ASTM D5110-22a
(Practice)Standard Practice for Calibration of Ozone Monitors and Certification of Ozone Transfer Standards Using Ultraviolet Photometry
Standard Practice for Calibration of Ozone Monitors and Certification of Ozone Transfer Standards Using Ultraviolet Photometry
SIGNIFICANCE AND USE
5.1 The reactivity and instability of O3 preclude the storage of O3 concentration standards for any practical length of time, and precludes direct certification of O3 concentrations as Standard Reference Materials (SRMs). Moreover, there is no available SRM that can be readily and directly adapted to the generation of O3 standards analogous to permeation devices and standard gas cylinders for sulfur dioxide and nitrogen oxides. Dynamic generation of O3 concentrations is relatively easy with a source of ultraviolet (UV) radiation. However, accurately certifying an O3 concentration as a primary standard requires assay of the concentration by a comprehensively specified analytical procedure, which must be performed every time a standard is needed (10).
5.2 This practice is not designed for the routine calibration of O3 monitors at remote locations (see Practices D5011).
SCOPE
1.1 This practice covers a means for calibrating ambient, workplace, or indoor ozone monitors, and for certifying transfer standards to be used for that purpose.
1.2 This practice describes means by which dynamic streams of ozone in air can be designated as primary ozone standards.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 8 for specific precautionary statements.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2022
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.03 - Ambient Atmospheres and Source Emissions
Relations
- Effective Date
- 01-Sep-2020
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Oct-2019
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Mar-2017
- Effective Date
- 15-Oct-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Dec-2014
- Effective Date
- 01-May-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Nov-2013
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-May-2011
Overview
ASTM D5110-22a Standard Practice for Calibration of Ozone Monitors and Certification of Ozone Transfer Standards Using Ultraviolet Photometry establishes a reliable method for calibrating ozone (O₃) monitors and certifying transfer standards. Developed by ASTM International, this standard is essential due to the reactive and unstable nature of ozone, which makes long-term storage or direct certification of ozone standards impractical. Instead, dynamic generation and UV photometric analysis are used for accurate calibration and certification processes. This practice is widely recognized in the fields of environmental monitoring, occupational health, and laboratory instrumentation.
Key Topics
- Dynamic Ozone Generation: Since ozone cannot be stored as a reference material, this standard describes procedures to dynamically generate ozone concentrations using ultraviolet (UV) light.
- Ultraviolet Photometry: Ozone concentrations are determined through the measurement of UV light absorption at 253.7 nm, based on the Beer-Lambert law.
- Primary and Transfer Standards: The practice provides means to designate dynamically-generated ozone streams as primary standards, and to certify transfer standards for field or laboratory calibration.
- System Requirements: The calibration system requires an ozone generator, UV photometer, zero air source, flow meters, temperature and pressure indicators, and nonreactive materials for all gas contact surfaces.
- Calibration Process: Standardized operating procedures guide the pre-conditioning, operation, and verification of ozone photometers, including system checks, linearity tests, and corrections for ozone losses.
- Safety Considerations: The standard addresses ozone's toxic properties and includes guidance on appropriate safety, environmental, and health practices.
Applications
This ASTM practice is designed for:
- Laboratory Calibration: Ensures high-accuracy calibration of ambient, indoor, and workplace ozone monitors in controlled settings.
- Transfer Standard Certification: Provides traceable certification for secondary standards used to calibrate ozone analyzers in the field or laboratory.
- Environmental Monitoring: Supports air quality monitoring efforts where precise measurement of ozone is critical, such as regulatory compliance and research stations.
- Workplace Exposure Assessment: Facilitates accurate monitoring in occupational environments where ozone exposure needs to be controlled and assessed.
- Instrument Verification: Laboratories and equipment manufacturers use this standard to validate and verify the performance and accuracy of ozone monitoring equipment.
Note: This standard is not intended for routine calibration of ozone monitors at remote locations. For such applications, consult ASTM D5011.
Related Standards
ASTM D5110-22a connects with several other key standards supporting atmospheric pollutant measurement and analyzer calibration, including:
- ASTM D5011 - Practices for Calibration of Ozone Monitors Using Transfer Standards
- ASTM D1356 - Terminology Relating to Sampling and Analysis of Atmospheres
- ASTM D3195 - Practice for Rotameter Calibration
- ASTM D3249 - Practice for General Ambient Air Analyzer Procedures
- ASTM D3631 - Test Methods for Measuring Surface Atmospheric Pressure
- ASTM E220 - Test Method for Calibration of Thermocouples By Comparison Techniques
- ASTM E644 - Test Methods for Testing Industrial Resistance Thermometers
Practical Value
Adopting ASTM D5110-22a assures traceable, repeatable, and documented calibration of ozone monitors, which is vital for air quality measurement, regulatory reporting, and workplace safety assessments. By specifying rigorous UV photometric techniques and quality control procedures, this practice supports data integrity and comparability across different laboratories and field sites, meeting international standards recognition and regulatory expectations.
Keywords: ozone calibration, UV photometry, ozone monitor certification, transfer standards, air quality, laboratory calibration, primary ozone standard, ASTM D5110-22a
Buy Documents
ASTM D5110-22a - Standard Practice for Calibration of Ozone Monitors and Certification of Ozone Transfer Standards Using Ultraviolet Photometry
REDLINE ASTM D5110-22a - Standard Practice for Calibration of Ozone Monitors and Certification of Ozone Transfer Standards Using Ultraviolet Photometry
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D5110-22a is a standard published by ASTM International. Its full title is "Standard Practice for Calibration of Ozone Monitors and Certification of Ozone Transfer Standards Using Ultraviolet Photometry". This standard covers: SIGNIFICANCE AND USE 5.1 The reactivity and instability of O3 preclude the storage of O3 concentration standards for any practical length of time, and precludes direct certification of O3 concentrations as Standard Reference Materials (SRMs). Moreover, there is no available SRM that can be readily and directly adapted to the generation of O3 standards analogous to permeation devices and standard gas cylinders for sulfur dioxide and nitrogen oxides. Dynamic generation of O3 concentrations is relatively easy with a source of ultraviolet (UV) radiation. However, accurately certifying an O3 concentration as a primary standard requires assay of the concentration by a comprehensively specified analytical procedure, which must be performed every time a standard is needed (10). 5.2 This practice is not designed for the routine calibration of O3 monitors at remote locations (see Practices D5011). SCOPE 1.1 This practice covers a means for calibrating ambient, workplace, or indoor ozone monitors, and for certifying transfer standards to be used for that purpose. 1.2 This practice describes means by which dynamic streams of ozone in air can be designated as primary ozone standards. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 8 for specific precautionary statements. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 The reactivity and instability of O3 preclude the storage of O3 concentration standards for any practical length of time, and precludes direct certification of O3 concentrations as Standard Reference Materials (SRMs). Moreover, there is no available SRM that can be readily and directly adapted to the generation of O3 standards analogous to permeation devices and standard gas cylinders for sulfur dioxide and nitrogen oxides. Dynamic generation of O3 concentrations is relatively easy with a source of ultraviolet (UV) radiation. However, accurately certifying an O3 concentration as a primary standard requires assay of the concentration by a comprehensively specified analytical procedure, which must be performed every time a standard is needed (10). 5.2 This practice is not designed for the routine calibration of O3 monitors at remote locations (see Practices D5011). SCOPE 1.1 This practice covers a means for calibrating ambient, workplace, or indoor ozone monitors, and for certifying transfer standards to be used for that purpose. 1.2 This practice describes means by which dynamic streams of ozone in air can be designated as primary ozone standards. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 8 for specific precautionary statements. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D5110-22a is classified under the following ICS (International Classification for Standards) categories: 13.040.01 - Air quality in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5110-22a has the following relationships with other standards: It is inter standard links to ASTM D1356-20a, ASTM D1356-20, ASTM E644-11(2019), ASTM D3249-95(2019), ASTM D5011-17, ASTM D3631-99(2017), ASTM D1356-15a, ASTM D1356-15, ASTM D1356-14b, ASTM D1356-14a, ASTM D1356-14, ASTM E220-13, ASTM D3631-99(2011), ASTM D3249-95(2011), ASTM E644-11. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5110-22a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5110 −22a
Standard Practice for
Calibration of Ozone Monitors and Certification of Ozone
Transfer Standards Using Ultraviolet Photometry
This standard is issued under the fixed designation D5110; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D5011Practices for Calibration of Ozone Monitors Using
Transfer Standards
1.1 This practice covers a means for calibrating ambient,
E220Test Method for Calibration of Thermocouples By
workplace, or indoor ozone monitors, and for certifying
Comparison Techniques
transfer standards to be used for that purpose.
E591Practice for Safety and Health Requirements Relating
1.2 This practice describes means by which dynamic
to Occupational Exposure to Ozone (Withdrawn 1990)
streams of ozone in air can be designated as primary ozone
E644Test Methods for Testing Industrial Resistance Ther-
standards.
mometers
1.3 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this 3. Terminology
standard.
3.1 Definitions—For definitions of terms used in this
1.4 This standard does not purport to address all of the
practice, refer to Terminology D1356.
safety concerns, if any, associated with its use. It is the
3.2 Definitions of Terms Specific to This Standard:
responsibility of the user of this standard to establish appro- 3.2.1 primary standard, n—a standard directly defined and
priate safety, health, and environmental practices and deter-
established by some authority, against which all secondary
mine the applicability of regulatory limitations prior to use. standards are compared.
See Section 8 for specific precautionary statements.
3.2.2 secondary standard, n—a standard used as a means of
1.5 This international standard was developed in accor-
comparison, but checked against a primary standard.
dance with internationally recognized principles on standard-
3.2.3 standard, n—an accepted reference sample or device
ization established in the Decision on Principles for the
used for establishing measurement of a physical quantity.
Development of International Standards, Guides and Recom-
3.2.4 transfer standard, n—a type of secondary standard. It
mendations issued by the World Trade Organization Technical
is a transportable device or apparatus that, together with
Barriers to Trade (TBT) Committee.
operational procedures, is capable of reproducing pollutant
2. Referenced Documents
concentration or producing acceptable assays of pollutant
concentrations.
2.1 ASTM Standards:
D1356Terminology Relating to Sampling and Analysis of 3.2.5 zero air, n—purified air that does not contain ozone,
Atmospheres
and does not contain any other component that may interfere
D3195Practice for Rotameter Calibration with the measurement (see 7.1).
D3249Practice for General Ambient Air Analyzer Proce-
dures 4. Summary of Practice
D3631Test Methods for Measuring Surface Atmospheric
4.1 Thispracticeisbasedonthephotometricassayofozone
Pressure
(O ) concentrations in a dynamic flow system. The concentra-
tion of O in an absorption cell is determined from a measure-
ment of the amount of 253.7 nm light absorbed by the sample.
This practice is under the jurisdiction of ASTM Committee D22 on Air
Qualityand is the direct responsibility of Subcommittee D22.03 on Ambient
This determination requires knowledge of (1) the absorption
Atmospheres and Source Emissions.
coefficient of O at 253.7 nm, (2) the optical path length
Current edition approved Sept. 1, 2022. Published September 2022. Originally
through the sample, (3) the transmittance of the sample at a
approved in 1990. Last previous edition approved in 2022 as D5110–22. DOI:
10.1520/D5110-22A.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on The last approved version of this historical standard is referenced on
the ASTM website. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5110 − 22a
wavelength of 253.7 nm, and (4) the temperature and pressure source of zero air, and other components as necessary. The
of the sample. The transmittance is defined as the ratio: configuration must provide a stable O concentration at the
systemoutputandallowthephotometertoassayaccuratelythe
I/I
o
output concentration to the precision specified for the photom-
where:
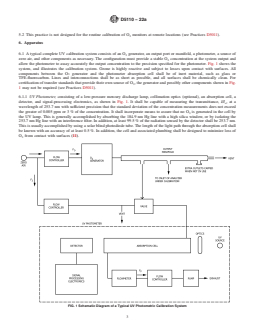
eter. Fig. 1 shows the system, and illustrates the calibration
I = the intensity of light that passes through the cell and is
system. Ozone is highly reactive and subject to losses upon
sensed by the detector when the cell contains an O
contact with surfaces.All components between the O genera-
sample, and
tor and the photometer absorption cell shall be of inert
I = the intensity of light that passes through the cell and is
o
material, such as glass or TFE-fluorocarbon. Lines and inter-
sensed by the detector when the cell contains zero air.
connections shall be as short as possible, and all surfaces shall
Itisassumedthatallconditionsofthesystem,exceptforthe be chemically clean. For certification of transfer standards that
contents of the absorption cell, are identical during measure- provide their own source of O , the generator and possibly
ments of I and I . The quantities defined above are related by
other components shown in Fig. 1 may not be required (see
o
the Beer-Lambert absorption law: Practices D5011).
2acd
6.1.1 UV Photometer, consisting of a low-pressure mercury
Transmittance 5 I/I 5 e (1)
o
discharge lamp, collimation optics (optional), an absorption
where:
cell, a detector, and signal-processing electronics, as shown in
a = absorption coefficient of O at 253.7 nm,
Fig. 1. It shall be capable of measuring the transmittance, I/I ,
o
−6 −1 −1
(304.39 60.94)×10 ppm cm at 0°C and 101.3
at a wavelength of 253.7 nm with sufficient precision that the
kPa (1 atm) (1-9),
standarddeviationoftheconcentrationmeasurementsdoesnot
c =O concentration, ppm, and
exceedthegreaterof0.005ppmor3%oftheconcentration.It
d = optical path length, cm.
shallincorporatemeanstoassurethatnoO isgeneratedinthe
4.1.1 Inpractice,astableO generator(see6.1.4)isusedto
cell by the UV lamp. This is generally accomplished by
produce O concentrations over the required range. Each O
3 3 absorbing the 184.9 nm Hg line with a high silica window, or
concentration is determined from the measurement of the
byisolatingthe253.7nmHglinewithaninterferencefilter.In
transmittanceofthesampleat253.7nm,andiscalculatedfrom
addition,atleast99.5%oftheradiationsensedbythedetector
the equation:
shall be 253.7 nm. This is usually accomplished by using a
solar blind photodiode tube. The length of the light path
I
2ln
throughtheabsorptioncellshallbeknownwithanaccuracyof
I
o
c 5 (2)
at least 0.5%. In addition, the cell and associated plumbing
~ad!
The calculated O concentrations must be corrected for O
shall be designed to minimize loss of O from contact with
3 3
losses, which may occur in the photometer, and for the tem-
surfaces (11).
perature and pressure of the sample.
6.1.2 Air Flow Controller,capableofregulatingairflowsas
necessarytomeettheoutputstabilityandphotometerprecision
5. Significance and Use
requirements.
5.1 The reactivity and instability of O preclude the storage
6.1.3 Flowmeters, calibrated in accordance with Practice
of O concentration standards for any practical length of time,
D3195.
and precludes direct certification of O concentrations as
6.1.4 Ozone Generator, capable of generating stable levels
Standard Reference Materials (SRMs). Moreover, there is no
of O over the required concentration range. It shall be stable
available SRM that can be readily and directly adapted to the 3
over short periods to facilitate the sequential photometric
generation of O standards analogous to permeation devices
measurement of I and I , and to allow for stability of the
and standard gas cylinders for sulfur dioxide and nitrogen
o
monitor or transfer standard connected to the output manifold.
oxides. Dynamic generation of O concentrations is relatively
Conventional UV-photolytic type generators may be adequate,
easy with a source of ultraviolet (UV) radiation. However,
but shall have line voltage and temperature regulation.
accuratelycertifyinganO concentrationasaprimarystandard
requires assay of the concentration by a comprehensively
6.1.5 Output Manifold, constructed of glass, TFE-
specifiedanalyticalprocedure,whichmustbeperformedevery
fluorocarbon, or other nonreactive material. It shall be of
time a standard is needed (10).
sufficient diameter to ensure a negligible pressure drop at the
photometer connection and other output ports. The output
5.2 This practice is not designed for the routine calibration
manifoldservesthefunctionofprovidinganinterfacebetween
of O monitors at remote locations (see Practices D5011).
the calibration system and other devices and systems that
utilize the output O concentrations. It shall have one or more
6. Apparatus
portsforconnectionoftheexternalinstrumentsorsystems,and
6.1 AtypicalcompleteUVcalibrationsystemconsistsofan
shallbesuchthatallportsprovidethesameO concentrations.
O generator, an output port or manifold, a photometer, a
The vent, which exhausts excess gas flow from the system and
insures that the manifold outlet ports are kept at atmospheric
4 pressure for all flowrates, shall be large enough to avoid
The boldface numbers in parentheses refer to the references listed at the end of
this practice. appreciable pressure drop, and shall be located downstream of
D5110 − 22a
FIG. 1 Schematic Diagram of a Typical UV Photometric Calibration System
the output ports to ensure that no ambient air enters the 6.1.8 BarometerorPressureIndicator,accurateto250Pa(2
manifold due to eddy currents, back diffusion, and so forth. torr). The barometer or pressure indicator is used to measure
6.1.6 Three-Way Valve, constructed ofTFE-fluorocarbon, to
the pressure of the gas in the cell to calculate a pressure
switch the flow through the absorption cell from zero air (for
correction. Most photometer cells operate at atmospheric
the I measurement) to manifold gas (for the I measurement).
o pressure. If there are no restrictions between the cell and the
6.1.7 Temperature Indicator, accurate to 61°C. This indi-
output manifold, the cell pressure should be very nearly the
cator is needed to measure the temperature of the gas in the
same as the local barometric pressure. A certified local baro-
photometric cell to calculate a temperature correction. In most
metric pressure reading can then be used for the pressure
photometers, particularly those whose cell is enclosed inside a
correction. If the cell pressure is different from the local
case or housing with other electrical or electronic components,
barometric pressure, some means of accurately measuring the
the cell operates at a temperature somewhat above ambient
cell pressure (manometer, pressure gauge, or pressure trans-
room temperature. Therefore, it is important to measure the
ducer) is required. This device shall be calibrated against a
temperature of the gas inside the cell, and not room tempera-
suitable pressure standard, in accordance with Test Methods
ture. A small thermocouple or thermistor, connected to an
D3631.
external readout device, may be attached to the cell wall or
6.1.9 Output Indicating Device, such as continuous strip
inserted through the cell wall to measure internal cell tempera-
chart recorder or digital volt meter.
ture. The point of temperature sensing shall be representative
of the average cell temperature. The temperature sensing
6.1.9.1 If a recorder is used, it shall have the following
deviceshallbecalibratedagainstaNISTcertifiedthermometer specifications:
initially, and at periodic intervals, subject to the laboratory
Accuracy ±0.25 % of span
quality control checks (12). See Test Method E220 or Test Chart width no less than 150 mm
Time for full-scale travel 1 s
Methods E644 for calibration procedures.
D5110 − 22a
6.1.9.2 If a digital volt meter is used, it shall have an and specifically not used for ambient monitoring or as a
accuracy of 60.25% of range. transfer standard. The reason for this requirement is that the
photometer must be intrinsically accurate and not exposed to
7. Reagents and Materials
outsideeffects.Ifitisusedforotherpurposes,itwilleventually
become dirty and will be prone to O losses and will give
7.1 Zero Air—Free of O and any substance that by itself or 3
erraticreadings.Reservingthephotometerforuseasaprimary
whose decomposition products from the ozonizer might react
standard, where only clean, dry, filtered gas passes through the
with O , absorb 255.7 nm light, or undergo photolysis (for
cell, will minimize loss of accuracy. A photometer used as a
example NO, NO , ethylene, and particulate matter). The air
transfer standard will be subjected to environmental
shall be purified to remove such substances. Dirty air shall be
conditions, which may have an effect on its output.
precleaned to remove particulate matter, oil mist, liquid water,
and so forth.
9.3 Photometer Verifications—Since the accuracy of the
7.1.1 The following describes a system that has been used
calibrationstandardsobtainedbythispracticedependsentirely
successfully: The air is dried with a membrane type dryer,
ontheaccuracyofthephotometer,itisimportanttoensurethat
followed by a column of indicating silica gel. The air is
the photometer is operating properly and accurately.
irradiated with a UV lamp to generate O , to convert NO to
9.3.1 A well designed and properly built photometer is a
NO andthenpassedthroughacolumnofactivatedcharcoal(6
precision instrument; once shown to operate adequately, it is
to 14 mesh) to remove NO,O , hydrocarbons, and various
2 3
likely to continue to do so for some time, particularly if it is
other substances, a column of molecular sieve (6 to 16 mesh,
held stationary and used intermittently under laboratory con-
type 4A), and a final particulate filter (2 µm) to remove
ditions.Therefore,theperformancechecksmaynotnecessarily
particulate matter. (Warning—An important requirement in
have to be conducted every time the photometer is used. The
photometer operation is that the zero air supplied to the
actual frequency of the checks is a trade-off between confi-
photometerduringtheI measurementisfromthesamesource
o
denceinthephotometerperformanceandthecostandeffortto
as that used for the generation of O .The impurities present in
conductthechecks.Thisisamatterofjudgment,subjecttothe
zero air from different sources can significantly affect the
laboratory quality control checks (12). One reasonable ap-
transmittance of an air sample. This requirement presents no
proach is to perform the checks very frequently with a new
problemiftheconfigurationshowninFig.1isused.However,
photometer, keeping a chronological record of each perfor-
there may be a problem in certifying O generator transfer
mance check, using the QA control chart, and to reduce the
standards that have their own source of zero air or O (see
frequencyasexperiencedictates.Evenwheretherecordshows
Practices D5011). The zero air produced in 7.1.1 is very dry.
excellent stability, the checks shall be performed at some
The O response of some measurement methods (for example,
minimum frequency (for example, once every 2 or 3 weeks)
ethylene chemiluminescence, KI bubblers) is affected by
because the possibility of malfunction is always present. A
changes in relative humidity.This dependence should be taken
regular schedule of checks will avoid the risk of losing long
into account when reporting results.)
periods of data due to photometer malfunction.
9.4 Instrument Manual—If
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5110 − 22 D5110 − 22a
Standard Practice for
Calibration of Ozone Monitors and Certification of Ozone
Transfer Standards Using Ultraviolet Photometry
This standard is issued under the fixed designation D5110; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers a means for calibrating ambient, workplace, or indoor ozone monitors, and for certifying transfer
standards to be used for that purpose.
1.2 This practice describes means by which dynamic streams of ozone in air can be designated as primary ozone standards.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. See Section 8 for specific precautionary statements.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1356 Terminology Relating to Sampling and Analysis of Atmospheres
D3195 Practice for Rotameter Calibration
D3249 Practice for General Ambient Air Analyzer Procedures
D3631 Test Methods for Measuring Surface Atmospheric Pressure
D5011 Practices for Calibration of Ozone Monitors Using Transfer Standards
E220 Test Method for Calibration of Thermocouples By Comparison Techniques
E591 Practice for Safety and Health Requirements Relating to Occupational Exposure to Ozone (Withdrawn 1990)
E644 Test Methods for Testing Industrial Resistance Thermometers
3. Terminology
3.1 Definitions—For definitions of terms used in this practice, refer to Terminology D1356.
3.2 Definitions of Terms Specific to This Standard:
This practice is under the jurisdiction of ASTM Committee D22 on Air Quality and is the direct responsibility of Subcommittee D22.03 on Ambient Atmospheres and
Source Emissions.
Current edition approved May 1, 2022Sept. 1, 2022. Published September 2022. Originally approved in 1990. Last previous edition approved in 20172022 as D5110 – 98
(2017).D5110 – 22. DOI: 10.1520/D5110-22.10.1520/D5110-22A.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5110 − 22a
3.2.1 primary standard, n—a standard directly defined and established by some authority, against which all secondary standards
are compared.
3.2.2 secondary standard, n—a standard used as a means of comparison, but checked against a primary standard.
3.2.3 standard, n—an accepted reference sample or device used for establishing measurement of a physical quantity.
3.2.4 transfer standard, n—a type of secondary standard. It is a transportable device or apparatus that, together with operational
procedures, is capable of reproducing pollutant concentration or producing acceptable assays of pollutant concentrations.
3.2.5 zero air, n—purified air that does not contain ozone, and does not contain any other component that may interfere with the
measurement (see 7.1).
4. Summary of Practice
4.1 This practice is based on the photometric assay of ozone (O ) concentrations in a dynamic flow system. The concentration of
O in an absorption cell is determined from a measurement of the amount of 253.7 nm light absorbed by the sample. This
determination requires knowledge of (1) the absorption coefficient of O at 253.7 nm, (2) the optical path length through the
sample, (3) the transmittance of the sample at a wavelength of 253.7 nm, and (4) the temperature and pressure of the sample. The
transmittance is defined as the ratio:
I/I
o
where:
I = the intensity of light that passes through the cell and is sensed by the detector when the cell contains an O sample, and
I = the intensity of light that passes through the cell and is sensed by the detector when the cell contains zero air.
o
It is assumed that all conditions of the system, except for the contents of the absorption cell, are identical during measurements
of I and I . The quantities defined above are related by the Beer-Lambert absorption law:
o
2acd
Transmittance 5 I/I 5 e (1)
o
where:
−6 −1 −1 4
a = absorption coefficient of O at 253.7 nm, (304.39 6 0.94) × 10 ppm cm at 0 °C and 101.3 kPa (1 atm) (1-9),
c = O concentration, ppm, and
d = optical path length, cm.
4.1.1 In practice, a stable O generator (see 6.1.4) is used to produce O concentrations over the required range. Each O
3 3 3
concentration is determined from the measurement of the transmittance of the sample at 253.7 nm, and is calculated from the
equation:
I
2ln
I
o
c 5 (2)
~ad!
The calculated O concentrations must be corrected for O losses, which may occur in the photometer, and for the temperature
3 3
and pressure of the sample.
5. Significance and Use
5.1 The reactivity and instability of O preclude the storage of O concentration standards for any practical length of time, and
3 3
precludes direct certification of O concentrations as Standard Reference Materials (SRMs). Moreover, there is no available SRM
that can be readily and directly adapted to the generation of O standards analogous to permeation devices and standard gas
cylinders for sulfur dioxide and nitrogen oxides. Dynamic generation of O concentrations is relatively easy with a source of
ultraviolet (UV) radiation. However, accurately certifying an O concentration as a primary standard requires assay of the
concentration by a comprehensively specified analytical procedure, which must be performed every time a standard is needed (10).
The boldface numbers in parentheses refer to the references listed at the end of this practice.
D5110 − 22a
5.2 This practice is not designed for the routine calibration of O monitors at remote locations (see Practices D5011).
6. Apparatus
6.1 A typical complete UV calibration system consists of an O generator, an output port or manifold, a photometer, a source of
zero air, and other components as necessary. The configuration must provide a stable O concentration at the system output and
allow the photometer to assay accurately the output concentration to the precision specified for the photometer. Fig. 1 shows the
system, and illustrates the calibration system. Ozone is highly reactive and subject to losses upon contact with surfaces. All
components between the O generator and the photometer absorption cell shall be of inert material, such as glass or
TFE-fluorocarbon. Lines and interconnections shall be as short as possible, and all surfaces shall be chemically clean. For
certification of transfer standards that provide their own source of O , the generator and possibly other components shown in Fig.
1 may not be required (see Practices D5011).
6.1.1 UV Photometer, consisting of a low-pressure mercury discharge lamp, collimation optics (optional), an absorption cell, a
detector, and signal-processing electronics, as shown in Fig. 1. It shall be capable of measuring the transmittance, I/I , at a
o
wavelength of 253.7 nm with sufficient precision that the standard deviation of the concentration measurements does not exceed
the greater of 0.005 ppm or 3 % of the concentration. It shall incorporate means to assure that no O is generated in the cell by
the UV lamp. This is generally accomplished by absorbing the 184.9 nm Hg line with a high silica window, or by isolating the
253.7 nm Hg line with an interference filter. In addition, at least 99.5 % of the radiation sensed by the detector shall be 253.7 nm.
This is usually accomplished by using a solar blind photodiode tube. The length of the light path through the absorption cell shall
be known with an accuracy of at least 0.5 %. In addition, the cell and associated plumbing shall be designed to minimize loss of
O from contact with surfaces (11).
FIG. 1 Schematic Diagram of a Typical UV Photometric Calibration System
D5110 − 22a
6.1.2 Air Flow Controller, capable of regulating air flows as necessary to meet the output stability and photometer precision
requirements.
6.1.3 Flowmeters, calibrated in accordance with Practice D3195.
6.1.4 Ozone Generator, capable of generating stable levels of O over the required concentration range. It shall be stable over short
periods to facilitate the sequential photometric measurement of I and I , and to allow for stability of the monitor or transfer standard
o
connected to the output manifold. Conventional UV-photolytic type generators may be adequate, but shall have line voltage and
temperature regulation.
6.1.5 Output Manifold, constructed of glass, TFE-fluorocarbon, or other nonreactive material. It shall be of sufficient diameter to
ensure a negligible pressure drop at the photometer connection and other output ports. The output manifold serves the function of
providing an interface between the calibration system and other devices and systems that utilize the output O concentrations. It
shall have one or more ports for connection of the external instruments or systems, and shall be such that all ports provide the same
O concentrations. The vent, which exhausts excess gas flow from the system and insures that the manifold outlet ports are kept
at atmospheric pressure for all flowrates, shall be large enough to avoid appreciable pressure drop, and shall be located downstream
of the output ports to ensure that no ambient air enters the manifold due to eddy currents, back diffusion, and so forth.
6.1.6 Three-Way Valve, constructed of TFE-fluorocarbon, to switch the flow through the absorption cell from zero air (for the I
o
measurement) to manifold gas (for the I measurement).
6.1.7 Temperature Indicator, accurate to 61°C. This indicator is needed to measure the temperature of the gas in the photometric
cell to calculate a temperature correction. In most photometers, particularly those whose cell is enclosed inside a case or housing
with other electrical or electronic components, the cell operates at a temperature somewhat above ambient room temperature.
Therefore, it is important to measure the temperature of the gas inside the cell, and not room temperature. A small thermocouple
or thermistor, connected to an external readout device, may be attached to the cell wall or inserted through the cell wall to measure
internal cell temperature. The point of temperature sensing shall be representative of the average cell temperature. The temperature
sensing device shall be calibrated against a NIST certified thermometer initially, and at periodic intervals, subject to the laboratory
quality control checks (12). See Test Method E220 or Test Methods E644 for calibration procedures.
6.1.8 Barometer or Pressure Indicator, accurate to 250 Pa (2 torr). The barometer or pressure indicator is used to measure the
pressure of the gas in the cell to calculate a pressure correction. Most photometer cells operate at atmospheric pressure. If there
are no restrictions between the cell and the output manifold, the cell pressure should be very nearly the same as the local barometric
pressure. A certified local barometric pressure reading can then be used for the pressure correction. If the cell pressure is different
from the local barometric pressure, some means of accurately measuring the cell pressure (manometer, pressure gauge, or pressure
transducer) is required. This device shall be calibrated against a suitable pressure standard, in accordance with Test Methods
D3631.
6.1.9 Output Indicating Device, such as continuous strip chart recorder or digital volt meter.
6.1.9.1 If a recorder is used, it shall have the following specifications:
Accuracy ±0.25 % of span
Chart width no less than 150 mm
Time for full-scale travel 1 s
6.1.9.2 If a digital volt meter is used, it shall have an accuracy of 60.25 % of range.
7. Reagents and Materials
7.1 Zero Air—Free of O and any substance that by itself or whose decomposition products from the ozonizer might react with
O , absorb 255.7 nm light, or undergo photolysis (for example NO, NO , ethylene, and particulate matter). The air shall be purified
3 2
to remove such substances. Dirty air shall be precleaned to remove particulate matter, oil mist, liquid water, and so forth.
D5110 − 22a
7.1.1 The following describes a system that has been used successfully: The air is dried with a membrane type dryer, followed
by a column of indicating silica gel. The air is irradiated with a UV lamp to generate O , to convert NO to NO and then passed
3 2
through a column of activated charcoal (6 to 14 mesh) to remove NO , O , hydrocarbons, and various other substances, a column
2 3
of molecular sieve (6 to 16 mesh, type 4A), and a final particulate filter (2 μm) to remove particulate matter. (Warning—An
important requirement in photometer operation is that the zero air supplied to the photometer during the I measurement is from
o
the same source as that used for the generation of O . The impurities present in zero air from different sources can significantly
affect the transmittance of an air sample. This requirement presents no problem if the configuration shown in Fig. 1 is used.
However, there may be a problem in certifying O generator transfer standards that have their own source of zero air or O (see
3 3
Practices D5011). The zero air produced in 7.1.1 is very dry. The O response of some measurement methods (for example,
ethylene chemiluminescence, KI bubblers) is affected by changes in relative humidity. This dependence should be taken into
account when reporting results.)
8. Hazards
8.1 Safety Hazards—See Practice D3249 for safety precautions on the use of monitors and electronic equipment.
8.1.1 Ozone is a toxic gaseous substance. See Practice E591 for biological effects, and for safety and health requirements.
8.1.2 The manifold vents and photometer and monitor exhausts must be vented to remove exhaust gases from the workplace. Care
shall be taken to avoid a back pressure in the cell and manifold, and in the monitor or transfer standard being calibrated.
9. Preparation of Apparatus
9.1 Conditioning—The system shall be conditioned by flowing O at maximum concentration for 10 to 30 min initially, and after
periods of time while the system was not used, prior to reuse.
9.2 General Operation—A photometer used as a primary standard shall be dedicated exclusively to calibration service and
specifically not used for ambient monitoring or as a transfer standard. The reason for this requirement is that the photometer must
be intrinsically accurate and not exposed to outside effects. If it is used for other purposes, it will eventually become dirty and will
be prone to O losses and will give erratic readings. Reserving the photometer for use as a primary standard, where only clean,
dry, filtered gas passes through the cell, will minimize loss of accuracy. A photometer used as a transfer standard will be subjected
to environmental conditions, which may have an effect on its output.
9.3 Photometer Verifications—Since the accuracy of the calibration standards obtained by this practice depends entirely on the
accuracy of the photometer, it is important to ensure that the photometer is operating properly and accurately.
9.3.1 A well designed and properly built photometer is a precision instrument; once shown to operate adequately, it is likely to
continue to do so for some time, particularly if it is held stationary and used intermittently under laboratory conditions. Therefore,
the performance checks may not necessarily have to be conducted every time the p
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...