ASTM E2475-23
(Guide)Standard Guide for Process Understanding Related to Pharmaceutical Manufacture and Control
Standard Guide for Process Understanding Related to Pharmaceutical Manufacture and Control
SCOPE
1.1 The purpose of this guide is to establish a framework and context for process understanding for pharmaceutical manufacturing using the principles of quality by design (QbD) (Juran, 1992;2 ICH Q8). The framework is applicable to both drug substance (DS) and drug product (DP) manufacturing. High (detailed) level process understanding can be used to facilitate production of product which consistently meets required specifications. It can also play a key role in continual process improvement efforts.
1.2 Process Analytical Technology (PAT) is one element that can be used for achieving control over those inputs determined to be critical to a process. It is important for the reader to recognize that PAT is defined as:
“…a system for designing, analyzing, and controlling manufacturing through timely measurements (i.e., during processing) of critical quality and performance attributes of raw and in process materials and processes, with the goal of ensuring final product quality. It is important to note that the term analytical in PAT is viewed broadly to include chemical, physical, microbiological, mathematical, and risk analysis conducted in an integrated manner. The goal of PAT is to enhance understanding and control the manufacturing process…” (USFDA PAT)
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Nov-2023
- Technical Committee
- E55 - Manufacture of Pharmaceutical and Biopharmaceutical Products
- Drafting Committee
- E55.11 - Process Design
Relations
- Effective Date
- 15-Nov-2023
- Effective Date
- 01-Apr-2022
- Effective Date
- 01-Apr-2022
- Effective Date
- 15-Nov-2023
- Effective Date
- 15-Nov-2023
Overview
ASTM E2475-23: Standard Guide for Process Understanding Related to Pharmaceutical Manufacture and Control provides a comprehensive framework for enhancing process understanding throughout pharmaceutical manufacturing. This guide is designed to apply to both drug substance (DS) and drug product (DP) processes. It facilitates the implementation of Quality by Design (QbD) principles and encourages the integration of Process Analytical Technology (PAT) as a key component to enable robust process control and continual improvement. Adhering to this standard helps manufacturers consistently produce pharmaceutical products that meet required specifications and supports initiatives for ongoing quality enhancement.
Key Topics
- Process Understanding: The standard defines process understanding as the logical, scientific, and statistical comprehension of how process inputs (such as materials, parameters, and environment) affect product quality attributes. High-level understanding helps minimize variability and risk.
- Quality by Design (QbD): Emphasizes a science- and risk-based approach to design processes that consistently deliver high-quality pharmaceuticals, referencing ICH Q8 and related guidelines.
- Process Analytical Technology (PAT): PAT is highlighted as a system that enables real-time monitoring and control of critical quality and performance attributes of raw and in-process materials, using integrated chemical, physical, microbiological, mathematical, and risk analysis tools.
- Levels of Process Knowledge: The document outlines a spectrum-descriptive, correlative, causal, mechanistic, and first principles knowledge-each adding depth to the understanding and control of manufacturing processes.
- Lifecycle Approach: The guide underscores the need for continual process review throughout product and process life cycles, promoting ongoing improvements and adaptation to changes in scale, raw materials, equipment, or personnel.
- Risk and Uncertainty: Focuses on maintaining risk at an acceptable level for stakeholders through robust understanding, predictive control, and regular evaluation.
Applications
ASTM E2475-23 is applicable across the pharmaceutical manufacturing sector, offering value for:
- Process Design and Development: Supports structured approaches for new or existing products, facilitating robust development, validation, and scale-up activities.
- Manufacturing Control: Assists in real-time monitoring and control, leveraging PAT to reduce variability and improve consistency in batch and continuous manufacturing environments.
- Regulatory Compliance: Aligns with international and national regulatory expectations, including those from the FDA and ICH, for evidence-based process control and risk management.
- Continuous Improvement: Aids organizations in establishing programs for ongoing process monitoring, knowledge accumulation, and risk reduction to sustain and improve product quality.
- Supplier and Raw Material Management: Provides direction for managing variability and detecting adulteration within the supply chain by understanding critical quality attributes of materials.
Related Standards
ASTM E2475-23 references and aligns with several key standards and guidance documents in pharmaceutical quality and process control, including:
- ASTM E456: Terminology Relating to Quality and Statistics
- ASTM E2281: Practice for Process Capability and Performance Measurement
- ASTM E2617: Practice for Validation of Empirically Derived Multivariate Calibrations
- ICH Q8: Pharmaceutical Development
- ICH Q9: Quality Risk Management
- ICH Q10: Pharmaceutical Quality Systems
- ICH Q11: Development and Manufacture of Drug Substances
- ISO 14971: Risk Management for Medical Devices
- FDA PAT Guidance: Guidance for Industry on PAT and Innovative Pharmaceutical Manufacturing
By implementing ASTM E2475-23, organizations can foster a science-based, risk-managed, and lifecycle-oriented approach to pharmaceutical process control-enabling improved product quality, regulatory readiness, and manufacturing efficiency.
Keywords: pharmaceutical manufacture, process understanding, process analytical technology, quality by design, risk management, process control, continual improvement, ASTM E2475-23.
Buy Documents
ASTM E2475-23 - Standard Guide for Process Understanding Related to Pharmaceutical Manufacture and Control
REDLINE ASTM E2475-23 - Standard Guide for Process Understanding Related to Pharmaceutical Manufacture and Control
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E2475-23 is a guide published by ASTM International. Its full title is "Standard Guide for Process Understanding Related to Pharmaceutical Manufacture and Control". This standard covers: SCOPE 1.1 The purpose of this guide is to establish a framework and context for process understanding for pharmaceutical manufacturing using the principles of quality by design (QbD) (Juran, 1992;2 ICH Q8). The framework is applicable to both drug substance (DS) and drug product (DP) manufacturing. High (detailed) level process understanding can be used to facilitate production of product which consistently meets required specifications. It can also play a key role in continual process improvement efforts. 1.2 Process Analytical Technology (PAT) is one element that can be used for achieving control over those inputs determined to be critical to a process. It is important for the reader to recognize that PAT is defined as: “…a system for designing, analyzing, and controlling manufacturing through timely measurements (i.e., during processing) of critical quality and performance attributes of raw and in process materials and processes, with the goal of ensuring final product quality. It is important to note that the term analytical in PAT is viewed broadly to include chemical, physical, microbiological, mathematical, and risk analysis conducted in an integrated manner. The goal of PAT is to enhance understanding and control the manufacturing process…” (USFDA PAT) 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SCOPE 1.1 The purpose of this guide is to establish a framework and context for process understanding for pharmaceutical manufacturing using the principles of quality by design (QbD) (Juran, 1992;2 ICH Q8). The framework is applicable to both drug substance (DS) and drug product (DP) manufacturing. High (detailed) level process understanding can be used to facilitate production of product which consistently meets required specifications. It can also play a key role in continual process improvement efforts. 1.2 Process Analytical Technology (PAT) is one element that can be used for achieving control over those inputs determined to be critical to a process. It is important for the reader to recognize that PAT is defined as: “…a system for designing, analyzing, and controlling manufacturing through timely measurements (i.e., during processing) of critical quality and performance attributes of raw and in process materials and processes, with the goal of ensuring final product quality. It is important to note that the term analytical in PAT is viewed broadly to include chemical, physical, microbiological, mathematical, and risk analysis conducted in an integrated manner. The goal of PAT is to enhance understanding and control the manufacturing process…” (USFDA PAT) 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2475-23 is classified under the following ICS (International Classification for Standards) categories: 11.120.01 - Pharmaceutics in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2475-23 has the following relationships with other standards: It is inter standard links to ASTM E2475-10(2016), ASTM E456-13a(2022)e1, ASTM E456-13a(2022), ASTM E2968-23, ASTM E3326-22. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2475-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2475 − 23
Standard Guide for
Process Understanding Related to Pharmaceutical
Manufacture and Control
This standard is issued under the fixed designation E2475; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
2.1 ASTM Standards:
1.1 The purpose of this guide is to establish a framework
E456 Terminology Relating to Quality and Statistics
and context for process understanding for pharmaceutical
E2281 Practice for Process Capability and Performance
manufacturing using the principles of quality by design (QbD)
2 Measurement
(Juran, 1992; ICH Q8). The framework is applicable to both
E2617 Practice for Validation of Empirically Derived Mul-
drug substance (DS) and drug product (DP) manufacturing.
tivariate Calibrations
High (detailed) level process understanding can be used to
2.2 U.S. Government Publications:
facilitate production of product which consistently meets
ICH Quality Implementation Working Group Points To
required specifications. It can also play a key role in continual
Consider (R2) ICH-Endorsed Guide for ICH Q8/Q9/Q10
process improvement efforts.
Implementation
ICH Q8 Pharmaceutical Development
1.2 Process Analytical Technology (PAT) is one element
ICH Q9 Quality Risk Management
that can be used for achieving control over those inputs
ICH Q10 Pharmaceutical Quality Systems
determined to be critical to a process. It is important for the
ICH Q11 Development and Manufacture of Drug Substances
reader to recognize that PAT is defined as:
ISO 14971 Medical devices—Application of risk manage-
“{a system for designing, analyzing, and controlling manufacturing through
ment to medical devices
timely measurements (i.e., during processing) of critical quality and performance
attributes of raw and in process materials and processes, with the goal of
USFDA PAT Guidance Document, Guidance for Industry
ensuring final product quality. It is important to note that the term analytical in
PAT—A Framework for Innovative Pharmaceutical
PAT is viewed broadly to include chemical, physical, microbiological,
Manufacturing and Quality Assurance
mathematical, and risk analysis conducted in an integrated manner. The goal of
PAT is to enhance understanding and control the manufacturing process{”
(USFDA PAT)
3. Terminology
1.3 This standard does not purport to address all of the
3.1 Definitions of Terms Specific to This Standard:
safety concerns, if any, associated with its use. It is the
3.1.1 critical inputs, n—critical process parameters and
responsibility of the user of this standard to establish appro-
critical raw material attributes for a given process.
priate safety, health, and environmental practices and deter-
3.1.2 empirical, adj—any conclusion based on experimental
mine the applicability of regulatory limitations prior to use.
data and past experience, rather than on theory.
1.4 This international standard was developed in accor-
3.1.3 expert system, n—an expert system is a computer
dance with internationally recognized principles on standard-
program that simulates the judgment and behavior of a human
ization established in the Decision on Principles for the
or an organization that has expert knowledge and experience in
Development of International Standards, Guides and Recom-
a particular field.
mendations issued by the World Trade Organization Technical
3.1.3.1 Discussion—Typically, such a system contains a
Barriers to Trade (TBT) Committee.
knowledge base containing accumulated experience and a set
of rules for applying the knowledge base to each particular
This guide is under the jurisdiction of ASTM Committee E55 on Manufacture
of Pharmaceutical and Biopharmaceutical Products and is the direct responsibility of For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Subcommittee E55.11 on Process Design. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved Nov. 15, 2023. Published December 2023. Originally Standards volume information, refer to the standard’s Document Summary page on
approved in 2010. Last previous edition approved in 2016 as E2475 – 10 (2016). the ASTM website.
DOI:10.1520/E2475-23. Available from U.S. Government Printing Office Superintendent of Documents,
Juran, J., Juran on Quality by Design: The New Steps for Planning Quality Into 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
Goods and Services, Free Press, New York, N.Y., 1992. www.access.gpo.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2475 − 23
situation that is described to the program. Sophisticated expert example, finite resources, time, and money), a process will
systems can be enhanced with additions to the knowledge base typically be commissioned as soon as the degree of process
or to the set of rules. understanding is sufficient to permit operation of the process
with an acceptably low, user specified, level of risk of
3.1.4 first principles, n—a calculation is said to be from first
producing out of specification product. While it may be
principles, or ab initio, if it starts directly at the level of
appropriate to commission a process once this minimum
established laws of physics and does not make assumptions
degree of process understanding is achieved, the risk that the
such as model and fitting parameters.
process may transition out of control steadily increases over
3.1.5 mechanistic, adj—(1) of, or relating to, theories that
time (for example, process drift), and could exceed the
explain phenomena in purely physical or deterministic terms: a
maximum acceptable risk without warning, unless an ongoing
mechanistic interpretation of nature.
program to enhance process understanding is in place.
3.1.6 process capability, n—statistical estimate of the out-
4.3.2 Accordingly, the improvement of process understand-
come of a characteristic from a process that has been demon-
ing should be treated as an ongoing exercise. Learning should
strated to be in a state of statistical control. E2281
continue throughout the product and process life cycle to
3.1.7 process inputs, n—the combination of all process
improve the level of process understanding to include process
parameters and raw material attributes for a given process. parameters and other factors (for example, environmental,
changes of scale, changes in raw materials, changes in person-
3.1.8 process understanding, v—to recall and comprehend
nel) which may have changed from the initial design of the
process knowledge such that product quality can be explained
chemical or biological DS or DP through manufacturing of the
logically or scientifically, or both, as a function of process
unit dose to final packaging. Work to enhance process under-
inputs and respond accordingly.
standing continuously throughout the life cycle of the product
3.1.9 quality attribute, n—a physical, chemical, biological,
and process can provide assurance that the process will
or microbiological property or characteristic of a product.
continue to have an acceptably low risk of producing out of
3.1.10 residual error, n—the difference between the ob-
specification results.
served result and the predicted value (estimated treatment
4.3.3 Manufacturers should have an ongoing program for
response); Observed Result minus Predicted Value. E456
monitoring and improving upon their operations to enhance
3.1.11 uncertainty, n—an indication of the variability asso- product quality.
ciated with a measured value that takes into account two major
4.4 Process Understanding for the Whole Process:
components of error: (1) bias, and (2) the random error
4.4.1 For each product, process understanding covers the
attributed to the imprecision of the measurement process. E456
process from the initial design of the chemical or biological DS
through manufacturing of the unit dose or device to final
4. Process Understanding
packaging. In addition, the critical quality attributes of the raw
4.1 From physical, chemical, biological, and microbiologi-
materials will in turn become inputs to the DP manufacturing
cal perspectives, a process is considered to be well understood
process, as will process parameters.
when:
4.4.2 Fig. 1 schematically illustrates that the performance of
(1) All critical sources of variability are identified and
any process output (Y) is a function of the inputs (X), which can
explained,
be classified into one of six categories (that is, operator,
(2) variability is managed by the process, and
equipment, measurements, methods, materials, and environ-
(3) product quality attributes can be accurately and reliably
mental conditions).
predicted over the design space established for materials used,
4.4.3 Comprehensive understanding of the relationships of
process parameters, manufacturing, environmental, and other
the process inputs and operating parameters to quality attri-
conditions.
butes of the resulting product is fundamental to developing a
4.2 Well-controlled processes result in the probability of
successful risk mitigation or control strategy, or both. Identi-
product not meeting required specifications at a level that is
fication of critical process parameters (CPPs) and critical raw
below the maximum acceptable limit as predetermined by the
material attributes (CMAs) should be carried out using suitable
user. Accordingly, process understanding requires the compre-
experimental and investigative techniques. An understanding
hension and recall of process knowledge sufficient for the
of these critical inputs (CPPs and CMAs), and their monitoring
logical, statistical, or scientific understanding, or combination
and control, is essential when designing a process that is able
thereof, of how significant process parameters and attributes of
to consistently and reliably deliver product of the desired
raw and in-process materials relate to, or impact the quality
quality.
attributes of, the product being produced. Sufficient process
4.4.4 One common method for achieving the desired state is
understanding should be achieved to reduce risk to an accept-
through multivariate analysis and control. The acceptable
able level for the patient, manufacturer, or any other stake-
operating envelope of the critical inputs defines the relationship
holder.
between input ranges and product quality.
4.3 A Lifecycle Commitment (Development and Commercial 4.4.5 Note that for raw materials, in addition to inherent
Manufacture): variability, an additional source of variability derives from the
4.3.1 Process understanding is fundamental to QbD. It is potential for adulteration. This requires that manufacturers
important to realize that due to commercial realities (for understand their incoming supply chain and suppliers quality
E2475 − 23
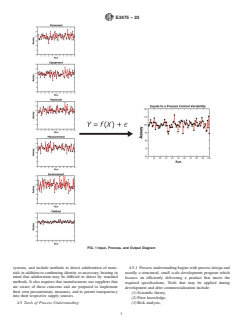
FIG. 1 Input, Process, and Output Diagram
systems, and include methods to detect adulteration of mate- 4.5.1 Process understanding begins with process design and
rials in addition to confirming identity as necessary, bearing in
usually a structured, small scale development program which
mind that adulteration may be difficult to detect by standard
focuses on efficiently delivering a product that meets the
methods. It also requires that manufacturers use suppliers that
required specifications. Tools that may be applied during
are aware of these concerns and are prepared to implement
development and after commercialization include:
their own precautionary measures, and to permit transparency
(1) Scientific theory,
into their respective supply sources.
(2) Prior knowledge,
4.5 Tools of Process Understanding: (3) Risk analysis,
E2475 − 23
(4) Design of experiments, 5.6 Within most organizations in the early stages of QbD
(5) Simulation of unit operations, implementation, process understanding tends to be based
(6) Selection of a suitable technology platform, mainly on descriptive and correlative and scientific knowledge.
(7) Mathematical models, The framework outlined in the USFDA’s “Pharmaceutical
(8) Validated empirical/statistical models, cGMPS for the 21st Century — A Risk-Based Approach”
(9) Appropriate instrumentation, and should encourage the pharmaceutical industry to enhance
(10) Appropriate analytical methods. understanding by adding process knowledge at the causal,
4.5.2 The measurement technologies encompass offline, mechanistic, and first principles levels.
atline, online, and inline technologies. Online and inline
5.7 Mechanistic and first principles process models can
measurement technologies can include but are not limited to
offer advantages over process models which are a combination
spectroscopic, acoustic, or other rapid sensor technologies. The
of only descriptive, correlative, and causative process knowl-
development of these and other advanced techniques will
edge. Proper evaluation of risk may be more challenging in the
continue to enable or enhance predictive control for commer-
absence of mechanistic or first principles process knowledge.
cial pharmaceutical processes.
The user is responsible for determining the level of process
The ability to measure process parameters and quality
knowledge which is appropriate for each specific circumstance.
attributes inline, online, or atline in real time can contribute to
5.8 The subsequent subsections provide greater detail and
process understanding and the ability to control the process.
discussion for each state of knowledge.
These technologies offer the development scientist, commer-
cial production engineer and manufacturing personnel the 5.9 This guide does not differentiate between programs to
opportunity for additional insight. This is achieved through the
develop understanding for products and processes for new
increased measurement frequency and availability of more molecular entities or existing products and processes. The tool
comprehensive data.
sets for each circumstance may be different considering the
available sources of data that can be used, such as existing
5. Process Knowledge
experimental data, historical databases or practical or logistical
5.1 Process knowledge is the cornerstone of process under-
constraints.
standing. There are various levels of process knowledge, and
5.10 The level of understanding that is chosen for each
these are listed from lowest to highest state of understanding:
product or process should be based on a sound science and
(1) Descriptive knowledge (what is occurring?),
risk-based approach. The desired level of understanding will
(2) Correlative knowledge (what correlations are empiri-
depend on a number of factors including quality, criticality,
cally observed?),
time and cost. Maintenance of comprehensive, contemporane-
(3) Causal knowledge (empirical, what causes what?),
ous documenta
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2475 − 10 (Reapproved 2016) E2475 − 23
Standard Guide for
Process Understanding Related to Pharmaceutical
Manufacture and Control
This standard is issued under the fixed designation E2475; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 The purpose of this guide is to establish a framework and context for process understanding for pharmaceutical manufacturing
using the principles of quality by design (QbD) (Juran, 1992; FDA/ICH Q8).ICH Q8). The framework is applicable to both active
pharmaceutical ingredient (API) and to drug substance (DS) and drug product (DP) manufacturing. High (detailed) level process
understanding can be used to facilitate production of product which consistently meets required specifications. It can also play a
key role in continuouscontinual process improvement efforts.
1.2 Process Analytical Technology (PAT) is one element that can be used for achieving control over those inputs determined to
be critical to a process. It is important for the reader to recognize that PAT is defined as:
“{a system for designing, analyzing, and controlling manufacturing through timely measurements (i.e., during processing)
of critical quality and performance attributes of raw and in process materials and processes, with the goal of ensuring final
product quality. It is important to note that the term analytical in PAT is viewed broadly to include chemical, physical,
microbiological, mathematical, and risk analysis conducted in an integrated manner. The goal of PAT is to enhance
understanding and control the manufacturing process{” (U.S. FDA PAT)
“{a system for designing, analyzing, and controlling manufacturing through timely measurements (i.e., during processing)
of critical quality and performance attributes of raw and in process materials and processes, with the goal of ensuring final
product quality. It is important to note that the term analytical in PAT is viewed broadly to include chemical, physical,
microbiological, mathematical, and risk analysis conducted in an integrated manner. The goal of PAT is to enhance
understanding and control the manufacturing process{” (USFDA PAT)
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E456 Terminology Relating to Quality and Statistics
This guide is under the jurisdiction of ASTM Committee E55 on Manufacture of Pharmaceutical and Biopharmaceutical Products and is the direct responsibility of
Subcommittee E55.11 on Process Design.
Current edition approved Sept. 1, 2016Nov. 15, 2023. Published September 2016December 2023. Originally approved in 2010. Last previous edition approved in 20102016
as E2475 – 10. DOI:10.1520/E2475-10R16.10 (2016). DOI:10.1520/E2475-23.
Juran, J., Juran on Quality by Design: The New Steps for Planning Quality Into Goods and Services, Free Press, New York, N.Y., 1992.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2475 − 23
E2281 Practice for Process Capability and Performance Measurement
E2474 Practice for Pharmaceutical Process Design Utilizing Process Analytical Technology (Withdrawn 2020)
E2617 Practice for Validation of Empirically Derived Multivariate Calibrations
2.2 U.S. Government Publications:
ICH Quality Implementation Working Group Points To Consider (R2) ICH-Endorsed Guide for ICH Q8/Q9/Q10 Implementa-
tion
FDA/ICH Q8ICH Q8 Pharmaceutical Development
ICH Q9 Quality Risk Management
FDA/ICH Q10ICH Q10 Pharmaceutical Quality Systems
ICH Q11 Development and Manufacture of Drug Substances
ISO 14971 Medical devices—Application of risk management to medical devices
U.S. FDA PATUSFDA PAT Guidance Document, Guidance for Industry PAT—A Framework for Innovative Pharmaceutical
Manufacturing and Quality Assurance
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 critical inputs, n—critical process parameters and critical raw material attributes for a given process.
American Society for Quality
3.1.2 empirical, adj—any conclusion based on experimental data and past experience, rather than on theory.
3.1.3 expert system, n—an expert system is a computer program that simulates the judgment and behavior of a human or an
organization that has expert knowledge and experience in a particular field.
3.1.3.1 Discussion—
Typically, such a system contains a knowledge base containing accumulated experience and a set of rules for applying the
knowledge base to each particular situation that is described to the program. Sophisticated expert systems can be enhanced with
additions to the knowledge base or to the set of rules.
3.1.4 first principles, n—a calculation is said to be from first principles, or ab initio, if it starts directly at the level of established
laws of physics and does not make assumptions such as model and fitting parameters.
3.1.5 mechanistic, adj—(1) of, or relating to, theories that explain phenomena in purely physical or deterministic terms: a
mechanistic interpretation of nature.
3.1.6 process capability, n—statistical estimate of the outcome of a characteristic from a process that has been demonstrated to
be in a state of statistical control. E2281
3.1.7 process inputs, n—the combination of all process parameters and raw material attributes for a given process.
3.1.8 process understanding, v—to recall and comprehend process knowledge such that product quality can be explained logically
or scientifically, or both, as a function of process inputs and respond accordingly.
3.1.9 quality attribute, n—a physical, chemical, biological, or microbiological property or characteristic of a product.
3.1.10 residual error, n—the difference between the observed result and the predicted value (estimated treatment response);
Observed Result minus Predicted Value. E456
3.1.11 uncertainty, n—an indication of the variability associated with a measured value that takes into account two major
components of error: (1) bias, and (2) the random error attributed to the imprecision of the measurement process. E456
Available from U.S. Government Printing Office Superintendent of Documents, 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
www.access.gpo.gov.
E2475 − 23
4. Process Understanding
4.1 From physical, chemical, biological, and microbiological perspectives, a process is considered to be well understood when:
(1) All significantcritical sources of variability in process inputs are identified and explained,
(2) variability is managed by the process, and
(3) The effect of these sources of variability on product quality attributes can be accurately and reliably estimated based on the
inputs to the process, andpredicted over the design space established for materials used, process parameters, manufacturing,
environmental, and other conditions.
(3) Significant process parameters are continuously managed and controlled to ensure that the process must produce product
which is continuously within required specifications to the user specified required degree or confidence.
4.2 A well-controlled process is a process where the risk of producing Well-controlled processes result in the probability of product
not meeting required specifications at a level that is below the maximum acceptable level of risk limit as predetermined by the user.
Accordingly, process understanding requires the comprehension and recall of process knowledge sufficient for the logical,
statistical, or scientific understanding, or combination thereof, of how significant process parameters and raw material attributes
attributes of raw and in-process materials relate to, or impact the quality attributes of, the product being produced. Sufficient
process understanding should be achieved to reduce risk to an acceptable level for the patient, manufacturer, or any other
stakeholder.
4.3 A Lifecycle Commitment (Development and Commercial Manufacture):
4.3.1 Process understanding is fundamental to QbD. It is important to realize that due to commercial realities (for example, finite
resources, time, and money), a process will typically be commissioned as soon as the degree of process understanding is sufficient
to permit operation of the process with an acceptably low, user specified, level of risk of producing out of specification product.
While it may be appropriate to commission a process once this minimum degree of process understanding is achieved, the risk that
the process may transition out of control steadily increases over time (for example, process drift), and could exceed the maximum
acceptable risk without warning, unless an ongoing program to enhance process understanding is in place.
4.3.2 Accordingly, the developmentimprovement of process understanding should be treated as an ongoing process.exercise.
Learning should continue throughout the product and process life cycle to improve the level of process understanding to include
process parameters and other factors (for example, environmental, changes of scale, changes in raw materials, changes in
personnel) which may have changed or which may have newly emerged since the time the process was first commissioned. from
the initial design of the chemical or biological DS or DP through manufacturing of the unit dose to final packaging. Work to
enhance process understanding continuously throughout the life cycle of the product and process can provide assurance that the
process will continue to have an acceptably low risk of producing out of specification results.
4.3.3 Manufacturers are encouraged to continuously monitor and improveshould have an ongoing program for monitoring and
improving upon their operations to enhance product quality.
4.4 Process Understanding for the Whole Process:
4.4.1 For each product, process understanding covers the process from the initial design of the chemical or biological drug
substance DS through manufacturing of the unit dose or device to final packaging. In addition, the critical quality attributes of the
raw materials will in turn become inputs to the drug product DP manufacturing process, as will process parameters.
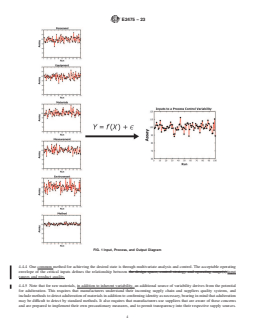
4.4.2 Fig. 1 schematically illustrates that the performance of any process output (Y) is a function of the inputs (X), which can be
classified into one of six categories (that is, operator, equipment, measurements, methods, materials, and environmental
conditions).
4.4.3 Comprehensive understanding of the relationships of the process inputs and operating parameters to quality attributes of the
resulting product is fundamental to developing a successful risk mitigation or control strategy, or both. Identification of critical
process parameters (CPPs) and critical raw material attributes (CMAs) should be carried out using suitable experimental and
investigative techniques. An understanding of these critical inputs (CPPs and critical raw material attributes), CMAs), and their
monitoring and control, is essential when designing a process that is able to consistently and reliably deliver product of the desired
quality.
E2475 − 23
FIG. 1 Input, Process, and Output Diagram
4.4.4 One common method for achieving the desired state is through multivariate analysis and control. The acceptable operating
envelope of the critical inputs defines the relationship between the design space, control strategy and operating range(s).input
ranges and product quality.
4.4.5 Note that for raw materials, in addition to inherent variability, an additional source of variability derives from the potential
for adulteration. This requires that manufacturers understand their incoming supply chain and suppliers quality systems, and
include methods to detect adulteration of materials in addition to confirming identity as necessary, bearing in mind that adulteration
may be difficult to detect by standard methods. It also requires that manufacturers use suppliers that are aware of these concerns
and are prepared to implement their own precautionary measures, and to permit transparency into their respective supply sources.
E2475 − 23
4.5 Tools of Process Understanding:
4.5.1 Process understanding begins with process design (Practice E2474) and usually a structured, small scale development
program which focuses on efficiently delivering a product meeting that meets the required specifications. Tools that may be applied
during development and after commercialization include:
(1) Scientific theory,
(2) Prior knowledge,
(3) Risk analysis,
(4) Design of experiments,
(5) Simulation of unit operations,
(6) Selection of a suitable technology platform,
(7) Mathematical models,
(8) Validated empirical/statistical models,
(9) Appropriate instrumentation, and
(10) Appropriate analytical methods.
4.5.2 The measurement technologies include but are not limited to spectroscopic, acoustic, or other rapid sensor technologies. The
development of these and other advanced techniques will continue to enable or enhance predictive control for commercial
pharmaceutical processes.
4.5.2 The measurement technologies encompass offline, atline, online, and inline technologies. Online and inline measurement
technologies can include but are not limited to spectroscopic, acoustic, or other rapid sensor technologies. The development of
these and other advanced techniques will continue to enable or enhance predictive control for commercial pharmaceutical
processes.
The ability to measure process parameters and quality attributes inline, online, or atline in real time can contribute to process
understanding and the ability to control the process. These technologies offer the development scientist, commercial production
engineer and manufacturing personnel the opportunity for additional insight. This is achieved through the increased measurement
frequency and availability of more comprehensive data.
5. Process Knowledge
5.1 Process knowledge is the cornerstone of process understanding. There are various levels of process knowledge, and these are
listed from lowest to highest state of understanding:
(1) Descriptive knowledge (what is occurring?),
(2) Correlative knowledge (what correlations are empirically observed?),
(3) Causal knowledge (empirical, what causes what?),
(4) Mechanistic knowledge (explanations for observed causality), and
(5) First principles knowledge (underlying physical, chemical, and biological phenomena of the mechanistic explanations).
5.2 Process knowledge is the accumulated facts about the process. This accumulated knowledge is generally embodied in a model
of the process. Accordingly, process model is often used synonymously with process knowledge.
5.3 Process understanding is demonstrated by the extent to which process knowledge can be used to predict and control the process
outcomes; a well understood process will combine knowledge from various sources to ensure a well controlled process and
consistent product quality.
5.4 At any point in time for any manufacturing process, the level of understanding will likely be a combination of various levels
of understanding. As more knowledge is obtained throughout the lifecycle of a product, the relative contribution to understanding
of the various levels is likely to change.
5.5 Prior knowledge is any knowledge that may be available through previous experience. Prior knowledge may c
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...