions for the storage and shipment of chemicals and chemical products.
- Regulatory Compliance: Assists manufacturers and operators in meeting international safety and health regulations regarding fire and explosion hazards.
- Research and Development: Facilitates the evaluation of new chemicals and formulations for process safety reviews.
Related Standards
For comprehensive flammability characterization and consistent laboratory methods, users may refer to these related ASTM standards and technical bulletins:
- ASTM E681 - Test Method for Concentration Limits of Flammability of Chemicals (Vapors and Gases)
- ASTM E2079 - Test Methods for Limiting Oxygen (Oxidant) Concentration in Gases and Vapors
- Bureau of Mines Bulletin 503 - Limits of Flammability of Gases and Vapors
- Bureau of Mines Bulletin 627 - Flammability Characteristics of Combustible Gases and Vapors
These standards collectively contribute to a comprehensive framework for assessing the flammability hazards of chemicals in laboratory and industrial contexts.
Keywords: ASTM E918-19, flammability limits, chemical process safety, elevated temperature, elevated pressure, vapor-oxidant mixtures, laboratory testing, lower flammable limit (LFL), upper flammable limit (UFL), process safety, chemical storage, fire hazard assessment.
Buy Documents
ASTM E918-19 - Standard Practice for Determining Limits of Flammability of Chemicals at Elevated Temperature and Pressure
REDLINE ASTM E918-19 - Standard Practice for Determining Limits of Flammability of Chemicals at Elevated Temperature and Pressure
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.
Sponsored listings
Frequently Asked Questions
ASTM E918-19 is a standard published by ASTM International. Its full title is "Standard Practice for Determining Limits of Flammability of Chemicals at Elevated Temperature and Pressure". This standard covers: SIGNIFICANCE AND USE 5.1 Knowledge of flammable limits at elevated temperatures and pressures is needed for safe and economical operation of some chemical processes. This information may be needed in order to start up a reactor without passing through a flammable range, to operate the reactor safely and economically, or to store or ship the product safely. 5.2 Limits of flammability data obtained in relatively clean vessels must be carefully interpreted and may not always be applicable to industrial conditions. Surface effects due to carbon deposits and other materials can significantly affect limits of flammability, especially in the fuel-rich region. Refer to Bulletin 503 and Bulletin 627. SCOPE 1.1 This practice covers the determination of the lower and upper concentration limits of flammability of combustible vapor-oxidant mixtures at temperatures up to 200°C and initial pressures up to as much as 1.38 MPa (200 psia). This practice is limited to mixtures which would have explosion pressures less than 13.79 MPa (2000 psia). 1.2 This practice should be used to measure and describe the properties of materials, products, or assemblies in response to heat and flame under controlled laboratory conditions and should not be used to describe or appraise the fire hazard or fire risk of materials, products, or assemblies under actual fire conditions. However, results of this test may be used as elements of a fire risk assessment which takes into account all of the factors which are pertinent to an assessment of the fire hazard of a particular end use. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Knowledge of flammable limits at elevated temperatures and pressures is needed for safe and economical operation of some chemical processes. This information may be needed in order to start up a reactor without passing through a flammable range, to operate the reactor safely and economically, or to store or ship the product safely. 5.2 Limits of flammability data obtained in relatively clean vessels must be carefully interpreted and may not always be applicable to industrial conditions. Surface effects due to carbon deposits and other materials can significantly affect limits of flammability, especially in the fuel-rich region. Refer to Bulletin 503 and Bulletin 627. SCOPE 1.1 This practice covers the determination of the lower and upper concentration limits of flammability of combustible vapor-oxidant mixtures at temperatures up to 200°C and initial pressures up to as much as 1.38 MPa (200 psia). This practice is limited to mixtures which would have explosion pressures less than 13.79 MPa (2000 psia). 1.2 This practice should be used to measure and describe the properties of materials, products, or assemblies in response to heat and flame under controlled laboratory conditions and should not be used to describe or appraise the fire hazard or fire risk of materials, products, or assemblies under actual fire conditions. However, results of this test may be used as elements of a fire risk assessment which takes into account all of the factors which are pertinent to an assessment of the fire hazard of a particular end use. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E918-19 is classified under the following ICS (International Classification for Standards) categories: 13.220.40 - Ignitability and burning behaviour of materials and products; 71.100.01 - Products of the chemical industry in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E918-19 has the following relationships with other standards: It is inter standard links to ASTM E918-83(2011), ASTM E2079-19, ASTM E2079-07(2013), ASTM E681-09, ASTM E2079-07, ASTM E681-04, ASTM E681-01, ASTM E2079-00, ASTM E2079-01, ASTM E1445-08(2015). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E918-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E918 − 19
Standard Practice for
Determining Limits of Flammability of Chemicals at Elevated
Temperature and Pressure
This standard is issued under the fixed designation E918; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E2079 Test Methods for Limiting Oxygen (Oxidant) Con-
centration in Gases and Vapors
1.1 This practice covers the determination of the lower and
2.2 Other Documents:
upper concentration limits of flammability of combustible
Bulletin 503 Bureau of Mines, “Limits of Flammability of
vapor-oxidant mixtures at temperatures up to 200°C and initial
Gases and Vapors,” NTIS AD701575
pressures up to as much as 1.38 MPa (200 psia). This practice
Bulletin 627 Bureau of Mines, “Flammability Characteris-
is limited to mixtures which would have explosion pressures
tics of Combustible Gases andVapors,” NTISAD701576
less than 13.79 MPa (2000 psia).
1.2 This practice should be used to measure and describe
3. Terminology
the properties of materials, products, or assemblies in response
3.1 Definitions:
to heat and flame under controlled laboratory conditions and
3.1.1 lower limit of flammability or lower flammable limit
should not be used to describe or appraise the fire hazard or
(LFL), n—the minimum concentration of a combustible sub-
fire risk of materials, products, or assemblies under actual fire
stance that is capable of propagating a flame through a
conditions. However, results of this test may be used as
homogeneous mixture of the combustible and a gaseous
elements of a fire risk assessment which takes into account all
oxidizer under the specified conditions of test.
of the factors which are pertinent to an assessment of the fire
3.1.2 upper limit of flammability or upper flammable limit
hazard of a particular end use.
(UFL), n—the maximum concentration of a combustible sub-
1.3 This standard does not purport to address all of the
stance that is capable of propagating a flame through a
safety concerns, if any, associated with its use. It is the
homogeneous mixture of the combustible and a gaseous
responsibility of the user of this standard to establish appro-
oxidizer under the specified conditions of test.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use. 3.2 Definitions of Terms Specific to This Standard:
3.2.1 propagation of flames, n—as used in this practice,a
1.4 This international standard was developed in accor-
dance with internationally recognized principles on standard- combustion reaction that produces at least a 7 % rise of the
initial absolute pressure:
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
P
^1.07
mendations issued by the World Trade Organization Technical
P
Barriers to Trade (TBT) Committee.
NOTE 1—This 7 % rise in pressure corresponds to 1 psia (0.007 MPa)
per atmosphere of initial pressure.
2. Referenced Documents
NOTE 2—The 7 % pressure criterion may not be appropriate for certain
fuelandoxidantmixtures.Thisisalsothecaseifthetestenclosurevolume
2.1 ASTM Standards:
is small, or when the ignition energy is substantially larger than 10 J. It is
E681 Test Method for Concentration Limits of Flammability
therefore a prudent practice to perform exploratory tests in the vicinity of
of Chemicals (Vapors and Gases) limit mixtures to evaluate the validity of the selected pressure rise
,5
criterion.
This practice is under the jurisdiction of ASTM Committee E27 on Hazard
Potential of Chemicals and is the direct responsibility of Subcommittee E27.04 on Available from National Technical Information Service (NTIS), 5301 Shawnee
Flammability and Ignitability of Chemicals. Rd., Alexandria, VA 22312, http://www.ntis.gov.
Current edition approved Dec. 1, 2019. Published January 2020. Originally Ural, E. A., and Brandes, E., “Towards A Global Standard For Flammability
approved in 1983. Last previous edition approved in 2011 as E918 – 83 (2011). Determination,” Proceedings of the 42nd Annual Loss Prevention Symposium,
DOI: 10.1520/E0918-19. Global Safety Congress, American Institute of Chemical Engineers, New Orleans,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or LA, Apr. 6–10, 2008.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Cashdollar, K. L., Zlochower, I. A., Green, G. M., Thomas, R. A., and
Standards volume information, refer to the standard’s Document Summary page on Hertzberg, M., “Flammability of Methane, Propane and Hydrogen Gases,” Journal
the ASTM website. of Loss Prevention in the Process Industry, Vol 13, 2000, pp. 327–340.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E918 − 19
4. Summary of Practice 7.3 In tests where the initial pressure is of the same order of
the atmospheric pressure or below, the test vessel must have a
4.1 A mixture of gaseous or vaporized fuel with a gaseous
volume of at least 4 L.
oxidizer is prepared in a steel or other appropriate metal vessel
at a controlled temperature and pressure. Proportions of the
8. Safety Precautions
components are determined by measurement of partial pres-
sures during filling of the vessel. Ignition of the mixture is 8.1 Adequate shielding must be provided to prevent injury
attempted with a fuse wire, and flammability is deduced from
in the event of equipment rupture. The apparatus is set up so
the pressure rise produced. Fuel concentration is varied be- that the operator is isolated by a blast-proof wall from the test
tween trials until the limits of flammability have been deter-
vesselwhilethevesselcontainsachargeofreactants,including
mined. Composition of the mixtures which fix the flammable the time while the vessel is being filled. The test apparatus
limits are confirmed by appropriate analysis.
should be equipped with interlocks so that the ignition source
cannot be activated unless the operator has taken necessary
5. Significance and Use
steps to protect personnel and equipment. Activation of the
5.1 Knowledgeofflammablelimitsatelevatedtemperatures
ignition source should be possible only from a position
and pressures is needed for safe and economical operation of
shielded from the test vessel.
some chemical processes. This information may be needed in
8.2 The test vessel shall be fitted with a rupture disk vented
order to start up a reactor without passing through a flammable
outside any enclosed area. Fuel may inadvertently be vented
range, to operate the reactor safely and economically, or to
inside the heated chamber or inside the enclosed area, so the
store or ship the product safely.
heated chamber should be fitted with an inert gas purge and the
5.2 Limits of flammability data obtained in relatively clean
area should be adequately ventilated to prevent buildup of an
vessels must be carefully interpreted and may not always be
explosive mixture in the large space.
applicable to industrial conditions. Surface effects due to
8.3 Undesirably energetic explosions may be produced if
carbon deposits and other materials can significantly affect
tests are made at high initial pressures with mixtures well
limits of flammability, especially in the fuel-rich region. Refer
within the flammable range. Very strong oxidizers greatly
to Bulletin 503 and Bulletin 627.
increase explosion severity and also greatly increase the
6. Limitations fuel-rich limit. To help in avoiding testing highly energetic
mixtures, limits of flammability should first be determined at
6.1 This practice is not applicable to mixtures which un-
atmospheric pressure. These limits are covered in Test Method
dergo spontaneous reaction before ignition is attempted.
E681. With this knowledge, the operator should proceed in
6.2 These test methods are limited to mixtures which have
cautious steps of initial pressure increase to work at higher
maximum deflagration pressures less than the maximum work-
pressures and temperatures.
ing pressure of the test apparatus.
9. Preparation of Apparatus
6.3 These test methods may be used up to the temperature
limit of the test system.
9.1 Clean and dry the test vessel and other gas-handling
6.4 Measurements of flammability are influenced by flame- equipment. Make sure that no oil, grease, or other combustible
quenching effects of the test vessel walls. Further surface is left inside the parts.
effects due to deposits of carbon or other materials can
9.2 Assemble the equipment as shown in Fig. 1. Purge the
significantly affect limits of flammability, especially in the
vessel with inert gas and then evacuate the system.
fuel-rich region. Refer to Bulletin 503 and Bulletin 627. For
9.3 Set the zero and gain on the pressure transducers so that
certain chemicals (for example, ammonia, halogenated
their output represents true pressure after the test vessel is at
materials, and certain amines) which have large ignition-
the working temperature.
quenching distances, tests may need to be conducted in vessels
larger than that specified below.
METHOD A
SAMPLE COMPONENTS WHICH HAVE ADEQUATE
7. Apparatus
VAPOR PRESSURE AT ROOM TEMPERATURE
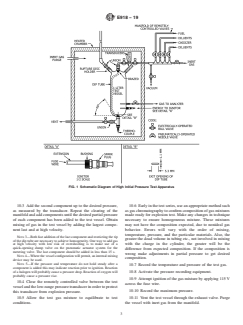
7.1 Fig. 1 is a schematic diagram of the high initial pressure
test apparatus; details and dimensions are presented in Annex
10. Procedure
A1. The apparatus consists of a metal pressure vessel with a
minimum volume of 1 Land a minimum inside diameter of 76 10.1 Attach pressure regulators to the supply cylinders of
gases to be used in the tests. Connect the regulators to the
mm (3 in.), an insulated chamber equipped with a source of
controlled-temperature inert gas, an ignition device with ap- manifold of remotely-controlled metering valves.
propriate power supply, remotely controlled valves, pressure
10.2 Flush each line from the supply cylinder to the meter-
measuring equipment, and a venting system for handling
ing valve. Evacuate the test vessel and manifold. By use of the
overpressuring.
remotely controlled valves, add to the test vessel the compo-
7.2 In tests where the initial pressure is of the same order of nent most appropriately added first; usually, this is the smallest
the atmospheric pressure or below, the apparatus described in component. Close the ball valve next to the test vessel and
Test Methods E2079 must be used. evacuate or purge the manifold.
E918 − 19
FIG. 1 Schematic Diagram of High Initial Pressure Test Apparatus
10.3 Add the second component up to the desired pressure, 10.6 Early in the test series, use an appropriate method such
as measured by the transducer. Repeat the clearing of the as gas chromatography to confirm composition of gas mixtures
manifold and add components until the desired partial pressure made ready for explosion test. Make any changes in technique
of each component has been added to the test vessel. Obtain necessary to ensure homogeneous mixture. These mixtures
mixing of gas in the test vessel by adding the largest compo- may not have the composition expected, due to nonideal gas
nent last and at high velocity. behavior. Errors will vary with the order of mixing,
temperature, pressure, and the particular materials. Also, the
NOTE 3—Both fast addition of the last component and restricting the tip
greater the dead volume in tubing etc., not involved in mixing
of the dip tube are necessary to achieve homogeneity. One way to add gas
with the charge in the cylinder, the greater will be the
at high velocity with low risk of overshooting is to make use of a
quick-opening dump valve on the pneumatic actuator system for the
difference from expected composition. If the composition is
metering valve. The last component should be added in less than 15 s.
wrong make adjustments in partial pressure to get desired
NOTE4—Wherethevesselconfigurationwillpermit,aninternalmixing
composition.
device may be used.
NOTE 5—If the pressure and temperature do not hold steady after a
10.7 Record the temperature and pressure of the test gas.
component is added this may indicate reaction prior to ignition. Reaction
of a halogen will probably cause a pressure drop. Reaction of oxygen will
10.8 Activate the pressure recording equipment.
probably cause a pressure rise.
10.9 Attempt ignition of the gas mixture by applying 115 V
10.4 Close the remotely controlled valve between the test
across the fuse wire.
vesselandthelow-rangepressuretransducerinordertoprotect
10.10 Record the maximum pressure.
this transducer from explosion pressure.
10.5 Allow the test gas mixture to equilibrate to test 10.11 Vent the test vessel through the exhaust valve. Purge
conditions. the vessel with inert gas from the manifold.
E918 − 19
NOTE8—Thecylinderofliquidfuelmustbefittedwithapressure-relief
10.12 Install another spark plug fitted with a fuse wire.
device which discharges outside the heated chamber and outsi
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E918 − 83 (Reapproved 2011) E918 − 19
Standard Practice for
Determining Limits of Flammability of Chemicals at Elevated
Temperature and Pressure
This standard is issued under the fixed designation E918; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers the determination of the lower and upper concentration limits of flammability of combustible
vapor-oxidant mixtures at temperatures up to 200°C and initial pressures up to as much as 1.38 MPa (200 psia). This practice is
limited to mixtures which would have explosion pressures less than 13.79 MPa (2000 psia).
1.2 This practice should be used to measure and describe the properties of materials, products, or assemblies in response to
heat and flame under controlled laboratory conditions and should not be used to describe or appraise the fire hazard or fire risk
of materials, products, or assemblies under actual fire conditions. However, results of this test may be used as elements of a fire
risk assessment which takes into account all of the factors which are pertinent to an assessment of the fire hazard of a particular
end use.
1.3 This standard may involve hazardous materials, operations, and equipment. This standard does not purport to address all
of the safety problems concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish
appropriate safety safety, health, and healthenvironmental practices and determine the applicability of regulatory limitations prior
to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E681 Test Method for Concentration Limits of Flammability of Chemicals (Vapors and Gases)
E2079 Test Methods for Limiting Oxygen (Oxidant) Concentration in Gases and Vapors
2.2 Other Documents:
Bulletin 503 Bureau of Mines, “Limits of Flammability of Gases and Vapors,” NTIS AD701575
Bulletin 627 Bureau of Mines, “Flammability Characteristics of Combustible Gases and Vapors,” NTIS AD701576
3. Terminology
3.1 Definitions:
3.1.1 lower limit of flammability or lower flammable limit (LFL)—(LFL), n—the minimum concentration of a combustible
substance that is capable of propagating a flame through a homogeneous mixture of the combustible and a gaseous oxidizer under
the specified conditions of test.
3.1.2 upper limit of flammability or upper flammable limit (UFL)—(UFL), n—the maximum concentration of a combustible
substance that is capable of propagating a flame through a homogeneous mixture of the combustible and a gaseous oxidizer under
the specified conditions of test.
3.2 Definitions of Terms Specific to This Standard:
This practice is under the jurisdiction of ASTM Committee E27 on Hazard Potential of Chemicals and is the direct responsibility of Subcommittee E27.04 on
Flammability and Ignitability of Chemicals.
Current edition approved Aug. 1, 2011Dec. 1, 2019. Published August 2011January 2020. Originally approved in 1983. Last previous edition approved in 20052011 as
E918 – 83 (2005).(2011). DOI: 10.1520/E0918-83R11. 10.1520/E0918-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
Available from National Technical Information Service (NTIS), 5301 Shawnee Rd., Alexandria, VA 22312, http://www.ntis.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E918 − 19
3.2.1 propagation of flames—flames, n—as used in this practice, a combustion reaction that produces at least a 7 % rise of the
initial absolute pressure,pressure:
P
^1.07.
P
P
^1.07
P
NOTE 1—This 7 % rise in pressure corresponds to 1 psia (0.007 MPa) per atmosphere of initial pressure.
NOTE 2—The 7 % pressure criterion may not be appropriate for certain fuel and oxidant mixtures. This is also the case if the test enclosure volume
is small, or when the ignition energy is substantially larger than 10 J. It is therefore a prudent practice to perform exploratory tests in the vicinity of limit
4, 5
mixtures to evaluate the validity of the selected pressure rise criterion.
4. Summary of Practice
4.1 A mixture of gaseous or vaporized fuel with a gaseous oxidizer is prepared in a steel or other appropriate metal vessel at
a controlled temperature and pressure. Proportions of the components are determined by measurement of partial pressures during
filling of the vessel. Ignition of the mixture is attempted with a fuse wire, and flammability is deduced from the pressure rise
produced. Fuel concentration is varied between trials until the limits of flammability have been determined. Composition of the
mixtures which fix the flammable limits are confirmed by appropriate analysis.
5. Significance and Use
5.1 Knowledge of flammable limits at elevated temperatures and pressures is needed for safe and economical operation of some
chemical processes. This information may be needed in order to start up a reactor without passing through a flammable range, to
operate the reactor safely and economically, or to store or ship the product safely.
5.2 Limits of flammability data obtained in relatively clean vessels must be carefully interpreted and may not always be
applicable to industrial conditions. Surface effects due to carbon deposits and other materials can significantly affect limits of
flammability, especially in the fuel-rich region. Refer to Bulletin 503 and Bulletin 627.
6. Limitations
6.1 This practice is not applicable to mixtures which undergo spontaneous reaction before ignition is attempted.
6.2 These test methods are limited to mixtures which have maximum deflagration pressures less than the maximum working
pressure of the test apparatus.
6.3 These test methods may be used up to the temperature limit of the test system.
6.4 Measured limits Measurements of flammability are influenced by flame-quenching effects of the test vessel walls. The vessel
described in this practice is suitable for use with most mixtures at elevated temperatures and pressures. For certain amines,
halogenated materials etc., Further surface effects due to deposits of carbon or other materials can significantly affect limits of
flammability, especially in the fuel-rich region. Refer to Bulletin 503 and Bulletin 627. For certain chemicals (for example,
ammonia, halogenated materials, and certain amines) which have large ignition-quenching distances, tests may need to be
conducted in larger diameter vessels.vessels larger than that specified below.
7. Apparatus
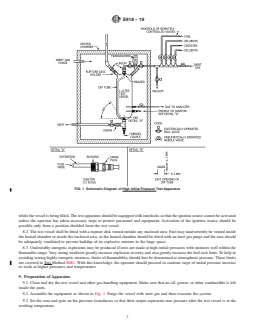
7.1 Fig. 1 is a schematic diagram of the high initial pressure test apparatus; details and dimensions are presented in Annex A1.
The apparatus consists of a metal pressure vessel with a minimum volume of 1 L and a minimum inside diameter of 76 mm (3
in.), an insulated chamber equipped with a source of controlled-temperature inert gas, an ignition device with appropriate power
supply, remotely controlled valves, pressure measuring equipment, and a venting system for handling overpressuring.
7.2 In tests where the initial pressure is of the same order of the atmospheric pressure or below, the apparatus described in Test
Methods E2079 must be used.
7.3 In tests where the initial pressure is of the same order of the atmospheric pressure or below, the test vessel must have a
volume of at least 4 L.
8. Safety Precautions
8.1 Adequate shielding must be provided to prevent injury in the event of equipment rupture. The apparatus is set up so that
the operator is isolated by a blast-proof wall from the test vessel while the vessel contains a charge of reactants, including the time
Ural, E. A., and Brandes, E., “Towards A Global Standard For Flammability Determination,” Proceedings of the 42nd Annual Loss Prevention Symposium, Global Safety
Congress, American Institute of Chemical Engineers, New Orleans, LA, Apr. 6–10, 2008.
Cashdollar, K. L., Zlochower, I. A., Green, G. M., Thomas, R. A., and Hertzberg, M., “Flammability of Methane, Propane and Hydrogen Gases,” Journal of Loss
Prevention in the Process Industry, Vol 13, 2000, pp. 327–340.
E918 − 19
FIG. 1 Schematic Diagram of High Initial Pressure Test Apparatus
while the vessel is being filled. The test apparatus should be equipped with interlocks so that the ignition source cannot be activated
unless the operator has taken necessary steps to protect personnel and equipment. Activation of the ignition source should be
possible only from a position shielded from the test vessel.
8.2 The test vessel shall be fitted with a rupture disk vented outside any enclosed area. Fuel may inadvertently be vented inside
the heated chamber or inside the enclosed area, so the heated chamber should be fitted with an inert gas purge and the area should
be adequately ventilated to prevent buildup of an explosive mixture in the large space.
8.3 Undesirably energetic explosions may be produced if tests are made at high initial pressures with mixtures well within the
flammable range. Very strong oxidizers greatly increase explosion severity and also greatly increase the fuel-rich limit. To help in
avoiding testing highly energetic mixtures, limits of flammability should first be determined at atmospheric pressure. These limits
are covered in Test Method E681. With this knowledge, the operator should proceed in cautious steps of initial pressure increase
to work at higher pressures and temperatures.
9. Preparation of Apparatus
9.1 Clean and dry the test vessel and other gas-handling equipment. Make sure that no oil, grease, or other combustible is left
inside the parts.
9.2 Assemble the equipment as shown in Fig. 1. Purge the vessel with inert gas and then evacuate the system.
9.3 Set the zero and gain on the pressure transducers so that their output represents true pressure after the test vessel is at the
working temperature.
E918 − 19
METHOD A—SAMPLEA
SAMPLE COMPONENTS WHICH HAVE ADEQUATE VAPOR PRESSURE AT ROOM TEMPERATURE
10. Procedure
10.1 Attach pressure regulators to the supply cylinders of gases to be used in the tests. Connect the regulators to the manifold
of remotely-controlled metering valves.
10.2 Flush each line from the supply cylinder to the metering valve. Evacuate the test vessel and manifold. By use of the
remotely controlled valves, add to the test vessel the component most appropriately added first; usually, this is the smallest
component. Close the ball valve next to the test vessel and evacuate or purge the manifold.
10.3 Add the second component up to the desired pressure, as measured by the transducer. Repeat the clearing of the manifold
and add components until the desired partial pressure of each component has been added to the test vessel. Obtain mixing of gas
in the test vessel by adding the largest component last and at high velocity.
NOTE 3—Both fast addition of the last component and restricting the tip of the dip tube are necessary to achieve homogeneity. One way to add gas
at high velocity with low risk of overshooting is to make use of a quick-opening dump valve on the pneumatic actuator system for the metering valve.
The last component should be added in less than 15 s.
NOTE 4—Where the vessel configuration will permit, an internal mixing device may be used.
NOTE 5—If the pressure and temperature do not hold steady after a component is added this may indicate reaction prior to ignition. Reaction of a
halogen will probably cause a pressure drop. Reaction of oxygen will probably cause a pressure rise.
10.4 Close the remotely controlled valve between the test vessel and the low-range pressure transducer in order to protect this
transducer from explosion pressure.
10.5 Allow the test gas mixture to equilibrate to test conditions.
10.6 Early in the test series, use an appropriate method such as gas chromatography to confirm composition of gas mixtures
made ready for explosion test. Make any changes in technique necessary to ensure homogeneous mixture. These mixtures may not
have the composition expected, due to nonideal gas behavior. Errors will vary with the order of mixing, temperature, pressure, and
the particular materials. Also, the greater the dead volume in tubing etc., not involved in mixing with the charge in the cylinder,
the greater will be the difference from expected composition. If the composition is wrong make adjustments in partial pressure to
get desired composition.
10.7 Record the temperature and pressure of the test gas.
10.8 Activate the pressure recording equipment.
10.9 Attempt ignition of the gas mixture by applying 115 V across the fuse wire.
10.10 Record the maximum pressure.
10.11 Vent the test vessel through the exhaust valve. Purge the vessel with inert gas from the manifold.
10.12 Install another spark plug fitted with a fuse wire.
NOTE 6—By having the spark plug positioned in f
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...