ASTM G170-06(2020)e1
(Guide)Standard Guide for Evaluating and Qualifying Oilfield and Refinery Corrosion Inhibitors in the Laboratory
Standard Guide for Evaluating and Qualifying Oilfield and Refinery Corrosion Inhibitors in the Laboratory
SIGNIFICANCE AND USE
5.1 Corrosion inhibitors continue to play a key role in controlling internal corrosion associated with oil and gas production and transportation. This results primarily from the industry’s extensive use of carbon and low alloy steels, which, for many applications, are economic materials of construction that generally exhibit poor corrosion resistance. As a consequence, there is a strong reliance on inhibitor deployment for achieving cost-effective corrosion control, especially for treating long flowlines and main export pipelines (1).5
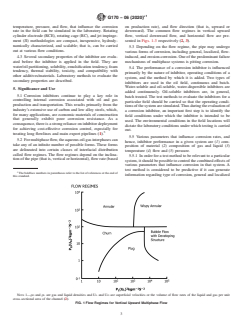
5.2 For multiphase flow, the aqueous-oil-gas interphases can take any of an infinite number of possible forms. These forms are delineated into certain classes of interfacial distribution called flow regimes. The flow regimes depend on the inclination of the pipe (that is, vertical or horizontal), flow rate (based on production rate), and flow direction (that is, upward or downward). The common flow regimes in vertical upward flow, vertical downward flow, and horizontal flow are presented in Figs. 1-3 respectively (2, 3).
5.14 To develop an inhibitor selection strategy, in addition to inhibitor efficiency, several other key performance factors need to be evaluated: (1) water/oil partitioning, (2) solubility, (3) emulsification tendency, (4) foaming tendency, (5) thermal stability, (6) toxicity, and (7) compatibility with other additives/materials.
SCOPE
1.1 This guide covers some generally accepted laboratory methodologies that are used for evaluating corrosion inhibitors for oilfield and refinery applications in well defined flow conditions.
1.2 This guide does not cover detailed calculations and methods, but rather covers a range of approaches which have found application in inhibitor evaluation.
1.3 Only those methodologies that have found wide acceptance in inhibitor evaluation are considered in this guide.
1.4 This guide is intended to assist in the selection of methodologies that can be used for evaluating corrosion inhibitors.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory requirements prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2020
- Technical Committee
- G01 - Corrosion of Metals
- Drafting Committee
- G01.05 - Laboratory Corrosion Tests
Relations
- Effective Date
- 01-May-2019
- Effective Date
- 15-Feb-2019
- Effective Date
- 01-May-2018
- Effective Date
- 01-Jan-2016
- Effective Date
- 15-Dec-2014
- Effective Date
- 01-Nov-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Aug-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 15-Nov-2012
Overview
ASTM G170-06(2020)e1 is the Standard Guide for Evaluating and Qualifying Oilfield and Refinery Corrosion Inhibitors in the Laboratory, published by ASTM International. The standard addresses the critical need for reliable evaluation methodologies for corrosion inhibitors in oil and gas production and transportation, particularly given the industry's reliance on carbon and low alloy steels, which offer poor intrinsic corrosion resistance. Proper evaluation and qualification in the laboratory are essential to ensure cost-effective corrosion control, particularly in complex multiphase flow environments typical of upstream and downstream operations.
Key Topics
- Laboratory Evaluation Methodologies: The standard outlines widely accepted laboratory approaches used for testing corrosion inhibitors under defined flow conditions. It focuses on practical, scalable test setups rather than detailed calculations or highly specialized methodologies.
- Multiphase Flow Dynamics: Recognizes the complexity of oil-water-gas multiphase flow, with varied flow regimes depending on pipe inclination, flow rate, and flow direction. Properly simulating these regimes in the laboratory is crucial for meaningful corrosion inhibitor assessment.
- Inhibitor Performance Factors: In addition to primary efficiency, several secondary characteristics are essential for real-world performance:
- Water/oil partitioning
- Solubility
- Emulsification tendency
- Foaming tendency
- Thermal stability
- Toxicity
- Compatibility with other additives and construction materials
- Corrosion Measurement Techniques: Both mass loss and electrochemical methods (such as linear polarization resistance and potentiodynamic polarization) are validated within a range of laboratory test setups.
Applications
- Oilfield and Refinery Operations: This standard is instrumental for any organization involved in oil and gas extraction or processing, where corrosion poses significant operational and safety risks. It assists in the selection and qualification of corrosion inhibitors for use in pipelines, flowlines, and related infrastructure.
- Laboratory Testing and Quality Control: Provides guidance for laboratory professionals evaluating inhibitor products, ensuring tests are relevant, realistic, and predictive of field performance.
- Material and Additive Compatibility Testing: By encompassing secondary performance factors, the guide helps prevent issues like emulsion stability, foaming, or additive incompatibility, which can disrupt operations or processing.
- Simulating Field Conditions: The guide supports designing laboratory experiments that closely replicate the environmental variables encountered in the field, including material composition, multiphase flow, temperature, and pressure.
Related Standards
ASTM G170-06(2020)e1 references and complements several other standards, including:
- ASTM G1: Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens
- ASTM G16: Guide for Applying Statistics to Analysis of Corrosion Data
- ASTM G31: Guide for Laboratory Immersion Corrosion Testing of Metals
- ASTM G46: Guide for Examination and Evaluation of Pitting Corrosion
- ASTM G59: Test Method for Conducting Potentiodynamic Polarization Resistance Measurements
- ASTM G96: Guide for Online Monitoring of Corrosion in Plant Equipment
- ISO 6614: Petroleum Products – Determination of Water Separability of Petroleum Oils and Synthetic Fluids
- NACE TM0196: Standard Test Method for Chemical Resistance of Polymeric Materials
Practical Value
Implementing ASTM G170 enables oilfield and refinery operators, laboratory specialists, and corrosion engineers to:
- Select the most suitable laboratory methods for evaluating corrosion inhibitors under representative field conditions.
- Compare inhibitor performance in a standardized, reproducible manner.
- Address both primary and secondary criteria to minimize risk of operational issues post-deployment.
- Ensure compliance with internationally recognized best practices for corrosion control, supporting safer, more reliable, and economically efficient operations.
By adhering to this guide, stakeholders can reliably evaluate corrosion inhibitor products, leading to better informed decisions, reduced corrosion-related failures, and optimized maintenance strategies in oilfield and refinery environments.
Buy Documents
ASTM G170-06(2020)e1 - Standard Guide for Evaluating and Qualifying Oilfield and Refinery Corrosion Inhibitors in the Laboratory
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM G170-06(2020)e1 is a guide published by ASTM International. Its full title is "Standard Guide for Evaluating and Qualifying Oilfield and Refinery Corrosion Inhibitors in the Laboratory". This standard covers: SIGNIFICANCE AND USE 5.1 Corrosion inhibitors continue to play a key role in controlling internal corrosion associated with oil and gas production and transportation. This results primarily from the industry’s extensive use of carbon and low alloy steels, which, for many applications, are economic materials of construction that generally exhibit poor corrosion resistance. As a consequence, there is a strong reliance on inhibitor deployment for achieving cost-effective corrosion control, especially for treating long flowlines and main export pipelines (1).5 5.2 For multiphase flow, the aqueous-oil-gas interphases can take any of an infinite number of possible forms. These forms are delineated into certain classes of interfacial distribution called flow regimes. The flow regimes depend on the inclination of the pipe (that is, vertical or horizontal), flow rate (based on production rate), and flow direction (that is, upward or downward). The common flow regimes in vertical upward flow, vertical downward flow, and horizontal flow are presented in Figs. 1-3 respectively (2, 3). 5.14 To develop an inhibitor selection strategy, in addition to inhibitor efficiency, several other key performance factors need to be evaluated: (1) water/oil partitioning, (2) solubility, (3) emulsification tendency, (4) foaming tendency, (5) thermal stability, (6) toxicity, and (7) compatibility with other additives/materials. SCOPE 1.1 This guide covers some generally accepted laboratory methodologies that are used for evaluating corrosion inhibitors for oilfield and refinery applications in well defined flow conditions. 1.2 This guide does not cover detailed calculations and methods, but rather covers a range of approaches which have found application in inhibitor evaluation. 1.3 Only those methodologies that have found wide acceptance in inhibitor evaluation are considered in this guide. 1.4 This guide is intended to assist in the selection of methodologies that can be used for evaluating corrosion inhibitors. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory requirements prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Corrosion inhibitors continue to play a key role in controlling internal corrosion associated with oil and gas production and transportation. This results primarily from the industry’s extensive use of carbon and low alloy steels, which, for many applications, are economic materials of construction that generally exhibit poor corrosion resistance. As a consequence, there is a strong reliance on inhibitor deployment for achieving cost-effective corrosion control, especially for treating long flowlines and main export pipelines (1).5 5.2 For multiphase flow, the aqueous-oil-gas interphases can take any of an infinite number of possible forms. These forms are delineated into certain classes of interfacial distribution called flow regimes. The flow regimes depend on the inclination of the pipe (that is, vertical or horizontal), flow rate (based on production rate), and flow direction (that is, upward or downward). The common flow regimes in vertical upward flow, vertical downward flow, and horizontal flow are presented in Figs. 1-3 respectively (2, 3). 5.14 To develop an inhibitor selection strategy, in addition to inhibitor efficiency, several other key performance factors need to be evaluated: (1) water/oil partitioning, (2) solubility, (3) emulsification tendency, (4) foaming tendency, (5) thermal stability, (6) toxicity, and (7) compatibility with other additives/materials. SCOPE 1.1 This guide covers some generally accepted laboratory methodologies that are used for evaluating corrosion inhibitors for oilfield and refinery applications in well defined flow conditions. 1.2 This guide does not cover detailed calculations and methods, but rather covers a range of approaches which have found application in inhibitor evaluation. 1.3 Only those methodologies that have found wide acceptance in inhibitor evaluation are considered in this guide. 1.4 This guide is intended to assist in the selection of methodologies that can be used for evaluating corrosion inhibitors. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory requirements prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM G170-06(2020)e1 is classified under the following ICS (International Classification for Standards) categories: 75.020 - Extraction and processing of petroleum and natural gas. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM G170-06(2020)e1 has the following relationships with other standards: It is inter standard links to ASTM G3-14(2019), ASTM G16-13(2019), ASTM G96-90(2018), ASTM D4410-16, ASTM G3-14, ASTM G5-14, ASTM G3-13, ASTM G16-13, ASTM G96-90(2013), ASTM G46-94(2013), ASTM G111-97(2013), ASTM G5-13e1, ASTM G5-13, ASTM G5-13e2, ASTM G5-12. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM G170-06(2020)e1 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
´1
Designation:G170 −06 (Reapproved 2020)
Standard Guide for
Evaluating and Qualifying Oilfield and Refinery Corrosion
Inhibitors in the Laboratory
This standard is issued under the fixed designation G170; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Units statement was inserted in 1.5, and Terminology G15 was replaced by Terminology G193 editorially, and
editorial changes were made throughout in December 2020.
1. Scope 2. Referenced Documents
1.1 This guide covers some generally accepted laboratory
2.1 ASTM Standards:
methodologies that are used for evaluating corrosion inhibitors
D1141 Practice for the Preparation of Substitute Ocean
for oilfield and refinery applications in well defined flow
Water
conditions.
D4410 Terminology for Fluvial Sediment
1.2 This guide does not cover detailed calculations and
G1 Practice for Preparing, Cleaning, and Evaluating Corro-
methods, but rather covers a range of approaches which have
sion Test Specimens
found application in inhibitor evaluation.
G3 Practice for Conventions Applicable to Electrochemical
Measurements in Corrosion Testing
1.3 Only those methodologies that have found wide accep-
G5 Reference Test Method for Making Potentiodynamic
tance in inhibitor evaluation are considered in this guide.
Anodic Polarization Measurements
1.4 This guide is intended to assist in the selection of
G16 Guide for Applying Statistics to Analysis of Corrosion
methodologies that can be used for evaluating corrosion
Data
inhibitors.
G31 Guide for Laboratory Immersion Corrosion Testing of
1.5 The values stated in SI units are to be regarded as
Metals
standard. No other units of measurement are included in this
G46 Guide for Examination and Evaluation of Pitting Cor-
standard.
rosion
1.6 This standard does not purport to address all of the
G59 Test Method for Conducting Potentiodynamic Polariza-
safety concerns, if any, associated with its use. It is the
tion Resistance Measurements
responsibility of the user of this standard to establish appro-
G96 Guide for Online Monitoring of Corrosion in Plant
priate safety, health, and environmental practices and deter-
Equipment (Electrical and Electrochemical Methods)
mine the applicability of regulatory requirements prior to use.
G102 Practice for Calculation of Corrosion Rates and Re-
1.7 This international standard was developed in accor-
lated Information from Electrochemical Measurements
dance with internationally recognized principles on standard-
G106 Practice for Verification of Algorithm and Equipment
ization established in the Decision on Principles for the
for Electrochemical Impedance Measurements
Development of International Standards, Guides and Recom-
G111 Guide for Corrosion Tests in High Temperature or
mendations issued by the World Trade Organization Technical
High Pressure Environment, or Both
Barriers to Trade (TBT) Committee.
G193 Terminology and Acronyms Relating to Corrosion
This guide is under the jurisdiction ofASTM Committee G01 on Corrosion of
Metals and is the direct responsibility of Subcommittee G01.05 on Laboratory
Corrosion Tests. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2020. Published December 2020. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2001. Last previous edition approved in 2012 as G170 – 06 (2012). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/G0170-06R20E01. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
´1
G170−06 (2020)
2.2 NACE Standards: 3.1.9 gas to oil ratio (GOR)—ratio of the amount of gas and
NACE-5A195 State-of-the-Art Report on Controlled-Flow oil transported through a pipe over a given time.
Laboratory Corrosion Test, Houston, TX, NACE Interna-
3.1.10 high-pressure—a pressure above ambient atmo-
tional Publication, Item No. 24187, December 1995
spheric pressure that cannot be contained in normal laboratory
NACE-ID196 Laboratory Test Methods for Evaluating Oil-
glassware. Typically, this is greater than 0.07 MPa (10 psig).
Field Corrosion Inhibitors, Houston, TX, NACE Interna-
3.1.11 high-temperature—temperatures above ambient
tional Publication, Item No. 24192, December 1996
laboratory temperature where sustained heating of the environ-
NACE-TM0196 Standard Test Method “Chemical Resis-
ment is required.
tance of Polymeric Materials by Periodic Evaluation,”
3.1.12 laboratory methodology—a small laboratory experi-
Houston, TX, NACE International Publication, Item No.
mental set up, that is used to generate the corrosion. Examples
21226, 1996
oflaboratorymethodologiesincluderotatingcylinderelectrode
2.3 ISO Standards:
(RCE), rotating cage (RC), and jet impingement (JI) under
ISO 696 Surface Active Agents — Measurements of Foam-
flowing conditions.
ing Power Modified Ross-Miles Method
ISO 6614 Petroleum Products — Determination of Water 3.1.13 live water—aqueous solution obtained from a pipe-
Separability of Petroleum Oils and Synthetic Fluids line or well. Usually live water is protected from atmospheric
oxygen.
3. Terminology
3.1.14 mass transfer coeffıcient (k, m/s)—the rate at which
3.1 Definitions of Terms Specific to This Standard:
the reactants (or products) are transferred to the surface (or
3.1.1 atmosphericpressureexperiment—anexperimentcon-
removed from the surface).
ducted at the ambient atmospheric pressure (typically less than
3.1.15 measuring technique—technique for determining the
0.07 MPa (10 psig)), using normal laboratory glassware.
rate of corrosion and the inhibitor efficiency. Examples of
3.1.2 batch inhibitor—an inhibitor that forms a film on the
measuring techniques are mass loss, linear polarization resis-
metal surface that persists to effect inhibition.
tance (LPR), electrochemical impedance spectroscopy (EIS),
3.1.3 batch treatment—a method of applying a batch inhibi- electrical resistance (ER), and potentiodynamic polarization
(PP) methods.
tor. Batch inhibitors are applied as a plug between pigs or as
slugs of chemical poured down the well bore. The batch
3.1.16 multiphase flow—simultaneous passage or transport
inhibitor is dissolved or dispersed in a medium, usually
of more than one phase, where the phases have different states
hydrocarbon and the inhibited solution is allowed to be in
(gas, liquid, and solid) or the same state (liquid), but different
contact with the surface that is to be protected for a fixed
fluid characteristics (viscosity, density, and specific gravity).
amountoftime.Duringthisperiod,theinhibitorfilmisformed
3.1.17 synthetic water—a synthetic solution prepared in the
on the surface and protects the surface during the passage of
laboratory using various chemicals. The composition is based
multiphase flow, for example, oil/water/gas.
on the composition of fluid found in an oil production system.
3.1.4 continuous inhibitor—an inhibitor that is continuously
3.1.18 Schmidt Number (Sc)—a measure of the ratio of the
injected into the system in order to effect inhibition. Since the
hydrodynamic boundary layer to the diffusion boundary layer.
surface receives full exposure to the inhibitor, the film repair is
This dimensionless parameter is equal to kinematic viscosity
continuous.
divided by diffusion coefficient.
3.1.5 emulsification-tendency—a property of an inhibitor
3.1.19 wall shear stress (τ, N/m )—a force per unit area on
that causes the water and hydrocarbon mixture to form an
the pipe due to fluid friction.
emulsion.Theemulsionformedcanbequitedifficulttoremove
3.2 The terminology used herein, if not specifically defined
and this will lead to separation difficulties in the production
otherwise, shall be in accordance with Terminology D4410 or
facilities.
G193. Definitions provided herein and not given in Terminol-
3.1.6 film persistency—ability of inhibitor film (usually
ogy D4410 or G193 are limited only to this guide.
batchinhibitor)towithstandtheforces(forexample,flow)that
tend to destroy the film over time.
4. Summary of Guide
3.1.7 flow loop—an experimental pipe that contains various
4.1 Inhibitor evaluation in the laboratory consists of two
corrosionprobestomonitorcorrosionrates.Aflowloopcanbe
steps(1)evaluationofinhibitorefficiencyand(2)evaluationof
constructed in the laboratory or attached to an operating
secondary inhibitor properties.
system.
4.2 Four laboratory methodologies, flow loop, rotating cyl-
3.1.8 foaming tendency—tendency of inhibitor in solution
inder electrode (RCE), rotating cage (RC), and jet impinge-
(water or hydrocarbon) to create and stabilize foam when gas
ment(JI)areavailabletoevaluatetheinhibitorefficiencyinthe
is purged through the solution.
laboratory. All four methodologies can be operated at atmo-
spheric and high pressure conditions. The corrosion rates can
AvailablefromNACEInternational(NACE),15835ParkTenPl.,Houston,TX
be measured using mass loss or electrochemical methods.
77084, http://www.nace.org.
Using the methodologies, several variables, compositions of
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
4th Floor, New York, NY 10036, http://www.ansi.org. material, composition of environment (gas and liquid),
´1
G170−06 (2020)
temperature, pressure, and flow, that influence the corrosion on production rate), and flow direction (that is, upward or
rate in the field can be simulated in the laboratory. Rotating downward). The common flow regimes in vertical upward
cylinder electrode (RCE), rotating cage (RC), and jet impinge- flow, vertical downward flow, and horizontal flow are pre-
ment (JI) methodologies are compact, inexpensive, hydrody- sented in Figs. 1-3 respectively (2, 3).
namically characterized, and scalable; that is, can be carried
5.3 Depending on the flow regime, the pipe may undergo
out at various flow conditions.
various forms of corrosion, including general, localized, flow-
4.3 Several secondary properties of the inhibitor are evalu- induced,anderosion-corrosion.Oneofthepredominantfailure
ated before the inhibitor is applied in the field. They are mechanisms of multiphase systems is pitting corrosion.
water/oil partitioning, solubility, emulsification tendency, foam
5.4 The performance of a corrosion inhibitor is influenced
tendency, thermal stability, toxicity, and compatibility with
primarily by the nature of inhibitor, operating conditions of a
other additives/materials. Laboratory methods to evaluate the
system, and the method by which it is added. Two types of
secondary properties are described.
inhibitors are used in the oil field, continuous and batch.
Water-soluble and oil-soluble, water-dispersible inhibitors are
5. Significance and Use
added continuously. Oil-soluble inhibitors are, in general,
5.1 Corrosion inhibitors continue to play a key role in
batch treated. The test methods to evaluate the inhibitors for a
controlling internal corrosion associated with oil and gas
particular field should be carried so that the operating condi-
production and transportation. This results primarily from the
tionsofthesystemaresimulated.Thusduringtheevaluationof
industry’s extensive use of carbon and low alloy steels, which,
a corrosion inhibitor, an important first step is to identify the
for many applications, are economic materials of construction
field conditions under which the inhibitor is intended to be
that generally exhibit poor corrosion resistance. As a
used. The environmental conditions in the field locations will
consequence,thereisastrongrelianceoninhibitordeployment
dictate the laboratory conditions under which testing is carried
for achieving cost-effective corrosion control, especially for
out.
treating long flowlines and main export pipelines (1).
5.5 Various parameters that influence corrosion rates, and
5.2 Formultiphaseflow,theaqueous-oil-gasinterphasescan
hence, inhibitor performance in a given system are (1) com-
take any of an infinite number of possible forms. These forms
position of material (2) composition of gas and liquid (3)
are delineated into certain classes of interfacial distribution
temperature (4) flow and (5) pressure.
called flow regimes. The flow regimes depend on the inclina-
5.5.1 In order for a test method to be relevant to a particular
tion of the pipe (that is, vertical or horizontal), flow rate (based
system, it should be possible to control the combined effects of
various parameters that influence corrosion in that system. A
test method is considered to be predictive if it can generate
The boldface numbers in parentheses refer to the list of references at the end of
information regarding type of corrosion, general and localized
this standard.
NOTE 1—ρ and ρ are gas and liquid densities and U and U are superficial velocities or the volume of flow rates of the liquid and gas per unit
G L L G
cross-sectional area of the channel (2).
FIG. 1Flow Regimes for Vertical Upward Multiphase Flow
´1
G170−06 (2020)
5.8 Flow is an indirect variable, and simulation of flow in
the laboratory is not direct. For this reason, the hydrodynamic
flow parameters are determined, and then the laboratory
corrosion tests are conducted under the calculated hydrody-
namic parameters. The fundamental assumption in this ap-
proach is that, when the hydrodynamic parameters of different
geometries are the same, then the corrosion mechanism will be
the same. Under these conditions, the corrosion rate and the
efficiency of corrosion inhibition in the laboratory and in the
field are similar. The commonly used hydrodynamic param-
eters are wall shear stress, Reynolds number, and mass transfer
coefficient (3, 5).
5.9 Neither the flow rate (m/s) nor dimensionless param-
eterscanbedirectlyrelatedtothelocalhydrodynamicforcesat
the material surface that may be responsible for accelerated
localized attack. Local hydrodynamic forces are influenced by
severalfactorsincludingpipeinclination,position(thatis,3,6,
9 o’clock), presence of bends, deposits, edges, welds,
FIG. 2Flow Regimes for Vertical Downward Flow (2)
expansion, and contraction. The flow rate and dimensionless
parameters describe only bulk, or average, properties of the
dynamic system. Thus the wall shear stress and mass transfer
corrosion rates, nature of inhibition, and life of inhibitor film
coefficient can be calculated only as averages at the surface
(or adsorbed layer). Rather than try to perfectly reproduce all
with an average surface roughness.
thefieldconditions,amorepracticalapproachistoidentifythe
5.10 Inhibitors are first screened in the laboratory, then
critical factors that determine/impact inhibitor performance
evaluated in the field, and finally used in engineering opera-
and then design experiments in a way which best evaluates
tions. The laboratory methodologies, therefore, should be
these factors.
carried out in a compact system with the capacity to evaluate
5.6 Composition of material, composition of gas and liquid
various products quickly with the flow pattern and regime
(oil and water), temperature, and pressure are direct variables.
characterized. The results obtained should be relevant to field
Simulation of them in the laboratory is direct. Laboratory
operation,shouldbepredictiveoffieldperformanceintermsof
experimentsarecarriedoutatthetemperatureofthefieldusing
inhibitor efficiency, and should be scalable, that is, the experi-
coupons or electrodes made out of the field material (for
ments can be carried out at various hydrodynamic conditions.
example, carbon steel). The effect of pressure is simulated by
5.11 Flow loops are used to evaluate corrosion inhibitors
using a gas mixture with a composition similar to the field for
either in the laboratory or by attaching to a live pipe. The loop
atmospheric experiments and by using partial pressures similar
simulates the flow regime, but the apparatus is relatively
to those in the field for high pressure experiments.
sophisticated, and experiments are expensive and time con-
5.7 In multiphase systems there are three phases, oil, aque-
suming. The loop is considered sophisticated to be an ideal
ous (brine water), and gas. Corrosion occurs at places where
laboratory methodology under the scope of this guide.
the aqueous phase contacts the material (for example, steel).
The corrosivity of the aqueous phase is influenced by the 5.12 This guide discusses test facilities and considers the
composition and the concentration of dissolved gases (for
necessary elements which need to be built into a laboratory
example, H S and CO ). In evaluating corrosion inhibitors in strategyfortestingcorrosioninhibitorsformultiphasesystems.
2 2
the laboratory, aqueous phase is usually used with a positive
The emphasis is on those methodologies that are compact and
pressure of gas mixture to simulate the gaseous phase. The oil
scalable, hydrodynamically well characterized, and relatively
may have a major effect on the corrosion rate and inhibitor inexpensive to use. The laboratory methodologies are (1)
efficiency. The presence of oil phase in the test environment
rotating cylinder electrode (RCE) (2) rotating cage (RC) and
can have significantly different effects (4). The primary effect (3) jet impingement (JI). These methodologies can be used
of the oil phase is apparently on the protectiveness of the
under both atmospheric and high pressure conditions. Detailed
corrosion inhibitor. The oil phase may have the following description of RCE and JI are presented in NACE-5A195.
effects: (1) partitioning of inhibitor between phases (2) chang-
5.13 Laboratory tests for inhibitor evaluation consist of two
ing the contact time of the aqueous phase on the pipe (3)
main components–laboratory methodology and measurement
changing the wetting behaviour of the pipe surface (4) intro-
technique. The combinations of laboratory methodology and
ducingprotectivecompoundsthatarenaturallyoccurringinthe
measurement technique for inhibitor evaluation for multiphase
oil.
systems are presented in Table 1.
5.7.1 Inhibitor evaluation in the absence of the oil phase
cannot give an accurate picture of the behaviour of steel in 5.14 To develop an inhibitor selection strategy, in addition
multiphase environments. Ideally, the oil phase should be to inhibitor efficiency, several other key performance factors
present when testing the inhibitor in the laboratory. need to be evaluated: (1) water/oil partitioning, (2) solubility,
´1
G170−06 (2020)
NOTE 1—Boundary conditions given by two studies are presented (2).
FIG. 3Flow Regimes for Horizontal Flow
TABLE 1 Laboratory Methodologies and Measurement
tice. Their composition should be specified in the work plan
Techniques for Corrosion Inhibitor Evaluation
and recorded in the laboratory logbook. Test solutions should
Laboratory Measurement Aqueous/Oil/ Remarks
be prepared using analytical grade reagents and deionized
Methodology Technique Gas Phase
water, unless otherwise specified. If other grades of chemicals
RCE mass loss, aqueous phase specimen is a
are used, their purity or grade should be recorded in the
electrochemical cylinder
laboratory logbook.
RCE mass loss aqueous/oil specimen is a
phase cylinder
6.2 The solutions should be deaerated by passing nitrogen
JI mass loss, aqueous phase specimen is a disc
electrochemical
(or any other inert gas) or carbon dioxide and kept under
JI mass loss aqueous/oil specimen is a disc
deaerated conditions. The solution pH before and after testing
phase
should be measured and recorded. If possible, the solution pH
JI electrochemical aqueous phase specimen is a ring
measurements
should be monitored continuously during the test. Solutions
RC mass loss aqueous phase electrochemical
should be transferred from the preconditioning vessel to the
or measurements
aqueous/oil cannot test vessel under positive nitrogen pressure to minimize air
phase be carried out
contamination during the transfer operation.
6.3 The appropriate composition of gas can be obtained by
mixing H S and CO streams from the standard laboratory gas
2 2
(3) emulsification tendency, (4) foaming tendency, (5) thermal
supply. Nitrogen can be used as a diluent to obtain the required
stability, (6) toxicity, and (7) compatibility with other
partial pressures of the corrosive gases. Alternatively, gas
additives/materials.
mixtures of the required compositions can be purchased from
suppliers of industrial gases. The concentrations of impurities,
6. Preparation of Test Solutions
particularly oxygen, should be kept as low as technically
6.1 Ideally, all solutions (oil and aqueous) should be ob-
possible (below 5 ppb, preferably under 1 ppb oxygen in
tained from the field for which the inhibitor is being evaluated.
solution). The solution oxygen concentration depends on the
It is important that live fluids do not already contain corrosion
quality of gases used to purge the electrolyte.
inhibitor. In the absence of live fluids, synthetic solutions
should be used, the composition of which, however, should be 6.4 Measure Inhibitor concentrations and report in %
based on field water analysis. Alternatively, standard brine (in weight/volume or ppm w/v (percentage or parts per million,
accordance with Practice D1141) should be employed. The weight in volume basis). The method of injecting the inhibitor
solutions should be prepared following good laboratory prac- into the test solution should reflect the actual field application
´1
G170−06 (2020)
that is being tested. Water-soluble inhibitors may be injected adecreaseofpressure,repressurizetheautoclaveagain.Repeat
neat (as-received) into the test solution (aqueous phase). To the process until no further pressure drop occurs. Then,
avoid the errors associated with handling small volumes of pressurize the autoclave with CO , by opening the CO gas
2 2
solution, an inhibitor stock solution may be prepared by cylinder at a pressure equal to the CO +H S partial pressure.
2 2
diluting the as-received chemical in an appropriate solvent. Leave it for 10 min. If there is a decrease of pressure,
The type of solvent and the concentration of the stock solution repressurize the autoclave again with CO gas. Repeat the
will depend on the characteristics of the inhibitor and on the process until no further pressure drop is observed. Finally,
specified test conditions. pressurizetheautoclavewiththeinertgas,byopeningtheinert
gas cylinder at the total gas pressure at which the experiments
6.5 Oil-soluble, water-dispersible inhibitor solutions are
are intended to be carried out.
prepared by the partition method. Place the required amounts
of crude oil, or condensate, and brine in the partitioning vessel
7. Materials
(usually a separation funnel). The relative volumes of hydro-
7.1 Methods for preparing specimens for tests and for
carbon and aqueous phases reflect the water cut to be tested. If
removing specimens after the test are described in Practice G1.
actual field condensate is not available, heptane, kerosene, or
Standardlaboratoryglasswareshouldbeusedforweighingand
any suitable hydrocarbon can be used as a substitute for the oil
measuring reagent volumes.
phase.Add the corrosion inhibitor to the oil phase. Vigorously
7.2 The specimen should be made of the material (for
shake the vessel to mix both phases thoroughly and allow the
example, carbon steel) for which the inhibitor is being evalu-
phases to separate. Heating to the minimum expected field
ated. Corrosion rates and inhibitor performance change by
temperature may help in the separation and will also provide
severalordersofmagnitudeassurfaceroughnesschangesfrom
more meaningful results; remove the aqueous phase and use as
rough to fine. The surface roughness should be kept the same
the test solution.
during inhibitor screening and, if possible, the surface rough-
6.6 Oil-soluble inhibitors (usually as batch inhibitors) are
nessofspecimensusedinthelaboratoryexperimentsshouldbe
applied in a separate procedure and the corrosion test is carried
related to that of field pipe.The specimens should be ground to
outafterthis.Theinhibitorisdissolvedintheoilphasetoform
a specified surface finish. The grinding should produce a
an inhibited oil-phase. The corrosion coupon or electrode is
reproducible surface finish, with no rust deposits, pits, or deep
exposed to this solution for a certain amount of time (usually
scratches. All sharp edges on the specimen should be ground.
30 min). The coupon or electrode is then removed and
All loose dirt particles should be wiped off using tissue paper.
introduced into the experimental vessel for the corrosion test.
7.3 Rinse the specimens with distilled water and then
6.7 Depending on the size of experimental vessel, heating
degrease the specimens by immersing in acetone (or methanol)
unit (mantle, bath, or wrapper around the vessel), difference
and ultrasonically cleaning for 1 min; dry the specimens with
between room and experimental temperatures, a range of
a paper towel. Do not touch the surface of the specimens with
temperature may prevail within the vessel. Exercise precaution
bare hands; and weigh the specimens to the nearest 0.1 mg.
to avoid or minimize the temperature differentials. The test
Measure the dimensions of the specimens to the nearest 1 mm,
vessels should be heated slowly to avoid overheating and, in
and calculate the area of each specimen.
the case of glass autoclaves, to prevent high thermal stresses
7.4 Ingeneral,specimensareheldinaninsulatingspecimen
between the inner and outer walls.The exact protocol followed
holder;thetypeofholdervarieswiththetest.Installthefreshly
will depend on the controller, the size and output of the heater,
prepared specimens in the synthetic materials holder and
and parameters such as vessel size, amount of liquid, thermal
tightenthem.Placethespecimenholderinthevessel,andclose
conductivity of liquid, and agitation.The pressure in the vessel
the lid. Fill the vessel with the preconditioned (deaerated) test
should be monitored during heating to make sure it does not
solution and continue deaerating for at least 1 h using nitrogen.
exceed the relief pressure. If necessary, some of the gas in the
vessel may be bled off to reduce the pressure. The test
8. Laboratory Methods for Evaluating Inhibitor
temperature should be maintained within 2 °C of the specified
Efficiency
temperature. Once the test temperature is reached, the test
8.1 Rotating Cylinder Electrode (RCE):
pressure should be adjusted to the predetermined value. The
8.1.1 The RCE test system is compact, relatively
pressure should be maintained within 610 % of the specified
inexpensive, and easily controlled (7). It operates in the
value for the duration of the test.
turbulent regime over a wide range of Reynolds numbers. The
6.8 For high-temperature, high-pressure experiments, using
apparatus operates under known and controlled hydrodynamic
a pre-mixed gas composition, pressurize the autoclave using
conditions. The experiments require small amounts of fluid,
the specified gas composition, and depressurize to approxi-
and mass loss and electrochemical measurements can be made.
mately 0.2 bar above atmospheric pressure. Repeat this cycle
General procedures for specimen preparation, methods of
of pressurizing/depressurizing at least twice to ensure that the
cleaning, corrosion rate measurements and evaluation of re-
gas cap has the required composition. Finally, pressurize the
sults are described in detail in Guides G16 and G31, Practices
autoclave to the test pressure.
G102 and G106, Test Method G59, and NACE-1D196.
6.9 For high-temperature, high-pressure experiments (6) 8.1.2 At very low electrode rotation speeds, the flow around
using individual gases, first pressurize the autoclave with H S the RCE is laminar and occurs in concentric circles around the
to the required partial pressure. Leave it for 10 min. If there is cylinder. At higher rotation speeds this simple flow pattern
´1
G170−06 (2020)
becomes unstable. Cellular motion is imposed on the flow
producing toroidal Taylor vortices containing a radial compo-
nent of velocity, but the bulk of the flow remains essentially
laminar. As rotational speeds increase further, the flow be-
comes fully turbulent and eddies increasingly break up the
regular flow pattern. The transition to fully turbulent flow
occurs at about Re 200. In the turbulent flow region, the RCE
can be applied to simulate flow behavior by hydrodynamic
analysis.
8.1.3 A typical RCE apparatus consists of a rotating unit
driven by a motor that is attached to a sample holder.Asystem
with a range of rotational speeds from 100 rpm to 10 000 rpm
with an accuracy of 62 rpm is typical. It is essential to be able
to rotate the electrode at both low and high speeds and to be
able to measure the speed and maintain it constant.At the side
of the sample holder, electrical connections to the electrodes
aremadebyabrushormercurycontact.Thecylindergeometry
FIG. 5Schematic Representation of RCE for Nonuniform Current
isusuallydefinedintermsofthelength-to-diameterratio.Both
and Potential Distribution
low and high ratios are used, with ratios varying between 0.3
(Below the Mass-Transfer-Limiting Current) (7)
and 3.0. The corrosion rates are measured using conventional
electrochemicalinstruments.Detailedproceduresaredescribed
in Practices G3, G102, and G106, Reference Test Method G5,
8.1.5 For RCE, the reaction rates may be mass transport
Test Method G59, and Guide G96. The rotating cylinder can
controlled. Provided the IR drop is constant in the cell, the
also be used as a mass loss coupon when the mass loss is
current distribution over the electrode surface may be uniform,
sufficiently large to be accurately measured using a conven-
and concentration (of reactants or product) changes may be
tional balance (with accuracy 0.1 mg).
calculated even though the fluid flow is generally turbulent.
8.1.4 In many designs, two electrodes, inner (rotating) and
Laminar flow is limited because, in the conventional
outer (stationary) electrodes are used. The outer electrode is
arrangement, the RCE is enclosed within a concentric cell and
usually the counter electrode. Below the mass-transfer-limited
Re ~ 200, corresponding to rotation speeds of <10 rpm.
crit
conditions, the current distribution is uniform if the electrode
Notwithstanding the instability of turbulent motion, the RCE
and the electrical isolation planes are at right angles, as shown
has found a wide variety of applications, especially when
in Fig. 4. If the electrodes are not placed in this way (as shown
naturallyturbulentindustrialprocesseshavetobesimulatedon
in Fig. 5), the current distribution is not uniform (7). When
a smaller scale or when mass transport must be maximized.
designing the rotating cylinder apparatus, the outer concentric
8.1.6 The limiting current density (i ) for turbulent flow in
L
electrodemustbeplacedseveralinner-cylinderdiametersaway
RCE is described as (7-8).
from the inner concentric electrode for Eq 1 to be valid (see
0.7 20.3 20.644
8.1.6). i 5 0.0791nFC ωr r/v v/D (1)
~ ! ~ ! ~ !
L
where:
n = number of electrons,
F = Faraday constant,
C = concentration of the reactant,
ω = angular velocity,
r = radius of the electrode,
v = kinematic-viscosity, and
D = diffusion coefficient.
8.1.7 When the wall shear stresses are equal in the two
geometries (that is, the RCE and the pipe), then similar
hydrodynamic conditions, for example, turbulence, are main-
tained. Under these conditions, the corrosion mechanism (not
the rate) is hypothesized to be the same in the two geometries.
8.1.8 The wall shear stress of RCE, τ is given as (9).
RCE
20.3 2 2
τ 5 0.0791Re ρr ω (2)
RCE
where:
Re = Reynolds number,
ρ = density,
FIG. 4Schematic Representation of RCE for Uniform Current
ω = angular velocity, and
and Potential Distribution
r = radius of the cylinder.
(Below the Mass-Transfer-Limiting Current) (7)
´1
G170−06 (2020)
8.1.9 Eq 2 can be used as a first approximation to establish 8.2.3 Depending on the rotation speed, the volume of the
the appropriate RCE velocity for modelling the desired system container, and the fluids used, the flow pattern can be divided
when evaluating corrosion inhibitors by single-phase flow. into four zones (Fig. 8):
There may be instances in which Eq 3 does not provide a good
8.2.3.1 Homogeneous Zone—Vortex dimensions that have
approximation. Equality of shear stress in RCE and pipe
been observed (length and width) increase with rotation speed.
systems does not result in equal mass-transfer coefficients, but
8.2.3.2 Side-wall Affected Zone—Vortex length increases,
relationships exist between mass-transfer coefficients and wall
but the width has reached the side and collides with the wall.
shear stress.
8.2.3.3 Turbulent Zone—Vortex length penetrates into the
rotating cage unit and creates turbulent flow.
8.2 Rotating Cage:
8.2.1 Therotatingcage(RC)provideshigherflowvelocities 8.2.3.4 Top-cover Affected Zone—The liquid level oscillates
than can usually be obtained simply by stirring the solution and rises to the top, pushing the flow pattern due to the
(10, 11). Fig. 6 shows the schematic diagram of the rotating backward movement of the fluids, and changing the flow
cage system. The vessel should be manufactured from an inert pattern (the rate of vortex length increases at a lower rate).
material. Glass and acrylic have been used. A typical rotating
8.2.4 The wall shear stress can be calculated using Eq 3
cage system is described below. A base is fitted at the bottom
(12):
of the container.At the center of the TFE-fluorocarbon base, a
20.3 2 2.3
τ 5 0.0791 Re ρr ω (3)
RC
hole is drilled into which the lower end of the stirring rod is
placed.This arrangement stabilizes the stirrer and the coupons. where:
Eight coupons (each of surface area about 36 cm ) are
r = the radius of the rotating cage.
supported between two TFE-fluorocarbon disks mounted 76
8.2.5 Eq 4 can be used to calculate the wall shear stresses in
mm apart on the stirring rod of the autoclave. Holes (diameter
the homogeneous zone only. In the turbulent zone, the wall
1cm)aredrilledinthetopandbottomTFE-fluorocarbonplates
shear stress may be higher than predicted by Eq 4; on the other
of the cage to increase the turbulence on the inside surface of
hand,intheside-affectedandtop-coveraffectedzones,thewall
the coupon. This experimental setup can be used at tempera-
shearstressmaybelessthanthatpredictedbyEq4,becauseof
tures up to 70 °C and rotation speeds up to 1 000 rpm. The
the movement of a portion of the fluid in the opposite direction
corrosion rates are determined by mass loss.
by the vortex-driven flow.
8.2.2 Rotatingcageexperimentsneedrelativelyinexpensive
8.2.6 The approach to correlate hydrodynamic relationships
facilities that can be easily duplicated to save investigation
between RC and another system (for example, pipe) is the
time. The tests are relatively simple to conduct. The flow
same as that used for RCE. When the wall shear stresses are
intensity is probably highest in the gap (grooves) between the
equal in the two geometries (for example, RC and pipe), then
coupons (Fig. 7). The grooves in the RC contribute to different
similar hydrodynamic conditions, for example, turbulence, are
types of corrosion artifacts. Local high turbulence at the
maintained.
leading and trailing edges of the grooves increases localized
corrosion rates. Procedures for examining and evaluating
8.3 Jet Impingement (JI):
pitting corrosion are described in Guide G46. A decrease in
8.3.1 Jet impingement is a widely used technique to study
corrosionmaybeobservedinsidethegroovewherethecoupon
flow-induced corrosion. The high turbulence associated with
is protected from the turbulent flow.
jet impingement is considered to simulate the turbulence
encountered at threaded joints, bends, valves, welds, and so
forth in tubulars, flowlines, and pipelines (13).
8.3.2 Jet impingement is a widely used test methodology to
study flow-accelerated corrosion and is a relatively new
methodology for evaluating the performance of corrosion
inhibitors. The jet impingement test can simulate reliably and
reproducibly high turbulence conditions in multiphase systems
(for example, oil, water and gas). It requires relatively small
volumes of test fluids and is controlled easily.
8.3.3 Jet impingement systems can be used to study the
effects of differential mass-transfer cells if the electrode diam-
eter is more than five times larger than the diameter of the jet
nozzle. Alternatively, a configuration or geometry of the
probe/electrode can be designed to account for a specific shear
stress region.
8.3.4 The advantage of using an impinging jet is that the
flow profile produced by an impinging jet is mathematically
well defined and expressed. By changing the jet velocity, the
nozzle diameter, and the distance between nozzle and test
coupon, the flow profile inside an impinging jet can easily be
FIG. 6Schematic Diagram of Rotating Cage (12) adjustedoverawiderangetosimulatevariousflowconditions.
´1
G170−06 (2020)
NOTE 1—The gap between the coupons (A) and the hole (B) introduce localized turbulence.
FIG. 7Photo of Rotating Cage Containing Coupons
in Fig. 9 (14). Under these conditions, a stagnation point exists
at the intersection of this axis with the plate and the flow is
symmetric about the axis. Because the flow is axis-symmetric,
only the flow and fluid properties in the radial plane normal to
the disk are considered.
8.3.6 RegionAis the region in which the flow is essentially
laminar near the plate and the principal velocity component
changes from axial to radial, with a stagnation point at the
center. Region A extends from the central axis to the point of
maximum velocity and minimum jet thickness at approxi-
mately r=2r . The local velocity field is complex, but is
o
mathematically definable. Because the flow vector is changing
rapidly as radial distance increases, this region is of little use
for correlation to field conditions.
8.3.7 Region B is a region of rapidly increasing turbulence,
with the flow developing into a wall jet; that is, the primary
flow vector is parallel to the solid surface. This region extends
radially to approximatelyr=4r . The flow pattern is charac-
o
terizedbyhighturbulence,alargevelocitygradientatthewall,
and high wall shear stress. Region B is of primary interest for
studying fluid flow effects on corrosion in high turbulence
areas and areas of flow disruption. The equation for the wall
shear stress in this region is:
22.0
r
2 20.182
τ 5 0.1788 3ρ 3U 3Re (4)
S D
w 0
r
where:
τ = wall shear stress (N/m ),
w
ρ = density (kg/m ),
U = velocity (m/s) of the flow at the position of leaving the
nozzle,
r = distance from stagnant point, m,
r = jet nozzle radius, m, and
Re = Reynold’s number.
8.3.8 The jet Reynolds number is defined as:
2r 3U
0 0
Re 5 (5)
ν
FIG. 8Flow Patterns in a Rotating Cage
where:
ν = the kinematic viscosity of the testing liquid, (m /s).
8.3.5 The typical flow field established by a jet impinging
on a flat plate with central axis normal to the plate is illustrated ν 5 µ/ρ (6)
~ !
´1
G170−06 (2020)
FIG. 9Different Flow Regions on a Jet Impingement
where: are located in the closed compartment of the cell. Up to four
multiple samples can be used simultaneously.
µ = viscosity, and
8.3.11 TheefficiencyofaninhibitorinJIandinthefieldcan
ρ = density.
be correlated using wall shear stress, as for RCE and RC.
8.3.9 In Region C, the bulk flow rate and turbulence decay
8.4 High Pressure Experiments:
rapidly as the thickness of the wall jet increases, momentum is
transferred away from the plate, and the surrounding fluid is 8.4.1 In order to simulate the effects of partial pressures of
corrosive gases (CO,H S), experiments should be carried out
entrained in the jet. This region is amenable to mathematical
2 2
characterization, but the flow cannot correlate to field under high pressure (see Guide G111).All laboratory method-
ologies used in atmospheric pressure tests can also be pressur-
conditions, since momentum transfer and fluid entrainment in
this region are in the opposite direction from pipe flow. ized to simulate high-pressure pipeline operation. The meth-
odologiesarehigh-temperature,high-pressurerotatingcylinder
8.3.10 One design of JI consists of a central cell with four
arms containing the nozzles. The impeller is housed in the cell electrode (HTHPRCE); high-temperature, high-pressure rotat-
ing cage (HTHPRC); and high-temperature, high-pressure jet
body and is driven by a motor magnetically coupled to the
impeller shaft. Fluid from the cell is forced by the impeller impingement (HTHPJI).
through the nozzles and is recirculated to the cell, as shown in 8.4.2 The analysis of corrosion inhibitors in high-pressure
Fig. 10. In this compact design, all moving parts of the pump experiments should be performed using an autoclave. The
FIG. 10Schematic Diagram of Jet Impingement
´1
G170−06 (2020)
autoclave is equipped with various measuring and regulating 9. Laboratory Methods for Determining Secondary
devices. Corrosion rates can be determined by mass loss (in Inhibitor Properties
HTHPRC and HTHPJI) and electrochemical methods (HTH-
9.1 Oil Water Partitioning:
PRCE and HTHPJI).
9.1.1 In oil and gas production, corrosion-related problems
8.4.3 A high-temperature, high-pressure system for electro-
are invariably attributed to the presence of aqueous phase.
chemical measurements should possess an electrically isolated
Therefore, in order to prevent corrosion, the inhibitor must be
electrode system, an electrically isolated motor for rotating the
present in the aqueous phase. This is achieved by partitioning
electrode,andavesselthatcanwithstandhighpressurewithout
or dispersion of the inhibitor from the hydrocarbon and is
leakage.
enabled by the inhibitor being water dispersible or, more
8.4.4 The design of a vessel that can be used under
commonly,watersoluble.Irrespectiveofthewatersolubilityof
pressurizedconditionsisshowninFig.11 (15, 16).Thestirring
the inhibitor, many factors influence the ability to partition
rodofanautoclavecanbemodifiedbydrillingaholeintherod
efficiently. Because of the need to address the performance of
into which an insulator, for example, can be inserted. O-rings
an inhibitor in a given environment, it is most appropriate to
are used to prevent leakage. Inside the TFE-fluorocarbon
test the partitioning characteristics based on a direct perfor-
insulator, a metal rod is introduced (Fig. 12). One end of the
mance measure. This is achieved by measuring the corrosivity
metal rod is threaded so that cylindrical (RCE) specimens can
ofanaqueousphasefollowingpartitionofacorrosioninhibitor
be attached (Fig. 13). The other end of the rod, projecting
from the hydrocarbon. Information gained from partitioning
slightlyabovethemotorunit,isattacheddirectlytotherotating
studies can be used as a guide to obtain accurate dosing levels
unit, through which the electrical connection is made. The
to achieve the desired degree of protection in areas of water
stainless steel rod is rotated by a separate motor connected to
drop.
the rod using a belt. The counter and reference electrodes are
inserted inside the autoclave. The corrosion rates can be 9.1.2 A known amount of corrosion inhibitor is allowed to
determined using conventional electrochemical measurements. distribute between a crude oil and an aqueous phase over a
8.4.5 The experimental setup similar to the one used for periodofupto24h.Typically,arangeofwater/crudeoilratios
atmospheric pressure RC and JI can be used to carry out (1:9, 1:1, 8:2 V/V) and corrosion inhibitor concentrations are
HTHPRC and HTHPJI except that an autoclave is used. The used. During each test, the fluids are visually examined to
hydrodynamic calculation of elevated pressure experiments is ensure that the corrosion inhibitor does not cause formation of
the same as that for atmospheric experiments. a stable emulsion or
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...