ASTM E3238-20
(Test Method)Standard Test Method for Quantitative Measurement of the Chemoattractant Capacity of a Nanoparticulate Material in vitro
Standard Test Method for Quantitative Measurement of the Chemoattractant Capacity of a Nanoparticulate Material <emph type="bdit">in vitro</emph>
SIGNIFICANCE AND USE
5.1 This test method will assess whether the test nanoparticulate material has chemoattractant activity.
5.2 This test method will provide a rapid and quantitative measure of the ability of nanoparticulate material to recruit immune cells.
5.3 Recruitment of immune cells by chemotaxis plays an important part in all phases of both humoral and cell-mediated immune responses.
5.4 Testing the capacity of a nanoparticulate material to recruit immune cells in vitro helps in predicting the influence of such material on the immune cell response.
SCOPE
1.1 This test method provides a protocol for rapid and quantitative measurement of the chemoattractant capacity of a nanoparticulate material (nanoparticles and their aggregates and agglomerates).
1.2 Immune cells recruitment (by chemotaxis) plays a central role in the immune system function especially in the inflammatory process.
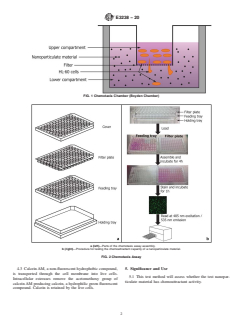
1.3 This test method uses an in vitro model. In this model, peripheral blood human acute promyelocytic leukemia cells HL-60 are separated from control chemoattractant or test nanoparticulate material by a 3-µm pore size filter; the cell migration through the filter is monitored and quantified using the fluorescent dye calcein AM (Figs. 1 and 2).
FIG. 1 Chemotaxis Chamber (Boyden Chamber)
FIG. 2 Chemotaxis Assay
a (left)—Parts of the chemotaxis assay assembly.
b (right)—Procedure for testing the chemoattractant capacity of a nanoparticulate material.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Dec-2019
- Technical Committee
- E56 - Nanotechnology
- Drafting Committee
- E56.08 - Nano-Enabled Medical Products
Relations
- Effective Date
- 01-Oct-2018
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Apr-2012
- Effective Date
- 01-Jun-2010
- Refers
ASTM F1903-10 - Standard Practice for Testing For Biological Responses to Particles <i>In Vitro</i> - Effective Date
- 01-Jun-2010
- Effective Date
- 01-Apr-2009
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Nov-2005
- Refers
ASTM F1903-98(2003) - Standard Practice for Testing for Biological Responses to Particles In Vitro - Effective Date
- 01-Nov-2003
- Effective Date
- 10-Apr-2003
- Effective Date
- 10-Jun-1998
- Effective Date
- 10-Apr-1998
Overview
ASTM E3238-20 is the internationally recognized standard test method for the quantitative measurement of the chemoattractant capacity of a nanoparticulate material in vitro. Developed by ASTM International, this standard provides a comprehensive protocol for researchers and product developers to assess whether a nanoparticle or its aggregates can recruit immune cells by chemotaxis. The method leverages a rapid and quantitative in vitro model, offering vital insights into the potential immunological effects of nanomaterials, particularly in the context of both humoral and cell-mediated immune responses.
Nanoparticles are increasingly used in medical, pharmaceutical, and material science applications, making reliable immune cell recruitment testing essential for evaluating safety and efficacy. ASTM E3238-20 enables standardized assessment, compliance, and comparison across laboratories.
Key Topics
Chemoattractant Activity Measurement
The standard outlines a protocol for determining whether a nanoparticulate material exhibits chemoattractant behavior, quantitatively evaluating the movement of immune cells in its presence.In Vitro Chemotaxis Assay
Using a well-established Boyden chamber setup, human HL-60 cells are separated from the nanoparticulate test sample by a 3-μm pore size filter. Cell migration is monitored via the fluorescent dye calcein AM, which accumulates in live, migrated cells.Quantitative and Rapid Results
The protocol delivers rapid and reproducible quantitative results for immune cell recruitment, supporting decision-making during preclinical evaluation of nanomaterials.Relevance to Immune Response and Inflammation
Chemotaxis - the directed movement of immune cells - is central to both innate and adaptive immunity, especially in inflammation. Measuring this property aids in predicting the immunomodulatory potential of test materials, which is crucial for applications involving nanomedicine, drug delivery, and immunotoxicological studies.Control and Precision Requirements
The standard specifies the necessary controls (positive, negative, background) and provides criteria for accepting data based on precision (coefficient of variation below specified thresholds).
Applications
Nanomedicine and Drug Delivery
Evaluate the immune cell recruitment potential of nanoparticle-based therapeutics, vaccines, or drug carriers prior to clinical development.Immunotoxicology Screening
Screen nanomaterials and formulations for unexpected immune activation or suppression, aiding in regulatory submissions and safety assessments.Research and Development
Compare different nanoparticle materials, coatings, or formulations to determine their impact on immune cell recruitment, supporting optimized material design.Quality Assurance
Employ standardized, repeatable procedures for batch-to-batch consistency checks in nanomaterial manufacturing.Academic Research
Support studies on the mechanisms of nanomaterial-immune system interaction, and explore the relationship between particle characteristics (size, charge, composition) and immune responses.
Related Standards
For a robust evaluation of nanoparticulate materials and their biological effects, ASTM E3238-20 should be considered alongside the following related standards:
- ASTM E2490 - Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Photon Correlation Spectroscopy (PCS)
- ASTM E2834 - Guide for Measurement of Particle Size Distribution of Nanomaterials in Suspension by Nanoparticle Tracking Analysis (NTA)
- ASTM F1877 - Practice for Characterization of Particles
- ASTM F1903 - Practice for Testing for Cellular Responses to Particles in vitro
These referenced standards enhance material characterization and the interpretation of biological testing results, supporting a comprehensive approach to nanomaterial safety and performance evaluation.
Keywords: chemoattractant, chemotaxis, immune cell recruitment, nanomaterial, nanoparticle, in vitro assay, ASTM E3238-20, immunotoxicity, nanomedicine standard, quantitative measurement, HL-60 cells
Buy Documents
ASTM E3238-20 - Standard Test Method for Quantitative Measurement of the Chemoattractant Capacity of a Nanoparticulate Material <emph type="bdit">in vitro</emph>
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E3238-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Quantitative Measurement of the Chemoattractant Capacity of a Nanoparticulate Material <emph type="bdit">in vitro</emph>". This standard covers: SIGNIFICANCE AND USE 5.1 This test method will assess whether the test nanoparticulate material has chemoattractant activity. 5.2 This test method will provide a rapid and quantitative measure of the ability of nanoparticulate material to recruit immune cells. 5.3 Recruitment of immune cells by chemotaxis plays an important part in all phases of both humoral and cell-mediated immune responses. 5.4 Testing the capacity of a nanoparticulate material to recruit immune cells in vitro helps in predicting the influence of such material on the immune cell response. SCOPE 1.1 This test method provides a protocol for rapid and quantitative measurement of the chemoattractant capacity of a nanoparticulate material (nanoparticles and their aggregates and agglomerates). 1.2 Immune cells recruitment (by chemotaxis) plays a central role in the immune system function especially in the inflammatory process. 1.3 This test method uses an in vitro model. In this model, peripheral blood human acute promyelocytic leukemia cells HL-60 are separated from control chemoattractant or test nanoparticulate material by a 3-µm pore size filter; the cell migration through the filter is monitored and quantified using the fluorescent dye calcein AM (Figs. 1 and 2). FIG. 1 Chemotaxis Chamber (Boyden Chamber) FIG. 2 Chemotaxis Assay a (left)—Parts of the chemotaxis assay assembly. b (right)—Procedure for testing the chemoattractant capacity of a nanoparticulate material. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method will assess whether the test nanoparticulate material has chemoattractant activity. 5.2 This test method will provide a rapid and quantitative measure of the ability of nanoparticulate material to recruit immune cells. 5.3 Recruitment of immune cells by chemotaxis plays an important part in all phases of both humoral and cell-mediated immune responses. 5.4 Testing the capacity of a nanoparticulate material to recruit immune cells in vitro helps in predicting the influence of such material on the immune cell response. SCOPE 1.1 This test method provides a protocol for rapid and quantitative measurement of the chemoattractant capacity of a nanoparticulate material (nanoparticles and their aggregates and agglomerates). 1.2 Immune cells recruitment (by chemotaxis) plays a central role in the immune system function especially in the inflammatory process. 1.3 This test method uses an in vitro model. In this model, peripheral blood human acute promyelocytic leukemia cells HL-60 are separated from control chemoattractant or test nanoparticulate material by a 3-µm pore size filter; the cell migration through the filter is monitored and quantified using the fluorescent dye calcein AM (Figs. 1 and 2). FIG. 1 Chemotaxis Chamber (Boyden Chamber) FIG. 2 Chemotaxis Assay a (left)—Parts of the chemotaxis assay assembly. b (right)—Procedure for testing the chemoattractant capacity of a nanoparticulate material. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E3238-20 is classified under the following ICS (International Classification for Standards) categories: 07.120 - Nanotechnologies; 11.020.20 - Medical science. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E3238-20 has the following relationships with other standards: It is inter standard links to ASTM F1903-18, ASTM F1877-16, ASTM E2834-12, ASTM F1877-05(2010), ASTM F1903-10, ASTM E2490-09, ASTM E2490-08, ASTM F1877-05e1, ASTM F1877-05, ASTM F1903-98(2003), ASTM F1877-98(2003)e1, ASTM F1903-98, ASTM F1877-98. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E3238-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E3238 − 20
Standard Test Method for
Quantitative Measurement of the Chemoattractant Capacity
of a Nanoparticulate Material in vitro
This standard is issued under the fixed designation E3238; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope of Nanomaterials in Suspension by Photon Correlation
Spectroscopy (PCS)
1.1 This test method provides a protocol for rapid and
E2834Guide for Measurement of Particle Size Distribution
quantitative measurement of the chemoattractant capacity of a
of Nanomaterials in Suspension by NanoparticleTracking
nanoparticulate material (nanoparticles and their aggregates
Analysis (NTA)
and agglomerates).
F1877Practice for Characterization of Particles
1.2 Immune cells recruitment (by chemotaxis) plays a
F1903Practice for Testing for Cellular Responses to Par-
central role in the immune system function especially in the
ticles in vitro
inflammatory process.
1.3 This test method uses an in vitro model. In this model, 3. Terminology
peripheral blood human acute promyelocytic leukemia cells
3.1 Acronyms:
HL-60 are separated from control chemoattractant or test
3.1.1 BSA—bovine serum albumin
nanoparticulate material by a 3-µm pore size filter; the cell
3.1.2 calcein AM—calcein acetoxymethyl ester
migration through the filter is monitored and quantified using
the fluorescent dye calcein AM (Figs. 1 and 2). 3.1.3 C —maximum serum concentration
max
3.1.4 CV—coefficient of variation
1.4 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this
3.1.5 FBS—fetal bovine serum
standard.
3.1.6 FU—fluorescence units
1.5 This standard does not purport to address all of the
3.1.7 g—relative centrifugal force
safety concerns, if any, associated with its use. It is the
3.1.8 NC—negative control
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
3.1.9 PBS—phosphate buffered saline
mine the applicability of regulatory limitations prior to use.
3.1.10 PC—positive control
1.6 This international standard was developed in accor-
3.1.11 PK—pharmacokinetic
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the 3.1.12 RBC—reagent background control
Development of International Standards, Guides and Recom-
3.1.13 RPMI—Roswell Park Memorial Institute
mendations issued by the World Trade Organization Technical
3.1.14 SD—standard deviation
Barriers to Trade (TBT) Committee.
3.1.15 SM—starvation medium
2. Referenced Documents
3.1.16 TS—test sample (of nanoparticulate material dis-
2.1 ASTM Standards: solved in starvation medium)
E2490Guide for Measurement of Particle Size Distribution
3.1.17 U—units
4. Summary of Test Method
This test method is under the jurisdiction of ASTM Committee E56 on
Nanotechnology and is the direct responsibility of Subcommittee E56.08 on
4.1 This test method describes a protocol for assessing and
Nano-Enabled Medical Products.
measuring the chemoattractant capacity of nanoparticulate
Current edition approved Jan. 1, 2020. Published January 2020. DOI: 10.1520/
material.
E3238-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
4.2 The migration of cells through a filter towards the
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
nanoparticulate material is monitored and quantified after
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. staining with calcein AM dye.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E3238 − 20
FIG. 1 Chemotaxis Chamber (Boyden Chamber)
a (left)—Parts of the chemotaxis assay assembly.
b (right)—Procedure for testing the chemoattractant capacity of a nanoparticulate material.
FIG. 2 Chemotaxis Assay
4.3 CalceinAM, a non-fluorescent hydrophobic compound, 5. Significance and Use
is transported through the cell membrane into live cells.
5.1 This test method will assess whether the test nanopar-
Intracellular esterases remove the acetomethoxy group of
ticulate material has chemoattractant activity.
calceinAM producing calcein, a hydrophilic green fluorescent
compound. Calcein is retained by the live cells.
E3238 − 20
5.2 This test method will provide a rapid and quantitative 9.6 Biohazard safety cabinetapprovedforLevelIIhandling
measure of the ability of nanoparticulate material to recruit of biological material.
immune cells.
9.7 Inverted microscope.
5.3 Recruitment of immune cells by chemotaxis plays an
9.8 Vortex mixer.
important part in all phases of both humoral and cell-mediated
9.9 Hemocytometer.
immune responses.
9.10 Fluorescence plate reader.
5.4 Testing the capacity of a nanoparticulate material to
recruit immune cells in vitro helps in predicting the influence
10. Preparation of Media, Calcein AM, Test Samples
of such material on the immune cell response.
and Controls
6. Materials
10.1 Three solutions of each nanoparticulate material test
concentration (referred to as test samples) and three solutions
6.1 Pipettes covering the range of 0.05 to 10 mL.
of each control (SM, PC, and NC) shall be independently
6.2 96-well filter plate, 3.0-µm pore size polycarbonate
prepared.
membrane, clear, sterile.
10.2 Preparation of Complete RPMI-1640 Medium—The
6.3 96-well feeding tray, clear, sterile (culture tray).
complete RPMI-1640 medium is a solution of RPMI-1640
6.4 Holding tray.
medium (basal medium) supplemented with 20 % FBS (heat-
inactivated), 4 mM L-glutamine, 1.5 g/L sodium bicarbonate,
6.5 Cover for holding tray.
50 µM β-mercaptoethanol, 100 U/mL penicillin, and 100
6.6 96-microwell clear bottom plates suitable for
µg/mL streptomycin sulfate. The medium should be stored at
fluorescent-based assays.
2–8°C protected from light for no longer than 1 month. Before
6.7 Polypropylene tubes, 5 and 15 mL.
use, warm in a water bath.
6.8 Cell culture flasks.
10.3 Preparation of Starvation Medium (SM)—The SM is a
solution of RPMI-1640 medium (basal medium) supplemented
7. Cell Line
with 0.2 % BSA, 4 mM L-glutamine, 1.5 g/L sodium
bicarbonate, 50 µM β-mercaptoethanol, 100 U/mL penicillin,
7.1 Peripheral blood human acute promyelocytic leukemia
and 100 µg/mL streptomycin sulfate. The medium is stored at
cells HL-60 (ATCC CCL-240 ).
2–8°C protected from light for no longer than 1 month. Before
use, warm in a water bath. Prepare three solutions of SM (see
8. Reagents
10.1).
8.1 Phosphate buffered saline (PBS), pH 7.4.
10.4 Postive Control (PC)—On the day of the experiment,
8.2 Bovine serum albumin (BSA), endotoxin-free and suit-
prepare a solution of RPMI-1640 medium supplemented with
able for cell culture.
20 % heat-inactivated FBS and 0.2 % BSA. Repeat twice to
8.3 Fetal bovine serum (FBS).
obtain three independently prepared solutions (see 10.1).
8.4 Commercial RPMI-1640 medium (basal medium).
10.5 Negative Control (NC)—Use PBS as a NC.
8.5 L-glutamine, 4 mM.
10.6 Calcein AM Working Solution—Dilute the calceinAM
1-mM stock solution in prewarmed (37°C) PBS to a final
8.6 Sodium bicarbonate.
concentration of 4 µM. The working dilution shall be prepared
8.7 β-mercaptoethanol, 50 µM.
immediately prior to use.
8.8 Calcein AM, 1 mM.
10.7 Specification of Nanoparticulate Material Test
8.9 Trypan blue solution.
Samples—The selected nanoparticulate material must be fully
dispersed in SM. This assay requires a minimum of 2.3 mLof
8.10 Penicillin.
nanoparticulate material dissolved/resuspended in SM. (This
8.11 Streptomycin sulfate.
volumeiscalculatedfromthenumberandconcentrationoftest
samplespreparedasspecifiedbelow,includingreplicates.)The
9. Apparatus
following factors shall be considered in choosing the nanopar-
9.1 Centrifuge.
ticulate material concentration:
(1)Dispersibility, stability, and homogeneity of the nano-
9.2 Water bath set at 37°C.
particulate material in a biocompatible buffer (that is, SM),
9.3 Refrigerator, 2–8°C.
(2)Maintaining the pH in the physiological range, and
9.4 Freezer, –20°C.
(3)Stability of nanoparticulate material during testing.
Before testing, the nanoparticulate material shall be charac-
9.5 Cell culture incubator with 5 % carbon dioxide (CO )
terized (for example, size, size distribution, and charge) under
and 95 % humidity.
physiological conditions in accordance with with standard
methods such as those recommended in Guides E2490 and
A trademark of ATCC in Manassas, VA. E2834 for nanomaterials and Practices F1877 and F1903 for
E3238 − 20
powders. In the absence of data from PK studies (see the 11.5 Place the feeding tray into the holding tray. Gently
following sentence), the highest concentration of nanoparticu- placethefilterplate(11.4)ontopofthefeedingtraycontaining
late material was selected to be 1.0 mg/mL. (When the the controls and test samples (11.3) and add the cover. Avoid
nanoparticulatematerial’splasmaconcentration C isknown shaking or tilting the chemotaxis assay assembly as it will
max
from in vivo PK studies, the highest concentration tested in disturb the concentration gradient.
vitro is chosen as 10×, 30×, or 100× of that C .)
max
11.6 Place the assembly in a cell culture incubator and
10.8 Preparation of Nanoparticulate Material Test incubate for4hwith5%CO and 95 % humidity. During
Samples—Prepare a stock solution containing nanoparticulate incubation, pre-warm PBS to 37°C and equilibrate calceinAM
material dissolved in SM at a concentration of 1.0 mg/mL. stock solution to room temperature.
Prepa
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...