ASTM E724-21
(Guide)Standard Guide for Conducting Static Short-Term Chronic Toxicity Tests Starting with Embryos of Four Species of Saltwater Bivalve Molluscs
Standard Guide for<brk type="line"/> Conducting Static Short-Term Chronic Toxicity Tests Starting with Embryos of Four Species of Saltwater Bivalve Molluscs
SIGNIFICANCE AND USE
5.1 An acute toxicity test is conducted to assess the effects of a short term exposure of organisms to a test material under specific experimental conditions. An acute toxicity test does not provide information concerning whether delayed effects will occur and typically evaluates effects on survival. A chronic test is typically longer in duration and includes a sublethal endpoint to assess effects on a population that might occur beyond the exposure period. Because the bivalve embryo development test includes a sublethal endpoint, but is also short in duration, these tests are considered to be short-term chronic tests.
5.2 Because embryos and larvae are usually assumed to be the most sensitive life stages of these bivalve mollusc species and because these species are commercially and recreationally important, results of these acute tests are often considered to be a good indication of the acceptability of pollutant concentrations to saltwater molluscan species in general. Results of these acute toxicity tests are often assumed to be an important consideration when assessing the hazard of materials to other saltwater organisms (see Guide E1023) or when deriving water quality criteria for saltwater organisms (3) .
5.3 Results of short-term chronic toxicity tests might be used to predict effects likely to occur to aquatic organisms in field situations as a result of exposure under comparable conditions, except that toxicity to benthic species might depend on sorption or settling of the test material onto the substrate.
5.4 Results of short-term chronic tests might be used to compare the sensitivities of different species to different test materials, and to determine the effects of various environmental factors on results of such tests.
5.5 Results of short-term chronic toxicity tests might be useful for studying biological availability of, and structure activity relationships between, test materials.
5.6 Results of any toxicity test will depend on temper...
SCOPE
1.1 This guide describes procedures for obtaining laboratory data concerning the acute effects of a test material on embryos and the resulting larvae of four species of saltwater bivalve molluscs (Pacific oyster, Crassostrea gigas Thunberg; eastern oyster, Crassostrea virginica Gmelin; quahog or hard clam, Mercenaria mercenaria Linnaeus; and the mussel species complex (Mytilus spp.) including the blue mussel, Mytilus edulis Linnaeus; the Mediterranean mussel, Mytilus galloprovincialis Lamark; and the Northern Bay Mussel, Mytilus trossulus Gould) during static 48-h exposures. These procedures will probably be useful for conducting static short-term chronic toxicity tests starting with embryos of other bivalve species (1)2 although modifications might be necessary.
1.2 Other modifications of these procedures might be justified by special needs or circumstances. Although using procedures appropriate to a particular species or special needs and circumstances is more important than following prescribed procedures, results of tests conducted by using unusual procedures are not likely to be comparable to results of many other tests. Comparison of results obtained by using modified and unmodified versions of these procedures might provide useful information concerning new concepts and procedures for conducting 48-h acute tests starting with embryos of bivalve molluscs.
1.3 These procedures are applicable to most chemicals, either individually or in formulations, commercial products, or known mixtures. With appropriate modifications these procedures can be used to conduct acute tests on temperature, dissolved oxygen, and pH and on such materials as aqueous effluents (see also Guide E1192), leachates, oils, particulate matter, sediments, and surface waters. Renewal tests might be preferable to static tests for materials that have a high oxygen demand, are highly volatile, are rapidly biologically or chemically transformed...
General Information
- Status
- Published
- Publication Date
- 14-Jan-2021
- Technical Committee
- E50 - Environmental Assessment, Risk Management and Corrective Action
- Drafting Committee
- E50.47 - Biological Effects and Environmental Fate
Relations
- Effective Date
- 01-Apr-2019
- Effective Date
- 01-Sep-2010
- Effective Date
- 01-Mar-2008
- Effective Date
- 01-Feb-2008
- Effective Date
- 01-Feb-2008
- Effective Date
- 01-Feb-2008
- Effective Date
- 01-Oct-2007
- Effective Date
- 01-Oct-2007
- Effective Date
- 15-Nov-2006
- Effective Date
- 15-Nov-2006
- Effective Date
- 10-Mar-2003
- Effective Date
- 10-Oct-1997
Overview
ASTM E724-21 is the Standard Guide for Conducting Static Short-Term Chronic Toxicity Tests Starting with Embryos of Four Species of Saltwater Bivalve Molluscs. Developed by ASTM, this standard provides comprehensive procedures for evaluating the acute effects of various materials, including chemicals and environmental samples, on the embryonic and larval stages of key saltwater bivalve species. The primary aim is to generate laboratory toxicity data crucial for environmental hazard assessment and regulatory compliance in marine and estuarine ecosystems.
This guide focuses on short-term, 48-hour static exposure tests with Pacific oyster (Crassostrea gigas), eastern oyster (Crassostrea virginica), hard clam (Mercenaria mercenaria), and the Mytilus mussel species complex. The methods assess both lethality and sublethal effects, offering a robust framework for determining pollutant acceptability and deriving water quality criteria to protect commercially and ecologically important species.
Key Topics
- Test Organisms: Protocols target early life stages (embryos and larvae) of four saltwater bivalve mollusc species, which are considered highly sensitive indicators of marine pollution.
- Static Short-Term Chronic Toxicity: Tests are designed for 48-hour exposures, evaluating both survival and developmental endpoints to provide more nuanced insight than traditional acute tests.
- Test Materials: Procedures are suitable for pure chemicals, formulations, environmental samples (effluents, surface waters, sediments, leachates), and under some modifications, for variables like pH, temperature, or dissolved oxygen.
- Dilution Water and Test Conditions: Guidance is provided for sourcing, treating, and characterizing high-quality dilution water, crucial for test validity, along with stringent requirements for test chamber design and cleaning.
- Reporting Endpoints: Results are typically reported as EC50 (effective concentration for 50% effect based on sublethal and lethal responses) and LC50 (lethal concentration for 50% mortality), supporting regulatory and risk assessment applications.
Applications
The standardized procedures in ASTM E724-21 are vital for:
- Marine Pollution Assessment: Determining the toxicity of waste discharges, chemical spills, or new substances before their release into marine and estuarine environments.
- Ecotoxicological Research: Comparing sensitivity among multiple saltwater species and materials to inform environmental monitoring programs.
- Regulatory Compliance: Supporting environmental impact assessments and establishment of water quality criteria by governments, industry, and regulatory agencies.
- Product Development: Evaluating the biological availability and ecological safety of chemicals, commercial formulations, and products in development or use near coastal waters.
- Comparative Studies: Assessing the influence of environmental factors (e.g., temperature, salinity, organic content) on toxicity test outcomes, and enabling comparison between standard and modified test procedures.
Related Standards
To ensure comprehensive environmental testing, ASTM E724-21 references and aligns with several related standards, including:
- ASTM E729: Guide for Conducting Acute Toxicity Tests on Test Materials with Fishes, Macroinvertebrates, and Amphibians.
- ASTM E1023: Guide for Assessing the Hazard of a Material to Aquatic Organisms.
- ASTM E1192: Guide for Conducting Acute Toxicity Tests on Aqueous Ambient Samples and Effluents.
- ASTM E943: Terminology Relating to Biological Effects and Environmental Fate.
- ASTM E1367 and E1706: Test methods for sediment-associated toxicity in estuarine, marine, and freshwater systems.
These complementary standards support a holistic approach to aquatic toxicity testing and reinforce best practices in laboratory and environmental assessment for saltwater organisms.
Keywords: ASTM E724-21, bivalve mollusc toxicity test, marine ecotoxicology, static chronic toxicity, embryo-larval assay, environmental assessment, water quality criteria, saltwater pollution, regulatory testing, marine bivalves, ASTM standards.
Buy Documents
ASTM E724-21 - Standard Guide for<brk type="line"/> Conducting Static Short-Term Chronic Toxicity Tests Starting with Embryos of Four Species of Saltwater Bivalve Molluscs
REDLINE ASTM E724-21 - Standard Guide for<brk type="line"/> Conducting Static Short-Term Chronic Toxicity Tests Starting with Embryos of Four Species of Saltwater Bivalve Molluscs
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

Bureau Veritas
Bureau Veritas is a world leader in laboratory testing, inspection and certification services.

DNV
DNV is an independent assurance and risk management provider.
Sponsored listings
Frequently Asked Questions
ASTM E724-21 is a guide published by ASTM International. Its full title is "Standard Guide for<brk type="line"/> Conducting Static Short-Term Chronic Toxicity Tests Starting with Embryos of Four Species of Saltwater Bivalve Molluscs". This standard covers: SIGNIFICANCE AND USE 5.1 An acute toxicity test is conducted to assess the effects of a short term exposure of organisms to a test material under specific experimental conditions. An acute toxicity test does not provide information concerning whether delayed effects will occur and typically evaluates effects on survival. A chronic test is typically longer in duration and includes a sublethal endpoint to assess effects on a population that might occur beyond the exposure period. Because the bivalve embryo development test includes a sublethal endpoint, but is also short in duration, these tests are considered to be short-term chronic tests. 5.2 Because embryos and larvae are usually assumed to be the most sensitive life stages of these bivalve mollusc species and because these species are commercially and recreationally important, results of these acute tests are often considered to be a good indication of the acceptability of pollutant concentrations to saltwater molluscan species in general. Results of these acute toxicity tests are often assumed to be an important consideration when assessing the hazard of materials to other saltwater organisms (see Guide E1023) or when deriving water quality criteria for saltwater organisms (3) . 5.3 Results of short-term chronic toxicity tests might be used to predict effects likely to occur to aquatic organisms in field situations as a result of exposure under comparable conditions, except that toxicity to benthic species might depend on sorption or settling of the test material onto the substrate. 5.4 Results of short-term chronic tests might be used to compare the sensitivities of different species to different test materials, and to determine the effects of various environmental factors on results of such tests. 5.5 Results of short-term chronic toxicity tests might be useful for studying biological availability of, and structure activity relationships between, test materials. 5.6 Results of any toxicity test will depend on temper... SCOPE 1.1 This guide describes procedures for obtaining laboratory data concerning the acute effects of a test material on embryos and the resulting larvae of four species of saltwater bivalve molluscs (Pacific oyster, Crassostrea gigas Thunberg; eastern oyster, Crassostrea virginica Gmelin; quahog or hard clam, Mercenaria mercenaria Linnaeus; and the mussel species complex (Mytilus spp.) including the blue mussel, Mytilus edulis Linnaeus; the Mediterranean mussel, Mytilus galloprovincialis Lamark; and the Northern Bay Mussel, Mytilus trossulus Gould) during static 48-h exposures. These procedures will probably be useful for conducting static short-term chronic toxicity tests starting with embryos of other bivalve species (1)2 although modifications might be necessary. 1.2 Other modifications of these procedures might be justified by special needs or circumstances. Although using procedures appropriate to a particular species or special needs and circumstances is more important than following prescribed procedures, results of tests conducted by using unusual procedures are not likely to be comparable to results of many other tests. Comparison of results obtained by using modified and unmodified versions of these procedures might provide useful information concerning new concepts and procedures for conducting 48-h acute tests starting with embryos of bivalve molluscs. 1.3 These procedures are applicable to most chemicals, either individually or in formulations, commercial products, or known mixtures. With appropriate modifications these procedures can be used to conduct acute tests on temperature, dissolved oxygen, and pH and on such materials as aqueous effluents (see also Guide E1192), leachates, oils, particulate matter, sediments, and surface waters. Renewal tests might be preferable to static tests for materials that have a high oxygen demand, are highly volatile, are rapidly biologically or chemically transformed...
SIGNIFICANCE AND USE 5.1 An acute toxicity test is conducted to assess the effects of a short term exposure of organisms to a test material under specific experimental conditions. An acute toxicity test does not provide information concerning whether delayed effects will occur and typically evaluates effects on survival. A chronic test is typically longer in duration and includes a sublethal endpoint to assess effects on a population that might occur beyond the exposure period. Because the bivalve embryo development test includes a sublethal endpoint, but is also short in duration, these tests are considered to be short-term chronic tests. 5.2 Because embryos and larvae are usually assumed to be the most sensitive life stages of these bivalve mollusc species and because these species are commercially and recreationally important, results of these acute tests are often considered to be a good indication of the acceptability of pollutant concentrations to saltwater molluscan species in general. Results of these acute toxicity tests are often assumed to be an important consideration when assessing the hazard of materials to other saltwater organisms (see Guide E1023) or when deriving water quality criteria for saltwater organisms (3) . 5.3 Results of short-term chronic toxicity tests might be used to predict effects likely to occur to aquatic organisms in field situations as a result of exposure under comparable conditions, except that toxicity to benthic species might depend on sorption or settling of the test material onto the substrate. 5.4 Results of short-term chronic tests might be used to compare the sensitivities of different species to different test materials, and to determine the effects of various environmental factors on results of such tests. 5.5 Results of short-term chronic toxicity tests might be useful for studying biological availability of, and structure activity relationships between, test materials. 5.6 Results of any toxicity test will depend on temper... SCOPE 1.1 This guide describes procedures for obtaining laboratory data concerning the acute effects of a test material on embryos and the resulting larvae of four species of saltwater bivalve molluscs (Pacific oyster, Crassostrea gigas Thunberg; eastern oyster, Crassostrea virginica Gmelin; quahog or hard clam, Mercenaria mercenaria Linnaeus; and the mussel species complex (Mytilus spp.) including the blue mussel, Mytilus edulis Linnaeus; the Mediterranean mussel, Mytilus galloprovincialis Lamark; and the Northern Bay Mussel, Mytilus trossulus Gould) during static 48-h exposures. These procedures will probably be useful for conducting static short-term chronic toxicity tests starting with embryos of other bivalve species (1)2 although modifications might be necessary. 1.2 Other modifications of these procedures might be justified by special needs or circumstances. Although using procedures appropriate to a particular species or special needs and circumstances is more important than following prescribed procedures, results of tests conducted by using unusual procedures are not likely to be comparable to results of many other tests. Comparison of results obtained by using modified and unmodified versions of these procedures might provide useful information concerning new concepts and procedures for conducting 48-h acute tests starting with embryos of bivalve molluscs. 1.3 These procedures are applicable to most chemicals, either individually or in formulations, commercial products, or known mixtures. With appropriate modifications these procedures can be used to conduct acute tests on temperature, dissolved oxygen, and pH and on such materials as aqueous effluents (see also Guide E1192), leachates, oils, particulate matter, sediments, and surface waters. Renewal tests might be preferable to static tests for materials that have a high oxygen demand, are highly volatile, are rapidly biologically or chemically transformed...
ASTM E724-21 is classified under the following ICS (International Classification for Standards) categories: 07.100.20 - Microbiology of water; 13.020.40 - Pollution, pollution control and conservation. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E724-21 has the following relationships with other standards: It is inter standard links to ASTM E1706-19, ASTM E1706-05(2010), ASTM E943-08, ASTM E1391-03(2008), ASTM E1192-97(2008), ASTM E1367-03(2008), ASTM E729-96(2007), ASTM E1023-84(2007), ASTM D4447-10, ASTM D4447-06, ASTM D4447-84(2003), ASTM D4447-84(1997). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E724-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E724 − 21
Standard Guide for
Conducting Static Short-Term Chronic Toxicity Tests
Starting with Embryos of Four Species of Saltwater Bivalve
Molluscs
This standard is issued under the fixed designation E724; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope matter, sediments, and surface waters. Renewal tests might be
preferable to static tests for materials that have a high oxygen
1.1 This guide describes procedures for obtaining laboratory
demand, are highly volatile, are rapidly biologically or chemi-
data concerning the acute effects of a test material on embryos
cally transformed in aqueous solution, or are removed from test
and the resulting larvae of four species of saltwater bivalve
solutions in substantial quantities by the test chambers or
molluscs (Pacific oyster, Crassostrea gigas Thunberg; eastern
organisms during the test.
oyster, Crassostrea virginica Gmelin; quahog or hard clam,
Mercenaria mercenaria Linnaeus; and the mussel species
1.4 Results of toxicity tests with embryos of bivalve mol-
complex (Mytilus spp.) including the blue mussel, Mytilus
luscs should usually be reported as the EC50 based on the total
edulis Linnaeus; the Mediterranean mussel, Mytilus gallopro-
incompletely developed and dead organisms. It might also be
vincialis Lamark; and the Northern Bay Mussel, Mytilus
desirable to report the LC50 based only on death. In some
trossulus Gould) during static 48-h exposures. These proce-
situations, it might only be necessary to determine whether a
dures will probably be useful for conducting static short-term
specific concentration is toxic to embryos or whether the EC50
chronic toxicity tests starting with embryos of other bivalve
is above or below a specific concentration.
species (1) although modifications might be necessary.
1.5 This guide is arranged as follows:
1.2 Other modifications of these procedures might be justi-
fied by special needs or circumstances. Although using proce-
dures appropriate to a particular species or special needs and
Section
circumstances is more important than following prescribed
Referenced Documents 2
procedures, results of tests conducted by using unusual proce-
Terminology 3
Summary of Guide 4
dures are not likely to be comparable to results of many other
Significance and Use 5
tests. Comparison of results obtained by using modified and
Hazards 6
unmodified versions of these procedures might provide useful
Apparatus 7
Facilities 7.1
information concerning new concepts and procedures for
Construction Materials 7.2
conducting 48-h acute tests starting with embryos of bivalve
Test Chambers 7.3
molluscs.
Cleaning 7.4
Acceptability 7.5
1.3 These procedures are applicable to most chemicals,
Dilution Water 8
either individually or in formulations, commercial products, or Requirements 8.1
Source 8.2
known mixtures. With appropriate modifications these proce-
Treatments 8.3
dures can be used to conduct acute tests on temperature,
Characterization 8.4
Test Material 9
dissolved oxygen, and pH and on such materials as aqueous
General 9.1
effluents (see also Guide E1192), leachates, oils, particulate
Stock Solution 9.2
Test Concentration(s) 9.3
Test Organisms 10
This guide is under the jurisdiction of ASTM Committee E50 on Environmental Species 10.1
Assessment, Risk Management and Corrective Action and is the direct responsibil- Age 10.2
Handling 10.3
ity of Subcommittee E50.47 on Biological Effects and Environmental Fate.
Brood Stock Source and Condition 10.4
Current edition approved Jan. 15, 2021. Published March 2021. Originally
Spawning and Fertilization 10.5
approved in 1980. Last previous edition approved in 2012 as E74 – 98 (2012). DOI:
Quality 10.6
10.1520/E0724-21.
Procedure 11
The boldface numbers in parentheses refer to the list of references at the end of
Experimental Design 11.1
this guide.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E724 − 21
Conducted Under ASTM Guidelines (Withdrawn 2022)
Dissolved Oxygen 11.2
Temperature 11.3
Beginning the Test 11.4
3. Terminology
Feeding 11.5
3.1 Definitions:
Duration of Test 11.6
Biological Data 11.7
3.1.1 The words “must,” “should,” “may,” “can,” and
Other Measurements 11.8
“might” have very specific meanings in this guide. “Must” is
Analytical Methods 12
Acceptability of Test 13 used to express an absolute requirement, that is, to state that the
Calculation of Results 14
test ought to be designed to satisfy the specified condition,
Report 15
unless the purpose of the test requires a different design.
Annex Annex A1
“Must” is only used in connection with factors that directly
relate to the acceptability of the test (see 13.1). “Should” is
used to state that the specified condition is recommended and
1.6 This standard does not purport to address all of the
ought to be met if possible. Although violation of one “should”
safety concerns, if any, associated with its use. It is the
statement is rarely a serious matter, violation of several will
responsibility of the user of this standard to establish appro-
often render the results questionable. Terms such as “is
priate safety and health practices and determine the applica-
desirable,” “is often desirable,” and “might be desirable” are
bility of regulatory limitations prior to use.Specific hazard
used in connection with less important factors. “May” is used
statements are given in Section 6.
to mean “is (are) allowed to,” “can” is used to mean “is (are)
1.7 This international standard was developed in accor- able to,” and “might” is used to mean “could possibly.” Thus
dance with internationally recognized principles on standard- the classic distinction between “may” and “can” is preserved,
ization established in the Decision on Principles for the and “might” is never used as a synonym for either “may” or
Development of International Standards, Guides and Recom-
“can.”
mendations issued by the World Trade Organization Technical 3.1.2 For definitions of other terms used in this guide, refer
Barriers to Trade (TBT) Committee.
to Guide E729, Terminology E943, and Guide E1023. For an
explanation of units and symbols, refer to Practice E380.
2. Referenced Documents 3.2 Definitions of Terms Specific to This Standard:
3.2.1 embryo—used herein to denote the stages between the
2.1 ASTM Standards:
fertilization of the egg and the trochophore (2) (Figs. 1 and 2).
E380 Practice for Use of the International System of Units
3.2.2 larva—used herein to include the trochophore and the
(SI) (the Modernized Metric System)
straight hinge stage (2) (Figs. 1 and 2).
D4447 Guide for Disposal of Laboratory Chemicals and
Samples
4. Summary of Guide
E729 Guide for Conducting Acute Toxicity Tests on Test
4.1 Adult saltwater bivalve molluscs are brought into the
Materials with Fishes, Macroinvertebrates, and Amphib-
laboratory, cleaned of detritus and fouling organisms, and
ians
identified to species. If the gonads are not ripe (see 10.4.5), the
E943 Terminology Relating to Biological Effects and Envi-
bivalves are conditioned to bring them into a suitable repro-
ronmental Fate
ductive state. Bivalves with ripe gonads are maintained under
E1023 Guide for Assessing the Hazard of a Material to
conditions that keep the gonads ripe without inducing unde-
Aquatic Organisms and Their Uses
sired spontaneous spawning or resorption of gametes. Mytilus
E1192 Guide for Conducting Acute Toxicity Tests on Aque-
spp. can be held for several months in a gravid state by keeping
ous Ambient Samples and Effluents with Fishes,
them in clean cold seawater without feeding. In order to start a
Macroinvertebrates, and Amphibians
test, spawning is induced by using one or more stimuli which
E1367 Test Method for Measuring the Toxicity of Sediment-
may be physical (for example, temperature), biological (for
Associated Contaminants with Estuarine and Marine In-
example, heat-killed bivalve sperm), or chemical (for example,
vertebrates
serotonin).
E1391 Guide for Collection, Storage, Characterization, and
Manipulation of Sediments for Toxicological Testing and
4.2 In each of two or more treatments, embryos and the
for Selection of Samplers Used to Collect Benthic Inver-
resulting larvae of one species are maintained for 48 h. In each
tebrates
of one or more control treatments, the embryos and resulting
E1706 Test Method for Measuring the Toxicity of Sediment-
larvae are maintained in dilution water to which no test
Associated Contaminants with Freshwater Invertebrates
material has been added in order to provide (a) a measure of
E1847 Practice for Statistical Analysis of Toxicity Tests
the acceptability of the test by giving an indication of the
quality of the organisms and the suitability of the dilution
water, test conditions, handling procedures, and so forth, and
3 (b) the basis for interpreting data obtained from the other
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM treatments. In each of one or more other treatments, the
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website.
4 5
Withdrawn. The last approved version of this historical standard is referenced The last approved version of this historical standard is referenced on
on www.astm.org. www.astm.org.
E724 − 21
FIG. 1 Drawings Exemplifying Five Key Developmental Stages of Bivalve Larvae Occurring During the First 48 h of Development
embryos and resulting larvae are maintained in dilution water 5. Significance and Use
to which a selected concentration of test material has been
5.1 An acute toxicity test is conducted to assess the effects
added. The 48-h EC50 is calculated based on the proportion of
of a short term exposure of organisms to a test material under
live larvae with completely developed shells in chambers
specific experimental conditions. An acute toxicity test does
containing the test material to live larvae with completely
not provide information concerning whether delayed effects
developed shells in the controls at the termination of the 48-h
will occur and typically evaluates effects on survival. A chronic
test.
E724 − 21
FIG. 2 Photomicrographs (×450) Exemplifying Five Key Developmental Stages of Bivalve Larvae Occurring During the First 48 h of De-
velopment
test is typically longer in duration and includes a sublethal a good indication of the acceptability of pollutant concentra-
endpoint to assess effects on a population that might occur tions to saltwater molluscan species in general. Results of these
beyond the exposure period. Because the bivalve embryo
acute toxicity tests are often assumed to be an important
development test includes a sublethal endpoint, but is also
consideration when assessing the hazard of materials to other
short in duration, these tests are considered to be short-term
saltwater organisms (see Guide E1023) or when deriving water
chronic tests.
quality criteria for saltwater organisms (3).
5.2 Because embryos and larvae are usually assumed to be
5.3 Results of short-term chronic toxicity tests might be
the most sensitive life stages of these bivalve mollusc species
used to predict effects likely to occur to aquatic organisms in
and because these species are commercially and recreationally
field situations as a result of exposure under comparable
important, results of these acute tests are often considered to be
E724 − 21
conditions, except that toxicity to benthic species might depend 6.7 To protect hands from being cut by sharp edges of oyster
on sorption or settling of the test material onto the substrate. shells, cotton work gloves should be worn over appropriate
protective gloves (see 6.1), if necessary, when oysters are
5.4 Results of short-term chronic tests might be used to
handled. When an oyster knife is used to open adults of any
compare the sensitivities of different species to different test
bivalve species, heavy rubber work gloves should be worn to
materials, and to determine the effects of various environmen-
protect hands from puncture.
tal factors on results of such tests.
6.8 Preservation of larvae to facilitate microscopic enu-
5.5 Results of short-term chronic toxicity tests might be
meration will be performed with a fixative agent such as
useful for studying biological availability of, and structure
buffered formalin, and biological stains (that is, Rose Bengal).
activity relationships between, test materials.
Appropriate safety precautions should be taken when handling.
5.6 Results of any toxicity test will depend on temperature,
composition of the dilution water, condition of the test
7. Apparatus
organisms, and other factors.
7.1 Facilities:
5.7 Results of short-term chronic toxicity tests might be
7.1.1 Flow-through troughs with appropriate trays (8)
used to predict effects likely to occur to aquatic organisms
should be available for holding and conditioning the brood
exposed to suspended particulates of dredged sediments dis-
stock. The water-supply system should be equipped for tem-
posed through the water column.
perature control and aeration (see 8.3), and should contain
strainers and air traps. Air used for aeration should be free of
5.8 Results of short-term chronic toxicity tests might be
fumes, oil, and water; filters to remove oil and water are
used to predict effects likely to occur to aquatic organisms
desirable. Test chambers should be in a constant-temperature
exposed to a bedded whole sediments.
room, incubator, or recirculating water bath. A dilution-water
tank or headbox, which might be used to prepare reconstituted
6. Hazards
water, is often elevated so that dilution water can be gravity fed
6.1 Many materials can adversely affect humans if precau-
into holding and conditioning troughs and test chambers. The
tions are inadequate. Therefore, skin contact with all test
facility should be well ventilated and free of fumes. To further
materials and solutions of them should be minimized by such
reduce the possibility of contamination of brood stock and test
means as wearing appropriate protective gloves (especially
organisms by test materials and other substances, especially
when washing equipment or putting hands in test solutions),
volatile ones, holding and conditioning troughs should not be
laboratory coats, aprons, and glasses. Special precautions, such
in a room in which toxicity tests are conducted, stock solutions
as covering test chambers and ventilating the area surrounding
or test solutions are prepared, or equipment is cleaned. During
the chambers, should be taken when conducting tests on
holding, conditioning, and testing, organisms should be
volatile materials. Information concerning toxicity to humans
shielded from disturbances with curtains or partitions to
(4), recommended handling procedures (5), and chemical and
prevent unnecessary stress.
physical properties of the test material should be studied before
7.1.2 It is probably desirable to include some safeguards in
a test is begun. Special procedures are necessary with radiola-
the system that supplies water to holding and conditioning
beled test materials (6) and with materials that are, or are
troughs. Monitors, possibly connected to auxiliary power
suspected of being, carcinogenic (7).
supplies, might be designed to initiate aeration, sound alarms,
6.2 Although in most cases disposal of stock solutions, test
or activate telephone auto-dialing alarms if water flow or
solutions, and test organisms poses no special problems, health temperature deviates from preset limits. If temperature be-
and safety precautions and applicable regulations should be
comes too high or low, corrective action should not cause the
considered before beginning a test (see Guide D4447). Re-
temperature of the water in holding and conditioning troughs to
moval or degradation of the test material might be desirable increase or decrease more than 2°C/day to reduce the chances
before disposal of stock and test solutions.
of spontaneous spawning.
6.3 Cleaning of equipment with a volatile solvent such as 7.2 Construction Materials—Equipment and facilities that
acetone should be performed only in a well-ventilated area in
contact stock solutions, test solutions, or any water into which
which no smoking is allowed and no open flame, such as a pilot brood stock or test organisms will be placed should not contain
light, is present.
substances that can be leached or dissolved by aqueous
solutions in amounts that adversely affect test organisms. In
6.4 Warning—An acidic solution should not be mixed with
addition, equipment and facilities that contact stock solutions
a hypochlorite solution because hazardous fumes might be
or test solutions should be chosen to minimize sorption of test
produced.
materials from water. Glass, Type 316 stainless steel, nylon,
6.5 To prepare dilute acid solutions, concentrated acid
and fluorocarbon plastics should be used whenever possible to
should be added to water, not vice versa. Opening a bottle of
minimize dissolution, leaching, and sorption, except that stain-
concentrated acid and adding concentrated acid to water should
less steel should not be used when testing metals. Concrete and
be performed only in a fume hood.
rigid plastics may be used for holding and conditioning tanks
6.6 Precaution—Use of ground fault systems and leak and in the water-supply system, but they should be soaked,
detectors is strongly recommended to help prevent electrical preferably in flowing dilution water, for a week or more before
shocks because salt water is a good conductor of electricity. use (9). Brass, copper, lead, galvanized metal, cast-iron pipe,
E724 − 21
and natural rubber should not contact dilution water, stock bivalve molluscs and their embryos and larvae, (b) be of
solutions, or test solutions before or during the test. Items made uniform quality, and (c) not unnecessarily affect results of the
of neoprene rubber and other materials not mentioned should test except as in 8.1.4.
not be used unless it has been shown that embryos and
8.1.1 The minimal requirement for an acceptable dilution
resulting larvae of the test species do not show more signs of
water for toxicity tests with embryos of oysters, clams and
stress, such as discoloration, incomplete shell development, or
mussels is that at least 70 % 60 %, or, 90 % respectively, of
death, when held for 48 h in static dilution water in which the
the embryos resulting from eggs and sperm produced by
item is soaking than when held in static dilution water that does appropriately conditioned adults result in live larvae with
not contain the item.
completely developed shells (normal in shape) while being
maintained in the dilution water for 48 h. Natural salt water
7.3 Test Chambers:
varies in quality enough that, even though it is usually
7.3.1 In a toxicity test with aquatic organisms, test chambers
acceptable, occasionally it might be toxic to embryos or larvae
are defined as the smallest physical units between which there
if, for example, certain toxic algae species are present such as
are no water connections. Chambers should be covered to keep
some dinoflagellates (12).
out extraneous contaminants and bacteria and to minimize
8.1.2 The quality of the dilution water should be uniform
evaporation of test solution and material. Substantial concen-
enough that the brood stock is held and conditioned and the test
trations of bacteria in the test solutions might severely reduce
is conducted in water of the same quality. In particular, the
the survival of the embryos and resulting larvae, whereas
salinity should always be between 18 and 34 g/kg or parts per
differences in the amount of evaporation among test chambers
thousand (ppt)(13) and within a test should not vary by more
will directly contribute to between-chamber variation in sur-
than 1 ppt among treatments or any renewals during a test.
vival. All chambers in a test must be identical.
When a test is conducted on an effluent, brine, drilling mud, or
7.3.2 Tests are usually conducted in glass chambers that are
other material that affects salinity when mixed with dilution
1 to 2 L in capacity. Very small test chambers, containing as
water, it might be desirable to adjust salinity by adding sea salt
little as 10 to 30 mL, and sealed test chambers (10) may be
(see 8.2.1.2) to raise the salinity or by adding distilled or
used if the survival and development of the embryos and
deionized water (see 8.2.1.2) to lower the salinity. If salts are
resulting larvae in the control(s) are acceptable (see 13.1.6).
added, the adjusted material should be well mixed and allowed
7.4 Cleaning—Test chambers and equipment used to pre-
to equilibrate for minimum of 2 h with gentle aeration (24 h
pare and store dilution water, stock solutions, and test solutions
preferred) and salinity confirmed as salts dissolve. The addition
should be cleaned before use. New items should be washed
of artificial salts can produce artifactual toxicity that is ame-
with detergent and rinsed with water, a water miscible organic
liorated with aging. If the salinity is adjusted, an additional salt
solvent, water, acid (such as 10 % concentrated hydrochloric
control, consisting of dilution water diluted with distilled water
acid), and at least twice with deionized, distilled, or dilution
to the salinity of the sample, and then adjusted back to the
water. (Some lots of some organic solvents might leave a film
appropriate salinity with artificial salts should be tested.
that is insoluble in water.) At the end of the test, all items that
8.1.3 The dilution water should not unnecessarily affect
are to be used again should be immediately (a) emptied, (b)
results of tests because of such things as sorption or complex-
rinsed with water, (c) cleaned by a procedure appropriate for
ation of test material. Therefore, except as per 8.1.4, concen-
removing the test material from the item (for example, acid for
trations of both total organic carbon (TOC) and particulate
removing metals and bases; detergent or organic solvent for
matter should be less than 5 mg/L in the dilution water. The
removing organic chemicals), and (d) rinsed at least twice with
concentrations of both TOC and particulate matter should be
deionized, distilled, or dilution water. Acid is often used to
greater than 5 mg/L in the water in which the brood stock is
remove mineral deposits. A hypochlorite solution, often rec-
held and conditioned in order to provide adequate food for the
ommended as a disinfection agent or to remove organic matter,
brood stock.
should not be used due to the extreme toxicity of chlorine-
8.1.4 If it is desired to study the effect of an environmental
produced oxidants to bivalve larvae (11). Test chambers should
factor such as TOC, particulate matter, or dissolved oxygen on
be rinsed with dilution water just before use.
the results of a test, it is necessary to use a water that is
7.5 Acceptability—Before a test is started with embryos of a
naturally or artificially high in TOC or particulate matter or low
bivalve mollusc in new test facilities, it is desirable to conduct
in dissolved oxygen. If such a water is used, it is important that
a nontoxicant test in which all test chambers contain dilution
adequate analyses be performed to characterize the water and
water with no added test material. This is desirable in order to
that a comparable test be available or be conducted in a more
determine (a) if embryos will survive and develop acceptably
usual dilution water to facilitate interpretation of the results
(see 13.1.6), (b) if the dilution water, handling procedures, and
obtained in the special water.
so forth, are acceptable, (c) if there are any location effects on
8.2 Source:
either survival or development, and (d) the magnitude of
8.2.1 Reconstituted Water:
between-chamber variance in the percentage of embryos that
8.2.1.1 Use of a reconstituted water is often not worth the
develop into live larvae with completely developed shells.
effort for tests starting with embryos of bivalve molluscs
8. Dilution Water
because of (a) the large volume needed for conditioning the
8.1 Requirements—Besides being available in adequate brood stock, (b) the necessity of providing adequate food for
supply, the dilution water should (a) be acceptable to adult the brood stock (see 10.4.8), and (c) frequently poor survival
E724 − 21
TABLE 1 Reconstituted Salt Water (14)
possibility of contamination and to maximize the concentration
of dissolved oxygen and healthy phytoplankton (see 10.4.8). A
NOTE 1—Add the following reagent-grade chemicals in the amounts
specially designed system is usually necessary to obtain salt
and order listed to 890 mL of water. Each chemical must be dissolved
A
before the next is added.
water from a natural water source (see Guide E729).
Chemical Amount
8.2.3 Chlorinated water should not be used as, or in the
B
NaF 3 mg
preparation of, saline dilution water because chlorine-produced
SrCl ·6H O 20 mg
2 2
oxidants are quite toxic to embryos and larvae of bivalve
H BO 30 mg
3 3
molluscs (7). Dechlorinated water should be used only as a last
KBr 100 mg
KCl 700 mg
resort because dechlorination is often incomplete. Sodium
CaCl ·2H O 1470 mg
2 2
bisulfite is probably better for dechlorinating water than
Na SO 4000 mg
2 4
sodium sulfite and both are more reliable than carbon filters,
MgCl ·6H O 10780 mg
2 2
NaCl 23500 mg
especially for removing chloramines (15). Some organic
Na SiO ·H O 20 mg
2 3 2
chloramines, however, react slowly with sodium bisulfite (16).
NaHCO 200 mg
In addition to residual chlorine, municipal drinking water often
A
If the resulting solution is diluted to 1 L, the salinity should be 34 ± 0.5 g/kg. The
contains high concentrations of copper, lead, zinc, and fluoride,
reconstituted water should be stripped of trace metals (15). If necessary, the water
should be diluted to the desired salinity at time of use.
and quality is often rather variable. The concentrations of most
B
The NaF should be omitted for tests with C. gigas and included for tests with C.
metals can usually be reduced by using a chelating resin (17),
virginica. Its value for or detriment to M. mercenaria and M. spp. is unknown.
but use of a different dilution water might be preferable.
8.3 Treatments:
8.3.1 Dilution water should be aerated intensively for 24 to
and development of the embryos and resulting larvae. Com-
48 h by such means as air stones, surface aerators, or column
mercially available sea salts have been used successfully in
aerators (18) before addition of test material. To prevent
bivalve embryo testing given proper conditioning of the water.
contamination with undesirable bacterial species during
Be sure to only use salts that are fast-dissolving and closely
aeration, the air used should be filtered through a 0.22-μm
match the ion mix found in natural seawater. A trial test with
bacterial filter, the container should be covered, and aeration
any new brand or batch of salt is recommended prior to any
should not last for more than 48 h. Adequate aeration will bring
testing of samples when feasible. Additionally, commercially
pH and concentrations of dissolved oxygen and other gases
available sea salts should be free of ethylenediaminetetraacetic
into equilibrium with air and minimize oxygen demand and
acid (EDTA) or other chelating agents that may remove or
concentrations of volatiles. The concentration of dissolved
mask toxicity in a sample that is being tested.
oxygen in dilution water should be between 90 and 100 % of
8.2.1.2 Reconstituted water is prepared by adding a com-
saturation (19) to help ensure that dissolved oxygen concen-
mercially avaiable sea salt or specified amounts of reagent-
6 trations are acceptable in test chambers. Supersaturation by
grade chemicals to high quality water (14) with (a) conduc-
dissolved gases, which can be caused by heating the dilution
tivity approximately <1 μS/cm and (b) either TOC <2 mg/L or
water, should be avoided to prevent gas-bubble disease (18,
chemical oxygen demand (COD) <5 mg/L. A formula for
20).
reconstituted water acceptable for use with bivalves is given in
8.3.2 The salinity and pH of dilution water may be adjusted
Table 1. Acceptable water for dissolution of sea salts can
by addition of appropriate reagent-grade chemicals, sea salt
usually be prepared by using a properly operated deionization,
(especially to prevent excessive decreases in salinity; see
distillation, or reverse osmosis unit. Conductivity should be
8.1.2), acid, base, and deionized or distilled water, if it has been
measured on each batch and TOC or COD should be measured
shown that the addition does not cause adverse effects on
at least twice a year and whenever substantial changes might be
embryos, larvae, and adults of the test species at the concen-
expected. If the water is prepared from a surface water, TOC or
tration used.
COD should be measured on each batch. Problems have been
8.3.3 Except possibly when holding and conditioning adult
encountered with some species in reconstituted salt water, but
bivalve molluscs (see 10.4.6), filtration through bag, sand,
sometimes these problems have been overcome by condition-
sock, or depth-type (honeycomb) cartridge filters may be used
ing (aging) and aerating the reconstituted water.
to keep the concentration of particulate matter acceptably low
8.2.2 Natural Dilution Water—If natural salt water is used,
(see 8.1.3) and as a pretreatment before ultraviolet sterilization
it should be obtained from an uncontaminated, uniform quality
or filtration through a finer filter.
source. The quality of saline well water is usually more
8.3.4 Water that might be contaminated with facultative
uniform than that of a saline surface water. If a surface water
pathogens may be passed through a properly maintained
is used, it should be obtained from an area known to support a
ultraviolet sterilizer (21) equipped with an intensity meter and
healthy, naturally reproducing population of bivalves. The
flow controls, or passed through a filter effective to 0.45 μm or
water intake should be positioned (for example, about 1 m
less.
below the surface) to minimize fluctuations in quality and the
8.3.5 Water from a surface-water source should be passed
through a graded series of filters, the finest effective to 1.0 μm
“Reagent Chemicals, American Chemical Society Specifications,” Am. Chemi-
or less to remove embryos and larvae of bivalve molluscs,
cal Soc., Washington, DC. For suggestions on the testing of reagents not listed by
parasites and predators. If bacteria are to be removed by
the American Chemical Society, see “Analar Standards for Laboratory U.K.
Chemicals,” BDH Ltd., Poole, Dorset, and the “United States Pharmacopeia.” filtration, a filter effective to 0.45 μm or less must be used.
E724 − 21
8.3.6 Filtration through activated carbon may be used to reagents might affect the pH of test solutions appreciably. Use
remove toxic algal exocrines and other organic chemicals. of a more soluble form of the test material, such as chloride or
sulfate salts of organic amines, sodium or potassium salts of
8.4 Characterization:
phenols and organic acids, and chloride or nitrate salts of
8.4.1 The following items should be measured at least twice
metals, might affect the pH more than use of the minimum
each year and more often if such measurements have not been
necessary amount of a strong acid or base.
made semiannually for at least 2 years or if a surface water is
used: salinity (or chlorinity), pH, particulate matter, TOC,
9.2.3 If a solvent other than dilution water is used, its
organophosphorus pesticides, organic chlorine (or organochlo-
concentration in test solutions should be kept to a minimum
rine pesticides, plus PCBs), chlorinated phenoxy herbicides,
and should be low enough that it does not affect the test
ammonia, cyanide, sulfide, bromide, fluoride, iodide, nitrate,
species. Because of its low toxicity to aquatic animals (24), low
phosphate, sulfate, calcium, magnesium, potassium,
volatility, and high ability to dissolve many organic chemicals,
aluminum, arsenic, beryllium, boron, cadmium, chromium,
triethylene glycol is often a good organic solvent for preparing
cobalt, copper, iron, lead, manganese, mercury, molybdenum,
stock solutions. Other water-miscible organic solvents such as
nickel, selenium, silver, and zinc.
methanol, ethanol, and acetone may also be used, but they
8.4.2 For each method used (see 12.2), the detection limit
might stimulate undesirable growths of microorganisms, and
should be below either (a) the concentration in the dilution
acetone is also quite volatile. If an organic solvent is used, it
water or (b) the lowest concentration that has been shown to
should be reagent-grade or better and its concentration in any
unacceptably affect embryos, larvae, or adults of saltwater
test solution should not exceed 0.5 mL/L. A surfactant should
bivalve molluscs (22).
not be used in the preparation of a stock solution because it
might affect the form and toxicity of the test material in the test
9. Test Material
solutions. (These limitations do not apply to any ingredient of
9.1 General—Test materials may include a range of sample
a mixture, formulation, or commercial product, unless an extra
types such as effluents, materials or products, specific
amount of solvent is used in the preparation of the stock
chemicals, solvents, oils, surface waters, drilling fluids,
solution.)
stormwater, and sediments (see Guide E1023). For chemical or
9.2.4 If no solvent other than water is used, only a dilution-
product testing studies the test material should be reagent-
grade or better, unless a test on a formulation, commercial water control must be included in the test. For oysters, at least
product, or technical-grade or use-grade material is specifically 70 % of the embryos introduced into the control treatment must
needed. Before a test is begun, the following should be known
result in live larvae with completely developed shells (normal)
about the test material: at the end of the test and at least 90 % of all embryos
9.1.1 Identities and concentrations of major ingredients and
introduced should be alive. For hard clams, at least 60 % of the
major impurities, that is, impurities that constitute more than
embryos introduced must result in live larvae with completely
about 1 % of the material,
developed shells at the end of the test, and at least 90 % of all
9.1.2 Solubility and stability in the dilution water,
embryos introduced should be alive. These values for live
9.1.3 Measured or estimated toxicity to an aquatic species,
larvae with completely developed shells reflect apparently
preferably the test species or another bivalve mollusc,
natural failure of up to 30 to 40 % of the larvae of oysters and
9.1.4 Precision and bias of the analytical method at the
hard clams to develop a shell. For mussels, at least 90 % of the
planned concentration(s) of the test material, if the test con-
embryos introduced into the control treatment must result in
centrations are to be measured,
live larvae with completely developed shells (normal) at the
9.1.5 Estimate of toxicity to humans, and
end of the test and at least 50 % of all embryos introduced
9.1.6 Recommended handling procedures (see 6.1).
should be alive. These criteria values for mussels are consistent
9.2 Stock Solution:
with USEPA guidance (25).
9.2.1 In some cases the test material can be added directly to
9.2.5 If a solvent other than water is used and the concen-
the dilution water, but often it is dissolved in a solvent to form
tration of solvent is the same in all test solutions that contain
a stock solution that is then added to the dilution water. If a
test material, a solvent control, containing the same concen-
stock solution is used, the concentration and stability of the test
tration of solvent as the test solutions and using solvent from
material in it should be determined before the beginning of the
the same batch used to make the stock solution, must be
test. If the test material is subject to photolysis, the stock
included in the test. In addition, a dilution-water control should
solution should be shielded from light.
be included in the test. The number of embryos that result in
9.2.2 Except possibly for tests on hydrolyzable, oxidizable,
live larvae with completely developed shells (normal) at the
and reducible materials, the preferred solvent is dilution water,
end of the test must be at least 70 % of the initial number in the
although filtration or sterilization, or both, of the water might
solvent control for oysters and 60 % for hard clams (see 9.2.4).
be necessary. If the salinity of the dilution water will not be
If a dilution-water control is included in the test, the number of
affected, deionized or distilled water may be used. Several
embryos that result in live larvae with completely developed
techniques have been specifically developed for preparing
shells at the end of the test should be at least 70 % of the initial
aqueous stock solutions of slightly soluble materials (23). The
number in the dilution-water control for oysters and 60 % for
minimum necessary amount of a strong acid or base may be
used in the preparation of an aqueous stock solution, but such hard clams.
E724 − 21
9.2.6 If a solvent other than water is used and the concen- Their use is encouraged to increase comparability of results
tration of solvent is not the same in all test solutions that and availability of much information about a few species rather
contain test material, both a solvent control, containing the
than little information about many species. The species used
highest concentration of solvent present in any other treatment
should be identified by using an appropriate taxonomic key.
and using solvent from the same batch used to make the stock
10.2 Age—The test must be begun with embryos within 4 h
solution, and a dilution-water control must be included in the
after fertilization when the embryos are in the 2-, 4-, and 8-cell
test. The number of embryos that result in live larvae with
stages.
completely developed shells at the end of the test must be at
least 70 % of the initial number in the solvent control and in the
10.3 Handling—Organisms should be handled as little as
dilution-water control for oysters and 60 % for hard clams (see
possible. When handling is necessary, it should be done
9.2.4).
carefully, gently, and quickly, so that organisms are not
9.2.7 If a solvent other than water is used to prepare a stock
unnecessarily stressed. Adults that are injured during handling
solution, it might be desirable to conduct simultaneous tests on
should be discarded. Gentle cleaning of the organisms is
the test material using two chemically unrelated solvents or
however recommended to remove other attached organisms
two different concentrations of the same solvent to obtain
and debris prior to sue for testing. Equipment used to transfer
information concerning possible effects of the solvent on the
embryos of bivalve molluscs should be cleaned between uses
results of the test.
by washing with detergent and rinsing with dilute acid and
9.3 Test Concentration(s):
distilled or deionized water. Hands should be washed before
9.3.1 If the test is intended to allow calculation of an EC50,
and after handling brood stock.
the test concentrations (see 11.1.1.1) should bracket the pre-
10.4 Brood Stock Source and Condition:
dicted EC50. The prediction might be based on the results of a
test with the same or a similar test material and the same or a 10.4.1 For any one test or a series of related tests, all
similar species. If a useful prediction is not available, it is
females and males in the brood stock must be collected from
usually desirable to conduct a range-finding test in which
the same location, which should be known precisely. Brood
embryos and resulting larvae are exposed for a total of 48 h to
stock may be obtained from a commercial source only if the
a control and three to five concentrations of the test material
original specific collection location of the bivalves can be
that differ by a factor of ten. The greater the similarity between
identified. Aquacultured shellfish that forced into triploidy
the range-finding test and the actual test, the more useful the
must not be utilized. To minimize the possibility of genetic or
range-finding test will be.
physiological adaptation to chemicals or aberrant water quality,
9.3.1.1 If necessary, concentrations above solubility should
organisms should be collected from a location that is not
be used because organisms in the real world are sometimes
subject to obvious point or nonpoint source pollution and has
exposed to concentrations above solubility. The use of concen-
water that is comparable in quality to the water that will be
trations that are more than ten times greater than solubility is
used for holding, conditioning, and testing. Gravid adult
probably not worthwhile. With some test materials, it might be
individuals with well developed normal shells should be
found that concentrations above solubility do not kill or affect
obtained. Molluscs possessing high condition factors (26) (see
a greater percentage of test organisms than does the concen-
Note 1), indicative of good food supply and water quality
tration that is the solubility limit; such information is certainly
throughout the year, should be obtained because they are easier
worth knowing.
to hold and condition, and they yield higher quality gametes
9.3.1.2 In some situations, usually related to regulatory
than molluscs with low condition factors. The investigator
activities, it is only necessary to determine whether (a) a
should be aware that sex ratios can differ substantially from
specific concentration of test material is toxic to embryos or
1:1. For example, Katkansky and Sparks (27) found that the
larvae of the test species or (b) the EC50 is above or below a
percentage of females approached 95 % in some groups of
specific concentration. For example, the specific concentration
4-year-old C. gigas due to protandric hermaphroditism. A
might be the concentration occurring in a surface water, the
similar phenomenon might occur in some populations of C.
concentration resulting from the direct application of the
virginica.
material to a body of water, or the solubility limit of the
material in water. When there is only interest in a specific
NOTE 1—The Condition Index (C. I.) for adult bivalves may be
concentration, it is often only necessary to test that concentra-
calculated as follows:
tion plus a control (see 11.1.1.2), and it is not necessary to
determine an EC50.
C. I. 5 100 tissue dry weight, g / volume of shell cavity, mL
~ ! ~ !
10. Test Organisms The sex ratio of the sample should be reported.
10.4.2 Adults may be obtained from distant locations during
10.1 Species—Whenever possible, either Pacific oysters
periods of the year when animals with mature gonads cannot be
(Crassostrea gigas), Eastern oysters (C. virginica), quahogs or
obtained in the vicinity of the test laboratory. A preferable
hard clams (Mercenaria mercenaria), or blue mussels (Mytilus
edulis) should be used as the test species. These species were means of extending the availability of spawnable bivalves is to
hold a population with mature gonads at an appropriate holding
selected on the basis of availability, commercial importance,
past successful use, and ease of handling in the laboratory. temperature (Table 2) after conditioning is complete. If done
E724 − 21
TABLE 2 Recommended Temperature (°C)
10.4.6 If the brood stock contains ripe gonads, the adults
Never to should be placed in cool water (see Table 2). If not maintained
Species Holding Conditioning Induction be Test
in cool water, a brood stock with ripe gametes is usable
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E724 − 98 (Reapproved 2012) E724 − 21
Standard Guide for
Conducting Static Acute Short-Term Chronic Toxicity Tests
Starting with Embryos of Four Species of Saltwater Bivalve
Molluscs
This standard is issued under the fixed designation E724; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide describes procedures for obtaining laboratory data concerning the acute effects of a test material on embryos and
the resulting larvae of four species of saltwater bivalve molluscs (Pacific oyster, Crassostrea gigas Thunberg; eastern oyster,
Crassostrea virginica Gmelin; quahog or hard clam, Mercenaria mercenaria Linnaeus; and the mussel species complex (Mytilus
spp.) including the blue mussel, Mytilus edulis Linnaeus) Linnaeus; the Mediterranean mussel, Mytilus galloprovincialis Lamark;
and the Northern Bay Mussel, Mytilus trossulus Gould) during static 48-h exposures. These procedures will probably be useful for
conducting static acute short-term chronic toxicity tests starting with embryos of other bivalve species (1) although modifications
might be necessary.
1.2 Other modifications of these procedures might be justified by special needs or circumstances. Although using procedures
appropriate to a particular species or special needs and circumstances is more important than following prescribed procedures,
results of tests conducted by using unusual procedures are not likely to be comparable to results of many other tests. Comparison
of results obtained by using modified and unmodified versions of these procedures might provide useful information concerning
new concepts and procedures for conducting 48-h acute tests starting with embryos of bivalve molluscs.
1.3 These procedures are applicable to most chemicals, either individually or in formulations, commercial products, or known
mixtures. With appropriate modifications these procedures can be used to conduct acute tests on temperature, dissolved oxygen,
and pH and on such materials as aqueous effluents (see also Guide E1192), leachates, oils, particulate matter, sediments, and
surface waters. Renewal tests might be preferable to static tests for materials that have a high oxygen demand, are highly volatile,
are rapidly biologically or chemically transformed in aqueous solution, or are removed from test solutions in substantial quantities
by the test chambers or organisms during the test.
1.4 Results of acute toxicity tests with embryos of bivalve molluscs should usually be reported as the EC50 based on the total
incompletely developed and dead organisms. It might also be desirable to report the LC50 based only on death. In some situations,
it might only be necessary to determine whether a specific concentration is acutely toxic to embryos or whether the EC50 is above
or below a specific concentration.
1.5 This guide is arranged as follows:
This guide is under the jurisdiction of ASTM Committee E50 on Environmental Assessment, Risk Management and Corrective Action and is the direct responsibility
of Subcommittee E50.47 on Biological Effects and Environmental Fate.
Current edition approved Dec. 1, 2012Jan. 15, 2021. Published December 2012March 2021. Originally approved in 1980. Last previous edition approved in 20042012
as E74 – 98 (2004).(2012). DOI: 10.1520/E0724-98R12.10.1520/E0724-21.
The boldface numbers in parentheses refer to the list of references at the end of this guide.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E724 − 21
Section
Referenced Documents 2
Terminology 3
Summary of Guide 4
Significance and Use 5
Hazards 6
Apparatus 7
Facilities 7.1
Construction Materials 7.2
Test Chambers 7.3
Cleaning 7.4
Acceptability 7.5
Dilution Water 8
Requirements 8.1
Source 8.2
Treatments 8.3
Characterization 8.4
Test Material 9
General 9.1
Stock Solution 9.2
Test Concentration(s) 9.3
Test Organisms 10
Species 10.1
Age 10.2
Source 10.3
Handling 10.4
Handling 10.3
Brood Stock Source and Condition 10.5
Brood Stock Source and Condition 10.4
Spawning and Fertilization 10.6
Spawning and Fertilization 10.5
Quality 10.7
Quality 10.6
Procedure 11
Experimental Design 11.1
Dissolved Oxygen 11.2
Temperature 11.3
Beginning the Test 11.4
Feeding 11.5
Duration of Test 11.6
Biological Data 11.7
Other Measurements 11.8
Analytical Methods 12
Acceptability of Test 13
Calculation of Results 14
Report 15
Annex Annex A1
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of
the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. Specific hazard statements are given in Section 6.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E380 Practice for Use of the International System of Units (SI) (the Modernized Metric System)
D4447 Guide for Disposal of Laboratory Chemicals and Samples
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Withdrawn. The last approved version of this historical standard is referenced on www.astm.org.
E724 − 21
E729 Guide for Conducting Acute Toxicity Tests on Test Materials with Fishes, Macroinvertebrates, and Amphibians
E943 Terminology Relating to Biological Effects and Environmental Fate
E1023 Guide for Assessing the Hazard of a Material to Aquatic Organisms and Their Uses
E1192 Guide for Conducting Acute Toxicity Tests on Aqueous Ambient Samples and Effluents with Fishes, Macroinvertebrates,
and Amphibians
E1367 Test Method for Measuring the Toxicity of Sediment-Associated Contaminants with Estuarine and Marine Invertebrates
E1391 Guide for Collection, Storage, Characterization, and Manipulation of Sediments for Toxicological Testing and for
Selection of Samplers Used to Collect Benthic Invertebrates
E1706 Test Method for Measuring the Toxicity of Sediment-Associated Contaminants with Freshwater Invertebrates
E1847 Practice for Statistical Analysis of Toxicity Tests Conducted Under ASTM Guidelines
3. Terminology
3.1 Definitions:
3.1.1 The words “must,” “should,” “may,” “can,” and “might” have very specific meanings in this guide. “Must” is used to express
an absolute requirement, that is, to state that the test ought to be designed to satisfy the specified condition, unless the purpose of
the test requires a different design. “Must” is only used in connection with factors that directly relate to the acceptability of the
test (see 13.1). “Should” is used to state that the specified condition is recommended and ought to be met if possible. Although
violation of one “should” statement is rarely a serious matter, violation of several will often render the results questionable. Terms
such as “is desirable,” “is often desirable,” and “might be desirable” are used in connection with less important factors. “May” is
used to mean “is (are) allowed to,” “can” is used to mean “is (are) able to,” and “might” is used to mean “could possibly.” Thus
the classic distinction between “may” and “can” is preserved, and “might” is never used as a synonym for either “may” or “can.”
3.1.2 For definitions of other terms used in this guide, refer to Guide E729, Terminology E943, and Guide E1023. For an
explanation of units and symbols, refer to Practice E380.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 embryo—used herein to denote the stages between the fertilization of the egg and the trochophore (2) (Figs. 1 and 2).
3.2.2 larva—used herein to include the trochophore and the straight hinge stage (2) (Figs. 1 and 2).
4. Summary of Guide
4.1 Adult saltwater bivalve molluscs are brought into the laboratory, cleaned of detritus and fouling organisms, and identified to
species. If the gonads are not ripe, ripe (see 10.4.5), the bivalves are conditioned to bring them into a suitable reproductive state.
Bivalves with ripe gonads are maintained under conditions that keep the gonads ripe without inducing undesired spontaneous
spawning or resorption of gametes. Mytilus spp. can be held for several months in a gravid state by keeping them in clean cold
seawater without feeding. In order to start a test, spawning is induced by using one or more stimuli which may be physical (for
example, temperature), biological (for example, heat-killed bivalve sperm), or chemical (for example, serotonin).
4.2 In each of two or more treatments, embryos and the resulting larvae of one species are maintained for 48 h. In each of one
or more control treatments, the embryos and resulting larvae are maintained in dilution water to which no test material has been
added in order to provide (a) a measure of the acceptability of the test by giving an indication of the quality of the organisms and
the suitability of the dilution water, test conditions, handling procedures, and so forth, and (b) the basis for interpreting data
obtained from the other treatments. In each of one or more other treatments, the embryos and resulting larvae are maintained in
dilution water to which a selected concentration of test material has been added. The 48-h EC50 is calculated based on the
proportion of live larvae with completely developed shells in chambers containing the test material to live larvae with completely
developed shells in the controls at the termination of the 48-h test.
5. Significance and Use
5.1 An acute toxicity test is conducted to obtain information concerning the acute assess the effects of a short term exposure of
organisms to a test material under specific experimental conditions. An acute toxicity test does not provide information concerning
whether delayed effects will occur. occur and typically evaluates effects on survival. A chronic test is typically longer in duration
and includes a sublethal endpoint to assess effects on a population that might occur beyond the exposure period. Because the
bivalve embryo development test includes a sublethal endpoint, but is also short in duration, these tests are considered to be
short-term chronic tests.
E724 − 21
FIG. 1 Drawings Exemplifying Five Key Developmental Stages of Bivalve Larvae Occurring During the First 48 h of Development
5.2 Because embryos and larvae are usually assumed to be the most sensitive life stages of these bivalve mollusc species and
because these species are commercially and recreationally important, results of these acute tests are often considered to be a good
indication of the acceptability of pollutant concentrations to saltwater molluscan species in general. Results of these acute toxicity
tests are often assumed to be an important consideration when assessing the hazard of materials to other saltwater organisms (see
Guide E1023) or when deriving water quality criteria for saltwater organisms (3).
5.3 Results of acute short-term chronic toxicity tests might be used to predict acute effects likely to occur to aquatic organisms
E724 − 21
FIG. 2 Photomicrographs (×450) Exemplifying Five Key Developmental Stages of Bivalve Larvae Occurring During the First 48 h of De-
velopment
in field situations as a result of exposure under comparable conditions, except that toxicity to benthic species might depend on
sorption or settling of the test material onto the substrate.
5.4 Results of acute short-term chronic tests might be used to compare the acute sensitivities of different species and the acute
toxicities of to different test materials, and to determine the effects of various environmental factors on results of such tests.
5.5 Results of acute short-term chronic toxicity tests might be useful for studying biological availability of, and structure activity
relationships between, test materials.
5.6 Results of acuteany toxicity teststest will depend on temperature, composition of the dilution water, condition of the test
organisms, and other factors.
E724 − 21
5.7 Results of short-term chronic toxicity tests might be used to predict effects likely to occur to aquatic organisms exposed to
suspended particulates of dredged sediments disposed through the water column.
5.8 Results of short-term chronic toxicity tests might be used to predict effects likely to occur to aquatic organisms exposed to
a bedded whole sediments.
6. Hazards
6.1 Many materials can adversely affect humans if precautions are inadequate. Therefore, skin contact with all test materials and
solutions of them should be minimized by such means as wearing appropriate protective gloves (especially when washing
equipment or putting hands in test solutions), laboratory coats, aprons, and glasses. Special precautions, such as covering test
chambers and ventilating the area surrounding the chambers, should be taken when conducting tests on volatile materials.
Information concerning toxicity to humans (4), recommended handling procedures (5), and chemical and physical properties of
the test material should be studied before a test is begun. Special procedures are necessary with radiolabeled test materials (6) and
with materials that are, or are suspected of being, carcinogenic (7).
6.2 Although in most cases disposal of stock solutions, test solutions, and test organisms poses no special problems, health and
safety precautions and applicable regulations should be considered before beginning a test. test (see Guide D4447). Removal or
degradation of the test material might be desirable before disposal of stock and test solutions.
6.3 Cleaning of equipment with a volatile solvent such as acetone should be performed only in a well-ventilated area in which
no smoking is allowed and no open flame, such as a pilot light, is present.
6.4 Warning—An acidic solution should not be mixed with a hypochlorite solution because hazardous fumes might be produced.
6.5 To prepare dilute acid solutions, concentrated acid should be added to water, not vice versa. Opening a bottle of concentrated
acid and adding concentrated acid to water should be performed only in a fume hood.
6.6 Precaution—Use of ground fault systems and leak detectors is strongly recommended to help prevent electrical shocks
because salt water is a good conductor of electricity.
6.7 To protect hands from being cut by sharp edges of oyster shells, cotton work gloves should be worn over appropriate protective
gloves (see 6.1), if necessary, when oysters are handled. When an oyster knife is used to open adults of any bivalve species, heavy
rubber work gloves should be worn to protect hands from puncture.
6.8 Preservation of larvae to facilitate microscopic enumeration will be performed with a fixative agent such as buffered formalin,
and biological stains (that is, Rose Bengal). Appropriate safety precautions should be taken when handling.
7. Apparatus
7.1 Facilities:
7.1.1 Flow-through troughs with appropriate trays (8) should be available for holding and conditioning the brood stock. The
water-supply system should be equipped for temperature control and aeration (see 8.3), and should contain strainers and air traps.
Air used for aeration should be free of fumes, oil, and water; filters to remove oil and water are desirable. Test chambers should
be in a constant-temperature room, incubator, or recirculating water bath. A dilution-water tank or headbox, which might be used
to prepare reconstituted water, is often elevated so that dilution water can be gravity fed into holding and conditioning troughs and
test chambers. The facility should be well ventilated and free of fumes. To further reduce the possibility of contamination of brood
stock and test organisms by test materials and other substances, especially volatile ones, holding and conditioning troughs should
not be in a room in which toxicity tests are conducted, stock solutions or test solutions are prepared, or equipment is cleaned.
During holding, conditioning, and testing, organisms should be shielded from disturbances with curtains or partitions to prevent
unnecessary stress.
7.1.2 It is probably desirable to include some safeguards in the system that supplies water to holding and conditioning troughs.
E724 − 21
Monitors, possibly connected to auxiliary power supplies, might be designed to initiate aeration, sound alarms, or activate
telephone auto-dialing alarms if water flow or temperature deviates from preset limits. If temperature becomes too high or low,
corrective action should not cause the temperature of the water in holding and conditioning troughs to increase or decrease more
than 2°C/day to reduce the chances of spontaneous spawning.
7.2 Construction Materials—Equipment and facilities that contact stock solutions, test solutions, or any water into which brood
stock or test organisms will be placed should not contain substances that can be leached or dissolved by aqueous solutions in
amounts that adversely affect test organisms. In addition, equipment and facilities that contact stock solutions or test solutions
should be chosen to minimize sorption of test materials from water. Glass, Type 316 stainless steel, nylon, and fluorocarbon plastics
should be used whenever possible to minimize dissolution, leaching, and sorption, except that stainless steel should not be used
when testing metals. Concrete and rigid plastics may be used for holding and conditioning tanks and in the water-supply system,
but they should be soaked, preferably in flowing dilution water, for a week or more before use (9). Brass, copper, lead, galvanized
metal, cast-iron pipe, and natural rubber should not contact dilution water, stock solutions, or test solutions before or during the
test. Items made of neoprene rubber and other materials not mentioned should not be used unless it has been shown that embryos
and resulting larvae of the test species do not show more signs of stress, such as discoloration, incomplete shell development, or
death, when held for 48 h in static dilution water in which the item is soaking than when held in static dilution water that does
not contain the item.
7.3 Test Chambers:
7.3.1 In a toxicity test with aquatic organisms, test chambers are defined as the smallest physical units between which there are
no water connections. Chambers should be covered to keep out extraneous contaminants and bacteria and to minimize evaporation
of test solution and material. Substantial concentrations of bacteria in the test solutions might severely reduce the survival of the
embryos and resulting larvae, whereas differences in the amount of evaporation among test chambers will directly contribute to
between-chamber variation in survival. All chambers in a test must be identical.
7.3.2 Tests are usually conducted in glass chambers that are 1 to 2 L in capacity. Very small test chambers, containing as little as
10 to 30 mL, and sealed test chambers (10) may be used if the survival and development of the embryos and resulting larvae in
the control(s) are acceptable (see 13.1.6).
7.4 Cleaning—Test chambers and equipment used to prepare and store dilution water, stock solutions, and test solutions should
be cleaned before use. New items should be washed with detergent and rinsed with water, a water miscible organic solvent, water,
acid (such as 10 % concentrated hydrochloric acid), and at least twice with deionized, distilled, or dilution water. (Some lots of
some organic solvents might leave a film that is insoluble in water.) At the end of the test, all items that are to be used again should
be immediately (a) emptied, (b) rinsed with water, (c) cleaned by a procedure appropriate for removing the test material from the
item (for example, acid for removing metals and bases; detergent or organic solvent for removing organic chemicals), and (d)
rinsed at least twice with deionized, distilled, or dilution water. Acid is often used to remove mineral deposits. A hypochlorite
solution, often recommended as a disinfection agent or to remove organic matter, should not be used due to the extreme toxicity
of chlorine-produced oxidants to bivalve larvae (11). Test chambers should be rinsed with dilution water just before use.
7.5 Acceptability—Before a test is started with embryos of a bivalve mollusc in new test facilities, it is desirable to conduct a
nontoxicant test in which all test chambers contain dilution water with no added test material. This is desirable in order to
determine (a) if embryos will survive and develop acceptably (see 13.1.6), (b) if the dilution water, handling procedures, and so
forth, are acceptable, (c) if there are any location effects on either survival or development, and (d) the magnitude of
between-chamber variance in the percentage of embryos that develop into live larvae with completely developed shells.
8. Dilution Water
8.1 Requirements—Besides being available in adequate supply, the dilution water should (a) be acceptable to adult bivalve
molluscs and their embryos and larvae, (b) be of uniform quality, and (c) not unnecessarily affect results of the test except as in
8.1.4.
8.1.1 The minimal requirement for an acceptable dilution water for acute toxicity tests starting with embryos of oysters or hard
clamsoysters, clams and mussels is that at least 70 % or 60 %, 70 % 60 %, or, 90 % respectively, of the embryos resulting from
eggs and sperm produced by appropriately conditioned adults result in live larvae with completely developed shells (normal in
shape) while being maintained in the dilution water for 48 h. Information is not presently available to provide similar guidance
E724 − 21
for the mussel. Natural salt water varies in quality enough that, even though it is usually acceptable, occasionally it might be toxic
to embryos or larvae if, for example, dinoflagellates are present certain toxic algae species are present such as some dinoflagellates
(12).
8.1.2 The quality of the dilution water should be uniform enough that the brood stock is held and conditioned and the test is
conducted in water of the same quality. In particular, the salinity should always be between 18 and 32 g/kg 34 g/kg or parts per
thousand (ppt)(13) and within a test should not vary by more than 1 g/kgppt among treatments or any renewals during a test. When
a test is conducted on an effluent, brine, drilling mud, or other material that affects salinity when mixed with dilution water, it might
be desirable to adjust salinity by adding sea salt (see 8.2.1.2) to raise the salinity or by adding distilled or deionized water (see
8.2.1.2) to lower the salinity. If salts are added, the adjusted material should be well mixed and allowed to equilibrate for minimum
of 2 h with gentle aeration (24 h preferred) and salinity confirmed as salts dissolve. The addition of artificial salts can produce
artifactual toxicity that is ameliorated with aging. If the salinity is adjusted, an additional salt control, consisting of dilution water
diluted with distilled water to the salinity of the sample, and then adjusted back to the appropriate salinity with artificial salts should
be tested.
8.1.3 The dilution water should not unnecessarily affect results of tests because of such things as sorption or complexation of test
material. Therefore, except as per 8.1.4, concentrations of both total organic carbon (TOC) and particulate matter should be less
than 5 mg/L in the dilution water. The concentrations of both TOC and particulate matter should be greater than 5 mg/L in the water
in which the brood stock is held and conditioned in order to provide adequate food for the brood stock.
8.1.4 If it is desired to study the effect of an environmental factor such as TOC, particulate matter, or dissolved oxygen on the
results of a test, it is necessary to use a water that is naturally or artificially high in TOC or particulate matter or low in dissolved
oxygen. If such a water is used, it is important that adequate analyses be performed to characterize the water and that a comparable
test be available or be conducted in a more usual dilution water to facilitate interpretation of the results obtained in the
specalspecial water.
8.2 Source:
8.2.1 Reconstituted Water:
8.2.1.1 Use of a reconstituted water is often not worth the effort for tests starting with embryos of bivalve molluscs because of
(a) the large volume needed for conditioning the brood stock, (b) the necessity of providing adequate food for the brood stock (see
10.5.810.4.8), and (c) frequently poor survival and development of the embryos and resulting larvae. Commercially available sea
salts have been used successfully in bivalve embryo testing given proper conditioning of the water. Be sure to only use salts that
are fast-dissolving and closely match the ion mix found in natural seawater. A trial test with any new brand or batch of salt is
recommended prior to any testing of samples when feasible. Additionally, commercially available sea salts should be free of
ethylenediaminetetraacetic acid (EDTA) or other chelating agents that may remove or mask toxicity in a sample that is being tested.
8.2.1.2 Reconstituted water is prepared by adding a commercially avaiable sea salt or specified amounts of reagent-grade
chemicals to high quality water (14) with (a) conductivity approximately <1 μΩ/cmμS/cm and (b) either TOC <2 mg/L or chemical
oxygen demand (COD) <5 mg/L. A formula for reconstituted water acceptable for use with bivalves is given in Table 1. Acceptable
water for dissolution of sea salts can usually be prepared by using a properly operated deionization, distillation, or reverse osmosis
unit. Conductivity should be measured on each batch and TOC or COD should be measured at least twice a year and whenever
substantial changes might be expected. If the water is prepared from a surface water, TOC or COD should be measured on each
batch. Problems have been encountered with some species in reconstituted salt water, but sometimes these problems have been
overcome by conditioning (aging) and aerating the reconstituted water.
8.2.2 Natural Dilution Water—If natural salt water is used, it should be obtained from an uncontaminated, uniform quality source.
The quality of saline well water is usually more uniform than that of a saline surface water. If a surface water is used, it should
be obtained from an area known to support a healthy, naturally reproducing population of bivalves. The water intake should be
positioned (for example, about 1 m below the surface) to minimize fluctuations in quality and the possibility of contamination and
to maximize the concentration of dissolved oxygen and healthy phytoplankton (see 10.5.810.4.8). A specially designed system is
usually necessary to obtain salt water from a natural water source (see Guide E729).
8.2.3 Chlorinated water should not be used as, or in the preparation of, saline dilution water because chlorine-produced oxidants
“Reagent Chemicals, American Chemical Society Specifications,” Am. Chemical Soc., Washington, DC. For suggestions on the testing of reagents not listed by the
American Chemical Society, see “Analar Standards for Laboratory U.K. Chemicals,” BDH Ltd., Poole, Dorset, and the “United States Pharmacopeia.”
E724 − 21
TABLE 1 Reconstituted Salt Water (14)
NOTE 1—Add the following reagent-grade chemicals in the amounts
and order listed to 890 mL of water. Each chemical must be dissolved
A
before the next is added.
Chemical Amount
B
NaF 3 mg
SrCl ·6H O 20 mg
2 2
H BO 30 mg
3 3
KBr 100 mg
KCl 700 mg
CaCl ·2H O 1470 mg
2 2
Na SO 4000 mg
2 4
MgCl ·6H O 10780 mg
2 2
NaCl 23500 mg
Na SiO ·H O 20 mg
2 3 2
NaHCO 200 mg
A
If the resulting solution is diluted to 1 L, the salinity should be 34 ± 0.5 g/kg. The
reconstituted water should be stripped of trace metals (15). If necessary, the water
should be diluted to the desired salinity at time of use.
B
The NaF should be omitted for tests with C. gigas and included for tests withC.
virginica. Its value for or detriment toM. mercenaria andM. edulisspp. is unknown.
are quite toxic to embryos and larvae of bivalve molluscs (7). Dechlorinated water should be used only as a last resort because
dechlorination is often incomplete. Sodium bisulfite is probably better for dechlorinating water than sodium sulfite and both are
more reliable than carbon filters, especially for removing chloramines (15). Some organic chloramines, however, react slowly with
sodium bisulfite (16). In addition to residual chlorine, municipal drinking water often contains high concentrations of copper, lead,
zinc, and fluoride, and quality is often rather variable. The concentrations of most metals can usually be reduced by using a
chelating resin (17), but use of a different dilution water might be preferable.
8.3 Treatments:
8.3.1 Dilution water should be aerated intensively for 24 to 48 h by such means as air stones, surface aerators, or column aerators
(18) before addition of test material. To prevent contamination with undesirable bacterial species during aeration, the air used
should be filtered through a 0.22-μm bacterial filter, the container should be covered, and aeration should not last for more than
48 h. Adequate aeration will bring pH and concentrations of dissolved oxygen and other gases into equilibrium with air and
minimize oxygen demand and concentrations of volatiles. The concentration of dissolved oxygen in dilution water should be
between 90 and 100 % of saturation (19) to help ensure that dissolved oxygen concentrations are acceptable in test chambers.
Supersaturation by dissolved gases, which can be caused by heating the dilution water, should be avoided to prevent gas-bubble
disease (18, 20).
8.3.2 The salinity and pH of dilution water may be adjusted by addition of appropriate reagent-grade chemicals, sea salt
(especially to prevent excessive decreases in salinity; see 8.1.2), acid, base, and deionized or distilled water, if it has been shown
that the addition does not cause adverse effects on embryos, larvae, and adults of the test species at the concentration used.
8.3.3 Except possibly when holding and conditioning adult bivalve molluscs (see 10.5.610.4.6), filtration through bag, sand, sock,
or depth-type (honeycomb) cartridge filters may be used to keep the concentration of particulate matter acceptably low (see 8.1.3)
and as a pretreatment before ultraviolet sterilization or filtration through a finer filter.
8.3.4 Water that might be contaminated with facultative pathogens may be passed through a properly maintained ultraviolet
sterilizer (21) equipped with an intensity meter and flow controls, or passed through a filter effective to 0.45 μm or less.
8.3.5 Water from a surface-water source should be passed through a graded series of filters, the finest effective to 1.0 μm or less
to remove embryos and larvae of bivalve molluscs, parasites and predators. If bacteria are to be removed by filtration, a filter
effective to 0.45 μm or less must be used.
8.3.6 Filtration through activated carbon may be used to remove toxic algal exocrines and other organic chemicals.
8.4 Characterization:
8.4.1 The following items should be measured at least twice each year and more often if such measurements have not been made
E724 − 21
semiannually for at least 2 years or if a surface water is used: salinity (or chlorinity), pH, particulate matter, TOC,
organophosphorus pesticides, organic chlorine (or organochlorine pesticides, plus PCBs), chlorinated phenoxy herbicides,
ammonia, cyanide, sulfide, bromide, fluoride, iodide, nitrate, phosphate, sulfate, calcium, magnesium, potassium, aluminum,
arsenic, beryllium, boron, cadmium, chromium, cobalt, copper, iron, lead, manganese, mercury, molybdenum, nickel, selenium,
silver, and zinc.
8.4.2 For each method used (see 12.2), the detection limit should be below either (a) the concentration in the dilution water or
(b) the lowest concentration that has been shown to unacceptably affect embryos, larvae, or adults of saltwater bivalve molluscs
(22).
9. Test Material
9.1 General—The Test materials may include a range of sample types such as effluents, materials or products, specific chemicals,
solvents, oils, surface waters, drilling fluids, stormwater, and sediments (see Guide E1023). For chemical or product testing studies
the test material should be reagent-grade or better, unless a test on a formulation, commercial product, or technical-grade or
use-grade material is specifically needed. Before a test is begun, the following should be known about the test material:
9.1.1 Identities and concentrations of major ingredients and major impurities, that is, impurities that constitute more than about
1 % of the material,
9.1.2 Solubility and stability in the dilution water,
9.1.3 Measured or estimated acute toxicity to an aquatic species, preferably the test species or another bivalve mollusc,
9.1.4 Precision and bias of the analytical method at the planned concentration(s) of the test material, if the test concentrations are
to be measured,
9.1.5 Estimate of toxicity to humans, and
9.1.6 Recommended handling procedures (see 6.1).
9.2 Stock Solution:
9.2.1 In some cases the test material can be added directly to the dilution water, but usuallyoften it is dissolved in a solvent to form
a stock solution that is then added to the dilution water. If a stock solution is used, the concentration and stability of the test material
in it should be determined before the beginning of the test. If the test material is subject to photolysis, the stock solution should
be shielded from light.
9.2.2 Except possibly for tests on hydrolyzable, oxidizable, and reducible materials, the preferred solvent is dilution water,
although filtration or sterilization, or both, of the water might be necessary. If the salinity of the dilution water will not be affected,
deionized or distilled water may be used. Several techniques have been specifically developed for preparing aqueous stock
solutions of slightly soluble materials (23). The minimum necessary amount of a strong acid or base may be used in the preparation
of an aqueous stock solution, but such reagents might affect the pH of test solutions appreciably. Use of a more soluble form of
the test material, such as chloride or sulfate salts of organic amines, sodium or potassium salts of phenols and organic acids, and
chloride or nitrate salts of metals, might affect the pH more than use of the minimum necessary amount of a strong acid or base.
9.2.3 If a solvent other than dilution water is used, its concentration in test solutions should be kept to a minimum and should be
low enough that it does not affect the test species. Because of its low toxicity to aquatic animals (24), low volatility, and high ability
to dissolve many organic chemicals, triethylene glycol is often a good organic solvent for preparing stock solutions. Other
water-miscible organic solvents such as methanol, ethanol, and acetone may also be used, but they might stimulate undesirable
growths of microorganisms, and acetone is also quite volatile. If an organic solvent is used, it should be reagent-grade or better
and its concentration in any test solution should not exceed 0.5 mL/L. A surfactant should not be used in the preparation of a stock
solution because it might affect the form and toxicity of the test material in the test solutions. (These limitations do not apply to
any ingredient of a mixture, formulation, or commercial product, unless an extra amount of solvent is used in the preparation of
the stock solution.)
9.2.4 If no solvent other than water is used, only a dilution-water control must be included in the test. For oysters, at least 70 %
of the embryos introduced into the control treatment must result in live larvae with completely developed shells (normal) at the
E724 − 21
end of the test and at least 90 % of all embryos introduced should be alive. For hard clams, at least 60 % of the embryos introduced
must result in live larvae with completely developed shells at the end of the test, and at least 90 % of all embryos introduced should
be alive. These values for live larvae with completely developed shells reflect apparently natural failure of up to 30 to 40 % of the
larvae of oysters and hard clams to develop a shell. These stipulations are species-specific and might be too high or too low for
the blue mussel as well asFor mussels, at least 90 % of the embryos introduced into the control treatment must result in live larvae
with completely developed shells (normal) at the end of the test and at least 50 % of all embryos introduced should be alive. These
criteria values for mussels are consistent with USEPA guidance bivalves (25not ).covered in this guide.
9.2.5 If a solvent other than water is used and the concentration of solvent is the same in all test solutions that contain test material,
a solvent control, containing the same concentration of solvent as the test solutions and using solvent from the same batch used
to make the stock solution, must be included in the test. In addition, a dilution-water control should be included in the test. The
number of embryos that result in live larvae with completely developed shells (normal) at the end of the test must be at least 70 %
of the initial number in the solvent control for oysters and 60 % for hard clams (see 9.2.4). If a dilution-water control is included
in the test, the number of embryos that result in live larvae with completely developed shells at the end of the test should be at
least 70 % of the initial number in the dilution-water control for oysters and 60 % for hard clams.
9.2.6 If a solvent other than water is used and the concentration of solvent is not the same in all test solutions that contain test
material, both a solvent control, containing the highest concentration of solvent present in any other treatment and using solvent
from the same batch used to make the stock solution, and a dilution-water control must be included in the test. The number of
embryos that result in live larvae with completely developed shells at the end of the test must be at least 70 % of the initial number
in the solvent control and in the dilution-water control for oysters and 60 % for hard clams (see 9.2.4).
9.2.7 If a solvent other than water is used to prepare a stock solution, it might be desirable to conduct simultaneous tests on the
test material using two chemically unrelated solvents or two different concentrations of the same solvent to obtain information
concerning possible effects of the solvent on the results of the test.
9.3 Test Concentration(s):
9.3.1 If the test is intended to allow calculation of an EC50, the test concentrations (see 11.1.1.1) should bracket the predicted
EC50. The prediction might be based on the results of a test with the same or a similar test material and the same or a similar
species. If a useful prediction is not available, it is usually desirable to conduct a range-finding test in which embryos and resulting
larvae are exposed for a total of 48 h to a control and three to five concentrations of the test material that differ by a factor of ten.
The greater the similarity between the range-finding test and the actual test, the more useful the range-finding test will be.
9.3.1.1 If necessary, concentrations above solubility should be used because organisms in the real world are sometimes exposed
to concentrations above solubility. The use of concentrations that are more than ten times greater than solubility is probably not
worthwhile. With some test materials, it might be found that concentrations above solubility do not kill or affect a greater
percentage of test organisms than does the concentration that is the solubility limit; such information is certainly worth knowing.
9.3.1.2 In some situations, usually related to regulatory activities, it is only necessary to determine whether (a) a specific
concentration of test material is acutely toxic to embryos or larvae of the test species or (b) the EC50 is above or below a specific
concentration. For example, the specific concentration might be the concentration occurring in a surface water, the concentration
resulting from the direct application of the material to a body of water, or the solubility limit of the material in water. When there
is only interest in a specific concentration, it is often only necessary to test that concentration plus a control (see 11.1.1.2), and it
is not necessary to determine an EC50.
10. Test Organisms
10.1 Species—Whenever possible, either Pacific oysters (Crassostrea gigas), Eastern oysters (C. virginica), quahogs or hard clams
(Mercenaria mercenaria), or blue mussels (Mytilus edulis) should be used as the test species. These species were selected on the
basis of availability, commercial importance, past successful use, and ease of handling in the laboratory. Their use is encouraged
to increase comparability of results and availability of much information about a few species rather than little information about
many species. The species used should be identified by using an appropriate taxonomic key.
10.2 Age—The test must be begun with embryos within 4 h after fertilization when the embryos are in the 2-, 4-, and 8-cell stages.
E724 − 21
TABLE 2 Recommended Temperature (°C)
Never to
Species Holding Conditioning Induction be Test
Exceeded
Crassostrea gigas 14–15 20 25–32 32 20
Crassostrea virginica 14–15 20–25 25–32 32 25
Mercenaria 14–15 20–25 25–32 32 25
mercenaria
Mytilus edulis 8 12–14 15–20 20 16
Mytilus spp. 8 12–15 12–18 25 15 or
10.3 Source—Embryos used to start a test should be obtained from females and males that had been maintained for at least 2 weeks
in the dilution water in the laboratory before they were stimulated to spawn.
10.3 Handling—Organisms should be handled as little as possible. When handling is necessary, it should be done carefully, gently,
and quickly, so that organisms are not unnecessarily stressed. Adults that are injured during handling should be discarded. Gentle
cleaning of the organisms is however recommended to remove other attached organisms and debris prior to sue for testing.
Equipment used to transfer embryos of bivalve molluscs should be cleaned between uses by washing with detergent and rinsing
with dilute acid and distilled or deionized water. Hands should be washed before and after handling brood stock.
10.4 Brood Stock Source and Condition:
10.4.1 For any one test or a series of related tests, all females and males in the brood stock must be collected from the same
location, which should be known precisely. Brood stock may be obtained from a commercial source only if the original specific
collection location of the bivalves can be identified. Aquacultured shellfish that forced into triploidy must not be utilized. To
minimize the possibility of genetic or physiological adaptation to chemicals or aberrant water quality, organisms should be
collected from a location that is not subject to obvious point or nonpoint source pollution and has water that is comparable in
quality to the water that will be used for holding, conditioning, and testing. Large Gravid adult individuals with well developed
normal shells should be obtained. Molluscs possessing high condition factors (2526) (see Note 1), indicative of good food supply
and water quality throughout the year, should be obtained be
...


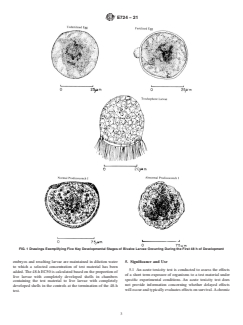
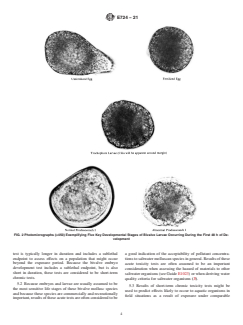



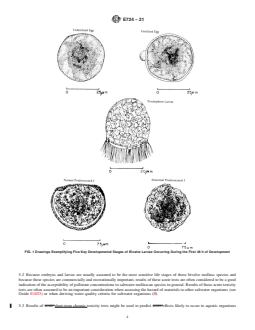
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...