IEC 60601-2-27:2011/COR1:2012
(Corrigendum)Corrigendum 1 - Medical electrical equipment - Part 2-27: Particular requirements for the basic safety and essential performance of electrocardiographic monitoring equipment
Corrigendum 1 - Medical electrical equipment - Part 2-27: Particular requirements for the basic safety and essential performance of electrocardiographic monitoring equipment
Corrigendum 1 - Appareils électromédicaux - Partie 2-27: Exigences particulières pour la sécurité de base et les performances essentielles des appareils de surveillance d'électrocardiographie
General Information
- Status
- Published
- Publication Date
- 15-May-2012
- Technical Committee
- SC 62D - Particular medical equipment, software, and systems
- Current Stage
- PPUB - Publication issued
- Start Date
- 16-May-2012
- Completion Date
- 30-Apr-2012
Relations
- Effective Date
- 05-Sep-2023
IEC 60601-2-27:2011/COR1:2012 - Corrigendum 1 - Medical electrical equipment - Part 2-27: Particular requirements for the basic safety and essential performance of electrocardiographic monitoring equipment Released:5/16/2012
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
IEC 60601-2-27:2011/COR1:2012 is a standard published by the International Electrotechnical Commission (IEC). Its full title is "Corrigendum 1 - Medical electrical equipment - Part 2-27: Particular requirements for the basic safety and essential performance of electrocardiographic monitoring equipment". This standard covers: Corrigendum 1 - Medical electrical equipment - Part 2-27: Particular requirements for the basic safety and essential performance of electrocardiographic monitoring equipment
Corrigendum 1 - Medical electrical equipment - Part 2-27: Particular requirements for the basic safety and essential performance of electrocardiographic monitoring equipment
IEC 60601-2-27:2011/COR1:2012 is classified under the following ICS (International Classification for Standards) categories: 11.040.50 - Radiographic equipment. The ICS classification helps identify the subject area and facilitates finding related standards.
IEC 60601-2-27:2011/COR1:2012 has the following relationships with other standards: It is inter standard links to IEC 60601-2-27:2011. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
IEC 60601-2-27:2011/COR1:2012 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
IEC 60601-2-27
(Third edition – 2011)
MEDICAL ELECTRICAL EQUIPMENT –
Part 2-27: Particular requirements for the basic safety and essential
performance of electrocardiographic monitoring equipment
CO RRI G E NDUM 1
Title
The correction applies to the French text only.
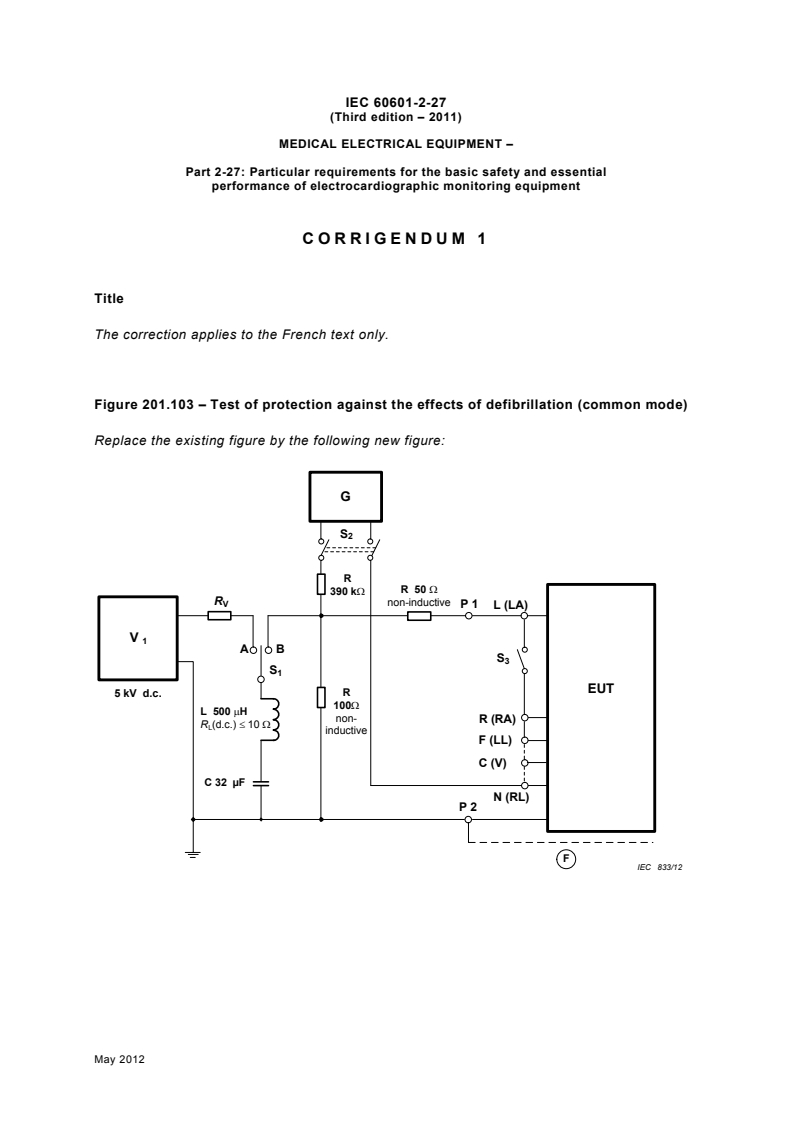
Figure 201.103 – Test of protection against the effects of defibrillation (common mode)
Replace the existing figure by the following new figure:
G
S
R
R 50 Ω
390 kΩ
R non-inductive
V
P 1 L (LA)
V
A B
S
S
EUT
R
5 kV d.c.
100Ω
L 500 µH
non-
R (RA)
R (d.c.) ≤ 10 Ω
L
inductive
F (LL)
C (V)
C 32 µF
N (RL)
P 2
F
IEC 833/12
May 2012
Components
G Sine wave generator 20 V peak-to-valley of 10 Hz
V
High voltage source 5 kV d.c.
Foil, simulating capacitance for CLASS II EQUIPMENT
Ⓕ
S
Switch; max. load 60 A, 5 kV
S
Switch connecting the signal source, 5 kV
S
Switch applying the signal source to LEAD WIRES
R
d.c. resistance of inductance L
L
R
Current limiting resistor
V
P1 Connecting point for EUT (includes PATIENT CABLES)
Connecting point for FUNCTIONAL EARTH TERMINAL and/or metal
P2
foil in contact with ENCLOSURE
Test to be conducted with MANUFACTURER’S recommended PATIENT CABLE and LEAD WIRES.
Figure 201.103 – Test of protection against the effects of defibrillation (common mode)
(see 201.8.5.5.1)
Figure 201.105 – General test circuit
Delete the symbol "F" between the lower part of the schematic and the legend beginning with
"Components".
201.12.1.101.15 * Heart rate range, accuracy, and QRS detection range
Replace the last sentence of the second paragraph with the following:
ECG input signals at rates above the upper limit of the specified display range, up to
300 1/min for adults and 350 1/min for neonatal and paediatric use shall not detect heart rates
lower than the specified upper limits.
_____________
May 2012
CEI 60601-2-27
(Troisième édition – 2011)
APPAREILS ÉLECTROMÉDICAUX –
Partie 2-27: Exigences particulières pour la sécurité de base et les performances
essentielles des appareils de surveillance d’électrocardiographie
CORRIGENDUM 1
Titre
Remplacer, sur la page de couvertu
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...