ASTM D5236-23
(Test Method)Standard Test Method for Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill Method)
Standard Test Method for Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill Method)
SIGNIFICANCE AND USE
5.1 This test method is one of a number of tests conducted on heavy hydrocarbon mixtures to characterize these materials for a refiner or a purchaser. It provides an estimate of the yields of fractions of various boiling ranges.
5.2 The fractions made by this test method can be used alone or in combination with other fractions to produce samples for analytical studies and quality evaluations.
5.3 Residues to be used in the manufacture of asphalt can also be made but may not always be suitable. The long heat soaking that occurs in this test method may alter some of the properties.
Note 1: While the practice of reblending distillates with residue can be done to produce a lighter residue, it is not recommended because it produces blends with irregular properties.
5.4 Details of cutpoints must be mutually agreed upon before the test begins.
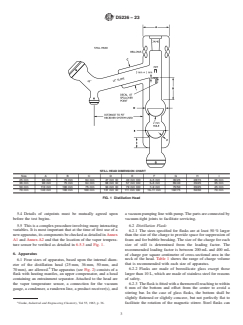
5.5 This is a complex procedure involving many interacting variables. It is most important that at the time of first use of a new apparatus, its components be checked as detailed in Annex A1 and Annex A2 and that the location of the vapor temperature sensor be verified as detailed in 6.5.3 and Fig. 1.
SCOPE
1.1 This test method covers the procedure for distillation of heavy hydrocarbon mixtures having initial boiling points greater than 150 °C (300 °F), such as heavy crude oils, petroleum distillates, residues, and synthetic mixtures. It employs a potstill with a low pressure drop entrainment separator operated under total takeoff conditions. Distillation conditions and equipment performance criteria are specified and typical apparatus is illustrated.
1.2 This test method details the procedures for the production of distillate fractions of standardized quality in the gas oil and lubricating oil range as well as the production of standard residue. In addition, it provides for the determination of standard distillation curves to the highest atmospheric equivalent temperature possible by conventional distillation.
1.3 The maximum achievable atmospheric equivalent temperature (AET) is dependent upon the heat tolerance of the charge. For most samples, a temperature up to 565 °C (1050 °F) can be attained. This maximum will be significantly lower for heat sensitive samples (for example, heavy residues) and might be somewhat higher for nonheat sensitive samples.
1.4 The recommended distillation method for crude oils up to cutpoint 400 °C (752 °F) AET is Test Method D2892. This test method can be used for heavy crude oils with initial boiling points greater than 150 °C (302 °F). However, distillation curves and fraction qualities obtained by these methods are not comparable.
1.5 This test method contains the following annexes:
1.5.1 Annex A1—Test Method for Determination of Temperature Response Time,
1.5.2 Annex A2—Practice for Calibration of Sensors,
1.5.3 Annex A3—Test Method for Dehydration of a Wet Sample of Oil,
1.5.4 Annex A4—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET), and
1.5.5 Annex A5—Test Method for Determination of Wettage.
1.6 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warnings, see 6.5.4.2, 6.5.6.3, 6.9.3, 9.5, 9.7, and A2.3.1.3.
1.8 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution when handling mercury and mercury-containing...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2023
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.08 - Volatility
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Jun-2020
- Effective Date
- 15-Jul-2017
Overview
ASTM D5236-23 is the Standard Test Method for Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill Method) developed by ASTM International. This method provides a detailed procedure for the vacuum distillation of heavy hydrocarbon mixtures, such as heavy crude oils, petroleum residues, and synthetic blends, with initial boiling points above 150 °C (300 °F). The purpose of this standard is to estimate the yields of various boiling fractions and to obtain distillate and residue samples for further analytical assessments and quality evaluations. The vacuum potstill approach allows for obtaining standardized fractions in the gas oil and lubricating oil range, as well as high-boiling residues, under controlled, low-pressure conditions that prevent thermal decomposition.
Key Topics
- Sample Characterization: The method is designed to characterize heavy hydrocarbon mixtures, providing refiners and purchasers with essential data regarding the boiling range distributions and yields.
- Fraction Yields: The procedure yields fractions that represent different boiling ranges, which can serve as standalone samples or be blended for in-depth studies.
- Distillation Procedure: Utilizes a potstill with a low pressure drop entrainment separator, operating under total takeoff and vacuum conditions, to safely and efficiently separate heavy hydrocarbon mixtures.
- Equipment Criteria: Specifies requirements for apparatus such as vacuum flasks, heating mantles, condensers, and temperature/pressure sensors to ensure accuracy and repeatability.
- Temperature and Pressure Control: Includes details about calibrating temperature and pressure sensors, and converting observed vapor temperatures to atmospheric equivalent temperatures.
- Safety and Quality: Stresses the importance of equipment inspection, operation within recommended parameters, and awareness of potential safety hazards, including those related to mercury.
Applications
- Refining and Petrochemicals: Widely used for characterizing heavy crude oils, petroleum distillates, and residues to assist in refining strategies and purchasing decisions.
- Quality Control: Enables laboratories to produce and analyze different distillation fractions to assess quality and suitability for downstream applications such as lubricants, fuels, and asphalt production.
- Analytical Studies: Provides fractions for further chemical analysis, such as density, viscosity, and composition determination.
- Product Development: Assists manufacturers in understanding the boiling profiles and properties of novel or synthetic heavy oil blends.
- Residue Evaluation: Supports the production of residues for potential asphalt manufacturing, while also noting that prolonged heating may alter residue properties.
Related Standards
- ASTM D2892: Standard Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column) - Recommended for crude oils with initial boiling points up to 400 °C AET.
- ASTM D4057, D4177: Standards for manual and automatic sampling of petroleum products.
- ASTM D941, D1217, D1298, D1480, D5002: Standards for measuring density and relative density of liquids.
- ASTM D1250: Guide for temperature and pressure-volume correction factors for crude oils and refined products.
Practical Value
Adopting ASTM D5236-23 ensures consistency in the distillation and analysis of heavy hydrocarbons, supporting critical decisions in refining, product specification, and quality control. By following this internationally recognized standard, organizations can:
- Obtain accurate boiling range distributions and distillate yields from heavy oil samples.
- Standardize laboratory and pilot plant studies for gas oil, lubricating oil, and residue production.
- Enhance reliability and comparability of results across organizations and regions.
Keywords: ASTM D5236, vacuum distillation, heavy oil, hydrocarbon mixtures, residue analysis, boiling point determination, petroleum refining, standard test method, potstill distillation, quality control, atmospheric equivalent temperature.
Buy Documents
ASTM D5236-23 - Standard Test Method for Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill Method)
REDLINE ASTM D5236-23 - Standard Test Method for Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill Method)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5236-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill Method)". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is one of a number of tests conducted on heavy hydrocarbon mixtures to characterize these materials for a refiner or a purchaser. It provides an estimate of the yields of fractions of various boiling ranges. 5.2 The fractions made by this test method can be used alone or in combination with other fractions to produce samples for analytical studies and quality evaluations. 5.3 Residues to be used in the manufacture of asphalt can also be made but may not always be suitable. The long heat soaking that occurs in this test method may alter some of the properties. Note 1: While the practice of reblending distillates with residue can be done to produce a lighter residue, it is not recommended because it produces blends with irregular properties. 5.4 Details of cutpoints must be mutually agreed upon before the test begins. 5.5 This is a complex procedure involving many interacting variables. It is most important that at the time of first use of a new apparatus, its components be checked as detailed in Annex A1 and Annex A2 and that the location of the vapor temperature sensor be verified as detailed in 6.5.3 and Fig. 1. SCOPE 1.1 This test method covers the procedure for distillation of heavy hydrocarbon mixtures having initial boiling points greater than 150 °C (300 °F), such as heavy crude oils, petroleum distillates, residues, and synthetic mixtures. It employs a potstill with a low pressure drop entrainment separator operated under total takeoff conditions. Distillation conditions and equipment performance criteria are specified and typical apparatus is illustrated. 1.2 This test method details the procedures for the production of distillate fractions of standardized quality in the gas oil and lubricating oil range as well as the production of standard residue. In addition, it provides for the determination of standard distillation curves to the highest atmospheric equivalent temperature possible by conventional distillation. 1.3 The maximum achievable atmospheric equivalent temperature (AET) is dependent upon the heat tolerance of the charge. For most samples, a temperature up to 565 °C (1050 °F) can be attained. This maximum will be significantly lower for heat sensitive samples (for example, heavy residues) and might be somewhat higher for nonheat sensitive samples. 1.4 The recommended distillation method for crude oils up to cutpoint 400 °C (752 °F) AET is Test Method D2892. This test method can be used for heavy crude oils with initial boiling points greater than 150 °C (302 °F). However, distillation curves and fraction qualities obtained by these methods are not comparable. 1.5 This test method contains the following annexes: 1.5.1 Annex A1—Test Method for Determination of Temperature Response Time, 1.5.2 Annex A2—Practice for Calibration of Sensors, 1.5.3 Annex A3—Test Method for Dehydration of a Wet Sample of Oil, 1.5.4 Annex A4—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET), and 1.5.5 Annex A5—Test Method for Determination of Wettage. 1.6 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warnings, see 6.5.4.2, 6.5.6.3, 6.9.3, 9.5, 9.7, and A2.3.1.3. 1.8 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution when handling mercury and mercury-containing...
SIGNIFICANCE AND USE 5.1 This test method is one of a number of tests conducted on heavy hydrocarbon mixtures to characterize these materials for a refiner or a purchaser. It provides an estimate of the yields of fractions of various boiling ranges. 5.2 The fractions made by this test method can be used alone or in combination with other fractions to produce samples for analytical studies and quality evaluations. 5.3 Residues to be used in the manufacture of asphalt can also be made but may not always be suitable. The long heat soaking that occurs in this test method may alter some of the properties. Note 1: While the practice of reblending distillates with residue can be done to produce a lighter residue, it is not recommended because it produces blends with irregular properties. 5.4 Details of cutpoints must be mutually agreed upon before the test begins. 5.5 This is a complex procedure involving many interacting variables. It is most important that at the time of first use of a new apparatus, its components be checked as detailed in Annex A1 and Annex A2 and that the location of the vapor temperature sensor be verified as detailed in 6.5.3 and Fig. 1. SCOPE 1.1 This test method covers the procedure for distillation of heavy hydrocarbon mixtures having initial boiling points greater than 150 °C (300 °F), such as heavy crude oils, petroleum distillates, residues, and synthetic mixtures. It employs a potstill with a low pressure drop entrainment separator operated under total takeoff conditions. Distillation conditions and equipment performance criteria are specified and typical apparatus is illustrated. 1.2 This test method details the procedures for the production of distillate fractions of standardized quality in the gas oil and lubricating oil range as well as the production of standard residue. In addition, it provides for the determination of standard distillation curves to the highest atmospheric equivalent temperature possible by conventional distillation. 1.3 The maximum achievable atmospheric equivalent temperature (AET) is dependent upon the heat tolerance of the charge. For most samples, a temperature up to 565 °C (1050 °F) can be attained. This maximum will be significantly lower for heat sensitive samples (for example, heavy residues) and might be somewhat higher for nonheat sensitive samples. 1.4 The recommended distillation method for crude oils up to cutpoint 400 °C (752 °F) AET is Test Method D2892. This test method can be used for heavy crude oils with initial boiling points greater than 150 °C (302 °F). However, distillation curves and fraction qualities obtained by these methods are not comparable. 1.5 This test method contains the following annexes: 1.5.1 Annex A1—Test Method for Determination of Temperature Response Time, 1.5.2 Annex A2—Practice for Calibration of Sensors, 1.5.3 Annex A3—Test Method for Dehydration of a Wet Sample of Oil, 1.5.4 Annex A4—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET), and 1.5.5 Annex A5—Test Method for Determination of Wettage. 1.6 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for information only and are not considered standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warnings, see 6.5.4.2, 6.5.6.3, 6.9.3, 9.5, 9.7, and A2.3.1.3. 1.8 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution when handling mercury and mercury-containing...
ASTM D5236-23 is classified under the following ICS (International Classification for Standards) categories: 71.080.01 - Organic chemicals in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5236-23 has the following relationships with other standards: It is inter standard links to ASTM D5236-18a, ASTM D2892-23, ASTM D2892-20, ASTM D1298-12b(2017)e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5236-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5236 − 23
Standard Test Method for
Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill
Method)
This standard is issued under the fixed designation D5236; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 1.5.4 Annex A4—Practice for Conversion of Observed Va-
por Temperature to Atmospheric Equivalent Temperature
1.1 This test method covers the procedure for distillation of
(AET), and
heavy hydrocarbon mixtures having initial boiling points
1.5.5 Annex A5—Test Method for Determination of Wet-
greater than 150 °C (300 °F), such as heavy crude oils,
tage.
petroleum distillates, residues, and synthetic mixtures. It em-
ploys a potstill with a low pressure drop entrainment separator
1.6 The values stated in SI units are to be regarded as
operated under total takeoff conditions. Distillation conditions
standard. The values given in parentheses after SI units are
and equipment performance criteria are specified and typical
provided for information only and are not considered standard.
apparatus is illustrated.
1.7 This standard does not purport to address all of the
1.2 This test method details the procedures for the produc-
safety concerns, if any, associated with its use. It is the
tion of distillate fractions of standardized quality in the gas oil
responsibility of the user of this standard to establish appro-
and lubricating oil range as well as the production of standard
priate safety, health, and environmental practices and deter-
residue. In addition, it provides for the determination of
mine the applicability of regulatory limitations prior to use.
standard distillation curves to the highest atmospheric equiva-
For specific warnings, see 6.5.4.2, 6.5.6.3, 6.9.3, 9.5, 9.7, and
lent temperature possible by conventional distillation.
A2.3.1.3.
1.3 The maximum achievable atmospheric equivalent tem-
1.8 WARNING—Mercury has been designated by many
perature (AET) is dependent upon the heat tolerance of the
regulatory agencies as a hazardous substance that can cause
charge. For most samples, a temperature up to 565 °C
serious medical issues. Mercury, or its vapor, has been dem-
(1050 °F) can be attained. This maximum will be significantly
onstrated to be hazardous to health and corrosive to materials.
lower for heat sensitive samples (for example, heavy residues)
Use Caution when handling mercury and mercury-containing
and might be somewhat higher for nonheat sensitive samples.
products. See the applicable product Safety Data Sheet (SDS)
1.4 The recommended distillation method for crude oils up
for additional information. The potential exists that selling
to cutpoint 400 °C (752 °F) AET is Test Method D2892. This
mercury or mercury-containing products, or both, is prohibited
test method can be used for heavy crude oils with initial boiling
by local or national law. Users must determine legality of sales
points greater than 150 °C (302 °F). However, distillation
in their location.
curves and fraction qualities obtained by these methods are not
1.9 This international standard was developed in accor-
comparable.
dance with internationally recognized principles on standard-
1.5 This test method contains the following annexes:
ization established in the Decision on Principles for the
1.5.1 Annex A1—Test Method for Determination of Tem-
Development of International Standards, Guides and Recom-
perature Response Time,
mendations issued by the World Trade Organization Technical
1.5.2 Annex A2—Practice for Calibration of Sensors,
Barriers to Trade (TBT) Committee.
1.5.3 Annex A3—Test Method for Dehydration of a Wet
Sample of Oil,
2. Referenced Documents
2.1 ASTM Standards:
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.08 on Volatility. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2023. Published November 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1992. Last previous edition approved in 2018 as D5236 – 18a. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D5236-23. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5236 − 23
D941 Test Method for Density and Relative Density (Spe- 3.1.8 loading, n—the volume of charge relative to the
cific Gravity) of Liquids by Lipkin Bicapillary Pycnom- cross-sectional area of the neck.
eter (Withdrawn 1993)
3.1.9 pressure drop, n—the difference between the operating
D1217 Test Method for Density and Relative Density (Spe-
pressure and the pressure measured in the distillation flask.
cific Gravity) of Liquids by Bingham Pycnometer
3.1.9.1 Discussion—It is a result of the friction developed
D1250 Guide for the Use of the Joint API and ASTM
by driving the vapors through the system expressed in kilopas-
Adjunct for Temperature and Pressure Volume Correction
cals (mm Hg).
Factors for Generalized Crude Oils, Refined Products, and
3.1.10 spillover point, n—the lowest point in the head above
Lubricating Oils: API MPMS Chapter 11.1
the entrainment separator over which the vapors can flow to the
D1298 Test Method for Density, Relative Density, or API
condensing region.
Gravity of Crude Petroleum and Liquid Petroleum Prod-
ucts by Hydrometer Method
3.1.11 static hold-up (or wettage), n—the amount of liquid
D1480 Test Method for Density and Relative Density (Spe-
material remaining on the inside of the walls of the apparatus
cific Gravity) of Viscous Materials by Bingham Pycnom-
after the distillation has been completed.
eter
3.1.11.1 Discussion—In this test method, it includes wettage
D2892 Test Method for Distillation of Crude Petroleum
of the distillation flask in the case of the steel flasks, but not in
(15-Theoretical Plate Column)
the case of glass flasks that are removed for weighing after the
D4057 Practice for Manual Sampling of Petroleum and
distillation is completed.
Petroleum Products
3.1.12 takeoff rate, n—the quantity of product removed per
D4177 Practice for Automatic Sampling of Petroleum and
unit time.
Petroleum Products
3.1.12.1 Discussion—It is approximately equal to the
D5002 Test Method for Density, Relative Density, and API
boil-up rate differing only by parasitic heat losses.
Gravity of Crude Oils by Digital Density Analyzer
4. Summary of Test Method
3. Terminology
3.1 Definitions of Terms Specific to This Standard: 4.1 A weighed volume of sample is distilled at absolute
3.1.1 atmospheric equivalent temperature (AET), n—the pressures between 6.6 kPa and 0.013 kPa (50 mm Hg and
temperature converted from the measured vapor temperature 0.1 mm Hg) at specified distillation rates. Cuts are taken at
using equations in Annex A4. preselected temperatures. Records of vapor temperature, oper-
3.1.1.1 Discussion—The AET is the expected vapor tem- ating pressure, and other variables are made at intervals,
perature if the distillation was performed at atmospheric including at each cutpoint.
pressure and there was no thermal decomposition.
4.2 The mass of each fraction is obtained. Distillation yields
3.1.2 boil-up rate, n—the quantity of vapor entering the
by mass are calculated from the mass of each fraction relative
distillation head per unit time.
to the total mass recovery.
3.1.2.1 Discussion—It is approximately equal to the takeoff
4.3 The density of each fraction is obtained. Distillation
rate, differing only by the parasitic heat losses. It is expressed
yields by volume are calculated from the volume computed for
in millilitres per hour for a head of any given internal diameter
each fraction at 15 °C (59 °F) relative to the total recovery.
or millilitres per hour per square centimetre of cross-sectional
area of the throat for comparative purposes. 4.4 Distillation curves of temperature versus mass or vol-
ume percent, or both, are drawn using the data from 4.2 and
3.1.3 condenser, n—the apparatus connected to the outlet of
4.3.
the distillation head in which condensation of the product
occurs.
5. Significance and Use
3.1.4 distillation flask, n—the flask, of glass or metal, in
5.1 This test method is one of a number of tests conducted
which the charge is boiled.
on heavy hydrocarbon mixtures to characterize these materials
3.1.4.1 Discussion—The flask is sometimes called a kettle
or pot. for a refiner or a purchaser. It provides an estimate of the yields
of fractions of various boiling ranges.
3.1.5 distillation head, n—the section immediately above
the distillation flask containing the entrainment separator.
5.2 The fractions made by this test method can be used
alone or in combination with other fractions to produce
3.1.6 distillation pressure (or operating pressure), n—the
samples for analytical studies and quality evaluations.
pressure measured in the distillation head just before the outlet
to the recovery system.
5.3 Residues to be used in the manufacture of asphalt can
3.1.7 distillation temperature (or vapor temperature),
also be made but may not always be suitable. The long heat
n—the temperature of the vapors in the distillation head at the soaking that occurs in this test method may alter some of the
point of measurement.
properties.
NOTE 1—While the practice of reblending distillates with residue can be
The last approved version of this historical standard is referenced on done to produce a lighter residue, it is not recommended because it
www.astm.org. produces blends with irregular properties.
D5236 − 23
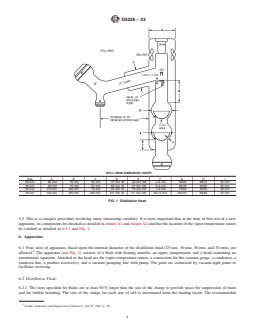
STILL HEAD DIMENSION CHART
Size A B C D E F G H I
25 mm 85 mm 75 mm 64 mm 47 mm ID 40 mm OD 4–5 mm 35/25 28/15 35 mm
36 mm 90 mm 75 mm 64 mm 68 mm ID 57 mm OD 5–6 mm 65/40 35/25 35 mm
50 mm 110 mm 100 mm 75 mm 94 mm ID 79 mm OD 7–9 mm 75/50 35/25 45 mm
70 mm 140 mm 100 mm 100 mm 131 mm ID 111 mm OD 10–11 mm 102/75 50/30 70 mm
FIG. 1 Distillation Head
5.4 Details of cutpoints must be mutually agreed upon a vacuum pumping line with pump. The parts are connected by
before the test begins. vacuum-tight joints to facilitate servicing.
5.5 This is a complex procedure involving many interacting
6.2 Distillation Flask:
variables. It is most important that at the time of first use of a
6.2.1 The sizes specified for flasks are at least 50 % larger
new apparatus, its components be checked as detailed in Annex
than the size of the charge to provide space for suppression of
A1 and Annex A2 and that the location of the vapor tempera-
foam and for bubble breaking. The size of the charge for each
ture sensor be verified as detailed in 6.5.3 and Fig. 1.
size of still is determined from the loading factor. The
recommended loading factor is between 200 mL and 400 mL
6. Apparatus
of charge per square centimetre of cross-sectional area in the
neck of the head. Table 1 shows the range of charge volume
6.1 Four sizes of apparatus, based upon the internal diam-
that is recommended with each size of apparatus.
eter of the distillation head (25 mm, 36 mm, 50 mm, and
6.2.2 Flasks are made of borosilicate glass except those
70 mm), are allowed. The apparatus (see Fig. 2) consists of a
larger than 10 L, which are made of stainless steel for reasons
flask with heating mantles, an upper compensator, and a head
of safety.
containing an entrainment separator. Attached to the head are
6.2.3 The flask is fitted with a thermowell reaching to within
the vapor temperature sensor, a connection for the vacuum
gauge, a condenser, a rundown line, a product receiver(s), and 6 mm of the bottom and offset from the center to avoid a
stirring bar. In the case of glass flasks, the bottom shall be
slightly flattened or slightly concave, but not perfectly flat to
Cooke, Industrial and Engineering Chemistry, Vol 55, 1963, p. 36. facilitate the rotation of the magnetic stirrer. Steel flasks can
D5236 − 23
FIG. 2 Apparatus
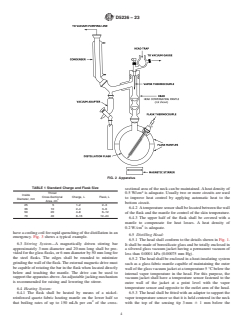
TABLE 1 Standard Charge and Flask Size
sectional area of the neck can be maintained. A heat density of
Throat 0.5 W/cm is adequate. Usually two or more circuits are used
Inside
Cross-Sectional Charge, L Flask, L
to improve heat control by applying automatic heat to the
Diameter, mm
Area, cm
bottom circuit.
25 5 1–2 2–3
6.4.2 A temperature sensor shall be located between the wall
36 10 2–4 3–6
50 20 4–8 6–12
of the flask and the mantle for control of the skin temperature.
70 40 8–16 12–24
6.4.3 The upper half of the flask shall be covered with a
mantle to compensate for heat losses. A heat density of
0.2 W ⁄cm is adequate.
have a cooling coil for rapid quenching of the distillation in an
6.5 Distilling Head:
emergency. Fig. 3 shows a typical example.
6.5.1 The head shall conform to the details shown in Fig. 1.
6.3 Stirring System—A magnetically driven stirring bar
It shall be made of borosilicate glass and be totally enclosed in
approximately 3 mm diameter and 20 mm long shall be pro-
a silvered glass vacuum jacket having a permanent vacuum of
vided for the glass flasks, or 6 mm diameter by 50 mm long for
less than 0.0001 kPa (0.00075 mm Hg).
the steel flasks. The edges shall be rounded to minimize
6.5.2 The head shall be enclosed in a heat-insulating system
grinding the wall of the flask. The external magnetic drive must
such as a glass fabric mantle capable of maintaining the outer
be capable of rotating the bar in the flask when located directly
wall of the glass vacuum jacket at a temperature 5 °C below the
below and touching the mantle. The drive can be used to
internal vapor temperature in the head. For this purpose, the
support the apparatus above. An adjustable jacking mechanism
vacuum jacket shall have a temperature sensor fastened to the
is recommended for raising and lowering the stirrer.
outer wall of the jacket at a point level with the vapor
temperature sensor and opposite to the outlet arm of the head.
6.4 Heating System:
6.4.1 The flask shall be heated by means of a nickel- 6.5.3 The head shall be fitted with an adapter to support the
reinforced quartz fabric heating mantle on the lower half so vapor temperature sensor so that it is held centered in the neck
that boiling rates of up to 150 mL/h per cm of the cross- with the top of the sensing tip 3 mm 6 1 mm below the
D5236 − 23
SYSTEM SIZE A B
25 mm 35/25 3 L
36 mm 65/40 6 L
50 mm 75/50 12 L
70 mm 102/75 24 L
FIG. 3 Distillation Flask
spillover point. This dimension can be checked by removing 6.5.4.3 Verification of the calibration of the vapor tempera-
the temperature sensor and inserting in its place a copper wire ture measuring devices is to be made on a regular basis.
having a short right angle bend at the bottom. By feeling for the Verification at least once a month is recommended. Verification
spillover point, the distance from the top joint of the adaptor of the calibration of the sensors can be accomplished poten-
can be found. Laying the wire on the temperature sensor will tiometrically by the use of standard precision resistance or by
then permit checking of this dimension. distilling a pure compound with accurately known boiling
6.5.4 The vapor temperature sensor shall be either a plati- point, as described in A2.2.3.
num resistance thermometer, a thermocouple with the junction 6.5.5 A head trap as illustrated in Fig. 4 shall be fitted to the
head fused to the lower tip of the well, or any other device adapter described in 6.5.3 for connection to the vacuum sensor.
which meets the requirements in 6.5.4 and 6.5.4.1. It shall have It shall be kept filled with crushed dry ice at all times while in
a response time of less than 60 s as described in Annex A1. service.
6.5.4.1 The vapor temperature measuring device shall have 6.5.6 A vacuum sensor shall be connected to the sidearm of
an accuracy of 0.5 °C or better and be measured with a the trap. The sensor shall be capable of reading the pressure
resolution of 0.1 °C or better. with a precision equal to or better than 0.00133 kPa (0.01 mm
6.5.4.2 The vapor temperature measuring device shall be Hg), whichever is greater. A non-tilting McLeod gauge or other
calibrated over the full range of useful temperatures in com- primary reference device can achieve this accuracy when
bination with its associated instrument at the time of first use properly used, but a mercury manometer will permit this
and at least once per year thereafter as described in A2.2.2. accuracy only down to a pressure of about 1 kPa and then only
Alternatively, certified sensors may be used, provided the when read with a good cathetometer (an instrument based on a
calibration of the sensor and its associated recording instru- telescope mounted on a vernier scale to determine levels very
ment can be traced back to a primary temperature standard. accurately). Also, electronic sensors of the diaphragm type
Recalibrate when either the sensor or the instrument is repaired have been found satisfactory. Vacuum gauges based on hot
or serviced. (Warning—Vapor temperature measurement is wires, radiation, or conductivity detectors are not recom-
one of the two major sources of error in distillation data.) mended.
D5236 − 23
FIG. 4 Head Trap and Temperature Sensor
6.5.6.1 Primary standards, such as the non-tilting McLeod
gauge, mercury manometer, or other analogous primary stan-
dard pressure devices, can be used without calibration when
properly used and maintained. Alternatively, a tensimeter or
certified electronic sensors may be used, provided the calibra-
tion of the sensor and its associated recording instrument can
be traced back to a primary pressure standard.
CONDENSER DIMENSION CHART
6.5.6.2 Noncertified gauges shall be calibrated from a non-
System Size A B C D
tilting McLeod gauge, another primary reference device, or a
25 mm 51 mm 28 mm 300 mm 35/25
secondary electronic standard traceable to a primary standard. 36 mm 75 mm 45 mm 300 mm 65/40
50 mm 80 mm 54 mm 400 mm 75/50
A basic calibration procedure is described in A2.3. Recalibrate
70 mm 120 mm 80 mm 400 mm 102/75
when either the sensor or the instrument is repaired or serviced.
6.5.6.3 Verification of the calibration of pressure sensors is
FIG. 5 Condenser
to be made on a regular basis. A frequency of at least once a
week is recommended. Verification of the calibration of the
sensors can be accomplished using the procedures described in 6.7.3 An isolation valve of a diameter at least equal to the
A2.3 or against a certified reference system. (Warning— diameter of the pumping line shall be connected between the
Measurement of vacuum (operating pressure) is one of the two surge tank and the vacuum pump.
major sources of error in the distillation procedure. It is 6.7.4 A dewar-type trap made of borosilicate glass, such as
therefore of prime importance that the instructions on calibra- that illustrated in Fig. 4, shall be placed between the top of the
tion and verification be followed with great care and on a distillation head and the vacuum sensor. It shall be kept filled
routine basis.) with crushed dry ice at all times during the distillation to
protect the vacuum system from contamination with residual
6.6 Condenser—A condenser made of borosilicate glass
vapors.
shall be connected to the outlet arm of the head (see Fig. 5). It
shall have sufficient capacity to condense essentially all vapors 6.8 Vacuum Source—A single-stage mechanical vacuum
and capable of operating at coolant temperatures up to 70 °C to pump capable of maintaining a steady pressure in the system at
prevent wax buildup. all operating pressures shall be connected to the pumping line.
Automatic or manual control can be used.
6.7 Pumping Line:
6.7.1 A pumping line shall be connected from the outlet of 6.9 Recovery System:
the condenser to the vacuum pump. The pumping line can be 6.9.1 The recovery system is connected to the lower outlet
made of heavy-walled rubber or light metal tubing, but its of the product condenser and consists of a vacuum adapter to
inside diameter must be greater than half the inside diameter of permit removal of distillate receivers without disturbing the
the outlet of the condenser and less than 2 m long. pressure in the system. A suitable manual device is illustrated
6.7.2 A surge tank of a size at least equal to the capacity of in Fig. 6.
the flask shall be inserted in the pumping line adjacent to the 6.9.2 Alternatively, either automatic or manual devices can
pump. be used to collect part or all of the fractions within the system
D5236 − 23
7.2 The sample must be in a closed container when received
and show no evidence of leakage.
7.3 If the sample looks waxy or has solidified, warm it
enough to liquefy it and ensure that it is thoroughly mixed
before using.
7.4 If, upon examination, there is evidence of water in the
sample, perform a preliminary distillation as described in
Annex A3.
8. Preparation of Apparatus
8.1 Clean and dry all glass parts and assemble them with
freshly lubricated joints as shown in Fig. 2. In the case of ball
joints, use only enough lubricant to produce a thin continuous
film. An excess of lubricant can promote leakage. The rings of
O-ring joints should be made of Vitron-A, or silicone of
equivalent hardness, and be lightly lubricated.
8.2 Tare the receivers to the nearest 0.1 % of the weight of
the charge.
8.3 To check for leaks, pump the system down to a pressure
of approximately 0.05 kPa (0.4 mm Hg) and isolate it from the
vacuum source. If, after 1 min, the rise in pressure is no greater
than 0.01 kPa (0.075 mm Hg), the system is acceptable. If the
rise in pressure is greater than 0.01 kPa (0.075 mm Hg) in
1 min, the gauge and its connections must be examined and
leaks corrected before proceeding.
8.4 Calibrate the temperature and pressure sensors as de-
scribed in Annex A2.
9. Procedure
INTERMEDIATE RECEIVER DIMENSION CHART
9.1 Determine the density of the sample by one of the
System Size A B C
following test methods: Test Method D941, D1217, D1480,
25 mm 45 mm 120 mm 35/25
36 mm 51 mm 120 mm 35/25
D5002, or D1298. Refer to Guide D1250 to correct densities to
50 mm 64 mm 150 mm 50/30
15 °C.
70 mm 75 mm 150 mm 50/30
9.2 Insert the stirring bar.
FIG. 6 Receiver System
9.3 From Table 1, determine the volume of the charge and
calculate the mass to be charged by multiplying its density by
the desired volume.
without disturbing the operating pressure until the end of the
run. Heating must be provided when needed to maintain the
9.4 Weigh this mass of charge into the flask to the nearest
product in the liquid state.
0.1 %. In the case of flasks too large to handle, the flask can be
6.9.3 The product receivers shall be made of borosilicate
put in place and the charge drawn in from a container (weighed
glass and large enough for the size of the fractions to be
with its transfer line) using a pressure of 90 kPa to 95 kPa in
collected. They shall be calibrated to the nearest 1 % from the
the still. The charge may need to be warmed to facilitate
bottom. (Warning—This apparatus operates under high
transfer. Its mass can be determined from the difference.
vacuum and high temperature. It is recommended that these
9.5 Attach the flask to the column (in the case of smaller
stills be kept in an enclosure to ensure that in case of an
flasks), and put on all the heating mantles. Put the stirring
implosion, the operator and others nearby are protected from
device in place and turn it on. (Warning—Ensure that the
flying debris, but that the front, at least, be transparent and
safety shield is in place.)
removable for access to controls and so forth. Automated stills,
9.6 A contiguous cutting scheme may be achieved in one of
which are left unattended for long periods, should be equipped
with an automatic fire extinguisher, automatic quench, and two ways, while remaining within the scope of the key aspects
of this method. It may be achieved by gradually reducing the
alarm.)
pressure over the course of the distillation (dynamic) or done
7. Sampling
stepwise, by slowing (or stopping) the takeoff rate to allow
7.1 Obtain the sample for distillation in accordance with
instructions given in Practice D4057 or Practice D4177. The
Vitron A is a registered trademark of DuPont E.I. De Nemours and Co.,
sample can also be a residue from Test Method D2892. Wilmington, DE 19898.
D5236 − 23
TABLE 2 Operating Pressures and Distillation Rates
Operating Boil-Up
Take-Off Rate, mL/h
Pressure, Rate, mL/
25 mm 36 mm 50 mm 70 mm
kPa (mm Hg) (h·×·cm )
6.67 (50) 90–150 450–750 900–1500 1800–3000 3600–6000
1.33 (10) 75–125 375–625 750–1250 1500–2500 3000–5000
0.133 (1) 45–75 225–375 450–750 900–1500 1800–3000
0.0400 (0.3) 30–50 150–250 300–500 600–1000 1200–2000
0.0133 (0.1) 10–20 50–100 100–200 200–400 400–800
lowering of the operating pressure to achieve the final cut and the differential between the vapor and pot temperature as
temperature. In each case, this must be done keeping in mind the operating envelope. Estimating that the difference between
the necessity to avoid starving the distillation (due to a slow the vapor and the pot temperature remain relatively constant,
takeoff rate), while at the same time avoiding entrainment (by determine if the final vapor temperature can be achieved at this
reducing too quickly the pressure applied to the system). pressure while remaining within the recommended limitations
of the flask temperature (see 9.18). If the final cutpoint cannot
9.7 Apply heat to the flask at a rate that will raise the
be achieved at the starting pressure, the pressure should be
temperature of the charge quickly, but no faster than 300 °C ⁄h
gradually lowered toward an operating pressure that will allow
(540 °F ⁄h). Do not exceed a skin temperature on the flask of
the final cut to be taken. This must be done bearing in mind the
400 °C (750 °F) or cracking may result on the walls of the
associated takeoff rates for the vacuum pressures indicated in
flask. (Warning—Some hydrocarbon mixtures cannot tolerate
Table 2 and the limitations of the pot temperature. The pressure
400 °C for any useful length of time. Reducing the skin
should be lowered enough to allow the takeoff rate to acceler-
temperature may be necessary in these cases.)
ate briefly while the operator is remaining vigilant to avoid
9.8 Turn on the head compensation mantle and maintain the
entrainment. The distillation rate at the operating pressure
outer wall of the glass vacuum jacket at a temperature
should fall within the recommendations stated in Table 2 and
approximately 40 °C below the temperature of the liquid in the
should be allowed to stabilize for at least 2 min before arriving
flask.
at a cutpoint. Experience has shown that reduced crude oil
samples typically run well at 0.133 kPa for up to 25 % to 30 %
9.9 Reduce the pressure in the system gradually to a suitable
starting pressure. Choose from Table 2 the highest pressure that of the charge volume. Subsequent lowering of the operating
pressure, as described above, has yielded satisfactory results.
is consistent with the expected initial boiling point as well as
the lowest pressure that is consistent with the maximum Repeat this procedure throughout the remainder of the distil-
lation until an operating pressure has been attained that will
cutpoint, using Fig. 7 as a guide. A pressure of 0.133 kPa
(1.0 mm Hg) has been
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5236 − 18a D5236 − 23
Standard Test Method for
Distillation of Heavy Hydrocarbon Mixtures (Vacuum Potstill
Method)
This standard is issued under the fixed designation D5236; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the procedure for distillation of heavy hydrocarbon mixtures having initial boiling points greater than
150 °C (300 °F), such as heavy crude oils, petroleum distillates, residues, and synthetic mixtures. It employs a potstill with a low
pressure drop entrainment separator operated under total takeoff conditions. Distillation conditions and equipment performance
criteria are specified and typical apparatus is illustrated.
1.2 This test method details the procedures for the production of distillate fractions of standardized quality in the gas oil and
lubricating oil range as well as the production of standard residue. In addition, it provides for the determination of standard
distillation curves to the highest atmospheric equivalent temperature possible by conventional distillation.
1.3 The maximum achievable atmospheric equivalent temperature (AET) is dependent upon the heat tolerance of the charge. For
most samples, a temperature up to 565 °C (1050 °F) can be attained. This maximum will be significantly lower for heat sensitive
samples (for example, heavy residues) and might be somewhat higher for nonheat sensitive samples.
1.4 The recommended distillation method for crude oils up to cutpoint 400 °C (752 °F) AET is Test Method D2892. This test
method can be used for heavy crude oils with initial boiling points greater than 150 °C (302 °F). However, distillation curves and
fraction qualities obtained by these methods are not comparable.
1.5 This test method contains the following annexes:
1.5.1 Annex A1—Test Method for Determination of Temperature Response Time,
1.5.2 Annex A2—Practice for Calibration of Sensors,
1.5.3 Annex A3—Test Method for Dehydration of a Wet Sample of Oil,
1.5.4 Annex A4—Practice for Conversion of Observed Vapor Temperature to Atmospheric Equivalent Temperature (AET), and
1.5.5 Annex A5—Test Method for Determination of Wettage.
1.6 The values stated in SI units are to be regarded as standard. The values given in parentheses after SI units are provided for
information only and are not considered standard.
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.08 on Volatility.
Current edition approved July 1, 2018Nov. 1, 2023. Published July 2018November 2023. Originally approved in 1992. Last previous edition approved in 2018 as D5236
– 18. DOI: 10.1520/D5236-18A.– 18a. DOI: 10.1520/D5236-23.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5236 − 23
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. For specific warnings, see 6.5.4.2, 6.5.6.3, 6.9.3, 9.5, 9.7, and A2.3.1.3.
1.8 WARNING—Mercury has been designated by many regulatory agencies as a hazardous substance that can cause serious
medical issues. Mercury, or its vapor, has been demonstrated to be hazardous to health and corrosive to materials. Use Caution
when handling mercury and mercury-containing products. See the applicable product Safety Data Sheet (SDS) for additional
information. The potential exists that selling mercury or mercury-containing products, or both, is prohibited by local or national
law. Users must determine legality of sales in their location.
1.9 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D941 Test Method for Density and Relative Density (Specific Gravity) of Liquids by Lipkin Bicapillary Pycnometer (Withdrawn
1993)
D1217 Test Method for Density and Relative Density (Specific Gravity) of Liquids by Bingham Pycnometer
D1250 Guide for the Use of the Joint API and ASTM Adjunct for Temperature and Pressure Volume Correction Factors for
Generalized Crude Oils, Refined Products, and Lubricating Oils: API MPMS Chapter 11.1
D1298 Test Method for Density, Relative Density, or API Gravity of Crude Petroleum and Liquid Petroleum Products by
Hydrometer Method
D1480 Test Method for Density and Relative Density (Specific Gravity) of Viscous Materials by Bingham Pycnometer
D2892 Test Method for Distillation of Crude Petroleum (15-Theoretical Plate Column)
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
D5002 Test Method for Density, Relative Density, and API Gravity of Crude Oils by Digital Density Analyzer
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 atmospheric equivalent temperature (AET), n—the temperature converted from the measured vapor temperature using
equations in Annex A4.
3.1.1.1 Discussion—
The AET is the expected vapor temperature if the distillation was performed at atmospheric pressure and there was no thermal
decomposition.
3.1.2 boil-up rate, n—the quantity of vapor entering the distillation head per unit time.
3.1.2.1 Discussion—
It is approximately equal to the takeoff rate, differing only by the parasitic heat losses. It is expressed in millilitres per hour for
a head of any given internal diameter or millilitres per hour per square centimetre of cross-sectional area of the throat for
comparative purposes.
3.1.3 condenser, n—the apparatus connected to the outlet of the distillation head in which condensation of the product occurs.
3.1.4 distillation flask, n—the flask, of glass or metal, in which the charge is boiled.
3.1.4.1 Discussion—
The flask is sometimes called a kettle or pot.
3.1.5 distillation head, n—the section immediately above the distillation flask containing the entrainment separator.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
D5236 − 23
3.1.6 distillation pressure (or operating pressure), n—the pressure measured in the distillation head just before the outlet to the
recovery system.
3.1.7 distillation temperature (or vapor temperature), n—the temperature of the vapors in the distillation head at the point of
measurement.
3.1.8 loading, n—the volume of charge relative to the cross-sectional area of the neck.
3.1.9 pressure drop, n—the difference between the operating pressure and the pressure measured in the distillation flask.
3.1.9.1 Discussion—
It is a result of the friction developed by driving the vapors through the system expressed in kilopascals (mm Hg).
3.1.10 spillover point, n—the lowest point in the head above the entrainment separator over which the vapors can flow to the
condensing region.
3.1.11 static hold-up (or wettage), n—the amount of liquid material remaining on the inside of the walls of the apparatus after the
distillation has been completed.
3.1.11.1 Discussion—
In this test method, it includes wettage of the distillation flask in the case of the steel flasks, but not in the case of glass flasks that
are removed for weighing after the distillation is completed.
3.1.12 takeoff rate, n—the quantity of product removed per unit time.
3.1.12.1 Discussion—
It is approximately equal to the boil-up rate differing only by parasitic heat losses.
4. Summary of Test Method
4.1 A weighed volume of sample is distilled at absolute pressures between 6.6 kPa and 0.013 kPa (50 mm Hg and 0.1 mm Hg)
at specified distillation rates. Cuts are taken at preselected temperatures. Records of vapor temperature, operating pressure, and
other variables are made at intervals, including at each cutpoint.
4.2 The mass of each fraction is obtained. Distillation yields by mass are calculated from the mass of each fraction relative to the
total mass recovery.
4.3 The density of each fraction is obtained. Distillation yields by volume are calculated from the volume computed for each
fraction at 15 °C (59 °F) relative to the total recovery.
4.4 Distillation curves of temperature versus mass or volume percent, or both, are drawn using the data from 4.2 and 4.3.
5. Significance and Use
5.1 This test method is one of a number of tests conducted on heavy hydrocarbon mixtures to characterize these materials for a
refiner or a purchaser. It provides an estimate of the yields of fractions of various boiling ranges.
5.2 The fractions made by this test method can be used alone or in combination with other fractions to produce samples for
analytical studies and quality evaluations.
5.3 Residues to be used in the manufacture of asphalt can also be made but may not always be suitable. The long heat soaking
that occurs in this test method may alter some of the properties.
NOTE 1—While the practice of reblending distillates with residue can be done to produce a lighter residue, it is not recommended because it produces
blends with irregular properties.
5.4 Details of cutpoints must be mutually agreed upon before the test begins.
D5236 − 23
STILL HEAD DIMENSION CHART
Size A B C D E F G H I
25 mm 85 mm 75 mm 64 mm 47 mm ID 40 mm OD 4–5 mm 35/25 28/15 35 mm
36 mm 90 mm 75 mm 64 mm 68 mm ID 57 mm OD 5–6 mm 65/40 35/25 35 mm
50 mm 110 mm 100 mm 75 mm 94 mm ID 79 mm OD 7–9 mm 75/50 35/25 45 mm
70 mm 140 mm 100 mm 100 mm 131 mm ID 111 mm OD 10–11 mm 102/75 50/30 70 mm
FIG. 1 Distillation Head
5.5 This is a complex procedure involving many interacting variables. It is most important that at the time of first use of a new
apparatus, its components be checked as detailed in Annex A1 and Annex A2 and that the location of the vapor temperature sensor
be verified as detailed in 6.5.3 and Fig. 1.
6. Apparatus
6.1 Four sizes of apparatus, based upon the internal diameter of the distillation head (25 mm, 36 mm, 50 mm, and 70 mm), are
allowed. The apparatus (see Fig. 2) consists of a flask with heating mantles, an upper compensator, and a head containing an
entrainment separator. Attached to the head are the vapor temperature sensor, a connection for the vacuum gauge, a condenser, a
rundown line, a product receiver(s), and a vacuum pumping line with pump. The parts are connected by vacuum-tight joints to
facilitate servicing.
6.2 Distillation Flask:
6.2.1 The sizes specified for flasks are at least 50 % larger than the size of the charge to provide space for suppression of foam
and for bubble breaking. The size of the charge for each size of still is determined from the loading factor. The recommended
Cooke, Industrial and Engineering Chemistry, Vol 55, 1963, p. 36.
D5236 − 23
FIG. 2 Apparatus
loading factor is between 200 mL and 400 mL of charge per square centimetre of cross-sectional area in the neck of the head. Table
1 shows the range of charge volume that is recommended with each size of apparatus.
6.2.2 Flasks are made of borosilicate glass except those larger than 10 L, which are made of stainless steel for reasons of safety.
6.2.3 The flask is fitted with a thermowell reaching to within 6 mm of the bottom and offset from the center to avoid a stirring
bar. In the case of glass flasks, the bottom shall be slightly flattened or slightly concave, but not perfectly flat to facilitate the
rotation of the magnetic stirrer. Steel flasks can have a cooling coil for rapid quenching of the distillation in an emergency. Fig.
3 shows a typical example.
6.3 Stirring System—A magnetically driven stirring bar approximately 3 mm diameter and 20 mm long shall be provided for the
glass flasks, or 6 mm diameter by 50 mm long for the steel flasks. The edges shall be rounded to minimize grinding the wall of
the flask. The external magnetic drive must be capable of rotating the bar in the flask when located directly below and touching
the mantle. The drive can be used to support the apparatus above. An adjustable jacking mechanism is recommended for raising
and lowering the stirrer.
6.4 Heating System:
6.4.1 The flask shall be heated by means of a nickel-reinforced quartz fabric heating mantle on the lower half so that boiling rates
2 2
of up to 150 mL/h per cm of the cross-sectional area of the neck can be maintained. A heat density of 0.5 W/cm is adequate.
Usually two or more circuits are used to improve heat control by applying automatic heat to the bottom circuit.
6.4.2 A temperature sensor shall be located between the wall of the flask and the mantle for control of the skin temperature.
D5236 − 23
TABLE 1 Standard Charge and Flask Size
Throat
Inside
Cross-Sectional Charge, L Flask, L
Diameter, mm
Area, cm
25 5 1–2 2–3
36 10 2–4 3–6
50 20 4–8 6–12
70 40 8–16 12–24
6.4.3 The upper half of the flask shall be covered with a mantle to compensate for heat losses. A heat density of 0.2 W ⁄cm is
adequate.
6.5 Distilling Head:
6.5.1 The head shall conform to the details shown in Fig. 1. It shall be made of borosilicate glass and be totally enclosed in a
silvered glass vacuum jacket having a permanent vacuum of less than 0.0001 kPa (0.00075 mm Hg).
6.5.2 The head shall be enclosed in a heat-insulating system such as a glass fabric mantle capable of maintaining the outer wall
of the glass vacuum jacket at a temperature 5 °C below the internal vapor temperature in the head. For this purpose, the vacuum
jacket shall have a temperature sensor fastened to the outer wall of the jacket at a point level with the vapor temperature sensor
and opposite to the outlet arm of the head.
6.5.3 The head shall be fitted with an adapter to support the vapor temperature sensor so that it is held centered in the neck with
the top of the sensing tip 3 mm 6 1 mm below the spillover point. This dimension can be checked by removing the temperature
sensor and inserting in its place a copper wire having a short right angle bend at the bottom. By feeling for the spillover point,
the distance from the top joint of the adaptor can be found. Laying the wire on the temperature sensor will then permit checking
of this dimension.
6.5.4 The vapor temperature sensor shall be either a platinum resistance thermometer, a thermocouple with the junction head fused
to the lower tip of the well, or any other device which meets the requirements in 6.5.4 and 6.5.4.1. It shall have a response time
of less than 60 s as described in Annex A1.
6.5.4.1 The vapor temperature measuring device shall have an accuracy of 0.5 °C or better and be measured with a resolution of
0.1 °C or better.
6.5.4.2 The vapor temperature measuring device shall be calibrated over the full range of useful temperatures in combination with
its associated instrument at the time of first use and at least once per year thereafter as described in A2.2.2. Alternatively, certified
sensors may be used, provided the calibration of the sensor and its associated recording instrument can be traced back to a primary
temperature standard. Recalibrate when either the sensor or the instrument is repaired or serviced. (Warning—Vapor temperature
measurement is one of the two major sources of error in distillation data.)
6.5.4.3 Verification of the calibration of the vapor temperature measuring devices is to be made on a regular basis. Verification
at least once a month is recommended. Verification of the calibration of the sensors can be accomplished potentiometrically by the
use of standard precision resistance or by distilling a pure compound with accurately known boiling point, as described in A2.2.3.
6.5.5 A head trap as illustrated in Fig. 4 shall be fitted to the adapter described in 6.5.3 for connection to the vacuum sensor. It
shall be kept filled with crushed dry ice at all times while in service.
6.5.6 A vacuum sensor shall be connected to the sidearm of the trap. The sensor shall be capable of reading the pressure with a
precision equal to or better than 0.00133 kPa (0.01 mm Hg), whichever is greater. A non-tilting McLeod gauge or other primary
reference device can achieve this accuracy when properly used, but a mercury manometer will permit this accuracy only down to
a pressure of about 1 kPa and then only when read with a good cathetometer (an instrument based on a telescope mounted on a
vernier scale to determine levels very accurately). Also, electronic sensors of the diaphragm type have been found satisfactory.
Vacuum gauges based on hot wires, radiation, or conductivity detectors are not recommended.
6.5.6.1 Primary standards, such as the non-tilting McLeod gauge, mercury manometer, or other analogous primary standard
D5236 − 23
SYSTEM SIZE A B
25 mm 35/25 3 L
36 mm 65/40 6 L
50 mm 75/50 12 L
70 mm 102/75 24 L
FIG. 3 Distillation Flask
FIG. 4 Head Trap and Temperature Sensor
pressure devices, can be used without calibration when properly used and maintained. Alternatively, a tensimeter or certified
electronic sensors may be used, provided the calibration of the sensor and its associated recording instrument can be traced back
to a primary pressure standard.
D5236 − 23
CONDENSER DIMENSION CHART
System Size A B C D
25 mm 51 mm 28 mm 300 mm 35/25
36 mm 75 mm 45 mm 300 mm 65/40
50 mm 80 mm 54 mm 400 mm 75/50
70 mm 120 mm 80 mm 400 mm 102/75
FIG. 5 Condenser
6.5.6.2 Noncertified gauges shall be calibrated from a non-tilting McLeod gauge, another primary reference device, or a secondary
electronic standard traceable to a primary standard. A basic calibration procedure is described in A2.3. Recalibrate when either the
sensor or the instrument is repaired or serviced.
6.5.6.3 Verification of the calibration of pressure sensors is to be made on a regular basis. A frequency of at least once a week is
recommended. Verification of the calibration of the sensors can be accomplished using the procedures described in A2.3 or against
a certified reference system. (Warning—Measurement of vacuum (operating pressure) is one of the two major sources of error
in the distillation procedure. It is therefore of prime importance that the instructions on calibration and verification be followed
with great care and on a routine basis.)
6.6 Condenser—A condenser made of borosilicate glass shall be connected to the outlet arm of the head (see Fig. 5). It shall have
sufficient capacity to condense essentially all vapors and capable of operating at coolant temperatures up to 70 °C to prevent wax
buildup.
6.7 Pumping Line:
6.7.1 A pumping line shall be connected from the outlet of the condenser to the vacuum pump. The pumping line can be made
of heavy-walled rubber or light metal tubing, but its inside diameter must be greater than half the inside diameter of the outlet of
the condenser and less than 2 m long.
D5236 − 23
INTERMEDIATE RECEIVER DIMENSION CHART
System Size A B C
25 mm 45 mm 120 mm 35/25
36 mm 51 mm 120 mm 35/25
50 mm 64 mm 150 mm 50/30
70 mm 75 mm 150 mm 50/30
FIG. 6 Receiver System
6.7.2 A surge tank of a size at least equal to the capacity of the flask shall be inserted in the pumping line adjacent to the pump.
6.7.3 An isolation valve of a diameter at least equal to the diameter of the pumping line shall be connected between the surge tank
and the vacuum pump.
6.7.4 A dewar-type trap made of borosilicate glass, such as that illustrated in Fig. 4, shall be placed between the top of the
distillation head and the vacuum sensor. It shall be kept filled with crushed dry ice at all times during the distillation to protect
the vacuum system from contamination with residual vapors.
6.8 Vacuum Source—A single-stage mechanical vacuum pump capable of maintaining a steady pressure in the system at all
operating pressures shall be connected to the pumping line. Automatic or manual control can be used.
6.9 Recovery System:
6.9.1 The recovery system is connected to the lower outlet of the product condenser and consists of a vacuum adapter to permit
removal of distillate receivers without disturbing the pressure in the system. A suitable manual device is illustrated in Fig. 6.
D5236 − 23
6.9.2 Alternatively, either automatic or manual devices can be used to collect part or all of the fractions within the system without
disturbing the operating pressure until the end of the run. Heating must be provided when needed to maintain the product in the
liquid state.
6.9.3 The product receivers shall be made of borosilicate glass and large enough for the size of the fractions to be collected. They
shall be calibrated to the nearest 1 % from the bottom. (Warning—This apparatus operates under high vacuum and high
temperature. It is recommended that these stills be kept in an enclosure to ensure that in case of an implosion, the operator and
others nearby are protected from flying debris, but that the front, at least, be transparent and removable for access to controls and
so forth. Automated stills, which are left unattended for long periods, should be equipped with an automatic fire extinguisher,
automatic quench, and alarm.)
7. Sampling
7.1 Obtain the sample for distillation in accordance with instructions given in Practice D4057 or Practice D4177. The sample can
also be a residue from Test Method D2892.
7.2 The sample must be in a closed container when received and show no evidence of leakage.
7.3 If the sample looks waxy or has solidified, warm it enough to liquefy it and ensure that it is thoroughly mixed before using.
7.4 If, upon examination, there is evidence of water in the sample, perform a preliminary distillation as described in Annex A3.
8. Preparation of Apparatus
8.1 Clean and dry all glass parts and assemble them with freshly lubricated joints as shown in Fig. 2. In the case of ball joints,
use only enough lubricant to produce a thin continuous film. An excess of lubricant can promote leakage. The rings of O-ring joints
should be made of Vitron-A, or silicone of equivalent hardness, and be lightly lubricated.
8.2 Tare the receivers to the nearest 0.1 % of the weight of the charge.
8.3 To check for leaks, pump the system down to a pressure of approximately 0.05 kPa (0.4 mm Hg) and isolate it from the vacuum
source. If, after 1 min, the rise in pressure is no greater than 0.01 kPa (0.075 mm Hg), the system is acceptable. If the rise in
pressure is greater than 0.01 kPa (0.075 mm Hg) in 1 min, the gauge and its connections must be examined and leaks corrected
before proceeding.
8.4 Calibrate the temperature and pressure sensors as described in Annex A2.
9. Procedure
9.1 Determine the density of the sample by one of the following test methods: Test Method D941, D1217, D1480, D5002, or
D1298. Refer to Guide D1250 to correct densities to 15 °C.
9.2 Insert the stirring bar.
9.3 From Table 1, determine the volume of the charge and calculate the mass to be charged by multiplying its density by the
desired volume.
9.4 Weigh this mass of charge into the flask to the nearest 0.1 %. In the case of flasks too large to handle, the flask can be put
in place and the charge drawn in from a container (weighed with its transfer line) using a pressure of 90 kPa to 95 kPa in the still.
The charge may need to be warmed to facilitate transfer. Its mass can be determined from the difference.
Vitron A is a registered trademark of DuPont E.I. De Nemours and Co., Wilmington, DE 19898.
D5236 − 23
TABLE 2 Operating Pressures and Distillation Rates
Operating Boil-Up
Take-Off Rate, mL/h
Pressure, Rate, mL/
25 mm 36 mm 50 mm 70 mm
kPa (mm Hg) (h·×·cm )
6.67 (50) 90–150 450–750 900–1500 1800–3000 3600–6000
1.33 (10) 75–125 375–625 750–1250 1500–2500 3000–5000
0.133 (1) 45–75 225–375 450–750 900–1500 1800–3000
0.0400 (0.3) 30–50 150–250 300–500 600–1000 1200–2000
0.0133 (0.1) 10–20 50–100 100–200 200–400 400–800
9.5 Attach the flask to the column (in the case of smaller flasks), and put on all the heating mantles. Put the stirring device in place
and turn it on. (Warning—Ensure that the safety shield is in place.)
9.6 A contiguous cutting scheme may be achieved in one of two ways, while remaining within the scope of the key aspects of
this method. It may be achieved by gradually reducing the pressure over the course of the distillation (dynamic) or done stepwise,
by slowing (or stopping) the takeoff rate to allow lowering of the operating pressure to achieve the final cut temperature. In each
case, this must be done keeping in mind the necessity to avoid starving the distillation (due to a slow takeoff rate), while at the
same time avoiding entrainment (by reducing too quickly the pressure applied to the system).
9.7 Apply heat to the flask at a rate that will raise the temperature of the charge quickly, but no faster than 300 °C ⁄h (540 °F ⁄h).
Do not exceed a skin temperature on the flask of 400 °C (750 °F) or cracking may result on the walls of the flask.
(Warning—Some hydrocarbon mixtures cannot tolerate 400 °C for any useful length of time. Reducing the skin temperature may
be necessary in these cases.)
9.8 Turn on the head compensation mantle and maintain the outer wall of the glass vacuum jacket at a temperature approximately
40 °C below the temperature of the liquid in the flask.
9.9 Reduce the pressure in the system gradually to a suitable starting pressure. Choose from Table 2 the highest pressure that is
consistent with the expected initial boiling point as well as the lowest pressure that is consistent with the maximum cutpoint, using
Fig. 7 as a guide. A pressure of 0.133 kPa (1.0 mm Hg) has been found satisfactory for starting a material having an initial boiling
point of 343 °C (650 °F) AET, such as residues from Test Method D2892 distillations.
NOTE 2—Degassing of the charge is sometimes evident before the actual distillation begins. This appears as bubbling at the surface without generation
of condensable vapors.
9.10 When distillation begins, evidenced by vapors entering the neck of the flask, reduce the heat input to a level that will maintain
the chosen distillation rate from Table 2 (see Note 2). Adjust the heat compensator on the head to maintain the outer wall of the
glass vacuum jacket at a temperature 5 °C below the vapor temperature.
NOTE 3—Although a range of distillation rates is permitted, 80 % of the maximum allowed is recommended.
9.11 In cases in which the observed initial vapor temperature will be 150 °C (302 °F) or lower, it is desirable to refrigerate the
first fraction receiver to ensure the retention of light ends. If solid, waxy material appears on the walls, warm the receiver with
an infrared heat lamp or hot air gun to liquify the product in the receiver in order to improve the accuracy of the reading. In
automatic operation, the receivers must be thermostated at a temperature high enough to ensure that no solidification takes place
and low enough to prevent evaporation of light material.
9.12 When using the dynamic method of pressure reduction, calculate a projected final cutpoint using the operating pressure and
the differential between the vapor and pot temperature as the operating envelope. Estimating that the difference between the vapor
and the pot temperature remain relatively constant, determine if the final vapor tempe
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...