ASTM F748-16
(Practice)Standard Practice for Selecting Generic Biological Test Methods for Materials and Devices
Standard Practice for Selecting Generic Biological Test Methods for Materials and Devices
SIGNIFICANCE AND USE
4.1 The objective of this practice is to recommend appropriate biological endpoint assessments (which may or may not require testing) to establish a reasonable level of confidence concerning the biological response to a material or device, while at the same time avoiding unnecessary testing.
4.2 This practice is intended to provide guidance to the materials investigator in selecting the proper procedures to be carried out for the screening of new or modified materials. Because each material and each implant situation involves its own unique circumstances, these recommendations should be modified as necessary and do not constitute the only assessment that will be required for a material. Nor should these guidelines be interpreted as minimum requirements for any particular situation. While an attempt has been made to provide recommendation for different implant circumstances, some of the recommended assessment may not be necessary or reasonable for a specific material or application.
SCOPE
1.1 This practice recommends generic biological test methods for materials and devices according to end-use applications. While chemical testing for extractable additives and residual monomers or residues from processing aids is necessary for most implant materials, such testing is not included as part of this practice. The reader is cautioned that the area of materials biocompatibility testing is a rapidly evolving field, and improved methods are evolving rapidly, so this practice is by necessity only a guideline. A thorough knowledge of current techniques and research is critical to a complete evaluation of new materials.
1.2 These test protocols are intended to apply to materials and medical devices for human application. Biological evaluation of materials and devices, and related subjects such as pyrogen testing, batch testing of production lots, and so on, are also discussed. Tests include those performed on materials, end products, and extracts. Rationale and comments on current state of the art are included for all test procedures described.
1.3 The biocompatibility of materials used in single or multicomponent medical devices for human use depends to a large degree on the particular nature of the end-use application. Biological reactions that are detrimental to the success of a material in one device application may have little or no bearing on the successful use of the material for a different application. It is, therefore, not possible to specify a set of biocompatibility test methods which will be necessary and sufficient to establish biocompatibility for all materials and applications.
1.4 The evaluation of tissue engineered medical products (TEMPs) may, in some cases, involve different or additional testing beyond those suggested for non-tissue-based materials and devices. Where appropriate, these differences are discussed in this practice and additional tests described.
1.5 The ethical use of research animals places the obligation on the individual investigator to determine the most efficient methods for performing the necessary testing without undue use of animals. Where adequate prior data exists to substantiate certain types of safety information, these guidelines should not be interpreted to mean that testing should be unnecessarily repeated.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2016
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.16 - Biocompatibility Test Methods
Relations
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Aug-2020
- Effective Date
- 01-Jun-2020
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Feb-2018
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Apr-2016
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-Oct-2013
Overview
ASTM F748-16: Standard Practice for Selecting Generic Biological Test Methods for Materials and Devices provides guidance for evaluating the biological response to materials and devices intended for human use. Developed by ASTM, this standard assists researchers, manufacturers, and regulators in selecting appropriate biological tests for assessing the biocompatibility of new or modified materials and medical devices. The standard emphasizes a risk-based approach to avoid unnecessary testing, encourages ethical use of animal studies, and supports efficient qualification of materials for healthcare applications.
Key Topics
- Biocompatibility Assessment: Guidance on selecting relevant biological endpoint assessments based on the intended use, type, and duration of material or device exposure within the human body.
- Test Selection Criteria: Recommends test methods for materials used in medical devices, considering device location (external, invasive, implantable), contact type (intact skin, breached surfaces, blood path), and expected duration of use (short-term, chronic, etc.).
- Biological Test Methods: Describes a range of biological tests, including:
- Cytotoxicity assays on cultured cells
- Sensitization and irritation testing (skin, mucous membranes)
- Systemic toxicity, hemolysis, and pyrogenicity
- Implantation studies for subcutaneous, intramuscular, and long-term applications
- Genotoxicity and carcinogenicity as needed based on device risk
- Complement activation and immunotoxicity testing for certain device categories
- Ethical Considerations: Promotes reduction of animal testing when possible by supplementing with literature review, validated in vitro methods, and prior data to avoid duplicative studies.
- Application-Specific Guidance: Recognizes that biocompatibility is use-dependent; a test necessary for one application may be irrelevant for another. Device classification by end-use and material type is central.
- Limitations: Clarifies that the standard serves as a guideline, not a checklist or minimum requirement, due to the evolving nature of biocompatibility science.
Applications
ASTM F748-16 is widely used in the medical device industry and by investigators involved in materials biocompatibility testing. Practical applications include:
- Screening New or Modified Materials: Assists researchers in choosing suitable biological evaluation strategies early in development, minimizing costly failures in later stages.
- Medical Device Qualification: Informs risk-based biological evaluation plans for regulatory submission, ensuring that devices meet relevant safety standards before human contact.
- Tissue-Engineered Products: Guides the assessment of tissue-engineered medical products, accounting for additional or unique biological compatibility questions.
- Process Validation and Change Management: Supports batch testing and the impact analysis of changes in material composition, processing, or sterilization on biological safety.
- Global Regulatory Alignment: Aligns with principles discussed in related ISO 10993-1 and FDA guidance, facilitating international market entry and compliance.
Related Standards
ASTM F748-16 references several key standards and guides used in combination for comprehensive biocompatibility testing:
- ISO 10993-1: Biological evaluation of medical devices - Evaluation and testing within a risk management process
- ASTM F619: Extraction of medical plastics
- ASTM F719 / F720 / F749 / F750 / F813 / F895: Various biological tests for skin irritation, sensitization, and cytotoxicity
- ASTM F981 / F1027 / F1408 / F1439: Implantation and carcinogenicity testing
- ASTM F756: Hemolytic properties of materials
- ASTM F2382 / F1984: Blood compatibility and complement activation testing
- General Program Memorandum #G95-1 and FDA Immunotoxicity Testing Guidance
- EN 10993-1: European equivalent of ISO 10993-1
Conclusion
ASTM F748-16 plays a central role in the responsible selection and execution of biological test methods for medical device materials and products. By enabling targeted testing, ethical practices, and risk-based evaluation, this standard streamlines the pathway to safer, compliant medical innovations worldwide. For optimal outcomes, users should incorporate current scientific advances and harmonize with related biocompatibility standards.
Buy Documents
ASTM F748-16 - Standard Practice for Selecting Generic Biological Test Methods for Materials and Devices
REDLINE ASTM F748-16 - Standard Practice for Selecting Generic Biological Test Methods for Materials and Devices
Frequently Asked Questions
ASTM F748-16 is a standard published by ASTM International. Its full title is "Standard Practice for Selecting Generic Biological Test Methods for Materials and Devices". This standard covers: SIGNIFICANCE AND USE 4.1 The objective of this practice is to recommend appropriate biological endpoint assessments (which may or may not require testing) to establish a reasonable level of confidence concerning the biological response to a material or device, while at the same time avoiding unnecessary testing. 4.2 This practice is intended to provide guidance to the materials investigator in selecting the proper procedures to be carried out for the screening of new or modified materials. Because each material and each implant situation involves its own unique circumstances, these recommendations should be modified as necessary and do not constitute the only assessment that will be required for a material. Nor should these guidelines be interpreted as minimum requirements for any particular situation. While an attempt has been made to provide recommendation for different implant circumstances, some of the recommended assessment may not be necessary or reasonable for a specific material or application. SCOPE 1.1 This practice recommends generic biological test methods for materials and devices according to end-use applications. While chemical testing for extractable additives and residual monomers or residues from processing aids is necessary for most implant materials, such testing is not included as part of this practice. The reader is cautioned that the area of materials biocompatibility testing is a rapidly evolving field, and improved methods are evolving rapidly, so this practice is by necessity only a guideline. A thorough knowledge of current techniques and research is critical to a complete evaluation of new materials. 1.2 These test protocols are intended to apply to materials and medical devices for human application. Biological evaluation of materials and devices, and related subjects such as pyrogen testing, batch testing of production lots, and so on, are also discussed. Tests include those performed on materials, end products, and extracts. Rationale and comments on current state of the art are included for all test procedures described. 1.3 The biocompatibility of materials used in single or multicomponent medical devices for human use depends to a large degree on the particular nature of the end-use application. Biological reactions that are detrimental to the success of a material in one device application may have little or no bearing on the successful use of the material for a different application. It is, therefore, not possible to specify a set of biocompatibility test methods which will be necessary and sufficient to establish biocompatibility for all materials and applications. 1.4 The evaluation of tissue engineered medical products (TEMPs) may, in some cases, involve different or additional testing beyond those suggested for non-tissue-based materials and devices. Where appropriate, these differences are discussed in this practice and additional tests described. 1.5 The ethical use of research animals places the obligation on the individual investigator to determine the most efficient methods for performing the necessary testing without undue use of animals. Where adequate prior data exists to substantiate certain types of safety information, these guidelines should not be interpreted to mean that testing should be unnecessarily repeated. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 4.1 The objective of this practice is to recommend appropriate biological endpoint assessments (which may or may not require testing) to establish a reasonable level of confidence concerning the biological response to a material or device, while at the same time avoiding unnecessary testing. 4.2 This practice is intended to provide guidance to the materials investigator in selecting the proper procedures to be carried out for the screening of new or modified materials. Because each material and each implant situation involves its own unique circumstances, these recommendations should be modified as necessary and do not constitute the only assessment that will be required for a material. Nor should these guidelines be interpreted as minimum requirements for any particular situation. While an attempt has been made to provide recommendation for different implant circumstances, some of the recommended assessment may not be necessary or reasonable for a specific material or application. SCOPE 1.1 This practice recommends generic biological test methods for materials and devices according to end-use applications. While chemical testing for extractable additives and residual monomers or residues from processing aids is necessary for most implant materials, such testing is not included as part of this practice. The reader is cautioned that the area of materials biocompatibility testing is a rapidly evolving field, and improved methods are evolving rapidly, so this practice is by necessity only a guideline. A thorough knowledge of current techniques and research is critical to a complete evaluation of new materials. 1.2 These test protocols are intended to apply to materials and medical devices for human application. Biological evaluation of materials and devices, and related subjects such as pyrogen testing, batch testing of production lots, and so on, are also discussed. Tests include those performed on materials, end products, and extracts. Rationale and comments on current state of the art are included for all test procedures described. 1.3 The biocompatibility of materials used in single or multicomponent medical devices for human use depends to a large degree on the particular nature of the end-use application. Biological reactions that are detrimental to the success of a material in one device application may have little or no bearing on the successful use of the material for a different application. It is, therefore, not possible to specify a set of biocompatibility test methods which will be necessary and sufficient to establish biocompatibility for all materials and applications. 1.4 The evaluation of tissue engineered medical products (TEMPs) may, in some cases, involve different or additional testing beyond those suggested for non-tissue-based materials and devices. Where appropriate, these differences are discussed in this practice and additional tests described. 1.5 The ethical use of research animals places the obligation on the individual investigator to determine the most efficient methods for performing the necessary testing without undue use of animals. Where adequate prior data exists to substantiate certain types of safety information, these guidelines should not be interpreted to mean that testing should be unnecessarily repeated. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM F748-16 is classified under the following ICS (International Classification for Standards) categories: 07.080 - Biology. Botany. Zoology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F748-16 has the following relationships with other standards: It is inter standard links to ASTM F748-06(2010), ASTM F1408-20a, ASTM F1408-20, ASTM F2382-18, ASTM E1262-88(2018), ASTM F720-17, ASTM F2382-17e1, ASTM F2382-17, ASTM F1877-16, ASTM F2147-01(2016), ASTM F756-13, ASTM F1984-99(2013), ASTM E1262-88(2013), ASTM F1439-03(2013), ASTM F1408-97(2013). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F748-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F748 − 16
Standard Practice for
Selecting Generic Biological Test Methods for Materials and
Devices
ThisstandardisissuedunderthefixeddesignationF748;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope methods for performing the necessary testing without undue
useofanimals.Whereadequatepriordataexiststosubstantiate
1.1 This practice recommends generic biological test meth-
certain types of safety information, these guidelines should not
ods for materials and devices according to end-use applica-
be interpreted to mean that testing should be unnecessarily
tions. While chemical testing for extractable additives and
repeated.
residual monomers or residues from processing aids is neces-
1.6 This standard does not purport to address all of the
sary for most implant materials, such testing is not included as
safety concerns, if any, associated with its use. It is the
part of this practice. The reader is cautioned that the area of
responsibility of the user of this standard to establish appro-
materials biocompatibility testing is a rapidly evolving field,
priate safety and health practices and determine the applica-
and improved methods are evolving rapidly, so this practice is
bility of regulatory limitations prior to use.
bynecessityonlyaguideline.Athoroughknowledgeofcurrent
techniques and research is critical to a complete evaluation of
2. Referenced Documents
new materials.
2.1 ASTM Standards:
1.2 These test protocols are intended to apply to materials
E1262 Guide for Performance of Chinese Hamster Ovary
and medical devices for human application. Biological evalu-
Cell/Hypoxanthine Guanine Phosphoribosyl Transferase
ation of materials and devices, and related subjects such as
Gene Mutation Assay
pyrogen testing, batch testing of production lots, and so on, are
F619 Practice for Extraction of Medical Plastics
alsodiscussed.Testsincludethoseperformedonmaterials,end
F719 Practice for Testing Biomaterials in Rabbits for Pri-
products, and extracts. Rationale and comments on current
mary Skin Irritation
state of the art are included for all test procedures described.
F720 PracticeforTestingGuineaPigsforContactAllergens:
1.3 The biocompatibility of materials used in single or
Guinea Pig Maximization Test
multicomponent medical devices for human use depends to a
F749 Practice for Evaluating Material Extracts by Intracuta-
largedegreeontheparticularnatureoftheend-useapplication.
neous Injection in the Rabbit
Biological reactions that are detrimental to the success of a
F750 Practice for Evaluating Material Extracts by Systemic
materialinonedeviceapplicationmayhavelittleornobearing
Injection in the Mouse
on the successful use of the material for a different application.
F756 Practice for Assessment of Hemolytic Properties of
It is, therefore, not possible to specify a set of biocompatibility
Materials
test methods which will be necessary and sufficient to establish
F763 Practice for Short-Term Screening of Implant Materi-
biocompatibility for all materials and applications.
als
F813 Practice for Direct Contact Cell Culture Evaluation of
1.4 The evaluation of tissue engineered medical products
Materials for Medical Devices
(TEMPs) may, in some cases, involve different or additional
F895 TestMethodforAgarDiffusionCellCultureScreening
testing beyond those suggested for non-tissue-based materials
for Cytotoxicity
anddevices.Whereappropriate,thesedifferencesarediscussed
F981 Practice for Assessment of Compatibility of Biomate-
in this practice and additional tests described.
rials for Surgical Implants with Respect to Effect of
1.5 Theethicaluseofresearchanimalsplacestheobligation
Materials on Muscle and Bone
on the individual investigator to determine the most efficient
F1027 Practice for Assessment of Tissue and Cell Compat-
ibility of Orofacial Prosthetic Materials and Devices
ThispracticeisunderthejurisdictionofASTMCommitteeF04onMedicaland
Surgical Materials and Devicesand is direct responsibility of Subcommittee F04.16
on Biocompatibility Test Methods. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved April 1, 2016. Published May 2016. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1982. Last previous edition approved in 2010 as F748 – 06 (2010). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/F0748-16. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F748 − 16
F1408 Practice for Subcutaneous Screening Test for Implant concerning the biological response to a material or device,
Materials while at the same time avoiding unnecessary testing.
F1439 Guide for Performance of Lifetime Bioassay for the
4.2 This practice is intended to provide guidance to the
Tumorigenic Potential of Implant Materials
materials investigator in selecting the proper procedures to be
F1877 Practice for Characterization of Particles
carried out for the screening of new or modified materials.
F1903 Practice for Testing For Biological Responses to
Because each material and each implant situation involves its
Particles In Vitro
own unique circumstances, these recommendations should be
F1904 Practice for Testing the Biological Responses to
modified as necessary and do not constitute the only assess-
Particles in vivo
ment that will be required for a material. Nor should these
F1983 Practice forAssessment of Selected Tissue Effects of
guidelines be interpreted as minimum requirements for any
Absorbable Biomaterials for Implant Applications
particularsituation.Whileanattempthasbeenmadetoprovide
F1984 Practice for Testing for Whole Complement Activa-
recommendation for different implant circumstances, some of
tion in Serum by Solid Materials
the recommended assessment may not be necessary or reason-
F2065 Practice for Testing forAlternative Pathway Comple-
able for a specific material or application.
ment Activation in Serum by Solid Materials (Withdrawn
2016)
5. Classification of Materials and Devices by End-Use
F2147 Practice for Guinea Pig: Split Adjuvant and Closed
Applications
Patch Testing for Contact Allergens
F2148 Practice for Evaluation of Delayed Contact Hyper-
5.1 General:
sensitivity Using the Murine Local Lymph Node Assay
5.1.1 When new materials are sought for a medical appli-
(LLNA)
cation for use on humans, the material(s) may comprise the
F2382 Test Method forAssessment of Intravascular Medical
whole final device product, or may be one of many component
Device Materials on Partial Thromboplastin Time (PTT)
materials in the device. The first step is a thorough literature
2.2 Other Referenced Documents:
search for previous use of the material or biocompatibility
ISO/AAMI/ANSI 10993-1 Biological Testing of Medical
testing studies to ensure that it has not been known to produce
andDentalMaterialsandDevices—Part1:Evaluationand
an adverse biological response that exceeds the expected
Testing within a Risk Management Process
benefit in the use of the device. Note that the final fabricated
EN 10993-1 Biological Testing of Medical and Dental Ma-
product may differ chemically, physically, or biologically from
terialsandDevices—Part1:EvaluationandTestingwithin
the raw materials used to fabricate the product due to process-
a Risk Management Process
ing and this has to be considered when conducting a biocom-
General Program Memorandum #G95-1 FDA
patibility evaluation and/or designing test protocols. For some
Immunotoxicity Testing Guidance-FDA
devices,iftestingisneeded,itmaybenecessaryordesirableto
take material test samples directly from the final device
3. Summary of Practice
product. Samples should be fully representative of the finished
3.1 A matrix listing biological endpoints relevant to a product in terms of processing, cleaning, packaging,
biocompatibility evaluation versus materials (devices) and
sterilization, and any other procedures that are performed on
their applications is included in Table 1.The expected duration the materials before the device is used.
of use of the device is also considered. Intraoperative is less
5.1.2 At this point, preliminary material screening may be
than 24 h, short-term is up to and including 30 days, and
employed, depending on the expertise of the organization(s)
chronic is greater than 30 days. The position of row and
evaluating the materials. Since preliminary screening is nor-
column intersection is marked to indicate whether assessment
mally an option to minimize the economic impact of a
of a biological endpoint is recommended for a material or
candidate material failing final biological tests after extensive
device for the specific application indicated.The terms relating
time and effort, it is not a required procedure. The investigator
to device or material type and application are addressed in
should be aware that, should an adverse tissue response be
Section 5. Discussion of applicability, current state of the art,
observed with a final product, it may be impossible to
and rationale for individual biological endpoint assessments
determine which component or process is responsible without
also appears in that section.
these initial screening tests.
5.1.3 This practice addresses two aspects of tissue-material
4. Significance and Use
interactions: duration and tissue type. A third aspect, which
4.1 The objective of this practice is to recommend appro-
shouldbeconsidered,istherelativesizedifferencebetweenthe
priate biological endpoint assessments (which may or may not
host and the material, that is, to how much material surface
require testing) to establish a reasonable level of confidence
area is the host exposed. The material surface area-to-body
weight ratio may become a significant factor for porous
materials, and devices of repeated short-term applications (for
The last approved version of this historical standard is referenced on
example, dialysis products). While this practice does not
www.astm.org.
addresstheissueof“intensityfactor”ofincreasedsurfacearea,
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the biocompatibility testing facility personnel should consider
4th Floor, New York, NY 10036, http://www.ansi.org.
Available from CDRH, 5600 Fishers Ln., Rockville, MD 20857. it in their material screening and testing protocol design.
F748 − 16
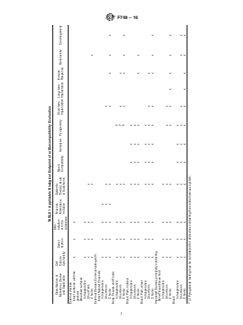
TABLE 1 Applicable Biological Endpoints for Biocompatibility Evaluation
Skin
Classification of Cell Mucous Systemic
Sensi- Irritation Blood Short-term Long-term Immune
Material or Device Culture Membrane Toxicity, Acute Hemolysis Pyrogenicity Genotoxicity Carcinogenicity
tization or Intra- Compatibility Implantation Implantation Response
and Application Cytotoxicity Irritation or Subchronic
cutaneous
External devices
Intact surfaces (all time xx x
periods)
Breached surfaces
Intraoperative x x x
Short-Term x x x x
Chronic x x x x x
External Devices Communicating with:
Intact Natural Channels
Intraoperative x x x x
Short-term x x x x x x
Chronic x x x x x x x x x
Body Tissues and Fluids
A
Intraoperative x x x x i
A
Short-term x x x x i xx
A
Chronic x x x x i xxx x
Blood Path, indirect
Intraoperative x x x x x x x
Short-term x x x x x x x
Chronic x x x x x x x x x
Blood Path, direct
Intraoperative x x x x x x x
Short-term x x x x x x x x x
Chronic x x x x x x x x x x x
Implanted Devices principally contacting
Bone/Tissue/tissue fluid
Intraoperative x x x x
Short-term x x x x x x
Chronic x x x x x x x x x x
Blood
Intraoperative x x x x x x x
Short-term x x x x x x x x x x
Chronic x x x x x x x x x x x x
A
(i) Pyrogenicity testing may be considered for all devices contacting the central nervous system.
F748 − 16
5.1.4 For the purposes of this practice, devices and the 5.3.3.1 Products that are used for >24 hours or that are used
materialsthatcomprisethemareclassifiedastoend-usehuman repeatedly in the same patient will be considered as chronic
application as outlined in 5.2 – 5.4. usage and should undergo extended testing.
5.3.4 Blood, Path, Direct—Single recirculating blood expo-
5.1.5 In general, the assessment for tissue engineered medi-
sure or product that is in the blood path, generally for less than
cal products (TEMPs) should address the same issues specific
24 hours. Examples include intravenous catheters,
to the type, location, and duration of use as other medical
oxygenators, extracorporeal oxygenator tubing and accesso-
devices and products. The selection of additional assessment
for compatibility criteria unique to these type of products ries.
5.3.5 Blood Path, Direct, Short Term, or Chronic, or re-
should be conducted with these recommendations in mind.
peated exposure—Examples include dialyzers or dialysis tub-
5.1.6 When assessing materials that are intended to degrade
ing and accessories, shunts.
and/or be metabolized while implanted in the body (both
synthetic and TEMPs), consideration should be given to the
5.4 Implanted Long-Term Devices:
degradation or metabolic products and appropriate modifica-
5.4.1 Devices Principally Contacting Bones—examples in-
tions made in test and sample selection so that the compatibil-
clude orthopedic pins, screws, replacement joints, bone
ity of degradation products as well as ungraded product are
prostheses, cements, and dental implants.
tested.
5.4.2 Devices Principally Residing in the Subcutaneous
Space—examples include pacemakers, neuromuscular
5.2 External Devices:
stimulators, facial augmentation devices, tissue expander
5.2.1 Devices That Contact Intact Body Surfaces Only—
devices, and breast prostheses.
examples include electrodes, splints, external prostheses, cer-
5.4.3 Devices Principally Contacting Soft Tissue and Tissue
taindressings,monitorsofvarioustypes,orostomyappliances.
Fluids—examples include drug supply devices, neuromuscular
5.2.2 Devices That Contact Breached Body Surfaces—
sensors, replacement tendons, penile, and other implants,
examples include ulcer, burn, and granulation tissue dressings,
cerebrospinalfluiddrains,artificiallarynx,vasdeferensvalves,
or healing devices.
and ligation clips.
5.3 Externally Communicating Devices:
5.4.4 Devices Principally Contacting Blood—examples in-
5.3.1 Devices Communicating with Intact Natural Chan-
clude pacemaker leads, artificial arteriovenous fistulae, heart
nels:
valves, vascular grafts, stents, blood monitors, internal drug
5.3.1.1 Intraoperative (<24 hours)—examples include in-
delivery catheters, and ventricular assist pumps.
traintestinal devices (such as sigmoidoscopes, colonoscopes,
stomach tubes, or gastroscopes), tracheal tubes, bronchoscopes
6. Selection of Test Procedures
and any parts of ancillary equipment that are in contact with
6.1 General—When it is determined that biocompatibility
materials entering the body, and irrigation sets.
testing is needed, the following should be considered:
5.3.1.2 Short-term(uptoandincluding30days)—examples
6.1.1 Biocompatibility testing involves tests of either the
include contact lenses, urinary catheters, and intravaginal
material itself, or an extract from it (see Practice F619), or
devices.
both,dependingonthenatureoftheend-useapplication.While
5.3.1.3 Chronic (>30 days)—examples include urinary
this practice does not address specific chemical methods for
catheters for chronic use and intrauterine devices.
evaluating the extractable substances or residuals from implant
5.3.2 Devices Communicating with Body Tissues and Flu-
materials, several of the recommended tests (see 6.2, 6.3, 6.6,
ids:
and 6.7) utilize extracts rather than the original material for
5.3.2.1 Intraoperative (<24 hours)—examples include hy-
testing. If sensitive chemical assay techniques (such as GC,
podermic needles, penetrating electrodes, biopsy instruments,
HPLC, and AA) should reveal no detectable substances being
arthroscopes, laparoscopes, irrigation equipment, surgical
extracted into the medium, consideration may be given to
instruments, trochars, and any parts of ancillary equipment that
deletion of these tests from the test battery. The investigator is
are in contact with materials entering the body.
cautioned, however, that the detection limit of the analytical
5.3.2.2 Short-term(uptoandincluding30days)—examples
chemistry procedures may not be adequate to detect trace
include cranial calipers, perfusion apparatus, drainage
extractables that may generate a tissue response. Before
apparatus, stabilizing orthopedic devices, and any parts of
analysis of extracts is substituted for actual biocompatibility
ancillary equipment that are in contact with material entering
testing of the extracts, validation procedures to show the
the body.
relative tissue response to levels of extractable which are
5.3.2.3 Chronic (>30 days)—examples include percutane-
slightly above the detection limit may be necessary. It is
ous electrodes, active penetrating electrodes, stapedectomy
particularly appropriate that animal testing involving extracts
prostheses, partial and total ossicular replacement prostheses,
be considered for deletion if there are no detectable substances
and tympanoplasty ventilation tubes. being extracted.
5.3.3 Blood Path, Indirect—Products contacting blood path 6.1.2 If the material to be tested is in the form of particles,
at one point (usually less than 24 hours), and that serve as a characterization of the particles in accordance with Practice
conduit for fluid entry into the vascular system. Examples F1877 should be performed so that the particles can be fully
include solution administration sets, extension sets, transfer described and their relevance to clinical usage situations
sets,
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F748 − 06 (Reapproved 2010) F748 − 16
Standard Practice for
Selecting Generic Biological Test Methods for Materials and
Devices
This standard is issued under the fixed designation F748; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice recommends generic biological test methods for materials and devices according to end-use applications.
While chemical testing for extractable additives and residual monomers or residues from processing aids is necessary for most
implant materials, such testing is not included as part of this practice. The reader is cautioned that the area of materials
biocompatibility testing is a rapidly evolving field, and improved methods are evolving rapidly, so this practice is by necessity only
a guideline. A thorough knowledge of current techniques and research is critical to a complete evaluation of new materials.
1.2 These test protocols are intended to apply to materials and medical devices for human application. Biological evaluation of
materials and devices, and related subjects such as pyrogen testing, batch testing of production lots, and so on, are also discussed.
Tests include those performed on materials, end products, and extracts. Rationale and comments on current state of the art are
included for all test procedures described.
1.3 The biocompatibility of materials used in single or multicomponent medical devices for human use depends to a large
degree on the particular nature of the end-use application. Biological reactions that are detrimental to the success of a material in
one device application may have little or no bearing on the successful use of the material for a different application. It is, therefore,
not possible to specify a set of biocompatibility test methods which will be necessary and sufficient to establish biocompatibility
for all materials and applications.
1.4 The evaluation of tissue engineered medical products (TEMPs) may, in some cases, involve different or additional testing
beyond those suggested for non-tissue-based materials and devices. Where appropriate, these differences are discussed in this
practice and additional tests described.
1.5 The ethical use of research animals places the obligation on the individual investigator to determine the most efficient
methods for performing the necessary testing without undue use of animals. Where adequate prior data exists to substantiate certain
types of safety information, these guidelines should not be interpreted to mean that testing should be unnecessarily repeated.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
E1202 Guide for Development of Micronucleus Assay Standards (Withdrawn 2013)
E1262 Guide for Performance of Chinese Hamster Ovary Cell/Hypoxanthine Guanine Phosphoribosyl Transferase Gene
Mutation Assay
E1263 Guide for Conduct of Micronucleus Assays in Mammalian Bone Marrow Erythrocytes (Withdrawn 2014)
E1280 Guide for Performing the Mouse Lymphoma Assay for Mammalian Cell Mutagenicity (Withdrawn 2014)
E1397 Practice for In Vitro Rat Hepatocyte DNA Repair Assay (Withdrawn 2013)
E1398 Practice for In Vivo Rat Hepatocyte DNA Repair Assay (Withdrawn 2013)
This practice is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devicesand is direct responsibility of Subcommittee F04.16 on
Biocompatibility Test Methods.
Current edition approved June 1, 2010April 1, 2016. Published September 2010May 2016. Originally approved in 1982. Last previous edition approved in 20062010 as
F748 – 06.F748 – 06 (2010). DOI: 10.1520/F0748-06R10.10.1520/F0748-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F748 − 16
F619 Practice for Extraction of Medical Plastics
F719 Practice for Testing Biomaterials in Rabbits for Primary Skin Irritation
F720 Practice for Testing Guinea Pigs for Contact Allergens: Guinea Pig Maximization Test
F749 Practice for Evaluating Material Extracts by Intracutaneous Injection in the Rabbit
F750 Practice for Evaluating Material Extracts by Systemic Injection in the Mouse
F756 Practice for Assessment of Hemolytic Properties of Materials
F763 Practice for Short-Term Screening of Implant Materials
F813 Practice for Direct Contact Cell Culture Evaluation of Materials for Medical Devices
F895 Test Method for Agar Diffusion Cell Culture Screening for Cytotoxicity
F981 Practice for Assessment of Compatibility of Biomaterials for Surgical Implants with Respect to Effect of Materials on
Muscle and Bone
F1027 Practice for Assessment of Tissue and Cell Compatibility of Orofacial Prosthetic Materials and Devices
F1408 Practice for Subcutaneous Screening Test for Implant Materials
F1439 Guide for Performance of Lifetime Bioassay for the Tumorigenic Potential of Implant Materials
F1877 Practice for Characterization of Particles
F1903 Practice for Testing For Biological Responses to Particles In Vitro
F1904 Practice for Testing the Biological Responses to Particles in vivo
F1905 Practice For Selecting Tests for Determining the Propensity of Materials to Cause Immunotoxicity (Withdrawn 2011)
F1906 Practice for Evaluation of Immune Responses In Biocompatibility Testing Using ELISA Tests, Lymphocyte Proliferation,
and Cell Migration (Withdrawn 2011)
F1983 Practice for Assessment of Selected Tissue Effects of Absorbable Biomaterials for Implant Applications
F1984 Practice for Testing for Whole Complement Activation in Serum by Solid Materials
F2065 Practice for Testing for Alternative Pathway Complement Activation in Serum by Solid Materials (Withdrawn 2016)
F2147 Practice for Guinea Pig: Split Adjuvant and Closed Patch Testing for Contact Allergens
F2148 Practice for Evaluation of Delayed Contact Hypersensitivity Using the Murine Local Lymph Node Assay (LLNA)
F2151 Practice for Assessment of White Blood Cell Morphology After Contact with Materials (Withdrawn 2007)
F2382 Test Method for Assessment of Intravascular Medical Device Materials on Partial Thromboplastin Time (PTT)
2.2 Other Referenced Documents:
ISO/AAMI/ANSI 10993-1 Biological Testing of Medical and Dental Materials and Devices - Part 1: Guidance on Selection of
TestsDevices—Part 1: Evaluation and Testing within a Risk Management Process
EN 30993–110993-1 Biological Testing of Medical and Dental Materials and Devices - Part 1: Guidance on Selection of
TestsDevices—Part 1: Evaluation and Testing within a Risk Management Process
General Program Memorandum #G95-1 FDA
Immunotoxicity Testing Guidance-FDA
3. Summary of Practice
3.1 A matrix listing biological test methods endpoints relevant to a biocompatibility evaluation versus materials (devices) and
their applications is included in Table 1. The expected duration of use of the device is also considered. Intraoperative is less than
24 h, short-term is up to and including 30 days, and chronic is greater than 30 days. The position of row and column intersection
is marked to indicate whether the test assessment of a biological endpoint is recommended for a material or device for the specific
application indicated. The terms relating to device or material type and application are addressed in Section 5. Discussion of
applicability, current state of the art, and rationale for individual test methods biological endpoint assessments also appears in that
section.
4. Significance and Use
4.1 The objective of this practice is to recommend sufficient biological testing appropriate biological endpoint assessments
(which may or may not require testing) to establish a reasonable level of confidence concerning the biological response to a
material or device, while at the same time avoiding unnecessary testing.
4.2 This practice is intended to provide guidance to the materials investigator in selecting the proper procedures to be carried
out for the screening of new or modified materials. Because each material and each implant situation involves its own unique
circumstances, these recommendations should be modified as necessary and do not constitute the only testingassessment that will
be required for a material normaterial. Nor should these guidelines be interpreted as minimum requirements for any particular
situation. While an attempt has been made to provide recommendation for different implant circumstances, some of the
recommended testingassessment may not be necessary or reasonable for a specific material or application.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Available from CDRH, 5600 Fishers Ln., Rockville, MD 20857.
F748 − 16
TABLE 1 Applicable TestsBiological Endpoints for Biocompatibility Evaluation
Skin
Classification of Cell Mucous Systemic
Sensi- Irritation Blood PyrogenPyrogenicityShort-term Long-term Immune
Material or Device Culture Membrane Toxicity, Acute Hemolysis Genotoxicity Carcinogenicity
tization or Intra- Compatibility Test Implantation Implantation Response
and Application Cytotoxicity Irritation or Subchronic
cutaneous
External devices
Intact surfaces (all time x x x
periods)
Breached surfaces
Intraoperative x x x
Short-Term x x x x
Chronic x x x x x
External Devices Communicating with:
Intact Natural Channels
Intraoperative x x x x
Short-term x x x x x x
Chronic x x x x x x x x x
Body Tissues and Fluids
A
Intraoperative x x x x i
A
Short-term x x x x i x x
A
Chronic x x x x i x x x x
Blood Path, indirect
Intraoperative x x x x x x x
Short-term x x x x x x x
Chronic x x x x x x x x x
Blood Path, direct
Intraoperative x x x x x x x
Short-term x x x x x x x x x
Chronic x x x x x x x x x x x
Implanted Devices principally contacting
Bone/Tissue/tissue fluid
Intraoperative x x x x
Short-term x x x x x x
Chronic x x x x x x x x x x
Blood
Intraoperative x x x x x x x
Short-term x x x x x x x x x x
Chronic x x x x x x x x x x x x
A
(i) Pyrogenicity testing may be considered for all devices contacting the central nervous system.
F748 − 16
5. Classification of Materials and Devices by End-Use Applications
5.1 General:
5.1.1 When new materials are sought for a medical application for use on humans, the material(s) may comprise the whole final
device product, or may be one of many component materials in the device. The first step is a thorough literature search for previous
use of the material or biocompatibility testing studies to ensure that it has not been known to produce an adverse biological
response that exceeds the expected benefit in the use of the device. Note that the final fabricated product may differ chemically,
physically, or biologically from the raw materials used to fabricate the product due to processing and this has to be considered when
conducting a biocompatibility evaluation and/or designing test protocols. For some devices, if testing is needed, it may be
necessary or desirable to take material test samples directly from the final device product. Samples should be fully representative
of the finished product in terms of processing, cleaning, packaging, sterilization, and any other procedures that are performed on
the materials before the device is used.
5.1.2 At this point, preliminary material screening may be employed, depending on the expertise of the organizationsorgani-
zation(s) evaluating the materials. Since preliminary screening is normally an option to minimize the economic impact of a
candidate material failing final biological tests after extensive time and effort, it is not a required procedure. The investigator should
be aware that, should an adverse tissue response be observed with a final product, it may be impossible to determine which
component or process is responsible without these initial screening tests.
5.1.3 This practice addresses two dimensionsaspects of tissue-material interactions: duration and tissue type. A third
dimension,aspect, which should be considered, is the relative size difference between the host and the material, that is, to how much
material surface area is the host exposed. The material surface area to body area-to-body weight ratio may become a significant
factor for porous materials, and devices of repeated short-term applications (for example, dialysis products). While this practice
does not address the issue of “intensity factor” of increased surface area, the biocompatibility testing facility personnel should
consider it in their material screening and testing protocol design.
5.1.4 For the purposes of this practice, devices and the materials that comprise them are classified as to end-use human
application as outlined in 5.2 – 5.4.
5.1.5 In general, the testingassessment for tissue engineered medical products (TEMPs) should address the same issues specific
to the type, location, and duration of use as other medical devices and products. The selection of additional testingassessment for
compatibility criteria unique to these type of products should be conducted with these recommendations in mind.
5.1.6 When testingassessing materials that are intended to degrade and/or be metabolized while implanted in the body (both
synthetic and TEMPs), consideration should be given to the degradation or metabolic products and appropriate modifications made
in test and sample selection so that the compatibility of degradation products is tested as well as the undegraded product.ungraded
product are tested.
5.2 External Devices:
5.2.1 Devices That Contact Intact Body Surfaces Only—examples include electrodes, splints, external prostheses, certain
dressings, monitors of various types, or ostomy appliances.
5.2.2 Devices That Contact Breached Body Surfaces—examples include ulcer, burn, and granulation tissue dressings, or healing
devices.
5.3 Externally Communicating Devices:
5.3.1 Devices Communicating with Intact Natural Channels:
5.3.1.1 Intraoperative (<24 hours)—examples include intraintestinal devices (such as sigmoidoscopes, colonoscopes, stomach
tubes, or gastroscopes), tracheal tubes, bronchoscopes and any parts of ancillary equipment that are in contact with materials
entering the body, and irrigation sets.
5.3.1.2 Short-term (up to and including 30 days)—examples include contact lenses, urinary catheters, and intravaginal devices.
5.3.1.3 Chronic (>30 days)—examples include urinary catheters for chronic use and intrauterine devices.
5.3.2 Devices Communicating with Body Tissues and Fluids:
5.3.2.1 Intraoperative (<24 hours)—examples include hypodermic needles, penetrating electrodes, biopsy instruments,
arthroscopes, laparoscopes, irrigation equipment, surgical instruments, trochars, and any parts of ancillary equipment that are in
contact with materials entering the body.
5.3.2.2 Short-term (up to and including 30 days)—examples include cranial calipers, perfusion apparatus, drainage apparatus,
stabilizing orthopedic devices, and any parts of ancillary equipment that are in contact with material entering the body.
5.3.2.3 Chronic (>30 days)—examples include percutaneous electrodes, active penetrating electrodes, stapedectomy prostheses,
partial and total ossicular replacement prostheses, orand tympanoplasty ventilation tubes.
5.3.3 Blood Path, Indirect—Products contacting blood path at one point for usually (usually less than 24 hours,hours), and
serves that serve as a conduit for fluid entry into the vascular system. Examples include solution administration sets, extension sets,
transfer sets, orand blood administration sets.
5.3.3.1 Products that are used for >24 hours or that are used repeatedly in the same patient will be considered as chronic usage
and should undergo extended testing.
F748 − 16
5.3.4 Blood, Path, Direct—Single recirculating blood exposure or product that is in the blood path, generally for less than 24
hours. Examples include intravenous catheters, oxygenators, extracorporeal oxygenator tubing and accessories.
5.3.5 Blood Path, Direct, Short Term, or Chronic, or repeated exposure—Examples include dialyzers or dialysis tubing and
accessories, shunts.
5.4 Implanted Long-Term Devices:
5.4.1 Devices Principally Contacting Bones—examples includdinclude orthopedic pins, screws, replacement joints, bone
prostheses, cements, orand dental implants.
5.4.2 Devices Principally Residing in the Subcutaneous Space—examples include pacemakers, neuromuscular stimulators,
facial augmentation devices, tissue expander devices, and breast prostheses.
5.4.3 Devices Principally Contacting Soft Tissue and Tissue Fluids—examples include drug supply devices, neuromuscular
sensors, replacement tendons, penile, and other implants, cerebrospinal fluid drains, artificial larynx, vas deferens valves, orand
ligation clips.
5.4.4 Devices Principally Contacting Blood—examples include pacemaker leads, artificial arteriovenous fistulae, heart valves,
vascular grafts, stents, blood monitors, internal drug delivery catheters, orand ventricular assist pumps.
6. Selection of Test Procedures
6.1 General: General—When it is determined that biocompatibility testing is needed, the following should be considered:
6.1.1 Biocompatibility testing involves tests of either the material itself, or an extract from it (see Practice F619), or both,
depending on the nature of the end-use application. While this practice does not address specific chemical methods for evaluating
the extractable substances or residuals from implant materials, several of the recommended tests (see 6.2, 6.76.3, 6.6, and 6.36.7)
utilize extracts rather than the original material for testing. If sensitive chemical assay techniques (such as GC, HPLC, and AA)
should reveal no detectable substances being extracted into the medium, consideration may be given to deletion of these tests from
the test battery. The investigator is cautioned, however, that the detection limit of the analytical chemistry procedures may not be
adequate to detect trace extractables that may generate a tissue response. Before analysis of extracts is substituted for actual
biocompatibility testing of the extracts, validation procedures may b
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...