ASTM E1251-17a
(Test Method)Standard Test Method for Analysis of Aluminum and Aluminum Alloys by Spark Atomic Emission Spectrometry
Standard Test Method for Analysis of Aluminum and Aluminum Alloys by Spark Atomic Emission Spectrometry
SIGNIFICANCE AND USE
5.1 The metallurgical properties of aluminum and its alloys are highly dependent on chemical composition. Precise and accurate analyses are essential to obtaining desired properties, meeting customer specifications, and helping to reduce scrap due to off-grade material.
5.2 This test method is applicable to chill cast specimens as defined in Practices E716 and can also be applied to other types of samples provided that suitable reference materials are available. Also, other sample forms can be melted and cast into a disk, using an appropriate mold, as described in Practices E716. However, it should be noted that some elements (for example, magnesium) readily form oxides, while some others (for example, sodium, lithium, calcium, and strontium) are volatile, and may be lost to varying degrees during the melting process.
SCOPE
1.1 This test method describes the analysis of aluminum and its alloys by spark-atomic emission spectrometry (Spark-AES). The aluminum specimen to be analyzed may be in the form of a chill cast disk, casting, foil, sheet, plate, extrusion, or some other wrought form or shape. The elements covered in the scope of this method are listed in the table below.
Element
Tested Mass Fraction Range
(Wt %)
Antimony
0.001 to 0.003
Arsenic
0.001 to 0.006
Beryllium
0.0004 to 0.24
Bismuth
0.03 to 0.6
Boron
0.0006 to 0.009
Calcium
0.0002 to –
Chromium
0.001 to 0.23
Cobalt
0.4 to –
Copper
0.001 to 5.5
Gallium
0.02 to –
Iron
0.2 to 0.5
Lead
0.04 to 0.6
Lithium
0.0003 to 2.1
Magnesium
0.03 to 5.4
Manganese
0.001 to 1.2
Nickel
0.005 to 2.6
Phosphorus
0.003 to –
Silicon
0.07 to 16
Sodium
0.003 to 0.02
Strontium
0.03 to –
Tin
0.03 to –
Titanium
0.001 to 0.12
Vanadium
0.002 to 0.022
Zinc
0.002 to 5.7
Zirconium
0.001 to 0.12
Note 1: The mass fraction ranges given in the above scope were established through cooperative testing (ILS) of selected reference materials. The range shown for each element does not demonstrate the actual usable analytical range for that element. The usable analytical range may be extended higher or lower based on individual instrument capability, spectral characteristics of the specific element wavelength being used, and the availability of appropriate reference materials.
Note 2: Mercury (Hg) is intentionally not included in the scope. Analysis of Hg in aluminum by Spark-AES is not recommended. Accurate analysis of Hg using this technique is compromised by the presence of an intense iron interference. Inaccurate reporting of Hg due to these interference effects can jeopardize the current designation of aluminum production as a mercury-free process. To demonstrate compliance with legislated Hg content limits, use of an alternate method capable of analysis with a minimum reporting limit of 0.0001% or lower is recommended. Suitable techniques include but are not limited to GD-MS, XRF (X-ray fluorescence), cold vapor AA, and ICP-MS.
1.2 This test method is suitable primarily for the analysis of chill cast disks as defined in Practices E716. Other forms may be analyzed, provided that: (1) they are sufficiently massive to prevent undue heating, (2) they allow machining to provide a clean, flat surface, which creates a seal between the specimen and the spark stand, and (3) reference materials of a similar metallurgical condition and chemical composition are available.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific safety and health statements are given in Section 10.
1.4 This international stand...
General Information
- Status
- Published
- Publication Date
- 30-Sep-2017
- Technical Committee
- E01 - Analytical Chemistry for Metals, Ores, and Related Materials
- Drafting Committee
- E01.04 - Aluminum and Magnesium
Relations
- Effective Date
- 01-Oct-2017
- Effective Date
- 01-Jan-2020
- Refers
ASTM E406-19 - Standard Practice for Using Controlled Atmospheres in Atomic Emission Spectrometry - Effective Date
- 01-Oct-2019
- Effective Date
- 15-May-2019
- Effective Date
- 01-Nov-2016
- Effective Date
- 15-May-2016
- Effective Date
- 01-Jul-2015
- Effective Date
- 15-May-2015
- Effective Date
- 15-Aug-2014
- Effective Date
- 01-Apr-2014
- Effective Date
- 15-Feb-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Dec-2012
Overview
ASTM E1251-17a is the established international standard for the analysis of aluminum and aluminum alloys by Spark Atomic Emission Spectrometry (Spark-AES). Developed by ASTM International, this test method is critical for accurately determining the chemical composition of aluminum products. From chill cast disks to wrought forms such as sheets, plates, extrusions, and castings, Spark-AES offers a robust analytical approach for the metals industry.
Precise analysis of aluminum alloys is vital because metallurgical properties and performance are highly influenced by the alloying elements present. This standard supports manufacturers and suppliers in achieving desired alloy characteristics, meeting customer specifications, and minimizing waste due to off-grade materials.
Key Topics
Scope of Application:
- Covers a broad range of aluminum and its alloys in forms such as chill cast disks, castings, sheets, plates, and extrusions.
- Suitable for analysis of elements like antimony, arsenic, beryllium, bismuth, boron, calcium, chromium, cobalt, copper, gallium, iron, lead, lithium, magnesium, manganese, nickel, phosphorus, silicon, sodium, strontium, tin, titanium, vanadium, zinc, and zirconium.
Analytical Procedure:
- Utilizes Spark-AES, where a controlled spark ablates the sample, and the emitted atomic spectra are analyzed for elemental composition.
- Calibration methods include binary-type, global-type, and alloy-type calibrations, enhancing flexibility and accuracy.
Sample Preparation:
- Recommends chill cast disks as per ASTM E716.
- Other forms can be analyzed if they are sufficiently massive and can be machined to fit the spark stand.
- Importance of using suitable reference materials and maintaining a freshly prepared surface for both sample and calibration standards.
Limitations:
- Mercury (Hg) analysis is not recommended due to interference and risk of inaccurate results.
- Special consideration is required for volatile elements (e.g., sodium, lithium, calcium, strontium) and those prone to forming oxides (e.g., magnesium) during sample preparation.
Applications
The practical value of ASTM E1251-17a spans several industries and scenarios:
Quality Control in Aluminum Production:
- Ensures products meet stringent chemical composition specifications required by automotive, aerospace, and construction sectors.
Supplier and Customer Assurance:
- Facilitates certification of material composition for procurement and supply chain management.
Process Optimization:
- Enables manufacturers to monitor and adjust processing parameters, reducing scrap, improving efficiency, and minimizing production costs.
Research and Development:
- Supports alloy development and innovation by providing reliable compositional data.
Regulatory Compliance:
- Helps demonstrate conformity to national and international standards, critical for product acceptance in global markets.
Related Standards
To effectively implement ASTM E1251-17a, users should be familiar with related documents, including:
- ASTM E716: Practices for sampling and sample preparation of aluminum and aluminum alloys for chemical composition determination by Spark-AES
- ASTM E135: Terminology relating to analytical chemistry for metals, ores, and related materials
- ASTM E305: Practice for establishing and controlling atomic emission spectrochemical analytical curves
- ASTM B985: Practice for sampling aluminum ingots, billets, castings, and finished or semi-finished wrought aluminum products for compositional analysis
- ANSI H35.1/H35.1M: American National Standard Alloy and Temper Designation Systems for Aluminum
ASTM E1251-17a is indispensable for modern aluminum analysis labs aiming for repeatable, accurate results and compliance with international best practices. By following its guidelines, organizations can enhance quality assurance, meet customer expectations, and maintain competitiveness in the global aluminum market.
Buy Documents
ASTM E1251-17a - Standard Test Method for Analysis of Aluminum and Aluminum Alloys by Spark Atomic Emission Spectrometry
REDLINE ASTM E1251-17a - Standard Test Method for Analysis of Aluminum and Aluminum Alloys by Spark Atomic Emission Spectrometry
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM E1251-17a is a standard published by ASTM International. Its full title is "Standard Test Method for Analysis of Aluminum and Aluminum Alloys by Spark Atomic Emission Spectrometry". This standard covers: SIGNIFICANCE AND USE 5.1 The metallurgical properties of aluminum and its alloys are highly dependent on chemical composition. Precise and accurate analyses are essential to obtaining desired properties, meeting customer specifications, and helping to reduce scrap due to off-grade material. 5.2 This test method is applicable to chill cast specimens as defined in Practices E716 and can also be applied to other types of samples provided that suitable reference materials are available. Also, other sample forms can be melted and cast into a disk, using an appropriate mold, as described in Practices E716. However, it should be noted that some elements (for example, magnesium) readily form oxides, while some others (for example, sodium, lithium, calcium, and strontium) are volatile, and may be lost to varying degrees during the melting process. SCOPE 1.1 This test method describes the analysis of aluminum and its alloys by spark-atomic emission spectrometry (Spark-AES). The aluminum specimen to be analyzed may be in the form of a chill cast disk, casting, foil, sheet, plate, extrusion, or some other wrought form or shape. The elements covered in the scope of this method are listed in the table below. Element Tested Mass Fraction Range (Wt %) Antimony 0.001 to 0.003 Arsenic 0.001 to 0.006 Beryllium 0.0004 to 0.24 Bismuth 0.03 to 0.6 Boron 0.0006 to 0.009 Calcium 0.0002 to – Chromium 0.001 to 0.23 Cobalt 0.4 to – Copper 0.001 to 5.5 Gallium 0.02 to – Iron 0.2 to 0.5 Lead 0.04 to 0.6 Lithium 0.0003 to 2.1 Magnesium 0.03 to 5.4 Manganese 0.001 to 1.2 Nickel 0.005 to 2.6 Phosphorus 0.003 to – Silicon 0.07 to 16 Sodium 0.003 to 0.02 Strontium 0.03 to – Tin 0.03 to – Titanium 0.001 to 0.12 Vanadium 0.002 to 0.022 Zinc 0.002 to 5.7 Zirconium 0.001 to 0.12 Note 1: The mass fraction ranges given in the above scope were established through cooperative testing (ILS) of selected reference materials. The range shown for each element does not demonstrate the actual usable analytical range for that element. The usable analytical range may be extended higher or lower based on individual instrument capability, spectral characteristics of the specific element wavelength being used, and the availability of appropriate reference materials. Note 2: Mercury (Hg) is intentionally not included in the scope. Analysis of Hg in aluminum by Spark-AES is not recommended. Accurate analysis of Hg using this technique is compromised by the presence of an intense iron interference. Inaccurate reporting of Hg due to these interference effects can jeopardize the current designation of aluminum production as a mercury-free process. To demonstrate compliance with legislated Hg content limits, use of an alternate method capable of analysis with a minimum reporting limit of 0.0001% or lower is recommended. Suitable techniques include but are not limited to GD-MS, XRF (X-ray fluorescence), cold vapor AA, and ICP-MS. 1.2 This test method is suitable primarily for the analysis of chill cast disks as defined in Practices E716. Other forms may be analyzed, provided that: (1) they are sufficiently massive to prevent undue heating, (2) they allow machining to provide a clean, flat surface, which creates a seal between the specimen and the spark stand, and (3) reference materials of a similar metallurgical condition and chemical composition are available. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific safety and health statements are given in Section 10. 1.4 This international stand...
SIGNIFICANCE AND USE 5.1 The metallurgical properties of aluminum and its alloys are highly dependent on chemical composition. Precise and accurate analyses are essential to obtaining desired properties, meeting customer specifications, and helping to reduce scrap due to off-grade material. 5.2 This test method is applicable to chill cast specimens as defined in Practices E716 and can also be applied to other types of samples provided that suitable reference materials are available. Also, other sample forms can be melted and cast into a disk, using an appropriate mold, as described in Practices E716. However, it should be noted that some elements (for example, magnesium) readily form oxides, while some others (for example, sodium, lithium, calcium, and strontium) are volatile, and may be lost to varying degrees during the melting process. SCOPE 1.1 This test method describes the analysis of aluminum and its alloys by spark-atomic emission spectrometry (Spark-AES). The aluminum specimen to be analyzed may be in the form of a chill cast disk, casting, foil, sheet, plate, extrusion, or some other wrought form or shape. The elements covered in the scope of this method are listed in the table below. Element Tested Mass Fraction Range (Wt %) Antimony 0.001 to 0.003 Arsenic 0.001 to 0.006 Beryllium 0.0004 to 0.24 Bismuth 0.03 to 0.6 Boron 0.0006 to 0.009 Calcium 0.0002 to – Chromium 0.001 to 0.23 Cobalt 0.4 to – Copper 0.001 to 5.5 Gallium 0.02 to – Iron 0.2 to 0.5 Lead 0.04 to 0.6 Lithium 0.0003 to 2.1 Magnesium 0.03 to 5.4 Manganese 0.001 to 1.2 Nickel 0.005 to 2.6 Phosphorus 0.003 to – Silicon 0.07 to 16 Sodium 0.003 to 0.02 Strontium 0.03 to – Tin 0.03 to – Titanium 0.001 to 0.12 Vanadium 0.002 to 0.022 Zinc 0.002 to 5.7 Zirconium 0.001 to 0.12 Note 1: The mass fraction ranges given in the above scope were established through cooperative testing (ILS) of selected reference materials. The range shown for each element does not demonstrate the actual usable analytical range for that element. The usable analytical range may be extended higher or lower based on individual instrument capability, spectral characteristics of the specific element wavelength being used, and the availability of appropriate reference materials. Note 2: Mercury (Hg) is intentionally not included in the scope. Analysis of Hg in aluminum by Spark-AES is not recommended. Accurate analysis of Hg using this technique is compromised by the presence of an intense iron interference. Inaccurate reporting of Hg due to these interference effects can jeopardize the current designation of aluminum production as a mercury-free process. To demonstrate compliance with legislated Hg content limits, use of an alternate method capable of analysis with a minimum reporting limit of 0.0001% or lower is recommended. Suitable techniques include but are not limited to GD-MS, XRF (X-ray fluorescence), cold vapor AA, and ICP-MS. 1.2 This test method is suitable primarily for the analysis of chill cast disks as defined in Practices E716. Other forms may be analyzed, provided that: (1) they are sufficiently massive to prevent undue heating, (2) they allow machining to provide a clean, flat surface, which creates a seal between the specimen and the spark stand, and (3) reference materials of a similar metallurgical condition and chemical composition are available. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific safety and health statements are given in Section 10. 1.4 This international stand...
ASTM E1251-17a is classified under the following ICS (International Classification for Standards) categories: 77.120.10 - Aluminium and aluminium alloys. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E1251-17a has the following relationships with other standards: It is inter standard links to ASTM E1251-17, ASTM E135-20, ASTM E406-19, ASTM E135-19, ASTM B985-12(2016), ASTM E135-16, ASTM E135-15a, ASTM E135-15, ASTM E135-14b, ASTM E135-14a, ASTM E135-14, ASTM E135-13a, ASTM E691-13, ASTM E826-08(2013), ASTM B985-12. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E1251-17a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E1251 − 17a
Standard Test Method for
Analysis of Aluminum and Aluminum Alloys by Spark
Atomic Emission Spectrometry
This standard is issued under the fixed designation E1251; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
AnalysisofHginaluminumbySpark-AESisnotrecommended.Accurate
1. Scope
analysis of Hg using this technique is compromised by the presence of an
1.1 Thistestmethoddescribestheanalysisofaluminumand
intense iron interference. Inaccurate reporting of Hg due to these inter-
itsalloysbyspark-atomicemissionspectrometry(Spark-AES). ference effects can jeopardize the current designation of aluminum
production as a mercury-free process. To demonstrate compliance with
The aluminum specimen to be analyzed may be in the form of
legislatedHgcontentlimits,useofanalternatemethodcapableofanalysis
a chill cast disk, casting, foil, sheet, plate, extrusion, or some
with a minimum reporting limit of 0.0001% or lower is recommended.
other wrought form or shape. The elements covered in the
Suitable techniques include but are not limited to GD-MS, XRF (X-ray
scope of this method are listed in the table below.
fluorescence), cold vapor AA, and ICP-MS.
Tested Mass Fraction Range
1.2 This test method is suitable primarily for the analysis of
Element
(Wt %)
chill cast disks as defined in Practices E716. Other forms may
Antimony 0.001 to 0.003
Arsenic 0.001 to 0.006 be analyzed, provided that: (1) they are sufficiently massive to
Beryllium 0.0004 to 0.24
prevent undue heating, (2) they allow machining to provide a
Bismuth 0.03 to 0.6
clean, flat surface, which creates a seal between the specimen
Boron 0.0006 to 0.009
Calcium 0.0002 to –
and the spark stand, and (3) reference materials of a similar
Chromium 0.001 to 0.23
metallurgical condition and chemical composition are avail-
Cobalt 0.4 to –
able.
Copper 0.001 to 5.5
Gallium 0.02 to –
1.3 This standard does not purport to address all of the
Iron 0.2 to 0.5
safety concerns, if any, associated with its use. It is the
Lead 0.04 to 0.6
Lithium 0.0003 to 2.1
responsibility of the user of this standard to establish appro-
Magnesium 0.03 to 5.4
priate safety, health, and environmental practices and deter-
Manganese 0.001 to 1.2
mine the applicability of regulatory limitations prior to use.
Nickel 0.005 to 2.6
Phosphorus 0.003 to –
Specific safety and health statements are given in Section 10.
Silicon 0.07 to 16
1.4 This international standard was developed in accor-
Sodium 0.003 to 0.02
dance with internationally recognized principles on standard-
Strontium 0.03 to –
Tin 0.03to–
ization established in the Decision on Principles for the
Titanium 0.001 to 0.12
Development of International Standards, Guides and Recom-
Vanadium 0.002 to 0.022
mendations issued by the World Trade Organization Technical
Zinc 0.002 to 5.7
Zirconium 0.001 to 0.12
Barriers to Trade (TBT) Committee.
NOTE 1—The mass fraction ranges given in the above scope were
2. Referenced Documents
established through cooperative testing (ILS) of selected reference mate-
rials. The range shown for each element does not demonstrate the actual
2.1 ASTM Standards:
usable analytical range for that element. The usable analytical range may
B985Practice for SamplingAluminum Ingots, Billets, Cast-
be extended higher or lower based on individual instrument capability,
spectralcharacteristicsofthespecificelementwavelengthbeingused,and ings and Finished or Semi-Finished Wrought Aluminum
the availability of appropriate reference materials.
Products for Compositional Analysis
NOTE 2—Mercury (Hg) is intentionally not included in the scope.
E29Practice for Using Significant Digits in Test Data to
Determine Conformance with Specifications
This test method is under the jurisdiction of ASTM Committee E01 on
Analytical Chemistry for Metals, Ores, and Related Materials and is the direct
responsibility of Subcommittee E01.04 on Aluminum and Magnesium. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Oct. 1, 2017. Published October 2017. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1988. Last previous edition approved in 2017 as E1251–17. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/E1251-17A. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1251 − 17a
E135Terminology Relating to Analytical Chemistry for izationmaterialsatorclosetothenominalcompositionforthe
Metals, Ores, and Related Materials particular alloy being analyzed. For best results, the standard-
E158Practice for Fundamental Calculations to Convert ization material being used should be within 610% of the
Intensities into Concentrations in Optical Emission Spec- composition(foreachrespectiveelement)ofthematerialbeing
trochemical Analysis (Withdrawn 2004) analyzed.
E172Practice for Describing and Specifying the Excitation
SourceinEmissionSpectrochemicalAnalysis(Withdrawn
4. Summary of Test Method
2001)
4.1 Acontrolledelectricaldischargeisproducedinanargon
E305Practice for Establishing and Controlling Atomic
atmosphere between the prepared flat surface of a specimen
Emission Spectrochemical Analytical Curves
and the tip of a semi-permanent counter electrode. The energy
E406Practice for Using Controlled Atmospheres in Spec-
ofthedischargeissufficienttoablatematerialfromthesurface
trochemical Analysis
ofthesample,breakthechemicalorphysicalbonds,andcause
E691Practice for Conducting an Interlaboratory Study to
the resulting atoms or ions to emit radiant energy. The radiant
Determine the Precision of a Test Method
energies of the selected analytical lines and the internal
E716Practices for Sampling and Sample Preparation of
standard line(s) are converted into electrical signals by either
Aluminum and Aluminum Alloys for Determination of
photomultiplier tubes (PMTs) or a suitable solid state detector.
Chemical Composition by Spark Atomic Emission Spec-
Thedetectorsignalsareelectricallyintegratedandconvertedto
trometry
a digitized value. The signals are ratioed to the proper internal
E826Practice for Testing Homogeneity of a Metal Lot or
standard signal and converted into mass fractions in accor-
Batch in Solid Form by Spark Atomic Emission Spec-
dance with Practice E158.
trometry
4.2 Three different methods of calibration defined in 3.2.2,
E1329PracticeforVerificationandUseofControlChartsin
3.2.3, and 3.2.1 are capable of giving the same precision,
Spectrochemical Analysis
accuracy, and detection limit.
E1507Guide for Describing and Specifying the Spectrom-
4.2.1 The first method, binary calibration, employs calibra-
eter of an Optical Emission Direct-Reading Instrument
tion curves that are determined using a large number of
2.2 ANSI Standard:
high-purity binary calibration materials. This approach is used
ANSI H35.1/H35.1MAmerican National Standard Alloy
when there is a need to analyze almost the entire range of
and Temper Designation Systems for Aluminum
aluminum alloys. Because binary calibration materials may
respond differently from alloy calibration materials, the latter
3. Terminology
are used to improve accuracy by applying a slope and/or
3.1 Definitions—For definitions of terms used in this
intercept correction to the observed readings.
Standard, refer to Terminology E135.
4.2.2 The second method, global calibration, employs cali-
3.2 Definitions of Terms Specific to This Standard:
bration curves that are determined using many different alloy
3.2.1 alloy-type calibration—calibration curves determined
calibration materials with a wide variety of compositions.
using calibration materials from alloys with similar composi-
Mathematical calculations are used to correct for both alloy
tions.
difference and inter-element effects. Like the method above,
3.2.2 binary-type calibration—calibration curves deter- specific alloy calibration materials may be used to apply a
minedusingbinarycalibrationmaterials(primaryaluminumto slope and/or intercept correction to the observed readings.
which has been added one specific element).
4.2.3 The third method, alloy calibration, employs calibra-
tioncurvesthataredeterminedusingdifferentalloycalibration
3.2.3 global-type calibration—calibration curves deter-
materials that have similar compositions.Again, specific alloy
mined using calibration materials from many different alloys
calibration materials may be used to apply a slope and/or
with considerable compositional differences.
intercept correction to the observed readings.
3.2.4 two-point drift correction—the practice of analyzing a
high and low standardization materials for each calibration
5. Significance and Use
curve and adjusting the intensities obtained back to the values
obtained on those particular standardization materials during 5.1 The metallurgical properties of aluminum and its alloys
the collection of the calibration data. The corrections are are highly dependent on chemical composition. Precise and
accomplishedmathematicallyandareappliedtoboththeslope accurate analyses are essential to obtaining desired properties,
and intercept. Improved precision may be obtained by using a meeting customer specifications, and helping to reduce scrap
multi-point drift correction as described in Practice E1329. due to off-grade material.
3.2.5 type standardization—mathematical adjustment of the
5.2 This test method is applicable to chill cast specimens as
calibration curve’s slope or intercept using a single standard-
definedinPracticesE716andcanalsobeappliedtoothertypes
of samples provided that suitable reference materials are
available.Also,othersampleformscanbemeltedandcastinto
The last approved version of this historical standard is referenced on
a disk, using an appropriate mold, as described in Practices
www.astm.org.
E716. However, it should be noted that some elements (for
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
4th Floor, New York, NY 10036, http://www.ansi.org. example, magnesium) readily form oxides, while some others
E1251 − 17a
(for example, sodium, lithium, calcium, and strontium) are excitation cycle. The excitation chamber will also be con-
volatile,andmaybelosttovaryingdegreesduringthemelting structed so that it is flushed automatically with argon gas
process. during the analytical burn cycle. The excitation chamber’s
design should allow for a flow of argon gas to prevent the
6. Recommended Analytical Lines and Potential
deposition of ablated metal dust on the inner-chamber win-
Interferences
dow(s). The excitation chamber will be equipped with an
6.1 Table 1 lists the analytical lines commonly used for
exhaustsystemthatwillsafelydisposeoftheargongasandthe
aluminum analysis. Other lines may be used if they give
metal dust created during the excitation cycle. For reasons of
comparableresults.Alsolistedarerecommendedmassfraction
health and cleanliness, the exhausted gas and dust should not
ranges,backgroundequivalentmassfractions,detectionlimits,
be vented directly into the laboratory. To help with this
useful linear ranges, and potential interferences. The values
situation, manufacturers have designed their instruments with
given in this table are typical; actual values obtained are
some type of exhaust/filter system tominimize this problem.
dependent on instrument design.
The exhaust can then be vented into an efficient hood system.
NOTE3—Thebackgroundequivalentmassfractionanddetectionlimits
7.4 Gas Flow System, will be designed so that it can deliver
listed in Table 1 have been attained with a spectrometer that has a
pure argon gas to the excitation chamber. The purity of the
reciprocal dispersion of 54 nm/mm and a working resolution of 3.5 nm,
using an entrance slit width of 25 µm and exit slit widths of 50 µm. argon gas will affect the precision of the results. Generally,
precision improves as the purity of the argon gas gets higher.
7. Apparatus
Argongaswithaminimumpurityof99.995%hasbeenfound
7.1 Specimen Preparation Equipment:
to be acceptable. The gas shall be delivered by a flow system
7.1.1 Sampling Molds, for aluminum and the techniques of
as described in Practice E406. The argon gas source can be
pouring a sample disk are described in Practices E716. Chill
from high-purity compressed gas cylinders, a cryogenic-type
cast samples, poured and cast as described within Practices
cylinder that contains liquid argon or possibly from a central
E716, shall be the recommended form in this test method.
supply (liquid only). It is essential that only argon gas meeting
7.1.2 Lathe, capable of machining a smooth, flat surface on
theminimumpurityof99.995%beused.Alowerpuritygrade
the reference materials and samples.Avariable speed cutter, a
of argon, such as a “welding grade,” should not be used. The
cemented carbide or polycrystalline diamond tool bit, and an
delivery system shall be composed of a two-stage type (high/
automaticcrossfeedarehighlyrecommended.Properdepthof
low pressure) regulator of all-metal construction with two
cut and desired surface finish are described in Practices E716.
pressuregages.Deliverytubingmustnotproduceanycontami-
7.1.3 Milling Machine, a milling machine can be used as an
nation of the argon stream. Refrigerator-grade copper tubing is
alternative to a lathe.
recommended. The gages on the regulator will allow for the
7.1.4 It is strongly recommended that the same preparation
adjustment of the gas pressure to the instrument. Delivery
machineryusedtopreparethestandardsisalsousedtoprepare
pressurespecificationswillvarywithinstrumentmanufacturer.
the samples. Differences in surface characteristics may influ-
Please note that the delivery tube connections should be made
ence the analysis.
withallmetalsealsandthedeliverytubingitselfshouldbekept
as short as possible. (All metal connections are strongly
7.2 Excitation Source—In today’s instrumentation, the ex-
recommended because the discharge is adversely affected by
citation source is computer controlled and is normally pro-
organic contamination, or by as little as 2 ppm of oxygen or a
grammed to produce: (1) a high-energy pre-spark (of some
few ppm of water vapor.) Argon supply shall be sufficient to
preset duration), (2) a spark-type discharge (of some preset
support required flow during analysis and bleed during idle
duration), (3) an arc-type discharge (of some preset duration),
periods. All connections must be leak-free.
and (4) a spark-type discharge, during which, time-resolved
measurements are made for improved detection limits (this
7.5 Spectrometer—For details on specifying the spectrom-
may be optional on some instruments).
eter please refer to Guide E1507.
7.2.1 Typical parameters and times are given in Table 2.It
7.6 Measuring and Control System of the instrument con-
should be emphasized that the information presented is given
sists of either photomultiplier and integrating electronics or
as an example only and parameters may vary with respect to
solid-state photosensitive arrays (CCD or CID) that convert
instrument model and manufacturer. For details on describing
observed light intensities to a digitizable signal. A dedicated
and specifying an excitation source, please refer to Practice
computer and/or microprocessor is used to control burn
E172.
conditions, source operation, data acquisition, and the conver-
7.3 Excitation Chamber, shall be designed with an upper
sion of intensity data to mass fractions. Data should be
plate that is smooth and flat so that it will mate (seal) perfectly
accessibletotheoperatorthroughoutallstepsofthecalculation
withthepreparedsurfaceofthesamplespecimen.Thesealthat
process. The instrument’s control software should include
is formed between the two will exclude atmospheric oxygen
functions for routine instrument drift correction
from entering the discharge chamber. The excitation chamber
(standardization), type standardization, and the application of
will contain a mounting clamp to hold the counter electrode.
these functions to subsequent analyses.
The excitation stand assembly will also have some type of
8. Materials
clamp or device designed to hold the sample firmly against the
topplate.Somemanufacturersmayprovideforthetopplateto 8.1 Counter Electrode—The counter electrode and speci-
be liquid cooled to minimize sample heating during the mensurfacearethetwoterminuspointsofthesparkdischarge.
E1251 − 17a
TABLE 1 Recommended Analytical Lines
Wavelength Recommended Background Calculated High
Interferences
Element in Air Mass Fraction Equivalent, Detection Mass Fraction
F
Element, λ(nm) and k, %
A B C,D E
(nm) Range, % % Limit, % Index, %
Aluminum 256.799 I 70-100
266.039 I 70-100
237.208 I 70-100
Antimony 231.147 I 0.001-0.5 0.17 0.0002 Co 231.166 0.6
259.806 I 0.001-0.5 0.0002 Fe 259.837
Mn 259.817 0.01
Arsenic 234.984 I 0.005-0.1
Beryllium 234.861 I 0.0001-0.05 0.001 0.00003
313.042 II 0.0001-0.05 0.0035 0.00001
332.134 I 0.0001-0.05 0.00001
Bismuth 306.772 I 0.001-0.7 0.04 0.0002
Boron 249.773 I 0.0001-0.05 0.002 0.0001* Fe 249.782 0.001
Mn 249.778 0.007
249.678 I 0.0001-0.05
208.959 I 0.0001-0.05 Mo 208.952 0.13
Cadmium 228.802 I 0.001-1 0.05 <0.0001 As 228.812
479.992 I 0.005-2 0.15 0.003
G
Calcium 393.367 II 0.001-0.05 0.001 0.00005 Fe 393.361
Chromium 425.435 I 0.001-1 0.015 <0.0001
267.716 II 0.001-1 0.004 0.0005*
G
276.654 II 0.005-1
Cobalt 345.351 I 0.0001-2 <0.0001
Copper 327.396 I 0.001-1.5 0.005 <0.0001 0.7
324.754 I 0.001-0.5
296.117 I 0.05-20 0.40 0.01* >20 Fe 296.128
224.700 II 0.01-5 0.03 0.0005* 5
510.554 I 0.05-20 0.32 0.01* >20
Gallium 294.364 I 0.001-0.05 0.015 <0.0001
G
417.206 I 0.001-0.05 Fe 417.213
Ti 417.190
Cr 417.167
Iron 238.204 II 0.001-1.5 0.015 0.0008 1.0
259.940 II 0.001-1.5 0.005 0.0004
259.957 I
273.955 II 0.01-3.5
G
374.949 I 0.001-3.5 0.0001
441.512 I 0.01-3.5 0.0004
438.355 I 0.005-3.5
Lead 405.782 I 0.002-0.7 0.04 0.0001 Mn 405.792 0.01
Mg 405.763 0.001
283.306 I 0.002-0.7 0.07 0.002
Lithium 610.364 I 0.0001-3
670.784 I 0.0001-0.02 0.0005 <0.0005
323.261 I 0.01-3 Fe 323.279
Sb 323.250
Magnesium 279.553 II 0.0005-0.3 0.0006 0.00003 0.04
285.213 I 0.0005-0.3 0.008 <0.0001 0.25
277.669 I 0.05-11 0.08 0.01 >11
G
383.231 I 0.01-11 0.015 0.002* >11
383.826 I 0.1-11
518.362 I 0.01-11 0.02 0.002* >11
G
Manganese 403.076 I 0.001-0.1 0.028 0.0001*
259.373 II 0.0005-0.5 0.004 0.00005 0.2
293.306 II 0.001-2 0.006 0.0002* >1.1
346.033 II 0.01-2
Nickel 341.476 I 0.001-2 0.02 <0.0001 >2.5 Zr 341.466 0.01
310.188 I 0.005-4 0.05 0.001* >5
231.604 II 0.001-2 0.015 0.0005* <2.5
H
Phosphorus 178.231 I 0.0001-0.1 0.084 0.0001
Silicon 288.158 I 0.001-1.5 0.01 0.0001 1.5 Cr 288.123
251.612 I 0.001-1.5 0.006 0.0001 1.5
G
390.553 I 0.05-24 0.25 0.01 >24 Cr 390.566 0.09
212.415 I 0.05-24 0.5 0.05 >24
Silver 328.068 I 0.0005-0.1
338.289 I 0.0001-0.1 >10
466.848 I 0.05-1.5
Sodium 588.995 I 0.0001-0.05 0.0015 <0.0001
G
Strontium 421.552 II 0.0001-0.1 0.0004 0.0001
460.733 I 0.0005-0.06
Tin 317.502 I 0.001-7.5 0.04 0.0001 >10
Titanium 334.904 II 0.0005-0.5 0.004 <0.0001
337.280 II 0.001-0.5 0.002 <0.00010
363.545 I 0.0005-0.05 0.030 0.003*
E1251 − 17a
TABLE 1 Continued
Wavelength Recommended Background Calculated High
Interferences
Element in Air Mass Fraction Equivalent, Detection Mass Fraction
F
Element, λ(nm) and k, %
A B C,D E
(nm) Range, % % Limit, % Index, %
Vanadium 318.341 I 0.001-0.15 0.06 0.0003*
437.924 I 0.001-0.25
310.230 II 0.001-0.15 0.014 <0.0001
Zinc 213.856 I 0.0005-0.1 0.035 0.0001* 0.05
334.502 I 0.001-10.0 0.065 0.0004 >8
481.053 I 0.01-10 0.07 0.001* >10
472.216 I 0.01-10 0.26 0.0015 >10
Zirconium 339.198 II 0.001-1 0.02 0.001*
G
349.621 II 0.001-1 0.006 <0.0001
A
I = atom line, II = ion line.
B
Background Equivalent Mass Fraction—The mass fraction at which the signal due to the element is equal to the signal due to the background.
C
In this test method, the calculated detection limit was measured by calculating the standard deviation of ten consecutive burns on a specimen with element mass
fraction(s) at levels below ten times the expected detection limit.
D
See footnote C. For values marked with an asterisk (*) the available data were for a mass fraction greater than ten (10) times but less than a hundred (100) times the
expected detection limit.
E
High Mass Fraction Index—The mass fraction at which the slope of the calibration curve drops below 0.75.
F
InterferenceFactor,k—The apparent increase in the mass fraction of the element being determined, expressed in percent, due to the presence of 1.0 % of the interfering
element.
G
Useful analytical lines with improved signal to background ratios due to the complete removal of C-N background by the argon atmosphere.
H
If phosphorus is to be determined, the most sensitive line appears to be the 178.231 nm in the second order which requires either a vacuum or a gas filled spectrometer.
The vacuum spectrometer should be operated at a pressure of 25 microtorr or less. The gas filled spectrometer will be charged with nitrogen to a positive pressure of slightly
over one atmosphere (101 k pa). Optimum results are obtained by using a background channel that has been profiled “off peak” of the first order 178.231 nm phosphorus
line as the internal standard. The ratio of P 178.231 nm (2nd) / background near the 178.231 nm (1st) is plotted against % phosphorus. Even with this compensation for
variability in background, alloys with highly different compositions of major alloying elements, particularly silicon, require separate reference materials and analytical curves.
TABLE 2 Typical Excitation Source Electrical Parameters
9.1.1 It is recommended that a calibration curve for any
High Energy particular element be composed of a minimum of four calibra-
Parameter Spark Arc
Pre-spark
tionmaterials.Themassfractionsofthesecalibrationmaterials
Resistance, Ω 11 15
should be fairly evenly spaced over the calibrated analytical
Inductance, µH 30 130 30
range so that a mathematically valid calibration curve can be
Volts, V 400 400 400
Frequency, Hz 300 300 300 established using all of the points.
Capacitance, µF 12 3 5
9.1.1.1 The calibration materials used shall be of sufficient
Time, s 10 5 5
quality, purchased from a recognized reputable source, and
have certified values to the required accuracy for the antici-
pated analytical tasks to be performed. A few SRMs are
available from the National Institute of Standards and Tech-
The counter electrode should be made from thoriated tungsten
nology (NIST). Also, there are other commercial sources for
or other suitable material and have a pointed end. The gap
aluminum reference materials.
distance between the specimen surface and the tip of the
9.1.2 For trace elements, reference materials that contain
counter electrode is specified by the manufacturer. The diam-
variablemassfractionsofthetraceelementinatypicalalloyof
eter and geometry of the counter electrode is also application
constant or nearly constant composition are available. These
and manufacturer dependent. If different designs and/or con-
reference materials can be used for establishing the analytical
figurations are offered, it is recommended that the prospective
curve, but will not reveal potential interferences from nearby
purchaser test each design to determine which performs the
lines of other elements, or matrix effects that change instru-
best for the intended analytical task. The counter electrode
mentresponseorbackground.Foroptimumusefulness,several
configuration and auxiliary gap distance must not be altered
of the calibration materials should have mass fractions for the
subsequent to spectrometer calibration or calibration adjust-
other elements that vary over the expected ranges in the
ments. Electrode maintenance (frequent brushing of the coun-
specimen to be analyzed.
ter electrode) to maintain its configuration, gap distance, and
9.1.3 Atomic emission analysis is a comparative technique
minimizesurfacecontaminationarecriticaltoaccurate,precise
that requires a close match of the metallurgy, structure, and
analytical results. It is recommended that the instrument
composition between the reference material and the test
purchaser order several spare counter electrodes so that they
material. Differences in structure, such as result from the
can be replaced when necessary.
sodium modification of high silicon alloys, or differences in
9. Reference Materials
metallurgicalhistory,duetoextruding,rolling,orheattreating,
induce a variety of effects that can influence the analytical
9.1 Calibration Materials—All calibration materials shall
results. To ensure analytical accuracy, care must be taken to
behomogeneousandfreeofcracksorporosity.Thesematerials
matchthecharacteristicsofthereferencematerialtothatofthe
should also possess a metallurgical condition that is similar to
test material or suitable corrections to adjust for these influ-
the material(s) that are being analyzed. The calibration mate-
ences must be established.
rials shall be used to produce the analytical curves f
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E1251 − 17 E1251 − 17a
Standard Test Method for
Analysis of Aluminum and Aluminum Alloys by Spark
Atomic Emission Spectrometry
This standard is issued under the fixed designation E1251; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope
1.1 This test method describes the analysis of aluminum and its alloys by atomicspark-atomic emission spectrometry.
spectrometry (Spark-AES). The aluminum specimen to be analyzed may be in the form of a chill cast disk, casting, foil, sheet,
plate, extrusion, or some other wrought form or shape. The elements covered in the scope of this method are listed in the table
below.
Tested Concentration Mass Fraction
Element Range
(Wt %)
Antimony 0.001 to 0.003
Arsenic 0.001 to 0.006
Beryllium 0.0004 to 0.24
Bismuth 0.03 to 0.6
Boron 0.0006 to 0.009
Calcium 0.0002 to –
Chromium 0.001 to 0.23
Cobalt 0.4 to –
Copper 0.001 to 5.5
Gallium 0.02 to –
Iron 0.2 to 0.5
Lead 0.04 to 0.6
Lithium 0.0003 to 2.1
Magnesium 0.03 to 5.4
Manganese 0.001 to 1.2
Nickel 0.005 to 2.6
Phosphorus 0.003 to –
Silicon 0.07 to 16
Sodium 0.003 to 0.02
Strontium 0.03 to –
Tin 0.03 to –
Titanium 0.001 to 0.12
Vanadium 0.002 to 0.022
Zinc 0.002 to 5.7
Zirconium 0.001 to 0.12
NOTE 1—The concentration mass fraction ranges given in the above scope were established through cooperative testing (ILS) of selected reference
materials. The range shown for each element does not demonstrate the actual usable analytical range for that element. The usable analytical range may
be extended higher or lower based on individual instrument capability, spectral characteristics of the specific element wavelength being used, and the
availability of appropriate reference materials.
NOTE 2—Mercury (Hg) is intentionally not included in the scope. Analysis of Hg in aluminum by spark atomic emission spectrometry (Spark-AES)
Spark-AES is not recommended. Accurate analysis of Hg using this technique is compromised by the presence of an intense iron interference. Inaccurate
reporting of Hg due to these interference effects can jeopardize the current designation of aluminum production as a mercury-free process. To demonstrate
compliance with legislated Hg content limits, use of an alternate method capable of analysis with a minimum reporting limit of 0.0001% or lower is
recommended. Suitable techniques include but are not limited to glow discharge mass spectrometry, XRF, and cold vapor AA.GD-MS, XRF (X-ray
fluorescence), cold vapor AA, and ICP-MS.
1.2 This test method is suitable primarily for the analysis of chill cast disks as defined in Practices E716. Other forms may be
analyzed, provided that: (1) they are sufficiently massive to prevent undue heating, (2) they allow machining to provide a clean,
This test method is under the jurisdiction of ASTM Committee E01 on Analytical Chemistry for Metals, Ores, and Related Materials and is the direct responsibility of
Subcommittee E01.04 on Aluminum and Magnesium.
Current edition approved Sept. 15, 2017Oct. 1, 2017. Published October 2017. Originally approved in 1988. Last previous edition approved in 20112017 as
E1251 – 11.E1251 – 17. DOI: 10.1520/E1251-17.10.1520/E1251-17A.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E1251 − 17a
flat surface, which creates a seal between the specimen and the spark stand, and (3) reference materials of a similar metallurgical
condition and chemical composition are available.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. Specific safety and health statements are given in Section 10.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
B985 Practice for Sampling Aluminum Ingots, Billets, Castings and Finished or Semi-Finished Wrought Aluminum Products for
Compositional Analysis
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E135 Terminology Relating to Analytical Chemistry for Metals, Ores, and Related Materials
E158 Practice for Fundamental Calculations to Convert Intensities into Concentrations in Optical Emission Spectrochemical
Analysis (Withdrawn 2004)
E172 Practice for Describing and Specifying the Excitation Source in Emission Spectrochemical Analysis (Withdrawn 2001)
E305 Practice for Establishing and Controlling Atomic Emission Spectrochemical Analytical Curves
E406 Practice for Using Controlled Atmospheres in Spectrochemical Analysis
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
E716 Practices for Sampling and Sample Preparation of Aluminum and Aluminum Alloys for Determination of Chemical
Composition by Spark Atomic Emission Spectrometry
E826 Practice for Testing Homogeneity of a Metal Lot or Batch in Solid Form by Spark Atomic Emission Spectrometry
E876 Practice for Use of Statistics in the Evaluation of Spectrometric Data (Withdrawn 2003)
E1329 Practice for Verification and Use of Control Charts in Spectrochemical Analysis
E1507 Guide for Describing and Specifying the Spectrometer of an Optical Emission Direct-Reading Instrument
2.2 ANSI Standard:
ANSI H35.1/H35.1M American National Standard Alloy and Temper Designation Systems for Aluminum
3. Terminology
3.1 Definitions—For definitions of terms used in this Standard, refer to Terminology E135.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 alloy-type calibration—calibration curves determined using calibrants calibration materials from alloys with similar
compositions.
3.2.2 binary-type calibration—calibration curves determined using binary calibrants calibration materials (primary aluminum
to which has been added one specific element).
3.2.3 global-type calibration—calibration curves determined using calibrants calibration materials from many different alloys
with considerable compositional differences.
3.2.4 two-point drift correction—the practice of analyzing a high and low standardant standardization materials for each
calibration curve and adjusting the counts or voltage values intensities obtained back to the values obtained on those particular
standardants standardization materials during the collection of the calibration data. The corrections are accomplished mathemati-
cally and are applied to both the slope and intercept. Improved precision may be obtained by using a multi-point drift correction
as described in Practice E1329.
3.2.5 type standardization—mathematical adjustment of the calibration curve’s slope or intercept using a single standardant
(reference material) standardization materials at or close to the nominal composition for the particular alloy being analyzed. For
best results, the standardant standardization material being used should be within 610 % of the composition (for each respective
element) of the material being analyzed.
4. Summary of Test Method
4.1 A unipolar triggered capacitor controlled electrical discharge is produced in an argon atmosphere between the prepared flat
surface of a specimen and the tip of a semi-permanent counter electrode. The energy of the discharge is sufficient to ablate material
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
E1251 − 17a
from the surface of the sample, break the chemical or physical bonds, and cause the resulting atoms or ions to emit radiant energy.
The radiant energies of the selected analytical lines and the internal standard line(s) are converted into electrical signals by either
photomultiplier tubes (PMTs) or a suitable solid state detector. The detector signals are electrically integrated and converted to a
digitized value. The signals are ratioed to the proper internal standard signal and converted into concentrations by a computer mass
fractions in accordance with Practice E158.
4.2 Three different methods of calibration defined in 3.2.2, 3.2.3, and 3.2.1 are capable of giving the same precision, accuracy,
and detection limit.
4.2.1 The first method, binary calibration, employs calibration curves that are determined using a large number of high-purity
binary calibrants. calibration materials. This approach is used when there is a need to analyze almost the entire range of aluminum
alloys. Because binary calibrants calibration materials may respond differently from alloy calibrants, calibration materials, the
latter are used to improve accuracy by applying a slope and/or intercept correction to the observed readings.
4.2.2 The second method, global calibration, employs calibration curves that are determined using many different alloy
calibrants calibration materials with a wide variety of compositions. Mathematical calculations are used to correct for both alloy
difference and inter-element effects. Like the method above, specific alloy calibrants calibration materials may be used to apply
a slope and/or intercept correction to the observed readings.
4.2.3 The third method, alloy calibration, employs calibration curves that are determined using different alloy calibrants
calibration materials that have similar compositions. Again, specific alloy calibrants calibration materials may be used to apply a
slope and/or intercept correction to the observed readings.
5. Significance and Use
5.1 The metallurgical properties of aluminum and its alloys are highly dependent on chemical composition. Precise and accurate
analyses are essential to obtaining desired properties, meeting customer specifications, and helping to reduce scrap due to off-grade
material.
5.2 This test method is applicable to chill cast specimens as defined in Practices E716 and can also be applied to other types
of samples provided that suitable reference materials are available. Also, other sample forms can be melted down and cast into a
disk, using an appropriate mold, as described in Practices E716. However, it should be noted that some elements (for example,
magnesium) readily form oxides, while some others (for example, sodium, lithium, calcium, and strontium) are volatile, and may
be lost to varying degrees during the melting process.
6. Recommended Analytical Lines and Potential Interferences
6.1 Table 1 lists the analytical lines commonly used for aluminum analysis. Other lines may be used if they give comparable
results. Also listed are recommended concentration mass fraction ranges, background equivalent concentrations (BEC),mass
fractions, detection limits, useful linear ranges, and potential interferences. The values given in this table are typical; actual values
obtained are dependent on instrument design.
NOTE 3—The BEC background equivalent mass fraction and detection limits listed in Table 1 have been attained with a spectrometer that has a
reciprocal dispersion of 54 nm/mm and a working resolution of 3.5 nm, using an entrance slit width of 25 μm and exit slit widths of 50 μm.
7. Apparatus
7.1 Specimen Preparation Equipment:
7.1.1 Sampling Molds, for aluminum and the techniques of pouring a sample disk are described in Practices E716. Chill cast
samples, poured and cast as described within Practices E716, shall be the recommended form in this test method.
7.1.2 Lathe, capable of machining a smooth, flat surface on the reference materials and samples. A variable speed cutter, a
cemented carbide or polycrystalline diamond tool bit, and an automatic cross feed are highly recommended. Proper depth of cut
and desired surface finish are described in Practices E716.
7.1.3 Milling Machine, a milling machine can be used as an alternative to a lathe.
7.1.4 Milling Machine, a milling machine can be used as an alternative to a lathe.It is strongly recommended that the same
preparation machinery used to prepare the standards is also used to prepare the samples. Differences in surface characteristics may
influence the analysis.
NOTE 4—It is strongly recommended that the same preparation machinery used to prepare the standards is also used to prepare the samples. Differences
in surface characteristics may influence the analysis.
7.2 Excitation Source, Source—capable of producing a unipolar, triggered capacitor discharge. In today’s instrumentation, the
excitation source is computer controlled and is normally programmed to produce: (1) a high-energy pre-spark (of some preset
duration), (2) a spark-type discharge (of some preset duration), (3) an arc-type discharge (of some preset duration), and (4) a
spark-type discharge, during which, time-resolved measurements are made for improved detection limits (this may be optional on
some instruments).
7.2.1 Typical parameters and exposure times are given in Table 2. It should be emphasized that the information presented is
given as an example only and parameters may vary with respect to instrument model and manufacturer. For details on describing
and specifying an excitation source, please refer to Practice E172.
E1251 − 17a
TABLE 1 Recommended Analytical Lines
Recommended High
Wavelength Background Calculated
ConcentrationMass ConcentrationMass Interferences
Element in Air Equivalent, Detection
F
Fraction Fraction Element, λ(nm) and k, %
A B C,D
(nm) % Limit, %
E
Range, % Index, %
Aluminum I 256.799 70-100
Aluminum 256.799 I 70-100
I 266.039 70-100
266.039 I 70-100
I 237.208 70-100
237.208 I 70-100
Antimony I 231.147 0.001-0.5 0.17 0.0002 Co 231.166 0.6
Antimony 231.147 I 0.001-0.5 0.17 0.0002 Co 231.166 0.6
I 259.806 0.001-0.5 0.0002 Fe 259.837
259.806 I 0.001-0.5 0.0002 Fe 259.837
Mn 259.817 0.01
Arsenic 234.984 I 0.005-0.1
Beryllium I 234.861 0.0001-0.05 0.001 0.00003
Beryllium 234.861 I 0.0001-0.05 0.001 0.00003
II 313.042 0.0001-0.05 0.0035 0.00001
313.042 II 0.0001-0.05 0.0035 0.00001
332.134 I 0.0001-0.05 0.00001
Bismuth I 306.772 0.001-0.7 0.04 0.0002
Bismuth 306.772 I 0.001-0.7 0.04 0.0002
Boron I 249.773 0.0001-0.05 0.002 0.0001* Fe 249.782 0.001
Boron 249.773 I 0.0001-0.05 0.002 0.0001* Fe 249.782 0.001
Mn 249.778 0.007
I 249.678 0.0001-0.05
249.678 I 0.0001-0.05
I 208.959 0.0001-0.05 Mo 208.952 0.13
208.959 I 0.0001-0.05 Mo 208.952 0.13
Cadmium I 228.802 0.001-1 0.05 <0.0001 As 228.812
Cadmium 228.802 I 0.001-1 0.05 <0.0001 As 228.812
I 479.992 0.005-2 0.15 0.003
479.992 I 0.005-2 0.15 0.003
G
Calcium II 393.367 0.001-0.05 0.001 0.00005 Fe 393.361
G
Calcium 393.367 II 0.001-0.05 0.001 0.00005 Fe 393.361
Chromium I 425.435 0.001-1 0.015 <0.0001
Chromium 425.435 I 0.001-1 0.015 <0.0001
II 267.716 0.001-1 0.004 0.0005*
267.716 II 0.001-1 0.004 0.0005*
G
II 276.654 0.005-1
G
276.654 II 0.005-1
Cobalt I 345.351 0.0001-2 <0.0001
Cobalt 345.351 I 0.0001-2 <0.0001
Copper I 327.396 0.001-1.5 0.005 <0.0001 0.7
Copper 327.396 I 0.001-1.5 0.005 <0.0001 0.7
I 324.754 0.001-0.5
324.754 I 0.001-0.5
I 296.117 0.05-20 0.40 0.01* >20 Fe 296.128
296.117 I 0.05-20 0.40 0.01* >20 Fe 296.128
II 224.700 0.01-5 0.03 0.0005* 5
224.700 II 0.01-5 0.03 0.0005* 5
I 510.554 0.05-20 0.32 0.01* >20
510.554 I 0.05-20 0.32 0.01* >20
Gallium I 294.364 0.001-0.05 0.015 <0.0001
Gallium 294.364 I 0.001-0.05 0.015 <0.0001
G
I 417.206 0.001-0.05 Fe 417.213
Ti 417.190
Cr 417.167
G
417.206 I 0.001-0.05 Fe 417.213
Ti 417.190
Cr 417.167
Iron II 238.204 0.001-1.5 0.015 0.0008 1.0
Iron 238.204 II 0.001-1.5 0.015 0.0008 1.0
II 259.940 0.001-1.5 0.005 0.0004
259.940 II 0.001-1.5 0.005 0.0004
I 259.957
259.957 I
II 273.955 0.01-3.5
273.955 II 0.01-3.5
G
I 374.949 0.001-3.5 0.0001
G
374.949 I 0.001-3.5 0.0001
I 441.512 0.01-3.5 0.0004
441.512 I 0.01-3.5 0.0004
I 438.355 0.005-3.5
438.355 I 0.005-3.5
E1251 − 17a
TABLE 1 Continued
Recommended High
Wavelength Background Calculated
ConcentrationMass ConcentrationMass Interferences
Element in Air Equivalent, Detection
F
Fraction Fraction Element, λ(nm) and k, %
A B C,D
(nm) % Limit, %
E
Range, % Index, %
Lead I 405.782 0.002-0.7 0.04 0.0001 Mn 405.792 0.01
Mg 405.763 0.001
Lead 405.782 I 0.002-0.7 0.04 0.0001 Mn 405.792 0.01
Mg 405.763 0.001
I 283.306 0.002-0.7 0.07 0.002
283.306 I 0.002-0.7 0.07 0.002
Lithium I 610.364 0.0001-3
Lithium 610.364 I 0.0001-3
I 670.784 0.0001-0.02 0.0005 <0.0005
670.784 I 0.0001-0.02 0.0005 <0.0005
I 323.261 0.01-3 Fe 323.279
Sb 323.250
323.261 I 0.01-3 Fe 323.279
Sb 323.250
Magnesium II 279.553 0.0005-0.3 0.0006 0.00003 0.04
Magnesium 279.553 II 0.0005-0.3 0.0006 0.00003 0.04
I 285.213 0.0005-0.3 0.008 <0.0001 0.25
285.213 I 0.0005-0.3 0.008 <0.0001 0.25
I 277.669 0.05-11 0.08 0.01 >11
277.669 I 0.05-11 0.08 0.01 >11
G
I 383.231 0.01-11 0.015 0.002* >11
G
383.231 I 0.01-11 0.015 0.002* >11
I 383.826 0.1-11
383.826 I 0.1-11
I 518.362 0.01-11 0.02 0.002* >11
518.362 I 0.01-11 0.02 0.002* >11
G
Manganese I 403.076 0.001-0.1 0.028 0.0001*
G
Manganese 403.076 I 0.001-0.1 0.028 0.0001*
II 259.373 0.0005-0.5 0.004 0.00005 0.2
II 293.306 0.001-2 0.006 0.0002* >1.1
259.373 II 0.0005-0.5 0.004 0.00005 0.2
293.306 II 0.001-2 0.006 0.0002* >1.1
II 346.033B 0.01-2
346.033 II 0.01-2
Nickel I 341.476 0.001-2 0.02 <0.0001 >2.5 Zr 341.466 0.01
Nickel 341.476 I 0.001-2 0.02 <0.0001 >2.5 Zr 341.466 0.01
I 310.188 0.005-4 0.05 0.001* >5
310.188 I 0.005-4 0.05 0.001* >5
II 231.604 0.001-2 0.015 0.0005* <2.5
231.604 II 0.001-2 0.015 0.0005* <2.5
H
Phosphorus I 178.231 0.0001-0.1 0.084 0.0001
H
Phosphorus 178.231 I 0.0001-0.1 0.084 0.0001
Silicon I 288.158 0.001-1.5 0.01 0.0001 1.5 Cr 288.123
Silicon 288.158 I 0.001-1.5 0.01 0.0001 1.5 Cr 288.123
I 251.612 0.001-1.5 0.006 0.0001 1.5
251.612 I 0.001-1.5 0.006 0.0001 1.5
G
I 390.553 0.05-24 0.25 0.01 >24 Cr 390.566 0.09
G
390.553 I 0.05-24 0.25 0.01 >24 Cr 390.566 0.09
I 212.415 0.05-24 0.5 0.05 >24
212.415 I 0.05-24 0.5 0.05 >24
Silver I 328.068 0.0005-0.1
Silver 328.068 I 0.0005-0.1
I 338.289 0.0001-0.1 >10
338.289 I 0.0001-0.1 >10
I 466.848 0.05-1.5
466.848 I 0.05-1.5
Sodium I 588.995 0.0001-0.05 0.0015 <0.0001
Sodium 588.995 I 0.0001-0.05 0.0015 <0.0001
G
Strontium II 421.552 0.0001-0.1 0.0004 0.0001
G
Strontium 421.552 II 0.0001-0.1 0.0004 0.0001
I 460.733 0.0005-0.06
460.733 I 0.0005-0.06
Tin I 317.502 0.001-7.5 0.04 0.0001 >10
Tin 317.502 I 0.001-7.5 0.04 0.0001 >10
Titanium II 334.904 0.0005-0.5 0.004 <0.0001
Titanium 334.904 II 0.0005-0.5 0.004 <0.0001
II 337.280 0.001-0.5 0.002 <0.00010
337.280 II 0.001-0.5 0.002 <0.00010
I 363.545 0.0005-0.05 0.030 0.003*
363.545 I 0.0005-0.05 0.030 0.003*
Vanadium I 318.341 0.001-0.15 0.06 0.0003*
Vanadium 318.341 I 0.001-0.15 0.06 0.0003*
E1251 − 17a
TABLE 1 Continued
Recommended High
Wavelength Background Calculated
ConcentrationMass ConcentrationMass Interferences
Element in Air Equivalent, Detection
F
Fraction Fraction Element, λ(nm) and k, %
A B C,D
(nm) % Limit, %
E
Range, % Index, %
I 437.924 0.001-0.25
437.924 I 0.001-0.25
II 310.230 0.001-0.15 0.014 <0.0001
310.230 II 0.001-0.15 0.014 <0.0001
Zinc I 213.856 0.0005-0.1 0.035 0.0001* 0.05
Zinc 213.856 I 0.0005-0.1 0.035 0.0001* 0.05
I 334.502 0.001-10.0 0.065 0.0004 >8
334.502 I 0.001-10.0 0.065 0.0004 >8
I 481.053 0.01-10 0.07 0.001* >10
481.053 I 0.01-10 0.07 0.001* >10
I 472.216 0.01-10 0.26 0.0015 >10
472.216 I 0.01-10 0.26 0.0015 >10
Zirconium II 339.198 0.001-1 0.02 0.001*
Zirconium 339.198 II 0.001-1 0.02 0.001*
G
II 349.621 0.001-1 0.006 <0.0001
G
349.621 II 0.001-1 0.006 <0.0001
A
I = atom line, II = ion line. Second (2nd) indicates that the second order shall be used, where available.
B
Background Equivalent Concentration (BEC)Mass Fraction—The concentration mass fraction at which the signal due to the element is equal to the signal due to the
background.
C
In this test method, the calculated detection limit was measured by calculating the standard deviation of ten consecutive burns on a specimen with element
concentration(s) mass fraction(s) at levels below ten times the expected detection limit.
D
See footnote C. For values marked with an asterisk (*) the available data were for a concentration mass fraction greater than ten (10) times but less than a hundred (100)
times the expected detection limit.
E
High Concentration Mass Fraction Index—The concentration mass fraction at which the slope of the calibration curve drops below 0.75.
F
Interference Factor, k—The apparent increase in the concentration mass fraction of the element being determined, expressed in percent, due to the presence of 1.0 %
of the interfering element.
G
Useful analytical lines with improved signal to background ratios due to the complete removal of C-N background by the argon atmosphere.
H
If phosphorus is to be determined, the most sensitive line appears to be the 178.231 nm in the second order which requires either a vacuum or a gas filled spectrometer.
The vacuum spectrometer should be operated at a pressure of 25 microtorr or less. The gas filled spectrometer will be charged with nitrogen to a positive pressure of slightly
over one atmosphere (101 k pa). Optimum results are obtained by using a background channel that has been profiled “off peak” of the first order 178.231 nm phosphorus
line as the internal standard. The ratio of P 178.231 nm (2nd) / background near the 178.231 nm (1st) is plotted against % phosphorus. Even with this compensation for
variability in background, alloys with highly different compositions of major alloying elements, particularly silicon, require separate reference materials and analytical curves.
TABLE 2 Typical Excitation Source Electrical Parameters
High Energy
Parameter Spark Arc
Pre-spark
Resistance, 1 1 15
Inductance, μH 30 130 30
Volts, V 400 400 400
Frequency, Hz 300 300 300
Capacitance, μF 12 3 5
Time, s 10 5 5
7.3 Excitation Chamber, shall be designed with an upper plate that is smooth and flat so that it will mate (seal) perfectly with
the prepared surface of the sample specimen. The seal that is formed between the two will exclude atmospheric oxygen from
entering the discharge chamber. The excitation chamber will contain a mounting clamp to hold the counter electrode. The
excitation stand assembly will also have some type of clamp or device designed to hold the sample firmly against the top plate.
Some manufacturers may provide for the top plate to be liquid cooled to minimize sample heat-upheating during the excitation
cycle. The excitation chamber will also be constructed so that it is flushed automatically with argon gas during the analytical burn
cycle. The excitation chamber’s design should allow for a flow of argon gas to prevent the deposition of ablated metal dust on the
inner-chamber quartz window(s). The excitation chamber will be equipped with an exhaust system that will safely dispose of the
argon gas and the metal dust created during the excitation cycle. For reasons of health and cleanliness, the exhausted gas and dust
should not be vented directly into the laboratory. To help with this situation, manufacturers have designed their instruments with
some type of exhaust/filter system to deal with tominimize this problem. The exhaust can then be vented into an efficient hood
system.
7.4 Gas Flow System, will be designed so that it can deliver pure argon gas to the excitation chamber. The purity of the argon
gas will affect the precision of the results. Generally, precision improves as the purity of the argon gas gets higher. Argon gas with
a minimum purity of 99.995 % has been found to be acceptable. The gas shall be delivered by a flow system as described in
Practice E406. The argon gas source can be from high-purity compressed gas cylinders, a cryogenic-type cylinder that contains
liquid argon or possibly from a central supply (liquid only). It is essential that only argon gas meeting the minimum purity of
99.995 % be used. A lower purity grade of argon, such as a “welding grade,” should not be used. The delivery system shall be
E1251 − 17a
composed of a two-stage type (high/low pressure) regulator of all-metal construction with two pressure gages. Delivery tubing
must not produce any contamination of the argon stream. Refrigerator-grade copper tubing is recommended. The gages on the
regulator will allow for the adjustment of the gas pressure to the instrument. Delivery pressure specifications will vary with
instrument manufacturer. Please note that the delivery tube connections should be made with all-metal all metal seals and the
delivery tubing itself should be kept as short as possible (possible. (AllNote 5). metal connections are strongly recommended
because the discharge is adversely affected by organic contamination, or by as little as 2 ppm of oxygen or a few ppm of water
vapor.) Argon supply shall be sufficient to support required flow during analysis and bleed during idle periods. All connections must
be leak-free.
NOTE 5—All-metal connections are strongly recommended because the discharge is adversely affected by organic contamination, or by as little as 2
ppm of oxygen or a few ppm of water vapor.
7.5 Spectrometer—For details on specifying the spectrometer please refer to Guide E1507.
7.6 Measuring and Control System of the instrument consists of either photomultiplier and integrating electronics or solid-state
photosensitive arrays (CCD or CID) that convert observed light intensities to a digitizable signal. A dedicated computer and/or
microprocessor is used to control burn conditions, source operation, data acquisition, and the conversion of intensity data to
concentrations. mass fractions. Data should be accessible to the operator throughout all steps of the calculation process.
Concentration data may be automatically transferred to a site mainframe computer or server for further data storage and
distribution. The instrument’s control software should include functions for routine instrument drift correction (standardization),
type standardization, and the application of these functions to subsequent analyses.
8. Materials
8.1 Counter Electrode—The counter electrode and specimen surface are the two terminus points of the spark discharge. The
counter electrode should be made from thoriated tungsten or silver other suitable material and have a pointed end. The gap distance
between the specimen surface and the tip of the counter electrode is specified by the manufacturer. The diameter and geometry
of the counter electrode is also application and vendormanufacturer dependent. If different designs and/or configurations are
offered, it is recommended that the prospective purchaser test each design to determine which one performs the best for the
intended analytical task. The counter electrode configuration and auxiliary gap distance must not be altered subsequent to
spectrometer calibration or calibration adjustments. Electrode maintenance (frequent brushing of the counter electrode) to maintain
its configuration, gap distance, and minimize surface contamination are critical to accurate, precise analytical results. It is
recommended that the instrument purchaser order several spare counter electrodes so that they can be replaced when necessary.
9. Reference Materials
9.1 Calibrants—Calibration Materials—All calibrants calibration materials shall be homogeneous and free of cracks or
porosity. These materials should also possess a metallurgical condition that is similar to the material(s) that are being analyzed.
The calibrants calibration materials shall be used to produce the analytical curves for the various elements being determined.
9.1.1 It is recommended that a calibration curve for any particular element be composed of a minimum of four calibrants. The
concentrations calibration materials. The mass fractions of these calibrants calibration materials should be fairly evenly spaced
over the calibrated analytical range so that a mathematically valid calibration curve can be established using all of the points.
9.1.1.1 The calibrants calibration materials used shall be of sufficient quality, purchased from a recognized reputable source, and
have certified values to the required accuracy for the anticipated analytical tasks to be performed. A few SRMs are available from
the National Institute of Standards and Technology (NIST). Also, there are other commercial sources for aluminum reference
materials.
9.1.2 For trace elements, reference materials that contain variable concentrations mass fractions of the trace element in a typical
alloy of constant or nearly constant composition are available. These reference materials can be used for establishing the analytical
curve, but will not reveal potential i
...



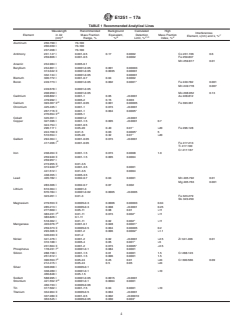




Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...