ASTM E1173-23
(Practice)Standard Practice for Evaluation of Preoperative, Precatheterization, or Preinjection Skin Preparations
Standard Practice for Evaluation of Preoperative, Precatheterization, or Preinjection Skin Preparations
SIGNIFICANCE AND USE
5.1 These procedures should be used to test topical antimicrobial-containing preparations that are intended to be fast-acting in reducing significantly the number of microorganisms on intact skin immediately and, for preoperative and vascular precatheterization preparations, maintenance of some degree of reduction for an extended time.
SCOPE
1.1 The practice is designed to measure the reduction of the microflora of the skin.
1.2 A knowledge of microbiological techniques is required for these procedures.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Exception—In this practice, metric units are used for all applications except for linear measure, in which case inches are used, and metric units follow in parentheses.
1.4 Performance of this procedure requires a knowledge of regulations pertaining to the protection of human subjects (1).2
Note 1: Importantly, it must be noted that the FDA currently does not accept data resulting from procedures in this Method for testing products for approval as Vascular Precatheterization Skin Preparations.
1.5 This standard does not purport to address all of the safety problems, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2023
- Technical Committee
- E35 - Pesticides, Antimicrobials, and Alternative Control Agents
- Drafting Committee
- E35.15 - Antimicrobial Agents
Relations
- Effective Date
- 01-Nov-2019
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-May-2010
- Effective Date
- 01-Oct-2009
- Effective Date
- 01-Apr-2008
- Effective Date
- 10-May-2002
- Effective Date
- 01-Jan-1997
Overview
ASTM E1173-23: Standard Practice for Evaluation of Preoperative, Precatheterization, or Preinjection Skin Preparations provides a uniform method for assessing the effectiveness of topical antimicrobial skin preparations. These preparations are intended to rapidly and significantly reduce the number of microorganisms on intact skin-immediately, and, in the case of preoperative and vascular precatheterization products, over extended durations. The standard outlines a practice for measuring the reduction of skin microflora using microbiological techniques, ensuring consistency in the evaluation of antimicrobial efficacy for products used prior to surgical procedures, catheterizations, or injections.
Key Topics
Purpose and Scope
- Measures reduction of skin microflora following application of antimicrobial skin preparations
- Applies to preparations used before surgery, vascular catheterization, or injection
- Requires familiarity with microbiological techniques and regulatory guidelines for human subject protection
Test Methodology
- Utilizes human volunteers with high baseline skin flora
- Involves standardized sample collection before and after product application
- Employs neutralizing fluids, sample randomization, and control formulations to ensure test validity
- Uses the "cup scrub technique" for microbiological sampling
Sampling and Evaluation
- Designates specific moist and dry anatomical test sites (e.g., inguinal region, abdomen, clavicular and cubital regions)
- Defines sampling schedules according to intended clinical use (e.g., preoperative, precatheterization, preinjection)
- Recommends statistical analysis methods, including parametric t-tests or nonparametric models
Safety and Compliance
- Adheres to international standardization principles and considers applicable safety, health, and environmental practices
- Notes that the U.S. FDA may not accept data from this method for certain product approvals
Applications
ASTM E1173-23 is valuable for:
- Medical device and pharmaceutical manufacturers: Standardizes testing for antimicrobial efficacy of skin prep solutions, ensuring reliable product evaluation before regulatory submission or market release.
- Hospitals and clinical laboratories: Provides a protocol for periodic verification of skin preparation product performance in surgical and procedural settings.
- Research laboratories: Supports scientific studies on skin antiseptics, offering a reproducible method for microbiological analysis and statistical evaluation.
- Quality control: Assists in validating batches of antimicrobial skin preparations for compliance with industry best practices.
- Regulatory and compliance professionals: Ensures that testing methodologies align with recognized international standards, facilitating global acceptance and harmonization.
Related Standards
Several related ASTM standards are commonly referenced alongside ASTM E1173-23 for comprehensive antimicrobial and microbiological testing:
- ASTM E1054 – Practices for Evaluation of Inactivators of Antimicrobial Agents
- ASTM E1874 – Practice for Recovery of Microorganisms From Skin using the Cup Scrub Technique
- ASTM E2756 – Terminology Relating to Antimicrobial and Antiviral Agents
ASTM E1173-23 is developed under the jurisdiction of ASTM Committee E35 on Pesticides, Antimicrobials, and Alternative Control Agents, aligning with international standardization principles set by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Employing ASTM E1173-23 improves the credibility and comparability of skin preparation product evaluations, supporting broader adoption and regulatory confidence in antimicrobial efficacy testing protocols.
Buy Documents
ASTM E1173-23 - Standard Practice for Evaluation of Preoperative, Precatheterization, or Preinjection Skin Preparations
REDLINE ASTM E1173-23 - Standard Practice for Evaluation of Preoperative, Precatheterization, or Preinjection Skin Preparations
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E1173-23 is a standard published by ASTM International. Its full title is "Standard Practice for Evaluation of Preoperative, Precatheterization, or Preinjection Skin Preparations". This standard covers: SIGNIFICANCE AND USE 5.1 These procedures should be used to test topical antimicrobial-containing preparations that are intended to be fast-acting in reducing significantly the number of microorganisms on intact skin immediately and, for preoperative and vascular precatheterization preparations, maintenance of some degree of reduction for an extended time. SCOPE 1.1 The practice is designed to measure the reduction of the microflora of the skin. 1.2 A knowledge of microbiological techniques is required for these procedures. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Exception—In this practice, metric units are used for all applications except for linear measure, in which case inches are used, and metric units follow in parentheses. 1.4 Performance of this procedure requires a knowledge of regulations pertaining to the protection of human subjects (1).2 Note 1: Importantly, it must be noted that the FDA currently does not accept data resulting from procedures in this Method for testing products for approval as Vascular Precatheterization Skin Preparations. 1.5 This standard does not purport to address all of the safety problems, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 These procedures should be used to test topical antimicrobial-containing preparations that are intended to be fast-acting in reducing significantly the number of microorganisms on intact skin immediately and, for preoperative and vascular precatheterization preparations, maintenance of some degree of reduction for an extended time. SCOPE 1.1 The practice is designed to measure the reduction of the microflora of the skin. 1.2 A knowledge of microbiological techniques is required for these procedures. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Exception—In this practice, metric units are used for all applications except for linear measure, in which case inches are used, and metric units follow in parentheses. 1.4 Performance of this procedure requires a knowledge of regulations pertaining to the protection of human subjects (1).2 Note 1: Importantly, it must be noted that the FDA currently does not accept data resulting from procedures in this Method for testing products for approval as Vascular Precatheterization Skin Preparations. 1.5 This standard does not purport to address all of the safety problems, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E1173-23 is classified under the following ICS (International Classification for Standards) categories: 11.080.01 - Sterilization and disinfection in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1173-23 has the following relationships with other standards: It is inter standard links to ASTM E2756-19, ASTM E2756-18, ASTM E2756-10, ASTM E1874-09, ASTM E1054-08, ASTM E1054-02, ASTM E1874-97. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1173-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1173 − 23
Standard Practice for
Evaluation of Preoperative, Precatheterization, or
Preinjection Skin Preparations
This standard is issued under the fixed designation E1173; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope E1054 Practices for Evaluation of Inactivators of Antimicro-
bial Agents
1.1 The practice is designed to measure the reduction of the
E1874 Practice for Recovery of Microorganisms From Skin
microflora of the skin.
using the Cup Scrub Technique
1.2 A knowledge of microbiological techniques is required
E2756 Terminology Relating to Antimicrobial and Antiviral
for these procedures.
Agents
1.3 The values stated in SI units are to be regarded as
3. Terminology
standard. No other units of measurement are included in this
standard.
3.1 Terms used in this standard are defined in E2756,
1.3.1 Exception—In this practice, metric units are used for Standard Terminology Relating to Antimicrobial and Antiviral
all applications except for linear measure, in which case inches
Agents. Others defined below are specific to their use in this
are used, and metric units follow in parentheses. document.
1.4 Performance of this procedure requires a knowledge of
3.2 Definitions of Terms Specific to This Standard:
regulations pertaining to the protection of human subjects (1). 3.2.1 active ingredient, n—a substance added to a formula-
NOTE 1—Importantly, it must be noted that the FDA currently does not
tion specifically for the inhibition or inactivation of microor-
accept data resulting from procedures in this Method for testing products
ganisms.
for approval as Vascular Precatheterization Skin Preparations.
3.2.2 inguen, n—groin: the junctional region between the
1.5 This standard does not purport to address all of the
abdomen and thigh; pl. inguina.
safety problems, if any, associated with its use. It is the
3.2.3 inguinal crease, n—the discrete region of flexure
responsibility of the user of this standard to establish appro-
between the abdomen and the thigh.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
3.2.4 sampling fluid, n—a recovery fluid that contains a
1.6 This international standard was developed in accor-
neutralizer demonstrated to inactivate or quench the active
dance with internationally recognized principles on standard-
ingredient(s) in the test formulation and the positive and
ization established in the Decision on Principles for the
negative control formulations. See Practices E1054.
Development of International Standards, Guides and Recom-
3.2.5 test formulation, n—a formulation containing an ac-
mendations issued by the World Trade Organization Technical
tive ingredient(s).
Barriers to Trade (TBT) Committee.
4. Summary of Test Method
2. Referenced Documents
4.1 This practice is conducted on human subjects selected
2.1 ASTM Standards:
randomly from a group of volunteers who, after refraining
voluntarily from using topical and oral antimicrobials for at
least two weeks (14 days), exhibit acceptably high normal flora
This practice is under the jurisdiction of ASTM Committee E35 on Pesticides,
counts on the skin sites to be used in testing (see Section 8).
Antimicrobials, and Alternative Control Agents and is the direct responsibility of
Subcommittee E35.15 on Antimicrobial Agents.
4.2 The antimicrobial activity of preoperative, vascular
Current edition approved April 1, 2023. Published May 2023. Originally
precatheterization, or preinjection skin preparations and of
approved in 1987. Last previous edition approved in 2015 as E1173 – 15. DOI:
controls is measured by comparing microbial counts, obtained
10.1520/E1173-23.
at various time intervals after application of a formulation to
The boldface numbers in parentheses refer to the list of references at the end of
this standard.
skin sites to counts obtained from those same sites prior to
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
application of a formulation. Skin sites recommended for use
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
in testing are: (1) the inguinal region and the abdomen for
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. preoperative skin preparations; (2) the inguinal region, the
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1173 − 23
subclavian (clavicular) region, and/or the median cubital re- 7.3 Scrubbing Cups—Autoclavable cylinders, height ap-
gion of the arm for vascular precatheterization preparations; proximately 1 in (2.5 cm) and inside diameter of a size
and (3) the median cubital region of the arm for preinjection convenient to placement on the skin of the anatomical area to
skin preparations. be sampled. Useful diameters range from approximately 0.5 to
4.2.1 Preoperative Skin Preparation—Microbial samples 1.5 in (1.3 to 3.8 cm), depending on sites to be sampled.
are collected from the test sites a minimum of three (3) times
7.4 Rubber Policeman, TFE-fluorocarbon Scrubbers, or
after treatment application on both moist and dry skin sites.
other appropriate device—Can be fashioned in the laboratory
The recommended sample times are 30 s, 10 min, and 6 h
or purchased from most laboratory supply houses. Whichever
post-treatment, but other relevant times may be selected.
type is selected, it should be used throughout the course of
4.2.2 Vascular Precatheterization Preparation—Microbial
testing.
samples are collected from the test sites a minimum of three (3)
7.5 Testing Formulation, including directions for use.
times after treatment application on skin sites. The recom-
mended sample times are “immediate,” 12 h, and 24 h
7.6 Sterile Gauge Pads—Used to cover treated skin sites.
post-treatment, but other relevant times may be selected. 4
7.7 Sterile Dressings —Used to cover treated skin sites.
4.2.3 Preinjection Preparation—Microbial samples are col-
7.8 Sampling Fluid—Dissolve 0.4 g KH PO , 10.1 g
lected from the skin sites 30 s post-treatment. 2 4
Na HPO , and 1.0 g isooctylphenoxypolyethoxyethanol in 1 L
2 4
4.3 The fluid used for sampling the test sites must effec-
of distilled water. Inactivator(s) specific for the antimicrobial
tively quench (neutralize) the antimicrobial action of all
active(s) in the test and control formulations must be included
formulations tested. The effectiveness of the inactivator must
(See Practices E1054). Adjust to pH 7.8. Dispense in appro-
be demonstrated prior to initiation of product-testing, as
priate volumes and sterilize.
described in Practices E1054, and using in-vivo techniques
7.9 Dilution Fluid—Butterfield’s (2) phosphate-buffered
consistent with the cup-scrub technique (see Section 10).
water adjusted to pH 7.2, or other suitable diluent, which must
4.4 A positive control formulation having performance
contain antimicrobial inactivators specific for the test, positive
characteristics known to the laboratory (internal validity of
and negative control formulations (see Practices E1054).
test) and a negative control (test formulation without active
7.10 Plating Medium—Soybean-casein digest agar (3), with
ingredient or a physiological saline solution) must be tested in
or without antimicrobial inactivators.
parallel with the test formulation.
7.11 Sterile Template Material—Used to demarcate the skin
5. Significance and Use
sites; made from paper, plastic, or cloth, for example.
5.1 These procedures should be used to test topical
7.12 Surgical Skin Marker—Used to delineate mark the skin
antimicrobial-containing preparations that are intended to be
sites to be used in testing.
fast-acting in reducing significantly the number of microorgan-
NOTE 2—Because some markers contain crystal violet or other fluids
isms on intact skin immediately and, for preoperative and
that are inhibitory to many skin microflora, a marker should be proven
vascular precatheterization preparations, maintenance of some
non-antimicrobial prior to use in testing.
degree of reduction for an extended time.
8. Skin Sites to be Used in Testing
6. Apparatus
8.1 Preoperative Skin Preparations:
6.1 Colony Counter—Any of several types may be used; for
8.1.1 The skin sites selected for evaluation of the effective-
example, Quebec colony counters and similar devices, or
ness of preoperative skin preparations should include both
automated, computerized plater/counter systems.
moist and dry skin areas. Bacterial baseline populations should
6.2 Incubator—Any incubator that can maintain a tempera-
be at least 3.0 log /cm greater on moist skin sites than the
ture of 30° 6 2°C may be used. detection limit of the sampling procedure, and at least 2.0 log
/cm greater than the detection limit on dry skin sites. The
6.3 Sterilizer—Any steam sterilizer that can produce the
preferred moist-skin areas are the inguina, in which skin-to-
conditions of sterilization is acceptable.
skin contact results in a moist environment conducive to higher
6.4 Timer (stopwatch)—One that displays hours, minutes,
populations of microflora. The preferred dry-skin area is the
and seconds.
lower abdomen adjacent to the umbilicus. These areas are
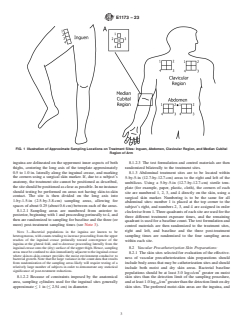
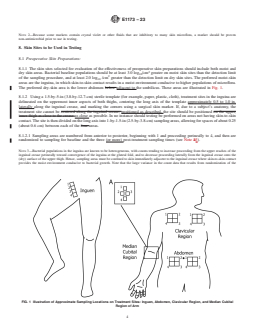
6.5 Examining Table—Any elevated surface, such as a illustrated in Fig. 1.
8.1.2 Using a 1.5-by-5-in (3.8-by-12.7-cm) sterile template
3-by-6-ft (0.9-by-1.8-meter) table with mattress or similar
padding to allow the subject to recline comfortably. (for example, paper, plastic, cloth), treatment sites in the
7. Reagents and Materials
The sole source of supply of the apparatus (TELFA non-adherent dressing, No.
7.1 Bacteriological Pipettes—10.0 and 2.2-mL or 1.1-mL
3279) known to the committee at this time is Kendall Co.; Hospital Products;
capacity, available from most laboratory supply houses.
Boston, MA 02101. This product is not sterile, but can be steam-sterilized prior to
use. If you are aware of alternative suppliers of appropriate dressings, please provide
7.2 Petri Dishes—100 mm by 15 mm for performing
this information to ASTM International Headquarters. Your comments will receive
standard plate counts, available from most laboratory supply
careful consideration at a meeting of the responsible technical committee, which
houses. you may attend.
E1173 − 23
FIG. 1 Illustration of Approximate Sampling Locations on Treatment Sites: Inguen, Abdomen, Clavicular Region, and Median Cubital
Region of Arm
inguina are delineated on the uppermost inner aspects of both 8.1.2.3 The test formulation and control materials are then
thighs, centering the long axis of the template approximately randomized bilaterally to the treatment sites.
0.5 to 1.0 in. laterally along the inguinal crease, and marking
8.1.3 Abdominal treatment sites are to be located within
the corners using a surgical skin marker. If, due to a subject’s
5-by-5-in (12.7-by-12.7-cm) areas to the right and left of the
anatomy, the treatment site cannot be positioned as described,
umbilicus. Using a 5-by-5-in (12.7-by-12.7-cm) sterile tem-
the site should be positioned as close as possible. In no instance
plate (for example, paper, plastic, cloth), the corners of each
should testing be performed on areas not having skin-to-skin
site are numbered 1, 2, 3, and 4 directly on the skin, using a
contact. The site is then divided on the long axis into
surgical skin marker. Numbering is to be the same for all
1-by-1.5-in (2.5-by-3.8-cm) sampling areas, allowing for
abdominal sites: number 1 is placed at the top corner to the
spaces of about 0.25 (about 0.6 cm) between each of the areas.
subject’s right, and numbers 2, 3, and 4 are assigned in order
8.1.2.1 Sampling areas are numbered from anterior to
clockwise from 1. Three quadrants of each site are used for the
posterior, beginning with 1 and proceeding perineally to 4, and
three different treatment exposure times, and the remaining
then are randomized to sampling for baseline and the three (or
quadrant is used for a baseline count. The test formulation and
more) post-treatment sampling times (see Note 3).
control materials are then randomized to the treatment sites,
right and left, and baseline and the three post-treatment
NOTE 3—Bacterial populations in the inguina are known to be
heterogeneous, with counts tending to increase proceeding from the upper sampling times are randomized to the four sampling areas
reaches of the inguinal crease perineally toward convergence of the
within each site.
inguina at the gluteal fold, and to decrease proceeding laterally from the
inguinal crease onto the (dry) surface of the upper thigh. Hence, sampling
8.2 Vascular Precatheterization Skin Preparations:
areas must be confined to skin immediately adjacent to the inguinal crease
8.2.1 The skin sites selected for evaluation of the effective-
where skin-to-skin contact provides the moist environment conducive to
ness of vascular precatheterization skin preparations should
bacterial growth. Note that the large variance in the count data that results
include body areas that may be catheterization sites and should
from randomization of the sampling areas likely will require testing of a
relatively large number of subjects in order to demonstrate any statistical
include both moist and dry skin areas. Bacterial baseline
significance of post-treatment reductions.
populations should be at least 3.0 log /cm greater on moist
8.1.2.2 Because of constraints imposed b
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1173 − 15 E1173 − 23
Standard Test Method Practice for
Evaluation of Preoperative, Precatheterization, or
Preinjection Skin Preparations
This standard is issued under the fixed designation E1173; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 The test method practice is designed to measure the reduction of the microflora of the skin.
1.2 A knowledge of microbiological techniques is required for these procedures.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Exception—In this test method, practice, metric units are used for all applications except for linear measure, in which case
inches are used, and metric units follow in parentheses.
1.4 Performance of this procedure requires a knowledge of regulations pertaining to the protection of human subjects (1).
NOTE 1—Importantly, it must be noted that the FDA currently does not accept data resulting from procedures in this Method for testing products for
approval as Vascular Precatheterization Skin Preparations.
1.5 This standard does not purport to address all of the safety problems, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E1054 Practices for Evaluation of Inactivators of Antimicrobial Agents
E1874 Practice for Recovery of Microorganisms From Skin using the Cup Scrub Technique
E2756 Terminology Relating to Antimicrobial and Antiviral Agents
This test method practice is under the jurisdiction of ASTM Committee E35 on Pesticides, Antimicrobials, and Alternative Control Agents and is the direct responsibility
of Subcommittee E35.15 on Antimicrobial Agents.
Current edition approved May 1, 2015April 1, 2023. Published July 2015May 2023. Originally approved in 1987. Last previous edition approved in 20092015 as
E1173 – 01E1173 – 15.(2009). DOI: 10.1520/E1173-15.10.1520/E1173-23.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1173 − 23
3. Terminology
3.1 Terms used in this standard are defined in E2756, Standard Terminology Relating to Antimicrobial and Antiviral Agents.
Others defined below are specific to their use in this document.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 active ingredient, n—a substance added to a formulation specifically for the inhibition or inactivation of microorganisms.
3.2.2 inguen, n—groin: the junctional region between the abdomen and thigh; pl. inguina.
3.2.3 inguinal crease—crease, n—the discrete region of flexure between the abdomen and the thigh.
3.2.4 sampling fluid—fluid, n—a recovery fluid that contains a neutralizer demonstrated to inactivate or quench the active
ingredient(s) in the test and reference formulation and the positive and negative control formulations. See Test Method Practices
E1054.
3.2.5 test formulation—formulation, n—a formulation containing an active ingredient(s).
4. Summary of Test Method
4.1 These test methods are This practice is conducted on human subjects selected randomly from a group of volunteers who, after
refraining voluntarily from using topical and oral antimicrobials for at least two weeks (14 days), exhibit acceptably high normal
flora counts on the skin sites to be used in testing (see Section 8).
4.2 The antimicrobial activity of preoperative, vascular precatheterization, or preinjection skin preparations and of controls is
measured by comparing microbial counts, obtained at various time intervals after application of a test formulation to skin sites,sites
to counts obtained from those same sites prior to application of the test a formulation. Skin sites recommended for use in testing
are: (1) the inguinal region and the abdomen for preoperative skin preparations; (2) the inguinal region, the subclavian (clavicular)
region, orand/or the median cubital region of the arm for vascular precatheterization preparations, or both; preparations; and (3)
the median cubital region of the arm for preinjection skin preparations.
4.2.1 Preoperative Skin Preparation—Microbial samples are collected from the test sites a minimum of three (3) times after
treatment application on both moist and dry skin sites. The recommended sample times are 30 s, 10 min, 30 min, and 6 h
post-treatment, but other relevant times may be selected.
4.2.2 Vascular Precatheterization Preparation—Microbial samples are collected from the test sites a minimum of three (3) times
after treatment application on both moist and dry skin sites. The recommended sample times are “immediate,” 12 h, and 24 h
post-treatment, but other relevant times may be selected. The immediate sample may be 30 s to 10 min, depending on the test
material evaluated.
4.2.3 Preinjection Preparation—A microbial sample is Microbial samples are collected from the test siteskin sites 30 s
post-treatment.
4.3 The fluid used for sampling the test sites must effectively quench (neutralize) the antimicrobial action of all formulations
tested. The effectiveness of the inactivator must be demonstrated prior to initiation of product-testing, as described in Test Method
Practices E1054, and using in-vivo techniques consistent with the cup-scrub technique (see Section 10).
4.4 To ensure the internal validity of the test, a reference control formulation having performance characteristics known to the
laboratory should A positive control formulation having performance characteristics known to the laboratory (internal validity of
test) and a negative control (test formulation without active ingredient or a physiological saline solution) must be tested in parallel
with the test formulation.
5. Significance and Use
5.1 These procedures should be used to test topical antimicrobial-containing preparations that are intended to be fast-acting in
E1173 − 23
reducing significantly the number of organismsmicroorganisms on intact skin immediately and, for preoperative and vascular
precatheterization preparations, to maintain reductions maintenance of some degree of reduction for an extended time.
6. Apparatus
6.1 Colony Counter—Any of several types may be used; for example, Quebec colony counters and similar devices, or automated,
computerized plater/counter systems.
6.2 Incubator—Any incubator that can maintain a temperature of 30° 6 2°C may be used.
6.3 Sterilizer—Any steam sterilizer that can produce the conditions of sterilization is acceptable.
6.4 Timer (stopwatch)—One that displays hours, minutes, and seconds.
6.5 Examining Table—Any elevated surface, such as a 3-by-6-ft (0.9-by-1.8-meter) table with mattress or similar padding to allow
the subject to recline.recline comfortably.
7. Reagents and Materials
7.1 Bacteriological Pipettes—10.0 and 2.2-mL or 1.1-mL capacity, available from most laboratory supply houses.
7.2 Petri Dishes—100 mm by 15 mm,mm for performing standard plate counts, available from most laboratory supply houses.
7.3 Scrubbing Cups—Autoclavable cylinders, height approximately 1 in (2.5 cm), cm) and inside diameter of a size convenient
to placement on the skin of the anatomical area to be sampled. Useful diameters range from approximately 0.5 to 1.5 in (1.3 to
3.8 cm), depending on sites to be sampled.
7.4 Rubber Policeman, TFE-fluorocarbon Scrubbers, or other appropriate advice—device—Can be fashioned in the laboratory or
purchased from most laboratory supply houses. Whichever type is selected, it should be used throughout the course of testing.
7.5 Testing Formulation, including directions for use.
7.6 Sterile Gauge Pads—Used to cover treated skin sites.
7.7 Sterile Dressings —Used to cover treated skin sites.
7.8 Sampling Fluid—Dissolve 0.4 g KH PO , 10.1 g Na HPO , and 1.0 g isooctylphenoxypolyethoxyethanol in 1 L of distilled
2 4 2 4
water. Inactivator(s) specific for the antimicrobial active(s) in the test and reference control formulations must be included (See
Test Method Practices E1054). Adjust to pH 7.8. Dispense in appropriate volumes and sterilize.
7.9 Dilution Fluid—Butterfield’s (2) phosphate-buffered water adjusted to pH 7.2, or other suitable diluent, which must contain
antimicrobial inactivators specific for the test and referencetest, positive and negative control formulations (see Test Method
Practices E1054).
7.10 Plating Medium—Soybean-casein digest agar (3), with or without antimicrobial inactivators.
7.11 Sterile Template Material—Used to demarcate the skin sites; made from paper, plastic, or cloth, for example.
7.12 Surgical Skin Marker—Used to delineate mark the skin sites to be used in testing.
The sole source of supply of the apparatus (TELFA non-adherent dressing, No. 3279) known to the committee at this time is Kendall Co.; Hospital Products; Boston,
MA 02101. This product is not sterile, but can be steam-sterilized prior to use. If you are aware of alternative suppliers of appropriate dressings, please provide this information
to ASTM International Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee, which you may attend.
E1173 − 23
NOTE 2—Because some markers contain crystal violet or other fluids that are inhibitory to many skin microflora, a marker should be proven
non-antimicrobial prior to use in testing.
8. Skin Sites to be Used in Testing
8.1 Preoperative Skin Preparations:
8.1.1 The skin sites selected for evaluation of the effectiveness of preoperative skin preparations should include both moist and
dry skin areas. Bacterial baseline populations should be at least 3.0 log /cm greater on moist skin sites than the detection limit
of the sampling procedure, and at least 2.0 log /cm greater than the detection limit on dry skin sites. The preferred moist-skin
areas are the inguina, in which skin-to-skin contact results in a moist environment conducive to higher populations of microflora.
The preferred dry-skin area is the lower abdomen below adjacent to the umbilicus. These areas are illustrated in Fig. 1.
8.1.2 Using a 1.5-by-5-in (3.8-by-12.7-cm) sterile template (for example, paper, plastic, cloth), treatment sites in the inguina are
delineated on the uppermost inner aspects of both thighs, centering the long axis of the template approximately 0.5 to 1.0 in.
laterally along the inguinal crease, and marking the corners using a surgical skin marker. If, due to a subject’s anatomy, the
treatment site cannot be centered along the inguinal crease, positioned as described, the site should be positioned on the upper,
inner thigh as close to the crease as close as possible. In no instance should testing be performed on areas not having skin-to-skin
contact. The site is then divided on the long axis into 1-by-1.5-in (2.5-by-3.8-cm) sampling areas, allowing for spaces of about 0.25
(about 0.6 cm) between each of the four areas.
8.1.2.1 Sampling areas are numbered from anterior to posterior, beginning with 1 and proceeding perineally to 4, and then are
randomized to sampling for baseline and the three (or more) post-treatment sampling times (see Note 23).
NOTE 3—Bacterial populations in the inguina are known to be heterogeneous, with counts tending to increase proceeding from the upper reaches of the
inguinal crease perineally toward convergence of the inguina at the gluteal fold, and to decrease proceeding laterally from the inguinal crease onto the
(dry) surface of the upper thigh. Hence, sampling areas must be confined to skin immediately adjacent to the inguinal crease where skin-to-skin contact
provides the moist environment conducive to bacterial growth. Note that the large variance in the count data that results from randomization of the
FIG. 1 Illustration of Approximate Sampling Locations on Treatment Sites: Inguen, Abdomen, Clavicular Region, and Median Cubital
Region of Arm
E1173 − 23
sampling areas likely will require testing of a relatively large number of subjects in order to demonstrate any statistical significance of post-treatment
reductions.
8.1.2.2 Because of constraints imposed by the anatomical area, sampling cylinders used for the inguinal sites must begenerally
approximate ≤ 1 in (≤ 2.54 cm) in diameter.
8.1.2.3 The test formulation and reference control materialmaterials are then randomized bilaterally to the treatment sites.
8.1.3 Abdominal treatment sites are to be located within 5-by-5-in (12.7-by-12.7-cm) sites below and areas to the right orand left
of the umbilicus, approximately midway between the umbilicus and the pubis. umbilicus. Using a 5-by-5-in (12.7-by-12.7-cm)
sterile template (for example, paper, plastic, cloth), the corners of each site are numbered 1, 2, 3, and 4 directly on the skin, using
a surgical skin marker. Numbering is to be the same for all abdominal sites: number 1 is placed at the top corner to the subject’s
right, and numbers 2, 3, and 4 are assigned in order clockwise from 1. Three quadrants of each site are used for the three different
treatment exposure times, and the remaining quadrant is used for a baseline count. The test formulation and reference control
materialmaterials are then randomized to the treatment sites, right and left, and baseline and the three post-treatment sampling
times are randomized to the four sampling areas within each site.
8.2 Vascular Precatheterization Skin Preparations:
8.2.1 The skin sites selected for evaluation of the effectiveness of vascular precatheterization skin preparations should include
body areas that may be catheterization sites and should include both moist and dry skin areas. Bacterial baseline populations should
2 2
be at least 3.0 log /cm greater on moist skin sites than the detection limit of the sampling procedure, and at least 1.0 log /cm
10 10
greater than the detection limit on dry skin sites. The preferred moist-skin areas are the inguina, and the preferred
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...