ASTM E1787-16
(Test Method)Standard Test Method for Anions in Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide) by Ion Chromatography
Standard Test Method for Anions in Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide) by Ion Chromatography
SIGNIFICANCE AND USE
4.1 Anion impurities in caustic soda and caustic potash are monitored by manufacturers and users for quality control of the products. Anions of primary interest are chloride, chlorate, and sulfate. This test method has determined precision estimates only for these three impurities.
SCOPE
1.1 This test method covers the determination of anionic impurities in 50 % caustic soda (sodium hydroxide) and 50 % caustic potash (potassium hydroxide) solutions using ion chromatography (IC). Anions that can be determined at concentrations of approximately 0.1 to 1000 ug/g (ppm) include: bromide, chlorate, chloride, fluoride, nitrate, phosphate, and sulfate.
1.2 By varying the sample size, this test method can be used for anhydrous caustic soda and caustic potash products, as well as other concentrations of liquid products.
1.3 This test method is not intended to be used to quantify chloride in caustic soda where the sodium chloride concentration is approximately 1 %. For the most accurate determinations, it is recommended that high concentrations of chloride be analyzed using a potentiometric titration procedure, such as the one described in Test Methods E291.
1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only.
1.5 Review the current appropriate Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 8.
General Information
- Status
- Published
- Publication Date
- 14-May-2016
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.12 - Caustics and Peroxides
Relations
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM E1787-16: Standard Test Method for Anions in Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide) by Ion Chromatography provides a robust procedure for detecting and quantifying anionic impurities in caustic soda (NaOH) and caustic potash (KOH) solutions. Using ion chromatography (IC), this method enables manufacturers and users to perform effective quality control for these widely used industrial chemicals. The principal anions detected include chloride, chlorate, and sulfate, with precision estimates determined for these critical impurities. The method is applicable for sample concentrations ranging from approximately 0.1 to 1000 µg/g (ppm) for a range of anions.
Key Topics
- Anionic Impurities Covered: The standard method addresses the analysis of bromide, chlorate, chloride, fluoride, nitrate, phosphate, and sulfate in caustic soda and caustic potash solutions.
- Applicable Concentrations: By adjusting sample size, the test is suitable for 50% solutions, other liquid concentrations, and anhydrous forms of NaOH and KOH.
- Analytical Technique: Ion chromatography is utilized to separate, detect, and quantify anionic species, offering high sensitivity and selectivity.
- Quality Control: Monitoring chloride, chlorate, and sulfate is essential for ensuring product purity required both during manufacturing and by end users.
- Limitations: The method is not recommended for quantifying chloride when sodium chloride is present at roughly 1% concentration; alternative techniques, such as potentiometric titration per ASTM E291, are suggested in such cases.
- Precision and Accuracy: The standard sets guidelines for acceptable repeatability and laboratory precision in testing, especially for chloride, chlorate, and sulfate.
- Safety Considerations: The procedure outlines safety precautions due to the caustic nature of the chemicals involved, recommending review of current Safety Data Sheets (SDS).
Applications
ASTM E1787-16 is critical in multiple scenarios involving caustic soda and caustic potash:
- Industrial Production: Ensuring raw caustic materials meet strict purity specifications to avoid downstream process disruptions or product contamination.
- Quality Assurance Laboratories: Routine impurity profiling to demonstrate compliance with internal or regulatory standards.
- Chemical Analysis: Research and development settings where precise detection of anions supports process optimization, troubleshooting, or new product development.
- Environmental and Safety Compliance: Verification of anionic impurities contributes to adherence to environmental discharge, workplace safety, and product labeling regulations.
- Process Troubleshooting: Diagnosing potential issues arising from anionic contamination in processes utilizing sodium hydroxide or potassium hydroxide.
Related Standards
- ASTM E291: Test Methods for Chemical Analysis of Caustic Soda and Caustic Potash, particularly recommended for high chloride concentrations.
- ASTM D1193: Specification for Reagent Water, ensuring the use of high-purity water for sample preparation and analysis.
- ASTM E180: Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals (withdrawn).
- Other Ion Chromatography Standards: Additional ASTM, ISO, or industry standards governing chromatographic determination of ions in industrial chemicals.
Keywords: ASTM E1787-16, ion chromatography, anions, caustic soda, caustic potash, sodium hydroxide, potassium hydroxide, chloride, chlorate, sulfate, quality control, chemical analysis, industrial chemicals, impurity detection.
Buy Documents
ASTM E1787-16 - Standard Test Method for Anions in Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide) by Ion Chromatography
REDLINE ASTM E1787-16 - Standard Test Method for Anions in Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide) by Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E1787-16 is a standard published by ASTM International. Its full title is "Standard Test Method for Anions in Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide) by Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 4.1 Anion impurities in caustic soda and caustic potash are monitored by manufacturers and users for quality control of the products. Anions of primary interest are chloride, chlorate, and sulfate. This test method has determined precision estimates only for these three impurities. SCOPE 1.1 This test method covers the determination of anionic impurities in 50 % caustic soda (sodium hydroxide) and 50 % caustic potash (potassium hydroxide) solutions using ion chromatography (IC). Anions that can be determined at concentrations of approximately 0.1 to 1000 ug/g (ppm) include: bromide, chlorate, chloride, fluoride, nitrate, phosphate, and sulfate. 1.2 By varying the sample size, this test method can be used for anhydrous caustic soda and caustic potash products, as well as other concentrations of liquid products. 1.3 This test method is not intended to be used to quantify chloride in caustic soda where the sodium chloride concentration is approximately 1 %. For the most accurate determinations, it is recommended that high concentrations of chloride be analyzed using a potentiometric titration procedure, such as the one described in Test Methods E291. 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only. 1.5 Review the current appropriate Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 8.
SIGNIFICANCE AND USE 4.1 Anion impurities in caustic soda and caustic potash are monitored by manufacturers and users for quality control of the products. Anions of primary interest are chloride, chlorate, and sulfate. This test method has determined precision estimates only for these three impurities. SCOPE 1.1 This test method covers the determination of anionic impurities in 50 % caustic soda (sodium hydroxide) and 50 % caustic potash (potassium hydroxide) solutions using ion chromatography (IC). Anions that can be determined at concentrations of approximately 0.1 to 1000 ug/g (ppm) include: bromide, chlorate, chloride, fluoride, nitrate, phosphate, and sulfate. 1.2 By varying the sample size, this test method can be used for anhydrous caustic soda and caustic potash products, as well as other concentrations of liquid products. 1.3 This test method is not intended to be used to quantify chloride in caustic soda where the sodium chloride concentration is approximately 1 %. For the most accurate determinations, it is recommended that high concentrations of chloride be analyzed using a potentiometric titration procedure, such as the one described in Test Methods E291. 1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only. 1.5 Review the current appropriate Safety Data Sheets (SDS) for detailed information concerning toxicity, first aid procedures, and safety precautions. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 8.
ASTM E1787-16 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis; 71.060.40 - Bases. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1787-16 has the following relationships with other standards: It is inter standard links to ASTM D1193-06, ASTM D1193-99e1, ASTM D1193-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1787-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1787 − 16
Standard Test Method for
Anions in Caustic Soda and Caustic Potash (Sodium
Hydroxide and Potassium Hydroxide) by Ion
Chromatography
This standard is issued under the fixed designation E1787; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.1 This test method covers the determination of anionic
impurities in 50% caustic soda (sodium hydroxide) and 50%
2. Referenced Documents
caustic potash (potassium hydroxide) solutions using ion
2.1 ASTM Standards:
chromatography (IC). Anions that can be determined at con-
D1193Specification for Reagent Water
centrations of approximately 0.1 to 1000 ug/g (ppm) include:
E180Practice for Determining the Precision of ASTM
bromide, chlorate, chloride, fluoride, nitrate, phosphate, and
Methods for Analysis and Testing of Industrial and Spe-
sulfate.
cialty Chemicals (Withdrawn 2009)
1.2 Byvaryingthesamplesize,thistestmethodcanbeused
E291Test Methods for Chemical Analysis of Caustic Soda
foranhydrouscausticsodaandcausticpotashproducts,aswell
and Caustic Potash (Sodium Hydroxide and Potassium
as other concentrations of liquid products.
Hydroxide)
1.3 This test method is not intended to be used to quantify
3. Summary of Test Method
chloride in caustic soda where the sodium chloride concentra-
tion is approximately 1 %. For the most accurate
3.1 Bromide, chlorate, chloride, fluoride, nitrate, phosphate
determinations, it is recommended that high concentrations of
and sulfate are measured in NaOH or KOH by ion chromatog-
chloridebeanalyzedusingapotentiometrictitrationprocedure,
raphy. The sample solution is diluted and injected onto a
such as the one described in Test Methods E291.
sample loop of an automated neutralization module. The
sample in the loop is pumped to a suppressor device (electro-
1.4 The values stated in SI units are to be regarded as
lyticallyorchemicallyregenerated)toneutralizethehydroxide
standard. The values given in parentheses are for information
ions. Anionic constituents of the neutralized sample are con-
only.
centrated on an anion concentrator column. After the concen-
1.5 Review the current appropriate Safety Data Sheets
tration they are separated into individual elution bands in the
(SDS) for detailed information concerning toxicity, first aid
eluent on a separator column.The conductivity of the eluent is
procedures, and safety precautions.
reduced with an anion suppression device, and the anions of
1.6 This standard does not purport to address all of the
interestaredetectedusingaconductivitydetector.Quantitation
safety concerns, if any, associated with its use. It is the
of the anions in the sample solution is achieved by calibrating
responsibility of the user of this standard to establish appro-
the IC with a series of standards containing known amounts of
priate safety, health, and environmental practices and deter-
the anions. These standards are also passed through the
mine the applicability of regulatory limitations prior to use.
neutralization device.
Specific hazards statements are given in Section 8.
4. Significance and Use
1.7 This international standard was developed in accor-
dance with internationally recognized principles on standard-
4.1 Anion impurities in caustic soda and caustic potash are
ization established in the Decision on Principles for the
monitoredbymanufacturersandusersforqualitycontrolofthe
Development of International Standards, Guides and Recom-
products.Anionsofprimaryinterestarechloride,chlorate,and
1 2
This test method is under the jurisdiction of ASTM Committee D16 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi- contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
bility of Subcommittee D16.12 on Caustics and Peroxides. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved May 15, 2016. Published May 2016. Originally the ASTM website.
approved in 1996. Last previous edition approved in 2008 as E1787– 08. DOI: The last approved version of this historical standard is referenced on
10.1520/E1787-16. www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1787 − 16
sulfate. This test method has determined precision estimates specifications are available. Other grades may be used pro-
only for these three impurities. vided it is first ascertained that the reagent is of sufficiently
high purity to permit its use without lessening the accuracy of
5. Interferences the determination.
7.2 Purity of Water—References to water meansType 1 (18
5.1 Substances that coelute with the anions of interest will
MΩ-cm deionized water) conforming to Specification D1193.
interfere.Ahigh concentration of one anion can interfere with
accurate quantitation of another anion if their retention times
7.3 Anion Stock Standards, 1000 ug/g (ppm):
are close and resolution is affected. For example, caustic soda
7.3.1 Bromide Stock Solution (1.00 mL = 1.00 mg
samples containing large concentrations of chloride can inter-
Bromide)—Dry sodium bromide (NaBr) for6hat 150°C and
fere with the quantitation of small amounts of fluoride.
cool in a desiccator. Dissolve 1.288 g of the dried NaBr in
Selection of a high capacity anion separator column will
water, dilute to 1 L with water, and mix well.
minimize this problem.
7.3.2 Chlorate Stock Solution (1.00 mL = 1.00 mg
Chlorate)—Dissolve 1.275 g of sodium chlorate (NaClO)in
5.2 Organic acids, surfactants, dyes, metals, etc., can cause
water, dilute to 1 L with water, and mix well.
fouling of the columns and membranes used in this test
7.3.3 Chloride Stock Solution (1.00 mL = 1.00 mg
method, resulting in interferences and decreased sensitivity. It
Chloride)—Dry sodium chloride (NaCl) for1hat 100°C and
is very important to follow the manufacturer’s recommenda-
cool in a desiccator. Dissolve 1.648 g of the dried NaCl in
tions for cleaning and maintaining the various parts of the IC
water, dilute to 1 L with water, and mix well.
system.
7.3.4 Fluoride Stock Solution (1.00 mL = 1.00 mg
5.3 The anion concentrator column in the neutralization
Fluoride)—Drysodiumfluoride(NaF)at105°Cforatleast8h
module has a finite capacity for trapping anions. Consult with
and cool in a desiccator. Dissolve 2.210 g of the dried NaF in
manufacturer for selection of concentrator column of suitable
500 mL of water, dilute to 1 L with water, and mix well.
capacity.Careshouldbetakennottoexceedthecapacityofthe
7.3.5 Nitrate Stock Solution (1.00 mL = 1.00 mg Nitrate)—
column. When the capacity of the column is exceeded, the
Dry sodium nitrate (NaNO ) at 105°C for 48 h and cool in a
stripping of anions will not be quantitative.
desiccator. Dissolve 1.371 g of the dried NaNO in water,
dilute to 1 L with water, and mix well.
6. Apparatus
7.3.6 Phosphate Stock Solution (1.00 mL = 1.00 mg
6.1 Ion Chromatograph, equipped with: Phosphate)—Dissolve 1.433 g of potassium dihydrogen phos-
6.1.1 Conductivity Detector, phate (KH PO ) in water, dilute to 1 L with water, and mix
2 4
well.
6.1.2 Anion Separator Column,
7.3.7 Sulfate Stock Solution (1.00 mL = 1.00 mg Sulfate)—
6.1.3 Guard Column,
Dry sodium sulfate (Na SO)for1hat 105°C and cool in a
6.1.4 100-µL Sample Loop,othersizesarepermittedsolong 2 4
desiccator. Dissolve 1.479 g of the Na SO in water, dilute to
as they do not degrade the precision of the method,
2 4
1 L with water, and mix well.
6.1.5 Autoneutralization Device, capable of neutralizing the
caustic sample prior to being directed through the separator
7.4 Eluent—The eluent used for the anion analysis will
column,
depend on the choice of separator column selected. Follow the
6.1.6 Post-Column Chemical Suppression Device, capable
manufacturer’s instructions for preparation of the eluent.
of reducing background conductivity due to the eluent, and
7.4.1 Eluent Concentrate, 0.27 mol/L (M) Sodium
6.1.7 Data Acquisition System, such as an integrator or Carbonate/0.03 mol/L (M) Sodium Bicarbonate (100 ×
computer system.
Concentrate)—Dissolve28.6gofsodiumcarbonateand2.52g
ofsodiumbicarbonateina1-Lvolumetricflaskcontaining800
6.2 100-mL Volumetric Flasks, for preparing sample solu-
mL of water. Dilute to volume with water and mix. Store in a
tions.
tightly capped polypropylene bottle.
6.3 Disposable 10-mL Syringes, for injecting solution into
7.4.2 Eluent, 2.7 mmol/L (mM) Sodium Carbonate/0.3
the IC.
mmol/L (mM) Sodium Bicarbonate Eluent—Pipet 20.0 mL of
6.3.1 IC Autosampler (optional), can be used as an alterna-
the eluent concentrate into a 2-Lvolumetric flask, dilute to the
tive to manually injecting samples.
mark with water, and mix.
7. Reagents
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
7.1 Purity of Reagents—Reagent grade chemicals should be
listed by the American Chemical Society, see Analar Standards for Laboratory
used in all tests. Unless otherwise indicated, all reagents must
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
conform to the specifications of the Committee on Analytical
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USP), Rockville,
Reagents of the American Chemical Society, where such MD.
E1787 − 16
8. Hazards
8.1 Sodium and potassium hydroxides are caustic alkalies,
which in their anhydrous or strong solution form, are hazard-
ous materials. In contact with the skin they produce burns that
may be quite serious unless promptly treated. Their action is
insidious since they produce no immediate stinging or burning
sensation, and damage may result before their presence is
realized.
8.2 Eyes are particularly vulnerable to severe damage from
these alkalies.
8.3 Use safety goggles or face shields and rubber gloves
when handling these alkalies, and avoid spillage on clothing.
These materials rapidly attack wool and leather.
8.4 Flushawayspilledcausticwithwaterwherepossible,or
coverwithabsorbentmaterial(suchassawdust,vermiculite,or
baking soda), and sweep up and discard in accordance with all
federal, state, and local health and environmental regulations.
Last traces may be neutralized with dilute acetic acid and the
area washed with water.
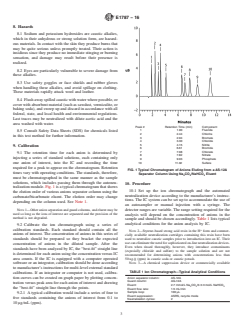
Peak # Retention Time (min) Component
1 1.90 Fluoride
8.5 Consult Safety Data Sheets (SDS) for chemicals listed
2 2.44 Chlorite
in this test method for further information.
3 2.94 Bromate
4 3.30 Chloride
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1787 − 08 E1787 − 16
Standard Test Method for
Anions in Caustic Soda and Caustic Potash (Sodium
Hydroxide and Potassium Hydroxide) by Ion
Chromatography
This standard is issued under the fixed designation E1787; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of anionic impurities in 50 % caustic soda (sodium hydroxide) and 50 % caustic
potash (potassium hydroxide) solutions using ion chromatography (IC). Anions that can be determined at concentrations of
approximately 0.1–1000 0.1 to 1000 ug/g (ppm) include: bromide, chlorate, chloride, fluoride, nitrate, phosphate, and sulfate.
1.2 By varying the sample size, this test method can be used for anhydrous caustic soda and caustic potash products, as well
as other concentrations of liquid products.
1.3 This test method is not intended to be used to quantify chloride in caustic soda where the sodium chloride concentration
is approximately 1 %. For the most accurate determinations, it is recommended that high concentrations of chloride be analyzed
using a potentiometric titration procedure, such as the one described in Test Methods E291.
1.4 The values stated in SI units are to be regarded as standard. The values given in parentheses are for information only.
1.5 Review the current appropriate Material Safety Data Sheets (MSDS)(SDS) for detailed information concerning toxicity, first
aid procedures, and safety precautions.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. Specific hazards statements are given in Section 8.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
E291 Test Methods for Chemical Analysis of Caustic Soda and Caustic Potash (Sodium Hydroxide and Potassium Hydroxide)
3. Summary of Test Method
3.1 Bromide, chlorate, chloride, fluoride, nitrate, phosphate and sulfate are measured in NaOH or KOH by ion chromatography.
The sample solution is diluted and injected onto a sample loop of an automated neutralization module. The sample in the loop is
pumped to a suppression device which uses electrolysis suppressor device (electrolytically or chemically regenerated) to neutralize
the hydroxide ions. The sample then is circulated through this device several times until it is completely neutralized. Anionic
constituents of the neutralized sample are concentrated on an anion concentrator column. After the concentration they are separated
into individual elution bands in the eluent on a separator column. The conductivity of the eluent is reduced with an anion
suppression device, and the anions of interest are detected using a conductivity detector. Quantitation of the anions in the sample
solution is achieved by calibrating the IC with a series of standards containing known amounts of the anions. These standards are
also passed through the neutralization device.
This test method is under the jurisdiction of ASTM Committee E15 on Industrial and Specialty Chemicals and is the direct responsibility of Subcommittee E15.01 on
General Standards.
Current edition approved April 1, 2008May 15, 2016. Published May 2008May 2016. Originally publishedapproved in 1996. Last previous edition approved in 20022008
as E1787-02.-08. DOI: 10.1520/E1787-08.10.1520/E1787-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1787 − 16
4. Significance and Use
4.1 Anion impurities in caustic soda and caustic potash are monitored by manufacturers and users for quality control of the
products. Anions of primary interest are chloride, chlorate, and sulfate. This test method has determined precision estimates only
for these three impurities.
5. Interferences
5.1 Substances that coelute with the anions of interest will interfere. A high concentration of one anion can interfere with
accurate quantitation of another anion if their retention times are close and resolution is affected. For example, caustic soda samples
containing large concentrations of chloride can interfere with the quantitation of small amounts of fluoride. Selection of a high
capacity anion separator column will minimize this problem.
5.2 Organic acids, surfactants, dyes, metals, etc., can cause fouling of the columns and membranes used in this test method,
resulting in interferences and decreased sensitivity. It is very important to follow the manufacturer’s recommendations for cleaning
and maintaining the various parts of the IC system.
5.3 The anion concentrator column in the neutralization module has a finite capacity for trapping anions, approximately 25
micro-equivalents per anions. Consult with manufacturer for selection of concentrator column of suitable capacity. Care should be
taken not to exceed the capacity of the column. When the capacity of the column is exceeded, the stripping of anions will not be
quantitative.
6. Apparatus
6.1 Ion Chromatograph, equipped with:
6.1.1 Conductivity Detector, Conductivity Detector,
6.1.2 Anion Separator Column, Anion Separator Column,
6.1.3 Guard Column, Guard Column,
6.1.4 100-μL Sample Loop, 100-μL Sample Loop, other sizes are permitted so long as they do not degrade the precision of the
method,
6.1.5 Autoneutralization Device, capable of neutralizing the caustic sample prior to being directed through the separator column,
6.1.6 Post-Column Chemical Suppression Device, capable of reducing background conductivity due to the eluent, and
6.1.7 Data Acquisition System, such as an integrator or computer system.
6.2 100-mL Volumetric Flasks, for preparing sample solutions.
6.3 Disposable 10-mL Syringes, for injecting solution into the IC.
6.3.1 IC Autosampler (optional), can be used as an alternative to manually injecting samples.
7. Reagents
7.1 Purity of Reagents—Reagent grade chemicals should be used in all tests. Unless otherwise indicated, all reagents must
conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where such
specifications are available. Other grades may be used provided it is first ascertained that the reagent is of sufficiently high purity
to permit its use without lessening the accuracy of the determination.
7.2 Purity of Water—References to water means Type 1 (18 MΩ-cm deionized water) conforming to Specification D1193.
7.3 Anion Stock Standards, 1000 ug/g (ppm):
7.3.1 Bromide Stock Solution (1.00 mL = 1.00 mg Bromide)—Dry sodium bromide (NaBr) for 6 h at 150°C and cool in a
desiccator. Dissolve 1.288 g of the dried NaBr in water, dilute to 1 L with water, and mix well.
7.3.2 Chlorate Stock Solution (1.00 mL = 1.00 mg Chlorate)—Dissolve 1.275 g of sodium chlorate (NaClO ) in water, dilute
to 1 L with water, and mix well.
7.3.3 Chloride Stock Solution (1.00 mL = 1.00 mg Chloride)—Dry sodium chloride (NaCl) for 1 h at 100°C and cool in a
desiccator. Dissolve 1.648 g of the dried NaCl in water, dilute to 1 L with water, and mix well.
7.3.4 Fluoride Stock Solution (1.00 mL = 1.00 mg Fluoride)—Dry sodium fluoride (NaF) at 105°C for at least 8 h 8 h and cool
in a desiccator. Dissolve 2.210 g of the dried NaF in 500 mL of water, dilute to 1 L with water, and mix well.
7.3.5 Nitrate Stock Solution (1.00 mL = 1.00 mg Nitrate)—Dry sodium nitrate (NaNO ) at 105°C for 48 h and cool in a
desiccator. Dissolve 1.371 g of the dried NaNO in water, dilute to 1 L with water, and mix well.
7.3.6 Phosphate Stock Solution (1.00 mL = 1.00 mg Phosphate)—Dissolve 1.433 g of potassium dihydrogen phosphate
(KH PO ) in water, dilute to 1 L with water, and mix well.
2 4
Reagent Chemicals, American Chemical Society Specifications, , American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),(USP), Rockville, MD.
E1787 − 16
7.3.7 Sulfate Stock Solution (1.00 mL = 1.00 mg Sulfate)—Dry sodium sulfate (Na SO ) for 1 h at 105°C and cool in a
2 4
desiccator. Dissolve 1.479 g of the Na SO in water, dilute to 1 L with water, and mix well.
2 4
7.4 Eluent—The eluent used for the anion analysis will depend on the choice of separator column selected. The eluent described
below is used with the Dionex AS-12A separator column.Follow the manufacturer’s instructions for preparation of the eluent.
7.4.1 Eluent Concentrate, 0.27 mol/L (M) Sodium Carbonate/0.03 mol/L (M) Sodium Bicarbonate (100× (100 × Concentrate)—
Dissolve 28.6 g of sodium carbonate and 2.52 g of sodium bicarbonate in a 1- L 1-L volumetric flask containing 800 mL of water.
Dilute to volume with water and mix. Store in a tightly capped polypropylene bottle.
7.4.2 Eluent, 2.7 mmol/L (mM) Sodium Carbonate/0.3 mmol/L (mM) Sodium Bicarbonate Eluent—Pipet 20.0 mL of the eluent
concentrate into a 2-L volumetric flask, dilute to the mark with water, and mix.
8. Hazards
8.1 Sodium and potassium hydroxides are caustic alkalies, which in their anhydrous or strong solution form, are hazardous
materials. In contact with the skin they produce burns that may be quite serious unless promptly treated. Their action is insidious
since they produce no immediate stinging or burning sensation, and damage may result before their presence is realized.
8.2 Eyes are particularly vulnerable to severe damage from these alkalies.
8.3 Use safety goggles or face shields and rubber gloves when handling these alkalies, and avoid spillage on clothing. These
materials rapidly attack wool and leather.
8.4 Flush away spilled caustic with water where possible, or cover with absorbent material (such as sawdust, vermiculite, or
baking soda), and sweep up and discard in accordance with all federal, state, and local health and environmental regulations. Last
traces may be neutralized with dilute acetic acid and the area washed with water.
8.5 Consult Material Safety Data Sheets (MSDS)(SDS) for chemicals listed in this test method for further information.
9. Calibration
9.1 The retention time for each anion is determined by injecting a series of standard solutions, each containing only one anion
of interest, into the IC and recording the time required for a peak to appear on the chromatogram. Retention times vary with
operating conditions. The standards, therefore, must be chromatographed
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...