ASTM D5086-20
(Test Method)Standard Test Method for Determination of Calcium, Magnesium, Potassium, and Sodium in Atmospheric Wet Deposition by Flame Atomic Absorption Spectrophotometry
Standard Test Method for Determination of Calcium, Magnesium, Potassium, and Sodium in Atmospheric Wet Deposition by Flame Atomic Absorption Spectrophotometry
SIGNIFICANCE AND USE
5.1 This test method may be used for the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition samples.
5.2 Emphasis is placed on the easily contaminated quality of atmospheric wet deposition samples due to the low concentration levels of dissolved metals commonly present.
SCOPE
1.1 This test method is applicable to the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition (rain, snow, sleet, and hail) by flame atomic absorption spectrophotometry (FAAS) (1).2
1.2 The concentration ranges are listed below. The range tested was confirmed using the interlaboratory collaborative test (see Table 1 for a statistical summary of the collaborative test).
MDL
(mg/L) (2)
Range of Method
(mg/L)
Range Tested
(mg/L)
Calcium
0.009
0.03–3.00
0.168–2.939
Magnesium
0.003
0.01–1.00
0.039–0.682
Potassium
0.003
0.01–1.00
0.029–0.499
Sodium
0.003
0.01–2.00
0.105–1.84
1.3 The method detection limit (MDL) as given in 1.2 is based on single operator precision. Detection limits vary by instrumentation. Laboratories may be able to achieve lower detection limits. The method detection limit for this method as described in 1.2 was determined in 1987 (2) .
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific warning statements are given in 8.3, 8.7, 12.1.8, and Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Mar-2020
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.03 - Ambient Atmospheres and Source Emissions
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Sep-2020
- Effective Date
- 01-May-2020
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Jan-2020
- Effective Date
- 01-Aug-2019
- Effective Date
- 15-Apr-2019
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Jan-2018
- Effective Date
- 15-Aug-2017
- Effective Date
- 01-Feb-2017
Overview
ASTM D5086-20 is the standard test method for the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition (such as rain, snow, sleet, and hail) using flame atomic absorption spectrophotometry (FAAS). Developed by ASTM International, this standard provides comprehensive guidance for laboratories analyzing low-concentration dissolved metals in precipitation samples. Accurate detection of these elements plays a crucial role in atmospheric chemistry, environmental monitoring, and regulatory compliance.
This method emphasizes minimizing contamination, given the typically low levels of metals found in wet deposition. The procedure includes sample collection, preservation, preparation, instrument calibration, and detection using FAAS, ensuring reliable and reproducible results.
Key Topics
Scope of Application
- Determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition samples
- Designed for rain, snow, sleet, and hail
- Applicable concentration ranges based on validated interlaboratory testing
Detection Limits
- Method detection limits (MDL) for each analyte are exceptionally low (Ca: 0.009 mg/L; Mg, K, Na: 0.003 mg/L)
- Laboratories may achieve even lower detection depending on instrumentation and protocols
Contamination Prevention
- Highlighted need for rigor in sample handling, storage, and analytical environment due to low analyte concentrations
- Guidance on clean laboratory practices, including use of laminar flow clean air workstations and specialized glassware cleaning
Instrumentation and Calibration
- Use of flame atomic absorption spectrophotometer with suitable lamps and detector configurations
- Preparation of certified calibration standards and use of suppressors or releasing agents to minimize interference
- Recommendations for instrument optimization and calibration curve construction for accurate quantification
Quality Control
- Regular instrument calibration checks
- Quality assurance/quality control (QA/QC) data reporting requirements
- Adherence to safety and environmental practices
Applications
Environmental Monitoring
- Assessment of atmospheric deposition trends for environmental protection agencies and research organizations
- Evaluation of nutrient and contaminant fluxes to ecosystems via precipitation
Regulatory Compliance
- Supports legislative and regulatory programs aimed at controlling atmospheric deposition of metals
- Provides data for compliance with national and international environmental standards
Atmospheric Research
- Study of sources, transport, and deposition processes of important cations in the environment
- Input into climate models and regional air quality management
Laboratory Accreditation and Data Quality
- Ensures consistent and comparable data across laboratories
- Facilitates inter-laboratory studies and reporting for governmental bodies and research consortia
Related Standards
For best practices in wet deposition analysis and laboratory methods, consider referencing these related ASTM and international standards:
- ASTM D1193 - Specification for Reagent Water
- ASTM D2777 - Practice for Determination of Precision and Bias
- ASTM D4453 - Practice for Handling High Purity Water Samples
- ASTM D4691 - Practice for Measuring Elements in Water by Flame Atomic Absorption Spectrophotometry
- ASTM D5012 - Guide for Materials Used for Collection and Preservation of Atmospheric Wet Deposition
- ISO 8655 - Piston-operated Volumetric Apparatus
Summary
ASTM D5086-20 sets the benchmark for quantifying calcium, magnesium, potassium, and sodium in atmospheric wet deposition using FAAS. Its stringent procedures are vital for obtaining high-quality, low-level measurements in environmental and atmospheric research. By following this standard, laboratories contribute valuable data for environmental monitoring programs, regulatory compliance, and scientific investigation into atmospheric processes. For comprehensive and reliable results, adherence to ASTM D5086-20 and its related standards is highly recommended.
Buy Documents
ASTM D5086-20 - Standard Test Method for Determination of Calcium, Magnesium, Potassium, and Sodium in Atmospheric Wet Deposition by Flame Atomic Absorption Spectrophotometry
REDLINE ASTM D5086-20 - Standard Test Method for Determination of Calcium, Magnesium, Potassium, and Sodium in Atmospheric Wet Deposition by Flame Atomic Absorption Spectrophotometry
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5086-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Calcium, Magnesium, Potassium, and Sodium in Atmospheric Wet Deposition by Flame Atomic Absorption Spectrophotometry". This standard covers: SIGNIFICANCE AND USE 5.1 This test method may be used for the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition samples. 5.2 Emphasis is placed on the easily contaminated quality of atmospheric wet deposition samples due to the low concentration levels of dissolved metals commonly present. SCOPE 1.1 This test method is applicable to the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition (rain, snow, sleet, and hail) by flame atomic absorption spectrophotometry (FAAS) (1).2 1.2 The concentration ranges are listed below. The range tested was confirmed using the interlaboratory collaborative test (see Table 1 for a statistical summary of the collaborative test). MDL (mg/L) (2) Range of Method (mg/L) Range Tested (mg/L) Calcium 0.009 0.03–3.00 0.168–2.939 Magnesium 0.003 0.01–1.00 0.039–0.682 Potassium 0.003 0.01–1.00 0.029–0.499 Sodium 0.003 0.01–2.00 0.105–1.84 1.3 The method detection limit (MDL) as given in 1.2 is based on single operator precision. Detection limits vary by instrumentation. Laboratories may be able to achieve lower detection limits. The method detection limit for this method as described in 1.2 was determined in 1987 (2) . 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific warning statements are given in 8.3, 8.7, 12.1.8, and Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method may be used for the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition samples. 5.2 Emphasis is placed on the easily contaminated quality of atmospheric wet deposition samples due to the low concentration levels of dissolved metals commonly present. SCOPE 1.1 This test method is applicable to the determination of calcium, magnesium, potassium, and sodium in atmospheric wet deposition (rain, snow, sleet, and hail) by flame atomic absorption spectrophotometry (FAAS) (1).2 1.2 The concentration ranges are listed below. The range tested was confirmed using the interlaboratory collaborative test (see Table 1 for a statistical summary of the collaborative test). MDL (mg/L) (2) Range of Method (mg/L) Range Tested (mg/L) Calcium 0.009 0.03–3.00 0.168–2.939 Magnesium 0.003 0.01–1.00 0.039–0.682 Potassium 0.003 0.01–1.00 0.029–0.499 Sodium 0.003 0.01–2.00 0.105–1.84 1.3 The method detection limit (MDL) as given in 1.2 is based on single operator precision. Detection limits vary by instrumentation. Laboratories may be able to achieve lower detection limits. The method detection limit for this method as described in 1.2 was determined in 1987 (2) . 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific warning statements are given in 8.3, 8.7, 12.1.8, and Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5086-20 is classified under the following ICS (International Classification for Standards) categories: 07.060 - Geology. Meteorology. Hydrology; 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5086-20 has the following relationships with other standards: It is inter standard links to ASTM E694-18(2024), ASTM D883-24, ASTM D883-23, ASTM D1356-20a, ASTM D1129-13(2020)e2, ASTM D1356-20, ASTM D883-20, ASTM D883-19c, ASTM D883-19a, ASTM D883-19, ASTM D883-18a, ASTM D883-18, ASTM E694-18, ASTM D883-17, ASTM D4453-17. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5086-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5086 − 20
Standard Test Method for
Determination of Calcium, Magnesium, Potassium, and
Sodium in Atmospheric Wet Deposition by Flame Atomic
Absorption Spectrophotometry
This standard is issued under the fixed designation D5086; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
1.1 This test method is applicable to the determination of
mendations issued by the World Trade Organization Technical
calcium, magnesium, potassium, and sodium in atmospheric
Barriers to Trade (TBT) Committee.
wet deposition (rain, snow, sleet, and hail) by flame atomic
absorption spectrophotometry (FAAS) (1).
2. Referenced Documents
1.2 The concentration ranges are listed below. The range
2.1 ASTM Standards:
tested was confirmed using the interlaboratory collaborative
D883 Terminology Relating to Plastics
test (see Table 1 for a statistical summary of the collaborative
D1129 Terminology Relating to Water
test).
D1193 Specification for Reagent Water
MDL Range of Method Range Tested
D1356 Terminology Relating to Sampling and Analysis of
(mg/L) (2) (mg/L) (mg/L)
Atmospheres
Calcium 0.009 0.03–3.00 0.168–2.939
D2777 Practice for Determination of Precision and Bias of
Magnesium 0.003 0.01–1.00 0.039–0.682
Applicable Test Methods of Committee D19 on Water
Potassium 0.003 0.01–1.00 0.029–0.499
D4453 Practice for Handling of High Purity Water Samples
Sodium 0.003 0.01–2.00 0.105–1.84
D4691 Practice for Measuring Elements in Water by Flame
1.3 The method detection limit (MDL) as given in 1.2 is
Atomic Absorption Spectrophotometry
based on single operator precision. Detection limits vary by
D5012 Practice for Preparation of Materials Used for the
instrumentation. Laboratories may be able to achieve lower
Collection and Preservation of Atmospheric Wet Deposi-
detection limits. The method detection limit for this method as
tion
described in 1.2 was determined in 1987 (2).
E131 Terminology Relating to Molecular Spectroscopy
1.4 The values stated in SI units are to be regarded as
E275 Practice for Describing and Measuring Performance of
standard. No other units of measurement are included in this
Ultraviolet and Visible Spectrophotometers
standard.
E694 Specification for Laboratory Glass Volumetric Appa-
1.5 This standard does not purport to address all of the
ratus
safety concerns, if any, associated with its use. It is the
IEEE/ASTM SI-10 Standard for Use of the International
responsibility of the user of this standard to establish appro-
System of Units (SI): The Modern Metric System
priate safety, health, and environmental practices and deter- 4
2.2 ISO Standard:
mine the applicability of regulatory limitations prior to use.
ISO 8655 Piston-Operated Volumetric Apparatus — Part 2:
Specific warning statements are given in 8.3, 8.7, 12.1.8, and
Piston Pipettes
Section 9.
1.6 This international standard was developed in accor-
3. Terminology
dance with internationally recognized principles on standard-
3.1 Definitions:
This test method is under the jurisdiction of ASTM Committee D22 on Air
Quality and is the direct responsibility of Subcommittee D22.03 on Ambient For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Atmospheres and Source Emissions. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved March 15, 2020. Published April 2020. Originally Standards volume information, refer to the standard’s Document Summary page on
approved in 1990. Last previous edition approved in 2013 as D5086 – 01 (2013). the ASTM website.
DOI: 10.1520/D5086-20. Available from International Organization for Standardization (ISO), ISO
The boldface numbers in parentheses refer to a list of references at the end of Central Secretariat, BIBC II, Chemin de Blandonnet 8, CP 401, 1214 Vernier,
this test method. Geneva, Switzerland, http://www.iso.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5086 − 20
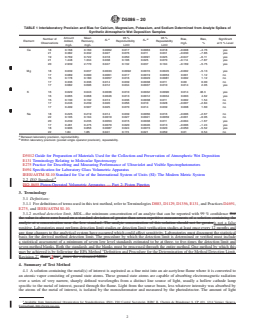
TABLE 1 Interlaboratory Precision and Bias for Calcium, Magnesium, Potassium, and Sodium Determined from Analyte Spikes of
Synthetic Atmospheric Wet Deposition Samples
Amount Mean 95 % 95 %
Number of Bias, Bias, Significant
A B
Element Added, Recovery, S Reproducibility S Repeatability
t o
Observations mg/L % at 5 % Level
mg/L mg/L Limit Limit
Ca 18 0.168 0.160 0.0062 0.017 0.0063 0.018 −0.008 −4.76 yes
21 0.382 0.332 0.027 0.076 0.011 0.031 −0.030 −7.85 yes
19 0.769 0.722 0.018 0.050 0.0091 0.025 −0.047 −6.11 yes
21 1.448 1.334 0.038 0.106 0.025 0.070 −0.114 −7.87 yes
20 2.939 2.770 0.047 0.132 0.037 0.104 −0.169 −5.75 yes
Mg 18 0.039 0.037 0.0033 0.0092 0.0016 0.0045 −0.002 −5.13 yes
17 0.089 0.090 0.0061 0.017 0.0019 0.0053 0.001 1.12 no
15 0.178 0.180 0.0057 0.016 0.0029 0.0081 0.002 1.12 no
17 0.336 0.336 0.014 0.039 0.0038 0.011 0.00 0.00 no
17 0.682 0.696 0.012 0.034 0.0037 0.010 0.014 2.05 yes
K 16 0.029 0.043 0.0036 0.010 0.0032 0.0090 0.014 48.3 yes
16 0.065 0.068 0.0046 0.013 0.0012 0.0034 0.003 4.62 yes
15 0.130 0.132 0.013 0.036 0.0038 0.011 0.002 1.54 no
17 0.246 0.239 0.020 0.056 0.010 0.028 −0.007 −2.84 no
17 0.499 0.507 0.025 0.070 0.014 0.039 0.008 1.60 no
Na 18 0.225 0.219 0.014 0.039 0.0056 0.016 −0.006 −2.67 no
22 0.105 0.104 0.0010 0.027 0.0021 0.0059 −0.001 −0.95 no
20 0.239 0.235 0.0053 0.015 0.0038 0.011 −0.004 −1.67 yes
17 0.481 0.475 0.0070 0.020 0.0046 0.013 −0.006 −1.24 yes
18 0.906 0.856 0.0087 0.024 0.0073 0.020 −0.050 −5.52 yes
22 1.84 1.85 0.041 0.115 0.021 0.059 0.01 0.54 no
A
Between laboratory precision, reproducibility.
B
Within laboratory precision (pooled single operator precision), repeatability.
3.1.1 For definitions of terms used in this test method, refer comparing the light transmitted through the flame to light
to Terminologies D883, D1129, D1356, E131, and Practices transmitted by a reference beam. The amount of light absorbed
D4691, E275, and IEEE/ASTM SI-10. in the flame is proportional to the concentration of the metal in
3.1.2 method detection limit, MDL—the minimum concen- solution. The relationship between absorption and concentra-
tration of an analyte that can be reported with 99 % confidence tion is expressed by Beer’s Law:
is not a false positive. Laboratories must perform detection
log I /I 5 abc 5 A (1)
~ !
o
limit studies or detection limit verification studies at least once
where:
every 12 months and any time changes to the analytical system
have occurred which could affect sensitivity. Laboratories must I = incident radiant power,
o
I = transmitted radiant power,
document the statistical basis for the derived method detection
a = absorptivity (constant for a given system),
limit. The procedure by which the detection limit is determined
b = sample path length,
or verified must include a statistical assessment of a minimum
c = concentration of absorbing species, and
of seven low level standards estimated to be at three- to
A = absorbance.
five-times the detection limit and seven method blanks. Both
The atomic absorption spectrophotometer is calibrated with
the standards and the blanks must be processed through the
standard solutions containing known concentrations of the
entire method. One method by which this may be achieved is
element(s) of interest. The concentration of each analyte in the
by following the EPA Method “Definition and Procedure for
unknown sample is determined from constructed calibration
the Determination of the Method Detection Limit, Revision 2”
(3). curves.
5. Significance and Use
4. Summary of Test Method
5.1 This test method may be used for the determination of
4.1 A solution containing the metal(s) of interest is aspirated
calcium, magnesium, potassium, and sodium in atmospheric
as a fine mist into an air acetylene flame where it is converted
wet deposition samples.
to an atomic vapor consisting of ground state atoms. These
ground state atoms are capable of absorbing electromagnetic
5.2 Emphasis is placed on the easily contaminated quality of
radiation over a series of very narrow, sharply defined wave-
atmospheric wet deposition samples due to the low concentra-
lengths from a distinct line source of light, usually a hollow
tion levels of dissolved metals commonly present.
cathode lamp specific to the metal of interest, passed through
6. Interferences
the flame. Light from the source beam, less whatever intensity
was absorbed by the atoms of the metal of interest, is isolated 6.1 A chemical interference can prevent, enhance, or sup-
by the monochromator and measured by the photodetector. The press the formation of ground state atoms in the flame. For
amount of light absorbed by the analyte is quantified by example, in the case of calcium determinations, the presence of
D5086 − 20
phosphate or sulfate can result in the formation of a salt that 7.3 Volumetric Flasks—Maintain a set of Class A volumetric
hinders proper atomization of the solution when it is aspirated flasks (see Specification E694) to be used only when making
into the flame. This decreases the number of free, ground state dilute calibration solutions for the analysis of atmospheric wet
atoms in the flame, resulting in lowered absorbance values. deposition samples.
7.3.1 The first time any glassware is used for making stock
Aluminum can cause a similar interference when measuring
magnesium. The addition of appropriate complexing agents, solutions and standards, clean with HCl (1+1) and rinse
thoroughly with water before use.
such as lanthanum, to the sample solution reduces or eliminates
chemical interferences and may increase the sensitivity of this 7.3.2 Store clean glassware filled with water and covered.
test method.
7.4 Laboratory Facilities—Laboratories used for the analy-
sis of atmospheric wet deposition samples should be free from
6.2 Alkali metals, such as potassium and sodium, can
external sources of contamination.
undergo ionization in an air-acetylene flame resulting in a
7.4.1 The use of laminar flow clean air workstations is
decrease in ground state atoms available for measurement by
recommended for sample processing and preparation to avoid
atomic absorption. The addition of a large excess of an easily
the introduction of airborne contaminants. If a clean air
ionizable element, such as cesium, will eliminate this problem,
workstations is unavailable, samples must be capped or cov-
since cesium will be preferentially ionized. The preferential
ered prior to analysis.
ionization of the cesium results in an enhanced atomic absorp-
7.4.2 A positive pressure environment within the laboratory
tion signal for both potassium and sodium.
is recommended to minimize the introduction of external
6.3 If a sample containing low concentrations of the metal
sources of contaminant gases and particulates. Windows within
being measured is analyzed immediately after a sample having
the laboratory should be kept closed at all times and sealed if
a concentration exceeding the concentration of the highest
leaks are apparent.
calibration standard, sample carryover can result in elevated
7.4.3 The use of disposable tacky floor mats at the entrance
readings due to residual metal from the previous sample. To
to the laboratory is helpful in reducing the particulate loading
prevent this interference, routinely aspirate water for about 15
within the room.
s after a high concentration sample. Depending on the concen-
tration of metal in the last sample analyzed, it may be
8. Reagents and Materials
necessary to rinse for longer time periods. Complete purging of
8.1 Purity of Reagents—Use reagent grade or better. All
the system is ascertained by aspirating water until the absor-
reagents shall conform to the specifications of the Committee
bance readout returns to the baseline.
on Analytical Reagents of the American Chemical Society
(ACS) where such specifications are available.
6.4 Atmospheric wet deposition samples are characterized
by low ionic strength and rarely contain enough salts to cause
8.2 Purity of Water—Unless otherwise indicated, references
interferences due to non-specific background absorbance. The
to water shall be understood to mean reagent water as defined
use of background correction techniques is not necessary and
by Type I of Specification D1193. Point of use 0.2 μm filters
will decrease the signal to noise ratio and lessen precision.
are recommended for all faucets supplying water to prevent the
introduction of bacteria and/or ion exchange resins into re-
7. Apparatus
agents.
8.3 Acetylene (Fuel)—Minimum acceptable acetylene pu-
7.1 Analytical Apparatus:
rity is 99.5 % (v/v). Change the cylinder when the pressure
7.1.1 Flame Atomic Absorption Spectrophotometer,
reaches 517 kPa (75 psig) if the acetylene is packed in acetone.
equipped with air/acetylene and nitrous oxide/acetylene burner
Pre-purified grades that contain a proprietary solvent can be
heads.
used to 207 kPa (30 psig) before replacement. Avoid introduc-
7.1.2 Hollow Cathode or Electrodeless Discharge Lamp, for
ing these solvents into the instrument. Damage to the instru-
each element to be determined.
ment’s plumbing system can result. To prevent solvent
7.1.3 Deuterium Continuum Lamp.
carryover, allow acetylene cylinders to stand for at least 24 h
7.1.4 Compressed Air—Appropriate pressure reducing regu-
before use. (Warning—Acetylene is a highly flammable gas.
lator with base connections (see instrument manufacturer’s
Follow the precautions in 9.3 – 9.6 regarding safe operating
instructions).
pressures, suitable plumbing, and operator safety.)
7.1.5 Acetylene Gas and Regulator—A cylinder of acety-
8.4 Cesium Solution (Ionization Suppressant)—Dissolve
lene equipped with a two-gage, two-stage pressure-reducing
126.7 g cesium chloride (CsCl), dried at 105°C for 1 h, in water
regulator with hose connections (see instrument manufactur-
and dilute to 1 L. Store at room temperature in a high density
er’s instructions).
7.2 Volumetric Pipets—Maintain a set of Class A volumetric
pipets (see Specification E694) to be used only when making
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
dilute calibration solutions for the analysis of atmospheric wet
DC. For suggestions on the testing of reagents not listed by the American Chemical
deposition samples. Alternatively, variable or fixed volume
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
piston operated disposable tip pipets may be used if they
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
comply with ISO 8655. copeial Convention, Inc. (USPC), Rockville, MD.
D5086 − 20
polyethylene or polypropylene container. Alternatively, com- 9.3 Acetylene is a flammable gas; take precautions when
mercially available Cesium ionization suppressants/buffers using it. To avoid explosions, never pass acetylene through
may be purchased. copper or high-copper alloy (brass, bronze) fittings or piping.
9.4 The operator must wear appropriate safety glasses to
8.5 Hydrochloric Acid (1+1, Volume to Volume)—Carefully
avoid eye damage from the ultraviolet light emitted by the
add one volume of concentrated hydrochloric acid (HCl, sp gr
flame.
1.19) to an equal volume of water.
9.5 To avoid in-line explosions, do not allow the pressure of
8.6 Hydrochloric Acid (1+19, Volume to Volume)—
the acetylene being delivered to exceed about 100 kPa (15
Carefully add 50 mL of concentrated hydrochloric acid (HCl,
psig). In the event of a flashback, turn off the gas control
sp gr 1.19) to 900 mL of water and dilute to 1 L.
switch, the instrument power, and the acetylene tanks.
8.7 Lanthanum Solution (Releasing Agent)—In a glass 1 L
9.6 Follow manufacturer’s operating guidelines carefully
volumetric flask, place 117.3 g of lanthanum oxide (La O ),
2 3
when optimizing gas flow rates. Too low gas flow rates can
dried at 105°C for 1 h. Wet with water and add HCl (1+1) in
result in a combustion within the gas mixing chamber and
small increments until a total of 500 mL of HCl (1+1) has been
therefore a flashback.
added. Cool the solution between additions. Dilute to 1 L with
water. Store at room temperature in a high density polyethylene
9.7 Check that the drain tube from the gas mixing chamber,
or polypropylene container. (Warning—Dissolving lanthanum
fitted with a safety trap, is filled with water before igniting the
oxide in hydrochloric acid is a strongly exothermic reaction;
flame. Keep the drain tube filled to prevent explosion in the
use extreme caution when dissolving the reagent. Refer to 9.1
chamber. The safety trap may be either looped or valved.
for proper safety precautions when preparing this solution.)
9.8 Avoid contact with a hot burner head to prevent serious
Alternatively, commercially available lanthanum releasing
tissue burns.
agent solutions may be purchased.
10. Sampling, Test Samples, and Test Units
8.8 Ox
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5086 − 01 (Reapproved 2013) D5086 − 20
Standard Test Method for
Determination of Calcium, Magnesium, Potassium, and
Sodium in Atmospheric Wet Deposition by Flame Atomic
Absorption Spectrophotometry
This standard is issued under the fixed designation D5086; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope
1.1 This test method is applicable to the determination of calcium, magnesium, potassium, and sodium in atmospheric wet
deposition (rain, snow, sleet, and hail) by flame atomic absorption spectrophotometry (FAAS) (1).
1.2 The concentration ranges are listed below. The range tested was confirmed using the interlaboratory collaborative test (see
Table 1 for a statistical summary of the collaborative test).
MDL Range of Method Range Tested
(mg/L) (2) (mg/L) (mg/L)
Calcium 0.009 0.03–3.00 0.168–2.939
Magnesium 0.003 0.01–1.00 0.039–0.682
Potassium 0.003 0.01–1.00 0.029–0.499
Sodium 0.003 0.01–2.00 0.105–1.84
1.3 The method detection limit (MDL) as given in 1.2 is based on single operator precisionprecision. (Detection2) and may be
higher or lower for other operators and laboratories. Many workers have found that this test method is reliable at lower levelslimits
vary by instrumentation. Laboratories may be able to achieve lower detection limits. The method detection limit for this method
as described in 1.2 than were tested, butwas determined in 1987 the (2precision ).and bias data presented are insufficient to justify
their use at lower levels.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. Specific warning statements are given in 8.3, 8.7, 12.1.8, and Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D883 Terminology Relating to Plastics
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D1356 Terminology Relating to Sampling and Analysis of Atmospheres
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D4453 Practice for Handling of High Purity Water Samples
D4691 Practice for Measuring Elements in Water by Flame Atomic Absorption Spectrophotometry
This test method is under the jurisdiction of ASTM Committee D22 on Air Quality and is the direct responsibility of Subcommittee D22.03 on Ambient Atmospheres
and Source Emissions.
Current edition approved Oct. 1, 2013March 15, 2020. Published October 2013April 2020. Originally approved in 1990. Last previous edition approved in 20082013 as
D5086 – 01 (2013).(2008). DOI: 10.1520/D5086-01R13.10.1520/D5086-20.
The boldface numbers in parentheses refer to a list of references at the end of this test method.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5086 − 20
TABLE 1 Interlaboratory Precision and Bias for Calcium, Magnesium, Potassium, and Sodium Determined from Analyte Spikes of
Synthetic Atmospheric Wet Deposition Samples
Amount Mean 95 % 95 %
A B
Number of S S Bias, Bias, Significant
t o
Element Added, Recovery, Reproducibility Repeatability
Observations mg/L % at 5 % Level
mg/L mg/L Limit Limit
Ca 18 0.168 0.160 0.0062 0.017 0.0063 0.018 −0.008 −4.76 yes
21 0.382 0.332 0.027 0.076 0.011 0.031 −0.030 −7.85 yes
19 0.769 0.722 0.018 0.050 0.0091 0.025 −0.047 −6.11 yes
21 1.448 1.334 0.038 0.106 0.025 0.070 −0.114 −7.87 yes
20 2.939 2.770 0.047 0.132 0.037 0.104 −0.169 −5.75 yes
Mg 18 0.039 0.037 0.0033 0.0092 0.0016 0.0045 −0.002 −5.13 yes
17 0.089 0.090 0.0061 0.017 0.0019 0.0053 0.001 1.12 no
15 0.178 0.180 0.0057 0.016 0.0029 0.0081 0.002 1.12 no
17 0.336 0.336 0.014 0.039 0.0038 0.011 0.00 0.00 no
17 0.682 0.696 0.012 0.034 0.0037 0.010 0.014 2.05 yes
K 16 0.029 0.043 0.0036 0.010 0.0032 0.0090 0.014 48.3 yes
16 0.065 0.068 0.0046 0.013 0.0012 0.0034 0.003 4.62 yes
15 0.130 0.132 0.013 0.036 0.0038 0.011 0.002 1.54 no
17 0.246 0.239 0.020 0.056 0.010 0.028 −0.007 −2.84 no
17 0.499 0.507 0.025 0.070 0.014 0.039 0.008 1.60 no
Na 18 0.225 0.219 0.014 0.039 0.0056 0.016 −0.006 −2.67 no
22 0.105 0.104 0.0010 0.027 0.0021 0.0059 −0.001 −0.95 no
20 0.239 0.235 0.0053 0.015 0.0038 0.011 −0.004 −1.67 yes
17 0.481 0.475 0.0070 0.020 0.0046 0.013 −0.006 −1.24 yes
18 0.906 0.856 0.0087 0.024 0.0073 0.020 −0.050 −5.52 yes
22 1.84 1.85 0.041 0.115 0.021 0.059 0.01 0.54 no
A
Between laboratory precision, reproducibility.
B
Within laboratory precision (pooled single operator precision), repeatability.
D5012 Guide for Preparation of Materials Used for the Collection and Preservation of Atmospheric Wet Deposition
E131 Terminology Relating to Molecular Spectroscopy
E275 Practice for Describing and Measuring Performance of Ultraviolet and Visible Spectrophotometers
E694 Specification for Laboratory Glass Volumetric Apparatus
IEEE/ASTM SI-10 Standard for Use of the International System of Units (SI): The Modern Metric System
2.2 ISO Standard:
ISO 8655 Piston-Operated Volumetric Apparatus — Part 2: Piston Pipettes
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this test method, refer to Terminologies D883, D1129, D1356, E131, and Practices D4691,
E275, and IEEE/ASTM SI-10.
3.1.2 method detection limit, MDL—the minimum concentration of an analyte that can be reported with 99 % confidence that
the value is above zero based on a standard deviation of greater than seven repetitive measurements of a solution containing the
analyte at a concentration near the low standard. The analyte concentration of this solution should not be greateris not a false
positive. Laboratories must perform detection limit studies or detection limit verification studies at least once every 12 months and
any time changes to the analytical system have occurred which could affect sensitivity. Laboratories must document the statistical
basis for the derived method detection limit. The procedure by which the detection limit is determined or verified must include
a statistical assessment of a minimum of seven low level standards estimated to be at three- to five-times the detection limit and
seven method blanks. Both the standards and the blanks must be processed through the entire method. One method by which this
may be achieved is by following the EPA Method “Definition and Procedure for the Determination of the Method Detection Limit,
Revision 2” than (3ten ).times the estimated MDL.
4. Summary of Test Method
4.1 A solution containing the metal(s) of interest is aspirated as a fine mist into an air acetylene flame where it is converted to
an atomic vapor consisting of ground state atoms. These ground state atoms are capable of absorbing electromagnetic radiation
over a series of very narrow, sharply defined wavelengths from a distinct line source of light, usually a hollow cathode lamp
specific to the metal of interest, passed through the flame. Light from the source beam, less whatever intensity was absorbed by
the atoms of the metal of interest, is isolated by the monochromator and measured by the photodetector. The amount of light
Available from International Organization for Standardization (ISO), ISO Central Secretariat, BIBC II, Chemin de Blandonnet 8, CP 401, 1214 Vernier, Geneva,
Switzerland, http://www.iso.org.
D5086 − 20
absorbed by the analyte is quantified by comparing the light transmitted through the flame to light transmitted by a reference beam.
The amount of light absorbed in the flame is proportional to the concentration of the metal in solution. The relationship between
absorption and concentration is expressed by Beer’sBeer’s Law:
log~I /I! 5 abc 5 A (1)
o
where:
I = incident radiant power,
o
I = transmitted radiant power,
a = absorptivity (constant for a given system),
b = sample path length,
c = concentration of absorbing species, and
A = absorbance.
The atomic absorption spectrophotometer is calibrated with standard solutions containing known concentrations of the
element(s) of interest. The concentration of each analyte in the unknown sample is determined from contructedconstructed
calibration curves.
5. Significance and Use
5.1 This test method may be used for the determination of calcium, magnesium, potassium, and sodium in atmospheric wet
deposition samples.
5.2 Emphasis is placed on the easily contaminated quality of atmospheric wet deposition samples due to the low concentration
levels of dissolved metals commonly present.
5.3 Annex A1 represents cumulative frequency percentile concentration plots of calcium, magnesium, potassium, and sodium
obtained from analyses of over five thousand wet deposition samples. These data may be used as an aid in the selection of
appropriate calibration standard concentrations. (3)
6. Interferences
6.1 A chemical interference can prevent, enhance, or suppress the formation of ground state atoms in the flame. For example,
in the case of calcium determinations, the presence of phosphate or sulfate can result in the formation of a salt that hinders proper
atomization of the solution when it is aspirated into the flame. This decreases the number of free, ground state atoms in the flame,
resulting in lowered absorbance values. Aluminum can cause a similar interference when measuring magnesium. The addition of
appropriate complexing agents, such as lanthanum, to the sample solution reduces or eliminates chemical interferences and may
increase the sensitivity of this test method.
6.2 Alkali metals, such as potassium and sodium, can undergo ionization in an air-acetylene flame resulting in a decrease in
ground state atoms available for measurement by atomic absorption. The addition of a large excess of an easily ionizable element,
such as cesium, will eliminate this problem, since cesium will be preferentially ionized. The preferential ionization of the cesium
results in an enhanced atomic absorption signal for both potassium and sodium.
6.3 If a sample containing low concentrations of the metal being measured is analyzed immediately after a sample having a
concentration exceeding the concentration of the highest calibration standard, sample carryover can result in elevated readings due
to residual metal from the previous sample. To prevent this interference, routinely aspirate water for about 15 s after a high
concentration sample. Depending on the concentration of metal in the last sample analyzed, it may be necessary to rinse for longer
time periods. Complete purging of the system is ascertained by aspirating water until the absorbance readout returns to the baseline.
6.4 Atmospheric wet deposition samples are characterized by low ionic strength and rarely contain enough salts to cause
interferences due to non-specific background absorbance. The use of background correction techniques is not necessary and will
decrease the signal to noise ratio and lessen precision.
7. Apparatus
7.1 Atomic Absorption Spectrophotometer—Analytical Apparatus: Select a double-beam instrument having a monochromator,
photodetector, pressure-reducing valves, adjustable spectral bandwidth, and a wavelength range of 190 to 800 nm. Peripheral
equipment may include a strip chart recorder or a suitable data system.
7.1.1 Burner—Flame Atomic Absorption Spectrophotometer, Use a long-path, single slot, air-acetylene burner head supplied by
the manufacturer of the spectrophotometer. equipped with air/acetylene and nitrous oxide/acetylene burner heads.
7.1.2 Hollow Cathode Lamps—or Electrodeless Discharge Lamp, Single element lamps are recommended. Multi-element
lamps are available but are not recommended. They have a shorter lifespan, are less sensitive, require a higher operating current,
and increase the chances of spectral interferences.for each element to be determined.
7.1.3 Deuterium Continuum Lamp.
7.1.4 Compressed Air—Appropriate pressure reducing regulator with base connections (see instrument manufacturer’s
instructions).
D5086 − 20
7.1.5 Monochromator—Acetylene Gas and Regulator—To increase the sensitivity for calcium and potassium measurements, a
monochromator A cylinder of acetylene equipped with a blaze grating in the range of 500 to 600 nm is recommended. For the
analysis of magnesium and sodium, a blaze grating in the range of 200 to 250 nm is adequate. two-gage, two-stage
pressure-reducing regulator with hose connections (see instrument manufacturer’s instructions).
7.1.4 Photomultiplier Tube—A wide spectral range (160 to 900 nm) photomultiplier tube is recommended. Select a red-sensitive
photomultiplier tube to detect potassium at 766.5 nm and to increase sensitivity for calcium at 422.7 nm.
7.2 Volumetric Pipets—Maintain a set of Class A volumetric pipets (see Specification E694) to be used only when making dilute
calibration solutions for the analysis of atmospheric wet deposition samples. Alternatively, variable or fixed volume piston operated
disposable tip pipets may be used.used if they comply with ISO 8655.
7.3 Volumetric Flasks—Maintain a set of Class A volumetric flasks (see Specification E694) to be used only when making dilute
calibration solutions for the analysis of atmospheric wet deposition samples.
7.3.1 The first time any glassware is used for making stock solutions and standards, clean with HCl (1+1) and rinse thoroughly
with water before use.
7.3.2 Store clean glassware filled with water and covered.
7.4 Laboratory Facilities—Laboratories used for the analysis of atmospheric wet deposition samples should be free from
external sources of contamination.
7.4.1 The use of laminar flow clean air workstations is recommended for sample processing and preparation to avoid the
introduction of airborne contaminants. If a clean air workstations is unavailable, samples must be capped or covered prior to
analysis.
7.4.2 A positive pressure environment within the laboratory is recommended to minimize the introduction of external sources
of contaminant gases and particulates. Windows within the laboratory should be kept closed at all times and sealed if leaks are
apparent.
7.4.3 The use of disposable tacky floor mats at the entrance to the laboratory is helpful in reducing the particulate loading within
the room.
8. Reagents and Materials
8.1 Purity of Reagents—Use reagent grade or higher grade chemicals for all solutions. better. All reagents shall conform to the
specifications of the Committee on Analytical Reagents of the American Chemical Society (ACS) where such specifications are
available.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water as defined by
Type I of Specification D1193. Point of use 0.2 μm filters are recommended for all faucets supplying water to prevent the
introduction of bacteria and/or ion exchange resins into reagents.
8.3 Acetylene (Fuel)—Minimum acceptable acetylene purity is 99.5 % (v/v). Change the cylinder when the pressure reaches 517
kPa (75 psig) if the acetylene is packed in acetone. Pre-purified grades that contain a proprietary solvent can be used to 207 kPa
(30 psig) before replacement. Avoid introducing these solvents into the instrument. Damage to the instrument’sinstrument’s
plumbing system can result. To prevent solvent carryover, allow acetylene cylinders to stand for at least 24 h before use.
(Warning—Acetylene is a highly flammable gas. Follow the precautions in 9.3 – 9.6 regarding safe operating pressures, suitable
plumbing, and operator safety.)
8.4 Cesium Solution (Ionization Suppressant)—Dissolve 126.7 g cesium chloride (CsCl), dried at 105°C for 1 h, in water and
dilute to 1 L. Store at room temperature in a high density polyethylene or polypropylene container. Alternatively, commercially
available Cesium ionization suppressants/buffers may be purchased.
8.5 Hydrochloric Acid (1+1)—(1+1, Volume to Volume)—Carefully add one volume of concentrated hydrochloric acid (HCl, sp
gr 1.19) to an equal volume of water.
8.6 Hydrochloric Acid (1+19)—(1+19, Volume to Volume)—Carefully add 50 mL of concentrated hydrochloric acid (HCl, sp
gr 1.19) to 900 mL of water and dilute to 1 L.
8.7 Lanthanum Solution (Releasing Agent)—In a glass 1 L volumetric flask, place 117.3 g of lanthanum oxide (La O ), dried
2 3
at 105°C for 1 h. Wet with water and add HCl (1+1) in small increments until a total of 500 mL of HCl (1+1) has been added.
Cool the solution between additions. Dilute to 1 L with water. Store at room temperature in a high density polyethylene or
polypropylene container. (Warning—Dissolving lanthanum oxide in hydrochloric acid is a strongly exothermic reaction; use
extreme caution when dissolving the reagent. Refer to 9.1 for proper safety precautions when preparing this solution.)
Alternatively, commercially available lanthanum releasing agent solutions may be purchased.
Reagent Chemicals, American Chemical Society SpecificationsACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, , American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
D5086 − 20
8.8 Oxidant (air)—The air may be provided by a compressor or commercially bottled supply. Remove oil, water, and other
foreign matter from the air using a filter recommended by the manufacturer. Refer to the manufacturer’smanufacturer’s guidelines
for recommended delivery pressure.
8.9 Stock Standard Solutions—Stock standard solutions may be purchased as certified solutions or prepared from ACS reagent
grade materials as detailed in 8.9.1 – 8.9.4. Store the solutions at room temperature in high density polyethylene or polypropylene
containers.
8.9.1 Calcium Solution, Stock (1.0 mL = 1.0 mg Ca)—Add 2.497 g of calcium carbonate (CaCO ), dried at 180°C for 1 h, to
approximately 600 mL of water. Add concentrated hydrochl
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...