ASTM F640-23
(Test Method)Standard Test Methods for Determining Radiopacity for Medical Use
Standard Test Methods for Determining Radiopacity for Medical Use
SIGNIFICANCE AND USE
5.1 These methods are intended to determine whether a material, product, or part of a product has the degree of radiopacity desired for its application as a medical device in the human body. This method allows for comparison with or without the use of a body mimic. Comparisons without the use of a body mimic should be used with caution as the relative radiopacity can be affected when imaging through the human body.
5.2 These methods allow for both qualitative and quantitative evaluation in different comparative situations.
SCOPE
1.1 These test methods cover the determination of the radiopacity of materials and products utilizing X-ray based techniques, including fluoroscopy, angiography, CT (computed tomography), and DEXA (dual energy X-ray absorptiometry), also known as DXA, The results of these measurements are an indication of the likelihood of locating the product within the human body.
1.2 Radiopacity is determined by (a) qualitatively comparing image(s) of a test specimen and a user-defined standard, with or without the use of a body mimic; or (b) quantitatively determining the specific difference in optical density or pixel intensity between the image of a test specimen and the image of a user-defined standard, with or without the use of a body mimic.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 28-Feb-2023
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.15 - Material Test Methods
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Dec-2019
- Effective Date
- 01-Mar-2019
- Effective Date
- 01-Jan-2018
- Effective Date
- 15-Jun-2017
- Effective Date
- 01-Feb-2017
- Effective Date
- 01-Aug-2016
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Dec-2015
- Effective Date
- 01-Sep-2015
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 15-Jun-2013
- Effective Date
- 01-Jun-2013
Overview
ASTM F640-23: Standard Test Methods for Determining Radiopacity for Medical Use establishes methods for evaluating the radiopacity of materials and products intended for medical applications within the human body. Developed by ASTM International, this standard is crucial for manufacturers and laboratories needing to ensure that medical devices and components can be accurately located using X-ray-based imaging techniques. The test methods included enable both qualitative and quantitative assessment of radiopacity, supporting critical decisions in the design and validation of surgical devices and implants.
Radiopacity is a key property required in many medical devices to ensure visibility during diagnostic or therapeutic procedures. Accurate measurement of this property allows for improved device safety, traceability, and regulatory compliance.
Key Topics
- Radiopacity Determination: Methods for establishing the radiopacity of materials using X-ray techniques, including fluoroscopy, angiography, computed tomography (CT), and dual energy X-ray absorptiometry (DEXA or DXA).
- Qualitative vs. Quantitative Analysis: These test methods allow for visual (qualitative) comparisons and digital (quantitative) evaluation based on optical density or pixel intensity measured against a user-defined standard.
- Use of Body Mimics: Tests may be conducted with or without a body mimic, such as phantoms, animal tissues, cadavers, or materials that simulate human tissue, to better replicate clinical conditions.
- Reporting and Documentation: Detailed requirements for reporting test conditions, specimen descriptions, imaging parameters, and results, supporting reproducibility and regulatory submissions.
- Apparatus and Materials: Guidance on the selection of X-ray imaging systems, digital analysis tools, standards for comparison, and ancillary materials such as step wedges and rubber blankets for image quality optimization.
Applications
- Medical Device Manufacturing: Ensuring that implants, surgical instruments, and other medical devices have adequate radiopacity for in vivo tracking and inspection.
- Quality Assurance and Regulatory Compliance: Providing validated data to demonstrate compliance with regulatory requirements for device visibility under X-ray, CT, or fluoroscopy.
- Research and Development: Aiding in the selection and formulation of materials for improved imaging characteristics in new medical device designs.
- Clinical Applications: Devices such as catheters, stents, shunt assemblies, and orthopedic implants must be readily identifiable in diagnostic images, facilitating precise placement, retrieval, or adjustment.
- Material Comparisons: Supporting material certification, supplier evaluation, and benchmarking of existing versus alternative device materials.
- Custom Standards: Flexibility to define user-specific standards or reference materials, adapted to the intended use and anatomy, enhances practical relevance in testing.
Related Standards
- ASTM B209/B209M: Specification for Aluminum and Aluminum-Alloy Sheet and Plate - referenced for material purity in step wedges.
- ASTM D3182: Practice for Rubber - covers materials and procedures for mixing compounds and preparing vulcanized sheets, relevant for sample preparation.
- ASTM E94/E94M: Guide for Radiographic Examination Using Industrial Radiographic Film - provides back-scatter protection recommendations.
- ASTM E1316: Terminology for Nondestructive Examinations - ensures consistent terminology use.
- ASTM F647: Practice for Evaluating and Specifying Implantable Shunt Assemblies for Neurosurgical Application - referenced for application-specific considerations.
Summary
By following the procedures detailed in ASTM F640-23, stakeholders in the medical device industry can confidently assess and document the radiopacity of products, optimizing patient safety and supporting robust compliance with international standards for radio-visible medical components. This standard ultimately enhances both the reliability of clinical procedures and the traceability of medical devices throughout their lifecycle.
Buy Documents
ASTM F640-23 - Standard Test Methods for Determining Radiopacity for Medical Use
REDLINE ASTM F640-23 - Standard Test Methods for Determining Radiopacity for Medical Use
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F640-23 is a standard published by ASTM International. Its full title is "Standard Test Methods for Determining Radiopacity for Medical Use". This standard covers: SIGNIFICANCE AND USE 5.1 These methods are intended to determine whether a material, product, or part of a product has the degree of radiopacity desired for its application as a medical device in the human body. This method allows for comparison with or without the use of a body mimic. Comparisons without the use of a body mimic should be used with caution as the relative radiopacity can be affected when imaging through the human body. 5.2 These methods allow for both qualitative and quantitative evaluation in different comparative situations. SCOPE 1.1 These test methods cover the determination of the radiopacity of materials and products utilizing X-ray based techniques, including fluoroscopy, angiography, CT (computed tomography), and DEXA (dual energy X-ray absorptiometry), also known as DXA, The results of these measurements are an indication of the likelihood of locating the product within the human body. 1.2 Radiopacity is determined by (a) qualitatively comparing image(s) of a test specimen and a user-defined standard, with or without the use of a body mimic; or (b) quantitatively determining the specific difference in optical density or pixel intensity between the image of a test specimen and the image of a user-defined standard, with or without the use of a body mimic. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 These methods are intended to determine whether a material, product, or part of a product has the degree of radiopacity desired for its application as a medical device in the human body. This method allows for comparison with or without the use of a body mimic. Comparisons without the use of a body mimic should be used with caution as the relative radiopacity can be affected when imaging through the human body. 5.2 These methods allow for both qualitative and quantitative evaluation in different comparative situations. SCOPE 1.1 These test methods cover the determination of the radiopacity of materials and products utilizing X-ray based techniques, including fluoroscopy, angiography, CT (computed tomography), and DEXA (dual energy X-ray absorptiometry), also known as DXA, The results of these measurements are an indication of the likelihood of locating the product within the human body. 1.2 Radiopacity is determined by (a) qualitatively comparing image(s) of a test specimen and a user-defined standard, with or without the use of a body mimic; or (b) quantitatively determining the specific difference in optical density or pixel intensity between the image of a test specimen and the image of a user-defined standard, with or without the use of a body mimic. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F640-23 is classified under the following ICS (International Classification for Standards) categories: 11.120.01 - Pharmaceutics in general; 83.080.01 - Plastics in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F640-23 has the following relationships with other standards: It is inter standard links to ASTM E1316-24, ASTM E1316-19b, ASTM E1316-19, ASTM E1316-18, ASTM E1316-17a, ASTM E1316-17, ASTM E1316-16a, ASTM E1316-16, ASTM E1316-15a, ASTM E1316-15, ASTM E1316-14e1, ASTM E1316-14, ASTM E1316-13d, ASTM E1316-13c, ASTM E1316-13b. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F640-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F640 − 23
Standard Test Methods for
Determining Radiopacity for Medical Use
This standard is issued under the fixed designation F640; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* Alloy Sheet and Plate
D3182 Practice for Rubber—Materials, Equipment, and Pro-
1.1 These test methods cover the determination of the
cedures for Mixing Standard Compounds and Preparing
radiopacity of materials and products utilizing X-ray based
Standard Vulcanized Sheets
techniques, including fluoroscopy, angiography, CT (computed
E94/E94M Guide for Radiographic Examination Using In-
tomography), and DEXA (dual energy X-ray absorptiometry),
dustrial Radiographic Film
also known as DXA, The results of these measurements are an
E1316 Terminology for Nondestructive Examinations
indication of the likelihood of locating the product within the
F647 Practice for Evaluating and Specifying Implantable
human body.
Shunt Assemblies for Neurosurgical Application
1.2 Radiopacity is determined by (a) qualitatively compar-
ing image(s) of a test specimen and a user-defined standard,
3. Terminology
with or without the use of a body mimic; or (b) quantitatively
determining the specific difference in optical density or pixel
3.1 Definitions—For definitions of terms relating to X-ray
intensity between the image of a test specimen and the image procedures, refer to Terminology E1316.
of a user-defined standard, with or without the use of a body
3.2 Descriptions of Terms:
mimic.
3.2.1 body mimic, n—a piece of material, a phantom, a
1.3 The values stated in SI units are to be regarded as cadaver, or an animal utilized to mimic the appropriate X-ray
standard. No other units of measurement are included in this attenuation through a particular part of the human body.
standard.
3.2.2 digital resolution, n—the number of pixels per inch in
1.4 This standard does not purport to address all of the
a digital image.
safety concerns, if any, associated with its use. It is the
3.2.2.1 Discussion—This may be different in the x and y
responsibility of the user of this standard to establish appro-
directions.
priate safety, health, and environmental practices and deter-
3.2.3 grayscale range, n—the number of levels in pixel
mine the applicability of regulatory limitations prior to use.
intensity resolved in the digital image.
1.5 This international standard was developed in accor-
dance with internationally recognized principles on standard-
3.2.3.1 Discussion—This range is normally 256 levels in an
ization established in the Decision on Principles for the
8-bit grayscale image, but 16-bit grayscale images can also be
Development of International Standards, Guides and Recom-
used.
mendations issued by the World Trade Organization Technical
3.2.4 pixel intensity, n—the grayscale level of a pixel
Barriers to Trade (TBT) Committee.
between 0 and 255, as determined by the digital analysis
program.
2. Referenced Documents
3.2.5 pixel intensity difference, n—the difference in gray-
2.1 ASTM Standards:
B209/B209M Specification for Aluminum and Aluminum- scale level between two regions or objects in an image,
reported to within the significance capability of the digital
analysis program.
These test methods are under the jurisdiction of ASTM Committee F04 on
Medical and Surgical Materials and Devices and are the direct responsibility of 3.2.6 user-defined standard, n—a comparison standard se-
Subcommittee F04.15 on Material Test Methods.
lected by the user. This standard could be a reference material
Current edition approved March 1, 2023. Published March 2023. Originally
or a predicate device.
approved in 1979. Last previous edition approved in 2020 as F640 – 20. DOI:
10.1520/F0640-23.
3.2.6.1 Discussion—This standard may be an existing medi-
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
cal product or a material in a particular form, it may be a
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
commercially available standard, or it may be one developed
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. by the user.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F640 − 23
4. Summary of Test Methods 6.3 Body Mimic (if used)—See Appendix X2 for discussion
of body mimics. (Note that this is not an all-inclusive list and
4.1 The test specimen is placed so it sits at or near the
other body mimics might be appropriate.)
middle of the X-ray image area in the X-ray imaging system.
6.3.1 Animal—An appropriate animal, or portion of appro-
X-ray images are made at specified voltages, times, and
priate animal, with which to perform the tests may be used.
currents that are typical of those used in the X-ray diagnosis of
6.3.2 Cadaver—A human body, or portion of human body,
humans. Preferred settings are those appropriate for the prod-
with which to perform the tests may be used.
uct and for the particular area of body interest (for example,
6.3.3 Metal, Plastic, or Composite—A metal, plastic, or
leg, heart, and so forth). The radiopacities of the test specimen
composite material of appropriate dimensions may be used.
and user-defined standard are evaluated in terms of the image
For example, a 5.0 mm, 10.0 mm, or 15.0 6 0.15 mm thick
background with or without the use of a body mimic. The
aluminum sheet might be appropriate. The aluminum sheet
radiopacity may be reported qualitatively or quantitatively.
shall be ≥99 % in purity, or type 1100 or purer, in accordance
with Specification B209/B209M.
5. Significance and Use
6.3.4 Phantom—An apparatus that mimics a portion of the
5.1 These methods are intended to determine whether a
body may be used; note that this apparatus may be as complex
material, product, or part of a product has the degree of
as a manufactured torso with appropriate densities representing
radiopacity desired for its application as a medical device in the
all portions of the anatomy within the torso, or may be as
human body. This method allows for comparison with or
simple as a defined thickness of water.
without the use of a body mimic. Comparisons without the use
6.3.5 Step Wedge—A step wedge may be used as a user-
of a body mimic should be used with caution as the relative
defined standard, if it has the requisite thickness steps. 1100
radiopacity can be affected when imaging through the human
aluminum shall be used to construct step wedges. Examples of
body.
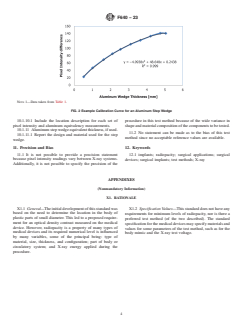
step wedges are shown in Fig. 1. Users may use other step
5.2 These methods allow for both qualitative and quantita- wedge dimensions as long as the step thickness is known.
tive evaluation in different comparative situations.
6.4 Rubber Blankets—Blankets incorporating X-ray absorb-
ers may be used to mask the image area not covered by the
6. Apparatus
body mimic material (this prevents undercutting). Lead sheets
6.1 X-Ray Imaging System. may also be used for masking.
6.2 X-Ray Film or Digital Image Acquisition System—The 6.5 Back-Scatter Protection, as described in Guide E94/
film or digital imaging system shall be appropriate for the E94M, or as appropriate with the specific X-ray imaging
system.
imaging conditions used. A grid may be used.
NOTE 1—The tolerance shall be 60.005 in. Units are in inches [mm]. 1100 aluminum shall be used. Users may use other step wedge dimensions as
long as the dimensions are known.
FIG. 1 Example Dimensions of Aluminum Step Wedges
F640 − 23
7. Test Specimens 9.4.1.4 Step Wedge Equivalency Calculation (if a step
wedge is used)—Plot a calibration curve of the pixel intensity
7.1 Material—The material may be in any form. For com-
difference of the step wedge versus step wedge thickness. See
paring results between materials, best results will be obtained
Table 1 and Fig. 2 for examples of the data and resulting
by utilizing the same form and dimensions for each material.
calibration plot. From the calibration curve and the sample
7.2 Product—The product or specific part or section of the
pixel intensity determined in 9.4.1.2, determine the equivalent
product may be utilized in any desired configuration.
step wedge thickness for each pixel intensity value.
NOTE 1—For plastics, a 2.0-mm thick sheet of material is often molded
especially for testing. For example, see the description of sample in
10. Report
Practice D3182.
10.1 The report shall include the following:
8. Imaging Conditions
10.1.1 All test equipment, including source type, filter type,
8.1 The test shall be performed at appropriate conditions for detector type, machine geometry, machine type and model
the imaging system, the product or material, and the area of the numbers, and film type and resolution (if film is used) or
body within which the product is intended for use. For imaging system resolution (if digital analysis is used).
example, each X-ray image is made at a specified voltage, 10.1.2 All test conditions, including the specific values of
current, time contrast, and brightness that are typical of those kVp and mA·s, the source-to-detector distance, the object-to-
used in the X-ray diagnosis of humans. Preferred settings are detector distance, and, if film is used, the focus-film distance
those appropriate for the product and for the particular area of and film exposure settings.
body interest (for example, leg, heart, and so forth). 10.1.3 If applicable, specification requirements, as listed in
the requirements of the standard specification for the medical
NOTE 2—Typical X-ray exposure conditions for various anatomical
3 d
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F640 − 20 F640 − 23
Standard Test Methods for
Determining Radiopacity for Medical Use
This standard is issued under the fixed designation F640; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 These test methods cover the determination of the radiopacity of materials and products utilizing X-ray based techniques,
including fluoroscopy, angiography, CT (computed tomography), and DEXA (dual energy X-ray absorptiometry), also known as
DXA, The results of these measurements are an indication of the likelihood of locating the product within the human body.
1.2 Radiopacity is determined by (a) qualitatively comparing image(s) of a test specimen and a user-defined standard, with or
without the use of a body mimic; or (b) quantitatively determining the specific difference in optical density or pixel intensity
between the image of a test specimen and the image of a user-defined standard, with or without the use of a body mimic.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
B209B209/B209M Specification for Aluminum and Aluminum-Alloy Sheet and Plate (Metric) B0209_B0209M
D3182 Practice for Rubber—Materials, Equipment, and Procedures for Mixing Standard Compounds and Preparing Standard
Vulcanized Sheets
E94/E94M Guide for Radiographic Examination Using Industrial Radiographic Film
E1316 Terminology for Nondestructive Examinations
F647 Practice for Evaluating and Specifying Implantable Shunt Assemblies for Neurosurgical Application
3. Terminology
3.1 Definitions—For definitions of terms relating to X-ray procedures, refer to Terminology E1316.
3.2 Descriptions of Terms:
These test methods are under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and are the direct responsibility of Subcommittee
F04.15 on Material Test Methods.
Current edition approved Oct. 1, 2020March 1, 2023. Published November 2020March 2023. Originally approved in 1979. Last previous edition approved in 20122020
as F640 – 12.F640 – 20. DOI: 10.1520/F0640-20.10.1520/F0640-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F640 − 23
3.2.1 body mimic, n—a piece of material, a phantom, a cadaver, or an animal utilized to mimic the appropriate X-ray attenuation
through a particular part of the human body.
3.2.2 digital resolution, n—the number of pixels per inch in a digital image.
3.2.2.1 Discussion—
This may be different in the x and y directions.
3.2.3 grayscale range, n—the number of levels in pixel intensity resolved in the digital image.
3.2.3.1 Discussion—
This range is normally 256 levels in an 8-bit grayscale image, but 16-bit grayscale images can also be used.
3.2.4 pixel intensity, n—the grayscale level of a pixel between 0 and 255, as determined by the digital analysis program.
3.2.5 pixel intensity difference, n—the difference in grayscale level between two regions or objects in an image, reported to within
the significance capability of the digital analysis program.
3.2.6 user-defined standard, n—a comparison standard selected by the user. This standard could be a reference material or a
predicate device.
3.2.6.1 Discussion—
This standard may be an existing medical product or a material in a particular form, it may be a commercially available standard,
or it may be one developed by the user.
4. Summary of Test Methods
4.1 The test specimen is placed so it sits at or near the middle of the X-ray image area in the X-ray imaging system. X-ray images
are made at specified voltages, times, and currents that are typical of those used in the X-ray diagnosis of humans. Preferred
settings are those appropriate for the product and for the particular area of body interest (for example, leg, heart, and so forth). The
radiopacities of the test specimen and user-defined standard are evaluated in terms of the image background with or without the
use of a body mimic. The radiopacity may be reported qualitatively or quantitatively.
5. Significance and Use
5.1 These methods are intended to determine whether a material, product, or part of a product has the degree of radiopacity desired
for its application as a medical device in the human body. This method allows for comparison with or without the use of a body
mimic. Comparisons without the use of a body mimic should be used with caution as the relative radiopacity can be affected when
imaging through the human body.
5.2 These methods allow for both qualitative and quantitative evaluation in different comparative situations.
6. Apparatus
6.1 X-Ray Imaging System.
6.2 X-Ray Film or Digital Image Acquisition System—The film or digital imaging system shall be appropriate for the imaging
conditions used. A grid may be used.
6.3 Body Mimic (if used)—See Appendix X2 for discussion of body mimics. (Note that this is not an all-inclusive list and other
body mimics might be appropriate.)
6.3.1 Animal—An appropriate animal, or portion of appropriate animal, with which to perform the tests may be used.
6.3.2 Cadaver—A human body, or portion of human body, with which to perform the tests may be used.
6.3.3 Metal, Plastic, or Composite—A metal, plastic, or composite material of appropriate dimensions may be used. For example,
F640 − 23
a 5.0 mm, 10.0 mm, or 15.0 6 0.15 mm thick aluminum sheet might be appropriate. The aluminum sheet shall be ≥99 % in purity,
or type 1100 or purer, in accordance with Specification B209B209/B209M.
6.3.4 Phantom—An apparatus that mimics a portion of the body may be used; note that this apparatus may be as complex as a
manufactured torso with appropriate densities representing all portions of the anatomy within the torso, or may be as simple as
a defined thickness of water.
6.3.5 Step Wedge—A step wedge may be used as a user-defined standard, if it has the requisite thickness steps. 1100 aluminum
shall be used to construct step wedges. Examples of step wedges are shown in Fig. 1. Users may use other step wedge dimensions
as long as the step thickness is known.
6.4 Rubber Blankets—Blankets incorporating X-ray absorbers may be used to mask the image area not covered by the body mimic
material (this prevents undercutting). Lead sheets may also be used for masking.
6.5 Back-Scatter Protection, as described in Guide E94/E94M, or as appropriate with the specific X-ray imaging system.
7. Test Specimens
7.1 Material—The material may be in any form. For comparing results between materials, best results will be obtained by utilizing
the same form and dimensions for each material.
7.2 Product—The product or specific part or section of the product may be utilized in any desired configuration.
NOTE 1—For plastics, a 2.0-mm thick sheet of material is often molded especially for testing. For example, see the description of sample in Practice
D3182.
8. Imaging Conditions
8.1 The test shall be performed at appropriate conditions for the imaging system, the product or material, and the area of the body
within which the product is intended for use. For example, each X-ray image is made at a specified voltage, current, time contrast,
NOTE 1—The tolerance shall be 60.005 in. Units are in inches [mm]. 1100 aluminum shall be used. Users may use other step wedge dimensions as
long as the dimensions are known.
FIG. 1 Example Dimensions of Aluminum Step Wedges
F640 − 23
and brightness that are typical of those used in the X-ray diagnosis of humans. Preferred settings are those appropriate for the
product and for the particular area of body interest (for example, leg, heart, and so forth).
NOTE 2—Typical X-ray exposure conditions for various anatomical parts are available in the literature (1).
8.2 Imaging conditions shall be described in the test report.
9. Procedure
9.1 Test Specimen Placement—Place the test specimen(s) and the user-defined standard at or near the middle of the X-ray imaging
area. If a body mimic is used, place the test specimen(s) and the user-defined standard as appropriate in, on, or under the body
mimic. As appropriate, the effect of the clinical X-ray table shall also be included with use of an appropriate material of thickness
similar to that used clinically.
9.2 X-Ray Exposure—Complete X-ray exposure using conditions typical of those used in the X-ray diagnosis of humans, the
product, and for the particular area of interest.
9.3 Qualitative Analysis—Visually compare the image(s) of the test specimen and the user-defined standard to the background on
the film or in the digital image (whether original or converted from film).
9.4 Quantitative Analysis:
9.4.1 Measurement of X-ray Density in Aluminum Thickness Equivalency:
9.4.1.1 Background—Measure the background pixel intensity in a region proximal to the test subject. Pixel intensities can be
measured using st
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...