ASTM F702-18
(Specification)Standard Specification for Polysulfone Resin for Medical Applications
Standard Specification for Polysulfone Resin for Medical Applications
ABSTRACT
This specification covers polysulfone resin (poly(oxy-p-phenylenesulfonyl-p-phenyleneoxy-p-phenyleneisopropylidene-p-phenylene)) for medical applications. Requirements and associated test methods for a form of this thermoplastic intended for use in manufacturing medical devices or components of medical devices are provided. The use of this resin in medical devices should be restricted to nonimplant applications until biocompatibility evaluations appropriate for the intended applications are successfully completed. The molecular weight of the resin shall be determined by osmotic pressure in monochlorobenzene. The polysulfone resin shall yield an infrared transmittance spectrum that exhibits major transmittance bands only at the same wavelengths as that of a reference spectrum. Medical devices made of polysulfone may be repeatedly sterilized through steam, ethylene oxide, irradiation, and dry heat sterilization, among others. The polysulfone resin shall be tested for nonvolatile content and melt flow, and shall conform to the specified electrical, physical and mechanical, and thermal properties.
SCOPE
1.1 This specification covers polysulfone resin (poly(oxy-1,4-phenylenesulfonyl-1,4–phenylene (dimethylmethylene)-1,4–phenylene)) as defined in ISO 25137–1, supplied by a vendor in virgin form (pellets, powder, fabricated forms and so forth) for medical applications. This specification provides requirements and associated test methods for this thermoplastic when it is intended for use in manufacturing medical devices or components of medical devices.
1.2 As with any material, some characteristics may be altered by the processing techniques (such as molding, extrusion, machining, sterilization, and so forth) required for the production of a specific part or device. Therefore, properties of fabricated forms of this resin should be evaluated using test methods which are appropriate to ensure safety and efficacy as agreed upon by the vendor, purchaser, and regulating bodies.
1.3 The standard allows for designation of polysulfone resin for all medical applications. The actual extent of performance and suitability for a specific application must be evaluated by the vendor, purchaser, and regulating bodies.
1.4 The properties included in this specification are those applicable for unfilled polysulfone (PSU) polymers with the addition of colorants and processing aids. Indicated properties are for injection molded forms. Forms containing fillers or other additives, as well as polymer blends which contain PSU, or reclaimed materials, are not covered by this specification.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 When evaluating material in accordance with this specification, hazardous materials, operations, and equipment may be involved. This standard does not purport to address all of the concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.11 - Polymeric Materials
Relations
- Effective Date
- 01-Apr-2018
- Effective Date
- 15-Aug-2017
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Apr-2012
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 15-May-2010
- Effective Date
- 01-May-2010
- Effective Date
- 01-Sep-2009
- Effective Date
- 01-Nov-2008
- Effective Date
- 01-Aug-2008
- Effective Date
- 15-Jun-2008
- Effective Date
- 01-Sep-2007
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Dec-2006
Overview
ASTM F702-18: Standard Specification for Polysulfone Resin for Medical Applications defines the requirements, test methods, and criteria for virgin, unfilled polysulfone (PSU) resin used in medical devices and components. This specification is vital for manufacturers, suppliers, and regulators, helping to ensure the quality, safety, and suitability of this high-performance thermoplastic in medical applications. The standard covers properties, processing considerations, biocompatibility assessments, and guidelines for the use of polysulfone resin, with special attention to its application in both implant and non-implant medical devices.
Key Topics
- Material Scope: Covers poly(oxy-p-phenylenesulfonyl-p-phenyleneoxy-p-phenyleneisopropylidene-p-phenylene) supplied in forms like pellets, powders, and fabricated shapes used in medical manufacturing.
- Test Methods and Properties: Specifies standard methods for evaluating:
- Mechanical (tensile strength, modulus, elongation)
- Thermal (deflection temperature under load)
- Chemical (infrared spectrum, melt flow)
- Electrical and physical characteristics
- Processing Guidelines: Accounts for material property changes due to molding, extrusion, machining, and sterilization.
- Contamination Control: Sets inspection protocols for foreign particle contamination, crucial for transparent PSU formulations.
- Certification and Statistical Control: Emphasizes the importance of process validation, lot traceability, and property certification, especially for critical medical uses.
- Biocompatibility: Recommends assessment of finished products per ASTM F748 or the ISO 10993 series, particularly after complete processing and sterilization.
Applications
Polysulfone resins as covered by ASTM F702-18 are widely used across the medical device sector due to their combination of chemical resistance, mechanical strength, and sterilization compatibility. Practical applications include:
- Non-Implant Medical Devices
- Handles and housings for surgical instruments
- Components exposed to aggressive cleaning or repeated sterilization
- Transparent parts requiring toughness and clarity, such as diagnostic equipment covers
- Implantable Devices
- With additional testing and compliance, limited to non load-bearing applications and subject to regulatory approval and biocompatibility evaluation
- Examples include battery housings for implantable electronics and ports for infusion devices (suitability to be determined case by case)
Note: Polysulfone is not generally recommended for long-term or load-bearing implant applications, especially where contact with lipid-containing biological fluids may lead to stress cracking.
- Multiple Sterilization Methods: Polysulfone resins accommodate repeated cycles of steam, ethylene oxide, irradiation, hydrogen peroxide gas plasma, and dry heat without significant degradation.
- Safe Medical Manufacturing: Consistent with Good Manufacturing Practices (GMP) and recommended for ISO 17025-accredited testing environments.
Related Standards
The specification references several key standards, ensuring consistent material performance and regulatory acceptance:
- ASTM D638: Tensile Properties of Plastics
- ASTM D256: Izod Pendulum Impact Resistance
- ASTM D648: Heat Deflection Temperature
- ASTM D792: Density and Specific Gravity
- ASTM D6394: Specification for Sulfone Plastics (SP)
- ASTM D7474: Residual Stresses in Sulfone Plastics
- ASTM F748: Biological Test Methods for Materials and Devices
- ISO 10993-1: Biological Evaluation of Medical Devices
- ISO 17025: Competence of Testing and Calibration Laboratories
- ISO 25137-1: Designation System for Sulfone Polymer Materials
Summary of Practical Value
Adhering to ASTM F702-18 ensures that medical devices made from polysulfone resin meet recognized standards for safety, consistency, and quality. The standard is widely regarded in the medical plastics industry for guiding material selection, validation, and processing. This facilitates efficient regulatory approval, optimizes device performance, and upholds patient safety across diverse medical applications.
Keywords: ASTM F702-18, polysulfone resin, PSU, medical device materials, biocompatibility, sterilizable plastics, medical grade polymers, ASTM medical standards, ISO 10993, sulfone plastics.
Buy Documents
ASTM F702-18 - Standard Specification for Polysulfone Resin for Medical Applications
REDLINE ASTM F702-18 - Standard Specification for Polysulfone Resin for Medical Applications
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F702-18 is a technical specification published by ASTM International. Its full title is "Standard Specification for Polysulfone Resin for Medical Applications". This standard covers: ABSTRACT This specification covers polysulfone resin (poly(oxy-p-phenylenesulfonyl-p-phenyleneoxy-p-phenyleneisopropylidene-p-phenylene)) for medical applications. Requirements and associated test methods for a form of this thermoplastic intended for use in manufacturing medical devices or components of medical devices are provided. The use of this resin in medical devices should be restricted to nonimplant applications until biocompatibility evaluations appropriate for the intended applications are successfully completed. The molecular weight of the resin shall be determined by osmotic pressure in monochlorobenzene. The polysulfone resin shall yield an infrared transmittance spectrum that exhibits major transmittance bands only at the same wavelengths as that of a reference spectrum. Medical devices made of polysulfone may be repeatedly sterilized through steam, ethylene oxide, irradiation, and dry heat sterilization, among others. The polysulfone resin shall be tested for nonvolatile content and melt flow, and shall conform to the specified electrical, physical and mechanical, and thermal properties. SCOPE 1.1 This specification covers polysulfone resin (poly(oxy-1,4-phenylenesulfonyl-1,4–phenylene (dimethylmethylene)-1,4–phenylene)) as defined in ISO 25137–1, supplied by a vendor in virgin form (pellets, powder, fabricated forms and so forth) for medical applications. This specification provides requirements and associated test methods for this thermoplastic when it is intended for use in manufacturing medical devices or components of medical devices. 1.2 As with any material, some characteristics may be altered by the processing techniques (such as molding, extrusion, machining, sterilization, and so forth) required for the production of a specific part or device. Therefore, properties of fabricated forms of this resin should be evaluated using test methods which are appropriate to ensure safety and efficacy as agreed upon by the vendor, purchaser, and regulating bodies. 1.3 The standard allows for designation of polysulfone resin for all medical applications. The actual extent of performance and suitability for a specific application must be evaluated by the vendor, purchaser, and regulating bodies. 1.4 The properties included in this specification are those applicable for unfilled polysulfone (PSU) polymers with the addition of colorants and processing aids. Indicated properties are for injection molded forms. Forms containing fillers or other additives, as well as polymer blends which contain PSU, or reclaimed materials, are not covered by this specification. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 When evaluating material in accordance with this specification, hazardous materials, operations, and equipment may be involved. This standard does not purport to address all of the concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This specification covers polysulfone resin (poly(oxy-p-phenylenesulfonyl-p-phenyleneoxy-p-phenyleneisopropylidene-p-phenylene)) for medical applications. Requirements and associated test methods for a form of this thermoplastic intended for use in manufacturing medical devices or components of medical devices are provided. The use of this resin in medical devices should be restricted to nonimplant applications until biocompatibility evaluations appropriate for the intended applications are successfully completed. The molecular weight of the resin shall be determined by osmotic pressure in monochlorobenzene. The polysulfone resin shall yield an infrared transmittance spectrum that exhibits major transmittance bands only at the same wavelengths as that of a reference spectrum. Medical devices made of polysulfone may be repeatedly sterilized through steam, ethylene oxide, irradiation, and dry heat sterilization, among others. The polysulfone resin shall be tested for nonvolatile content and melt flow, and shall conform to the specified electrical, physical and mechanical, and thermal properties. SCOPE 1.1 This specification covers polysulfone resin (poly(oxy-1,4-phenylenesulfonyl-1,4–phenylene (dimethylmethylene)-1,4–phenylene)) as defined in ISO 25137–1, supplied by a vendor in virgin form (pellets, powder, fabricated forms and so forth) for medical applications. This specification provides requirements and associated test methods for this thermoplastic when it is intended for use in manufacturing medical devices or components of medical devices. 1.2 As with any material, some characteristics may be altered by the processing techniques (such as molding, extrusion, machining, sterilization, and so forth) required for the production of a specific part or device. Therefore, properties of fabricated forms of this resin should be evaluated using test methods which are appropriate to ensure safety and efficacy as agreed upon by the vendor, purchaser, and regulating bodies. 1.3 The standard allows for designation of polysulfone resin for all medical applications. The actual extent of performance and suitability for a specific application must be evaluated by the vendor, purchaser, and regulating bodies. 1.4 The properties included in this specification are those applicable for unfilled polysulfone (PSU) polymers with the addition of colorants and processing aids. Indicated properties are for injection molded forms. Forms containing fillers or other additives, as well as polymer blends which contain PSU, or reclaimed materials, are not covered by this specification. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 When evaluating material in accordance with this specification, hazardous materials, operations, and equipment may be involved. This standard does not purport to address all of the concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F702-18 is classified under the following ICS (International Classification for Standards) categories: 11.100.99 - Other standards related to laboratory medicine; 83.080.20 - Thermoplastic materials. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F702-18 has the following relationships with other standards: It is inter standard links to ASTM D648-18, ASTM D7474-17, ASTM F748-16, ASTM D7474-12, ASTM D6394-11, ASTM F748-06(2010), ASTM D638-10, ASTM D6394-10, ASTM D6394-09, ASTM D6394-08, ASTM D7474-08, ASTM D792-08, ASTM D6394-07, ASTM D256-06ae1, ASTM D256-06a. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F702-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:F702 −18
Standard Specification for
Polysulfone Resin for Medical Applications
ThisstandardisissuedunderthefixeddesignationF702;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoptionor,inthecaseofrevision,theyearoflastrevision.Anumberinparenthesesindicatestheyearoflastreapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
1.1 This specification covers polysulfone resin (poly(oxy-1,
1.7 This international standard was developed in accor-
4-phenylenesulfonyl-1,4–phenylene (dimethylmethylene)-1,
dance with internationally recognized principles on standard-
4–phenylene)) as defined in ISO 25137–1, supplied by a
ization established in the Decision on Principles for the
vendorinvirginform(pellets,powder,fabricatedformsandso
Development of International Standards, Guides and Recom-
forth) for medical applications. This specification provides
mendations issued by the World Trade Organization Technical
requirementsandassociatedtestmethodsforthisthermoplastic
Barriers to Trade (TBT) Committee.
whenitisintendedforuseinmanufacturingmedicaldevicesor
components of medical devices.
2. Referenced Documents
1.2 As with any material, some characteristics may be 2
2.1 ASTM Standards:
altered by the processing techniques (such as molding,
D256Test Methods for Determining the Izod Pendulum
extrusion, machining, sterilization, and so forth) required for
Impact Resistance of Plastics
the production of a specific part or device. Therefore, proper-
D638Test Method for Tensile Properties of Plastics
ties of fabricated forms of this resin should be evaluated using
D648Test Method for Deflection Temperature of Plastics
test methods which are appropriate to ensure safety and
Under Flexural Load in the Edgewise Position
efficacy as agreed upon by the vendor, purchaser, and regulat-
D792Test Methods for Density and Specific Gravity (Rela-
ing bodies.
tive Density) of Plastics by Displacement
1.3 Thestandardallowsfordesignationofpolysulfoneresin D6394Classification System for and Basis for Specification
for all medical applications. The actual extent of performance for Sulfone Plastics (SP)
and suitability for a specific application must be evaluated by D7474Practice for Determining Residual Stresses in Ex-
the vendor, purchaser, and regulating bodies. trudedorMoldedSulfonePlastic(SP)PartsbyImmersion
in Various Chemical Reagents
1.4 The properties included in this specification are those
F748PracticeforSelectingGenericBiologicalTestMethods
applicable for unfilled polysulfone (PSU) polymers with the
for Materials and Devices
addition of colorants and processing aids. Indicated properties
2.2 ISO Standards:
are for injection molded forms. Forms containing fillers or
ISO 10993–1Biological Evaluation of Medical Devices—
other additives, as well as polymer blends which contain PSU,
Part1:EvaluationandTestingWithinaRiskManagement
or reclaimed materials, are not covered by this specification.
Process
1.5 The values stated in SI units are to be regarded as
ISO 17025General Requirements for the Competence of
standard. No other units of measurement are included in this
Testing and Calibration Laboratories
standard.
ISO 25137–1Plastics—Sulfone Polymer Moulding and Ex-
1.6 When evaluating material in accordance with this trusion Materials—Part I: Designation System and Basis
specification, hazardous materials, operations, and equipment for Specifications
maybeinvolved.Thisstandarddoesnotpurporttoaddressall
3. Significance and Use
of the concerns, if any, associated with its use. It is the
responsibility of the user of this standard to establish appro-
3.1 This specification is designed to recommend test meth-
odstoestablishareasonablelevelofconfidenceconcerningthe
1 2
This specification is under the jurisdiction of ASTM Committee F04 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Medical and Surgical Materials and Devices and is the direct responsibility of contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Subcommittee F04.11 on Polymeric Materials. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Dec. 1, 2018. Published February 2019. Originally the ASTM website.
approved in 1981. Last previous edition approved in 2010 as F702–10. DOI: Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
10.1520/F0702-18. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F702−18
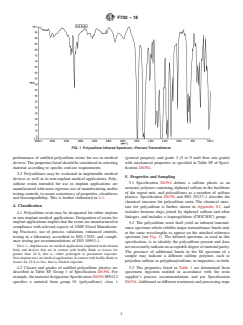
FIG. 1 Polysulfone Infrared Spectrum—Percent Transmittance
performance of unfilled polysulfone resins for use in medical (general purpose), and grade 2 (5 to 9 melt flow rate grade)
devices.Thepropertieslistedshouldbeconsideredinselecting with mechanical properties as specified in Table SP of Speci-
material according to specific end-use requirements. fication D6394.
3.2 Polysulfones may be evaluated in implantable medical
5. Properties and Sampling
devices as well as in non-implant medical applications. Poly-
5.1 Specification D6394 defines a sulfone plastic as an
sulfone resins intended for use in implant applications are
aromatic polymer containing diphenyl sulfone in the backbone
manufactured with more rigorous use of manufacturing and/or
of the repeat unit, and polysulfones as a member of sulfone
testingcontrols,toassureconsistencyofproperties,cleanliness
plastics. Specification D6394 and ISO 25137–1 describe the
and biocompatibility. This is further elaborated in 4.1.
chemical structure for polysulfone resin. The chemical struc-
4. Classification
ture for polysulfone is further shown in Appendix X1, and
includes benzene rings joined by diphenyl sulfone and ether
4.1 Polysulfone resin may be designated for either implant
linkages, and includes a isopropylidene (CH3CH3C) group.
or non-implant medical applications. Designation of resins for
implantapplicationsimpliesthattheresinsaremanufacturedin
5.2 The polysulfone resin shall yield an infrared transmit-
compliance with relevant aspects of GMP (Good Manufactur-
tance spectrum which exhibits major transmittance bands only
ing Practices), use of process validation, enhanced controls,
at the same wavelengths as appear on the attached reference
testing in a laboratory accredited to ISO 17025, and compli-
spectrum (see Fig. 1). The infrared spectrum, as used in this
ance testing per recommendations of ISO 10993–1.
specification, is to identify the polysulfone present and does
NOTE1—Implantusesaremedicalapplicationsimplantedinthehuman
notnecessarilyindicateanacceptabledegreeofmaterialpurity.
body and devices that are in contact with bodily fluids or tissues for
The presence of additional bands in the IR spectrum of a
greater than 24 h, that is, either prolonged or permanent exposure.
sample may indicate a different sulfone polymer, such as
Non-implantusesaremedicalapplicationsincontactwithbodilyfluidsor
polyether sulfone or polyphenylsulfone, or impurities, or both.
tissues for 24 h or less, that is, limited exposure.
4.2 Classes and grades of unfilled polysulfone plastics are 5.3 The properties listed in Table 1 are determined from
described in Table SP, Group 1 of Specification D6394. For specimens injection molded in accordance with the resin
example,thematerialdesignationSpecificationD6394SP0112 supplier’s process recommendations and per Specification
specifies a material from group 01 (polysulfone), class 1 D6394.Additional or different treatments and processing steps
F702−18
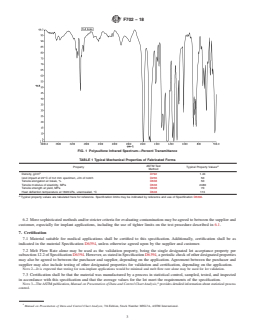
TABLE 1 Typical Mechanical Properties of Fabricated Forms
ASTM Test
A
Property Typical Property Values
Method
Density, g/cm D792 1.24
Izod impact at 22°C of 3.2 mm specimen, J/m of notch D256 50
Tensile elongation at break, % D638 50
Tensile modulus of elasticity, MPa D638 2480
Tensile strength at yield, MPa D638 70
Heat deflection temperature at 1820 kPa, unannealed, °C D648 174
A
Typical property values are tabulated here for reference. Specification limits may be indicated by reference and use of Specification D6394.
(such as extrusion, molding, machining, sterilization, and so 7.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F702 − 10 F702 − 18
Standard Specification for
Polysulfone Resin for Medical Applications
This standard is issued under the fixed designation F702; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This specification covers polysulfone resin (poly(oxy-1,4-phenylenesulfonyl-1,4–phenylene (dimethylmethylene)-1,
4–phenylene)) as defined in ISO 25137–1, supplied by a vendor in virgin form (pellets, powder, fabricated forms and so forth) for
medical applications. This specification provides requirements and associated test methods for this thermoplastic when it is
intended for use in manufacturing medical devices or components of medical devices.
1.2 As with any material, some characteristics may be altered by the processing techniques (such as molding, extrusion,
machining, sterilization, and so forth) required for the production of a specific part or device. Therefore, properties of fabricated
forms of this resin should be evaluated using test methods which are appropriate to ensure safety and efficacy as agreed upon by
the vendor, purchaser, and regulating bodies.
1.3 The standard allows for designation of polysulfone resin for all medical applications. The actual extent of performance and
suitability for a specific application must be evaluated by the vendor, purchaser, and regulating bodies.
1.4 The properties included in this specification are those applicable for unfilled polysulfone (PSU) polymers with the addition
of colorants and processing aids. Indicated properties are for injection molded forms. Forms containing fillers or other additives,
as well as polymer blends which contain PSU, or reclaimed materials, are not covered by this specification.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 When evaluating material in accordance with this specification, hazardous materials, operations, and equipment may be
involved. This standard does not purport to address all of the concerns, if any, associated with its use. It is the responsibility of
the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D256 Test Methods for Determining the Izod Pendulum Impact Resistance of Plastics
D638 Test Method for Tensile Properties of Plastics
D648 Test Method for Deflection Temperature of Plastics Under Flexural Load in the Edgewise Position
D792 Test Methods for Density and Specific Gravity (Relative Density) of Plastics by Displacement
D6394 Specification for Sulfone Plastics (SP)
D7474 Practice for Determining Residual Stresses in Extruded or Molded Sulfone Plastic (SP) Parts by Immersion in Various
Chemical Reagents
F748 Practice for Selecting Generic Biological Test Methods for Materials and Devices
2.2 ISO Standards:
ISO 1099310993–1 Biological Evaluation of Medical DevicesDevices—Part 1: Evaluation and Testing Within a Risk
Management Process
This specification is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee
F04.11 on Polymeric Materials.
Current edition approved Sept. 1, 2010Dec. 1, 2018. Published October 2010February 2019. Originally approved in 1981. Last previous edition approved in 20032010
as F702 – 98a (2003).F702 – 10. DOI: 10.1520/F0702-10.10.1520/F0702-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F702 − 18
ISO 17025 General Requirements for the Competence of Testing and Calibration Laboratories
ISO 25137–1 Plastics—Sulfone Polymer Moulding and Extrusion Materials—Part I: Designation System and Basis for
Specifications
3. Significance and Use
3.1 This specification is designed to recommend test methods to establish a reasonable level of confidence concerning the
performance of unfilled polysulfone resins for use in medical devices. The properties listed should be considered in selecting
material according to specific end-use requirements.
3.2 Polysulfones may be evaluated in implantable medical devices as well as in non-implant medical applications. Polysulfone
resins intended for use in implant applications are manufactured with more rigorous use of manufacturing and/or testing controls,
to assure consistency of properties, cleanliness and biocompatibility. This is further elaborated in 4.1.
4. Classification
4.1 Polysulfone resin may be designated for either implant or non-implant medical applications. Designation of resins for
implant applications implies that the resins are manufactured in compliance with relevant aspects of GMP (Good Manufacturing
Practices), use of process validation, enhanced controls, testing in a laboratory accredited to ISO 17025, and compliance testing
to ISO 10993:5 (cytotoxicity) and ISO 10993:18 (physiochemical testing). per recommendations of ISO 10993–1.
NOTE 1—Implant uses are medical applications implanted in the human body and devices that are in contact with bodily fluids or tissues for greater
than 24 h, that is, either prolonged or permanent exposure. Non-implant uses are medical applications in contact with bodily fluids or tissues for 24 h
or less, that is, limited exposure.
4.2 Classes and grades of unfilled polysulfone plastics are described in Table SP, Group 1 of Specification D6394. For example,
the material designation Specification D6394 SP0112 specifies a material from group 01 (polysulfone), class 1 (general purpose),
and grade 2 (5 to 9 melt flow rate grade) with mechanical properties as specified in Table SP of Specification D6394.
5. Properties and Sampling
5.1 Specification D6394 defines a sulfone plastic as an aromatic polymer containing diphenyl sulfone in the backbone of the
repeat unit, and polysulfones as a member of sulfone plastics. Specification D6394 and ISO 25137–1 describe the chemical
structure for polysulfone resin. The chemical structure for polysulfone is further shown in Appendix X1, and includes benzene
rings joined by diphenyl sulfone and ether linkages, and includes a isopropylidene (CH3CH3C) group.
5.2 The polysulfone resin shall yield an infrared transmittance spectrum which exhibits major transmittance bands only at the
same wavelengths as appear on the attached reference spectrum (see Fig. 1). The infrared spectrum, as used in this specification,
is to identify the polysulfone present and does not necessarily indicate an acceptable degree of material purity. The presence of
additional bands in the IR spectrum of a sample may indicate a different sulfone polymer, such as polyether sulfone or
polyphenylsulfone, or impurities, or both.
5.3 The properties listed in Table 1 are determined from specimens injection molded in accordance with the resin supplier’s
process recommendations and per Specification D6394. Additional or different treatments and processing steps (such as extrusion,
molding, machining, sterilization, and so forth) may alter the material properties. Table 1 lists typical properties of non-sterilized
fabricated forms.
5.4 Sampling shall be statistically adequate to satisfy the requirements of 7.3. The material shall be sampled with commonly
accepted sampling procedures or other sampling techniques as agreed upon between the customer and the supplier.
5.5 The quality of fabricated parts can be affected by residual stress from typical processing methods. Reduction of chemical
resistance of Polysulfone parts is a well-known effect of high residual stress. Fabricators and end-users may use Practice D7474
as a reference on evaluation of fabricated parts to help optimize processing parameters and improve environmental stress crack
resi
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...