ASTM D8310-20
(Test Method)Standard Test Method for Analysis of Target Phenols (TPs) in Soil by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
Standard Test Method for Analysis of Target Phenols (TPs) in Soil by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
SIGNIFICANCE AND USE
5.1 This test method developed for the analysis of TPs in soil and sediment samples is based upon an LC/MS/MS analysis. Any type of coupled liquid chromatography/mass spectrometry system may be used that meets the study objectives of the individual project. These may include, but are not limited to: trap, single quadrupoles, time-of-flight, high resolution, and others not mentioned here.
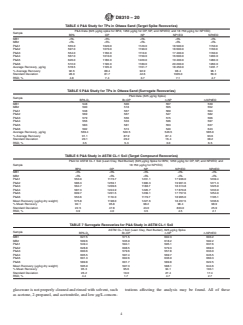
5.2 The MDL and reporting range for TPs are listed in Table 1. This SOP has been tested on Ottawa sand, four ASTM soil types, biosolid sample, and one commercial soil. The P&A QC acceptance criteria are listed in Table 3. Tables 4-17 display the TC and surrogate recoveries in the various soil types. 40 CFR Part 136, Appendix B was used as a guide to determine the MDLs. The 40 CFR Part 136 MDL criteria were not met for NP2EO; this does not affect the method because the SOP only reports to the RL and is not a regulatory method. All site sample results are not reported below the RL using this method. RLCS concentrations may be reported below the RL because they are spiked at or near the RL. (A) Uncertainty calculation based upon 95 % confidence interval and a two-tailed Student t distribution.
Uncertainty = Student t Value [(standard deviation) / (number of LCS)1/2]. (A) P&A values are after subtraction of average of MB if ≥RL. (A) P&A values are after subtraction of average of MB if ≥RL. (A)
5.3 The RL for a specific soil sample may differ from that listed depending on the nature of the interferences in the sample matrix. Variability in historical LCS spike recovery may be used to estimate uncertainty. The estimate of minimum laboratory contribution to measurement uncertainty of this test method for each analyte is listed in Table 3. These values are derived from P&A samples from the initial IDOC study for this test method. The uncertainty will be greater near the RL and much greater near the DL. Also, uncertainty estimated based on variability in LCS recov...
SCOPE
1.1 This test method covers analysis of nonylphenol (NP), nonylphenol monoethoxylate (NP1EO), nonylphenol diethoxylate (NP2EO), octylphenol (OP), and bisphenol A (BPA), referred to collectively as target phenols (TPs), in soil, sediments, and biosolids by extraction with acetone, filtration, dilution with water, and analysis by liquid chromatography/tandem mass spectrometry. The sample extracts are prepared in a solution of 75 % acetone and 25 % water because TPs have an affinity for surfaces and particles that is more pronounced at lower concentrations. The range of applicability of the test method is shown in Table 1. The method may be extended outside of these ranges depending on additional performance studies not undertaken here.
1.2 Units—The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2020
- Technical Committee
- D34 - Waste Management
- Drafting Committee
- D34.01.06 - Analytical Methods
Relations
- Effective Date
- 01-Sep-2018
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM D8310-20 is the internationally recognized standard test method for the analysis of target phenols (TPs) in soil, sediments, and biosolids using Liquid Chromatography/Mass Spectrometry (LC/MS/MS) with Multiple Reaction Monitoring (MRM). The method focuses on the quantitative detection of key phenolic environmental contaminants-namely, nonylphenol (NP), nonylphenol monoethoxylate (NP1EO), nonylphenol diethoxylate (NP2EO), octylphenol (OP), and bisphenol A (BPA)-collectively termed target phenols. This standard supports robust environmental monitoring and contamination assessment initiatives, enabling laboratories to generate precise and reliable results.
Key Topics
Scope and Purpose
- Enables the analysis of TPs in a range of matrices, including soil, sediments, and biosolids.
- Designed for use with a variety of coupled liquid chromatography/mass spectrometry systems that meet specific project requirements.
- Provides clear guidance for spiking, extraction, filtration, and quantification, ensuring consistent application.
Sample Preparation and Analysis
- Extraction using 75% acetone and 25% water, maximizing analyte recovery due to strong surface affinity of TPs at low concentrations.
- Filtration and dilution precede LC/MS/MS analysis, minimizing interferences and ensuring consistent chromatography.
Quality Control and Uncertainty
- Includes rigorous QC criteria: method blanks, laboratory control samples, matrix spikes, and surrogate recoveries.
- Reporting limits (RLs) are set methodically based on matrix interference and method detection limits (MDLs).
- Measurement uncertainty is estimated and controlled as part of initial and ongoing demonstration of capability.
Safety and Compliance
- Emphasizes the need for proper laboratory safety, with explicit warnings about chemical hazards and the importance of using reagent-grade chemicals.
- Developed under internationally recognized standardization principles, ensuring global applicability and alignment with WTO TBT requirements.
Applications
Environmental Monitoring
- Used for tracking the presence and concentration of key endocrine disrupting chemicals and industrial byproducts in terrestrial matrices.
- Supports site assessments, remediation projects, and regulatory compliance activities related to soil and sediment quality.
Waste Management and Research
- Essential tool for analyzing biosolids and soils in waste management facilities and environmental research studies.
- Provides reproducible, quantitative data for determining contamination levels of BPA, nonylphenols, and octylphenols.
Regulatory and Industry Compliance
- Can assist laboratories and organizations in meeting local, national, and international environmental monitoring requirements.
- Ideal for research institutions, consulting laboratories, and regulatory agencies focused on hazardous substances in environmental matrices.
Related Standards
- ASTM D7858: Test Method for Determination of Bisphenol A in Soil, Sludge, and Biosolids.
- ASTM D1193: Specification for Reagent Water.
- US EPA 40 CFR Part 136, Appendix B: Definitions and procedures for method detection limits.
- OSHA 29 CFR Part 1910: Occupational Safety and Health Standards related to laboratory safety.
These related references further support comprehensive monitoring and analysis strategies for phenolic contaminants.
By adhering to ASTM D8310-20, laboratories gain a standardized approach to the detection and quantification of target phenols in soils and sediments, supporting data quality, environmental stewardship, and regulatory compliance. For optimal SEO, keywords such as target phenols analysis, LC/MS/MS, environmental monitoring, nonylphenol, bisphenol A, soil testing standard, and ASTM D8310-20 have been integrated throughout this summary.
Buy Documents
ASTM D8310-20 - Standard Test Method for Analysis of Target Phenols (TPs) in Soil by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D8310-20 is a standard published by ASTM International. Its full title is "Standard Test Method for Analysis of Target Phenols (TPs) in Soil by Multiple Reaction Monitoring Liquid Chromatography/Mass Spectrometry (LC/MS/MS)". This standard covers: SIGNIFICANCE AND USE 5.1 This test method developed for the analysis of TPs in soil and sediment samples is based upon an LC/MS/MS analysis. Any type of coupled liquid chromatography/mass spectrometry system may be used that meets the study objectives of the individual project. These may include, but are not limited to: trap, single quadrupoles, time-of-flight, high resolution, and others not mentioned here. 5.2 The MDL and reporting range for TPs are listed in Table 1. This SOP has been tested on Ottawa sand, four ASTM soil types, biosolid sample, and one commercial soil. The P&A QC acceptance criteria are listed in Table 3. Tables 4-17 display the TC and surrogate recoveries in the various soil types. 40 CFR Part 136, Appendix B was used as a guide to determine the MDLs. The 40 CFR Part 136 MDL criteria were not met for NP2EO; this does not affect the method because the SOP only reports to the RL and is not a regulatory method. All site sample results are not reported below the RL using this method. RLCS concentrations may be reported below the RL because they are spiked at or near the RL. (A) Uncertainty calculation based upon 95 % confidence interval and a two-tailed Student t distribution. Uncertainty = Student t Value [(standard deviation) / (number of LCS)1/2]. (A) P&A values are after subtraction of average of MB if ≥RL. (A) P&A values are after subtraction of average of MB if ≥RL. (A) 5.3 The RL for a specific soil sample may differ from that listed depending on the nature of the interferences in the sample matrix. Variability in historical LCS spike recovery may be used to estimate uncertainty. The estimate of minimum laboratory contribution to measurement uncertainty of this test method for each analyte is listed in Table 3. These values are derived from P&A samples from the initial IDOC study for this test method. The uncertainty will be greater near the RL and much greater near the DL. Also, uncertainty estimated based on variability in LCS recov... SCOPE 1.1 This test method covers analysis of nonylphenol (NP), nonylphenol monoethoxylate (NP1EO), nonylphenol diethoxylate (NP2EO), octylphenol (OP), and bisphenol A (BPA), referred to collectively as target phenols (TPs), in soil, sediments, and biosolids by extraction with acetone, filtration, dilution with water, and analysis by liquid chromatography/tandem mass spectrometry. The sample extracts are prepared in a solution of 75 % acetone and 25 % water because TPs have an affinity for surfaces and particles that is more pronounced at lower concentrations. The range of applicability of the test method is shown in Table 1. The method may be extended outside of these ranges depending on additional performance studies not undertaken here. 1.2 Units—The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method developed for the analysis of TPs in soil and sediment samples is based upon an LC/MS/MS analysis. Any type of coupled liquid chromatography/mass spectrometry system may be used that meets the study objectives of the individual project. These may include, but are not limited to: trap, single quadrupoles, time-of-flight, high resolution, and others not mentioned here. 5.2 The MDL and reporting range for TPs are listed in Table 1. This SOP has been tested on Ottawa sand, four ASTM soil types, biosolid sample, and one commercial soil. The P&A QC acceptance criteria are listed in Table 3. Tables 4-17 display the TC and surrogate recoveries in the various soil types. 40 CFR Part 136, Appendix B was used as a guide to determine the MDLs. The 40 CFR Part 136 MDL criteria were not met for NP2EO; this does not affect the method because the SOP only reports to the RL and is not a regulatory method. All site sample results are not reported below the RL using this method. RLCS concentrations may be reported below the RL because they are spiked at or near the RL. (A) Uncertainty calculation based upon 95 % confidence interval and a two-tailed Student t distribution. Uncertainty = Student t Value [(standard deviation) / (number of LCS)1/2]. (A) P&A values are after subtraction of average of MB if ≥RL. (A) P&A values are after subtraction of average of MB if ≥RL. (A) 5.3 The RL for a specific soil sample may differ from that listed depending on the nature of the interferences in the sample matrix. Variability in historical LCS spike recovery may be used to estimate uncertainty. The estimate of minimum laboratory contribution to measurement uncertainty of this test method for each analyte is listed in Table 3. These values are derived from P&A samples from the initial IDOC study for this test method. The uncertainty will be greater near the RL and much greater near the DL. Also, uncertainty estimated based on variability in LCS recov... SCOPE 1.1 This test method covers analysis of nonylphenol (NP), nonylphenol monoethoxylate (NP1EO), nonylphenol diethoxylate (NP2EO), octylphenol (OP), and bisphenol A (BPA), referred to collectively as target phenols (TPs), in soil, sediments, and biosolids by extraction with acetone, filtration, dilution with water, and analysis by liquid chromatography/tandem mass spectrometry. The sample extracts are prepared in a solution of 75 % acetone and 25 % water because TPs have an affinity for surfaces and particles that is more pronounced at lower concentrations. The range of applicability of the test method is shown in Table 1. The method may be extended outside of these ranges depending on additional performance studies not undertaken here. 1.2 Units—The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D8310-20 is classified under the following ICS (International Classification for Standards) categories: 13.080.10 - Chemical characteristics of soils. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8310-20 has the following relationships with other standards: It is inter standard links to ASTM D7858-13(2018), ASTM D7858-13, ASTM D1193-06, ASTM D1193-99e1, ASTM D1193-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8310-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D8310 − 20
Standard Test Method for
Analysis of Target Phenols (TPs) in Soil by Multiple
Reaction Monitoring Liquid Chromatography/Mass
Spectrometry (LC/MS/MS)
This standard is issued under the fixed designation D8310; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope D7858Test Method for Determination of Bisphenol A in
Soil, Sludge, and Biosolids by Pressurized Fluid Extrac-
1.1 This test method covers analysis of nonylphenol (NP),
tion and Analyzed by Liquid Chromatography/Tandem
nonylphenolmonoethoxylate(NP1EO),nonylphenoldiethoxy-
Mass Spectrometry
late (NP2EO), octylphenol (OP), and bisphenol A (BPA),
2.2 Federal Standards:
referred to collectively as target phenols (TPs), in soil,
29CFRPart1910OccupationalSafetyandHealthStandards
sediments, and biosolids by extraction with acetone, filtration,
40 CFR Part 136,Appendix BDefinition and Procedure for
dilution with water, and analysis by liquid chromatography/
the Determination of the Method Detection Limit—
tandemmassspectrometry.Thesampleextractsarepreparedin
Revision 1.11
a solution of 75% acetone and 25% water because TPs have
anaffinityforsurfacesandparticlesthatismorepronouncedat
3. Terminology
lower concentrations. The range of applicability of the test
3.1 Definitions of Terms Specific to This Standard:
method is shown in Table 1. The method may be extended
3.1.1 batch quality control, n—all the quality control (QC)
outside of these ranges depending on additional performance
samples and standards included in an analytical procedure.
studies not undertaken here.
3.1.2 bisphenol A, BPA—defined in Test Method D7858.
1.2 Units—The values stated in SI units are to be regarded
3.1.3 bisphenol A (propane-D6), BPA-D —defined in Test
as the standard. No other units of measurement are included in
Method D7858.
this standard.
3.1.4 2-bromo-4-(1,1,3,3-tetramethylbutyl)phenol, Br-OP,
1.3 This standard does not purport to address all of the
n—used in this test method as a surrogate.
safety concerns, if any, associated with its use. It is the
3.1.4.1 Discussion—2-bromo-4-(1,1,3,3-tetramethylbutyl)
responsibility of the user of this standard to establish appro-
phenol is not produced commercially and is not expected to be
priate safety, health, and environmental practices and deter-
found in the environment. It was reported that compounds in
mine the applicability of regulatory limitations prior to use.
highlychlorinated,bromide-richwastewaterscouldpotentially
1.4 This international standard was developed in accor-
interfere with the Br-OP surrogate. If this interference is
dance with internationally recognized principles on standard-
encountered, n-nonylphenol is suggested as an alternative
ization established in the Decision on Principles for the
surrogate.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical 3.1.5 nonylphenol, NP, n—mixture of branched
Barriers to Trade (TBT) Committee. p-nonylphenol isomers.
3.1.5.1 Discussion—Commercial NP is produced by the
2. Referenced Documents
reaction of phenol with commercial nonene. Commercial
2.1 ASTM Standards: nonene is not simply a linear C H alpha olefin; it is a
9 18
complex mixture of predominantly nine-carbon olefins, called
D1193Specification for Reagent Water
propylene trimer, containing no linear isomers. This synthesis
results in a mixture of various branched nonylphenol isomers
This test method is under the jurisdiction ofASTM Committee D34 on Waste
rather than a discrete chemical structure. The branched nonyl
Management and is the direct responsibility of Subcommittee D34.01.06 on
Analytical Methods.
group is positioned predominantly in the para position on the
CurrenteditionapprovedFeb.1,2020.PublishedFebruary2020.DOI:10.1520/
phenol ring.
D8310-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Available from U.S. Government Printing Office, Superintendent of
Standards volume information, refer to the standard’s Document Summary page on Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://
the ASTM website. www.access.gpo.gov.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8310 − 20
TABLE 1 Tested Method Parameters of the Standard
3.2.19 MSP—method specific parameter
ESI MDL Reporting
3.2.20 NA—not available
Analyte
A
Mode (µg/kg) Range (µg/kg)
Bisphenol A Negative 15.5 100–2500 3.2.21 NP1EO—nonylphenol monoethoxylate
Octylphenol Negative 44.2 200–5000
3.2.22 NP2EO—nonylphenol diethoxylate
Nonylphenol Negative 30.4 100–2500
Nonylphenol Monoethoxylate Positive 931.2 3000–45 000
3.2.23 OP—octylphenol
Nonylphenol Diethoxylate Positive 7.4 100–2500
A 3.2.24 P&A—precision and accuracy
MDL is calculated based upon nine spiked samples.
3.2.25 PPB—parts per billion
3.2.26 PPM—parts per million
3.2.27 PPT—parts per trillion
3.1.6 nonylphenol diethoxylate, NP2EO, n—branched non-
3.2.28 PTFE—polytetrafluoroethylene
ylphenol diethoxylate.
3.2.29 PVDF—poly-vinylidene dichloride
3.1.7 nonylphenol monoethoxylate, NP1EO, n—branched
nonylphenol monoethoxylate.
3.2.30 QA—quality assurance
3.1.8 normal nonylphenol, n-NP, n—normal straight chain
3.2.31 QC—quality control
nonylphenol.
3.2.32 QMP—quality management plan
3.1.8.1 Discussion—n-NP is used in this test method as a
3.2.33 REC—percent recovery
surrogate. It is not produced commercially and is not expected
3.2.34 RL—reporting limit
to be found in the environment.
3.2.35 RLCS—reporting limit check sample
3.1.9 normal nonylphenol diethoxylate, n-NP2EO,
n—normal straight chain nonylphenol diethoxylate.
3.2.36 RSD—relative standard deviation
3.1.9.1 Discussion—n-NP2EOisusedinthistestmethodas
3.2.37 RT—retention time
a surrogate. It is not produced commercially and is not
3.2.38 RTS—retention time shift
expected to be found in the environment.
3.2.39 SOP—standard operating procedure
3.1.10 octylphenol, OP, n—produced by the reaction of
3.2.40 SRM—single reaction monitoring
phenol and diisobutylene to produce predominantly the 4-(1,
1,3,3-tetramethylbutyl)phenol isomer.
3.2.41 SS—surrogate standard
3.1.11 reporting limit check sample, RLCS, n—this sample
3.2.42 TC—target compound
verifies that if the analyte was present at the reporting limit, it
3.2.43 TCL—target compound limit
would be confidently identified.
3.2.44 TP—target phenols
3.1.12 target phenols, TPs, n—in this test method, NP,
3.2.45 UPLC—ultra performance liquid chromatography
NP1EO, NP2EO, OP, and BPA, collectively.
3.2.46 VOA—volatile organic analysis
3.2 Abbreviations:
3.2.1 ADOC—analyst demonstration of capability
4. Summary of Test Method
3.2.2 BPA—bisphenol A
4.1 Asample (~2 g) is transferred to aVOAvial and spiked
3.2.3 Br-OP—2-bromo-4-(1,1,3,3-tetramethylbutyl)phenol
with surrogates (all samples) and TPs (laboratory control and
3.2.4 CAS—chemical abstract service
matrix spike samples) and then extracted with 7.5 mL of
3.2.5 CCC—continuing calibration check acetone by tumbling on a rotator for 2 h. Any device may be
usedthatmixesthesample;arotatordevicewaschoseninthis
3.2.6 DL—detection limit
case because it inverts the sample, allowing the soil to be in a
3.2.7 EPA—U.S. Environmental Protection Agency
constant fluid motion and not clumped. The samples are
3.2.8 IC—initial calibration
centrifugedat1900rpmfor10minandthenfilteredthroughan
3.2.9 IDOC—initial demonstration of capability Acrodisc GxF/0.2 µm PVDF membrane syringe-driven filter
unit. Of Specification D1193Type 1 water, 2.5 mLis added to
3.2.10 LC—liquid chromatography
the filtered extract and then analyzed by LC/MS/MS. All
3.2.11 LCS—laboratory control sample
concentrations reported, only to the RL, using this method are
3.2.12 LCSD—laboratory control sample duplicate
based upon a dry-weight basis.
3.2.13 LIMS—relational laboratory information manage-
4.2 TheTCsareidentifiedbycomparingtheSRMtransition
ment system
and RT. BPA has a confirmatory SRM transition also. The
3.2.14 MDL—method detection limit
confirmatory SRM transition will be correlated to the known
standard SRM transition for identification of BPA (Table 2).
3.2.15 MI—matrix interference
The RT for the analyte of interest shall also agree with the RT
3.2.16 MRM—multiple reaction monitoring
ofthemid-levelstandardby 65%.TheTCisquantitatedusing
3.2.17 MS—mass spectrometry
the SRM transition of the TC using external calibration.As an
3.2.18 MS/MSD—matrix spike/matrix spike duplicate additional QC measure, non-labeled surrogates (listed in 8.2)
D8310 − 20
TABLE 2 Variable Mass Spectrometer Parameters
Retention Time, SRM Mass Transition Cone Voltage, Collision Energy,
Analyte ESI Mode
min (Parent > Product) V eV
BPA negative 5.58 227.1 > 211.9 40 18
A
BPA confirmatory negative 5.58 227.1 > 132.7 40 25
OP negative 8.53 205.1 > 132.7 40 24
NP negative 9.70 219.3 > 133.1 40 28
NP1EO positive 9.71 282.3 > 126.9 20 9
NP2EO positive 9.65 326.3 > 182.9 25 11
BPA-D (surrogate) negative 5.57 233.3 > 214.9 40 19
A
BPA-D confirmatory (surrogate) negative 5.57 233.3 > 137.8 40 25
Br-OP (surrogate) negative 9.56 283.1 > 78.6 40 25
n-NP (surrogate) negative 10.71 219.3 > 105.6 40 20
n-NP2EO (surrogate) positive 10.69 326.4 > 88.8 25 15
A
Confirmatory transitions are optional but should be included for added qualitative information.
recoveries are monitored; the percent recovery of each should laboratory contribution to measurement uncertainty of this test
fall within the control limits of the method. The final report method for each analyte is listed in Table 3. These values are
issued for each sample lists concentration of TPs and derivedfromP&AsamplesfromtheinitialIDOCstudyforthis
surrogates, if detected, or non-detect at the RLif not detected, test method. The uncertainty will be greater near the RL and
in microgram/kilogram on a dry-weight basis. muchgreaterneartheDL.Also,uncertaintyestimatedbasedon
variability in LCS recovery is conservative because some
5. Significance and Use
sources of variability are not included, such as subsample
5.1 This test method developed for the analysis of TPs in variability and matrix analyte recovery. This SOP covers
multiple soil matrices and the uncertainty among the various
soil and sediment samples is based upon an LC/MS/MS
analysis. Any type of coupled liquid chromatography/mass matrices is variable.
spectrometry system may be used that meets the study objec-
6. Interferences
tives of the individual project. These may include, but are not
limited to: trap, single quadrupoles, time-of-flight, high 6.1 Methodinterferencesmaybecausedbycontaminantsin
resolution, and others not mentioned here. solvents, reagents, glassware, LC vials/caps, disposable
pipettes, and other apparatus that lead to discrete artifacts or
5.2 TheMDLandreportingrangeforTPsarelistedinTable
elevated baselines in the selected ion current profiles. The
1. This SOP has been tested on Ottawa sand, four ASTM soil
presence and magnitude of method interferences are deter-
types,biosolidsample,andonecommercialsoil.TheP&AQC
mined by analysis of solvent and laboratory blanks.
acceptancecriteriaarelistedinTable3.Tables4-17displaythe
TC and surrogate recoveries in the various soil types. 40 CFR 6.2 Matrix interferences may be caused by contaminants
Part 136, Appendix B was used as a guide to determine the from the sample, sampling devices, or storage containers. The
MDLs. The 40 CFR Part 136 MDL criteria were not met for extent of matrix interferences will vary considerably from
NP2EO; this does not affect the method because the SOPonly samplesourcetosamplesourcedependingonvariationsofthe
reports to the RL and is not a regulatory method. All site sample matrix. The analysis of matrix spikes is critical for
sample results are not reported below the RL using this determining the impact of matrix interferences.
method. RLCS concentrations may be reported below the RL
6.3 Warnings:
because they are spiked at or near the RL.
6.3.1 All reagents and solvents should be of pesticide
5.3 The RL for a specific soil sample may differ from that residue purity or higher to minimize interference problems,
listed depending on the nature of the interferences in the preferably LC/MS grade.
sample matrix. Variability in historical LCS spike recovery 6.3.2 Contaminants have been found in improperly cleaned
maybeusedtoestimateuncertainty.Theestimateofminimum glassware and glass syringes. TPs stick to surfaces if the
A
TABLE 3 QC Acceptance Criteria and Uncertainty
Standard Deviation
Average Recovery, Number of Lower Control Limit Upper Control Limit Uncertainty (95 %
Parameter of Percentage
% Replicates, n (LCL), % (UCL), % Confidence Interval)
Recovery
BPA 92.6 4.5 6 70 130 4.7
OP 88.4 6.5 6 60 130 6.9
NP 92.9 3.4 6 70 130 3.6
NP1EO 98.4 7.0 6 70 130 7.3
NP2EO 96.1 4.5 6 70 130 4.7
BPA-D (surrogate) 91.1 3.2 8 70 130 2.7
Br-OP (surrogate) 87.9 4.7 8 70 130 3.9
n-NP (surrogate) 87.4 2.6 8 70 130 2.2
n-NP2EO (surrogate) 93.6 6.1 8 70 130 5.1
A
Uncertainty calculation based upon 95 % confidence interval and a two-tailed Student t distribution.
½
Uncertainty = Student t Value [(standard deviation) / (number of LCS) ].
D8310 − 20
TABLE 4 P&A Study for TPs in Ottawa Sand (Target Spike Recoveries)
P&A Data (625 µg/kg spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and 18 750 µg/kg for NP1EO)
Sample
BPA OP NP NP1EO NP2EO
MB1
MB2
P&A1 555.0 1020.0 1120.0 18 500.0 1150.0
P&A2 597.0 1070.0 1190.0 18 600.0 1160.0
P&A3 554.0 1160.0 1110.0 17 400.0 1160.0
P&A4 567.0 1010.0 1150.0 16 600.0 1200.0
P&A5 626.0 1180.0 1220.0 19 400.0 1280.0
P&A6 572.0 1190.0 1180.0 20 200.0 1260.0
Average Recovery, µg/kg 578.5 1105.0 1161.7 18 450.0 1201.7
% Average Recovery 92.6 88.4 92.9 98.4 96.1
Standard Deviation 28.0 81.7 42.6 1305.0 56.0
RSD, % 4.8 7.4 3.7 7.1 4.7
TABLE 5 P&A Study for TPs in Ottawa Sand (Surrogate Recoveries)
P&A Data (625 µg/kg Spike)
Sample
BPA-D Br-OP n-NP n-NP2EO
MB1 548 500 557 539
MB2 590 518 555 552
P&A1 538 567 569 564
P&A2 569 567 547 582
P&A3 579 556 515 566
P&A4 556 533 536 597
P&A5 583 581 553 637
P&A6 592 574 540 643
Average Recovery, µg/kg 569.4 549.5 546.5 585.0
% Average Recovery 91.1 87.9 87.4 93.6
Standard Deviation 20.1 29.2 16.4 38.2
RSD, % 3.5 5.3 3.0 6.5
TABLE 6 P&A Study in ASTM CL-1 Soil (Target Compound Recoveries)
P&A for ASTM CL-1 Soil (Lean Clay, Red Bucket) (625 µg/kg Spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and
Sample 18 750 µg/kg for NP1EO)
BPA OP NP NP1EO NP2EO
MB1
MB2
P&A1 553.0 1173.7 1231.1 17 936.0 1239.4
P&A2 586.3 1259.1 1300.3 19 901.6 1271.3
P&A3 564.7 1209.6 1188.7 18 313.8 1225.9
P&A4 587.3 1224.9 1226.7 17 870.8 1233.9
P&A5 609.7 1201.5 1239.1 17 757.6 1254.0
P&A6 553.6 1119.0 1179.7 18 965.0 1195.1
Mean Recovery (µg/kg dry weight) 575.8 1198.0 1227.6 18 457.5 1236.6
% Mean Recovery 92.1 95.8 98.2 98.4 98.9
Standard Deviation 22.5 47.8 43.0 833.0 25.9
RSD, % 3.9 4.0 3.5 4.5 2.1
TABLE 7 Surrogate Recoveries for P&A Study in ASTM CL-1 Soil
ASTM CL-1 Soil (Lean Clay, Red Bucket), 625 µg/kg Spike
Sample
BPA-D Br-OP n-NP n-NP2EO
MB1 627.5 571.5 602.3 603.2
MB2 599.6 535.9 618.2 592.2
P&A1 549.4 594.1 595.1 637.6
P&A2 628.8 608.5 579.3 669.0
P&A3 600.6 579.6 557.8 633.0
P&A4 605.5 597.4 569.7 645.5
P&A5 601.3 602.6 636.9 660.3
P&A6 589.8 601.7 589.8 622.5
Mean Recovery (µg/kg dry weight) 595.9 597.3 588.1 644.6
% Mean Recovery 95.3 95.6 94.1 103.1
Standard Deviation 26.2 10.0 27.4 17.4
RSD, % 4.4 1.7 4.7 2.7
glasswareisnotproperlycleanedandrinsedwithsolvent,such trations affecting the analysis may be found. All of these
as acetone, 2-propanol, and acetonitrile, and low µg/Lconcen-
D8310 − 20
TABLE 8 P&A Study in ASTM CH-1 Soil (Target Compound Recoveries)
P&A Data for ASTM CH-1 Soil (Fat Clay, White Bucket) (625 µg/kg Spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and
Sample 18 750 µg/kg for NP1EO)
BPA OP NP NP1EO NP2EO
MB1
MB2
P&A1 622.3 1245.6 1220.7 19 408.3 1272.0
P&A2 639.1 1173.9 1192.9 19 910.7 1267.6
P&A3 666.6 1236.3 1260.1 20 325.8 1301.7
P&A4 604.4 1178.0 1200.8 19 328.7 1232.3
P&A5 644.1 1164.7 1327.7 19 230.7 1325.0
P&A6 664.5 1338.0 1268.8 19 386.1 1350.5
Mean Recovery (µg/kg dry weight) 640.2 1222.7 1245.1 19 598.4 1291.5
% Mean Recovery 102.4 97.8 99.6 104.5 103.3
Standard Deviation 24.1 65.9 50.8 428.0 42.8
RSD, % 3.8 5.4 4.1 2.2 3.3
TABLE 9 Surrogate Recoveries for P&A Study in ASTM CH-1 Soil
ASTM CH-1 Soil (Fat Clay, White Bucket), 625 µg/kg Spike
Sample
BPA-D Br-OP n-NP n-NP2EO
MB1 645.2 537.0 603.7 571.1
MB2 635.6 572.0 645.5 591.9
P&A1 634.1 602.6 553.9 660.0
P&A2 609.7 593.7 569.1 644.4
P&A3 632.4 650.5 628.1 686.3
P&A4 623.4 590.2 577.3 631.9
P&A5 661.0 645.6 659.3 660.9
P&A6 672.2 649.6 686.4 702.2
Mean Recovery (µg/kg dry weight) 638.8 622.0 612.3 664.3
% Mean Recovery 102.2 99.5 98.0 106.3
Standard Deviation 23.5 29.4 53.8 26.0
RSD, % 3.7 4.7 8.8 3.9
TABLE 10 P&A Study in ASTM SP-1 Soil (Target Compound Recoveries)
P&A Data (625 µg/kg Spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and 18 750 µg/kg for NP1EO)
Sample
BPA OP NP NP1EO NP2EO
MB1
MB2
P&A1 611.5 1231.4 1205.7 17 023.3 1205.9
P&A2 631.0 1234.1 1252.3 18 426.6 1229.1
P&A3 574.3 1067.6 1140.7 17 788.8 1153.3
P&A4 626.9 1262.1 1211.8 18 010.4 1232.4
P&A5 586.7 1213.2 1200.2 17 549.9 1230.1
P&A6 578.4 1230.4 1184.4 16 480.0 1113.1
Mean Recovery (µg/kg dry weight) 601.5 1206.5 1199.2 17 546.5 1194.0
% Mean Recovery 96.2 96.5 95.9 93.6 95.5
Standard Deviation 24.9 69.8 36.5 701.4 49.7
RSD, % 4.1 5.8 3.0 4.0 4.2
TABLE 11 Surrogate Recoveries for P&A Study in ASTM SP-1 Soil
ASTM SP-1 Soil (Sand, Yellow Bucket), 625 µg/kg Spike
Sample
BPA-D Br-OP n-NP n-NP2EO
MB1 568.1 518.1 605.1 533.2
MB2 610.1 548.3 545.4 553.0
P&A1 629.8 589.7 567.6 622.4
P&A2 608.8 588.7 596.6 601.7
P&A3 543.8 534.5 513.0 594.4
P&A4 576.1 589.2 593.4 601.3
P&A5 559.4 616.7 590.1 629.7
P&A6 552.5 532.5 548.5 551.9
Mean Recovery (µg/kg dry weight) 578.4 575.2 568.2 600.2
% Mean Recovery 92.5 92.0 90.9 96.0
Standard Deviation 34.1 34.0 32.7 27.3
RSD, % 5.9 5.9 5.8 4.6
materials are routinely demonstrated to be free from interfer- conditions as the samples. If found, measures should be taken
ences by analyzing laboratory reagent blanks under the same to remove the contamination or data should be qualified.
D8310 − 20
TABLE 12 P&A Study in ASTM ML-1 Soil (Target Compound Recoveries)
P&A Data for ASTM ML-1 Soil (Silt, Green Bucket) (625 µg/kg Spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and
Sample 18 750 µg/kg for NP1EO)
BPA OP NP NP1EO NP2EO
MB1
MB2
P&A1 559.5 1117.4 1076.1 16 702.3 1124.2
P&A2 547.9 1179.6 1091.9 17 457.8 1106.4
P&A3 573.0 1148.2 1146.7 17 540.1 1152.7
P&A4 550.3 1193.2 1108.3 16 291.8 1111.0
P&A5 549.0 1151.9 1121.4 16 871.6 1131.9
P&A6 568.6 1121.2 1102.0 16 855.4 1139.1
Mean Recovery (µg/kg dry weight) 558.0 1151.9 1107.7 16 953.2 1127.6
% Mean Recovery 89.3 92.2 88.6 90.4 90.2
Standard Deviation 10.8 30.4 24.4 472.4 17.4
RSD, % 1.9 2.6 2.2 2.8 1.5
TABLE 13 Surrogate Recoveries for P&A Study in ASTM ML-1 Soil
ASTM ML-1 Soil (Silt, Green Bucket), 625 µg/kg Spike
Sample
BPA-D Br-OP n-NP n-NP2EO
MB1 555.6 479.8 513.4 522.6
MB2 504.3 499.8 492.9 525.8
P&A1 530.0 522.7 512.2 571.7
P&A2 518.3 538.0 516.1 563.2
P&A3 553.1 551.1 512.9 580.9
P&A4 527.9 519.3 532.0 548.6
P&A5 527.2 537.0 554.7 551.0
P&A6 551.5 522.6 524.5 588.0
Mean Recovery (µg/kg dry weight) 534.7 531.8 525.4 567.2
% Mean Recovery 85.5 85.1 84.1 90.8
Standard Deviation 14.2 12.4 16.2 15.9
RSD, % 2.7 2.3 3.1 2.8
TABLE 14 P&A Study in Fairfield Biosolid Samples (Target Compound Recoveries)
P&A Data for Fairfield Biosolid Samples (625 µg/kg Spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and 18 750 µg/kg
Sample for NP1EO)
A A
BPA OP NP NP1EO NP2EO
MB1
MB2
P&A1 457.7 821.8 1727.7 13 795.6 931.3
P&A2 514.1 889.4 1329.6 15 307.5 996.4
P&A3 496.8 1002.2 2243.0 15 135.0 1007.5
P&A4 496.0 1007.7 2148.3 15 158.7 1005.0
P&A5 471.7 1114.0 6930.9 14 673.7 982.7
P&A6 456.5 1018.1 3984.5 14 310.2 966.2
Mean Recovery (µg/kg dry weight) 482.1 975.5 3060.7 14 730.1 981.5
% Mean Recovery 77.1 78.0 244.9 78.6 78.5
Standard Deviation 23.6 103.7 2102.7 588.8 29.0
RSD, % 4.9 10.6 68.7 4.0 3.0
A
P&A values are after subtraction of average of MB if$RL.
6.3.3 Glass syringes are used with this test method. A 7.1.1 Liquid Chromatograph (LC) System—An ultra-
thoroughly cleaned 10 or 20-mL hypodermic glass syringe performance LC (UPLC) with flow-through needle design.
with a PVDF filter unit is used to filter samples and has been 7.1.2 Analytical Column—An analytical column that will
shown to perform well. achieve adequate results that meet or exceed this test method.
6.3.4 Nonylphenol was found in polypropylene. The filter 7.1.3 Mass Spectrometer (MS) System—Atriple quadrupole
units shall be rinsed with at least 10 mL of acetonitrile and mass spectrometer is used. A mass spectrometer capable of
10mL of methanol before use to remove nonylphenol. MRManalysiswithfastenoughcycletimetoobtainatleastten
6.3.5 The procedure described in 7.3.8 should be followed scans over a peak is needed with adequate sensitivity.
to make glassware free from interferences.Alkylphenol deter- 7.1.4 Data Backup Device—A data archival unit to archive
gents shall not be used. Detergent may containTPs, precursor, data.
or breakdown products and these shall be avoided.
AWaters Acquity UPLC® BEH C18, 2.1 × 100 mm and 1.7 µm particle size
7. Apparatus
was used to develop this test method. Any column may be used that meets or
7.1 Equipment: exceeds the performance of this test method.
D8310 − 20
TABLE 15 Surrogate Recoveries for P&A Study in Fairfield Biosolid Samples
Fairfield Biosolid Samples, 625 µg/kg Spike
Sample
BPA-D Br-OP n-NP n-NP2EO
MB1 447.1 387.8 176.8 431.0
MB2 466.9 417.6 214.5 449.7
P&A1 413.3 391.4 179.6 450.8
P&A2 432.4 420.8 190.2 486.9
P&A3 456.1 419.7 177.0 496.5
P&A4 460.1 449.1 195.1 507.0
P&A5 433.6 408.5 151.4 468.0
P&A6 436.2 425.1 165.8 492.2
Mean Recovery (µg/kg dry weight) 438.6 419.1 176.5 483.5
% Mean Recovery 70.2 67.1 28.2 77.4
Standard Deviation 17.2 19.1 16.1 20.6
RSD, % 3.9 4.6 9.1 4.3
TABLE 16 P&A Study in Commercial Soil (Milorganite)
P&A Data for Milorganite (625 µg/kg Spike for BPA; 1250 µg/kg for OP, NP, and NP2EO; and 18 750 µg ⁄kg for NP1EO)
Sample
A A A A A
BPA OP NP NP1EO NP2EO
MB1 109.7 194.2 3460.9 7875.8 4971.7
MB2 89.3 196.5 3512.1 7637.2 4587.9
P&A1 454.9 801.7 729.7 17 038.9 1832.0
P&A2 432.1 783.8 590.0 15 539.7 1452.2
P&A3 422.7 785.6 555.3 15 936.5 1453.7
P&A4 428.5 836.5 713.5 15 928.4 1550.2
P&A5 402.9 790.4 569.2 16 070.2 1306.2
P&A6 433.1 843.0 756.4 16 216.7 1669.0
Mean Recovery (µg/kg dry weight) 429.0 806.8 652.3 16 121.7 1543.9
% Mean Recovery 68.6 64.5 52.2 86.0 123.5
Standard Deviation 16.9 26.3 90.3 502.6 185.3
RSD, % 3.9 3.3 13.8 3.1 12.0
A
P&A values are after subtraction of average of MB if$RL.
TABLE 17 Surrogate Recoveries for Precision and Accuracy Study in Commercial Soil (Milorganite)
Milorganite, 625 µg/kg Spike
Sample
BPA-D Br-OP n-NP n-NP2EO
A
MB1 402.2 324.1
MB2 425.1 335.5
P&A1 431.0 341.7
P&A2 363.5 325.9
P&A3 405.8 330.3
P&A4 354.4 312.1
P&A5 371.3 309.4
P&A6 404.9 329.1
Mean Recovery (µg/kg dry weight) 388.5 324.8 No recovery 552.8
% Mean Recovery 62.2 52.0 NA 88.4
Standard Deviation 29.8 12.1 NA 7.2
RSD, % 7.7 3.7 NA 1.3
A
7.1.5 Data System—Software shall be interfaced to the 7.3.3 Class A volumetric glassware.
LC/MS/MS that allows the continuous acquisition and storage
7.3.4 VOA Vials—20, 40, or 60 mL.
on machine-readable media of all mass spectra obtained
7.3.5 GxF/0.2 µm PVDF Membrane Syringe-Driven Filter
throughout the duration of the chromatographic program.
Unit—The filters are washed with 10 mLacetonitrile followed
Software is used for all data quantitation.
by 10 mL methanol before use.
7.2 Calibrated Support Equipment:
7.3.6 Syringe—Use 10 to 25-mL filter-adaptable glass sy-
7.2.1 Graduated Syringes—Use 10, 25, 50, 100, 250, 500,
ringe with luer lock.
and 1000-µL graduated syringes.
7.3.7 Disposable glass pipettes. (Warning—Syringes shall
7.2.2 Analytical Balance (Certified Annually)—Use an ana-
be cleaned with copious amounts of warm tap water, distilled
lytical balance accurate to 60.1 % of sample mass with Class
water, and thoroughly rinsed with acetonitrile and methanol
3 or 4 weights to verify calibration.
between uses.)
7.3 Glassware and Miscellaneous Supplies: 7.3.8 Glassware Cleaning Instructions—All glassware is
7.3.1 Vials—Use 2-mL autosampler vials with pre-slit washedinhotwater(above50°C)withdetergent,rinsedinhot
PTFE/silicone septa or equivalent. water, and rinsed with distilled water. The glassware is then
7.3.2 Gases—Ultra-pure argon and nitrogen. dried in an oven for up to 1 h (except volumetric glassware,
D8310 − 20
which is air dried). All glassware is subsequently rinsed with – 8.3.4.4. The calibration levels (prepared from the high-level
an organic solvent such as acetone, methanol, and acetonitrile. calibration stock), laboratory control samples, matrix spike
(Warning—Detergents containing alkylphenolic compounds samples, and duplicates are made for each batch of samples.
shall not be used.) They are usually prepared and analyzed immediately. A CCC
standard is also prepared for each batch of samples.
8. Reagents and Materials
8.3.4.1 TP Surrogate Spiking Solution—A surrogate stan-
dard solution containing Br-OP, n-NP, BPA-D , and n-NP2EO
8.1 Purity of Reagents—Reagent-grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that is added to each 2-g soil sample. Br-OP and n-NP are used as
thesurrogatesforNPandOP,BPA-D isusedasasurrogatefor
all reagents conform to the specifications of the Committee on
Analytical Reagents of theAmerican Chemical Society, where BPA, and n-NP2EO is used as a surrogate for NP1EO and
NP2EO. A stock surrogate spiking solution is prepared in
such specifications are available. Other grades may be used,
provided it is first ascertained that the reagent is of sufficiently methanol at 12.5 mg/L for Br-OP, n-NP, BPA-D , and
n-NP2EO.The surrogates are added to each sample to achieve
high purity to permit its use without lessening the accuracy of
the determination. a concentration of 625 µg/L (that is, 100 µL of a 12.5-mg/L
methanol solution containing Br-OP, n-NP, BPA-D , and
8.2 Solvents and Reagents:
n-NP2EO is added to a 2-g soil sample).
8.2.1 Acetonitrile.
8.3.4.2 MS/MSD and LCS/LCSD Spiking Solution (Target
8.2.2 Water—HPLC mass spectrometry pesticide quality.
Spike Solution)—Each ~2-g sample, approximately 2 g and
8.2.2.1 Purity of Water—Unless otherwise indicated, refer-
weighed to the hundredths, MS/MSD or LCS/LCSD sample is
ences to water shall be understood to mean reagent water as
spiked with TPs containing BPA at 12.5 mg/L, NP, OP and
defined by Type I of Specification D1193.
NP2EO at 25 mg/L, and NP1EO at 375 mg/L to achieve a
8.2.3 Methanol.
concentration of 1250 µg/Lfor OP, NP, and NP2EO, 625 µg/L
8.2.4 Isopropyl alcohol.
for BPA, and 18750 µg/L for NP1EO (that is, 100 µL of a
8.2.5 Ammonium acetate.
methanol solution containing BPA at 12.5 mg/L, NP, OP, and
8.2.6 Acetone.
NP2EO at 25 mg/Land NP1EO at 375 mg/Lis added to a 2-g
8.2.7 Nonylphenol monoethoxylate.
soil sample).
8.2.8 Nonylphenol diethoxylate.
8.3.4.3 RL Check Spiking Solution—The RLCS is prepared
8.2.9 Nonylphenol.
by spiking the RLCS with the RL check standard near RL. A
8.2.10 Octylphenol.
50-µL volume of a methanolic RL check standard containing
8.2.11 n-Nonylphenol diethoxylate.
BPAat2mg/L;NP,OP,andNP2EOat4mg/L;andNP1EOat
8.2.12 n-Nonylphenol.
60 mg/L is added to RLCS to achieve a concentration of 50
8.2.13 2-bromo-4-(1,1,3,3-tetramethylbutyl)phenol.
µg/L for BPA; 100 µg/L for OP, NP, and NP2EO; and 1500
8.2.14 Bisphenol A.
µg/L for NP1EO.
8.2.15 Bisphenol A (propane-D6).
8.3.4.4 Calibration Standards—Calibration stock standard
8.3 Reagents and Standard Preparation:
SolutionAispreparedfromtargetspikingsolutionscontaining
8.3.1 All calibration standard preparations shall be re-
BPA at 12.5 mg/L; NP, OP, and NP2EO at 25 mg/L; and
corded.Allcalibrationsolutionsarepreparedwithacetone:wa-
NP1EOat375mg/Landsurrogatespikingsolutionscontaining
ter (75:25) unless otherwise stated. The concentrated stock
Br-OP, n-NP, BPA-D ; and n-NP2EO at 12.5 mg/L.Add 1000
standard concentration can vary when preparing from neat
µLofthetargetspikesolutionandsurrogatespikingsolutionto
material, usually between 50 to 100 mg/L of each TP in
a 25-mL volumetric flask and diluted to 25 mL volume with
methanol.
75:25 (acetone:water) (Solution B). Stock standard SolutionA
8.3.2 Expiration time of a prepared standard is six months
(Level8,Tables18and19)containingBPAandeachsurrogate
from the time prepared. The standards can be used for more
at 500µg⁄L; OP, NP, and NP2EO at 1000 µg/L; and NP1EO at
than six months if they fall within 630 % from the calibration
15000µg⁄Lis diluted to prepare Levels 1 through 7 as shown
standard that is less than six months old. Presently, there is no
in Tables 18 and 19. All calibration standards should contain
holding time study for this standard and the six months is an
75:25 acetone to water in 2-mL LC vials. The CCC is a
estimate with no laboratory data supporting this time period.
mid-level calibration standard.
8.3.3 Label all standards and verify the correct grade of
9. Hazards
solvents were used. Traceability of standards is established
using the manufacturer’s specifications provided at the time of
9.1 Users of this test method should operate a formal safety
purchase.
program.
8.3.4 The specific instructions for the preparation of stock
9.2 Warnings—Health Hazards—The toxicity and carcino-
standards, spiking solutions, and QC batch are listed in 8.3.4.1
genicityofeachreagentusedinthistestmethodhavenotbeen
preciselydefined;however,eachchemicalcompoundistreated
Reagent Chemicals, American Chemical
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...