ASTM E2629-20
(Guide)Standard Guide for Verification of Process Analytical Technology (PAT) Enabled Control Systems
Standard Guide for Verification of Process Analytical Technology (PAT) Enabled Control Systems
SIGNIFICANCE AND USE
5.1 This guide supports the principles of Guide E2500 and extends these principles to the verification of PAT-enabled control systems.
5.2 This guide clarifies what is important for verification of PAT-enabled control systems. Such systems are often complex and require multidisciplinary and cross-functional teams to achieve optimum results. This guide provides a common basis for understanding requirements for all involved disciplines such as control engineering, development, manufacturing, and process validation.
SCOPE
1.1 This guide describes the verification of process analytical technology (PAT) enabled control systems using a science- and risk-based approach. It establishes principles for determining the scope and extent of verification activities necessary to ensure that the PAT-enabled control system is fit for purpose, properly implemented, and functions as expected.
1.2 In this guide, a PAT-enabled control system is considered to be the system that adjusts the manufacturing process using timely measurements (that is, during processing) of attributes of raw and in-process materials to determine responses that assure the process remains within specified boundaries and minimizes variability in the output material. The overall aim of the PAT-enabled control system is to ensure product quality. The PAT-enabled control system of a manufacturing process provides the capability to determine the current status of the process and drive the process to ensure the output material has the desired quality characteristics. The control system should be able to respond to process variations in a timely manner, providing corrections that ensure that the process follows the desired process trajectory to reach the desired outcome. PAT-enabled control systems may use process models based on first principles understanding or empirical models derived from experimental investigations or both. In addition to automated controls, a PAT-enabled control system may include components where there is manual intervention.
1.3 Principles described in this guide may be applied regardless of the complexity or scale of the PAT-enabled control system or whether applied to batch or continuous processing, or both. The intention of this standard is to describe and support the implementation of a PAT enabled Control Strategy, as described in ICH Q8(R2).
1.4 The principles described in this guide are applicable to a PAT-enabled control system and also to its component subsystems. This guide does not cover the requirements for continuous quality verification of the overall process, which are covered in Guide E2537, or for validation of PAT methods, which is covered in Guide E2898.
1.5 For information on science- and risk-based approaches in the pharmaceutical industry, reference should be made to ICH Q8(R2), ICH Q9, and ICH Q10. For guidance on PAT systems in the pharmaceutical industry, reference should be made to FDA Guidance for Industry—PAT and FDA Guidance for Industry—Process Validation, as well as EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use and EU Guideline on Process Validation for Finished Products.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jul-2020
- Technical Committee
- E55 - Manufacture of Pharmaceutical and Biopharmaceutical Products
- Drafting Committee
- E55.13 - Process Evaluation and Control
Relations
- Refers
ASTM E2898-20 - Standard Guide for Risk-Based Validation of Analytical Methods for PAT Applications - Effective Date
- 15-May-2020
- Refers
ASTM E2898-14 - Standard Guide for Risk-Based Validation of Analytical Methods for PAT Applications - Effective Date
- 01-Jun-2014
- Refers
ASTM E2898-13 - Standard Guide for Risk-Based Validation of Analytical Methods for PAT Applications - Effective Date
- 01-Nov-2013
- Effective Date
- 15-Oct-2012
- Effective Date
- 01-Aug-2011
- Effective Date
- 01-Aug-2009
- Effective Date
- 15-May-2009
- Effective Date
- 01-Jan-2008
- Effective Date
- 01-Oct-2007
- Effective Date
- 01-Jun-2007
- Effective Date
- 15-Jul-2006
- Effective Date
- 15-Jan-2006
- Effective Date
- 15-Oct-2005
- Effective Date
- 15-Aug-2005
- Effective Date
- 01-Mar-2005
Overview
ASTM E2629-20: Standard Guide for Verification of Process Analytical Technology (PAT) Enabled Control Systems provides a comprehensive, science- and risk-based framework for verifying PAT-enabled control systems in pharmaceutical and biopharmaceutical manufacturing. Developed by ASTM International, this guide supports and extends the principles of ASTM E2500, focusing on ensuring that PAT-enabled control systems are fit for purpose, properly implemented, and function as intended to assure product quality.
PAT-enabled control systems are critical for real-time monitoring and control of manufacturing processes using timely measurement of material attributes. These systems play a central role in maintaining product quality by adjusting processes dynamically to minimize variability and keep output within specified parameters.
Key Topics
- Science- and Risk-Based Verification: Verification activities are driven by quality risk management to ensure that systems and components function as intended within the specified process design space.
- Multidisciplinary Collaboration: Effective verification requires contributions from control engineering, process development, manufacturing, and validation teams.
- Three-Stage Verification Process:
- Verification Planning: Establishes scope, strategy, technical assessment, sensitivity analysis, and acceptance criteria.
- Testing and Confirmation: Involves measurement system validation, process and control model testing, in-situ system challenges, and documentation.
- Continued Verification: Ongoing assurance of system performance in commercial manufacturing (supported by continuous quality verification guides).
- Critical Components and Concepts:
- Measurement Systems: Validation and calibration of sensors and analytical instruments.
- Process and Control Models: Mathematical models used for process and parameter control, including first-principles and empirical approaches.
- Sample Representativeness: Ensuring data collected are representative of actual process material.
- System Stability and Capability: Ensuring that feedback and adaptive controls respond appropriately to process disturbances.
- Levels of Control Strategy: From dynamic, real-time process control supporting real-time release, to traditional end-product testing, clarifying the scope relevant to PAT-enabled systems.
Applications
ASTM E2629-20 is primarily used in the development, validation, and routine operation of pharmaceutical and biopharmaceutical manufacturing processes employing PAT-enabled control systems. Key applications include:
- Implementation of PAT Control Strategies: Assuring process and product quality by monitoring and controlling critical process parameters and quality attributes in real time.
- Risk Management: Applying science- and risk-based approaches to determine the extent of verification required for each system component.
- Regulatory Compliance: Supporting requirements in alignment with ICH Q8(R2), ICH Q9, ICH Q10, and relevant FDA and EU good manufacturing practice (GMP) guidelines.
- Quality Assurance: Providing a structured basis for verification of process models, measurement systems, and control algorithms to enable data-driven process control.
- Continuous Process Improvement: Facilitating ongoing system evaluation and adaptation to ensure processes remain in a state of control, minimizing the risk of out-of-specification material.
Related Standards
For a holistic approach to PAT-enabled systems and process verification, the following standards and guidance documents are often referenced in conjunction with ASTM E2629-20:
- ASTM E2500: Specification, design, and verification of pharmaceutical and biopharmaceutical manufacturing systems and equipment.
- ASTM E2537: Guide for application of continuous process verification.
- ASTM E2898: Guide for risk-based validation of analytical methods for PAT.
- ASTM E2476: Guide for risk assessment and risk control impacting PAT process design and operation.
- ICH Q8(R2), Q9, and Q10: Guidance documents covering pharmaceutical development, risk management, and pharmaceutical quality systems.
- FDA Guidance on PAT and Process Validation, and EU GMP Guidelines.
Keywords: PAT-enabled control, process analytical technology, process control, verification, risk-based approach, pharmaceutical manufacturing, biopharmaceutical, quality assurance, process validation, ASTM standards, GMP compliance.
Buy Documents
ASTM E2629-20 - Standard Guide for Verification of Process Analytical Technology (PAT) Enabled Control Systems
REDLINE ASTM E2629-20 - Standard Guide for Verification of Process Analytical Technology (PAT) Enabled Control Systems
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E2629-20 is a guide published by ASTM International. Its full title is "Standard Guide for Verification of Process Analytical Technology (PAT) Enabled Control Systems". This standard covers: SIGNIFICANCE AND USE 5.1 This guide supports the principles of Guide E2500 and extends these principles to the verification of PAT-enabled control systems. 5.2 This guide clarifies what is important for verification of PAT-enabled control systems. Such systems are often complex and require multidisciplinary and cross-functional teams to achieve optimum results. This guide provides a common basis for understanding requirements for all involved disciplines such as control engineering, development, manufacturing, and process validation. SCOPE 1.1 This guide describes the verification of process analytical technology (PAT) enabled control systems using a science- and risk-based approach. It establishes principles for determining the scope and extent of verification activities necessary to ensure that the PAT-enabled control system is fit for purpose, properly implemented, and functions as expected. 1.2 In this guide, a PAT-enabled control system is considered to be the system that adjusts the manufacturing process using timely measurements (that is, during processing) of attributes of raw and in-process materials to determine responses that assure the process remains within specified boundaries and minimizes variability in the output material. The overall aim of the PAT-enabled control system is to ensure product quality. The PAT-enabled control system of a manufacturing process provides the capability to determine the current status of the process and drive the process to ensure the output material has the desired quality characteristics. The control system should be able to respond to process variations in a timely manner, providing corrections that ensure that the process follows the desired process trajectory to reach the desired outcome. PAT-enabled control systems may use process models based on first principles understanding or empirical models derived from experimental investigations or both. In addition to automated controls, a PAT-enabled control system may include components where there is manual intervention. 1.3 Principles described in this guide may be applied regardless of the complexity or scale of the PAT-enabled control system or whether applied to batch or continuous processing, or both. The intention of this standard is to describe and support the implementation of a PAT enabled Control Strategy, as described in ICH Q8(R2). 1.4 The principles described in this guide are applicable to a PAT-enabled control system and also to its component subsystems. This guide does not cover the requirements for continuous quality verification of the overall process, which are covered in Guide E2537, or for validation of PAT methods, which is covered in Guide E2898. 1.5 For information on science- and risk-based approaches in the pharmaceutical industry, reference should be made to ICH Q8(R2), ICH Q9, and ICH Q10. For guidance on PAT systems in the pharmaceutical industry, reference should be made to FDA Guidance for Industry—PAT and FDA Guidance for Industry—Process Validation, as well as EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use and EU Guideline on Process Validation for Finished Products. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This guide supports the principles of Guide E2500 and extends these principles to the verification of PAT-enabled control systems. 5.2 This guide clarifies what is important for verification of PAT-enabled control systems. Such systems are often complex and require multidisciplinary and cross-functional teams to achieve optimum results. This guide provides a common basis for understanding requirements for all involved disciplines such as control engineering, development, manufacturing, and process validation. SCOPE 1.1 This guide describes the verification of process analytical technology (PAT) enabled control systems using a science- and risk-based approach. It establishes principles for determining the scope and extent of verification activities necessary to ensure that the PAT-enabled control system is fit for purpose, properly implemented, and functions as expected. 1.2 In this guide, a PAT-enabled control system is considered to be the system that adjusts the manufacturing process using timely measurements (that is, during processing) of attributes of raw and in-process materials to determine responses that assure the process remains within specified boundaries and minimizes variability in the output material. The overall aim of the PAT-enabled control system is to ensure product quality. The PAT-enabled control system of a manufacturing process provides the capability to determine the current status of the process and drive the process to ensure the output material has the desired quality characteristics. The control system should be able to respond to process variations in a timely manner, providing corrections that ensure that the process follows the desired process trajectory to reach the desired outcome. PAT-enabled control systems may use process models based on first principles understanding or empirical models derived from experimental investigations or both. In addition to automated controls, a PAT-enabled control system may include components where there is manual intervention. 1.3 Principles described in this guide may be applied regardless of the complexity or scale of the PAT-enabled control system or whether applied to batch or continuous processing, or both. The intention of this standard is to describe and support the implementation of a PAT enabled Control Strategy, as described in ICH Q8(R2). 1.4 The principles described in this guide are applicable to a PAT-enabled control system and also to its component subsystems. This guide does not cover the requirements for continuous quality verification of the overall process, which are covered in Guide E2537, or for validation of PAT methods, which is covered in Guide E2898. 1.5 For information on science- and risk-based approaches in the pharmaceutical industry, reference should be made to ICH Q8(R2), ICH Q9, and ICH Q10. For guidance on PAT systems in the pharmaceutical industry, reference should be made to FDA Guidance for Industry—PAT and FDA Guidance for Industry—Process Validation, as well as EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use and EU Guideline on Process Validation for Finished Products. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2629-20 is classified under the following ICS (International Classification for Standards) categories: 71.120.01 - Equipment for the chemical industry in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2629-20 has the following relationships with other standards: It is inter standard links to ASTM E2898-20, ASTM E2898-14, ASTM E2898-13, ASTM E2500-07(2012), ASTM E122-09e1, ASTM E122-09, ASTM E2476-09, ASTM E2537-08, ASTM E122-07, ASTM E2500-07, ASTM E2363-06a, ASTM E2363-06, ASTM E2363-05b, ASTM E2363-05a, ASTM E2363-05. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2629-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2629 − 20
Standard Guide for
Verification of Process Analytical Technology (PAT) Enabled
Control Systems
This standard is issued under the fixed designation E2629; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope subsystems. This guide does not cover the requirements for
continuous quality verification of the overall process, which
1.1 This guide describes the verification of process analyti-
are covered in Guide E2537, or for validation of PAT methods,
cal technology (PAT) enabled control systems using a science-
which is covered in Guide E2898.
and risk-based approach. It establishes principles for determin-
ing the scope and extent of verification activities necessary to
1.5 For information on science- and risk-based approaches
ensure that the PAT-enabled control system is fit for purpose, in the pharmaceutical industry, reference should be made to
properly implemented, and functions as expected.
ICH Q8(R2), ICH Q9, and ICH Q10. For guidance on PAT
systems in the pharmaceutical industry, reference should be
1.2 In this guide, a PAT-enabled control system is consid-
made to FDA Guidance for Industry—PAT and FDA Guidance
ered to be the system that adjusts the manufacturing process
for Industry—Process Validation, as well as EU Guidelines for
using timely measurements (that is, during processing) of
Good Manufacturing Practice for Medicinal Products for
attributes of raw and in-process materials to determine re-
Human and Veterinary Use and EU Guideline on Process
sponses that assure the process remains within specified
Validation for Finished Products.
boundaries and minimizes variability in the output material.
The overall aim of the PAT-enabled control system is to ensure
1.6 This standard does not purport to address all of the
product quality. The PAT-enabled control system of a manu-
safety concerns, if any, associated with its use. It is the
facturing process provides the capability to determine the
responsibility of the user of this standard to establish appro-
current status of the process and drive the process to ensure the
priate safety, health, and environmental practices and deter-
output material has the desired quality characteristics. The
mine the applicability of regulatory limitations prior to use.
control system should be able to respond to process variations 1.7 This international standard was developed in accor-
in a timely manner, providing corrections that ensure that the
dance with internationally recognized principles on standard-
process follows the desired process trajectory to reach the ization established in the Decision on Principles for the
desired outcome. PAT-enabled control systems may use pro-
Development of International Standards, Guides and Recom-
cess models based on first principles understanding or empiri- mendations issued by the World Trade Organization Technical
cal models derived from experimental investigations or both.
Barriers to Trade (TBT) Committee.
In addition to automated controls, a PAT-enabled control
2. Referenced Documents
system may include components where there is manual inter-
vention.
2.1 ASTM Standards:
1.3 Principles described in this guide may be applied E122 Practice for Calculating Sample Size to Estimate, With
regardless of the complexity or scale of the PAT-enabled Specified Precision, the Average for a Characteristic of a
control system or whether applied to batch or continuous Lot or Process
E2363 Terminology Relating to Manufacturing of Pharma-
processing, or both. The intention of this standard is to describe
and support the implementation of a PAT enabled Control ceutical and Biopharmaceutical Products in the Pharma-
ceutical and Biopharmaceutical Industry
Strategy, as described in ICH Q8(R2).
E2476 Guide for Risk Assessment and Risk Control as it
1.4 The principles described in this guide are applicable to
Impacts the Design, Development, and Operation of PAT
a PAT-enabled control system and also to its component
Processes for Pharmaceutical Manufacture
This guide is under the jurisdiction of ASTM Committee E55 on Manufacture
of Pharmaceutical and Biopharmaceutical Products and is the direct responsibility of
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Subcommittee E55.13 on Process Evaluation and Control.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved Aug. 1, 2020. Published August 2020. Originally
approved in 2011. Last previous edition approved in 2019 as E2629 – 19. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/E2629-20. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2629 − 20
E2500 Guide for Specification, Design, and Verification of to operate in the desired manner.
Pharmaceutical and Biopharmaceutical Manufacturing Perry’s Handbook of Chemical Engineering
Systems and Equipment
3.1.4 measurement system, n—system of sensors,
E2537 Guide for Application of Continuous Process Verifi-
instruments, and/or analyzers that collects signals generated by
cation to Pharmaceutical and Biopharmaceutical Manu-
passive or active interaction with process material or process
facturing
equipment and converts those signals into data.
E2898 Guide for Risk-Based Validation of Analytical Meth-
3.1.5 parameter, n—measureable or quantifiable character-
ods for PAT Applications
istic of a system or process. E2363
2.2 Regulatory Guidance and Other Documents:
3.1.6 process model, n—mathematical expression (algo-
ICH Q2(R1) Validation of Analytical Procedures: Text and
rithm) that uses data from the measurement system(s) (inputs
Methodology
to the process model) to calculate the value of one or more of
ICH Q8(R2) Pharmaceutical Development
the process material attributes (outputs from the process
ICH Q9 Risk Management
model) at the time the measurement was taken.
ICH Q10 Pharmaceutical Quality System
3.1.6.1 Discussion—The process model typically will have
FDA Guidance for Industry—PAT A Framework for Inno-
to handle sets of orthogonal or nonorthogonal attributes. The
vative Pharmaceutical Development, Manufacturing and
mathematical algorithm will ideally represent first-principle
Quality Assurance
understanding of the process being modelled. However, when
FDA Guidance for Industry—Process Validation General
sufficient first-principles understanding is unavailable, an em-
Principles and Practices
pirical model may also be used.
EU Guidelines for Good Manufacturing Practice for Medici-
3.2 Acronyms:
nal Products for Human and Veterinary Use Annex 15:
3.2.1 CCP—Critical control parameter
Qualification and Validation
3.2.2 CPP—Critical process parameter
EU Guideline on Process Validation for Finished Products
3.2.3 CQA—Critical quality attribute
3. Terminology
3.2.4 CQV—Continuous quality verification
3.1 Definitions—See also Terminology E2363 for other PAT
3.2.5 FDA—Food and Drug Administration
terms.
3.2.6 ICH—International Conference on Harmonization of
3.1.1 attribute, n—characteristic or inherent quality or
Technical Requirements for Registration of Pharmaceuticals
feature. E2363
for Human Use
3.1.2 control model, n—procedure or mathematical expres-
3.2.7 ISA—International Society of Automation
sion (algorithm) that uses the outputs of the process model
3.2.8 LOD—Limit of detection
combined with any other data inputs required to calculate
3.2.9 MBC—Model-based control
values for the critical control parameters for the process; it uses
3.2.10 MVA—Multivariate analysis
input data from the process to generate an actionable command
or commands that are issued to the control system.
3.2.11 PAT—Process analytical technology
3.1.2.1 Discussion—The control model may define what
3.2.12 PID—Proportional integral derivative
actions to take when specific attribute values are detected. The
3.2.13 PP—Process parameter
control model may be complex or simple, for example, it may
3.2.14 QA—Quality attribute
be predictive, as in the case of model-based control (MBC) in
which it is desired to manage the operation of the process along
4. Summary of Practice
a particular trajectory; it may be a single proportional integral
derivative (PID) loop controller; or it may be anything in 4.1 To aid reader understanding, a diagram of the data flows
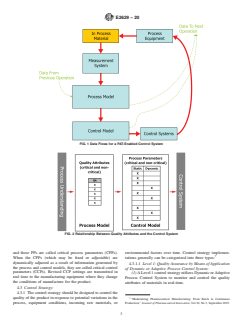
between. in a PAT-enabled control system is shown in Fig. 1. The
diagram shows how process and control models can be used in
3.1.3 control system, n—system that responds to inputs
a closed loop control paradigm (with decisions being made
signals from the process, its associated equipment, other
based on action limits set in the control model) but also for
programmable systems or an operator or both, and generates
feed-forward control to downstream process steps/operations.
output signals causing the process and its associated equipment
4.2 Fig. 2 shows how the quality attributes (QAs), noncriti-
cal as well as critical, are fed into the control model via the
process model. Each process has process parameters (PPs).
Available from International Conference on Harmonisation of Technical
Based on process understanding, some PPs are held static and
Requirements for Registration of Pharmaceuticals for Human Use (ICH), ICH
Secretariat, c/o IFPMA, 15 ch. Louis-Dunant, P.O. Box 195, 1211 Geneva 20, others are subject to dynamic adjustment. Some of the PPs
Switzerland, http://www.ich.org.
directly or indirectly impact critical quality attributes (CQAs)
Available from Office of Training and Communication, Division of Drug
Information, HFD-240, Center for Drug Evaluation and Research, Food and Drug
Administration, 5600 Fishers Lane, Rockville, MD 20857, http://www.fda.gov.
5 6
Available from the European Commission (European Union (EU)), https:// Perry’s Handbook of Chemical Engineering, see BPCS–Basic Process Control
ec.europa.eu. System, McGraw Hill, 2007.
E2629 − 20
FIG. 1 Data Flows for a PAT-Enabled Control System
FIG. 2 Relationship Between Quality Attributes and the Control System
and these PPs are called critical process parameters (CPPs). environmental factors over time. Control strategy implemen-
When the CPPs (which may be fixed or adjustable) are tations generally can be categorized into three types:
dynamically adjusted as a result of information generated by
4.3.1.1 Level 1: Quality Assurance by Means of Application
the process and control models, they are called critical control
of Dynamic or Adaptive Process Control System:
parameters (CCPs). Revised CCP settings are transmitted in
(1) A Level 1 control strategy utilizes Dynamic or Adaptive
real time to the manufacturing equipment where they change
Process Control System to monitor and control the quality
the conditions of manufacture for the product.
attributes of materials in real-time.
4.3 Control Strategy:
4.3.1 The control strategy should be designed to control the
quality of the product in response to potential variations in the
“Modernizing Pharmaceutical Manufacturing: From Batch to Continuous
process, equipment conditions, incoming raw materials, or Production,” Journal of Pharmaceutical Innovation, Vol 10, No 3, September 2015.
E2629 − 20
(2) In Dynamic or Adaptive Process Control, system pro- 5. Significance and Use
cess parameters are monitored and may be adjusted in response
5.1 This guide supports the principles of Guide E2500 and
to disturbances to ensure that the quality attributes consistently
extends these principles to the verification of PAT-enabled
conform to the established acceptance criteria.
control systems.
(3) The successful application of a Dynamic or Adaptive
5.2 This guide clarifies what is important for verification of
Process Control system represents a high degree of product and
PAT-enabled control systems. Such systems are often complex
process understanding as the design of an engineering control
and require multidisciplinary and cross-functional teams to
system entails expressing the dynamic relationships among
achieve optimum results. This guide provides a common basis
process parameters, raw material and product attributes in a
for understanding requirements for all involved disciplines
quantitative and predictive manner.
such as control engineering, development, manufacturing, and
(4) The ability of a Dynamic or Adaptive Process Control
process validation.
System to compensate for variation in the raw material
attributes or external disturbances to the process condition
6. Principles To Be Considered for Verification of PAT-
significantly reduces the risk of producing of out of specifica- Enabled Control Systems
tion material and hence the requirement for routine
6.1 Verification should be science and risk based. Quality
segregation/diversion of out-of-specification is also reduced.
risk management should drive the verification process. Practice
(5) Statistical monitoring tools, for example Univariate or
E2476 provides additional guidance on risk assessments for
Multivariate SPC, may be used to demonstrate that the Dy-
PAT systems.
namic or Adaptive Process control system is ensuring that the
6.2 Verification should use the most efficient and effective
process is operating in a State of Control where there is a very
method available to achieve the specified results, choosing
low probability of out of specification being produced.
from, for example, simulation, testing, first principle modeling,
(6) Successful implementation of a Dynamic or Adaptive
or other approaches or combinations of these.
Process Control system directly supports a real-time release
6.3 Verification should cover the design space of the manu-
strategy.
facturing process. This will include all those ranges in which it
4.3.1.2 Level 2: Quality Assurance by Means of Operation
is necessary that the control system will be able to bring the
Within an Established Design Space and Confirmatory End
process back into its intended operating range. The verification
Product and In-Process Material Testing:
can occur during Process Performance Qualification or prior
(1) Product Quality may be assured by combination of
(during development phase), or both, given the process system
appropriate end product testing together with appropriate
is in a qualified state.
monitoring of controlled raw material attributes and process
6.4 Verification of the control systems should always in-
parameters.
clude verification of the system as a whole. It may also include
(2) The product and process understanding obtained
verification of individual system components.
through the establishment of a multivariate design space
facilitates the identification of potential sources of raw material
6.5 The verification process should confirm that relevant
and process variability that can impact product quality.
quality attributes will be controlled concurrently.
(3) Understanding the impact that variability from these
6.6 Verification should ensure that the control system is
sources has on in-process materials, downstream processing,
stable throughout the range of operation.
and drug product quality provides an opportunity to shift
6.7 Each component of the PAT-enabled control system
controls upstream and to reduce the reliance on end-product
should generate outputs with sufficient frequency, accuracy,
testing.
and precision to make the necessary level of process control
(4) The absence of real time corrective action within the
practical, meaningful and value-added.
control system increases the probability for production of out
of specification material and hence requires appropriate mecha-
6.8 Process and control models and the control system
nisms for ensuring rejection/diversion of any out-of-
should be verified as applicable to the scale of manufacture at
specification product.
which they will be used.
4.3.1.3 Level 3: Quality Assurance by Means of Operation
6.9 All stages of the verification should be appropriately
Within a Validated and Tightly Constrained Material Attributes
demonstrated and clearly documented in accordance with
and Process Parameters and Release Based on End Product
relevant requirements.
Testing:
7. Verification Process for PAT-Enabled Control Systems
(1) A Level 3, Control Strategy does not use PAT for either
feedforward or feedback control and is therefore outside t
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2629 − 19 E2629 − 20
Standard Guide for
Verification of Process Analytical Technology (PAT) Enabled
Control Systems
This standard is issued under the fixed designation E2629; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide describes the verification of process analytical technology (PAT) enabled control systems using a science- and
risk-based approach. It establishes principles for determining the scope and extent of verification activities necessary to ensure that
the PAT-enabled control system is fit for purpose, properly implemented, and functions as expected.
1.2 In this guide, a PAT-enabled control system is considered to be the system that adjusts the manufacturing process using
timely measurements (that is, during processing) of attributes of raw and in-process materials to determine responses that assure
the process remains within specified boundaries and minimizes variability in the output material. The overall aim of the
PAT-enabled control system is to ensure product quality. The PAT-enabled control system of a manufacturing process provides the
capability to determine the current status of the process and drive the process to ensure the output material has the desired quality
characteristics. The control system should be able to respond to process variations in a timely manner, providing corrections that
ensure that the process follows the desired process trajectory to reach the desired outcome. PAT-enabled control systems may use
process models based on first principles understanding or empirical models derived from experimental investigations or both. In
addition to automated controls, a PAT-enabled control system may include components where there is manual intervention.
1.3 Principles described in this guide may be applied regardless of the complexity or scale of the PAT-enabled control system
or whether applied to batch or continuous processing, or both. The intention of this standard is to describe and support the
implementation of a PAT enabled Control Strategy, as described in ICH Q8(R2).
1.4 The principles described in this guide are applicable to a PAT-enabled control system and also to its component subsystems.
This guide does not cover the requirements for continuous quality verification of the overall process, which are covered in Guide
E2537, or for validation of PAT methods, which is covered in Guide E2898.
1.5 For information on science- and risk-based approaches in the pharmaceutical industry, reference should be made to
ICH Q8(R2), ICH Q9, and ICH Q10. For guidance on PAT systems in the pharmaceutical industry, reference should be made to
FDA Guidance for Industry—PAT and FDA Guidance for Industry—Process Validation, as well as EU Guidelines for Good
Manufacturing Practice for Medicinal Products for Human and Veterinary Use and EU Guideline on Process Validation for
Finished Products.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E122 Practice for Calculating Sample Size to Estimate, With Specified Precision, the Average for a Characteristic of a Lot or
Process
This guide is under the jurisdiction of ASTM Committee E55 on Manufacture of Pharmaceutical and Biopharmaceutical Products and is the direct responsibility of
Subcommittee E55.01 on Process Understanding and PAT System Management, Implementation and Practice.
Current edition approved June 15, 2019Aug. 1, 2020. Published July 2019August 2020. Originally approved in 2011. Last previous edition approved in 20112019 as E2629
– 11.19. DOI: 10.1520/E2629-19.10.1520/E2629-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2629 − 20
E2363 Terminology Relating to Process Analytical Technology in the Pharmaceutical Industry
E2476 Guide for Risk Assessment and Risk Control as it Impacts the Design, Development, and Operation of PAT Processes
for Pharmaceutical Manufacture
E2629 − 20
E2500 Guide for Specification, Design, and Verification of Pharmaceutical and Biopharmaceutical Manufacturing Systems and
Equipment
E2537 Guide for Application of Continuous Process Verification to Pharmaceutical and Biopharmaceutical Manufacturing
E2898 Guide for Risk-Based Validation of Analytical Methods for PAT Applications
2.2 Regulatory Guidance and Other Documents:
ICH Q2(R1) Validation of Analytical Procedures: Text and Methodology
ICH Q8(R2) Pharmaceutical Development
ICH Q9 Risk Management
ICH Q10 Pharmaceutical Quality System
FDA Guidance for Industry—PAT A Framework for Innovative Pharmaceutical Development, Manufacturing and Quality
Assurance
FDA Guidance for Industry—Process Validation General Principles and Practices
EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use Annex 15: Qualification
and Validation
EU Guideline on Process Validation for Finished Products
3. Terminology
3.1 Definitions—See also Terminology E2363 for other PAT terms.
3.1.1 attribute, n—characteristic or inherent quality or feature. E2363
3.1.2 control model, n—procedure or mathematical expression (algorithm) that uses the outputs of the process model combined
with any other data inputs required to calculate values for the critical control parameters for the process; it uses input data from
the process to generate an actionable command or commands that are issued to the control system.
3.1.2.1 Discussion—
The control model may define what actions to take when specific attribute values are detected. The control model may be complex
or simple, for example, it may be predictive, as in the case of model-based control (MBC) in which it is desired to manage the
operation of the process along a particular trajectory; it may be a single proportional integral derivative (PID) loop controller; or
it may be anything in between.
3.1.3 control system, n—system that responds to inputs signals from the process, its associated equipment, other programmable
systems or an operator or both, and generates output signals causing the process and its associated equipment to operate in the
desired manner.
Perry’s Handbook of Chemical Engineering
3.1.4 measurement system, n—system of sensors, instruments, and/or analyzers that collects signals generated by passive or
active interaction with process material or process equipment and converts those signals into data.
3.1.5 parameter, n—measureable or quantifiable characteristic of a system or process. E2363
3.1.6 process model, n—mathematical expression (algorithm) that uses data from the measurement system(s) (inputs to the
process model) to calculate the value of one or more of the process material attributes (outputs from the process model) at the time
the measurement was taken.
3.1.6.1 Discussion—
The process model typically will have to handle sets of orthogonal or nonorthogonal attributes. The mathematical algorithm will
ideally represent first-principle understanding of the process being modelled. However, when sufficient first-principles
understanding is unavailable, an empirical model may also be used.
3.2 Acronyms:
3.2.1 CCP—Critical control parameter
3.2.2 CPP—Critical process parameter
3.2.3 CQA—Critical quality attribute
Available from International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), ICH Secretariat, c/o
IFPMA, 15 ch. Louis-Dunant, P.O. Box 195, 1211 Geneva 20, Switzerland, http://www.ich.org.
Available from Office of Training and Communication, Division of Drug Information, HFD-240, Center for Drug Evaluation and Research, Food and Drug
Administration, 5600 Fishers Lane, Rockville, MD 20857, http://www.fda.gov.
Available from the European Commission (European Union (EU)), https://ec.europa.eu.
Perry’s Handbook of Chemical Engineering, see BPCS–Basic Process Control System, McGraw Hill, 2007.
E2629 − 20
3.2.4 CQV—Continuous quality verification
3.2.5 FDA—Food and Drug Administration
3.2.6 ICH—International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for
Human Use
3.2.7 ISA—International Society of Automation
3.2.8 LOD—Limit of detection
3.2.9 MBC—Model-based control
3.2.10 MVA—Multivariate analysis
3.2.11 PAT—Process analytical technology
3.2.12 PID—Proportional integral derivative
3.2.13 PP—Process parameter
3.2.14 QA—Quality attribute
4. Summary of Practice
4.1 To aid reader understanding, a diagram of the data flows in a PAT-enabled control system is shown in Fig. 1. The diagram
shows how process and control models can be used in a closed loop control paradigm (with decisions being made based on action
limits set in the control model) but also for feed-forward control to downstream process steps/operations.
4.2 Fig. 2 shows how the quality attributes (QAs), noncritical as well as critical, are fed into the control model via the process
model. Each process has process parameters (PPs). Based on process understanding, some PPs are held static and others are subject
to dynamic adjustment. Some of the PPs directly or indirectly impact critical quality attributes (CQAs) and these PPs are called
critical process parameters (CPPs). When the CPPs (which may be fixed or adjustable) are dynamically adjusted as a result of
information generated by the process and control models, they are called critical control parameters (CCPs). Revised CCP settings
are transmitted in real time to the manufacturing equipment where they change the conditions of manufacture for the product.
4.3 Control Strategy:
FIG. 1 Data Flows for a PAT-Enabled Control System
E2629 − 20
FIG. 2 Relationship Between Quality Attributes and the Control System
4.3.1 The control strategy should be designed to control the quality of the product in response to potential variations in the
process, equipment conditions, incoming raw materials, or environmental factors over time. Control strategy implementations
generally can be categorized into three types:
4.3.1.1 Level 1: Quality Assurance by Means of Application of Dynamic or Adaptive Process Control System:
(1) A Level 1 control strategy utilizes Dynamic or Adaptive Process Control System to monitor and control the quality
attributes of materials in real-time.
(2) In Dynamic or Adaptive Process Control, system process parameters are monitored and may be adjusted in response to
disturbances to ensure that the quality attributes consistently conform to the established acceptance criteria.
(3) The successful application of a Dynamic or Adaptive Process Control system represents a high degree of product and
process understanding as the design of an engineering control system entails expressing the dynamic relationships among process
parameters, raw material and product attributes in a quantitative and predictive manner.
(4) The ability of a Dynamic or Adaptive Process Control System to compensate for variation in the raw material attributes
or external disturbances to the process condition significantly reduces the risk of producing of out of specification material and
hence the requirement for routine segregation/diversion of out-of-specification is also reduced.
(5) Statistical monitoring tools, for example Univariate or Multivariate SPC, may be used to demonstrate that the Dynamic or
Adaptive Process control system is ensuring that the process is operating in a State of Control where there is a very low probability
of out of specification being produced.
(6) Successful implementation of a Dynamic or Adaptive Process Control system directly supports a real-time release strategy.
4.3.1.2 Level 2: Quality Assurance by Means of Operation Within an Established Design Space and Confirmatory End Product
and In-Process Material Testing:
(1) Product Quality may be assured by combination of appropriate end product testing together with appropriate monitoring
of controlled raw material attributes and process parameters.
(2) The product and process understanding obtained through the establishment of a multivariate design space facilitates the
identification of potential sources of raw material and process variability that can impact product quality.
(3) Understanding the impact that variability from these sources has on in-process materials, downstream processing, and drug
product quality provides an opportunity to shift controls upstream and to reduce the reliance on end-product testing.
(4) The absence of real time corrective action within the control system increases the probability for production of out of
specification material and hence requires appropriate mechanisms for ensuring rejection/diversion of any out-of-specification
product.
4.3.1.3 Level 3: Quality Assurance by Means of Operation Within a Validated and Tightly Constrained Material Attributes and
Process Parameters and Release Based on End Product Testing:
(1) A Level 3, Control Strategy does not use PAT for either feedforward or feedback control and is therefore outside the scope
of this standard. Process analytics may however be used as a measurement of intermediate CQA as a form of in-process control
(to verify the process has remained in a state of control) or in place of end-of-line testing.
5. Significance and Use
5.1 This guide supports the principles of Guide E2500 and extends these principles to the verification of PAT-enabled control
systems.
“Modernizing Pharmaceutical Manufacturing: From Batch to Continuous Production,” Journal of Pharmaceutical Innovation, Vol 10, No 3, September 2015.
E2629 − 20
5.2 This guide clarifies what is important for verification of PAT-enabled control systems. Such systems are often complex and
require multidisciplinary and cross-functional teams to achieve optimum results. This guide provides a common basis for
understanding requirements for all involved disciplines such as control engineering, development, manufacturing, and process
validation.
6. Principles To Be Considered for Verification of PAT-Enabled Control Systems
6.1 Verification should be science and risk based. Quality risk management should drive the verification process. Practice E2476
provides additional guidance on risk assessments for PAT systems.
6.2 Verification should use the most efficient and effective method available to achieve the specified results, choosing from, for
example, simulation, testing, first principle modeling, or other approaches or combinations of these.
6.3 Verification should cover the design space of the manufacturing process. This will include all those ranges in which it is
necessary that the control system will be able to bring the process back into its intended operating range. The verification can occur
during Process Performance Qualification or prior (during development phase), or both, given the process system is in a qualified
state.
6.4 Verification of the control systems should always include verification of the system as a whole. It may also include
verification of individual system components.
6.5 The verification process should confirm that relevant quality attributes will be controlled concurrently.
6.6 Verification should ensure that the control system is stable throughout the range of operation.
6.7 Each component of the PAT-enabled control system should generate outputs with sufficient frequency, accuracy, and
precision to make the necessary level of pro
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...