ASTM E2656-16
(Practice)Standard Practice for Real-time Release Testing of Pharmaceutical Water for the Total Organic Carbon Attribute
Standard Practice for Real-time Release Testing of Pharmaceutical Water for the Total Organic Carbon Attribute
SIGNIFICANCE AND USE
5.1 Pharmaceutical water is the most common component or ingredient used in pharmaceutical and biopharmaceutical manufacturing. Acceptable purity of the water is important to the quality of the final pharmaceutical product. TOC concentration is a key indicator and attribute of the purity of this water and also an important monitor of the overall performance of the water purification system. TOC analysis is the measurement of all the covalently bound carbon present in the water, not including carbon in the form of carbon dioxide (CO2), bicarbonate icon (HCO3 –), or carbonate ion (CO3 2–), and is reported as the mass of organic carbon per volume.
5.2 Application of this practice provides pertinent information to make informed decisions on the release of water meeting pharmaceutical TOC concentration specifications.
SCOPE
1.1 This practice establishes an approach to the real-time release testing (RTRT) of pharmaceutical water based on the total organic carbon (TOC) attribute using on-line total organic carbon (OLTOC) instrumentation that is in agreement with current regulatory thinking.
1.2 This practice is harmonized with or supports the concepts of relevant ASTM International Committee E55 on Manufacture of Pharmaceutical Products standards, ICH Harmonized Tripartite Guidelines, the U.S. FDA PAT Guidance, and U.S. FDA Pharmaceutical cGMPs.
1.3 This practice does not provide general guidance information for pharmaceutical procedures that are considered standard practice in the pharmaceutical industry. This practice provides specific guidance for non-standardized procedures.
1.4 This practice does not address the user’s various internal procedures for risk, change, or quality management systems. The overall project effort associated with this practice shall be proportional to the overall risk of failing the pharmaceutical water’s TOC concentration specification.
1.5 This practice does not purport to establish how to comply with pharmacopeias. The RTRT methodology selected must assure compliance with the user’s current required pharmacopeias. However, compliance with pharmacopeia TOC methods is not necessarily sufficient to meet current regulatory expectations for RTRT.
1.6 This practice does not purport to substitute for or replace compendial bioburden testing requirements. It is strictly applicable to the TOC attribute of water quality.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2016
- Technical Committee
- E55 - Manufacture of Pharmaceutical and Biopharmaceutical Products
- Drafting Committee
- E55.12 - Process Applications
Relations
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Feb-2017
- Effective Date
- 01-May-2015
- Effective Date
- 15-Oct-2012
- Effective Date
- 01-Oct-2012
- Effective Date
- 01-May-2011
- Effective Date
- 01-Oct-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Apr-2008
- Effective Date
- 01-Jan-2008
- Effective Date
- 15-Jun-2007
- Effective Date
- 01-Jun-2007
- Effective Date
- 15-Jul-2006
- Effective Date
- 15-Jan-2006
Overview
ASTM E2656-16 - Standard Practice for Real-time Release Testing of Pharmaceutical Water for the Total Organic Carbon Attribute provides a comprehensive framework for the real-time release testing (RTRT) of pharmaceutical water by monitoring the total organic carbon (TOC) attribute. As pharmaceutical water is the most widely used ingredient in pharmaceutical and biopharmaceutical manufacturing, maintaining its purity is crucial for the quality and safety of final pharmaceutical products. TOC concentration serves as a key indicator of water purity and the performance of the water purification system. This standard outlines the use of on-line total organic carbon (OLTOC) instrumentation in alignment with regulatory and industry best practices for ensuring compliance with TOC specifications.
Key Topics
- TOC as a Water Quality Attribute: TOC analysis measures all covalently bound carbon in water, serving as a sensitive, timely indicator of organic impurities. Proper TOC limits help assure water quality for manufacturing and product safety.
- Real-time Release Testing (RTRT): RTRT enables immediate release of pharmaceutical water by continuously monitoring TOC, reducing delays and supporting real-time decision-making during production.
- On-line TOC Instrumentation (OLTOC): The standard emphasizes configuring and validating OLTOC instruments at suitable sampling points within the water distribution system, often at or after the point-of-use (POU).
- Risk-based Approach: Implementation efforts, from system design to data quality assurance, should be proportional to the risk of failing TOC specifications. Comprehensive risk assessments guide instrument placement, frequency of testing, and control strategies.
- Regulatory Alignment: The practice harmonizes with ASTM Committee E55, ICH Guidelines (Q2, Q7, Q8, Q9, Q10), U.S. FDA PAT Guidance, and cGMPs, ensuring consistency with current global regulatory expectations.
Applications
Adhering to ASTM E2656-16 allows pharmaceutical and biopharmaceutical manufacturers to:
- Ensure Acceptable Water Purity: Continuous TOC monitoring helps maintain water quality, reducing the risk of product contamination and regulatory non-compliance.
- Enhance Operational Efficiency: Real-time detection of TOC deviations supports immediate corrective actions, minimizing production downtime.
- Support Regulatory Compliance: Following this standard aids in meeting FDA, ICH, and Pharmacopoeia requirements for TOC in pharmaceutical water, providing documented proof of compliance.
- Improve Process Control: Automated data collection, analysis, and reporting facilitate proactive quality management and ongoing process improvement.
- Reduce Laboratory Testing Limitations: On-line and real-time TOC analysis avoid issues associated with off-line sample collection, storage, and manual analysis, resulting in more reliable, lower-variability data.
Related Standards
Following ASTM E2656-16 can be complemented by other relevant standards and guidelines, such as:
- ASTM E2281: Process Capability and Performance Measurement
- ASTM E2363: Terminology Relating to Process Analytical Technology
- ASTM E2500: Specification, Design, and Verification of Manufacturing Systems and Equipment
- ASTM E2537: Application of Continuous Quality Verification
- ASTM D4839, D5173, D5904, D5997, D6317: Methods for TOC Measurement in Water
- ICH Guidelines: Q2 (Validation), Q7 (GMP), Q8 (Development), Q9 (Risk Management), Q10 (Quality System)
- ISO 15839: Water Quality-On-line Sensors/Analyzing Equipment
- USP Chapters <643>, <1225>, <1226>, <1231>: TOC Analysis and Water for Pharmaceutical Purposes
- Pharmacopoeial Chapters: (Ph. Eur., JP, USP) for TOC requirements and validation
By implementing ASTM E2656-16, organizations can realize robust pharmaceutical water quality management, ensure patient safety, and meet rigorous regulatory demands through effective TOC-based real-time release testing.
Buy Documents
ASTM E2656-16 - Standard Practice for Real-time Release Testing of Pharmaceutical Water for the Total Organic Carbon Attribute
REDLINE ASTM E2656-16 - Standard Practice for Real-time Release Testing of Pharmaceutical Water for the Total Organic Carbon Attribute
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E2656-16 is a standard published by ASTM International. Its full title is "Standard Practice for Real-time Release Testing of Pharmaceutical Water for the Total Organic Carbon Attribute". This standard covers: SIGNIFICANCE AND USE 5.1 Pharmaceutical water is the most common component or ingredient used in pharmaceutical and biopharmaceutical manufacturing. Acceptable purity of the water is important to the quality of the final pharmaceutical product. TOC concentration is a key indicator and attribute of the purity of this water and also an important monitor of the overall performance of the water purification system. TOC analysis is the measurement of all the covalently bound carbon present in the water, not including carbon in the form of carbon dioxide (CO2), bicarbonate icon (HCO3 –), or carbonate ion (CO3 2–), and is reported as the mass of organic carbon per volume. 5.2 Application of this practice provides pertinent information to make informed decisions on the release of water meeting pharmaceutical TOC concentration specifications. SCOPE 1.1 This practice establishes an approach to the real-time release testing (RTRT) of pharmaceutical water based on the total organic carbon (TOC) attribute using on-line total organic carbon (OLTOC) instrumentation that is in agreement with current regulatory thinking. 1.2 This practice is harmonized with or supports the concepts of relevant ASTM International Committee E55 on Manufacture of Pharmaceutical Products standards, ICH Harmonized Tripartite Guidelines, the U.S. FDA PAT Guidance, and U.S. FDA Pharmaceutical cGMPs. 1.3 This practice does not provide general guidance information for pharmaceutical procedures that are considered standard practice in the pharmaceutical industry. This practice provides specific guidance for non-standardized procedures. 1.4 This practice does not address the user’s various internal procedures for risk, change, or quality management systems. The overall project effort associated with this practice shall be proportional to the overall risk of failing the pharmaceutical water’s TOC concentration specification. 1.5 This practice does not purport to establish how to comply with pharmacopeias. The RTRT methodology selected must assure compliance with the user’s current required pharmacopeias. However, compliance with pharmacopeia TOC methods is not necessarily sufficient to meet current regulatory expectations for RTRT. 1.6 This practice does not purport to substitute for or replace compendial bioburden testing requirements. It is strictly applicable to the TOC attribute of water quality. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 Pharmaceutical water is the most common component or ingredient used in pharmaceutical and biopharmaceutical manufacturing. Acceptable purity of the water is important to the quality of the final pharmaceutical product. TOC concentration is a key indicator and attribute of the purity of this water and also an important monitor of the overall performance of the water purification system. TOC analysis is the measurement of all the covalently bound carbon present in the water, not including carbon in the form of carbon dioxide (CO2), bicarbonate icon (HCO3 –), or carbonate ion (CO3 2–), and is reported as the mass of organic carbon per volume. 5.2 Application of this practice provides pertinent information to make informed decisions on the release of water meeting pharmaceutical TOC concentration specifications. SCOPE 1.1 This practice establishes an approach to the real-time release testing (RTRT) of pharmaceutical water based on the total organic carbon (TOC) attribute using on-line total organic carbon (OLTOC) instrumentation that is in agreement with current regulatory thinking. 1.2 This practice is harmonized with or supports the concepts of relevant ASTM International Committee E55 on Manufacture of Pharmaceutical Products standards, ICH Harmonized Tripartite Guidelines, the U.S. FDA PAT Guidance, and U.S. FDA Pharmaceutical cGMPs. 1.3 This practice does not provide general guidance information for pharmaceutical procedures that are considered standard practice in the pharmaceutical industry. This practice provides specific guidance for non-standardized procedures. 1.4 This practice does not address the user’s various internal procedures for risk, change, or quality management systems. The overall project effort associated with this practice shall be proportional to the overall risk of failing the pharmaceutical water’s TOC concentration specification. 1.5 This practice does not purport to establish how to comply with pharmacopeias. The RTRT methodology selected must assure compliance with the user’s current required pharmacopeias. However, compliance with pharmacopeia TOC methods is not necessarily sufficient to meet current regulatory expectations for RTRT. 1.6 This practice does not purport to substitute for or replace compendial bioburden testing requirements. It is strictly applicable to the TOC attribute of water quality. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM E2656-16 is classified under the following ICS (International Classification for Standards) categories: 11.120.10 - Medicaments; 13.060.50 - Examination of water for chemical substances. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2656-16 has the following relationships with other standards: It is inter standard links to ASTM D5904-02(2024), ASTM D4839-03(2024), ASTM D5904-02(2017), ASTM D6317-15, ASTM E2500-07(2012), ASTM E2281-08a(2012)e1, ASTM D4839-03(2011), ASTM D5997-96(2009), ASTM E2281-08a, ASTM E2281-08, ASTM E2537-08, ASTM D5173-97(2007), ASTM E2500-07, ASTM E2363-06a, ASTM E2363-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2656-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2656 − 16
Standard Practice for
Real-time Release Testing of Pharmaceutical Water for the
Total Organic Carbon Attribute
This standard is issued under the fixed designation E2656; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This practice establishes an approach to the real-time 2.1 ASTM Standards:
release testing (RTRT) of pharmaceutical water based on the E2281 Practice for Process Capability and Performance
total organic carbon (TOC) attribute using on-line total organic Measurement
carbon (OLTOC) instrumentation that is in agreement with E2363 Terminology Relating to Manufacturing of Pharma-
current regulatory thinking. ceutical and Biopharmaceutical Products in the Pharma-
ceutical and Biopharmaceutical Industry
1.2 This practice is harmonized with or supports the con-
E2500 Guide for Specification, Design, and Verification of
cepts of relevant ASTM International Committee E55 on
Pharmaceutical and Biopharmaceutical Manufacturing
Manufacture of Pharmaceutical Products standards, ICH Har-
Systems and Equipment
monized Tripartite Guidelines, the U.S. FDA PAT Guidance,
E2537 Guide for Application of Continuous Process Verifi-
and U.S. FDA Pharmaceutical cGMPs.
cation to Pharmaceutical and Biopharmaceutical Manu-
1.3 This practice does not provide general guidance infor-
facturing
mation for pharmaceutical procedures that are considered
D4839 Test Method for Total Carbon and Organic Carbon in
standard practice in the pharmaceutical industry. This practice
Water by Ultraviolet, or Persulfate Oxidation, or Both, and
provides specific guidance for non-standardized procedures.
Infrared Detection
1.4 This practice does not address the user’s various internal D5173 Guide for On-Line Monitoring of Total Organic
Carbon in Water by Oxidation and Detection of Resulting
procedures for risk, change, or quality management systems.
The overall project effort associated with this practice shall be Carbon Dioxide
D5904 Test Method for Total Carbon, Inorganic Carbon, and
proportional to the overall risk of failing the pharmaceutical
Organic Carbon in Water by Ultraviolet, Persulfate
water’s TOC concentration specification.
Oxidation, and Membrane Conductivity Detection
1.5 This practice does not purport to establish how to
D5997 Test Method for On-Line Monitoring of Total
comply with pharmacopeias. The RTRT methodology selected
Carbon, Inorganic Carbon in Water by Ultraviolet, Persul-
must assure compliance with the user’s current required
fate Oxidation, and Membrane Conductivity Detection
pharmacopeias. However, compliance with pharmacopeia TOC
D6317 Test Method for Low Level Determination of Total
methods is not necessarily sufficient to meet current regulatory
Carbon, Inorganic Carbon and Organic Carbon in Water
expectations for RTRT.
by Ultraviolet, Persulfate Oxidation, and Membrane Con-
1.6 This practice does not purport to substitute for or replace
ductivity Detection
compendial bioburden testing requirements. It is strictly appli-
2.2 Pharmacopoeia Documents:
cable to the TOC attribute of water quality.
ICH Q2 (R1) Validation of Analytical Procedures: Text and
1.7 This standard does not purport to address all of the
Methodology
safety concerns, if any, associated with its use. It is the
ICH Q7 Good Manufacturing Practice Guide for Active
responsibility of the user of this standard to establish appro-
Pharmaceutical Ingredients
priate safety and health practices and determine the applica-
bility of regulatory limitations prior to use.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
This practice is under the jurisdiction of ASTM Committee E55 on Manufac- Standards volume information, refer to the standard’s Document Summary page on
ture of Pharmaceutical and Biopharmaceutical Products and is the direct responsi- the ASTM website.
bility of Subcommittee E55.12 on Process Applications. Available from International Conference on Harmonisation of Technical
Current edition approved Nov. 1, 2016. Published November 2016. Originally Requirements for Registration of Pharmaceuticals for Human Use (ICH), ICH
approved in 2010. Last previous edition approved in 2010 as E2656 – 10. DOI: Secretariat, c/o IFPMA, 15 ch. Louis-Dunant, P.O. Box 195, 1211 Geneva 20,
10.1520/E2656-16. Switzerland, http://www.ich.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2656 − 16
ICH Q8 (R2) Pharmaceutical Development guidelines. The following steps are required to meet the
ICH Q9 Quality Risk Management objectives of this practice.
ICH Q10 Pharmaceutical Quality System 4.1.1 Technical Evaluation—Evaluate and understand water
ISO 15839 Water Quality — On-line Sensors/Analyzing systems, TOC measurement technologies, and the related
Equipment for Water: Specifications and Performance regulatory requirements.
Tests 4.1.2 Risk Assessment—Perform quality risk analysis on the
JP Chapter <2.59> Test for Total Organic Carbon prospective RTRT system designs to establish the sampling
Ph. Eur. Chapter <2.2.44> Total Organic Carbon in Water locations representative of the point-of-use.
for Pharmaceutical Use 4.1.3 Data Quality—Ensure the quality of the data from the
U.S. FDA Part 11 Guidance Guidance for Industry: Part 11, TOC measurement system is suitable for the intended use in
Electronic Records; Electronic Signatures — Scope and the water RTRT system. Ensure equivalency/consistency to
Application data from existing TOC measurement systems used to release
U.S. FDA PAT Guidance Guidance for Industry: PAT — A water to the TOC attribute, if they exist.
Framework for Innovative Pharmaceutical Development, 4.1.4 Implementation Strategies—Develop process to as-
Manufacturing, and Quality Assurance sure successful implementation of RTRT.
U.S. FDA Pharmaceutical cGMPs Pharmaceutical cGMPs 4.1.5 Continuous Verification Procedures—Develop quality
for the 21st Century — A Risk-Based Approach control strategies to ensure consistent system performance.
U.S. FDA Procedures and Methods Validation Guidance for 4.1.6 Continuous Process Improvement—Assess and imple-
Industry: Analytical Procedures and Methods Validation ment process improvement practices.
Chemistry, Manufacturing, and Controls Documentation
5. Significance and Use
U.S. FDA Process Validation Guidance Guidance for Indus-
try: Process Validation: General Principles and Practices
5.1 Pharmaceutical water is the most common component
USP Chapter <643> Total Organic Carbon (TOC)
or ingredient used in pharmaceutical and biopharmaceutical
USP Chapter <1225> Validation of Compendial Procedures
manufacturing. Acceptable purity of the water is important to
USP Chapter <1226> Verification of Compendial Proce-
the quality of the final pharmaceutical product. TOC concen-
dures
tration is a key indicator and attribute of the purity of this water
USP Chapter <1231> Water for Pharmaceutical Purposes
and also an important monitor of the overall performance of the
USP Guidance <1058> Analytical Instrument Qualification
water purification system. TOC analysis is the measurement of
all the covalently bound carbon present in the water, not
3. Terminology
including carbon in the form of carbon dioxide (CO ), bicar-
– 2–
3.1 For definitions of terms specific to this standard, refer to
bonate icon (HCO ), or carbonate ion (CO ), and is reported
3 3
the Terminology sections of Practice E2281, Terminology as the mass of organic carbon per volume.
E2363, and Guide E2500. Refer to ICH Q2 (R1) for method
5.2 Application of this practice provides pertinent informa-
validation terminology.
tion to make informed decisions on the release of water
meeting pharmaceutical TOC concentration specifications.
4. Summary of Practice
4.1 This practice provides the user with sufficient guidance
6. Procedure
for developing the scientific and risk-based information nec-
6.1 Technical Evaluation:
essary to make informed decisions on the implementation,
6.1.1 The overall project scope shall be proportional to the
continuous verification, and continuous improvement of a
associated risk of exceeding the pharmaceutical water TOC
system to provide the real-time release testing of pharmaceu-
concentration specifications. Knowledge and understanding of
tical water using on-line total organic carbon (RTRT-OLTOC)
the TOC concentration in the water system, the OLTOC
instrumentation that meets pharmaceutical water TOC specifi-
measurement system technology performance, and the phar-
cations. This guidance is based on Practice E2281, Terminol-
maceutical water system design shall be acquired to minimize
ogy E2363, and Guide E2500 standards as well as
risk, ensure correct quality decisions, and maximize return on
ICH Q2 (R1), ICH Q7, ICH Q8 (R1), ICH Q9, and ICH Q10
investment (USP Chapter <1231> and (1-7) ). TOC measure-
ment technologies are referenced in Test Methods D4839,
D5904, D5997, and D6317, and Guide D5173.
Available from International Organization for Standardization (ISO), 1, ch. de
6.1.2 Technical assessments should be conducted to evalu-
la Voie-Creuse, Case postale 56, CH-1211, Geneva 20, Switzerland, http://
ate and develop a low-risk, science-based RTRT-OLTOC
www.iso.ch.
Available from Japanese Pharmacopoeia (JP), Standards Division, Office of
system design. Knowledge of related information from avail-
Compliance and Standards, Pharmaceuticals and Medical Devices Agency (PMDA),
able sources should be used to understand, interpret, and
Shin-kasumigaseki Building, 3-3-2, Kasumigaseki, Chiyoda-ku, Tokyo 100-0013,
implement the results of the technical assessments. Information
Japan, http://www.std.pmda.go.jp.
Available from European Pharmacopoeia (Ph. Eur.), 7 allée Kastner, CS 30026, on general and specific RTRT-OLTOC system design
F67081 Strasbourg, France, http://www.pheur.org.
Available from Food and Drug Administration (FDA), 5600 Fishers Ln.,
Rockville, MD 20857, http://www.fda.gov.
8 9
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, The boldface numbers in parentheses refer to a list of references at the end of
MD 20852-1790, http://www.usp.org. this standard.
E2656 − 16
considerations, performance characteristics, and validation procedure must be verified under actual conditions of use.
should be found in published documents and texts (8-15). Users shall use USP Chapter <1226>, ICH Q2 (R1), or equiva-
6.1.3 For existing water purification systems, the user lent to verify compendial procedures.
should assess historical, current, and potential organic contami-
6.1.5.2 The procedure for validation and verification of the
nation. Evaluation of potential organic contamination should
TOC analytical method shall depend on the analytical proce-
be based on a realistic assessment of water system design and
dure classification in ICH Q2 (R1), USP Chapter <1225>, or
components to determine the probability of a specific or a
the U.S. FDA Procedures and Methods Validation. The mea-
broad spectrum of organic contaminants reaching the water
surement of the TOC attribute in water shall be classified as an
distribution system. The user should consult with TOC instru-
impurity test. Under impurity tests are two additional
mentation vendors to determine if the TOC measurement
classifications, quantitative and limit test. For each of these,
system will meet the requirements of the intended application
there are recommended lists of validation tests to perform. All
in light of any organic contamination assessment.
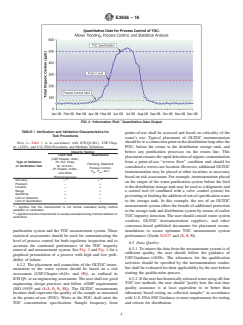
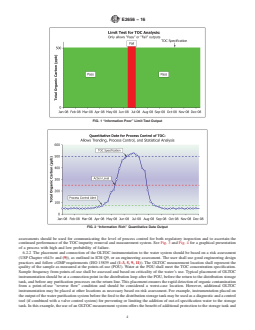
pharmacopeia TOC test methods are limit tests. Limit testing
6.1.4 For new water purification systems, the presence of
produces only a pass or fail output as graphically represented
potential problematic compounds in the pharmaceutical water
by Fig. 1. To control, trend, and monitor on-line systems and to
system shall be addressed during the design and qualification
release water in real time using quantitative data, the analytical
and validation activities and correction/mitigation/preventive
method requires the use of quantitative data, so the analytical
actions shall be implemented accordingly.
method shall be validated to the requirements of quantitative
6.1.5 TOC measurement system technology assessments
tests (U.S. FDA PAT Guidance). Quantitative data use is
shall be achieved by meeting regulatory guidance requirements
graphically represented in Fig. 2. Classifications and recom-
on analytical procedure verifications and validations
mended tests are shown in Table 1. Additional helpful infor-
(ICH Q2 (R1), USP Chapter <1225>, and U.S. FDA Proce-
mation can be found in ISO 15839.
dures and Methods Validation). The requirements shall depend
6.1.5.3 The U.S. FDA considers “real-time release to be
on the use of the data and the intended use of the instrumen-
comparable to Alternative Analytical Procedures” and the U.S.
tation.
Regulation CFR 211.165 requires that the accuracy, sensitivity,
6.1.5.1 Legal U.S. Requirements and Verification of
specificity, and reproducibility of the alternative analytical test
USP Chapter <643>—The use of USP Chapter <643> TOC is
methods or procedures used for process control purposes be
legally recognized to meet the requirements for testing the
validated and documented appropriately (U.S. FDA PAT Guid-
TOC attribute in pharmaceutical water. The users of
ance and U.S. FDA Procedures and Methods Validation).
USP Chapter <643> TOC are not required to validate this
6.2 Risk Assessment:
practice, but they shall verify it is suitable under actual
conditions of use. The user shall understand that Section 6.2.1 If the TOC concentration data is to be used in a
501(b) of the U.S. Food, Drug, and Cosmetic Act (the Act) quantitative way for trending, process control, or process
legally recognizes the analytical procedures in the U.S. statistical analysis, a statistical assessment of the process
Pharmacopeia/National Formulary (USP/NF) for purposes of performance should be done to estimate the risk of the process
determining compliance with this Act (U.S. FDA Procedures
failing the specification requirement. This information should
and Methods Validation). The U.S. Federal Regulation CFR be used in the project implementation phase to understand and
211.194(a)(2) states: the suitability of a compendial analytical improve, if necessary, the combined performance of the water
FIG. 1 “Information Poor” Limit Test Output
E2656 − 16
FIG. 2 “Information Rich” Quantitative Data Output
TABLE 1 Verification and Validation Characteristics for
points-of-use shall be assessed and based on criticality of the
Test Procedures
water’s use. Typical placement of OLTOC instrumentation
NOTE 1—Table 1 is in accordance with ICH Q2 (R1), USP Chap- should be at a connection point in the distribution loop after the
ter <1225>, and U.S. FDA Procedures and Methods Validation.
POU, before the return to the distribution storage tank, and
Impurity Testing
before any purification processes on the return line. This
Limit Test Quantitative
placement ensures the rapid detection of organic contamination
(USP Chapter <643>,
from a point-of-use “reverse flow” condition and should be
Type of Validation Ph. Eur. Chap-
(Trending, Statistical
or Verification Test ter <2.2.44>,
considered a worst-case location. However, additional OLTOC
Process Control,
JP Chapter <2.59>,
C , P , etc.)
pk pk instrumentation may be placed at other locations as necessary
and Other
Pharmacopoeias) based on risk assessment. For example, instrumentation placed
A B
Accuracy – +
on the output of the water purification system before the feed
Precision – +
to the distribution storage tank may be used as a diagnostic and
Linearity – +
Range – +
a control tool (if combined with a valve control system) for
Specificity + +
preventing or limiting the addition of out-of-specification water
Limit of Detection + –
Limit of Quantitation – + to the storage tank. In this example, the use of an OLTOC
A
measurement system offers the benefit of additional protection
– signifies that this characteristic is not normal evaluated during method
validation or verification.
to the storage tank and distribution system by means of earlier
B
+ signifies that this characteristic is usually evaluated during method validation or
TOC impurity detection. The user should consult water system
verification.
vendors, OLTOC instrumentation suppliers, and other
consensus-based published documents for placement recom-
mendations to assure optimum TOC measurement system
purification system and the TOC measurement system. These
performance (Guide E2537 and (3, 5, 9)).
statistical assessments should be used for communicating the
level of process control for both regulatory inspection and to
6.3 Data Quality:
ascertain the continued performance of the TOC impurity
6.3.1 To ensure the data from the measurement system is of
removal and measurement system. See Fig. 3 and Fig. 4 for a
sufficient quality, the user should follow the guidance of
graphical presentation of a process with high and low prob-
USP Guidance <1058>. The tolerances for the qualification
ability of failure.
activities should be specified by the instrumentation vendor,
6.2.2 The placement and connection of the OLTOC instru-
but shall be evaluated for their applicability by the user before
mentation to the water system should be based on a risk
starting the qualification process.
assessment (USP Chapter <643> and (9)), as outlined in
6.3.2 If the user has historically released water using off-line
ICH Q9, or an engineering assessment. The user shall use good
TOC test methods, the user should “justify how the real time
engineering design practices and follow cGMP requirements
quality assurance is at least equivalent to or better than
(ISO 15839 and (1-3, 5, 9, 11)). The OLTOC measurement
location shall represent the quality of the sample as measured laboratory based testing on collected samples” in accordance
with U.S. FDA PAT Guidance to meet requirements for testing
at the points-of-use (POU). Water at the POU shall meet the
TOC concentration specification.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2656 − 10 E2656 − 16
Standard Practice for
Real-time Release Testing of Pharmaceutical Water for the
Total Organic Carbon Attribute
This standard is issued under the fixed designation E2656; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice establishes an approach to the real-time release testing (RTRT) of pharmaceutical water based on the total
organic carbon (TOC) attribute using on-line total organic carbon (OLTOC) instrumentation that is in agreement with current
regulatory thinking.
1.2 This practice is harmonized with or supports the concepts of relevant ASTM International Committee E55 on Manufacture
of Pharmaceutical Products standards, ICH Harmonized Tripartite Guidelines, the USU.S. FDA PAT Guidance, and USU.S. FDA
Pharmaceutical cGMPs.
1.3 This practice does not provide general guidance information for pharmaceutical procedures that are considered standard
practice in the pharmaceutical industry. This practice provides specific guidance for non-standardized procedures.
1.4 This practice does not address the user’s various internal procedures for risk, change, or quality management systems. The
overall project effort associated with this practice shall be proportional to the overall risk of failing the pharmaceutical water’s TOC
concentration specification.
1.5 This practice does not purport to establish how to comply with pharmacopeias. The RTRT methodology selected must assure
compliance with the user’s current required pharmacopeias. However, compliance with pharmacopeia TOC methods is not
necessarily sufficient to meet current regulatory expectations for RTRT.
1.6 This practice does not purport to substitute for or replace compendial bioburden testing requirements. It is strictly applicable
to the TOC attribute of water quality.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
E2281 Practice for Process Capability and Performance Measurement
E2363 Terminology Relating to Process Analytical Technology in the Pharmaceutical Industry
E2500 Guide for Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing Systems and
Equipment
E2537 Guide for Application of Continuous Quality Verification to Pharmaceutical and Biopharmaceutical Manufacturing
D4839 Test Method for Total Carbon and Organic Carbon in Water by Ultraviolet, or Persulfate Oxidation, or Both, and Infrared
Detection
D5173 Guide for On-Line Monitoring of Total Organic Carbon in Water by Oxidation and Detection of Resulting Carbon
Dioxide
D5904 Test Method for Total Carbon, Inorganic Carbon, and Organic Carbon in Water by Ultraviolet, Persulfate Oxidation, and
Membrane Conductivity Detection
This practice is under the jurisdiction of ASTM Committee E55 on Manufacture of Pharmaceutical and Biopharmaceutical Products and is the direct responsibility of
Subcommittee E55.03 on General Pharmaceutical Standards.
Current edition approved Aug. 1, 2010Nov. 1, 2016. Published October 2010November 2016. Originally approved in 2010. Last previous edition approved in 2010 as
E2656 – 10. DOI: 10.1520/E2656-10.10.1520/E2656-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2656 − 16
D5997 Test Method for On-Line Monitoring of Total Carbon, Inorganic Carbon in Water by Ultraviolet, Persulfate Oxidation,
and Membrane Conductivity Detection
D6317 Test Method for Low Level Determination of Total Carbon, Inorganic Carbon and Organic Carbon in Water by
Ultraviolet, Persulfate Oxidation, and Membrane Conductivity Detection
2.2 Pharmacopoeia Documents:
ICH Q2 (R1) Validation of Analytical Procedures: Text and Methodology
ICH Q7 Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients
ICH Q8 (R1)ICH Q8 (R2) Pharmaceutical Development
ICH Q9 Quality Risk Management
ICH Q10 Pharmaceutical Quality System
ISO 15839 Water Quality — On-line Sensors/Analyzing Equipment for Water: Specifications and Performance Tests
JP Chapter <2.59> Test for Total Organic Carbon
Ph. Eur. Chapter <2.2.44> Total Organic Carbon in Water for Pharmaceutical Use
U.S. FDA Part 11 Guidance Guidance for Industry: Part 11, Electronic Records; Electronic Signatures — Scope and Application
USU.S. FDA PAT Guidance Guidance for Industry: PAT — A Framework for Innovative Pharmaceutical Development,
Manufacturing, and Quality Assurance
USU.S. FDA Pharmaceutical cGMPs Pharmaceutical cGMPs for the 21st Century — A Risk-Based Approach
USU.S. FDA Procedures and Methods Validation Guidance for Industry: Analytical Procedures and Methods Validation
Chemistry, Manufacturing, and Controls Documentation
U.S. FDA Process Validation Guidance Guidance for Industry: Process Validation: General Principles and Practices
USP Chapter <643> Total Organic Carbon (TOC)
USP Chapter <1225> Validation of Compendial Procedures
USP Chapter <1226> Verification of Compendial Procedures
USP Chapter <1231> Water for Pharmaceutical Purposes
USP Guidance <1058> Analytical Instrument Qualification
3. Terminology
3.1 For definitions of terms specific to this standard, refer to the Terminology sections of Practice E2281, Terminology E2363,
and Guide E2500. Refer to ICH Q2 (R1) for method validation terminology.
4. Summary of Practice
4.1 This practice provides the user with sufficient guidance for developing the scientific and risk-based information necessary
to make informed decisions on the implementation, continuous verification, and continuous improvement of a system to provide
the real-time release testing of pharmaceutical water using on-line total organic carbon (RTRT-OLTOC) instrumentation that meets
pharmaceutical water TOC specifications. This guidance is based on Practice E2281, Terminology E2363, and Guide E2500
standards as well as ICH Q2 (R1), ICH Q7, ICH Q8 (R1), ICH Q9, and ICH Q10 guidelines. The following steps are required to
meet the objectives of this practice.
4.1.1 Technical Evaluation—Evaluate and understand water systems, TOC measurement technologies, and the related
regulatory requirements.
4.1.2 Risk Assessment—Perform quality risk analysis on the prospective RTRT system designs to establish the sampling
locations representative of the point-of-use.
4.1.3 Data Quality—Ensure the quality of the data from the TOC measurement system is suitable for the intended use in the
water RTRT system. Ensure equivalency/consistency to data from existing TOC measurement systems used to release water to the
TOC attribute, if they exist.
4.1.4 Implementation Strategies—Develop process to assure successful implementation of RTRT.
4.1.5 Continuous Verification Procedures—Develop quality control strategies to ensure consistent system performance.
4.1.6 Continuous Process Improvement—Assess and implement process improvement practices.
5. Significance and Use
5.1 Pharmaceutical water is the most common component or ingredient used in pharmaceutical and biopharmaceutical
manufacturing. Acceptable purity of the water is important to the quality of the final pharmaceutical product. TOC concentration
Available from International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), ICH Secretariat, c/o
IFPMA, 15 ch. Louis-Dunant, P.O. Box 195, 1211 Geneva 20, Switzerland, http://www.ich.org.
Available from International Organization for Standardization (ISO), 1, ch. de la Voie-Creuse, Case postale 56, CH-1211, Geneva 20, Switzerland, http://www.iso.ch.
Available from Japanese Pharmacopoeia (JP), Standards Division, Office of Compliance and Standards, Pharmaceuticals and Medical Devices Agency (PMDA),
Shin-kasumigaseki Building, 3-3-2, Kasumigaseki, Chiyoda-ku, Tokyo 100-0013, Japan, http://www.std.pmda.go.jp.
Available from European Pharmacopoeia (Ph. Eur.), 7 allée Kastner, CS 30026, F67081 Strasbourg, France, http://www.pheur.org.
Available from Food and Drug Administration (FDA), 5600 Fishers Ln., Rockville, MD 20857, http://www.fda.gov.
Available from U.S. Pharmacopeia (USP), 12601 Twinbrook Pkwy., Rockville, MD 20852-1790, http://www.usp.org.
E2656 − 16
is a key indicator and attribute of the purity of this water and also an important monitor of the overall performance of the water
purification system. TOC analysis is the measurement of all the covalently bound carbon present in the water, not including carbon
– 2–
in the form of carbon dioxide (CO ), bicarbonate icon (HCO ), or carbonate ion (CO ), and is reported as the mass of organic
2 3 3
carbon per volume.
5.2 Application of this practice provides pertinent information to make informed decisions on the release of water meeting
pharmaceutical TOC concentration specifications.
6. Procedure
6.1 Technical Evaluation:
6.1.1 The overall project scope shall be proportional to the associated risk of exceeding the pharmaceutical water TOC
concentration specifications. Knowledge and understanding of the TOC concentration in the water system, the OLTOC
measurement system technology performance, and the pharmaceutical water system design shall be acquired to minimize risk,
ensure correct quality decisions, and maximize return on investment (USP Chapter <1231> and (1-7) ). TOC measurement
technologies are referenced in Test Methods D4839, D5173, D5904, D5997, and D6317, and Guide D5173.
6.1.2 Technical assessments should be conducted to evaluate and develop a low-risk, science-based RTRT-OLTOC system
design. Knowledge of related information from available sources should be used to understand, interpret, and implement the results
of the technical assessments. Information on general and specific RTRT-OLTOC system design considerations, performance
characteristics, and validation should be found in published documents and texts (8-15).
6.1.3 For existing water purification systems, the user should assess historical, current, and potential organic contamination.
Evaluation of potential organic contamination should be based on a realistic assessment of water system design and components
to determine the probability of a specific or a broad spectrum of organic contaminants reaching the water distribution system. The
user should consult with TOC instrumentation vendors to determine if the TOC measurement system will meet the requirements
of the intended application in light of any organic contamination assessment.
6.1.4 For new water purification systems, the presence of potential problematic compounds in the pharmaceutical water system
shall be addressed during the design and qualification and validation activities and correction/mitigation/preventive actions shall
be implemented accordingly.
6.1.5 TOC measurement system technology assessments shall be achieved by meeting regulatory guidance requirements on
analytical procedure verifications and validations (ICH Q2 (R1), USP Chapter <1225>, and USU.S. FDA Procedures and Methods
Validation). The requirements shall depend on the use of the data and the intended use of the instrumentation.
6.1.5.1 Legal USAU.S. Requirements and Verification of USP Chapter <643>—The use of USP Chapter <643> TOC is legally
recognized to meet the requirements for testing the TOC attribute in pharmaceutical water. The users of USP Chapter <643> TOC
are not required to validate this method,practice, but they shall verify it is suitable under actual conditions of use. The user shall
understand that Section 501(b) of the United States U.S. Food, Drug, and Cosmetic Act (the Act) legally recognizes the analytical
procedures in the United States U.S. Pharmacopeia/National Formulary (USP/NF) for purposes of determining compliance with
this Act (US(U.S. FDA Procedures and Methods Validation). The USA federal regulationU.S. Federal Regulation CFR
211.194(a)(2) states: the suitability of a compendial analytical procedure must be verified under actual conditions of use. Users
shall use USP Chapter <1226>, ICH Q2 (R1), or equivalent to verify compendial procedures.
6.1.5.2 The procedure for validation and verification of the TOC analytical method shall depend on the analytical procedure
classification in ICH Q2 (R1), USP Chapter <1225>, or the USU.S. FDA Procedures and Methods Validation. The measurement
of the TOC attribute in water shall be classified as an impurity test. Under impurity tests are two additional classifications,
quantitative and limit test. For each of these, there are recommended lists of validation tests to perform. All pharmacopeia TOC
test methods are limit tests. Limit testing produces only a pass or fail output as graphically represented by Fig. 1. To control, trend,
and monitor on-line systems and to release water in real time using quantitative data, the analytical method requires the use of
quantitative data, so the analytical method shall be validated to the requirements of quantitative tests (US(U.S. FDA PAT
Guidance). Quantitative data use is graphically represented in Fig. 2. Classifications and recommended tests are shown in Table
1. Additional helpful information can be found in ISO 15839.
6.1.5.3 The USU.S. FDA considers “real-time release to be comparable to Alternative Analytical Procedures” and the USU.S.
Regulation CFR 211.165 requires that the accuracy, sensitivity, specificity, and reproducibility of the alternative analytical test
methods or procedures used for process control purposes be validated and documented appropriately (US(U.S. FDA PAT Guidance
and USU.S. FDA Procedures and Methods Validation).
6.2 Risk Assessment:
6.2.1 If the TOC concentration data is to be used in a quantitative way for trending, process control, or process statistical
analysis, a statistical assessment of the process performance should be done to estimate the risk of the process failing the
specification requirement. This information should be used in the project implementation phase to understand and improve, if
necessary, the combined performance of the water purification system and the TOC measurement system. These statistical
The boldface numbers in parentheses refer to a list of references at the end of this standard.
E2656 − 16
FIG. 1 “Information Poor” Limit Test Output
FIG. 2 “Information Rich” Quantitative Data Output
assessments should be used for communicating the level of process control for both regulatory inspection and to ascertain the
continued performance of the TOC impurity removal and measurement system. See Fig. 3 and Fig. 4 for a graphical presentation
of a process with high and low probability of failure.
6.2.2 The placement and connection of the OLTOC instrumentation to the water system should be based on a risk assessment
(USP Chapter <643> and (9)), as outlined in ICH Q9, or an engineering assessment. The user shall use good engineering design
practices and follow cGMP requirements (ISO 15839 and (1-3, 5, 9, 11)). The OLTOC measurement location shall represent the
quality of the sample as measured at the points-of-use (POU). Water at the POU shall meet the TOC concentration specification.
Sample frequency from points-of-use shall be assessed and based on criticality of the water’s use. Typical placement of OLTOC
instrumentation should be at a connection point in the distribution loop after the POU, before the return to the distribution storage
tank, and before any purification processes on the return line. This placement ensures the rapid detection of organic contamination
from a point-of-use “reverse flow” condition and should be considered a worst-case location. However, additional OLTOC
instrumentation may be placed at other locations as necessary based on risk assessment. For example, instrumentation placed on
the output of the water purification system before the feed to the distribution storage tank may be used as a diagnostic and a control
tool (if combined with a valve control system) for preventing or limiting the addition of out-of-specification water to the storage
tank. In this example, the use of an OLTOC measurement system offers the benefit of additional protection to the storage tank and
E2656 − 16
TABLE 1 Verification and Validation Characteristics for
Test Procedures
NOTE 1—Table 1 is in accordance with ICH Q2 (R1), USP Chap-
ter <1225>, and USU.S. FDA Procedures and Methods Validation.
Impurity Testing
Limit Test Quantitative
(USP Chapter <643>,
Type of Validation Ph. Eur. Chap-
(Trending, Statistical
or Verification Test ter <2.2.44>,
Process Control,
JP Chapter <2.59>,
C , P , etc.)
pk pk
and Other
Pharmacopoeias)
A B
Accuracy – +
Precision – +
Linearity – +
Range – +
Specificity + +
Limit of Detection + –
Limit of Quantitation – +
A
– signifies that this characteristic is not normal evaluated during method
validation or verification.
B
+ signifies that this characteristic is usually evacuatedevaluated during method
validation or verification.
distribution system by means of earlier TOC impurity detection. The user should consult water system vendors, OLTOC
instrumentation suppliers, and other consensus-based published documents for placement recommendations to assure optimum
TOC measurement system performance (Guide E2537 and (3, 5, 9)).
6.3 Data Quality:
6.3.1 To ensure the data from the measurement system is of sufficient quality, the user should follow the guidance of
USP Guidance <1058>. The tolerances for the qualification activities should be specified by the instrumentation vendor, but shall
be evaluated for their applicability by the user before starting the qualification process.
6.3.2 If the user has historically released water using off-line TOC test methods, the user should “justify how the real time
quality assurance is at least equivalent to or better than laboratory based testing on collected samples” in accordance with USU.S.
FDA PAT Guidance to meet requirements for testing and release for distribution.
6.3.3 Alternatively, if the user has not historically rele
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...