ASTM D8084-17
(Test Method)Standard Test Method for Photoelectrochemical Oxygen Demand of Freshwater Sources for Drinking Water Treatment Plants and Treated Drinking Water
Standard Test Method for Photoelectrochemical Oxygen Demand of Freshwater Sources for Drinking Water Treatment Plants and Treated Drinking Water
SIGNIFICANCE AND USE
5.1 This test method describes a rapid method to determine the maximum quantity of oxygen that may be consumed by impurities in water. As outlined in Test Methods D1252, chemical oxygen demand is typically used to monitor and control oxygen-consuming pollutants, both organic and inorganic, in domestic and industrial wastewaters. This photoelectrochemical oxygen demand test method is specific for measuring organics and inorganics in freshwater sources for drinking water treatment plants and treated drinking water matrices. This photoelectrochemical oxygen demand test method is not intended for domestic and industrial wastewaters to replace Test Methods D1252.
5.2 This test method does not require the use of the hazardous reagents, such as mercuric sulfate, potassium dichromate and silver sulfate, that are associated with chemical oxygen demand. It can also provide a result more rapidly than chemical oxygen demand as samples do not require reflux.
SCOPE
1.1 This test method covers a protocol for the determination of the photoelectrochemical oxygen demand of freshwater sources for drinking water treatment plants and treated drinking water in the range of 0.7 mg/L to 20 mg/L. Higher levels may be determined by sample dilution.
1.2 Photoelectrochemical oxygen demand is determined using the current generated from the photoelectrochemical oxidation of the sample using titanium dioxide (TiO2) irradiated with ultraviolet (UV) light from a light-emitting diode (LED).
1.3 This test method does not require the use of the hazardous reagents, such as mercuric sulfate, potassium dichromate and silver sulfate, that are often associated with the determination of chemical oxygen demand (that is, Test Methods D1252). It can also provide a result rapidly, as samples do not require reflux.
1.4 Determination of photoelectrochemical oxygen demand in freshwater sources for drinking water treatment plants and treated drinking water matrices has important implications for assessing treatment efficacy. Photoelectrochemical oxygen demand can be used as a bulk surrogate measure of natural organic matter, a key target for drinking water treatment. In aerobic biological treatment processes, determination of photoelectrochemical oxygen demand can provide an estimation of the oxygen required by microorganisms to degrade organic matter. This test method is complementary to existing natural organic matter (NOM) monitoring techniques and will help scientists and engineers further the understanding of NOM in water with a rapid oxygen demand test.
1.5 This test method was used successfully with reagent grade water spiked with pure compounds, freshwater sources for drinking water treatment plants and treated drinking water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices.
1.6 This test method is applicable to oxidizable matter,
Note 1: This test method can be performed (1) immediately in the field or laboratory on an unpreserved sample, and (2) in the laboratory on a properly preserved sample following the stated hold times.
1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jul-2017
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 15-Jun-2012
- Effective Date
- 15-Jun-2012
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jan-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 01-Sep-2006
- Effective Date
- 01-Sep-2006
- Effective Date
- 15-Aug-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 15-Feb-2006
- Refers
ASTM D1252-06 - Standard Test Methods for Chemical Oxygen Demand (Dichromate Oxygen Demand) of Water - Effective Date
- 15-Feb-2006
- Effective Date
- 01-Mar-2004
Overview
ASTM D8084-17 is the Standard Test Method for Photoelectrochemical Oxygen Demand (PEOD) of Freshwater Sources for Drinking Water Treatment Plants and Treated Drinking Water. Developed by ASTM International, this standard provides a rapid and practical method for quantifying the maximum oxygen consumption potential of impurities in freshwater sources used in drinking water treatment facilities. Unlike traditional chemical oxygen demand (COD) methods, this test utilizes photoelectrochemical oxidation and does not require hazardous reagents, providing faster results and improved safety for laboratory and field use.
PEOD is a valuable tool for water quality professionals, scientists, and engineers monitoring the efficacy of drinking water treatment processes, as it measures both organic and inorganic oxygen-consuming pollutants in a targeted and efficient manner.
Key Topics
- Purpose and Scope: This method covers the determination of photoelectrochemical oxygen demand for freshwaters and treated drinking water in concentrations ranging from 0.7 mg/L to 20 mg/L, with higher levels measurable by dilution.
- Testing Principle: PEOD is determined by measuring the current generated during the photoelectrochemical oxidation of water samples. This process uses a titanium dioxide (TiO₂) electrode irradiated by ultraviolet (UV) light from an LED.
- Safety Advantages: The method eliminates the use of hazardous chemicals such as mercuric sulfate, potassium dichromate, and silver sulfate associated with classic COD analysis. No refluxing is required, making the process both safer and quicker.
- Testing Flexibility: The standard can be applied in the field or in the lab, either on unpreserved or properly preserved samples, following recommended hold times.
- Treatment Process Assessment: Results can help estimate oxygen requirements for biological processes and serve as a surrogate measurement of natural organic matter (NOM), aiding operational decisions in water utilities.
Applications
- Drinking Water Source Analysis: Assess the oxygen demand in raw water sources prior to treatment, aiding in the selection and optimization of treatment strategies.
- Treatment Efficacy Monitoring: Evaluate the removal of oxygen-demanding substances during filtration, biological treatment, or chemical processes in drinking water treatment plants.
- Rapid Water Quality Assessment: Deliver near real-time results for oxygen demand, supporting operational adjustments and ensuring regulatory compliance.
- Natural Organic Matter (NOM) Characterization: Provide an indirect but reliable estimation of NOM in water, which is crucial for managing disinfection by-product formation and ensuring safe drinking water.
- Complement to Existing Techniques: Use alongside other monitoring techniques, such as traditional COD, for a more comprehensive understanding of water quality in treatment processes.
Related Standards
- ASTM D1252: Test Methods for Chemical Oxygen Demand (Dichromate Oxygen Demand) of Water-traditional method for measuring COD with hazardous reagents.
- ASTM D1129: Terminology Relating to Water-provides terminology used across water analysis standards.
- ASTM D1193: Specification for Reagent Water-establishes water purity requirements.
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water.
- ASTM D3370: Practices for Sampling Water from Closed Conduits-guidelines for representative sample collection.
- ASTM D5847: Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis.
By adopting ASTM D8084-17, water utilities and laboratories benefit from a safe, rapid, and reliable method for assessing the oxygen demand in water intended for human consumption, ensuring high standards of public health and operational efficiency.
Keywords: photoelectrochemical oxygen demand, PEOD, ASTM D8084-17, water quality, drinking water treatment, organics, inorganics, oxygen-consuming pollutants, natural organic matter, NOM, chemical oxygen demand alternative, rapid water analysis.
Buy Documents
ASTM D8084-17 - Standard Test Method for Photoelectrochemical Oxygen Demand of Freshwater Sources for Drinking Water Treatment Plants and Treated Drinking Water
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D8084-17 is a standard published by ASTM International. Its full title is "Standard Test Method for Photoelectrochemical Oxygen Demand of Freshwater Sources for Drinking Water Treatment Plants and Treated Drinking Water". This standard covers: SIGNIFICANCE AND USE 5.1 This test method describes a rapid method to determine the maximum quantity of oxygen that may be consumed by impurities in water. As outlined in Test Methods D1252, chemical oxygen demand is typically used to monitor and control oxygen-consuming pollutants, both organic and inorganic, in domestic and industrial wastewaters. This photoelectrochemical oxygen demand test method is specific for measuring organics and inorganics in freshwater sources for drinking water treatment plants and treated drinking water matrices. This photoelectrochemical oxygen demand test method is not intended for domestic and industrial wastewaters to replace Test Methods D1252. 5.2 This test method does not require the use of the hazardous reagents, such as mercuric sulfate, potassium dichromate and silver sulfate, that are associated with chemical oxygen demand. It can also provide a result more rapidly than chemical oxygen demand as samples do not require reflux. SCOPE 1.1 This test method covers a protocol for the determination of the photoelectrochemical oxygen demand of freshwater sources for drinking water treatment plants and treated drinking water in the range of 0.7 mg/L to 20 mg/L. Higher levels may be determined by sample dilution. 1.2 Photoelectrochemical oxygen demand is determined using the current generated from the photoelectrochemical oxidation of the sample using titanium dioxide (TiO2) irradiated with ultraviolet (UV) light from a light-emitting diode (LED). 1.3 This test method does not require the use of the hazardous reagents, such as mercuric sulfate, potassium dichromate and silver sulfate, that are often associated with the determination of chemical oxygen demand (that is, Test Methods D1252). It can also provide a result rapidly, as samples do not require reflux. 1.4 Determination of photoelectrochemical oxygen demand in freshwater sources for drinking water treatment plants and treated drinking water matrices has important implications for assessing treatment efficacy. Photoelectrochemical oxygen demand can be used as a bulk surrogate measure of natural organic matter, a key target for drinking water treatment. In aerobic biological treatment processes, determination of photoelectrochemical oxygen demand can provide an estimation of the oxygen required by microorganisms to degrade organic matter. This test method is complementary to existing natural organic matter (NOM) monitoring techniques and will help scientists and engineers further the understanding of NOM in water with a rapid oxygen demand test. 1.5 This test method was used successfully with reagent grade water spiked with pure compounds, freshwater sources for drinking water treatment plants and treated drinking water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices. 1.6 This test method is applicable to oxidizable matter, Note 1: This test method can be performed (1) immediately in the field or laboratory on an unpreserved sample, and (2) in the laboratory on a properly preserved sample following the stated hold times. 1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9. 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method describes a rapid method to determine the maximum quantity of oxygen that may be consumed by impurities in water. As outlined in Test Methods D1252, chemical oxygen demand is typically used to monitor and control oxygen-consuming pollutants, both organic and inorganic, in domestic and industrial wastewaters. This photoelectrochemical oxygen demand test method is specific for measuring organics and inorganics in freshwater sources for drinking water treatment plants and treated drinking water matrices. This photoelectrochemical oxygen demand test method is not intended for domestic and industrial wastewaters to replace Test Methods D1252. 5.2 This test method does not require the use of the hazardous reagents, such as mercuric sulfate, potassium dichromate and silver sulfate, that are associated with chemical oxygen demand. It can also provide a result more rapidly than chemical oxygen demand as samples do not require reflux. SCOPE 1.1 This test method covers a protocol for the determination of the photoelectrochemical oxygen demand of freshwater sources for drinking water treatment plants and treated drinking water in the range of 0.7 mg/L to 20 mg/L. Higher levels may be determined by sample dilution. 1.2 Photoelectrochemical oxygen demand is determined using the current generated from the photoelectrochemical oxidation of the sample using titanium dioxide (TiO2) irradiated with ultraviolet (UV) light from a light-emitting diode (LED). 1.3 This test method does not require the use of the hazardous reagents, such as mercuric sulfate, potassium dichromate and silver sulfate, that are often associated with the determination of chemical oxygen demand (that is, Test Methods D1252). It can also provide a result rapidly, as samples do not require reflux. 1.4 Determination of photoelectrochemical oxygen demand in freshwater sources for drinking water treatment plants and treated drinking water matrices has important implications for assessing treatment efficacy. Photoelectrochemical oxygen demand can be used as a bulk surrogate measure of natural organic matter, a key target for drinking water treatment. In aerobic biological treatment processes, determination of photoelectrochemical oxygen demand can provide an estimation of the oxygen required by microorganisms to degrade organic matter. This test method is complementary to existing natural organic matter (NOM) monitoring techniques and will help scientists and engineers further the understanding of NOM in water with a rapid oxygen demand test. 1.5 This test method was used successfully with reagent grade water spiked with pure compounds, freshwater sources for drinking water treatment plants and treated drinking water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices. 1.6 This test method is applicable to oxidizable matter, Note 1: This test method can be performed (1) immediately in the field or laboratory on an unpreserved sample, and (2) in the laboratory on a properly preserved sample following the stated hold times. 1.7 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9. 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D8084-17 is classified under the following ICS (International Classification for Standards) categories: 13.060.20 - Drinking water. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8084-17 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D1252-06(2012), ASTM D2777-12, ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D2777-08, ASTM D3370-07, ASTM D1129-06a, ASTM D1129-06ae1, ASTM D2777-06, ASTM D1193-06, ASTM D1129-06, ASTM D1252-06, ASTM D1129-04. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8084-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8084 − 17
Standard Test Method for
Photoelectrochemical Oxygen Demand of Freshwater
Sources for Drinking Water Treatment Plants and Treated
Drinking Water
This standard is issued under the fixed designation D8084; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope It is the user’s responsibility to ensure the validity of this test
method for waters of untested matrices.
1.1 This test method covers a protocol for the determination
of the photoelectrochemical oxygen demand of freshwater 1.6 This test method is applicable to oxidizable matter, <50
sources for drinking water treatment plants and treated drink- µm that can be introduced into the sensor.
ing water in the range of 0.7 mg/L to 20 mg/L. Higher levels
NOTE 1—This test method can be performed (1) immediately in the
may be determined by sample dilution.
field or laboratory on an unpreserved sample, and (2) in the laboratory on
a properly preserved sample following the stated hold times.
1.2 Photoelectrochemical oxygen demand is determined
using the current generated from the photoelectrochemical
1.7 The values stated in SI units are to be regarded as
oxidation of the sample using titanium dioxide (TiO ) irradi-
standard. No other units of measurement are included in this
ated with ultraviolet (UV) light from a light-emitting diode
standard.
(LED).
1.8 This standard does not purport to address all of the
1.3 This test method does not require the use of the
safety concerns, if any, associated with its use. It is the
hazardous reagents, such as mercuric sulfate, potassium di- responsibility of the user of this standard to establish appro-
chromate and silver sulfate, that are often associated with the
priate safety and health practices and determine the applica-
determination of chemical oxygen demand (that is, Test Meth-
bility of regulatory limitations prior to use. For specific hazard
ods D1252). It can also provide a result rapidly, as samples do
statements, see Section 9.
not require reflux.
1.9 This international standard was developed in accor-
dance with internationally recognized principles on standard-
1.4 Determination of photoelectrochemical oxygen demand
ization established in the Decision on Principles for the
in freshwater sources for drinking water treatment plants and
Development of International Standards, Guides and Recom-
treated drinking water matrices has important implications for
mendations issued by the World Trade Organization Technical
assessing treatment efficacy. Photoelectrochemical oxygen de-
Barriers to Trade (TBT) Committee.
mand can be used as a bulk surrogate measure of natural
organic matter, a key target for drinking water treatment. In
2. Referenced Documents
aerobic biological treatment processes, determination of pho-
toelectrochemicaloxygendemandcanprovideanestimationof
2.1 ASTM Standards:
the oxygen required by microorganisms to degrade organic
D1129 Terminology Relating to Water
matter. This test method is complementary to existing natural
D1193 Specification for Reagent Water
organic matter (NOM) monitoring techniques and will help
D1252 Test Methods for Chemical Oxygen Demand (Di-
scientists and engineers further the understanding of NOM in
chromate Oxygen Demand) of Water
water with a rapid oxygen demand test.
D2777 Practice for Determination of Precision and Bias of
Applicable Test Methods of Committee D19 on Water
1.5 This test method was used successfully with reagent
D3370 Practices for Sampling Water from Closed Conduits
grade water spiked with pure compounds, freshwater sources
D5847 Practice for Writing Quality Control Specifications
for drinking water treatment plants and treated drinking water.
for Standard Test Methods for Water Analysis
This test method is under the jurisdiction of ASTM Committee D19 on Water
andisthedirectresponsibilityofSubcommitteeD19.06onMethodsforAnalysisfor For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Organic Substances in Water. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Current edition approved July 15, 2017. Published August 2017. DOI: 10.1520/ Standards volume information, refer to the standard’s Document Summary page on
D8084-17. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8084 − 17
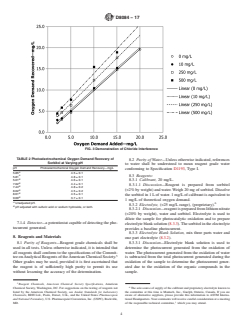
TABLE 1 Photoelectrochemical Oxygen Demand Recovery of
3. Terminology
A
Various Pure Compounds in Reagent Grade Water
3.1 Definitions—For definitions of terms used in this
Range of Theoretical
standard, refer to Terminology D1129.
Pure Compound Oxygen Demand Slope (mx+b)
Added—mg/L
3.1.1 chemical oxygen demand, n—the amount of oxygen
Caffeine 1.2–24.2 0.69x – 0.36
required under specified test conditions for the oxidation of
Phenylalanine 0.9–17.8 1.14x – 1.18
water borne organic and inorganic matter.
Sodium Acetate 0.7–13.3 0.79x – 1.34
Sodium Formate 0.3–6.7 0.96x – 0.58
3.2 Definitions of Terms Specific to This Standard:
Sodium Oxalate 0.2–3.3 1.47x – 0.13
3.2.1 theoretical oxygen demand, n—the calculated amount Tryptophan 0.9–18.8 1.01x – 0.62
Tyrosine 0.9–17.0 1.01x – 0.87
of oxygen required to oxidize a compound to its final oxidation
A
Adapted from Stoddart,A., K., and Gagnon, G.A., “Application of Photoelectro-
products based on stoichiometry.
chemical Chemical Oxygen Demand to Drinking Water,” Journal of American
Water Works Association, Vol 106, No. 9, 2014, pp. 383–390.
4. Summary of Test Method
4.1 Photoelectrochemical oxygen demand is determined by
photoelectrochemical oxidation. A sample is mixed with an
electrolyte containing lithium nitrate (LiNO ) and introduced
5. Significance and Use
into a sensor containing TiO nanoparticles immobilized on a
5.1 This test method describes a rapid method to determine
thin film. The TiO is irradiated with a UV LED, oxidizing the
the maximum quantity of oxygen that may be consumed by
sample and generating an electrical signal proportional to the
impurities in water. As outlined in Test Methods D1252,
chemical oxygen demand of the sample. A schematic of this
chemical oxygen demand is typically used to monitor and
working principle is provided in Fig. 1.
control oxygen-consuming pollutants, both organic and
4.2 Photoelectrochemical oxygen demand recovery of
inorganic, in domestic and industrial wastewaters. This photo-
known amounts of theoretical oxygen demand (as pure com-
electrochemical oxygen demand test method is specific for
pound) in reagent grade water are shown in Table 1.
measuring organics and inorganics in freshwater sources for
4.2.1 A slope of unity for a given compound in Table 1
drinking water treatment plants and treated drinking water
demonstrates that photoelectrochemical oxygen demand was a
matrices. This photoelectrochemical oxygen demand test
complete predictor of theoretical oxygen demand over the
methodisnotintendedfordomesticandindustrialwastewaters
theoreticaloxygendemandrangetested.Eachrangecomprised
to replace Test Methods D1252.
five concentrations.
5.2 This test method does not require the use of the
4.3 Photoelectrochemical oxygen demand recovery of hazardous reagents, such as mercuric sulfate, potassium di-
known amounts of theoretical oxygen demand (as sorbitol) in chromate and silver sulfate, that are associated with chemical
reagent grade water, surface water, filtered drinking water and oxygen demand. It can also provide a result more rapidly than
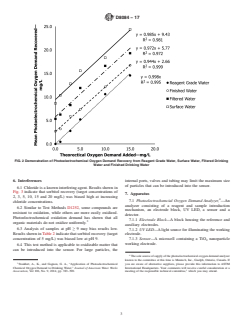
finished drinking water are shown in Fig. 2. chemical oxygen demand as samples do not require reflux.
Adapted from Zhao, H., Jiang, D., Zhang, S., Catterall, K., and John, R.,
“Development of a Direct Photoelectrochemical Method for Determination of
Chemical Oxygen Demand,” Analytical Chemistry, Vol 76, No. 1, 2004, pp.
155–160.
FIG. 1 Working Principle of Photoelectrochemcial Oxygen De-
mand Determination
D8084 − 17
FIG. 2 Demonstration of Photoelectrochemical Oxygen Demand Recovery from Reagent Grade Water, Surface Water, Filtered Drinking
Water and Finished Drinking Water
6. Interferences internal ports, valves and tubing may limit the maximum size
of particles that can be introduced into the sensor.
6.1 Chloride is a known interfering agent. Results shown in
Fig. 3 indicate that sorbitol recovery (target concentrations of
7. Apparatus
2, 3, 5, 10, 15 and 20 mg/L) was biased high at increasing
7.1 Photoelectrochemcial Oxygen Demand Analyzer, —An
chloride concentrations.
analyzer consisting of a reagent and sample introduction
6.2 Similar to Test Methods D1252, some compounds are
mechanism, an electrode block, UV LED, a sensor and a
resistant to oxidation, while others are more easily oxidized.
detector.
Photoelectrochemical oxidation demand has shown that all
7.1.1 Electrode Block—A block housing the reference and
organic materials do not oxidize uniformly.
auxiliary electrodes.
6.3 Analysis of samples at pH ≥ 9 may bias results low.
7.1.2 UV LED—Alight source for illuminating the working
Results shown in Table 2 indicate that sorbitol recovery (target
electrode.
concentration of 5 mg/L) was biased low at pH 9.
7.1.3 Sensor—A microcell containing a TiO nanoparticle
working electrode.
6.4 This test method is applicable to oxidizable matter that
can be introduced into the sensor. For large particles, the
Thesolesourceofsupplyofthephotoelectrochemicaloxygendemandanalyzer
known to the committee at this time is Mantech, Inc., Guelph, Ontario, Canada. If
Stoddart, A., K., and Gagnon, G. A., “Application of Photoelectrochemical you are aware of alternative suppliers, please provide this information to ASTM
Chemical Oxygen Demand to Drinking Water,” Journal of American Water Works International Headquarters. Your comments will receive careful consideration at a
Association, Vol 106, No. 9, 2014, pp. 383–390. meeting of the responsible technical committee, which you may attend.
D8084 − 17
FIG. 3 Demonstration of Chloride Interference
TABLE 2 Photoelectrochemical Oxygen Demand Recovery of
8.2 Purity of Water—Unless otherwise indicated, references
Sorbitol at Varying pH
to water shall be understood to mean reagent grade water
pH Photoelectrochemical Oxygen Demand Recovery—mg/L
conforming to Specification D1193, Type I.
A
5.85 4.5±0.1
A 8.3 Reagents:
5.81 4.8±0.1
B
5.01 4.6±0.1
8.3.1 Calibrant, 20 mg/L.
B
7.02 4.4±0.1
8.3.1.1 Discussion—Reagent is prepared from sorbitol
B
7.44 4.8±0.2
B
(<2% by weight) and water.Weigh 20 mg of sorbitol. Dissolve
8.02 4.5±0.2
B
8.61 4.5±0.1
the sorbitol in 1 Lof water. 1 mg/Lof calibrant is equivalent to
B
9.00 3.7±0.1
1 mg/L of theoretical oxygen demand.
A
Unadjusted pH.
8.3.2 Electrolyte, (<25 mg/L range), (proprietary).
B
pH adjusted with sulfuric acid or sodium hydroxide, or both.
8.3.2.1 Discussion—reagent is prepared from lithium nitrate
(<20% by weight), water and sorbitol. Electrolyte is used to
dilute the sample for photocatalytic oxidation and to prepare
7.1.4 Detector—a potentiostat capable of detecting the pho-
electrolyteblanksolution(8.3.3).Thesorbitolintheelectrolyte
tocurrent generated.
provides a baseline photocurrent.
8.3.3 Electrolyte Blank Solution, mix three parts water and
8. Reagents and Materials
one part electrolyte (8.3.2).
8.1 Purity of Reagents—Reagent grade chemicals shall be 8.3.3.1 Discussion—Electrolyte blank solution is used to
used in all tests. Unless otherwise indicated, it is intended that determine the photocurrent generated from the oxidation of
all reagents shall conform to the specifications of the Commit- water. The photocurrent generated from the oxidation of water
teeonAnalyticalReagentsoftheAmericanChemicalSociety. is subtracted from the total photocurrent generated during the
Other grades may be used, provided it is first ascertained that oxidation of the sample to determine the photocurrent gener-
the reagent is of sufficiently high purity to permit its use ated due to the oxidation of the organic compounds in the
without lessening the accuracy of the determination. sample.
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not The sole source of supply of the calibrant and proprietary electrolyte known to
listed by the American Chemical Society, see Analar Standards for Laboratory the committee at this time is Mantech, Inc., Guelph, Ontario, Canada. If you are
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia aware of alternative suppliers, please provide this information to ASTM Interna-
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, tional Headquarters.Your comments will receive careful consideration at a meeting
MD. of the responsible technical committee, which you may attend.
D8084 − 17
NOTE 2—Follow manufacturers’instructions regarding product storage
photoelectrochemical oxygen demand analyzer (7.1). Ensure
and shelf life.
pump volume settings are within the limits recommended by
8.4 Materials: the manufacturer.
8.4.1 Sample Vials, disposable 50-mL centrifuge tubes.
11.4 Introduce the electrolyte blank solution (8.3.3)tothe
8.4.2 Racks, suitable for sample vials.
blank solution port of the photoelectrochemical oxygen de-
8.4.3 Macropipette, 1–10 mL
mand analyzer (7.1) and prime the flow line three times.
8.4.4 Graduated Cylinders, various sizes.
12. Calibration and Standardization
9. Hazards
12.1 Calibration:
9.1 Warning—The UV light is automatically turned off
12.1.1 Using a macropipette (8.4.3), prepare an adequate
when the photoelectrochemical oxygen demand analyzer is
volume of calibrant for calibration by mixing three parts
opened.However,asasafetymeasure,donotlookdirectlyinto
calibrant (8.3.1) with one part electrolyte (8.3.2) in a sample
the UV light source at any time.
vial (8.4.1).
12.1.1.1 The rate of use of the calibrant (8.3.1) will depend
9.2 Warning—The electrolyte solution contains lithium
on the pump volume settings of the photoelectrochemical
nitrate, which is a skin irritant.
oxygen demand analyzer (7.1). Ensure pump volume settings
are within the lim
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...