ASTM D4519-16
(Test Method)Standard Test Method for On-Line Determination of Anions and Carbon Dioxide in High Purity Water by Cation Exchange and Degassed Cation Conductivity
Standard Test Method for On-Line Determination of Anions and Carbon Dioxide in High Purity Water by Cation Exchange and Degassed Cation Conductivity
SIGNIFICANCE AND USE
5.1 This test method can be a useful diagnostic tool in measuring the impurities and detecting their sources in high purity water, boiler feed water and steam condensate of high pressure power plants, and in the process water of certain industries requiring high purity water.
5.2 The measurement of such impurities is most important to these industries since plant outages or product contamination can result from events such as condenser leakage. Also, water quality deviations can occur from condensate polishing and makeup water equipment malfunctions.
5.3 The continuous measurement and trends provided by this test method are of particular interest and can indicate the need for corrections in water treating or operating procedures and equipment. The equipment for this test method can be considered more rugged and adaptable to installation under plant operating conditions than the more accurate laboratory methods, such as ion chromatography and atomic absorption.
SCOPE
1.1 This on-line test method includes hydrogen exchange and degassing by heating or gas stripping and provides means for determining anions (such as Cl−, SO4—, NO3−, and F−) at levels as low as 2 μg/L (2 ppb) and carbon dioxide at the level of 0.01 to 10 mg/L (ppm) at 25°C in high purity water and in steam and water samples in power plants by measuring electrical conductivity.
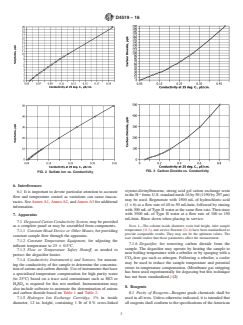
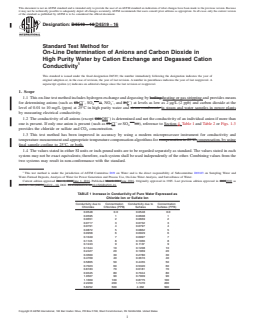
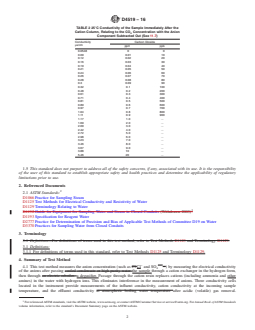
1.2 The conductivity of all anions (except OH−) is determined and not the conductivity of an individual anion if more than one is present. If only one anion is present (such as Cl− or SO4—−), reference to Section 4, Table 1 and Table 2 or Figs. 1-3 provides the chloride or sulfate and CO2 concentration.
1.3 This test method has been improved in accuracy by using a modern microprocessor instrument for conductivity and temperature measurement and appropriate temperature compensation algorithms for compensation, by using final sample cooling to 25°C, or both.
1.4 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-May-2016
- Technical Committee
- D19 - Water
Relations
- Effective Date
- 01-Jun-2016
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 15-Jun-2012
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 15-Dec-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
Overview
ASTM D4519-16 is the standard test method for the on-line determination of anions and carbon dioxide in high purity water using cation exchange and degassed cation conductivity. Developed by ASTM International, this method allows for continuous monitoring and quantification of key impurities-particularly chloride (Cl−), sulfate (SO₄²−), nitrate (NO₃−), fluoride (F−), and carbon dioxide (CO₂)-in water systems where the highest purity is vital. Industries such as power generation and electronics manufacturing rely on this standard for the precise, real-time assessment of water quality in boiler feedwater, steam condensate, and process water.
This test method uses cation exchange followed by degassing to remove volatile acids and measure the electrical conductivity of the resulting sample. The method is especially valued as it is rugged, well-suited to harsh industrial environments, and adaptable for continuous online water quality monitoring.
Key Topics
- Measurement Principle: Uses hydrogen ion exchange and degassing (by either heating or gas stripping) to remove volatile interferences, followed by conductivity measurement.
- Detection Sensitivity: Capable of detecting anions at levels as low as 2 μg/L (2 ppb) and carbon dioxide from 0.01 to 10 mg/L at 25°C.
- Continuous Monitoring: Provides real-time, ongoing measurement and trend data for prompt detection of water quality deviations.
- Robust Instrumentation: Utilizes modern microprocessor-based conductivity and temperature measurement technologies along with temperature compensation algorithms.
- Practical Calibration: Can be calibrated using standard chloride or sulfate solutions to verify system performance and ensure measurement accuracy.
- Diagnostic Utility: Helps trace sources of contamination, detect condenser leaks, and monitor the performance of water treatment and purification systems.
Applications
- Power Plants: Essential for high pressure boiler and steam condensate monitoring, ensuring early detection of condenser leaks and process upsets that could lead to costly shutdowns or damage.
- Electronics and Pharmaceutical Manufacturing: Critical in maintaining ultrapure water standards across cleaning, rinsing, and process operations.
- Industrial Process Water: Supports continuous assurance of water purity in facilities where trace anion or volatile contamination can compromise product quality or system performance.
- Water Treatment Diagnostics: Used to validate the functionality of condensate polishing, makeup water production, and demineralization equipment.
- Compliance and Quality Control: Supports regulatory compliance by providing documentation and real-time data on water quality.
Related Standards
- ASTM D1066: Practice for Sampling Steam-procedures for accurate sampling of steam for chemical analysis.
- ASTM D1125: Test Methods for Electrical Conductivity and Resistivity of Water-foundational methods for electrical measurements in water analysis.
- ASTM D1129: Terminology Relating to Water-standardized vocabulary for water-related testing and analysis.
- ASTM D1193: Specification for Reagent Water-defines quality for water used in reagent preparation and analytical testing.
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water.
- ASTM D3370: Practices for Sampling Water from Closed Conduits-sampling techniques from pipelines and pressurized lines.
Practical Value
Implementing ASTM D4519-16 enables industries to maintain optimal operation by minimizing the risk of plant outages and product contamination due to water quality issues. The standard’s adaptability for online, real-time measurement reduces reliance on complicated laboratory analyses, providing fast, actionable results where and when they are needed most. This ensures higher operational efficiency, reduced downtime, and assurance of compliance with stringent water purity requirements.
Keywords: ASTM D4519, high purity water, boiler feedwater, condensate, anions, carbon dioxide, cation exchange, degassed cation conductivity, online water analysis, power plant water chemistry.
Buy Documents
ASTM D4519-16 - Standard Test Method for On-Line Determination of Anions and Carbon Dioxide in High Purity Water by Cation Exchange and Degassed Cation Conductivity
REDLINE ASTM D4519-16 - Standard Test Method for On-Line Determination of Anions and Carbon Dioxide in High Purity Water by Cation Exchange and Degassed Cation Conductivity
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM D4519-16 is a standard published by ASTM International. Its full title is "Standard Test Method for On-Line Determination of Anions and Carbon Dioxide in High Purity Water by Cation Exchange and Degassed Cation Conductivity". This standard covers: SIGNIFICANCE AND USE 5.1 This test method can be a useful diagnostic tool in measuring the impurities and detecting their sources in high purity water, boiler feed water and steam condensate of high pressure power plants, and in the process water of certain industries requiring high purity water. 5.2 The measurement of such impurities is most important to these industries since plant outages or product contamination can result from events such as condenser leakage. Also, water quality deviations can occur from condensate polishing and makeup water equipment malfunctions. 5.3 The continuous measurement and trends provided by this test method are of particular interest and can indicate the need for corrections in water treating or operating procedures and equipment. The equipment for this test method can be considered more rugged and adaptable to installation under plant operating conditions than the more accurate laboratory methods, such as ion chromatography and atomic absorption. SCOPE 1.1 This on-line test method includes hydrogen exchange and degassing by heating or gas stripping and provides means for determining anions (such as Cl−, SO4—, NO3−, and F−) at levels as low as 2 μg/L (2 ppb) and carbon dioxide at the level of 0.01 to 10 mg/L (ppm) at 25°C in high purity water and in steam and water samples in power plants by measuring electrical conductivity. 1.2 The conductivity of all anions (except OH−) is determined and not the conductivity of an individual anion if more than one is present. If only one anion is present (such as Cl− or SO4—−), reference to Section 4, Table 1 and Table 2 or Figs. 1-3 provides the chloride or sulfate and CO2 concentration. 1.3 This test method has been improved in accuracy by using a modern microprocessor instrument for conductivity and temperature measurement and appropriate temperature compensation algorithms for compensation, by using final sample cooling to 25°C, or both. 1.4 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 This test method can be a useful diagnostic tool in measuring the impurities and detecting their sources in high purity water, boiler feed water and steam condensate of high pressure power plants, and in the process water of certain industries requiring high purity water. 5.2 The measurement of such impurities is most important to these industries since plant outages or product contamination can result from events such as condenser leakage. Also, water quality deviations can occur from condensate polishing and makeup water equipment malfunctions. 5.3 The continuous measurement and trends provided by this test method are of particular interest and can indicate the need for corrections in water treating or operating procedures and equipment. The equipment for this test method can be considered more rugged and adaptable to installation under plant operating conditions than the more accurate laboratory methods, such as ion chromatography and atomic absorption. SCOPE 1.1 This on-line test method includes hydrogen exchange and degassing by heating or gas stripping and provides means for determining anions (such as Cl−, SO4—, NO3−, and F−) at levels as low as 2 μg/L (2 ppb) and carbon dioxide at the level of 0.01 to 10 mg/L (ppm) at 25°C in high purity water and in steam and water samples in power plants by measuring electrical conductivity. 1.2 The conductivity of all anions (except OH−) is determined and not the conductivity of an individual anion if more than one is present. If only one anion is present (such as Cl− or SO4—−), reference to Section 4, Table 1 and Table 2 or Figs. 1-3 provides the chloride or sulfate and CO2 concentration. 1.3 This test method has been improved in accuracy by using a modern microprocessor instrument for conductivity and temperature measurement and appropriate temperature compensation algorithms for compensation, by using final sample cooling to 25°C, or both. 1.4 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the two systems may result in non-conformance with the standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM D4519-16 is classified under the following ICS (International Classification for Standards) categories: 17.220.20 - Measurement of electrical and magnetic quantities. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4519-16 has the following relationships with other standards: It is inter standard links to ASTM D4519-10, ASTM D1129-13(2020)e2, ASTM D1066-18e1, ASTM D1066-18, ASTM D2777-12, ASTM D1066-11, ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1066-06, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4519-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4519 − 16
Standard Test Method for
On-Line Determination of Anions and Carbon Dioxide in
High Purity Water by Cation Exchange and Degassed Cation
Conductivity
This standard is issued under the fixed designation D4519; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 This on-line test method includes hydrogen exchange
Barriers to Trade (TBT) Committee.
and degassing by heating or gas stripping and provides means
− − − −
for determining anions (such as Cl,SO ,NO , and F)at
4 3
2. Referenced Documents
levels as low as 2 µg/L(2 ppb) and carbon dioxide at the level
2.1 ASTM Standards:
of 0.01 to 10 mg/L (ppm) at 25°C in high purity water and in
D1066Practice for Sampling Steam
steam and water samples in power plants by measuring
D1125Test Methods for Electrical Conductivity and Resis-
electrical conductivity.
tivity of Water
−
1.2 The conductivity of all anions (except OH ) is deter-
D1129Terminology Relating to Water
mined and not the conductivity of an individual anion if more
D1193Specification for Reagent Water
−
thanoneispresent.Ifonlyoneanionispresent(suchasCl or
D2777Practice for Determination of Precision and Bias of
−
SO ), reference to Section 4, Table 1 and Table 2 or Figs. 1-3
Applicable Test Methods of Committee D19 on Water
provides the chloride or sulfate and CO concentration.
D3370Practices for Sampling Water from Closed Conduits
1.3 This test method has been improved in accuracy by
3. Terminology
using a modern microprocessor instrument for conductivity
and temperature measurement and appropriate temperature
3.1 Definitions:
compensation algorithms for compensation, by using final
3.1.1 For definitions of terms used in this standard, refer to
sample cooling to 25°C, or both.
Test Methods D1125 and Terminology D1129.
1.4 The values stated in either SI units or inch-pound units
4. Summary of Test Method
are to be regarded separately as standard. The values stated in
each system may not be exact equivalents; therefore, each
4.1 Thistestmethodmeasurestheanionconcentration(such
− −
system shall be used independently of the other. Combining
as Cl and SO ) by measuring the electrical conductivity of
values from the two systems may result in non-conformance
the anions after passing the sample through a cation exchanger
with the standard.
in the hydrogen form, then through a degasifier. Passage
through the cation resin replaces cations (including ammonia
1.5 This standard does not purport to address all of the
and other amines) in the water with hydrogen ions. This
safety concerns, if any, associated with its use. It is the
eliminates interference in the measurement of anions. Three
responsibility of the user of this standard to establish appro-
conductivity cells located in the instrument provide measure-
priate safety, health, and environmental practices and deter-
ments of the influent conductivity, cation conductivity at the
mine the applicability of regulatory limitations prior to use.
incoming sample temperature, and the effluent conductivity
1.6 This international standard was developed in accor-
after acidic (volatile) gas removal. The sample is then either
dance with internationally recognized principles on standard-
cooled to 25°C or conductivity values are compensated to
ization established in the Decision on Principles for the
25°C. While the influent conductivity measurement is not
necessary in determining the total anion conductivity, its
determination provides a more complete evaluation of the
This test method is under the jurisdiction ofASTM Committee D19 on Water
and is the direct responsibility of Subcommittee D19.03 on Sampling Water and
Water-Formed Deposits,Analysis of Water for Power Generation and Process Use,
On-Line Water Analysis, and Surveillance of Water. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved June 1, 2016. Published June 2016. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1985. Last previous edition approved in 2010 as D4519–10. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D4519-16. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4519 − 16
TABLE 1 Increase in Conductivity of Pure Water Expressed as
Chloride Ion or Sulfate Ion
Conductivity due to Concentration Conductivity due to Concentration
Chlorides Chlorides (PPB) Sulfates Sulfates (PPB)
0.0548 0.0 0.0548 0.0
0.0595 1 0.0608 1
0.0651 2 0.0669 2
0.0717 3 0.0732 3
0.0791 4 0.0797 4
0.0872 5 0.0862 5
0.0958 6 0.0929 6
0.1049 7 0.0997 7
0.1145 8 0.1066 8
0.1243 9 0.1137 9
0.1344 10 0.1208 10
0.2427 20 0.1969 20
0.3560 30 0.2780 30
0.4709 40 0.3616 40
0.5865 50 0.4455 50
0.7023 60 0.5320 60
0.8183 70 0.6181 70
0.9345 80 0.7044 80
1.0507 90 0.7909 90
1.1669 100 0.8775 100
2.2209 200 1.7470 200
5.8252 500 4.362 500
TABLE 2 25°C Conductivity of the Sample Immediately After the
Cation Column, Relating to the CO Concentration with the Anion
Component Subtracted Out (See 11.7)
Conductivity Carbon Dioxide
µs/cm
ppm ppb
0.0548 0 0
0.09 0.01 10
0.12 0.02 20
0.16 0.03 30
0.19 0.04 40
FIG. 1 Chloride Ion vs. Conductivity
0.21 0.05 50
0.24 0.06 60
0.26 0.07 70
4.2 In-depth studies provide additional background and
0.28 0.08 80
0.3 0.09 90
updated experience with the degassed cation conductivity
0.32 0.1 100
technique (1).
0.48 0.2 200
0.61 0.3 300
5. Significance and Use
0.71 0.4 400
0.81 0.5 500
5.1 This test method can be a useful diagnostic tool in
0.89 0.6 600
0.97 0.7 700 measuring the impurities and detecting their sources in high
1.04 0.8 800
purity water, boiler feed water and steam condensate of high
1.11 0.9 900
pressure power plants, and in the process water of certain
1.17 1.0 .
1.69 2.0 . industries requiring high purity water.
2.09 3.0 .
5.2 The measurement of such impurities is most important
2.42 4.0 .
2.72 5.0 .
totheseindustriessinceplantoutagesorproductcontamination
2.98 6.0 .
can result from events such as condenser leakage.Also, water
3.23 7.0 .
quality deviations can occur from condensate polishing and
3.46 8.0 .
3.67 9.0 .
makeup water equipment malfunctions.
3.88 10 .
5.46 20 . 5.3 The continuous measurement and trends provided by
this test method are of particular interest and can indicate the
need for corrections in water treating or operating procedures
and equipment. The equipment for this test method can be
considered more rugged and adaptable to installation under
sample, which can also include an estimation of the amine
plant operating conditions than the more accurate laboratory
content. Measurement of the cation and degassed sample
methods, such as ion chromatography and atomic absorption.
conductivities are necessary in determining the composition of
the influent (total anions and acidic gas content). Reference to
Table 1 and Table 2 or Figs. 1-3, or both, are then necessary to
The boldface numbers in parentheses refer to a list of references at the end of
complete the determinations. this standard.
D4519 − 16
FIG. 3 Carbon Dioxide vs. Conductivity
FIG. 2 Sulfate Ion vs. Conductivity
6. Interferences
styrene-divinylbenzene, strong acid gel cation exchange resin
6.1 It is important to devote particular attention to accurate
+
intheH form;U.S.standardmesh16by50(1190by297µm)
flow and temperature control as variations can cause inaccu-
may be used. Regenerate with 1500 mL of hydrochloric acid
racies.SeeAnnexA1,AnnexA2,andAnnexA3foradditional
(1+6) at a flow rate of 40 to 50 mL/min, followed by rinsing
information.
with300mLofTypeIIwateratthesameflowrate.Thenrinse
with 3500 mL of Type II water at a flow rate of 100 to 150
7. Apparatus
mL/min. Rinse down when placing in service.
7.1 Degassed Cation Conductivity System,maybeprovided
NOTE 1—The column inside diameter, resin bed height, inlet sample
as a complete panel or may be assembled from components.
temperature (11.3), and service flowrate (11.4) have been standardized to
7.1.1 Constant Head Device or Other Means, for providing
provide comparable results. They may not be the optimum values. The
constant sample flow through the apparatus.
user should realize that those parameters affect the measurement.
7.1.2 Constant Temperature Equipment, for adjusting the
7.1.6 Degasifier, for removing carbon dioxide from the
influent temperature to 25 6 0.5°C.
sample. The degasifier may operate by heating the sample to
7.1.3 Flow or Temperature Safety Shutoff, as needed to
near boiling temperature with a reboiler or by sparging with a
protect the degasifier heater.
CO -free gas such as nitrogen. Following a reboiler, a cooler
7.1.4 Conductivity Instrument(s) and Sensors, for measur-
may be used to reduce the sample temperature and potential
ing the conductivity of the sample to determine the concentra-
errors in temperature compensation. (Membrane gas stripping
tionofanionsandcarbondioxide.Useofinstrumentsthathave
has been used experimentally for degassing but this technique
a specialized temperature compensation for high purity water
has not been standardized.) (2)
(to 25°C) based on a trace acid contaminant such as HCl or
H SO is required for this test method. Instrumentation may
2 4
8. Reagents
also include software to automate the determination of anions
and carbon dioxide based on Table 1 and Table 2. 8.1 Purity of Reagents—Reagent grade chemicals shall be
7.1.5 Hydrogen Ion Exchange Cartridge, 1 ⁄8 in. inside used in all tests. Unless otherwise indicated, it is intended that
diameter, 12 in. height, containing 1 lb of 8% cross-linked allreagentsshallconformtothespecificationsoftheAmerican
D4519 − 16
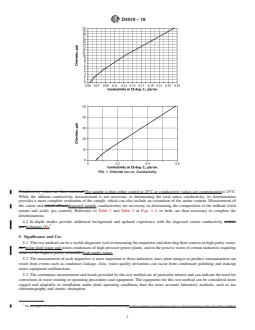
FIG. 4 Block Diagram of Measurement Apparatus
Chemical Society, where such specifications are available. 10.2 If it is desired to calibrate the instrument using known
Other grades may be used, provided it is first ascertained that chloride solutions, the range of 0.001 to 0.010 mg/L (1 to 10
the reagent is of sufficiently high purity to permit its use ppb) of chloride may be covered by application of 0.2 to 2.0
without lessening the accuracy of the determination. mL/min of the standard chloride solution (8.4.1) to the 200
mL/min flow by injecting the flow from a small peristaltic
8.2 Purity of Water—Unless otherwise indicated, references
pump to a hypodermic needle inserted in a plastic tubing
towaterthatisusedforreagentpreparation,rinsingordilution
connection.
shall be understood to mean water that conforms to the
quantitative specifications of Type II reagent water of Specifi-
11. Procedure
cation D1193.
−
11.1 Flush out the piping thoroughly by flow of high purity
8.3 Chloride Solution, Stock (1 mL = 0.1 mg Cl )—
water through the instrument before starting the flow of water
Dissolve in water 0.1649 g of sodium chloride (NaCl) dried to
to be tested.
constant weight at 105°C, and dilute to 1 L in a thoroughly
cleaned polyethylene flask.
11.2 Refer to the manufacturer’s instruction booklet before
startingtheinstrument.Becauseofthenatureofthismeasuring
8.4 Chloride Solution, Standard (1.00 mL=0.001 mg)—
technique, it is very important to follow the manufacturer’s
Dilute 10.00 mL of chloride stock solution (8.3) to 1 L with
instructions very carefully in order to achieve accurate results.
water.
See Annex A1 – Annex A4 for a discussion on the effects
8.4.1 This standard chloride solution is to be used in the
caused by variations in operating conditions and interfering
calibration of the instrument if desired, or reference can be
substances.
made to the instruction booklet furnished with the instrument.
11.3 Connect the sample line to be tested to the instrument
8.5 Hydrochloric Acid (1+6)—Add 100 mL concentrated
influent conne
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4519 − 10 D4519 − 16
Standard Test Method for
On-Line Determination of Anions and Carbon Dioxide in
High Purity Water by Cation Exchange and Degassed Cation
Conductivity
This standard is issued under the fixed designation D4519; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This on-line test method includes hydrogen exchange and degassing by boiling heating or gas stripping and provides means
− −− — − −
for determining anions (such as Cl Cl , SO , NO , and F F ) at levels as low as 2 μg/L (2 ppb) and carbon dioxide at the
4 3
level of 0.01 to 10 mg/L (ppm) at 25°C in high purity water and steam condensate in steam and water samples in power plants
by measuring electrical conductivity.
−
1.2 The conductivity of all anions (except OH OH ) is determined and not the conductivity of an individual anion if more than
− −−—−
one is present. If only one anion is present (such as Cl Cl or SO ), reference to Section 4, Table 1 and Table 2 or Figs. 1-3
provides the chloride or sulfate and CO concentration.
1.3 This test method has been improved in accuracy by using a modern microprocessor instrument for conductivity and
temperature measurement and appropriate temperature compensation algorithms for compensation to 25°C.compensation, by using
final sample cooling to 25°C, or both.
1.4 The values stated in either SI units or inch-pound units are to be regarded separately as standard. The values stated in each
system may not be exact equivalents; therefore, each system shall be used independently of the other. Combining values from the
two systems may result in non-conformance with the standard.
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.03 on Sampling Water and
Water-Formed Deposits, Analysis of Water for Power Generation and Process Use, On-Line Water Analysis, and Surveillance of Water.
Current edition approved Dec. 1, 2010June 1, 2016. Published March 2011June 2016. Originally approved in 1985. Last previous edition approved in 20052010 as
D4519 – 94 (2005).D4519 – 10. DOI: 10.1520/D4519-10.10.1520/D4519-16.
TABLE 1 Increase in Conductivity of Pure Water Expressed as
Chloride Ion or Sulfate Ion
Conductivity due to Concentration Conductivity due to Concentration
Chlorides Chlorides (PPB) Sulfates Sulfates (PPB)
0.0548 0.0 0.0548 0.0
0.0595 1 0.0608 1
0.0651 2 0.0669 2
0.0717 3 0.0732 3
0.0791 4 0.0797 4
0.0872 5 0.0862 5
0.0958 6 0.0929 6
0.1049 7 0.0997 7
0.1145 8 0.1066 8
0.1243 9 0.1137 9
0.1344 10 0.1208 10
0.2427 20 0.1969 20
0.3560 30 0.2780 30
0.4709 40 0.3616 40
0.5865 50 0.4455 50
0.7023 60 0.5320 60
0.8183 70 0.6181 70
0.9345 80 0.7044 80
1.0507 90 0.7909 90
1.1669 100 0.8775 100
2.2209 200 1.7470 200
5.8252 500 4.362 500
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4519 − 16
TABLE 2 25°C Conductivity of the Sample Immediately After the
Cation Column, Relating to the CO Concentration with the Anion
Component Subtracted Out (See 11.7)
Conductivity Carbon Dioxide
μs/cm
ppm ppb
0.0548 0 0
0.09 0.01 10
0.12 0.02 20
0.16 0.03 30
0.19 0.04 40
0.21 0.05 50
0.24 0.06 60
0.26 0.07 70
0.28 0.08 80
0.3 0.09 90
0.32 0.1 100
0.48 0.2 200
0.61 0.3 300
0.71 0.4 400
0.81 0.5 500
0.89 0.6 600
0.97 0.7 700
1.04 0.8 800
1.11 0.9 900
1.17 1.0 .
1.69 2.0 .
2.09 3.0 .
2.42 4.0 .
2.72 5.0 .
2.98 6.0 .
3.23 7.0 .
3.46 8.0 .
3.67 9.0 .
3.88 10 .
5.46 20 .
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
D1066 Practice for Sampling Steam
D1125 Test Methods for Electrical Conductivity and Resistivity of Water
D1129 Terminology Relating to Water
D1192 Guide for Equipment for Sampling Water and Steam in Closed Conduits (Withdrawn 2003)
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Closed Conduits
3. Terminology
3.1 Definitions—For definitions of terms used in this test method, refer to Test Methods D1125 and Terminology D1129.
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Test Methods D1125 and Terminology D1129.
4. Summary of Test Method
− −− —
4.1 This test method measures the anion concentration (such as Cl Cl and SO ) by measuring the electrical conductivity
of the anions after passing cooled condensate or high purity water the sample through a cation exchanger in the hydrogen form,
then through an electric reboiler. a degasifier. Passage through the cation resin replaces cations (including ammonia and other
amines) in the water with hydrogen ions. This eliminates interference in the measurement of anions. Three conductivity cells
located in the instrument provide measurements of the influent conductivity, cation conductivity at the incoming sample
temperature, and the effluent conductivity at atmospheric boiling water temperature after acidic (volatile) gas removal.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
D4519 − 16
FIG. 1 Chloride Ion vs. Conductivity
Conductivity values are then corrected The sample is then either cooled to 25°C or conductivity values are compensated to 25°C.
While the influent conductivity measurement is not necessary in determining the total anion conductivity, its determination
provides a more complete evaluation of the sample, which can also include an estimation of the amine content. Measurement of
the cation and reboil effluentdegassed sample conductivities are necessary in determining the composition of the influent (total
anions and acidic gas content). Reference to Table 1 and Table 2 or Figs. 1-3, or both, are then necessary to complete the
determinations.
4.2 In-depth studies provide additional background and updated experience with the degassed cation conductivity techni-
que.technique (1).
5. Significance and Use
5.1 This test method can be a useful diagnostic tool in measuring the impurities and detecting their sources in high purity water,
the boiler feed water and steam condensate of high pressure power plants, and in the process water of certain industries requiring
water of the highest purity attainable.high purity water.
5.2 The measurement of such impurities is most important to these industries since plant outages or product contamination can
result from events such as condenser leakage. Also, water quality deviations can occur from condensate polishing and makeup
water equipment malfunctions.
5.3 The continuous measurement and trends provided by this test method are of particular interest and can indicate the need for
corrections in water treating or operating procedures and equipment. The equipment for this test method can be considered more
rugged and adaptable to installation under plant operating conditions than the more accurate laboratory methods, such as ion
chromatography and atomic absorption.
The last approved version of this historical standard is referenced on www.astm.org.boldface numbers in parentheses refer to a list of references at the end of this standard.
D4519 − 16
FIG. 2 Sulfate Ion vs. Conductivity
6. Interferences
6.1 It is important to devote particular attention to accurate flow and temperature control as variations can cause inaccuracies.
See Annex A1, Annex A2, and Annex A3 for additional information on this subject.information.
7. Apparatus
7.1 Mechanical Ion Exchanger-Degasser Instrument.
7.2 Constant Head Device, for providing approximately 1.5 m (5 ft) head pressure for water entering the instrument.
7.3 Constant Temperature Equipment, for adjusting the influent temperature to 25°C 6 0.5°C.
7.4 Conductivity Instruments and Sensors, for measuring the conductivity of the sample to determine the concentration of
anions and carbon dioxide. Use of instruments that have a specialized temperature compensation for high purity water (to 25°C)
based on an acid such as HCl or H SO is required for this test method.
2 4
7.1 Hydrogen Exchange Cartridge ,Degassed Cation Conductivity System, 1 ⁄8 in. inside diameter, 12 in. height, containing 1
+
lb of 8 % cross-linked styrene-divinylbenzene, strong acid gel cation exchange resin in the H form; U.S. standard mesh 16 by
50 (1190 by 297 μm) may be used. Regenerate with 1500 mL of hydrochloric acid (1 + 6) at a flow rate of 40 to 50 mL/min,
followed by rinsing with 300 mL of Type II water at the same flow rate. Then rinse with 3500 mL of Type II water at a flow rate
of 100 to 150 mL/min. Rinse down when placing in service.may be provided as a complete panel or may be assembled from
components.
NOTE 1—The column inside diameter, resin bed height, inlet sample temperature (11.3), and service flowrate (11.4) have been standardized to provide
comparable results. They may not be the optimum values. The user should realize that those parameters affect the measurement.
7.1.1 Constant Head Device or Other Means, for providing constant sample flow through the apparatus.
7.1.2 Constant Temperature Equipment, for adjusting the influent temperature to 25 6 0.5°C.
7.1.3 Flow or Temperature Safety Shutoff, as needed to protect the degasifier heater.
D4519 − 16
FIG. 3 Carbon Dioxide vs. Conductivity
7.1.4 Conductivity Instrument(s) and Sensors, for measuring the conductivity of the sample to determine the concentration of
anions and carbon dioxide. Use of instruments that have a specialized temperature compensation for high purity water (to 25°C)
based on a trace acid contaminant such as HCl or H SO is required for this test method. Instrumentation may also include software
2 4
to automate the determination of anions and carbon dioxide based on Table 1 and Table 2.
7.1.5 Hydrogen Ion Exchange Cartridge, 1 ⁄8 in. inside diameter, 12 in. height, containing 1 lb of 8 % cross-linked
+
styrene-divinylbenzene, strong acid gel cation exchange resin in the H form; U.S. standard mesh 16 by 50 (1190 by 297 μm) may
be used. Regenerate with 1500 mL of hydrochloric acid (1 + 6) at a flow rate of 40 to 50 mL/min, followed by rinsing with 300
mL of Type II water at the same flow rate. Then rinse with 3500 mL of Type II water at a flow rate of 100 to 150 mL/min. Rinse
down when placing in service.
NOTE 1—The column inside diameter, resin bed height, inlet sample temperature (11.3), and service flowrate (11.4) have been standardized to provide
comparable results. They may not be the optimum values. The user should realize that those parameters affect the measurement.
7.1.6 Degasifier, for removing carbon dioxide from the sample. The degasifier may operate by heating the sample to near boiling
temperature with a reboiler or by sparging with a CO -free gas such as nitrogen. Following a reboiler, a cooler may be used to
reduce the sample temperature and potential errors in temperature compensation. (Membrane gas stripping has been used
experimentally for degassing but this technique has not been standardized.) (2)
7.6 Software to automate the determination of anions and carbon dioxide is available.
8. Reagents
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the American Chemical Society, where such specifications are available. Other
grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening
the accuracy of the determination.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmaceutical Convention, Inc. (USPC), Rockville, MD.
D4519 − 16
FIG. 4 Flow Diagram for Mechanical-Ion Exchanger-Degasser InstrumentBlock Diagram of Measurement Apparatus
8.2 Purity of Water—Unless otherwise indicated, references to water that is used for reagent preparation, rinsing or dilution shall
be understood to mean water that conforms to the quantitative specifications of Type II reagent water of Specification D1193.
−
8.3 Chloride Solution, Stock (1 mL = 0.1 mg Cl Cl )—Dissolve in water 0.1649 g of sodium chloride (NaCl) dried to constant
weight at 105°C, and dilute to 1 L in a thoroughly cleaned polyethylene flask.
8.4 Chloride Solution, Standard (1.00 mL = 0.001 mg)—Dilute 10.00 mL of chloride stock solution (8.3) to 1 L with water.
8.4.1 This standard chloride solution is to be used in the calibration of the instrument if desired, or reference can be made to
the instruction booklet furnished with the instrument.
8.5 Hydrochloric Acid (1 + 6)—Add 100 mL concentrated HCl (sp. gr. 1.19) to 600 mL water.
9. Sampling
9.1 Collect the sampleEstablish sampling conditions in accordance with the applicable ASTM standards: Practice D1066,
Specification D1192, and Practices D3370.
10. Calibration
− −−−
10.
...




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...