ASTM D7938-21
(Practice)Standard Practice for Sampling of C-14 in Gaseous Effluents
Standard Practice for Sampling of C-14 in Gaseous Effluents
SIGNIFICANCE AND USE
5.1 This practice was developed4 for the purpose of sampling gaseous effluent streams from a facility that releases 14C in either organic or inorganic forms.
5.1.1 For many years 14C was not included in gaseous and liquid effluent measurements used for effluent dose calculations at nuclear power facilities. U.S. NRC Regulatory Guide 1.21 now requires 14C analysis (either estimated by calculation or actual measurement) and its impact on annual dose in the environs of nuclear plants be evaluated. Based on the revisions to the Regulatory Guide and NRC guidance to licensees, 14C activity will need to be reported and evaluated for dose contribution based on the activity concentration and chemical form of the 14C in the release.
5.2 While 14C releases may be estimated, the measurement of actual 14C emissions provides a more reliable and accurate means of reporting emissions. The chemical form of 14C that yields the greatest dose significance due to uptake by living organisms is the inorganic form. Thus the distribution of 14C chemical forms in plant effluents is important in assessing the overall dose impact.
5.3 Use of this sampling practice has identified that for pressurized water reactors (PWRs) >90 % of all 14C released may be in the organic form during operation, and for boiling water reactors (BWRs) 14C released may be in the organic form during operation.
5.3.1 Some power plants have catalytic hydrogen recombiners in the waste gas processing system. These can also oxidize organic carbon to CO2, increasing the percentage of 14CO2 in the effluent release.
5.3.2 During refueling outages, oxidizing conditions exist in the reactor cavity due to air saturation and radiolytic reactions by the nuclear fuel. The combination of these two effects has been shown to increase the 14CO2 content of the sampled atmosphere inside the containment building.
5.4 The sampling methodology described in this practice is not capable of discriminating between different org...
SCOPE
1.1 The intended use of this practice is for sampling of gasses containing 14C in inorganic, organic or particulate forms. This sampling practice captures the 14C in a media that can be submitted to a laboratory for analysis, typically by liquid scintillation counting (LSC)
1.2 This practice does not include the needed steps for the liberation of 14C from the media on which it was adsorbed or those for the preparation for LSC sample preparation in the laboratory prior to liquid scintillation analysis. This practice does not include the methodology used to analyze the prepared samples by LSC.
1.3 The overall 14C analytical detection capability is impacted by a number of factors including the volume sampled, the method used to desorb the 14C from the media, and the analytical method used the measure 14C from the media. This practice only directly addresses the volume of the gas stream from which any present 14C would be adsorbed.
1.4 The values stated in pCi units are to be regarded as standard given the reporting requirements of the U.S. NRC Regulatory Guide 1.21. The Bq values given in parenthesis are mathematical conversions to SI units that are provided for information only and are not considered standard. Other values stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Dec-2021
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.04 - Methods of Radiochemical Analysis
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-May-2020
- Effective Date
- 01-Feb-2018
- Effective Date
- 01-Feb-2016
- Effective Date
- 15-Jan-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Mar-2010
- Effective Date
- 15-Dec-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Feb-2006
- Effective Date
- 01-Mar-2004
- Effective Date
- 01-Mar-2004
- Effective Date
- 10-Aug-2003
- Effective Date
- 10-Mar-2003
Overview
ASTM D7938-21: Standard Practice for Sampling of C-14 in Gaseous Effluents provides essential guidance for collecting samples containing carbon-14 (C-14) in gaseous effluents. This standard is published by ASTM International and is designed for use at facilities, such as nuclear power plants, that may release C-14 in either organic or inorganic forms into the atmosphere. The practice aims to support regulatory compliance-particularly with U.S. NRC Regulatory Guide 1.21-by enabling reliable assessment of the distribution and concentration of C-14, thereby supporting accurate dose calculations for environmental and public health monitoring.
Key Topics
- Sampling Methodology: Defines procedures for collecting C-14 samples in various chemical and physical forms, including inorganic (CO₂), organic, and particulate.
- Effluent Monitoring: Addresses the differentiation and quantification of both inorganic and organic C-14 in plant emissions, which is key for accurate dose and risk assessment.
- Compliance with Regulations: Supports regulatory reporting requirements, such as those set by the U.S. Nuclear Regulatory Commission, which now require the evaluation of C-14 contributions to environmental radiation doses.
- Sample Handling: Outlines handling of collected samples for laboratory analysis, commonly via liquid scintillation counting, ensuring representativeness and quality control.
- Quality Assurance: Specifications for field blanks, duplicates, and use of reference materials to validate the reliability and precision of the sampling and analytical process.
- Interference Mitigation: Addresses potential interferences from radionuclides such as radon progeny and tritium, and prescribes steps to minimize such effects.
Applications
ASTM D7938-21 is highly relevant for:
- Nuclear Power Plants and Facilities: Enables the monitoring of C-14 emissions in compliance with regulatory obligations and supports environmental stewardship by verifying the form and amount of radioactive carbon released.
- Environmental Monitoring Programs: Used in radiological environmental monitoring (REMP) to evaluate the impact of plant emissions on the public and environment, helping inform dose assessments and mitigation strategies.
- Laboratory Analysis Preparation: Establishes a consistent method for sample collection suitable for transfer to laboratories performing detailed radiochemical analysis.
- Process Optimization in Waste Management: Insight into the chemical forms of C-14 aids in optimizing plant systems, such as catalytic hydrogen recombiners or gas processing systems, to reduce the radiological dose impact.
- Refueling Outage Monitoring: Provides practices for accurate sampling during refueling outages, when the chemical form distribution of C-14 can change due to altered plant conditions.
Related Standards
- ASTM D1129: Terminology Relating to Water, for supporting definitions.
- ASTM D7282: Practice for Set-up, Calibration, and Quality Control of Instruments Used for Radioactivity Measurements, relevant for analytical assurance.
- ASTM D7902: Terminology for Radiochemical Analyses, providing additional reference descriptions.
- U.S. NRC Regulatory Guide 1.21: Key regulatory basis for measuring, evaluating, and reporting radioactive material in liquid and gaseous effluents.
By following ASTM D7938-21, organizations can ensure accurate and defensible sampling of C-14 in gaseous effluents, meeting regulatory demands while protecting human health and the environment. The standard's focus on differentiation of chemical forms, quality control, and robust methodology makes it a cornerstone for effective radioactive effluent management.
Keywords: C-14, carbon-14, gaseous effluent, ASTM D7938, radioactive emissions, nuclear power, environmental monitoring, regulatory compliance, radiochemistry, liquid scintillation counting
Buy Documents
ASTM D7938-21 - Standard Practice for Sampling of C-14 in Gaseous Effluents
REDLINE ASTM D7938-21 - Standard Practice for Sampling of C-14 in Gaseous Effluents
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D7938-21 is a standard published by ASTM International. Its full title is "Standard Practice for Sampling of C-14 in Gaseous Effluents". This standard covers: SIGNIFICANCE AND USE 5.1 This practice was developed4 for the purpose of sampling gaseous effluent streams from a facility that releases 14C in either organic or inorganic forms. 5.1.1 For many years 14C was not included in gaseous and liquid effluent measurements used for effluent dose calculations at nuclear power facilities. U.S. NRC Regulatory Guide 1.21 now requires 14C analysis (either estimated by calculation or actual measurement) and its impact on annual dose in the environs of nuclear plants be evaluated. Based on the revisions to the Regulatory Guide and NRC guidance to licensees, 14C activity will need to be reported and evaluated for dose contribution based on the activity concentration and chemical form of the 14C in the release. 5.2 While 14C releases may be estimated, the measurement of actual 14C emissions provides a more reliable and accurate means of reporting emissions. The chemical form of 14C that yields the greatest dose significance due to uptake by living organisms is the inorganic form. Thus the distribution of 14C chemical forms in plant effluents is important in assessing the overall dose impact. 5.3 Use of this sampling practice has identified that for pressurized water reactors (PWRs) >90 % of all 14C released may be in the organic form during operation, and for boiling water reactors (BWRs) 14C released may be in the organic form during operation. 5.3.1 Some power plants have catalytic hydrogen recombiners in the waste gas processing system. These can also oxidize organic carbon to CO2, increasing the percentage of 14CO2 in the effluent release. 5.3.2 During refueling outages, oxidizing conditions exist in the reactor cavity due to air saturation and radiolytic reactions by the nuclear fuel. The combination of these two effects has been shown to increase the 14CO2 content of the sampled atmosphere inside the containment building. 5.4 The sampling methodology described in this practice is not capable of discriminating between different org... SCOPE 1.1 The intended use of this practice is for sampling of gasses containing 14C in inorganic, organic or particulate forms. This sampling practice captures the 14C in a media that can be submitted to a laboratory for analysis, typically by liquid scintillation counting (LSC) 1.2 This practice does not include the needed steps for the liberation of 14C from the media on which it was adsorbed or those for the preparation for LSC sample preparation in the laboratory prior to liquid scintillation analysis. This practice does not include the methodology used to analyze the prepared samples by LSC. 1.3 The overall 14C analytical detection capability is impacted by a number of factors including the volume sampled, the method used to desorb the 14C from the media, and the analytical method used the measure 14C from the media. This practice only directly addresses the volume of the gas stream from which any present 14C would be adsorbed. 1.4 The values stated in pCi units are to be regarded as standard given the reporting requirements of the U.S. NRC Regulatory Guide 1.21. The Bq values given in parenthesis are mathematical conversions to SI units that are provided for information only and are not considered standard. Other values stated in SI units are to be regarded as standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This practice was developed4 for the purpose of sampling gaseous effluent streams from a facility that releases 14C in either organic or inorganic forms. 5.1.1 For many years 14C was not included in gaseous and liquid effluent measurements used for effluent dose calculations at nuclear power facilities. U.S. NRC Regulatory Guide 1.21 now requires 14C analysis (either estimated by calculation or actual measurement) and its impact on annual dose in the environs of nuclear plants be evaluated. Based on the revisions to the Regulatory Guide and NRC guidance to licensees, 14C activity will need to be reported and evaluated for dose contribution based on the activity concentration and chemical form of the 14C in the release. 5.2 While 14C releases may be estimated, the measurement of actual 14C emissions provides a more reliable and accurate means of reporting emissions. The chemical form of 14C that yields the greatest dose significance due to uptake by living organisms is the inorganic form. Thus the distribution of 14C chemical forms in plant effluents is important in assessing the overall dose impact. 5.3 Use of this sampling practice has identified that for pressurized water reactors (PWRs) >90 % of all 14C released may be in the organic form during operation, and for boiling water reactors (BWRs) 14C released may be in the organic form during operation. 5.3.1 Some power plants have catalytic hydrogen recombiners in the waste gas processing system. These can also oxidize organic carbon to CO2, increasing the percentage of 14CO2 in the effluent release. 5.3.2 During refueling outages, oxidizing conditions exist in the reactor cavity due to air saturation and radiolytic reactions by the nuclear fuel. The combination of these two effects has been shown to increase the 14CO2 content of the sampled atmosphere inside the containment building. 5.4 The sampling methodology described in this practice is not capable of discriminating between different org... SCOPE 1.1 The intended use of this practice is for sampling of gasses containing 14C in inorganic, organic or particulate forms. This sampling practice captures the 14C in a media that can be submitted to a laboratory for analysis, typically by liquid scintillation counting (LSC) 1.2 This practice does not include the needed steps for the liberation of 14C from the media on which it was adsorbed or those for the preparation for LSC sample preparation in the laboratory prior to liquid scintillation analysis. This practice does not include the methodology used to analyze the prepared samples by LSC. 1.3 The overall 14C analytical detection capability is impacted by a number of factors including the volume sampled, the method used to desorb the 14C from the media, and the analytical method used the measure 14C from the media. This practice only directly addresses the volume of the gas stream from which any present 14C would be adsorbed. 1.4 The values stated in pCi units are to be regarded as standard given the reporting requirements of the U.S. NRC Regulatory Guide 1.21. The Bq values given in parenthesis are mathematical conversions to SI units that are provided for information only and are not considered standard. Other values stated in SI units are to be regarded as standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D7938-21 is classified under the following ICS (International Classification for Standards) categories: 13.040.40 - Stationary source emissions. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7938-21 has the following relationships with other standards: It is inter standard links to ASTM D7902-20, ASTM D1129-13(2020)e2, ASTM D7902-18, ASTM D7902-16, ASTM D7902-14e1, ASTM D7902-14, ASTM D1129-10, ASTM D7282-06, ASTM D1129-06ae1, ASTM D1129-06a, ASTM D1129-06, ASTM D1129-04e1, ASTM D1129-04, ASTM D1129-03a, ASTM D1129-03. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7938-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7938 − 21
Standard Practice for
Sampling of C-14 in Gaseous Effluents
This standard is issued under the fixed designation D7938; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 The intended use of this practice is for sampling of 2.1 ASTM Standards:
D1129 Terminology Relating to Water
gasses containing C in inorganic, organic or particulate
D7282 Practice for Set-up, Calibration, and Quality Control
forms. This sampling practice captures the C in a media that
of Instruments Used for Radioactivity Measurements
can be submitted to a laboratory for analysis, typically by
D7902 Terminology for Radiochemical Analyses
liquid scintillation counting (LSC)
2.2 U.S. NRC Publications:
1.2 This practice does not include the needed steps for the
U.S. NRC Regulatory Guide 1.21 “Measuring, Evaluating,
liberation of C from the media on which it was adsorbed or
and Reporting Radioactive Material in Liquid and Gas-
those for the preparation for LSC sample preparation in the
eous Effluents and Solid Waste,” revision 2, June 2009
laboratory prior to liquid scintillation analysis. This practice
does not include the methodology used to analyze the prepared
3. Terminology
samples by LSC.
3.1 Definitions:
1.3 The overall C analytical detection capability is im- 3.1.1 For definitions of terms used in this standard, refer to
pacted by a number of factors including the volume sampled, Terminologies D1129 and D7902.
the method used to desorb the C from the media, and the 3.2 Definitions of Terms Specific to This Standard:
14 14 14
analytical method used the measure C from the media. This 3.2.1 organic C, n—any gaseous, chemical C form
practice only directly addresses the volume of the gas stream (including CO) that is not particulate and not CO .
from which any present C would be adsorbed. 3.2.1.1 Discussion—Although no specific organic form is
determined, the major contributors are likely to be CH,C H ,
4 2 6
1.4 The values stated in pCi units are to be regarded as
C H , CO, and C H .
2 4 2 2
standard given the reporting requirements of the U.S. NRC
14 14
3.2.2 inorganic C, n—the gaseous, chemical form of C
Regulatory Guide 1.21. The Bq values given in parenthesis are
as CO .
mathematical conversions to SI units that are provided for 2
3.2.2.1 Discussion—These chemical form categorizations
information only and are not considered standard. Other values
are based on U.S. NRC Regulatory Guide 1.21.
stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the
4. Summary of Practice
safety concerns, if any, associated with its use. It is the
4.1 A sample of a flowing gaseous stream is extracted at a
responsibility of the user of this standard to establish appro-
flowrateof30to3000mL/min.Thesampleisfilteredandsplit
priate safety, health, and environmental practices and deter-
into two parallel flow paths. One flow path is passed through a
mine the applicability of regulatory limitations prior to use. 14
furnace to convert all carbon to CO .This will yield a total C
1.6 This international standard was developed in accor-
content of the sample. The other flow path collects only the
dance with internationally recognized principles on standard-
CO fraction of the gaseous stream. This yields the inorganic
ization established in the Decision on Principles for the 14
C content of the gaseous stream. The calculated difference
Development of International Standards, Guides and Recom-
between the measured total and inorganic carbon content is the
mendations issued by the World Trade Organization Technical
organic content (U.S. NRC Regulatory Guide 1.21). The
Barriers to Trade (TBT) Committee. 14
concentration of C in the particulate matter may also be
determined.
1 2
This practice is under the jurisdiction of ASTM Committee D19 on Water and For referenced ASTM standards, visit the ASTM website, www.astm.org, or
is the direct responsibility of Subcommittee D19.04 on Methods of Radiochemical contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Analysis. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Dec. 15, 2021. Published February 2022. Originally the ASTM website.
approved in 2015. Last previous edition approved in 2015 as D7938 – 15. DOI: Available from U.S. Nuclear Regulatory Commission, Washington, DC 20555-
10.1520/D7938-21. 0001, http://www.nrc.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7938 − 21
5. Significance and Use sample volume ensuring that the adsorbent does not become
4 saturated with stable CO , allowing bypass of the CO .
2 2
5.1 This practice was developed for the purpose of sam-
14 14
pling gaseous effluent streams from a facility that releases C 6.3 Tritium can interfere with the analysis of C by LSC.
in either organic or inorganic forms. Tritium (when present as part of a water molecule) is effec-
5.1.1 For many years C was not included in gaseous and tively removed during the sampling collection process using a
liquideffluentmeasurementsusedforeffluentdosecalculations desiccant upstream of the carbon dioxide trap.
at nuclear power facilities. U.S. NRC Regulatory Guide 1.21 6.3.1 Tritium removal can also be accomplished during the
now requires C analysis (either estimated by calculation or sample preparation step in the laboratory by using a water trap
actual measurement) and its impact on annual dose in the with HCl prior to the final carbon dioxide trap (see Fig. X1.1).
environs of nuclear plants be evaluated. Based on the revisions
6.4 High levels of moisture in the gaseous effluent would
to the Regulatory Guide and NRC guidance to licensees, C
limit the sample volume raising the detection limit. A method
activity will need to be reported and evaluated for dose
of monitoring the level of depletion of the desiccant material
contribution based on the activity concentration and chemical
should be employed in this method.
form of the C in the release.
7. Apparatus
5.2 While C releases may be estimated, the measurement
of actual C emissions provides a more reliable and accurate
7.1 Combustion Furnace—Capable of attaining at least
means of reporting emissions. The chemical form of C that
600°C with the air flow at a maximum of 3000 mL/min.
yields the greatest dose significance due to uptake by living
7.1.1 The performance of the converter was determined by
organisms is the inorganic form. Thus the distribution of C
independent laboratory tests by measuring the conversion
chemical forms in plant effluents is important in assessing the
efficiency of methane to CO . The conversion efficiency was
overall dose impact.
found to be >95 % over a sample flow rate of 300 to 3000 mL
per minute. No reduction in conversion efficiency was ob-
5.3 Use of this sampling practice has identified that for
served as a function of methane concentration or flow rate.
pressurized water reactors (PWRs) >90 % of all C released
may be in the organic form during operation, and for boiling
7.2 Air Sampling Pump—Capacity in the range of 30 to
water reactors (BWRs) <30 % of all C released may be in the
3000 mL/min.
organic form during operation.
7.3 Two Flow Monitors—Calibrated for air flow in the
5.3.1 Somepowerplantshavecatalytichydrogenrecombin-
desired flow range.
ers in the waste gas processing system. These can also oxidize
7.4 Particulate Filter Holder—Normally sized for a 47–mm
organic carbon to CO , increasing the percentage of CO in
2 2
diameter filter but may be sized to fit the sample flow.
the effluent release.
5.3.2 Duringrefuelingoutages,oxidizingconditionsexistin
7.5 Desiccant Tube—Sized for the amount of desiccant
the reactor cavity due to air saturation and radiolytic reactions
needed for the volume of air to be sampled.
by the nuclear fuel. The combination of these two effects has
8. Reagents and Materials
been shown to increase the CO content of the sampled
atmosphere inside the containment building.
8.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
5.4 The sampling methodology described in this practice is
not capable of discriminating between different organic forms all reagents shall conform to the specifications of the Commit-
tee onAnalytical Reagents of theAmerican Chemical Society,
of C.
where such specifications are available. Other grades may be
used, provided it is first ascertained that the reagent is of
6. Interferences
sufficiently high purity to permit its use without increasing the
6.1 Based on the chemical methodology used in this sam-
background of the measurement.
pling practice, interference from non-volatile radionuclides is
8.1.1 Somechemicals,evenofhighpurity,containnaturally
eliminated. However, the radon progeny of the uranium
222 220 219 occurring radioactive elements, for example, uranium,
( Rn), thorium ( Rn), and actinium ( Rn) series may be
actinium,thorium,rareearthsandpotassiumcompounds.Also,
possible interferences. Their contribution to the liquid scintil-
some chemical reagents, including organic compounds, have
lation activity of the samples should be assessed by an
been found to be contaminated with artificially produced
independent radiological method (for example, gamma-ray
radionuclides. Consequently, when carrier chemicals are used
spectrometry, or monitoring the liquid scintillation region
in the analysis of low-radioactivity samples, the radioactivity
above the C window).
of the carriers shall be determined under identical analytical
6.2 Gaseous flow paths with carbon dioxide concentrations
conditions as used for the sample. The radioactivity of the
greater than those found in routine atmospheric samples may
raise the detection limit for C. This would require a lower
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
4 14
Holtzclaw, J., “Sample and Analysis Protocol for C in Gaseous Effluents,” Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
Radioactive Effluent Technical Specifications (RETS) and Radiological Environ- U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
mental Monitoring Programs (REMP) Workshop, San Jose, CA, June 28–30, 2010. copeial Convention, Inc. (USPC), Rockville, MD.
D7938 − 21
reagentsmaybeconsideredasbackgroundandsubtractedfrom Particulate C captured on the filtration medium is analyzed
the test sample counting rate. This increased background separately. If the measurement quality objectives require only
reduces the sensitivity of the measurement. total C analysis, it is still recommended that the sample be
6 filtered. The particulate filter should be treated separately to
8.2 Ascarite II, commercially available solid material for
ensure the conversion to carbon dioxide will be complete prior
adsorption of CO .
to radiochemical analysis.
8.2.1 The use of another material which will trap carbon
dioxide will be acceptable. However, validation of the ability 10.5 The preceding steps in this section of this practice
of the medium to retain carbon dioxide
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7938 − 15 D7938 − 21
Standard Practice for
Sampling of C-14 in Gaseous Effluents
This standard is issued under the fixed designation D7938; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 The intended use of this practice is for sampling of gasses containing C in inorganic, organic or particulate forms. This
sampling practice captures the C in a media that can be submitted to a laboratory for analysis, typically by liquid scintillation
counting (LSC)
1.2 This practice does not include the needed steps for the liberation of C from the media on which it was adsorbed or those
for the preparation for LSC sample preparation in the laboratory prior to liquid scintillation analysis. This practice does not include
the methodology used to analyze the prepared samples by LSC.
1.3 The overall C analytical detection capability is impacted by a number of factors including the volume sampled, the method
14 14
used to desorb the C from the media, and the analytical method used the measure C from the media. This practice only directly
addresses the volume of the gas stream from which any present C would be adsorbed.
1.4 The values stated in pCi units are to be regarded as standard given the reporting requirements of the U.S. NRC Regulatory
Guide 1.21. The Bq values given in parenthesis are mathematical conversions to SI units that are provided for information only
and are not considered standard. Other values stated in SI units are to be regarded as standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D7282 Practice for Set-up, Calibration, and Quality Control of Instruments Used for Radioactivity Measurements
D7902 Terminology for Radiochemical Analyses
2.2 U.S. NRC Publications:
U.S. NRC Regulatory Guide 1.21 “Measuring, Evaluating, and Reporting Radioactive Material in Liquid and Gaseous Effluents
and Solid Waste,” revision 2, June 2009
This practice is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.04 on Methods of Radiochemical Analysis.
Current edition approved Dec. 15, 2015Dec. 15, 2021. Published February 2016February 2022. Originally approved in 2015. Last previous edition approved in 2015 as
D7938 – 15. DOI: 10.1520/D7938-15.10.1520/D7938-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from U.S. Nuclear Regulatory Commission, Washington, DC 20555-0001, http://www.nrc.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7938 − 21
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminologies D1129 and D7902.
3.2 Definitions of Terms Specific to This Standard:
14 14
3.2.1 organic C, n—any gaseous, chemical C form (including CO) that is not particulate and not CO .
3.2.1.1 Discussion—
Although no specific organic form is determined, the major contributors are likely to be CH , C H , C H , CO, and C H .
4 2 6 2 4 2 2
14 14
3.2.2 inorganic C, n—the gaseous, chemical form of C as CO .
3.2.2.1 Discussion—
These chemical form categorizations are based on U.S. NRC Regulatory Guide 1.21.
4. Summary of Practice
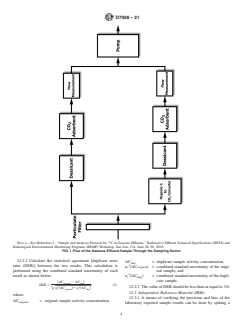
4.1 A sample of a flowing gaseous stream is extracted at a flow rate of 30 to 3000 mL/min. The sample is filtered,filtered and split
into two parallel flow paths. One flow path is passed through a furnace to convert all carbon to CO . This will yield a total C
content of the sample. The other flow path collects only the CO fraction of the gaseous stream. This yields the inorganic C
content of the gaseous stream. The calculated difference between the measured total and inorganic carbon content is the organic
content (U.S. NRC Regulatory Guide 1.21). The concentration of C in the particulate matter may also be determined.
5. Significance and Use
4 14
5.1 This practice was developed for the purpose of sampling gaseous effluent streams from a facility that releases C in either
organic or inorganic forms.
5.1.1 For many years C was not included in gaseous and liquid effluent measurements used for effluent dose calculations at
nuclear power facilities. U.S. NRC Regulatory Guide 1.21 now requires C analysis (either estimated by calculation or actual
measurement) and its impact on annual dose in the environs of nuclear plants be evaluated. Based on the revisions to the
Regulatory Guide and NRC guidance to licensees, C activity will need to be reported and evaluated for dose contribution based
on the activity concentration and chemical form of the C in the release.
14 14
5.2 While C releases may be estimated, the measurement of actual C emissions provides a more reliable and accurate means
of reporting emissions. The chemical form of C that yields the greatest dose significance due to uptake by living organisms is
the inorganic form. Thus the distribution of C chemical forms in plant effluents is important in assessing the overall dose impact.
5.3 Use of this sampling practice has identified that for pressurized water reactors (PWRs) >90 % of all C released may be in
the organic form during operation, and for boiling water reactors (BWRs) <30 % of all C released may be in the organic form
during operation.
5.3.1 Some power plants have catalytic hydrogen recombiners in the waste gas processing system. These can also oxidize organic
carbon to CO , increasing the percentage of CO in the effluent release.
2 2
5.3.2 During refueling outages, oxidizing conditions exist in the reactor cavity due to air saturation and radiolytic reactions by the
nuclear fuel. The combination of these two effects has been shown to increase the CO content of the sampled atmosphere inside
the containment building.
5.4 The sampling methodology described in this practice is not capable of discriminating between different organic forms of C.
6. Interferences
6.1 Based on the chemical methodology used in this sampling practice, interference from non-volatile radionuclides is eliminated.
Holtzclaw, J., “Sample and Analysis Protocol for C in Gaseous Effluents,” Radioactive Effluent Technical Specifications (RETS) and Radiological Environmental
Monitoring Programs (REMP) Workshop, San Jose, CA, June 28–30, 2010.
D7938 − 21
222 220 219
However, the radon progeny of the uranium ( Rn), thorium ( Rn), and actinium ( Rn) series may be possible interferences.
Their contribution to the liquid scintillation activity of the samples should be assessed by an independent radiological method (for
example, gamma ray gamma-ray spectrometry, or monitoring the liquid scintillation region above the C window).
6.2 Gaseous flow paths with carbon dioxide concentrations greater than those found in routine atmospheric samples may raise the
detection limit for C. This would require a lower sample volume ensuring that the adsorbent does not become saturated with
stable CO , allowing bypass of the CO .
2 2
6.3 Tritium can interfere with the analysis of C by LSC. Tritium (when present as part of a water molecule) is effectively
removed during the sampling collection process using a desiccant upstream of the carbon dioxide trap.
6.3.1 Tritium removal can also be accomplished during the sample preparation step in the laboratory by using a water trap with
HCl prior to the final carbon dioxide trap (see Fig. X1.1).
6.4 High levels of moisture in the gaseous effluent would limit the sample volume raising the detection limit. A method of
monitoring the level of depletion of the desiccant material should be employed in this method.
7. Apparatus
7.1 Combustion Furnace—Capable of attaining at least 600°C with the air flow at a maximum of 3000 mL/min.
7.1.1 The performance of the converter was determined by independent laboratory tests by measuring the conversion efficiency
of methane to CO . The conversion efficiency was found to be >95 % over a sample flow rate of 300 to 3000 mL per minute. No
reduction in conversion efficiency was observed as a function of methane concentration or flow rate.
7.2 Air Sampling Pump—Capacity in the range of 30 to 3,0003000 mL/min.
7.3 Two Flow Monitors—Calibrated for air flow in the desired flow range.
7.4 Particulate Filter Holder—Normally sized for a 47–mm diameter filter but may be sized to fit the sample flow.
7.5 Desiccant Tube—Sized for the amount of desiccant needed for the volume of air to be sampled.
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without increasing the background of the measurement.
8.1.1 Some chemicals, even of high purity, contain naturally occurring radioactive elements, for example, uranium, actinium,
thorium, rare earths and potassium compounds. Also, some chemical reagents, including organic compounds, have been found to
be contaminated with artificially produced radionuclides. Consequently, when carrier chemicals are used in the analysis of
low-radioactivity samples, the radioactivity of the carriers shall be determined under identical analytical conditions as used for the
sample. The radioactivity of the reagents may be considered as background and subtracted from the test sample counting rate. This
increased background reduces the sensitivity of the measurement.
8.2 Ascarite II, commercially available solid material for adsorption of CO .
Reagent Chemicals, American Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference Materials,
American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see ‘AnalaR’Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. PharmaceuticalPharmacopeial Convention, Inc.
(USPC), Rockville, MD.
The sole source of supply of the apparatus known to the committee at this time is Arthur H. Thomas Company in Swedesboro, NJ, 08085. If you are aware of alternative
suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical
committee, which you may attend.
D7938 − 21
8.2.1 The use of another material which will trap carbon dioxide will be acceptable. However, validation of the mediums’ ability
ability of the medium to retain carbon dioxide should be verified.
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...