ASTM F3369-19e1
(Guide)Standard Guide for Assessing the Skeletal Myoblast Phenotype
Standard Guide for Assessing the Skeletal Myoblast Phenotype
SIGNIFICANCE AND USE
5.1 This guide describes markers involved in myoblast differentiation that can be used to screen stem cells to help define myogenic capacity. Stem cells include pluripotent and multipotent stem cells capable of differentiating into several different mesenchymal cells, including skeletal muscle myoblasts.
5.2 To assess myogenesis in cells derived and not derived from muscle, markers are measured to accurately define the changes in transcription and structural proteins that regulate differentiation, fusion, and myotube formation. Discussion of these markers is important to understand why they are recommended.
5.3 Myogenic Differentiation:
5.3.1 Myogenic differentiation is a highly regulated process controlled by paired box (Pax) transcription factors and the myogenic regulatory factor (MRF) family. During early differentiation in adults, myogenic progenitors such as activated satellite cells or myoblasts express Pax3 and Pax7. Pax3 and Pax7 transcription factors switch the cells toward a myogenic fate, and repress myocyte differentiation (2), priming the cell for later MRFs. To form muscle, the family of MRFs is required to terminally differentiate myoblasts and form myofibers. These regulatory proteins belong to a superfamily of basic helix-loop-helix transcription factors that consists of myogenic differentiation factor 1 (Myod1), myogenic factor 5 (Myf5), myogenin (Myog), and myogenic factor 6 (Myf6). In the initial stages of myogenic differentiation, Myod1 and Myf5 are the first MRFs to be expressed, and trigger increased production of Myog and Myf6 (3). Increased intracellular Myog and Myf6 induces terminal differentiation of myoblasts into myocytes, leading to fused myotubes.
5.4 Forming Myotubes:
5.4.1 While myogenic markers describe differentiation, fusion into multinucleated myotubes is an important factor in muscle biology. Myoblasts differentiate into a fusogenic phenotype characterized by multiple fusion markers. One marker of note is m-c...
SCOPE
1.1 Myogenic differentiation is a process regulated by specific transcription factors and signaling molecules that have been shown to induce a myogenic phenotype. Transcription factors mark the stages of myogenesis and act as benchmarks for use in myogenic assays.
1.2 This guide applies to mammalian cells but does not apply to non-mammalian cells as the myogenic markers for non-mammalian cells can be different than those described here.
1.3 This guide proposes appropriate markers to measure when conducting myogenic differentiation assays. This guide describes the stages for multipotent stem cell differentiation toward myoblasts and myotubes. This guide provides information about the appropriate methods to determine myogenic differentiation. This guide does not provide information about media, supplements, or substrates that drive differentiation toward a myogenic phenotype.
1.4 The purpose of this guide is to act as an aid for work performed in the area of skeletal myogenesis. Using this guide, researchers should be able to understand which skeletal muscle markers are best suited for experiments. This guide will improve consistency for studies of myogenic differentiation of multipotent stem cells by identifying appropriate markers for each stage leading to myocyte differentiation. It should be noted that myoblast differentiation in vitro may not be predictive of results that may be obtained in vivo.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Gu...
General Information
- Status
- Published
- Publication Date
- 31-Jan-2019
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.43 - Cells and Tissue Engineered Constructs for TEMPs
Relations
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Apr-2011
- Effective Date
- 01-Jun-2010
- Effective Date
- 01-Nov-2004
- Effective Date
- 10-Sep-2003
Overview
ASTM F3369-19e1: Standard Guide for Assessing the Skeletal Myoblast Phenotype is a vital reference for researchers and laboratories involved in skeletal muscle biology, stem cell differentiation, and tissue engineering. Developed by ASTM International, this guide defines the recommended markers and methodologies for evaluating myogenic differentiation in mammalian stem cells, specifically those with the potential to adopt a skeletal muscle cell fate. By establishing standardized criteria and assays for identifying the skeletal myoblast phenotype, ASTM F3369 enhances consistency, reproducibility, and confidence in studies related to muscle regeneration, stem cell therapy, and tissue-engineered constructs.
Key Topics
Myogenic Differentiation Markers: The guide outlines key transcription factors and structural proteins involved in myoblast differentiation:
- Paired box factors: Pax3 and Pax7, crucial in early muscle progenitors and satellite cells.
- Myogenic regulatory factors (MRFs): Myod1, Myf5, Myogenin (Myog), Myf6-important for muscle lineage commitment and differentiation.
- Structural and fusion proteins: Integrin α7, desmin, and m-cadherin, critical for myotube formation and muscle-specific structural identity.
Differentiation Assessment Methods:
- Gene expression: Quantitative PCR (qPCR) and RT-PCR to measure transcriptional activity of myogenic markers.
- Protein analysis: Immunoblotting and ELISA assays to assess production of key muscle proteins.
- Microscopy: Morphological assessment for myotube formation, cell fusion, and identification of malformed fibers.
Marker Categories:
- Primary markers: Regulate or are closely associated with myoblast differentiation and myotube formation.
- Secondary markers: Expressed in skeletal muscle but also found in other tissues, making them less specific for definitive identification.
Experimental Recommendations:
- Use well-established protocols and standardized conditions for data reliability.
- Interpret marker expression in the context of cell type and developmental stage; no single marker conclusively defines myogenic differentiation.
Applications
- Stem Cell Screening: Evaluate pluripotent and multipotent stem cells for their myogenic differentiation capacity, crucial for stem cell research and regenerative medicine.
- Muscle Regeneration Studies: Monitor and characterize the progression of satellite cells and myogenic progenitors in muscle repair and disease models.
- Tissue Engineering: Assess engineered constructs for skeletal muscle tissue, supporting development and quality control of cell-based therapies and medical devices.
- Basic Muscle Biology: Provide a framework for research on muscle cell lineage, differentiation pathways, and functional muscle development in vitro.
- Preclinical Testing: Facilitate consistent and reproducible protocols for in vitro assays prior to animal or clinical studies.
Related Standards
- ASTM F2312 – Terminology Relating to Tissue Engineered Medical Products: Offers definitions crucial for consistency in the field of tissue engineering.
- Additional standards may cover cell culture methods, scaffold materials, and other assays for tissue-engineered constructs.
Practical Value
Adhering to ASTM F3369-19e1 ensures that researchers use science-based, consensus-driven criteria for assessing skeletal myoblast phenotypes. This approach:
- Improves cross-study comparability and reproducibility.
- Facilitates regulatory and publication compliance by following international standardization principles.
- Supports innovation in muscle tissue engineering and regenerative medicine by providing a validated assessment strategy.
For professionals in cell biology, regenerative medicine, and tissue engineering, ASTM F3369-19e1 is an indispensable tool for robust and standardized evaluation of the skeletal myoblast phenotype.
Keywords: myoblasts, myogenic differentiation, stem cell assays, muscle regeneration, myotubes, tissue engineering, marker assessment.
Buy Documents
ASTM F3369-19e1 - Standard Guide for Assessing the Skeletal Myoblast Phenotype
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3369-19e1 is a guide published by ASTM International. Its full title is "Standard Guide for Assessing the Skeletal Myoblast Phenotype". This standard covers: SIGNIFICANCE AND USE 5.1 This guide describes markers involved in myoblast differentiation that can be used to screen stem cells to help define myogenic capacity. Stem cells include pluripotent and multipotent stem cells capable of differentiating into several different mesenchymal cells, including skeletal muscle myoblasts. 5.2 To assess myogenesis in cells derived and not derived from muscle, markers are measured to accurately define the changes in transcription and structural proteins that regulate differentiation, fusion, and myotube formation. Discussion of these markers is important to understand why they are recommended. 5.3 Myogenic Differentiation: 5.3.1 Myogenic differentiation is a highly regulated process controlled by paired box (Pax) transcription factors and the myogenic regulatory factor (MRF) family. During early differentiation in adults, myogenic progenitors such as activated satellite cells or myoblasts express Pax3 and Pax7. Pax3 and Pax7 transcription factors switch the cells toward a myogenic fate, and repress myocyte differentiation (2), priming the cell for later MRFs. To form muscle, the family of MRFs is required to terminally differentiate myoblasts and form myofibers. These regulatory proteins belong to a superfamily of basic helix-loop-helix transcription factors that consists of myogenic differentiation factor 1 (Myod1), myogenic factor 5 (Myf5), myogenin (Myog), and myogenic factor 6 (Myf6). In the initial stages of myogenic differentiation, Myod1 and Myf5 are the first MRFs to be expressed, and trigger increased production of Myog and Myf6 (3). Increased intracellular Myog and Myf6 induces terminal differentiation of myoblasts into myocytes, leading to fused myotubes. 5.4 Forming Myotubes: 5.4.1 While myogenic markers describe differentiation, fusion into multinucleated myotubes is an important factor in muscle biology. Myoblasts differentiate into a fusogenic phenotype characterized by multiple fusion markers. One marker of note is m-c... SCOPE 1.1 Myogenic differentiation is a process regulated by specific transcription factors and signaling molecules that have been shown to induce a myogenic phenotype. Transcription factors mark the stages of myogenesis and act as benchmarks for use in myogenic assays. 1.2 This guide applies to mammalian cells but does not apply to non-mammalian cells as the myogenic markers for non-mammalian cells can be different than those described here. 1.3 This guide proposes appropriate markers to measure when conducting myogenic differentiation assays. This guide describes the stages for multipotent stem cell differentiation toward myoblasts and myotubes. This guide provides information about the appropriate methods to determine myogenic differentiation. This guide does not provide information about media, supplements, or substrates that drive differentiation toward a myogenic phenotype. 1.4 The purpose of this guide is to act as an aid for work performed in the area of skeletal myogenesis. Using this guide, researchers should be able to understand which skeletal muscle markers are best suited for experiments. This guide will improve consistency for studies of myogenic differentiation of multipotent stem cells by identifying appropriate markers for each stage leading to myocyte differentiation. It should be noted that myoblast differentiation in vitro may not be predictive of results that may be obtained in vivo. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Gu...
SIGNIFICANCE AND USE 5.1 This guide describes markers involved in myoblast differentiation that can be used to screen stem cells to help define myogenic capacity. Stem cells include pluripotent and multipotent stem cells capable of differentiating into several different mesenchymal cells, including skeletal muscle myoblasts. 5.2 To assess myogenesis in cells derived and not derived from muscle, markers are measured to accurately define the changes in transcription and structural proteins that regulate differentiation, fusion, and myotube formation. Discussion of these markers is important to understand why they are recommended. 5.3 Myogenic Differentiation: 5.3.1 Myogenic differentiation is a highly regulated process controlled by paired box (Pax) transcription factors and the myogenic regulatory factor (MRF) family. During early differentiation in adults, myogenic progenitors such as activated satellite cells or myoblasts express Pax3 and Pax7. Pax3 and Pax7 transcription factors switch the cells toward a myogenic fate, and repress myocyte differentiation (2), priming the cell for later MRFs. To form muscle, the family of MRFs is required to terminally differentiate myoblasts and form myofibers. These regulatory proteins belong to a superfamily of basic helix-loop-helix transcription factors that consists of myogenic differentiation factor 1 (Myod1), myogenic factor 5 (Myf5), myogenin (Myog), and myogenic factor 6 (Myf6). In the initial stages of myogenic differentiation, Myod1 and Myf5 are the first MRFs to be expressed, and trigger increased production of Myog and Myf6 (3). Increased intracellular Myog and Myf6 induces terminal differentiation of myoblasts into myocytes, leading to fused myotubes. 5.4 Forming Myotubes: 5.4.1 While myogenic markers describe differentiation, fusion into multinucleated myotubes is an important factor in muscle biology. Myoblasts differentiate into a fusogenic phenotype characterized by multiple fusion markers. One marker of note is m-c... SCOPE 1.1 Myogenic differentiation is a process regulated by specific transcription factors and signaling molecules that have been shown to induce a myogenic phenotype. Transcription factors mark the stages of myogenesis and act as benchmarks for use in myogenic assays. 1.2 This guide applies to mammalian cells but does not apply to non-mammalian cells as the myogenic markers for non-mammalian cells can be different than those described here. 1.3 This guide proposes appropriate markers to measure when conducting myogenic differentiation assays. This guide describes the stages for multipotent stem cell differentiation toward myoblasts and myotubes. This guide provides information about the appropriate methods to determine myogenic differentiation. This guide does not provide information about media, supplements, or substrates that drive differentiation toward a myogenic phenotype. 1.4 The purpose of this guide is to act as an aid for work performed in the area of skeletal myogenesis. Using this guide, researchers should be able to understand which skeletal muscle markers are best suited for experiments. This guide will improve consistency for studies of myogenic differentiation of multipotent stem cells by identifying appropriate markers for each stage leading to myocyte differentiation. It should be noted that myoblast differentiation in vitro may not be predictive of results that may be obtained in vivo. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Gu...
ASTM F3369-19e1 is classified under the following ICS (International Classification for Standards) categories: 11.020.20 - Medical science; 11.100.99 - Other standards related to laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3369-19e1 has the following relationships with other standards: It is inter standard links to ASTM F2312-11(2020), ASTM F2312-11, ASTM F2312-10, ASTM F2312-04, ASTM F2312-03. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3369-19e1 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
´1
Designation: F3369 − 19
Standard Guide for
Assessing the Skeletal Myoblast Phenotype
This standard is issued under the fixed designation F3369; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
ε NOTE—Table 1 was reformatted in May 2019.
1. Scope Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 Myogenic differentiation is a process regulated by
Barriers to Trade (TBT) Committee.
specifictranscriptionfactorsandsignalingmoleculesthathave
been shown to induce a myogenic phenotype. Transcription
2. Referenced Documents
factors mark the stages of myogenesis and act as benchmarks
2.1 ASTM Standards:
for use in myogenic assays.
F2312Terminology Relating to Tissue Engineered Medical
1.2 This guide applies to mammalian cells but does not
Products
apply to non-mammalian cells as the myogenic markers for
non-mammalian cells can be different than those described
3. Terminology
here.
3.1 Unless provided otherwise in 3.2, terminology shall be
1.3 This guide proposes appropriate markers to measure
in conformance with Terminology F2312.
when conducting myogenic differentiation assays. This guide
3.2 Definitions:
describes the stages for multipotent stem cell differentiation
3.2.1 myoblasts, n—myoblasts repair or replace damaged
toward myoblasts and myotubes.This guide provides informa-
muscle fibers by differentiating and fusing either with an
tion about the appropriate methods to determine myogenic
existing muscle fiber or with each other to form multi-
differentiation. This guide does not provide information about
nucleated myotubes. Proliferating myoblasts are able to self-
media, supplements, or substrates that drive differentiation
renew, but lose that ability once they exit the mitotic cycle.
toward a myogenic phenotype.
3.2.2 myogenic cells, n—myogenic cells express a class of
1.4 The purpose of this guide is to act as an aid for work
markers that suggest a muscle cell lineage fate or a cell’s
performedintheareaofskeletalmyogenesis.Usingthisguide,
involvement in muscle fiber formation, or both.
researchersshouldbeabletounderstandwhichskeletalmuscle
3.2.3 myogenic differentiation, n—myogenic differentiation
markers are best suited for experiments. This guide will
refers to the normal process by which early muscle precursor
improve consistency for studies of myogenic differentiation of
cells and myoblasts become more specialized cells capable of
multipotent stem cells by identifying appropriate markers for
fusing with mature myoblasts or existing myotubes.
each stage leading to myocyte differentiation. It should be
Furthermore, the process of differentiation can be measured
noted that myoblast differentiation in vitro may not be predic-
using markers to help describe cell behavior during culture.
tive of results that may be obtained in vivo.
This guide will describe those different markers.
1.5 This standard does not purport to address all of the
3.2.4 myogenicterminaldifferentiation,n—myogenictermi-
safety concerns, if any, associated with its use. It is the
nal differentiation refers to the normal end stage of myoblast
responsibility of the user of this standard to establish appro-
maturation, where myoblasts can no longer proliferate and are
priate safety, health, and environmental practices and deter-
able to fuse into myotubes. Experimental data to quantify this
mine the applicability of regulatory limitations prior to use.
stage of differentiation is necessary in gaining additional
1.6 This international standard was developed in accor-
information regarding cell behavior during culture.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the 3.2.5 myotubes, n—myotubes are long tubular structures
with multiple nuclei, originating from mature myoblasts that
This guide is under the jurisdiction ofASTM Committee F04 on Medical and
Surgical Materials and Devices and is the direct responsibility of Subcommittee For referenced ASTM standards, visit the ASTM website, www.astm.org, or
F04.43 on Cells and Tissue Engineered Constructs for TEMPs. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved Feb. 1, 2019. Published March 2019. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
F3369-19E01. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
´1
F3369 − 19
fusedtogether.Nucleiarecentrallylocatedfollowingtheinitial 5. Significance and Use
fiber formation and as the myotube matures into a functional
5.1 This guide describes markers involved in myoblast
fiber the nuclei translocate to the periphery.
differentiation that can be used to screen stem cells to help
3.2.6 primary myotube, n—primary myotubes are multi-
define myogenic capacity. Stem cells include pluripotent and
nucleated muscle cells present early during development.
multipotent stem cells capable of differentiating into several
different mesenchymal cells, including skeletal muscle myo-
3.2.7 satellite cells, n—satellitecellsarenormallyquiescent
blasts.
cells that reside between the basement membrane and the
sarcolemma of muscle fibers. Satellite cells are activated by
5.2 To assess myogenesis in cells derived and not derived
extracellular cues associated with local damage. Once
from muscle, markers are measured to accurately define the
activated,satellitecellswillre-enterthecellcycletoproliferate
changes in transcription and structural proteins that regulate
and supply a population of progeny (myoblasts). Satellite cells
differentiation, fusion, and myotube formation. Discussion of
can adopt divergent cell fates that expand or maintain the
these markers is important to understand why they are recom-
satellite pool, or expand the number of myoblasts, or both.
mended.
Proliferatingmyoblastsareresponsibleformuscleregeneration
5.3 Myogenic Differentiation:
in vivo by fusing with other myoblasts or myotubes.
5.3.1 Myogenic differentiation is a highly regulated process
3.2.8 secondary myotube, n—seconary myotubes are multi-
controlled by paired box (Pax) transcription factors and the
nucleatedmusclecellspresentlaterinembryonicdevelopment
myogenic regulatory factor (MRF) family. During early differ-
that devleop in close apposition to primary myotubes.
entiation in adults, myogenic progenitors such as activated
3.2.9 stem cells, n—progenitor cells capable of self- satellite cells or myoblasts express Pax3 and Pax7. Pax3 and
Pax7 transcription factors switch the cells toward a myogenic
replication, proliferation, and differentiation (1).
fate, and repress myocyte differentiation (2), priming the cell
for later MRFs. To form muscle, the family of MRFs is
4. Summary of Guide
required to terminally differentiate myoblasts and form myo-
4.1 This guide consists of (1) recommended markers that
fibers. These regulatory proteins belong to a superfamily of
are good predictors of myogenic differentiation, (2) markers
basic helix-loop-helix transcription factors that consists of
that are not consistent predictors and are better used as
myogenic differentiation factor 1 (Myod1), myogenic factor 5
complementary data, and (3) common methods that will
(Myf5), myogenin (Myog), and myogenic factor 6 (Myf6). In
enhance research results and data analysis such as real-time
the initial stages of myogenic differentiation, Myod1 and Myf5
quantitative reverse transcription polymerase chain reaction,
are the first MRFs to be expressed, and trigger increased
immunoblot, enzyme-linked immunosorbent assays, and mi-
productionof Myogand Myf6 (3).Increasedintracellular Myog
croscopy.
and Myf6 induces terminal differentiation of myoblasts into
4.2 This guide proposes a list of myogenic markers that
myocytes, leading to fused myotubes.
have demonstrated a clear role in myogenic differentiation and
5.4 Forming Myotubes:
muscle development both in vitro and in vivo.
5.4.1 While myogenic markers describe differentiation, fu-
4.3 Identifying markers in muscle development and func-
sion into multinucleated myotubes is an important factor in
tionwouldenhanceconfidencefollowingdataacquisition.This
muscle biology. Myoblasts differentiate into a fusogenic phe-
guide will separate markers into two categories: primary and
notype characterized by multiple fusion markers. One marker
secondary.
ofnoteism-cadherin.M-cadherinisreportedtobeinvolvedin
myoblast fusion and to regulate myotube development (4).
4.4 Primary markers regulate myoblast differentiation and
Therefore, assessment of fusion markers in addition to myo-
myotube formation.
genic differentiation markers would favor a cell phenotype
4.5 Secondary markers are expressed and involved in
capable of forming muscle. In support of this, studies have
muscle, but do not regulate myogenic differentiation, or are
shown that despite expression of myogenic differentiation
difficult to distinguish from other tissues that express them as
genes,cellsnotexpressingm-cadherinwereunabletofuseand
well.
form muscle. These results suggest that in addition to myo-
4.6 Methods used to measure and assess myogenic differ-
genic differentiation markers, fusion markers should be con-
entiationinstemcellandprogenitorcellculturesareaddressed
sideredgiventheirimportanceasindicatorsofwhetheracellis
in this guide, including (1) gene expression using polymerase
able to fuse (5). This guide will enumerate published methods
chain reaction (PCR); (2) protein assessment using immunob-
to measure and quantify myoblast fusion markers.
lot; and (3) imaging.
6. Primary Myogenic Markers
4.7 Expression of primary markers does not guarantee
NOTE 1—The markers discussed below are considered important
myoblast differentiation or myotube formation, and should not
regulators of a myogenic phenotype including satellite cell, myoblast,
mature myoblast, and myotube.
be considered entirely sufficient until proven experimentally.
6.1 Paired Box 3 (Pax3) and Paired Box 7 (Pax7):
6.1.1 In adult skeletal muscle, Pax3-positive and Pax7-
positive cells positioned adjacent to the myofiber and under-
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
this standard. neath the basal lamina are called satellite cells. These are
´1
F3369 − 19
important in muscle regeneration and hypertrophy, and studies assess the myogenic phenotype. In support of this, reports
have shown that Pax7-positive cells strongly influence muscle using hydrogels of different stiffness values to culture mesen-
regeneration (6). Pax7hasbeenshowntoplayacriticalrolein
chymal stem cells demonstrated an increase in other myogenic
adultskeletalmuscle;progressivelossofPax7andsatellitecell
regulatory factors, but were negative for Pax7. Lastly, when
fl/CreERT2
populations impaired muscle regeneration in Pax7
considering animal studies, spatial distribution of Pax7- and
mice (7). This role is supported by demonstration that in vitro
Pax3-positive cells should be carefully considered given their
siRNA-mediated suppression of Pax7 resulted in loss of Myf5
distribution in the body as noted above.
expression, a critical step in myogenesis (8). More, Pax7
6.2 MyogenicFactor5(Myf5)andMyogenicDifferentiation
transcription is an essential coactivator of other downstream
Factor 1 (Myod1):
muscle genes such as Myod1 (9), and was shown to be
sufficient for myogenic specification of muscle-derived stem
6.2.1 Expression of both Myod1 and Myf5 is a key step in
cells (10). Like Pax7, Pax3 is expressed in early muscle
commitment of multipotential somite cells to the myogenic
progenitor cells, and its absence causes severe muscle deficits.
lineage. Myf5 expression is induced in the dorsal-medial
Pax3 is one of the earliest markers expressed during muscle
somites and is followed by expression of Myod1.As Myod1
development and its expression continues in postnatal muscle,
expression increases, Myf5 expression Myod1 is reduced. In
but in specific locations (11). For instance, Pax3 β-galactoside
animal knockouts, neither Myod1 nor Myf5 knockout mice
positive cells were identified in mouse forelimbs but were
alone disrupt normal muscle development, but when Myod1
sparse in the hindlimbs, indicating that Pax3 is spatially
and Myf5 are deleted, muscle development is inhibited (12).
dependent.
ThesefindingsunderscorethespecificityofMyf5andMyod1in
6.1.2 Taken together, Pax3 and Pax7 could indicate a
themyogenicprogram,andhighlightstheirexpressioninearly
myogenic phenotype, and their level of expression should be
stagemyogeniccells.Itisforthisreasonthat Myod1and Myf5
taken in reference along with additional experimental data to
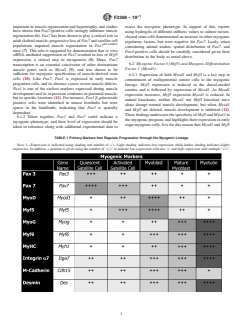
TABLE 1 Primary Markers that Regulate Progression through the Myogenic Lineage
NOTE 1—Expression is indicated using shading and number of +’s. Light shading indicates low expression while darker shading indicates higher
expression. In addition, a gradient is given using the number of “+’s” to indicate low expression with one ‘+’and high expression with multiple “+’s”.
´1
F3369 − 19
are used so often in myogenic assays to determine relative differentiating myocytes, located at the periphery of the Z-disk
expression of these genes. in muscle fibers and aids in muscle contraction. It’s expression
is required for functional skeletal muscle to be formed. The
6.2.2 Studies have also demonstrated that forced expression
first recognizable step in skeletal muscle myogenesis is the
of Myod1 induced a myoblast-like phenotype in non-muscle
initiation of desmin synthesis in replicating myoblasts (26).
cells. Myod1 is a master regulator (13) and functions by
Further, the amount and localization of desmin makes it an
activating a feed-forward loop to control myogenic gene
easy target to determine whether or not the differentiating cell
expression. Myod1 has been shown to promote expression and
possesses a skeletal myogenic phenotype (27).While there are
production of Myog and Myf6. To demonstrate importance,
studies that demonstrate desmin synthesis in cardiac and
Myod1hasbeentransfectedintomanynon-musclespecificcell
smooth muscle cells, smooth muscle desmin is located at the
typesandgeneratedexpressionofdownstreammyogenicgenes
dense bodies of the cells, while in cardiac and skeletal muscle
like myogenin (14, 15). While forced expression of Myod1
desmin is located at the Z-disk (28, 29). To determine differ-
caused transdifferentiation of non-muscle cells into myoblast-
ences between cardiac and skeletal muscle cells, the level of
like cells, non-muscle specific genes can still be expressed.
protein production can be examined. Skeletal muscle has
When evaluating stem cells or progenitor cells derived from
elevated levels of desmin when compared to cardiac muscle.
muscle and non-muscle sources, Myod1 characterization is
Lastly, it should be noted that mesenchymal stem cells also
recommended to ensure that a gene which regulates several
were reported to produce desmin filaments (30), suggesting
muscle pathways is considered. Likewise, Myf5 expression is
that close attention should be given to experimental data
also considered an important gene regulator and is recom-
following various differentiation and cell culture protocols.
meded as a potential marker to evaluate myogenic phenotype.
Desmin is a protein that if assessed improperly could be
6.3 Myogenin (Myog):
interpretedincorrectly.Thus,thisguiderecommendstheuseof
6.3.1 Myogenin is a key developmental regulator for skel-
desmin with Western blot or microscropy, or both, as an easy
etal muscle formation, is a marker of mature myoblasts, and
and effective way to distinguish between smooth, cardiac, and
while expression is reduced after muscle forms, it continues to
skeletal muscle cells.
beexpressedinfullymaturemusclefibers.Theroleof Myogis
6.6 M-Cadherin (Cdh15):
highlighted by the fact that knockout mice fail to form muscle
6.6.1 M-cadherin is a component of muscular adherens
during development (16, 17). This phenotype is consistent in
junctions and provides a physical interaction between myo-
cells that demonstrate reduced Myog expression and protein
blasts. When production of m-cadherin is suppressed, myo-
production,failingtofuseandformmyotubes (18).Inaddition,
blastspossessareducedabilitytofuse (31).Interestingly,when
loss of Myog during postnatal life still yields normal muscle
m-cadherin expression was deleted in mice, muscle still
development, but those developed fibers are significantly
developed due to alternative pathways available through other
smaller compared to normal mice (19). Together, these data
cadherins (32), indicating the overall importance of cadherins
indicate that Myog might be critical in early muscle precursor
in muscle fiber formation. Based on these reports, which
cells and stem cells to form myotubes, but its role and
demonstrate that m-cadherin is a muscle-specific protein that
downstream effect might change with maturation in either
regulatestubeformation,thisguidesuggeststhatconsideration
myoblasts or myotubes. Therefore, to assess a myogenic
be given to m-cadherin as a marker of myogenic phenotype in
phenotype it is recommended that Myog be carefully consid-
cells cultured for the purpose of myogenesis.
ered as a marker of differentiation and maturation.
6.4 Integrin α7 subunit (Itga7):
7. Secondary Complementary Markers
6.4.1 In the basal lamina, satellite cells remain quiescent,
7.1 This list of markers in Table 2 is suggested to be used
expressing Pax7 and low levels of integrin α7(Itga7). A
secondarytothemarkerslistedinTable1.Whilethesemarkers
common marker of satellite cell isolations from muscle, Itga7
are present in skeletal muscle cells they are also present in
is essential for myoblast immobilization and fusion (20), and
manyothercelltypes.Thus,theirspecificityinmuscleislower
increases as satellite cells differentiate into mature myoblasts.
making their use to assess myogenesis more difficult.
The role of Itga7 in muscle development is emphasized by its
ability to cause muscular dystrophy in Itga7-null mice. Dele-
7.2 Secondary Markers of Myogenesis:
tioninmicehasbeenshowntodisruptmusclehealing,leading
7.2.1 Vimentin (Vim) is an intermediate filament protein
to fibrosis and exacerbating dystrophic muscle development
presentinmuscle. Vimisstronglyexpressedindevelopingfetal
(21, 22). Further, increases in eccentric exe
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...