ASTM D7318-19e1
(Test Method)Standard Test Method for Existent Inorganic Sulfate in Ethanol by Potentiometric Titration
Standard Test Method for Existent Inorganic Sulfate in Ethanol by Potentiometric Titration
SIGNIFICANCE AND USE
5.1 Ethanol is used as a blending agent added to gasoline. Sulfates are indicated in filter plugging deposits and fuel injector deposits. When fuel ethanol is burned, sulfates may contribute to sulfuric acid emissions. Ethanol acceptability for use depends on the sulfate content. Sulfate content, as measured by this test method, can be used as one measure of determination of the acceptability of ethanol for automotive spark-ignition engine fuel use.
SCOPE
1.1 This test method covers a potentiometric titration procedure for determining the existent inorganic sulfate content of hydrous, anhydrous ethanol, and anhydrous denatured ethanol, which is added as a blending agent with spark ignition fuels. It is intended for the analysis of denatured ethanol samples containing between 1.0 mg/kg to 20 mg/kg existent inorganic sulfate.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards prior to usage.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 13-Feb-2019
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.03 - Elemental Analysis
Relations
- Effective Date
- 01-Dec-2023
- Effective Date
- 15-Dec-2017
- Effective Date
- 15-Nov-2017
- Effective Date
- 01-Jan-2017
- Effective Date
- 01-Oct-2013
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Nov-2009
- Effective Date
- 15-Oct-2008
- Effective Date
- 01-Nov-2007
- Effective Date
- 01-Nov-2007
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Jun-2002
Overview
ASTM D7318-19e1 - Standard Test Method for Existent Inorganic Sulfate in Ethanol by Potentiometric Titration - provides a robust and reliable procedure for determining the concentration of existent inorganic sulfate in ethanol. This method is essential in assessing the suitability of hydrous and anhydrous ethanol, including denatured ethanol, as a blending agent for spark-ignition engine fuels. The standard employs potentiometric titration, using a lead ion-selective electrode, to detect sulfate concentrations within the 1.0 mg/kg to 20 mg/kg range. Maintaining low sulfate content in ethanol minimizes the risk of equipment fouling, corrosion, and sulfuric acid emissions in automotive engines.
Key Topics
- Scope of Testing: The standard specifies the use of potentiometric titration to quantify inorganic sulfate species that are present without oxidative conversion. The method is applicable to hydrous, anhydrous, and denatured ethanol samples.
- Test Method Details:
- Utilizes a lead ion-selective electrode and a double junction reference electrode.
- Employs perchloric acid to mitigate interference from carbonates.
- Results reported in SI units (mg/kg).
- Analytical Range: Designed for samples containing between 1.0 mg/kg and 20 mg/kg of inorganic sulfate.
- Quality Assurance: Recommends regular quality control checks and control charting to verify instrument and procedural consistency.
- Precision: Outlines repeatability and reproducibility values to guide laboratories in maintaining method accuracy.
Applications
- Fuel Quality Assessment: Used widely by fuel blenders, ethanol producers, and refineries to confirm ethanol compliance with regulatory and internal standards for sulfate content. Acceptance of ethanol for use in automotive fuels is often contingent upon sulfate analysis results.
- Environmental Impact Reduction: Limiting sulfate concentrations in fuel ethanol helps reduce the formation of filter and injector deposits and curbs sulfuric acid emissions during combustion, contributing to cleaner air.
- Industrial Quality Control: The method supports routine monitoring and batch release testing, ensuring that ethanol shipments meet industry requirements and do not compromise engine function or emissions control systems.
Related Standards
- ASTM D4057 - Practice for Manual Sampling of Petroleum and Petroleum Products: Guidance on sample collection to ensure representativity and avoid cross-contamination.
- ASTM D4177 - Practice for Automatic Sampling of Petroleum and Petroleum Products: Procedures for obtaining automatic, representative samples for analysis.
- ASTM D4052 - Test Method for Density, Relative Density, and API Gravity of Liquids by Digital Density Meter: Important for calculating the conversion of sulfate concentrations from mg/kg to mg/L.
- ASTM D1193 - Specification for Reagent Water: Specifies the quality of water required for reagent preparation and analysis.
- ASTM D6299 - Practice for Applying Statistical Quality Assurance and Control Charting Techniques: Provides the statistical framework for maintaining process control and data integrity in analytical laboratories.
Practical Value
Adhering to ASTM D7318-19e1 ensures accurate determination of existent inorganic sulfate, which aids in:
- Protecting fuel system components from sulfate-induced fouling and corrosion.
- Minimizing operational hazards by identifying and controlling sulfate impurities before ethanol blending.
- Facilitating regulatory compliance and supporting environmentally responsible fuel formulation.
- Strengthening the reliability and reputation of fuel ethanol supply chains through standardized, internationally recognized analytical practices.
By implementing this test method, laboratories and ethanol producers can maintain high standards for product quality and contribute to both engine performance and emission reduction initiatives. For organizations involved in fuel quality control and environmental compliance, ASTM D7318-19e1 is a critical reference for effective inorganic sulfate analysis in ethanol.
Buy Documents
ASTM D7318-19e1 - Standard Test Method for Existent Inorganic Sulfate in Ethanol by Potentiometric Titration
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

ECOCERT
Organic and sustainability certification.

Element Materials Technology
Materials testing and product certification.
Sponsored listings
Frequently Asked Questions
ASTM D7318-19e1 is a standard published by ASTM International. Its full title is "Standard Test Method for Existent Inorganic Sulfate in Ethanol by Potentiometric Titration". This standard covers: SIGNIFICANCE AND USE 5.1 Ethanol is used as a blending agent added to gasoline. Sulfates are indicated in filter plugging deposits and fuel injector deposits. When fuel ethanol is burned, sulfates may contribute to sulfuric acid emissions. Ethanol acceptability for use depends on the sulfate content. Sulfate content, as measured by this test method, can be used as one measure of determination of the acceptability of ethanol for automotive spark-ignition engine fuel use. SCOPE 1.1 This test method covers a potentiometric titration procedure for determining the existent inorganic sulfate content of hydrous, anhydrous ethanol, and anhydrous denatured ethanol, which is added as a blending agent with spark ignition fuels. It is intended for the analysis of denatured ethanol samples containing between 1.0 mg/kg to 20 mg/kg existent inorganic sulfate. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards prior to usage. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Ethanol is used as a blending agent added to gasoline. Sulfates are indicated in filter plugging deposits and fuel injector deposits. When fuel ethanol is burned, sulfates may contribute to sulfuric acid emissions. Ethanol acceptability for use depends on the sulfate content. Sulfate content, as measured by this test method, can be used as one measure of determination of the acceptability of ethanol for automotive spark-ignition engine fuel use. SCOPE 1.1 This test method covers a potentiometric titration procedure for determining the existent inorganic sulfate content of hydrous, anhydrous ethanol, and anhydrous denatured ethanol, which is added as a blending agent with spark ignition fuels. It is intended for the analysis of denatured ethanol samples containing between 1.0 mg/kg to 20 mg/kg existent inorganic sulfate. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Material Safety Data Sheets are available for reagents and materials. Review them for hazards prior to usage. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7318-19e1 is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis; 75.160.20 - Liquid fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7318-19e1 has the following relationships with other standards: It is inter standard links to ASTM D6299-23a, ASTM D6299-17b, ASTM D6299-17a, ASTM D6299-17, ASTM D6299-13e1, ASTM D4057-06(2011), ASTM D6299-10e2, ASTM D6299-10, ASTM D6299-09, ASTM D6299-08, ASTM D6299-07, ASTM D6299-07e1, ASTM D1193-06, ASTM D6299-02, ASTM D6299-02e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7318-19e1 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
´1
Designation: D7318 − 19
Standard Test Method for
Existent Inorganic Sulfate in Ethanol by Potentiometric
Titration
This standard is issued under the fixed designation D7318; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
NOTE—Subsection 11.2.1 was corrected and the yeardate changed on February 14, 2019.
Footnote 1 was corrected editorially in December 2019.
1. Scope* D4057 Practice for Manual Sampling of Petroleum and
Petroleum Products
1.1 This test method covers a potentiometric titration pro-
D4177 Practice for Automatic Sampling of Petroleum and
cedure for determining the existent inorganic sulfate content of
Petroleum Products
hydrous, anhydrous ethanol, and anhydrous denatured ethanol,
D6299 Practice for Applying Statistical Quality Assurance
which is added as a blending agent with spark ignition fuels. It
and Control Charting Techniques to Evaluate Analytical
is intended for the analysis of denatured ethanol samples
Measurement System Performance
containing between 1.0 mg⁄kg to 20 mg⁄kg existent inorganic
sulfate.
3. Terminology
1.2 The values stated in SI units are to be regarded as
3.1 Definitions:
standard. No other units of measurement are included in this
3.1.1 existent inorganic sulfate, n—inorganic sulfate species
standard.
actually present in the sample at the time of analysis with no
1.3 This standard does not purport to address all of the
oxidation treatment.
safety concerns, if any, associated with its use. It is the
3.1.1.1 Discussion—Specifically in this test method, inor-
responsibility of the user of this standard to establish appro-
ganic sulfate is present as sulfate in ethanol.
priate safety, health, and environmental practices and deter-
–2
3.1.2 inorganic sulfate, n—sulfate (SO ) species present
mine the applicability of regulatory limitations prior to use.
as sulfuric acid, ionic salts of this acid, or mixtures of these.
Material Safety Data Sheets are available for reagents and
materials. Review them for hazards prior to usage.
4. Summary of Test Method
1.4 This international standard was developed in accor-
4.1 An ethanol sample containing inorganic sulfate is
dance with internationally recognized principles on standard-
titrated in ethanolic medium with a standard lead nitrate
ization established in the Decision on Principles for the
solution. Lead sulfate precipitate is formed during the titration.
Development of International Standards, Guides and Recom-
Perchloric acid is added to remove possible interference from
mendations issued by the World Trade Organization Technical
carbonate. The endpoint is signaled by an increase in lead ion
Barriers to Trade (TBT) Committee.
activity, as measured by a lead-selective electrode.
2. Referenced Documents
5. Significance and Use
2.1 ASTM Standards:
D1193 Specification for Reagent Water
5.1 Ethanol is used as a blending agent added to gasoline.
D4052 Test Method for Density, Relative Density, and API
Sulfates are indicated in filter plugging deposits and fuel
Gravity of Liquids by Digital Density Meter
injector deposits. When fuel ethanol is burned, sulfates may
contribute to sulfuric acid emissions. Ethanol acceptability for
use depends on the sulfate content. Sulfate content, as mea-
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of sured by this test method, can be used as one measure of
Subcommittee D02.03 on Elemental Analysis.
determination of the acceptability of ethanol for automotive
Current edition approved Feb. 14, 2019. Published February 2019. Originally
spark-ignition engine fuel use.
approved in 2007. Last previous edition approved in 2013 as D7318 – 13. DOI:
10.1520/D7318-19E01.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or 6. Apparatus
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
6.1 Potentiometric Titration Assembly—A titration assem-
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. bly consisting of an automatic titrator fitted with a lead
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
´1
D7318 − 19
ion-selective electrode, a double-junction reference electrode, 7.6 Lithium Chloride—99+ %, A.C.S. reagent grade.
buret, and stirring is used. Stirring may be accomplished by
7.7 Water—Type III reagent water conforming to Specifica-
means of magnetic or propeller type stirrer mechanisms. The
tion D1193.
buret size should ideally be 10 mL or 20 mL.
7.8 Anhydrous Calcium Sulfate Desiccant.
6.2 Reference Electrode—A double junction reference elec-
trode with the inner electrode composed of silver/silver chlo-
8. Preparation of Standard Solutions
ride with a potassium chloride solution as internal electrolyte.
8.1 Lead Nitrate Titrant, 0.0025 M—Dissolve 0.833 g lead
The external solution is composed of 1 M lithium chloride in
nitrate in 300 mL water. Pour into a 1 L bottle and fill with
ethanol. This configuration is used to prevent silver ion, a lead
denatured ethanol and mix well. Standardize in accordance
electrodepoison,fromleachingintotheanalytesolutionduring
with 10.1.
titration. Preferred electrolytes for use in double junction
8.1.1 Alternatively, this solution may be purchased from a
electrodes may vary with the manufacturer; use the manufac-
commercial vendor, and its exact molarity shall be determined
turer’s recommended electrolytes for the application. Other
in accordance with 10.1.
types of reference electrodes may be considered with some
8.2 Aqueous Sulfate Standard, 0.01 M—Dry 5 g anhydrous
caveats (for example, single junction, combination, or glassy
sodium sulfate at 110 °C for 1 h. Remove it from the oven, and
carbon), but the data presented in this test method were
allow it to cool in a desiccator over anhydrous calcium sulfate.
generated using exclusively a double junction electrode, which
Accurately weigh about 0.70 g on an analytical balance to the
is the best choice for this determination.
nearest tenth of a milligram, and place it in a 500 mL
6.3 Lead Electrode—Alead sulfide-based crystalline sensor
volumetric flask. Add Type III water to dissolve the sodium
type lead ion selective electrode (ISE) is used.
sulfate, then dilute to volume. Calculate the exact concentra-
tion in accordance with Eq 1.
6.4 Drying Oven—A drying oven for drying sodium sulfate
at 110 °C is required.
G
5 Molarity (1)
142.02 0.500
6.5 Pipets or Volumetric Transferring Devices—Class A ~ !~ !
glass pipets or their equivalent.
where:
6.6 Polishing Material—Lead sulfide based crystalline sen-
Molarity = molarity of sulfate standard solution, mol/L,
sor electrodes require polishing to remove oxidation products.
G = weight in grams of Na SO , dissolved in
2 4
These materials are supplied with the electrode from the
500 mL, and
manufacturer.
142.02 = gram molecular weight of Na SO .
2 4
6.7 pH Test Strips—Test strips in the range of pH 1 to pH 7. 8.3 Aqueous Sulfate Stock Solution for Standards in
Ethanol, 2000 mg⁄L—Accurately weigh 2.95 g anhydrous so-
6.8 Titration Vessels—Standard glass beakers or titration
dium sulfate to the nearest tenth of a milligram and transfer it
vessels supplied with titration equipment.
to a 1 L volumetric flask. (Dried anhydrous sodium sulfate
shouldbestoredinadesiccator.)AddTypeIIIwatertodissolve
7. Reagents and Materials
the sodium sulfate, and make to volume. Calculate the concen-
7.1 Lead Nitrate—Reagent grade, 99 % minimum purity.
tration of sulfate in the solution in accordance with Eq 2.
(Warning—Poison, harmful by inhalation and if ingested.
gNa SO 0.6764 1000 mg/g
~ !~ !~ !
2 4
Avoid contact with the skin.) Dispose of this material in
Aqueous Stock Sulfate mg/L 5
~ !
1L
accordance with accepted local requirements.
(2)
7.2 Sodium Sulfate—Anhydrous, reagent grade, 99 % mini-
where:
mum purity. (Warning—Do not ingest. Avoid unnecessary
gNa SO = weight in grams of Na SO dissolved in 1 L,
exposure.)
2 4 2 4
and
7.3 Perchloric Acid 70 %—A.C.S. reagent grade, minimum
0.6764 = fraction of sulfate in Na SO .
2 4
purity with sulfate concentration <0.001 % (m/M). Dispose of
8.4 Sulfate Standards in Ethanol—Ethanol (denatured con-
thismaterialinaccordancewithacceptedlocalrequirements.It
taining no measurable sulfate) is weighed into a container
must contain no measurable sulfate. (Warning—Corrosive;
(equipped with a closure to prevent evaporation) in accordance
keep away from skin and eyes. Perchloric acid is a strong
with Table 1 to achieve the desired standard. Aqueous sulfate
oxidizer.)
7.4 Ethanol—Denatured with methanol, formula 3A or his-
tological grade ethanol, anhydrous, denatured with ethyl TABLE 1 Preparation of Sulfate Standards in Ethanol
acetate, methyl isobutyl ketone and hydrocarbon naphtha. It
Ethanolic Sulfate Standard, Ethanol, Aqueous Sulfate Stock
mg sulfate/kg ethanol g Solution, mL
must be free of any measurable sulfate. (Warning—
50 975 25
Flammable, toxic, may be harmful or fatal if ingested or
20 990 10
inhaled. Avoid skin contact.)
10 995 5
5 997.5 2.5
7.5 Ethanol—Absolute, 200 proof, 99.5 %, A.C.S. reagent
1 999.5 0.5
grade.
´1
D7318 − 19
stock solution from 8.3 is added to the solution in accordance 9. Titration Equipment Preparation
with Table 1, and the final weight of the solution recorded.
9.1 Titrator—Prepare the titrator by filling the titrator res-
Standards should be remade weekly or if recovery of less than
ervoir with lead titrant (8.1). Follow the manufacturer’s pro-
90 % is noted. The concentration of the standard is calculated
cedure for filling the buret. Perform titrations in monotonic
by dividing the number of milligrams sulfate from the sulfate
titration mode, using either 25 µL or 50 µL titrant addition
stock solution and dividing by the final solution weight in
increments. Use a 10 mV⁄min drift condition or 20 s wait time
accordance with Eq 3.
between additions (whichever is achieved first).
V 3C
EtOH Sulfate Standard mg/kg 5 (3) 9.2 Electrode Preparation—Proper care of the lead-
~ !
W
selective electrode is essential for obtaining high-quality titra-
where:
tion curves. Preparation of the lead electrode should be
V = volume of aqueous sulfate stock (8.3), mL, performed as specified by the manufacturer. A lead electrode
C = concentration of aqueous sulfate stock (8.3), mg/L, and utilizing a solid crystal sensor requires polishing when perfor-
W = final weight of ethanol and aqueous sulfate stock
mance deteriorates. The voltage range for a 10 ppm sulfate in
aliquot, g.
ethanol titration should span at least 50 mV and produce an
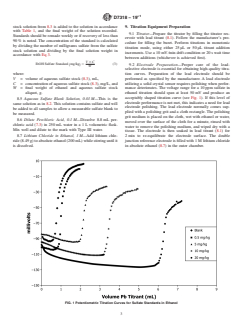
acceptably shaped titration curve (see Fig. 1). If this level of
8.5 Aqueous Sulfate Blank Solution, 0.01 M—This is the
electrode performance is not met, this indicates a need for lead
same solution as in 8.2. This solution contains sulfate and will
electrode polishing. The lead electrode normally comes sup-
be added to all samples to allow a measurable sulfate blank to
plied with a polishing grit and a cloth rectangle. The polishing
be measured.
grit medium is placed on the cloth, wet with ethanol or water,
8.6 Dilute Perchloric Acid, 0.1
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...