ASTM C1894-22
(Guide)Standard Guide for Microbially Induced Corrosion of Concrete Products
Standard Guide for Microbially Induced Corrosion of Concrete Products
SCOPE
1.1 This guide discusses microbially induced corrosion (MIC) of concrete products and laboratory test methods for determining the resistance of concrete to MIC. Although the guide is intended for concrete products, it also covers cementitious mortar and paste that are used in specialized applications or laboratory investigations.
1.2 While this guide discusses concrete materials and admixtures, the document is not intended to specifically address field exposure conditions or sewage pipe, concrete tank, or concrete riser network design.
1.3 This guide does not cover live trial tests where concrete coupons or other specimens are monitored in sewers.
1.4 This guide does not cover concrete deterioration due to chemical sulfate attack, which is caused by the reaction of sulfate compounds that exist in wastewater with the hydration products of cement. Test methods for assessing sulfate attack are provided by Test Methods C452 and C1012/C1012M.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 The text of this guide references notes and footnotes that provide explanatory material. These notes and footnotes (excluding those in tables and figures) shall not be considered as requirements of the standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 28-Feb-2022
- Technical Committee
- C13 - Concrete Pipe
Relations
- Refers
ASTM C31/C31M-24a - Standard Practice for Making and Curing Concrete Test Specimens in the Field - Effective Date
- 01-Apr-2024
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Feb-2024
- Refers
ASTM C31/C31M-24 - Standard Practice for Making and Curing Concrete Test Specimens in the Field - Effective Date
- 01-Jan-2024
- Effective Date
- 01-May-2020
- Effective Date
- 15-Dec-2019
- Effective Date
- 01-Oct-2019
- Effective Date
- 15-Jun-2019
- Effective Date
- 01-Apr-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-Jan-2019
- Effective Date
- 15-Dec-2018
- Effective Date
- 15-Dec-2018
- Effective Date
- 01-Oct-2018
- Refers
ASTM D4262-05(2018) - Standard Test Method for pH of Chemically Cleaned or Etched Concrete Surfaces - Effective Date
- 01-Aug-2018
Overview
ASTM C1894-22: Standard Guide for Microbially Induced Corrosion of Concrete Products provides essential guidance on microbially induced corrosion (MIC) in concrete structures commonly found within wastewater and sewage infrastructure. This guide addresses laboratory test methods for evaluating the resistance of concrete, cementitious mortar, and paste to MIC, while also offering practical information on concrete components and antimicrobial admixtures related to corrosion resistance.
MIC is a critical durability concern for concrete exposed to environments where specific microorganisms facilitate the production of corrosive acids, especially in wastewater treatment systems. Understanding MIC and how to test materials for resistance are vital for manufacturers, engineers, and facility operators seeking to ensure the longevity and performance of concrete products in aggressive environments.
Key Topics
Mechanisms of MIC
The guide describes a multi-stage process of MIC:- Initial alkalinity reduction (Stage I)
- Formation of biofilms and further pH decrease (Stage II)
- Acid attack and material degradation (Stage III)
Laboratory Test Methods
Several laboratory approaches are detailed for simulating and measuring MIC:- Biogenic (biotic) acidification tests, with advanced chamber and benchtop methods
- Acid immersion tests for Stage III damage simulation
- Surface pH measurement techniques
- Screening methodologies for antimicrobial and admixture effectiveness
Material Selection and Admixtures
The guide discusses the impact of cement types, supplementary cementitious materials, differing aggregate compositions (such as calcareous limestone vs. siliceous aggregates), and chemical or antimicrobial admixtures on MIC resistance.Limitations
The document clarifies that while various laboratory methods provide valuable data, they cannot fully replicate the complexity of real-world field conditions, such as those found in sewer systems.
Applications
ASTM C1894-22 is valuable for professionals involved in the design, evaluation, and production of:
- Concrete pipes, tanks, manholes, and wastewater infrastructure, where exposure to MIC is a concern.
- Manufacturers developing or specifying concrete products and mixtures for municipal or industrial wastewater environments.
- Testing laboratories performing standard resistance and qualification assessments on new materials, especially those incorporating antimicrobial admixtures.
- Quality control and assurance teams seeking to evaluate long-term performance of cementitious products in biogenically aggressive scenarios.
The standard is particularly useful in:
- Research and development for innovative or MIC-resistant materials
- Comparative assessment of aggregates, binders, and admixtures
- Simulating MIC progression for material improvement or performance documentation
Related Standards
ASTM C1894-22 references several critical standards to support MIC testing and terminology, including:
- ASTM C31/C31M - Practice for Making and Curing Concrete Test Specimens in the Field
- ASTM C33/C33M - Specification for Concrete Aggregates
- ASTM C42/C42M - Methods for Testing Drilled Cores of Concrete
- ASTM C150/C150M, C595/C595M, C1600/C1600M - Specifications for various cements
- ASTM C1904 - Biogenic acidification immersion test procedures
- ASTM C1898 - Chemical acid resistance testing for concrete
- ISO 22196 - Measurement of antibacterial activity on non-porous surfaces
Please consult all relevant ASTM and ISO standards for full methodological details and material requirements related to MIC in concrete.
Keywords: microbially induced corrosion, MIC, concrete corrosion, concrete pipe, wastewater infrastructure, biogenic acid attack, laboratory test methods, antimicrobial admixtures, sulfate-reducing bacteria, sulfate-oxidizing bacteria, biofilm, ASTM C1894-22.
Buy Documents
ASTM C1894-22 - Standard Guide for Microbially Induced Corrosion of Concrete Products
REDLINE ASTM C1894-22 - Standard Guide for Microbially Induced Corrosion of Concrete Products
Get Certified
Connect with accredited certification bodies for this standard

ICC Evaluation Service
Building products evaluation and certification.

QAI Laboratories
Building and construction product testing and certification.

Aboma Certification B.V.
Specialized in construction, metal, and transport sectors.
Sponsored listings
Frequently Asked Questions
ASTM C1894-22 is a guide published by ASTM International. Its full title is "Standard Guide for Microbially Induced Corrosion of Concrete Products". This standard covers: SCOPE 1.1 This guide discusses microbially induced corrosion (MIC) of concrete products and laboratory test methods for determining the resistance of concrete to MIC. Although the guide is intended for concrete products, it also covers cementitious mortar and paste that are used in specialized applications or laboratory investigations. 1.2 While this guide discusses concrete materials and admixtures, the document is not intended to specifically address field exposure conditions or sewage pipe, concrete tank, or concrete riser network design. 1.3 This guide does not cover live trial tests where concrete coupons or other specimens are monitored in sewers. 1.4 This guide does not cover concrete deterioration due to chemical sulfate attack, which is caused by the reaction of sulfate compounds that exist in wastewater with the hydration products of cement. Test methods for assessing sulfate attack are provided by Test Methods C452 and C1012/C1012M. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 The text of this guide references notes and footnotes that provide explanatory material. These notes and footnotes (excluding those in tables and figures) shall not be considered as requirements of the standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SCOPE 1.1 This guide discusses microbially induced corrosion (MIC) of concrete products and laboratory test methods for determining the resistance of concrete to MIC. Although the guide is intended for concrete products, it also covers cementitious mortar and paste that are used in specialized applications or laboratory investigations. 1.2 While this guide discusses concrete materials and admixtures, the document is not intended to specifically address field exposure conditions or sewage pipe, concrete tank, or concrete riser network design. 1.3 This guide does not cover live trial tests where concrete coupons or other specimens are monitored in sewers. 1.4 This guide does not cover concrete deterioration due to chemical sulfate attack, which is caused by the reaction of sulfate compounds that exist in wastewater with the hydration products of cement. Test methods for assessing sulfate attack are provided by Test Methods C452 and C1012/C1012M. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 The text of this guide references notes and footnotes that provide explanatory material. These notes and footnotes (excluding those in tables and figures) shall not be considered as requirements of the standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM C1894-22 is classified under the following ICS (International Classification for Standards) categories: 91.100.30 - Concrete and concrete products. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM C1894-22 has the following relationships with other standards: It is inter standard links to ASTM C31/C31M-24a, ASTM C1012/C1012M-24, ASTM C989/C989M-24, ASTM C31/C31M-24, ASTM C1898-20, ASTM C125-19a, ASTM C497-19a, ASTM C452-19, ASTM C150/C150M-19, ASTM C125-19, ASTM C497-19, ASTM C1600/C1600M-18, ASTM C1012/C1012M-18b, ASTM C125-18b, ASTM D4262-05(2018). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM C1894-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: C1894 − 22
Standard Guide for
Microbially Induced Corrosion of Concrete Products
This standard is issued under the fixed designation C1894; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This guide discusses microbially induced corrosion 2.1 ASTM Standards:
(MIC) of concrete products and laboratory test methods for C31/C31M Practice for Making and Curing Concrete Test
determining the resistance of concrete to MIC. Although the Specimens in the Field
guide is intended for concrete products, it also covers cemen- C33/C33M Specification for Concrete Aggregates
titious mortar and paste that are used in specialized applica- C42/C42M Test Method for Obtaining and Testing Drilled
tions or laboratory investigations. Cores and Sawed Beams of Concrete
C125 Terminology Relating to Concrete and Concrete Ag-
1.2 While this guide discusses concrete materials and
gregates
admixtures, the document is not intended to specifically
C150/C150M Specification for Portland Cement
address field exposure conditions or sewage pipe, concrete
C192/C192M Practice for Making and Curing ConcreteTest
tank, or concrete riser network design.
Specimens in the Laboratory
1.3 This guide does not cover live trial tests where concrete
C260/C260M Specification for Air-Entraining Admixtures
coupons or other specimens are monitored in sewers.
for Concrete
1.4 This guide does not cover concrete deterioration due to C294 Descriptive Nomenclature for Constituents of Con-
crete Aggregates
chemical sulfate attack, which is caused by the reaction of
sulfate compounds that exist in wastewater with the hydration C452 Test Method for Potential Expansion of Portland-
Cement Mortars Exposed to Sulfate
products of cement. Test methods for assessing sulfate attack
are provided by Test Methods C452 and C1012/C1012M. C494/C494M Specification for Chemical Admixtures for
Concrete
1.5 The values stated in SI units are to be regarded as
C497 Test Methods for Concrete Pipe, Concrete Box
standard. No other units of measurement are included in this
Sections, Manhole Sections, or Tile
standard.
C595/C595M Specification for Blended Hydraulic Cements
1.6 Thetextofthisguidereferencesnotesandfootnotesthat
C618 Specification for Coal Fly Ash and Raw or Calcined
provide explanatory material. These notes and footnotes (ex-
Natural Pozzolan for Use in Concrete
cluding those in tables and figures) shall not be considered as
C822 Terminology Relating to Concrete Pipe and Related
requirements of the standard.
Products
1.7 This standard does not purport to address all of the C989/C989M Specification for Slag Cement for Use in
safety concerns, if any, associated with its use. It is the Concrete and Mortars
responsibility of the user of this standard to establish appro- C1012/C1012M Test Method for Length Change of
priate safety, health, and environmental practices and deter- Hydraulic-Cement Mortars Exposed to a Sulfate Solution
mine the applicability of regulatory limitations prior to use. C1017/C1017M Specification for Chemical Admixtures for
1.8 This international standard was developed in accor- Use in Producing Flowing Concrete (Withdrawn 2022)
dance with internationally recognized principles on standard- C1240 Specification for Silica Fume Used in Cementitious
ization established in the Decision on Principles for the Mixtures
Development of International Standards, Guides and Recom- C1600/C1600M Specification for Rapid Hardening Hydrau-
mendations issued by the World Trade Organization Technical lic Cement
Barriers to Trade (TBT) Committee.
1 2
This test method is under the jurisdiction of ASTM Committee C13 on For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Concrete Pipe and is the direct responsibility of Subcommittee C13.03 on contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Determining the Effects of Biogenic SulfuricAcid on Concrete Pipe and Structures. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved March 1, 2022. Published March 2022. Originally the ASTM website.
approved in 2019. Last previous edition approved in 2019 as C1570 – 19. DOI: The last approved version of this historical standard is referenced on
10.1520/C1894-22. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1894 − 22
C1898 Test Methods for Determining the Chemical Resis- ence and activities of bacteria within wastewater collection,
tance of Concrete Products to Acid Attack storage and treatment infrastructure. Also referred to as bio-
C1904 Test Methods for Determination of the Effects of genic sulfuric acid (BSA) corrosion, and biological corrosion
Biogenic Acidification on Concrete Antimicrobial Addi- of concrete, hydrogen sulfide corrosion, microbial corrosion of
tives and/or Concrete Products concrete.
D4262 TestMethodforpHofChemicallyCleanedorEtched
3.2.11 chemical oxidation, n—chemical reaction in which
Concrete Surfaces
the atoms in a molecule lose electrons and the net valence of
D4783 Test Methods for Resistance of Adhesive Prepara-
the molecule is correspondingly increased, commonly associ-
tions in Container toAttack by Bacteria, Yeast, and Fungi
ated with addition of molecular oxygen to the chemical
G21 Practice for Determining Resistance of Synthetic Poly-
composition of an ‘oxidized’ material.
meric Materials to Fungi
3.2.12 sulfate oxidizing bacteria (SOB), n—bacteriathatcan
2.2 Other Standards:
convert hydrogen sulfide (H S) into elemental sulfur (S) by
ISO 22196 Measurement of antibacterial activity on plastics
2–
partial oxidation, or sulfate (SO ).
and other non-porous surfaces
3.2.13 sulfate reducing bacteria (SRB), n—bacteria that can
3. Terminology
obtain energy by oxidizing organic compounds or molecular
hydrogen while reducing sulfate to hydrogen sulfide. Most
3.1 Definitions:
sulfate reducing bacteria can also reduce other oxidized inor-
3.1.1 For definitions of terms used in this practice, refer to
ganic sulfur compounds, such as sulfite, thiosulfate/elemental
Terminology standards C125 and C822.
sulfur. A common mechanism for anaerobic bacterial for
3.2 Definitions of Terms Specific to This Standard:
respiration in the absence of oxygen.
3.2.1 antimicrobial admixture, n—EPA registered chemical
3.2.14 Thiobacillus species (for example, Thiobacillus
admixture that is intended to inhibit microorganism growth
thioparus, Starkeya novella, Halothiobacillus neapolitanus,
(-static effect) or kill microorganisms (-cidal effect). Antimi-
Thiomonas intermedia and Acidithiobacillus thiooxidans.),
crobial admixtures are registered according to the organisms
n—a genus of gram negative bacteria, known for using sulfur
they are effective against and typically, due to their chemical
and sulfur compounds as part of their respiration cycle (sulfur
nature for industrial use, have broad spectrum effectiveness
a.k.a. thio-).
against many organism types, including bacteria, fungi and
3.2.15 turbulence, n—violent or unsteady movement of air
algae.
or water, or of some other fluid.
3.2.2 aerobic bacteria, n—bacteria that have a metabolic
requirement for the presence of available oxygen to grow and
4. Microbially Induced Corrosion (MIC) of Concrete
thrive.
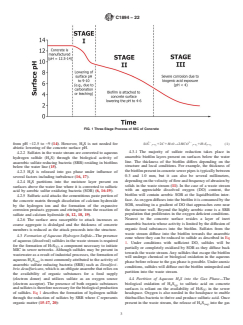
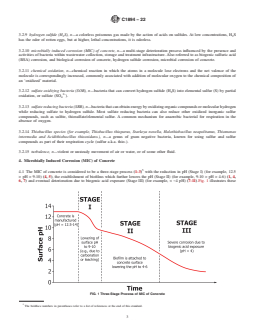
4.1 The MIC of concrete is considered to be a three-stage
3.2.3 anaerobic bacteria, n—bacteria that do not live or
process (1-3) with the reduction in pH (Stage I) (for example,
grow when oxygen is present.
12.5 > pH > 9-10) (4, 5), the establishment of biofilms which
3.2.4 biofilm, n—a complex mixture of established
furtherlowersthepH(StageII)(forexample,9-10>pH>4-6)
microorganisms, microorganism components (extra-cellular
(1, 4, 6, 7) and eventual deterioration due to biogenic acid
matrix) and environmental detritus.
exposure (Stage III) (for example, < ~4 pH) (7-11). Fig. 1
3.2.5 biogenic (biotic) acidification, n—process of produc-
illustrates these stages that have been observed in laboratory
tion of mixture of inorganic and organic acids from respiring
testing.Testing procedures are described that simulate all three
organisms resulting in acidification of the microbial environ-
stages or individual stages. This document clarifies the stages
ment.
where each test applies.
3.2.6 chemical (abiotic) acidification, n—when compounds
4.2 This section provides a brief summary of the commonly
like ammonia, nitrogen oxides and sulphur dioxides are con-
accepted chain of events that lead to the initiation and
verted in a chemical reaction into acidic substances.
propagation of MIC in wastewater collection networks. Addi-
3.2.7 Desulfovibrio desulfuricans, n—anaerobic dissimila- tional details are provided in 4.3.
tory sulfate-reducing bacterium. 4.2.1 Abiotic lowering of the concrete surface pH takes
place before colonization by bacteria can occur. Carbonation,
3.2.8 dissolved oxygen (DO) content, n—oxygen (O ) mol-
the process by which atmospheric carbon dioxide reacts with
ecules available for respiration to aquatic organisms.
calciumhydroxideandwaterwithinthecementmicrostructure,
3.2.9 hydrogen sulfide (H S), n—a colorless poisonous gas
is typically credited with the initial reduction in surface pH of
made by the action of acids on sulfides.At low concentrations,
the concrete. Leaching of calcium hydroxide through contact
H S has the odor of rotten eggs, but at higher, lethal
with wastewater may also lead to a reduction in pH near the
concentrations, it is odorless.
concrete surface (12, 13). It is also claimed that H S undergoes
3.2.10 microbially induced corrosion (MIC) of concrete,
inorganic chemical reaction to lower the initial pH of concrete
n—a multi-stage deterioration process influenced by the pres-
4 5
Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St., The boldface numbers in parentheses refer to a list of references at the end of
4th Floor, New York, NY 10036, http://www.ansi.org. this standard.
C1894 − 22
FIG. 1 Three-Stage Process of MIC of Concrete
22 32
from pH ~12.5 to ~9 (14). However, H S is not needed for SO 12C1H O→2HCO 1H S (1)
4 aq 2 aq 2 aq
2 ~ ! ~ ! ~ !
abiotic lowering of the concrete surface pH.
4.3.1 The majority of sulfate reduction takes place in
4.2.2 Sulfates in the waste stream are converted to aqueous
anaerobic biofilm layers present on surfaces below the water
hydrogen sulfide (H S) through the biological activity of
line. The thickness of the biofilm differs depending on the
anaerobic sulfate reducing bacteria (SRB) residing in biofilms
structure and local conditions. For example, the thickness of
below the water line (15).
thebiofilmpresentinconcretesewerpipesistypicallybetween
4.2.3 H S is released into gas phase under influence of
0.3 and 1.0 mm, but it can also be several millimeters,
several factors including turbulence (16, 17).
dependingonthevelocityofflowandfrequencyofabrasionby
4.2.4 H S partitions into the moisture layer present on
solids in the waste stream (11). In the case of a waste stream
surfaces above the water line where it is converted to sulfuric
with an appreciable dissolved oxygen (DO) content, the
acid by aerobic sulfur oxidizing bacteria (SOB) (6, 16-19).
biofilm will contain aerobic SOB at the liquid/biofilm inter-
4.2.5 Sulfuric acid attacks the cementitious paste portion of
face.As oxygen diffuses into the biofilm it is consumed by the
the concrete matrix through dissolution of calcium hydroxide
SOB, resulting in a gradient of DO that approaches zero near
by the hydrogen ion and the formation of the expansive
the structure wall. Beyond the highly aerobic zone is a SRB
corrosion products gypsum and ettringite from the reaction of
population that proliferates in the oxygen deficient conditions.
sulfate and calcium hydroxide (6, 12, 18, 19).
Nearest to the concrete surface resides a layer of inert
4.2.6 The surface area susceptible to attack increases as
anaerobic bacteria whose activity is limited by the diffusion of
coarse aggregate is dislodged and the thickness of concrete
organic food substances into the biofilm. Sulfates from the
members is reduced as the attack proceeds into the structure.
waste stream diffuse into the biofilm towards the anaerobic
4.3 Formation of Aqueous Hydrogen Sulfide—The presence
zone where they can be reduced to sulfide as described in Eq
of aqueous (dissolved) sulfides in the waste stream is required
1. Under conditions with sufficient DO, sulfides will be
for the formation of H S , a component necessary to initiate
2 (g)
partially or completely oxidized by SOB as they diffuse back
MIC in sewer networks. Although sulfides may be present in
towards the waste stream.Any sulfides that escape the biofilm
wastewater as a result of industrial processes, the formation of
will undergo chemical or biological oxidation in the aqueous
aqueous H S is most commonly attributed to the activity of
2 (aq)
phase before release to the gas phase is possible. Under anoxic
anaerobic sulfate reducing bacteria (SRB) such as Desulfovi-
conditions, sulfides will diffuse out the biofilm unimpeded and
brio desulfuricans, which is an obligate anaerobe that relies on
partition into the waste stream.
the availability of organic substances for a food supply
(electron donor) and utilizes sulfate as an oxygen source 4.4 Partition of Aqueous H S into the Gas Phase—The
(electron acceptor). The presence of both organic substances biological oxidation of H S to sulfuric acid on concrete
2 (g)
andsulfatesisthereforenecessaryforthebiologicalproduction surfaces is reliant on the availability of H S in the sewer
2 (g)
of sulfides. Eq 1 describes the formation of hydrogen sulfide headspace. Oxygen is also needed in the headspace to enable
through the reduction of sulfates by SRB where C represents thiobacillus bacteria to thrive and produce sulfuric acid. Once
organic matter (15-17, 20): present in the waste stream, the release of H S into the gas
2 (aq)
C1894 − 22
phase will be heavily influenced by the pH of the wastewater, 4.6.1 The end product of the oxidation of H S by SOB is
the equilibrium conditions between gas and liquid phases, sulfuric acid. The chemical composition of hydrated portland
temperature, and the turbulence of the flow. Ventilation condi- cement makes concrete susceptible to degradation when ex-
tions above the water line will influence the sustained concen- posed to acidic conditions. The volume occupied by hydrated
tration of H S in the headspace. cement paste is generally composed of the following propor-
2 (g)
tions of four solid products: 50-60 % calcium silica hydrate
4.5 Oxidation of H S(g) to Sulfuric Acid—Once present in
(C-S-H), 20-25 % calcium hydroxide (CH), 15-20 % calcium
the sewer headspace, H S is free to partition into moisture
2 (g)
sulfoaluminates, and varying amounts of unhydrated cement
films present on surfaces above the water line. Back in
grains. Exposure to acid results in the decalcification of these
solution, H S is subject to both biological and chemical
2 (aq)
hydrated products, beginning with CH, and the eventual
conversion to multiple oxidation states, ultimately leading to
breakdownofthemicrostructureresultinginincreasedporosity
the production of sulfuric acid and the corrosion of the cement
and decrease in mechanical properties. After decalcification,
paste portion of the concrete matrix. Sulfur oxidation states are
calcium ions either diffuse out of the microstructure or com-
dependent on the local pH and the type and activity of SOB
bine with the salt of the acid to form insoluble calcium salts of
present. Several Thiobacillus species have been identified as
little structural value. The presence of these products results in
contributors to MIC in concrete wastewater networks. Multiple
the formation of a porous layer on the concrete surface.
species may be present on sewer walls at pH values of 3.0-8.0.
Degradation continues as hydrated products become more
Thiobacillus thioparus makes use of sulfides, elemental sulfur
unstable with decreasing alkalinity in the system. The degra-
and thiosulfate in the production of sulfuric acid. Thiomonas
dation mechanisms and severity of acid attack on concrete are
intermedia and Starkeya novella are the next species to
dependent on the type of attack, strength and type of acid. The
colonize the surface, relying mainly on thiosulfate as a sub-
ability of concrete to resist acid attack is related to acid
strate.As pH is reduced to below 7, Halothiobacillus neapoli-
neutralization capacity, composition of hydrated products, and
tanus becomes prevalent until surface pH is reduced to around
porosity.
3. Being highly acidophilic, Acidithiobacillus thiooxidans
4.6.2 The following reactions summarize the decalcification
thrivesatpHvaluesbelow3whereitoxidizesbothsulfidesand
of C-S-H gel (Eq 2) and the dissolution of calcium hydroxide
elemental sulfur to sulfuric acid. Acidithiobacillus thiooxidans
by sulfuric acid (Eq 3) to form gypsum, as well as the
continuetolowerthesurfacepHuntilacidproductionbecomes
formation of ettringite (Eq 4 and 5) (21):
self-inhibitory at pH values from 0.5 to 1.0. Preferred sub-
strates and pH ranges for SOB involved in MIC in concrete 3CaO·2SiO ·3H O13H SO→3~CaSO ·2H O!12SiO (2)
2 2 2 4 4 2 2
sewers are given in Table 1.
Ca~OH! 1H SO→CaSO ·2H O (3)
2 2 4 4 2
4.5.1 Fresh concrete is highly alkaline, often exhibiting pH
3 CaSO ·2H O 13CaO·Al O
~ !
4 2 2 3
between 12.5 and 14 (13). Abiotic lowering of the concrete
126H O→3CaO·Al O ·3CaSO ·32H O (4)
2 2 3 4 2
surface pH is therefore necessary before colonization by
Thiobacillus can occur. Carbonation, the process by which 2~CaSO ·2H O!13CaO·Al O ·CaSO ·12H O
4 2 2 3 4 2
atmospheric carbon dioxide reacts with calcium hydroxide and
116H O→3CaO·Al O ·3CaSO ·32H O (5)
2 2 3 4 2
water within the cement microstructure, is typically credited
with the initial reduction in surface pH of the concrete.
5. Test Methods for Evaluating Concrete Resistance to
Leaching of calcium hydroxide through contact with wastewa-
MIC
ter (12, 13)orinorganicreactionofH Sontheconcretesurface
5.1 Laboratory Investigations:
(14) may also lead to a reduction in pH near the concrete
5.1.1 General—Reproducing MIC in the laboratory is one
surface. Once the concrete surface reaches a pH value of 9-10,
way to investigate specific mechanisms of attack or evaluate
colonization by SOB can potentially begin. After SOB are
the corrosion resistance of cementitious materials; however,
established, abiotic lowering of the concrete pH is no longer
the complex nature of MIC makes laboratory reproduction and
relevant as biological production of sulfuric acid governs the
the design of straightforward testing techniques difficult. The
surface pH. Initial SOB colonization is followed by a succes-
use of microorganisms requires knowledge of microbiology
sive establishment of more acidophilic species of Thiobacillus.
and introduces a level of variability that makes repeatability of
4.6 Acid Degradation of Cementitious Systems:
experimental conditions difficult to achieve.
5.1.2 Materials:
5.1.2.1 Materials that are described here are intended to be
used with both chemical and biogenic acidification tests.
TABLE 1 Preferred Substrates and pH Ranges for SOB Involved
Modifications and exceptions to these sections are provided
with MIC in Concrete Sewers
under each test method.
Species Preferred Substrate Preferred pH Growth
5.1.2.2 Concrete—Unless otherwise specified by a specific
Range
0 2–
Thiobacillus thioparus H S, S ,S O 5-9
2 2 3
test method, concrete can be prepared following Practice
2–
Starkeya novella S O 2.5-8
2 3
C192/C192M, Practice C31/C31M, or obtained from existing
2–
Thiomonas intermedia S O 2.5-8
2 3
0 2–
Halothiobacillus S ,S O 3-7 structures following Test Method C42/C42M, Test Methods
2 3
neopolitanus
C497.Othermethodsofconcreteproductionorextractionfr
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: C1894 − 19 C1894 − 22
Standard Guide for
Microbially Induced Corrosion of Concrete Products
This standard is issued under the fixed designation C1894; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide discusses microbially induced corrosion (MIC) of concrete products and laboratory test methods for determining
the resistance of concrete to MIC. Although the guide is intended for concrete products, it also covers cementitious mortar and
paste that are used in specialized applications or laboratory investigations.
1.2 While this guide discusses concrete materials and admixtures, the document is not intended to specifically address field
exposure conditions or sewage pipe, concrete tank, or concrete riser network design.
1.3 This guide does not cover live trial tests where concrete coupons or other specimens are monitored in sewers.
1.4 This guide does not cover concrete deterioration due to chemical sulfate attack, which is caused by the reaction of sulfate
compounds that exist in wastewater with the hydration products of cement. Test methods for assessing sulfate attack are provided
by Test Methods C452 and C1012/C1012M.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 The text of this guide references notes and footnotes that provide explanatory material. These notes and footnotes (excluding
those in tables and figures) shall not be considered as requirements of the standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C31/C31M Practice for Making and Curing Concrete Test Specimens in the Field
C33/C33M Specification for Concrete Aggregates
C42/C42M Test Method for Obtaining and Testing Drilled Cores and Sawed Beams of Concrete
This test method is under the jurisdiction of ASTM Committee C13 on Concrete Pipe and is the direct responsibility of Subcommittee C13.03 on Determining the Effects
of Biogenic Sulfuric Acid on Concrete Pipe and Structures.
Current edition approved Oct. 1, 2019March 1, 2022. Published October 2019March 2022. Originally approved in 2019. Last previous edition approved in 2019 as
C1570 – 19. DOI: 10.1520/C1894-1910.1520/C1894-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
C1894 − 22
C125 Terminology Relating to Concrete and Concrete Aggregates
C150/C150M Specification for Portland Cement
C192/C192M Practice for Making and Curing Concrete Test Specimens in the Laboratory
C260/C260M Specification for Air-Entraining Admixtures for Concrete
C267 Test Methods for Chemical Resistance of Mortars, Grouts, and Monolithic Surfacings and Polymer Concretes
C294 Descriptive Nomenclature for Constituents of Concrete Aggregates
C452 Test Method for Potential Expansion of Portland-Cement Mortars Exposed to Sulfate
C494/C494M Specification for Chemical Admixtures for Concrete
C497 Test Methods for Concrete Pipe, Concrete Box Sections, Manhole Sections, or Tile
C595/C595M Specification for Blended Hydraulic Cements
C618 Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete
C822 Terminology Relating to Concrete Pipe and Related Products
C989/C989M Specification for Slag Cement for Use in Concrete and Mortars
C1012/C1012M Test Method for Length Change of Hydraulic-Cement Mortars Exposed to a Sulfate Solution
C1017/C1017M Specification for Chemical Admixtures for Use in Producing Flowing Concrete (Withdrawn 2022)
C1240 Specification for Silica Fume Used in Cementitious Mixtures
C1600/C1600M Specification for Rapid Hardening Hydraulic Cement
C1898 Test Methods for Determining the Chemical Resistance of Concrete Products to Acid Attack
C1904 Test Methods for Determination of the Effects of Biogenic Acidification on Concrete Antimicrobial Additives and/or
Concrete Products
D4262 Test Method for pH of Chemically Cleaned or Etched Concrete Surfaces
D4783 Test Methods for Resistance of Adhesive Preparations in Container to Attack by Bacteria, Yeast, and Fungi
G21 Practice for Determining Resistance of Synthetic Polymeric Materials to Fungi
2.2 Other Standards:
ISO 22196 Measurement of antibacterial activity on plastics and other non-porous surfaces
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this practice, refer to Terminology standards C125 and C822.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 antimicrobial admixture, n—EPA registered chemical admixture that is intended to inhibit microorganism growth (-static
effect) or kill microorganisms (-cidal effect). Antimicrobial admixtures are registered according to the organisms they are effective
against and typically, due to their chemical nature for industrial use, have broad spectrum effectiveness against many organism
types, including bacteria, fungi and algae.
3.2.2 aerobic bacteria, n—bacteria that have a metabolic requirement for the presence of available oxygen to grow and thrive.
3.2.3 anaerobic bacteria, n—bacteria that do not live or grow when oxygen is present.
3.2.4 biofilm, n—a complex mixture of established microorganisms, microorganism components (extra-cellular matrix) and
environmental detritus.
3.2.5 biogenic (biotic) acidification, n—process of production of mixture of inorganic and organic acids from respiring organisms
resulting in acidification of the microbial environment.
3.2.6 chemical (abiotic) acidification, n—when compounds like ammonia, nitrogen oxides and sulphur dioxides are converted in
a chemical reaction into acidic substances.
3.2.7 Desulfovibrio desulfuricans, n—anaerobic dissimilatory sulfate-reducing bacterium.
3.2.8 dissolved oxygen (DO) content, n—oxygen (O ) molecules available for respiration to aquatic organisms.
The last approved version of this historical standard is referenced on www.astm.org.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
C1894 − 22
3.2.9 hydrogen sulfide (H S), n—a colorless poisonous gas made by the action of acids on sulfides. At low concentrations, H S
2 2
has the odor of rotten eggs, but at higher, lethal concentrations, it is odorless.
3.2.10 microbially induced corrosion (MIC) of concrete, n—a multi-stage deterioration process influenced by the presence and
activities of bacteria within wastewater collection, storage and treatment infrastructure. Also referred to as biogenic sulfuric acid
(BSA) corrosion, and biological corrosion of concrete, hydrogen sulfide corrosion, microbial corrosion of concrete.
3.2.11 chemical oxidation, n—chemical reaction in which the atoms in a molecule lose electrons and the net valence of the
molecule is correspondingly increased, commonly associated with addition of molecular oxygen to the chemical composition of
an ‘oxidized’ material.
3.2.12 sulfate oxidizing bacteria (SOB), n—bacteria that can convert hydrogen sulfide (H S) into elemental sulfur (S) by partial
2–
oxidation, or sulfate (SO ).
3.2.13 sulfate reducing bacteria (SRB), n—bacteria that can obtain energy by oxidizing organic compounds or molecular hydrogen
while reducing sulfate to hydrogen sulfide. Most sulfate reducing bacteria can also reduce other oxidized inorganic sulfur
compounds, such as sulfite, thiosulfate/elemental sulfur. A common mechanism for anaerobic bacterial for respiration in the
absence of oxygen.
3.2.14 Thiobacillus species (for example, Thiobacillus thioparus, Starkeya novella, Halothiobacillus neapolitanus, Thiomonas
intermedia and Acidithiobacillus thiooxidans.), n—a genus of gram negative bacteria, known for using sulfur and sulfur
compounds as part of their respiration cycle (sulfur a.k.a. thio-).
3.2.15 turbulence, n—violent or unsteady movement of air or water, or of some other fluid.
4. Microbially Induced Corrosion (MIC) of Concrete
4.1 The MIC of concrete is considered to be a three-stage process (1-3) with the reduction in pH (Stage I) (for example, 12.5
> pH > 9-10) (4, 5), the establishment of biofilms which further lowers the pH (Stage II) (for example, 9-10 > pH > 4-6) (1, 4,
6, 7) and eventual deterioration due to biogenic acid exposure (Stage III) (for example, < ~4 pH) (7-11).Fig. 1 illustrates these
FIG. 1 Three-Stage Process of MIC of Concrete
The boldface numbers in parentheses refer to a list of references at the end of this standard.
C1894 − 22
stages that have been observed in laboratory testing. Testing procedures are described that simulate all three stages or individual
stages. This document clarifies the stages where each test applies.
4.2 This section provides a brief summary of the commonly accepted chain of events that lead to the initiation and propagation
of MIC in wastewater collection networks. Additional details are provided in 4.3.
4.2.1 Abiotic lowering of the concrete surface pH takes place before colonization by bacteria can occur. Carbonation, the process
by which atmospheric carbon dioxide reacts with calcium hydroxide and water within the cement microstructure, is typically
credited with the initial reduction in surface pH of the concrete. Leaching of calcium hydroxide through contact with wastewater
may also lead to a reduction in pH near the concrete surface (12, 13). It is also claimed that H S undergoes inorganic chemical
reaction to lower the initial pH of concrete from pH ~12.5 to ~9 (14). However, H S is not needed for abiotic lowering of the
concrete surface pH.
4.2.2 Sulfates in the waste stream are converted to aqueous hydrogen sulfide (H S) through the biological activity of anaerobic
sulfate reducing bacteria (SRB) residing in biofilms below the water line (15).
4.2.3 H S is released into gas phase under influence of several factors including turbulence (16, 17).
4.2.4 H S partitions into the moisture layer present on surfaces above the water line where it is converted to sulfuric acid by
aerobic sulfur oxidizing bacteria (SOB) (6, 16-19).
4.2.5 Sulfuric acid attacks the cementitious paste portion of the concrete matrix through dissolution of calcium hydroxide by the
hydrogen ion and the formation of the expansive corrosion products gypsum and ettringite from the reaction of sulfate and calcium
hydroxide (6, 12, 18, 19).
4.2.6 The surface area susceptible to attack increases as coarse aggregate is dislodged and the thickness of concrete members is
reduced as the attack proceeds into the structure.
4.3 Formation of Aqueous Hydrogen Sulfide—The presence of aqueous (dissolved) sulfides in the waste stream is required for the
formation of H S , a component necessary to initiate MIC in sewer networks. Although sulfides may be present in wastewater
2 (g)
as a result of industrial processes, the formation of aqueous H S is most commonly attributed to the activity of anaerobic sulfate
2 (aq)
reducing bacteria (SRB) such as Desulfovibrio desulfuricans, which is an obligate anaerobe that relies on the availability of organic
substances for a food supply (electron donor) and utilizes sulfate as an oxygen source (electron acceptor). The presence of both
organic substances and sulfates is therefore necessary for the biological production of sulfides. Eq 1 describes the formation of
hydrogen sulfide through the reduction of sulfates by SRB where C represents organic matter (15-17, 20):
22 32
SO 12C1H O→2HCO 1H S (1)
4 a q 2 a q 2 a q
~ ! ~ ! ~ !
4.3.1 The majority of sulfate reduction takes place in anaerobic biofilm layers present on surfaces below the water line. The
thickness of the biofilm differs depending on the structure and local conditions. For example, the thickness of the biofilm present
in concrete sewer pipes is typically between 0.3 and 1.0 mm, but it can also be several millimeters, depending on the velocity of
flow and frequency of abrasion by solids in the waste stream (11). In the case of a waste stream with an appreciable dissolved
oxygen (DO) content, the biofilm will contain aerobic SOB at the liquid/biofilm interface. As oxygen diffuses into the biofilm it
is consumed by the SOB, resulting in a gradient of DO that approaches zero near the structure wall. Beyond the highly aerobic
zone is a SRB population that proliferates in the oxygen deficient conditions. Nearest to the concrete surface resides a layer of inert
anaerobic bacteria whose activity is limited by the diffusion of organic food substances into the biofilm. Sulfates from the waste
stream diffuse into the biofilm towards the anaerobic zone where they can be reduced to sulfide as described in Eq 1. Under
conditions with sufficient DO, sulfides will be partially or completely oxidized by SOB as they diffuse back towards the waste
stream. Any sulfides that escape the biofilm will undergo chemical or biological oxidation in the aqueous phase before release to
the gas phase is possible. Under anoxic conditions, sulfides will diffuse out the biofilm unimpeded and partition into the waste
stream.
4.4 Partition of Aqueous H S into the Gas Phase—The biological oxidation of H S to sulfuric acid on concrete surfaces is reliant
2 2 (g)
on the availability of H S in the sewer headspace. Oxygen is also needed in the headspace to enable thiobacillus bacteria to thrive
2 (g)
and produce sulfuric acid. Once present in the waste stream, the release of H S into the gas phase will be heavily influenced
2 (aq)
by the pH of the wastewater, the equilibrium conditions between gas and liquid phases, temperature, and the turbulence of the flow.
Ventilation conditions above the water line will influence the sustained concentration of H S in the headspace.
2 (g)
C1894 − 22
4.5 Oxidation of H S(g) to Sulfuric Acid—Once present in the sewer headspace, H S is free to partition into moisture films
2 2 (g)
present on surfaces above the water line. Back in solution, H S is subject to both biological and chemical conversion to multiple
2 (aq)
oxidation states, ultimately leading to the production of sulfuric acid and the corrosion of the cement paste portion of the concrete
matrix. Sulfur oxidation states are dependent on the local pH and the type and activity of SOB present. Several Thiobacillus species
have been identified as contributors to MIC in concrete wastewater networks. Multiple species may be present on sewer walls at
pH values of 3.0-8.0. Thiobacillus thioparus makes use of sulfides, elemental sulfur and thiosulfate in the production of sulfuric
acid. Thiomonas intermedia and Starkeya novella are the next species to colonize the surface, relying mainly on thiosulfate as a
substrate. As pH is reduced to below 7, Halothiobacillus neapolitanus becomes prevalent until surface pH is reduced to around
3. Being highly acidophilic, Acidithiobacillus thiooxidans thrives at pH values below 3 where it oxidizes both sulfides and
elemental sulfur to sulfuric acid. Acidithiobacillus thiooxidans continue to lower the surface pH until acid production becomes
self-inhibitory at pH values from 0.5 to 1.0. Preferred substrates and pH ranges for SOB involved in MIC in concrete sewers are
given in Table 1.
4.5.1 Fresh concrete is highly alkaline, often exhibiting pH between 12.5 and 14 (13). Abiotic lowering of the concrete surface
pH is therefore necessary before colonization by Thiobacillus can occur. Carbonation, the process by which atmospheric carbon
dioxide reacts with calcium hydroxide and water within the cement microstructure, is typically credited with the initial reduction
in surface pH of the concrete. Leaching of calcium hydroxide through contact with wastewater (12, 13) or inorganic reaction of
H S on the concrete surface (14) may also lead to a reduction in pH near the concrete surface. Once the concrete surface reaches
a pH value of 9-10, colonization by SOB can potentially begin. After SOB are established, abiotic lowering of the concrete pH
is no longer relevant as biological production of sulfuric acid governs the surface pH. Initial SOB colonization is followed by a
successive establishment of more acidophilic species of Thiobacillus.
4.6 Acid Degradation of Cementitious Systems:
4.6.1 The end product of the oxidation of H S by SOB is sulfuric acid. The chemical composition of hydrated portland cement
makes concrete susceptible to degradation when exposed to acidic conditions. The volume occupied by hydrated cement paste is
generally composed of the following proportions of four solid products: 50-60 % calcium silica hydrate (C-S-H), 20-25 % calcium
hydroxide (CH), 15-20 % calcium sulfoaluminates, and varying amounts of unhydrated cement grains. Exposure to acid results in
the decalcification of these hydrated products, beginning with CH, and the eventual breakdown of the microstructure resulting in
increased porosity and decrease in mechanical properties. After decalcification, calcium ions either diffuse out of the microstructure
or combine with the salt of the acid to form insoluble calcium salts of little structural value. The presence of these products results
in the formation of a porous layer on the concrete surface. Degradation continues as hydrated products become more unstable with
decreasing alkalinity in the system. The degradation mechanisms and severity of acid attack on concrete are dependent on the type
of attack, strength and type of acid. The ability of concrete to resist acid attack is related to acid neutralization capacity,
composition of hydrated products, and porosity.
4.6.2 The following reactions summarize the decalcification of C-S-H gel (Eq 2) and the dissolution of calcium hydroxide by
sulfuric acid (Eq 3) to form gypsum, as well as the formation of ettringite (Eq 4 and 5) (21):
3CaO·2SiO ·3H O13H SO →3 CaSO · 2H O 12SiO (2)
~ !
2 2 2 4 4 2 2
Ca O H 1H SO →CaSO ·2H O (3)
~ !
2 4 4 2
3~CaSO · 2H O!13CaO·Al O 126H O→3CaO·Al O ·3CaSO ·32H O (4)
4 2 2 3 2 2 3 4 2
2~CaSO · 2H O!13CaO·Al O ·CaSO ·12H O116H O→3CaO·Al O ·3CaSO ·32H O (5)
4 2 2 3 4 2 2 2 3 4 2
TABLE 1 Preferred Substrates and pH Ranges for SOB Involved
with MIC in Concrete Sewers
Species Preferred Substrate Preferred pH Growth
Range
0 2–
Thiobacillus thioparus H S, S , S O 5-9
2 2 3
2–
Starkeya novella S O 2.5-8
2 3
2–
Thiomonas intermedia S O 2.5-8
2 3
0 2–
Halothiobacillus S , S O 3-7
2 3
neopolitanus
Acidithiobacillus H S, SO 0.5-3
thiooxidans
C1894 − 22
5. Test Methods for Evaluating Concrete Resistance to MIC
5.1 Laboratory Investigations:
5.1.1 General—Reproducing MIC in the laboratory is one way to investigate specific mechanisms of attack or evaluate the
corrosion resistance of cementitious materials; however, the complex nature of MIC makes laboratory reproduction and the design
of straightforward testing techniques difficult. The use of microorganisms requires knowledge of microbiology and introduces a
level of variability that makes repeatability of experimental conditions difficult to achieve.
5.1.2 Materials:
5.1.2.1 Materials that are described here are intended to be used with both chemical and biogenic acidification tests. Modifications
and exceptions to these sections are provided under each test method.
5.1.2.2 Concrete—Unless otherwise specified by a specific test method, concrete can be prepared following Practice C192/
C192M, Practice C31/C31M, or obtained from existing structures following Test Method C42/C42M, Test Methods C497. Other
methods of concrete production or extraction from existing structures are possible, as long as these procedures and applicable
standards are specified as part of the reporting process. Mixture proportions and curing methodology shall also be documented if
procedures described in the cited standards are not followed.
5.1.2.3 Cementitious Materials—Concrete may contain ASTM C150/C150M portland cements (non-air entrained), ASTM
C595/C595M blended portland cements, and ASTM C1600/C1600M rapid hardening hydraulic cements. Additionally, supple-
mentary cementitious materials (SCM) may be added to non-air entrained portland cements following Specification C150/C150M.
These SCM include ASTM C618 coal fly ash and raw and calcined natural pozzolans for use in concrete, ASTM C989/C989M
slag cement for use in concrete and mortars, and ASTM C1240 silica fume used in cementitious mixtures. Material specification
reports for all cements and SCM shall be part of the reporting process.
5.1.2.4 Admixtur
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...