ASTM E2738-23
(Practice)Standard Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE) for Computed Radiography (CR) Test Methods
Standard Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE) for Computed Radiography (CR) Test Methods
SIGNIFICANCE AND USE
5.1 Personnel that are responsible for the creation, transfer, and storage of computed radiography NDE data will use this practice. This practice will define a set of information modules that along with the Practice E2339 and the DICOM standard will provide a standard means to organize CR inspection data. The CR inspection data may be displayed and analyzed on any device that conforms to the standard. Personnel wishing to view any CR inspection data stored in accordance with Practice E2339 may use this practice to help them decode and display the data contained in the DICONDE compliant inspection record.
SCOPE
1.1 This practice covers the interoperability of computed radiography (CR) imaging and data acquisition equipment by specifying image data transfer and archival storage methods in commonly accepted terms. This practice is intended to be used in conjunction with Practice E2339 on Digital Imaging and Communication in Nondestructive Evaluation (DICONDE). Practice E2339 defines an industrial adaptation of NEMA PS3 / ISO 12052 (DICOM, see http://medical.nema.org), an international standard for image data acquisition, review, storage, and archival storage. The goal of Practice E2339, commonly referred to as DICONDE, is to provide a standard that facilitates the display and analysis of NDE results on any system conforming to the DICONDE standard. Toward that end, Practice E2339 provides a data dictionary and a set of information modules that are applicable to all NDE modalities. This practice supplements Practice E2339 by providing information object definitions, information modules and a data dictionary that are specific to computed radiography test methods.
1.2 This practice has been developed to overcome the issues that arise when analyzing or archiving data from CR test equipment using proprietary data transfer and storage methods. As digital technologies evolve, data must remain decipherable through the use of open, industry-wide methods for data transfer and archival storage. This practice defines a method where all standard CR technique parameters and test results are communicated and stored in a standard manner regardless of changes in digital technology.
1.3 This practice does not specify:
1.3.1 A testing or validation procedure to assess an implementation's conformance to the standard.
1.3.2 The implementation details of any features of the standard on a device claiming conformance.
1.3.3 The overall set of features and functions to be expected from a system implemented by integrating a group of devices each claiming DICONDE conformance.
1.4 Although this practice contains no values that require units, it does describe methods to store and communicate data that do require units to be properly interpreted. The SI units required by this practice are to be regarded as standard. No other units of measurement are included in this practice.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Jun-2023
- Technical Committee
- E07 - Nondestructive Testing
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Dec-2019
- Effective Date
- 01-Mar-2019
- Effective Date
- 01-Jan-2018
- Effective Date
- 15-Jun-2017
- Effective Date
- 01-Feb-2017
- Effective Date
- 01-Aug-2016
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Dec-2015
- Effective Date
- 01-Sep-2015
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 15-Jun-2013
- Effective Date
- 01-Jun-2013
Overview
ASTM E2738-23 establishes a standardized practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE) for Computed Radiography (CR) test methods. Developed by ASTM International, this standard addresses the interoperability of CR imaging and data acquisition equipment by defining image data transfer and archival storage in universally recognized terms. It supplements ASTM E2339, which provides an industrial adaptation of the DICOM (Digital Imaging and Communications in Medicine/ISO 12052) standard, tailoring it specifically for CR modalities used in nondestructive evaluation (NDE).
By enabling the consistent organization, communication, and storage of CR inspection data, ASTM E2738-23 helps ensure that nondestructive testing (NDT) results remain accessible, interpretable, and compatible with any DICONDE-conformant system, reducing reliance on proprietary systems.
Key Topics
- Interoperability in Computed Radiography: Ensures CR image data and associated inspection parameters are encoded, transferred, and archived using standardized, widely-accepted formats.
- Data Communication and Storage: Utilizes a common data dictionary and communication protocols to facilitate data exchange between equipment and systems from different manufacturers.
- Information Object Definitions (IODs): Specifies the structure and content required for CR images in NDE applications, extending the generic DICONDE model with CR-specific modules.
- Information Modules: Defines detailed modules for attributes such as NDE CR Series, NDE Source Radiography, and NDE CR Image, covering critical technical data like part examined, radiographic view, filter types, exposure parameters, and equipment details.
- SI Units and Data Consistency: Mandates the use of SI units for precise interpretation and standardization across international applications.
- Change Management: Addresses updates and corrections to data models and modules, ensuring continuous alignment with practical needs.
Applications
ASTM E2738-23 is widely applicable across industries where computed radiography plays a critical role in nondestructive evaluation, including but not limited to:
- Aerospace and Defense: For inspection of composite structures, welds, and critical components in compliance with government and industry regulations.
- Oil & Gas: Used for pipeline integrity assessments and equipment inspections, improving data transfer and long-term storage reliability.
- Automotive Manufacturing: Supports high-quality standards in parts verification and quality assurance tests.
- Power Generation: Enables efficient sharing and analysis of CR data for maintenance of turbines, boilers, and critical assets.
- Research and Development: Facilitates the interoperability of R&D imaging equipment, supporting collaborative analysis and data archiving.
With ASTM E2738-23, organizations benefit from enhanced data integrity, easier system integration, improved equipment compatibility, and reduced barriers to transitioning between different digital platforms.
Related Standards
- ASTM E2339 - Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE): The foundational standard for DICONDE, outlining the general data dictionary and modules applicable to all NDE modalities.
- ISO 12052 / NEMA PS3 (DICOM) - The international standard for digital image communication, originally developed for the medical field but adapted for industrial use in DICONDE.
- ASTM E1316 - Terminology for Nondestructive Examinations: Provides standardized terms used across NDT and NDE standards.
Adoption of ASTM E2738-23 enhances digital imaging workflows for nondestructive testing professionals, ensuring that computed radiography images and supporting data are readily accessible, interpretable, and preserved for the future. Use of this standard helps organizations meet global quality requirements, supports regulatory compliance, and streamlines inspection data interoperability.
Buy Documents
ASTM E2738-23 - Standard Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE) for Computed Radiography (CR) Test Methods
REDLINE ASTM E2738-23 - Standard Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE) for Computed Radiography (CR) Test Methods
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

NYCE
Mexican standards and certification body.
Sponsored listings
Frequently Asked Questions
ASTM E2738-23 is a standard published by ASTM International. Its full title is "Standard Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE) for Computed Radiography (CR) Test Methods". This standard covers: SIGNIFICANCE AND USE 5.1 Personnel that are responsible for the creation, transfer, and storage of computed radiography NDE data will use this practice. This practice will define a set of information modules that along with the Practice E2339 and the DICOM standard will provide a standard means to organize CR inspection data. The CR inspection data may be displayed and analyzed on any device that conforms to the standard. Personnel wishing to view any CR inspection data stored in accordance with Practice E2339 may use this practice to help them decode and display the data contained in the DICONDE compliant inspection record. SCOPE 1.1 This practice covers the interoperability of computed radiography (CR) imaging and data acquisition equipment by specifying image data transfer and archival storage methods in commonly accepted terms. This practice is intended to be used in conjunction with Practice E2339 on Digital Imaging and Communication in Nondestructive Evaluation (DICONDE). Practice E2339 defines an industrial adaptation of NEMA PS3 / ISO 12052 (DICOM, see http://medical.nema.org), an international standard for image data acquisition, review, storage, and archival storage. The goal of Practice E2339, commonly referred to as DICONDE, is to provide a standard that facilitates the display and analysis of NDE results on any system conforming to the DICONDE standard. Toward that end, Practice E2339 provides a data dictionary and a set of information modules that are applicable to all NDE modalities. This practice supplements Practice E2339 by providing information object definitions, information modules and a data dictionary that are specific to computed radiography test methods. 1.2 This practice has been developed to overcome the issues that arise when analyzing or archiving data from CR test equipment using proprietary data transfer and storage methods. As digital technologies evolve, data must remain decipherable through the use of open, industry-wide methods for data transfer and archival storage. This practice defines a method where all standard CR technique parameters and test results are communicated and stored in a standard manner regardless of changes in digital technology. 1.3 This practice does not specify: 1.3.1 A testing or validation procedure to assess an implementation's conformance to the standard. 1.3.2 The implementation details of any features of the standard on a device claiming conformance. 1.3.3 The overall set of features and functions to be expected from a system implemented by integrating a group of devices each claiming DICONDE conformance. 1.4 Although this practice contains no values that require units, it does describe methods to store and communicate data that do require units to be properly interpreted. The SI units required by this practice are to be regarded as standard. No other units of measurement are included in this practice. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Personnel that are responsible for the creation, transfer, and storage of computed radiography NDE data will use this practice. This practice will define a set of information modules that along with the Practice E2339 and the DICOM standard will provide a standard means to organize CR inspection data. The CR inspection data may be displayed and analyzed on any device that conforms to the standard. Personnel wishing to view any CR inspection data stored in accordance with Practice E2339 may use this practice to help them decode and display the data contained in the DICONDE compliant inspection record. SCOPE 1.1 This practice covers the interoperability of computed radiography (CR) imaging and data acquisition equipment by specifying image data transfer and archival storage methods in commonly accepted terms. This practice is intended to be used in conjunction with Practice E2339 on Digital Imaging and Communication in Nondestructive Evaluation (DICONDE). Practice E2339 defines an industrial adaptation of NEMA PS3 / ISO 12052 (DICOM, see http://medical.nema.org), an international standard for image data acquisition, review, storage, and archival storage. The goal of Practice E2339, commonly referred to as DICONDE, is to provide a standard that facilitates the display and analysis of NDE results on any system conforming to the DICONDE standard. Toward that end, Practice E2339 provides a data dictionary and a set of information modules that are applicable to all NDE modalities. This practice supplements Practice E2339 by providing information object definitions, information modules and a data dictionary that are specific to computed radiography test methods. 1.2 This practice has been developed to overcome the issues that arise when analyzing or archiving data from CR test equipment using proprietary data transfer and storage methods. As digital technologies evolve, data must remain decipherable through the use of open, industry-wide methods for data transfer and archival storage. This practice defines a method where all standard CR technique parameters and test results are communicated and stored in a standard manner regardless of changes in digital technology. 1.3 This practice does not specify: 1.3.1 A testing or validation procedure to assess an implementation's conformance to the standard. 1.3.2 The implementation details of any features of the standard on a device claiming conformance. 1.3.3 The overall set of features and functions to be expected from a system implemented by integrating a group of devices each claiming DICONDE conformance. 1.4 Although this practice contains no values that require units, it does describe methods to store and communicate data that do require units to be properly interpreted. The SI units required by this practice are to be regarded as standard. No other units of measurement are included in this practice. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E2738-23 is classified under the following ICS (International Classification for Standards) categories: 35.240.80 - IT applications in health care technology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E2738-23 has the following relationships with other standards: It is inter standard links to ASTM E1316-24, ASTM E1316-19b, ASTM E1316-19, ASTM E1316-18, ASTM E1316-17a, ASTM E1316-17, ASTM E1316-16a, ASTM E1316-16, ASTM E1316-15a, ASTM E1316-15, ASTM E1316-14e1, ASTM E1316-14, ASTM E1316-13d, ASTM E1316-13c, ASTM E1316-13b. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E2738-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E2738 − 23
Standard Practice for
Digital Imaging and Communication in Nondestructive
Evaluation (DICONDE) for Computed Radiography (CR) Test
Methods
This standard is issued under the fixed designation E2738; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope* 1.3.2 The implementation details of any features of the
standard on a device claiming conformance.
1.1 This practice covers the interoperability of computed
1.3.3 The overall set of features and functions to be ex-
radiography (CR) imaging and data acquisition equipment by
pected from a system implemented by integrating a group of
specifying image data transfer and archival storage methods in
devices each claiming DICONDE conformance.
commonly accepted terms. This practice is intended to be used
in conjunction with Practice E2339 on Digital Imaging and 1.4 Although this practice contains no values that require
Communication in Nondestructive Evaluation (DICONDE). units, it does describe methods to store and communicate data
Practice E2339 defines an industrial adaptation of NEMA that do require units to be properly interpreted. The SI units
PS3 ⁄ ISO 12052 (DICOM, see http://medical.nema.org), an required by this practice are to be regarded as standard. No
international standard for image data acquisition, review, other units of measurement are included in this practice.
storage, and archival storage. The goal of Practice E2339,
1.5 This standard does not purport to address all of the
commonly referred to as DICONDE, is to provide a standard
safety concerns, if any, associated with its use. It is the
that facilitates the display and analysis of NDE results on any
responsibility of the user of this standard to establish appro-
system conforming to the DICONDE standard. Toward that
priate safety, health, and environmental practices and deter-
end, Practice E2339 provides a data dictionary and a set of
mine the applicability of regulatory limitations prior to use.
information modules that are applicable to all NDE modalities.
1.6 This international standard was developed in accor-
This practice supplements Practice E2339 by providing infor-
dance with internationally recognized principles on standard-
mation object definitions, information modules and a data
ization established in the Decision on Principles for the
dictionary that are specific to computed radiography test
Development of International Standards, Guides and Recom-
methods.
mendations issued by the World Trade Organization Technical
Barriers to Trade (TBT) Committee.
1.2 This practice has been developed to overcome the issues
that arise when analyzing or archiving data from CR test
2. Referenced Documents
equipment using proprietary data transfer and storage methods.
As digital technologies evolve, data must remain decipherable 2.1 ASTM Standards:
through the use of open, industry-wide methods for data E1316 Terminology for Nondestructive Examinations
transfer and archival storage. This practice defines a method E2339 Practice for Digital Imaging and Communication in
where all standard CR technique parameters and test results are Nondestructive Evaluation (DICONDE)
communicated and stored in a standard manner regardless of 2.2 Other Documentation:
changes in digital technology.
DICOM NEMA PS3 / ISO 12052 Digital Imaging and
Communications in Medicine (DICOM)
1.3 This practice does not specify:
1.3.1 A testing or validation procedure to assess an imple-
3. Terminology
mentation’s conformance to the standard.
3.1 Definitions:
1 2
This practice is under the jurisdiction of ASTM Committee E07 on Nonde- For referenced ASTM standards, visit the ASTM website, www.astm.org, or
structive Testing and is the direct responsibility of Subcommittee E07.11 on Digital contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Imaging and Communication in Nondestructive Evaluation (DICONDE). Standards volume information, refer to the standard’s Document Summary page on
Current edition approved June 15, 2023. Published July 2023. Originally the ASTM website.
approved in 2010. Last previous edition approved in 2018 as E2738 – 18. DOI: Available from National Electrical Manufacturers Association (NEMA), 1300
10.1520/E2738-23. N. 17th St., Suite 1752, Rosslyn, VA 22209, http://www.dicomstandard.org/
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2738 − 23
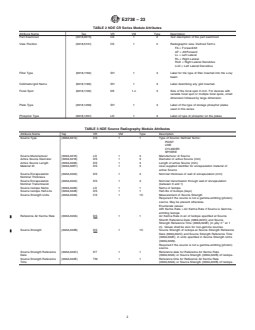
TABLE 1 NDE CR Image Information Object Definition
DICOM Module DICONDE Module Reference Usage
Patient Component E2339, Section 7 M
Clinical Trial Subject Not Applicable
General Study Component Study E2339, Section 7 M
Patient Study Not Applicable
Clinical Trial Study Not Applicable
General Series Component Series E2339, Section 7 M
CR Series NDE CR Series Section 7.1 M
Clinical Trial Series Not Applicable U
General Equipment NDE Equipment E2339, Section 7 M
Contrast/bolous Not Applicable U
CR image NDE CR image Section 7.3 M
NDE Indication E2339, Section 7 U
NDE Geometry E2339, Section 7 U
NDE Source Radiography Section 7.2 U
NDE Data Element Label Dictionary E2339, Section 7 U
NDE Geolocation E2339, Section 7 U
3.1.1 Nondestructive evaluation terms used in this practice device that conforms to the standard. Personnel wishing to
can be found in Terminology E1316. view any CR inspection data stored in accordance with Practice
3.1.2 DICONDE terms used in this practice are defined in E2339 may use this practice to help them decode and display
Practice E2339. the data contained in the DICONDE compliant inspection
record.
4. Summary of Practice
6. Information Object Definitions
4.1 A fundamental principle of DICONDE is the use of
standard definitions and attribute formats for data communica- 6.1 NDE Computed Radiography Image IOD Description:
tion and storage. This means all systems that are DICONDE 6.1.1 The NDE Computed Radiography (CR) Image Infor-
compliant use a common data dictionary and common com- mation Object Definition specifies an image that has been
munication protocols. To further standardization, the elements created by a computed radiography imaging device for NDE
in the data dictionary are organized into common groups purposes. The IOD definition will follow that for CR Images
referred to as information modules. The data dictionary and found in Part 3, Section A.2 of the DICOM standard except as
information modules common to all NDE modalities are noted in Table 1. Table 1 is not stand-alone and must be used
defined in Practice E2339. in conjunction with Part 3, Section A.2 of the DICOM standard
to have a complete definition of the NDE CR information
4.2 The data dictionary and information modules specified
object.
in Practice E2339 do not cover the information storage
6.1.2 This IOD will use the Service-Object Pair (SOP) class
requirements for each individual modality (CT, DDA, CR, UT,
for the CR IOD as defined in Part 4, Section B.5 of the DICOM
etc.). Additions to the data dictionary and information modules
standard.
are required to support the individual modalities. This practice
contains the additions to the DICONDE data dictionary and
7. Information Modules
information modules necessary for CR inspection.
7.1 NDE CR Series Module:
4.3 The highest organizational level in the DICONDE
7.1.1 Table 2 specifies the attributes that describe NDE
information model is the information object definition (IOD).
Computed Radiography images.
An information object definition is a collection of the infor-
7.2 NDE Source Radiography Module:
mation modules necessary to represent a set of test results from
7.2.1 Table 3 specifies the attributes that describe NDE
a specific modality. This practice contains information object
Source Radiography Module.
definitions for CR inspection.
7.3 NDE CR Image Module:
5. Significance and Use
7.3.1 Table 4 specifies the attributes that describe NDE CR
5.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E2738 − 18 E2738 − 23
Standard Practice for
Digital Imaging and Communication in Nondestructive
Evaluation (DICONDE) for Computed Radiography (CR) Test
Methods
This standard is issued under the fixed designation E2738; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope*
1.1 This practice facilitatescovers the interoperability of computed radiography (CR) imaging and data acquisition equipment by
specifying image data transfer and archival storage methods in commonly accepted terms. This practice is intended to be used in
conjunction with Practice E2339 on Digital Imaging and Communication in Nondestructive Evaluation (DICONDE). Practice
E2339 defines an industrial adaptation of the NEMA StandardsPS3 Publication titled Digital Imaging and Communications in
Medicine ⁄ ISO 12052 (DICOM, see http://medical.nema.org), an international standard for image data acquisition, review, storage,
and archival storage. The goal of Practice E2339, commonly referred to as DICONDE, is to provide a standard that facilitates the
display and analysis of NDE results on any system conforming to the DICONDE standard. Toward that end, Practice E2339
provides a data dictionary and a set of information modules that are applicable to all NDE modalities. This practice supplements
Practice E2339 by providing information object definitions, information modules and a data dictionary that are specific to
computed radiography test methods.
1.2 This practice has been developed to overcome the issues that arise when analyzing or archiving data from CR test equipment
using proprietary data transfer and storage methods. As digital technologies evolve, data must remain decipherable through the use
of open, industry-wide methods for data transfer and archival storage. This practice defines a method where all standard CR
technique parameters and test results are communicated and stored in a standard manner regardless of changes in digital
technology.
1.3 This practice does not specify:
1.3.1 A testing or validation procedure to assess an implementation’s conformance to the standard.
1.3.2 The implementation details of any features of the standard on a device claiming conformance.
1.3.3 The overall set of features and functions to be expected from a system implemented by integrating a group of devices each
claiming DICONDE conformance.
1.4 Although this practice contains no values that require units, it does describe methods to store and communicate data that do
require units to be properly interpreted. The SI units required by this practice are to be regarded as standard. No other units of
measurement are included in this practice.
This practice is under the jurisdiction of ASTM Committee E07 on Nondestructive Testing and is the direct responsibility of Subcommittee E07.11 on Digital Imaging
and Communication in Nondestructive Evaluation (DICONDE).
Current edition approved Nov. 1, 2018June 15, 2023. Published November 2018July 2023. Originally approved in 2010. Last previous edition approved in 20132018 as
ɛ01
E2738 - 13E2738 . DOI: 10.1520/E2738-18. – 18. DOI: 10.1520/E2738-23.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E2738 − 23
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E1316 Terminology for Nondestructive Examinations
E2339 Practice for Digital Imaging and Communication in Nondestructive Evaluation (DICONDE)
2.2 Other Documentation:
DICOM NEMA PS3 / ISO 12052 National Electrical Manufacturers Association Standard for Digital Imaging and Communi-
cationCommunications in Medicine (DICOM), 2011.(DICOM)
3. Terminology
3.1 Definitions:
3.1.1 Nondestructive evaluation terms used in this practice can be found in Terminology E1316.
3.1.2 DICONDE terms used in this practice are defined in Practice E2339.
4. Summary of Practice
4.1 A fundamental principle of DICONDE is the use of standard definitions and attribute formats for data communication and
storage. This means all systems that are DICONDE compliant use a common data dictionary and common communication
protocols. To further standardization, the elements in the data dictionary are organized into common groups referred to as
information modules. The data dictionary and information modules common to all NDE modalities are defined in Practice E2339.
4.2 The data dictionary and information modules specified in Practice E2339 do not cover the information storage requirements
for each individual modality (CT, DDA, CR, UT, etc.). Additions to the data dictionary and information modules are required to
support the individual modalities. This practice contains the additions to the DICONDE data dictionary and information modules
necessary for CR inspection.
4.3 The highest organizational level in the DICONDE information model is the information object definition (IOD). An
information object definition is a collection of the information modules necessary to represent a set of test results from a specific
modality. This practice contains information object definitions for CR inspection.
5. Significance and Use
5.1 Personnel that are responsible for the creation, transfer, and storage of computed radiography NDE data will use this practice.
This practice will define a set of information modules that along with the Practice E2339 and the DICOM standard will provide
a standard means to organize CR inspection data. The CR inspection data may be displayed and analyzed on any device that
conforms to the standard. Personnel wishing to view any CR inspection data stored in accordance with Practice E2339 may use
this practice to help them decode and display the data contained in the DICONDE compliant inspection record.
6. Information Object Definitions
6.1 NDE Computed Radiography Image IOD Description:
6.1.1 The NDE Computed Radiography (CR) Image Information Object Definition specifies an image that has been created by
a computed radiography imaging device for NDE purposes. The IOD definition will follow that for CR Images found in Part 3,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from National Electrical Manufacturers Association (NEMA), 1300 N. 17th St., Suite 1752, Rosslyn, VA 22209, http://www.dicomstandard.org/
E2738 − 23
TABLE 1 NDE CR Image Information Object Definition
DICOM Module DICONDE Module Reference Usage
Patient Component E2339, Section 7 M
Clinical Trial Subject Not Applicable
General Study Component Study E2339, Section 7 M
Patient Study Not Applicable
Clinical Trial Study Not Applicable
General Series Component Series E2339, Section 7 M
CR Series NDE CR Series Section 7.1 M
CR Series NDE CR Series Section 7.1 M
Clinical Trial Series Not Applicable U
General Equipment NDE Equipment E2339, Section 7 M
Contrast/bolous Not Applicable U
CR image NDE CR image Section 7.3 M
NDE Indication E2339, Section 7 U
NDE Geometry E2339, Section 7 U
NDE Source Radiography Section 7.2 U
NDE Data Element Label Dictionary E2339, Section 7 U
NDE Geolocation E2339, Section 7 U
Section A.2 of the DICOM standard except as noted in Table 1. Table 1 is not stand-alone and must be used in conjunction with
Part 3,
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...