ASTM E263-18
(Test Method)Standard Test Method for Measuring Fast-Neutron Reaction Rates by Radioactivation of Iron
Standard Test Method for Measuring Fast-Neutron Reaction Rates by Radioactivation of Iron
SIGNIFICANCE AND USE
5.1 Refer to Guide E844 for guidance on the selection, irradiation, and quality control of neutron dosimeters.
5.2 Refer to Practice E261 for a general discussion of the determination of fast-neutron fluence rate with threshold detectors.
5.3 Pure iron in the form of foil or wire is readily available and easily handled.
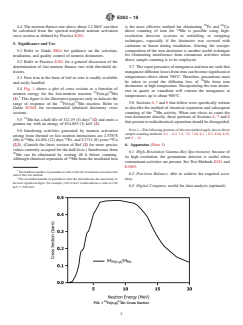
5.4 Fig. 1 shows a plot of cross section as a function of neutron energy for the fast-neutron reaction 54Fe(n,p)54Mn (1).3 This figure is for illustrative purposes only to indicate the range of response of the 54Fe(n,p)54Mn reaction. Refer to Guide E1018 for recommended tabulated dosimetry cross sections.
FIG. 1 54Fe(n,p)54Mn Cross Section
5.5 54Mn has a half-life of 312.19 (3) days4 (2) and emits a gamma ray with an energy of 834.855 (3) keV (2).
5.6 Interfering activities generated by neutron activation arising from thermal or fast neutron interactions are 2.57878 (46)-h 56Mn, 44.494 (12) days 59Fe, and 5.2711 (8) years 60Co (2,3). (Consult the latest version of Ref (2) for more precise values currently accepted for the half-lives.) Interference from 56Mn can be eliminated by waiting 48 h before counting. Although chemical separation of 54Mn from the irradiated iron is the most effective method for eliminating 59Fe and 60Co, direct counting of iron for 54Mn is possible using high-resolution detector systems or unfolding or stripping techniques, especially if the dosimeter was covered with cadmium or boron during irradiation. Altering the isotopic composition of the iron dosimeter is another useful technique for eliminating interference from extraneous activities when direct sample counting is to be employed.
5.7 The vapor pressures of manganese and iron are such that manganese diffusion losses from iron can become significant at temperatures above about 700°C. Therefore, precautions must be taken to avoid the diffusion loss of 54Mn from iron dosimeters at high temperature. Encapsulating the iron dosimeter in quartz o...
SCOPE
1.1 This test method describes procedures for measuring reaction rates by the activation reaction 54Fe(n,p)54Mn.
1.2 This activation reaction is useful for measuring neutrons with energies above approximately 2.2 MeV and for irradiation times up to about three years, provided that the analysis methods described in Practice E261 are followed. If dosimeters are analyzed after irradiation periods longer than three years, the information inferred about the fluence during irradiation periods more than three years before the end of the irradiation should not be relied upon without supporting data from dosimeters withdrawn earlier.
1.3 With suitable techniques, fission-neutron fluence rates above 108 cm−2·s−1 can be determined. However, in the presence of a high thermal-neutron fluence rate (for example, >2 × 1014 cm−2·s −1) 54Mn depletion should be investigated.
1.4 Detailed procedures describing the use of other fast-neutron detectors are referenced in Practice E261.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2018
- Technical Committee
- E10 - Nuclear Technology and Applications
- Drafting Committee
- E10.05 - Nuclear Radiation Metrology
Relations
- Effective Date
- 01-Dec-2018
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Jun-2018
- Effective Date
- 01-Jun-2017
- Effective Date
- 01-Oct-2016
- Effective Date
- 15-Feb-2016
- Effective Date
- 01-Sep-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Jun-2015
- Effective Date
- 15-Mar-2015
- Effective Date
- 15-Oct-2014
- Effective Date
- 01-Sep-2014
- Refers
ASTM E844-09(2014)e2 - Standard Guide for Sensor Set Design and Irradiation for Reactor Surveillance - Effective Date
- 01-Jun-2014
- Effective Date
- 01-Jun-2014
Overview
ASTM E263-18, titled Standard Test Method for Measuring Fast-Neutron Reaction Rates by Radioactivation of Iron, defines methodologies for determining fast-neutron reaction rates using the activation reaction 54Fe(n,p)54Mn. Developed by ASTM International, this standard provides precise guidance for employing high-purity iron foils or wires as neutron dosimeters, enabling accurate measurement of fast-neutron fluence rates in nuclear environments. This test method applies to applications needing assessment of neutron fields, surveillance of reactor components, and nuclear facility quality assurance.
The standard supports safe and accurate neutron dosimetry, ensuring compliance with internationally recognized best practices. It is especially useful when evaluating neutron energies above approximately 2.2 MeV, with recommended irradiation periods up to about three years.
Key Topics
Fast-Neutron Reaction Measurement
The method centers on the radioactivation of 54Fe, resulting in the formation of radioactive 54Mn, which emits a characteristic gamma ray. The quantity of 54Mn produced reflects the fast-neutron fluence.Iron Dosimeter Preparation
High-purity iron samples, in foil or wire form, are irradiated and later analyzed via gamma-spectrometry or chemical separation techniques to quantify 54Mn.Interference and Decontamination
The standard addresses the elimination of potential measurement interferences by:- Allowing short-lived isotopes, such as 56Mn, to decay prior to measurement
- Using chemical separation or direct counting with high-resolution detectors to distinguish 54Mn from other activation products (e.g., 59Fe, 60Co)
Temperature Precautions
To prevent diffusion losses of manganese, special attention is given to dosimeter encapsulation, especially at temperatures above 700°C.Data Analysis and Reporting
Includes guidance on measurement, uncertainty estimation, and proper reporting using referenced ASTM practices.
Applications
ASTM E263-18 finds practical application in various sectors where fast-neutron measurement is crucial, such as:
Reactor Surveillance
- Monitoring neutron exposure in nuclear reactor vessels and associated structures
- Support for lifespan assessment and embrittlement studies
Neutron Field Characterization
- Quality assurance and validation of neutron spectra in research and power reactors
- Critical for routine safety assessments and calibration of other detectors
Nuclear Facility Quality Control
- Verifying shielding integrity and neutron flux distributions
- Supporting compliance with regulatory requirements regarding radiation protection
Research and Development
- Benchmarking neutron dosimetry and supporting material science studies involving neutron irradiation
Related Standards
For a comprehensive approach to neutron dosimetry and nuclear measurement, ASTM E263-18 should be used in conjunction with several related standards:
- ASTM E844 – Guide for Sensor Set Design and Irradiation for Reactor Surveillance
- ASTM E261 – Practice for Determining Neutron Fluence, Fluence Rate, and Spectra by Radioactivation Techniques
- ASTM E1018 – Guide for Application of ASTM Evaluated Cross Section Data File
- ASTM E181 – Test Methods for Detector Calibration and Analysis of Radionuclides
- ASTM E944 – Guide for Application of Neutron Spectrum Adjustment Methods in Reactor Surveillance
- ASTM E1005 – Test Method for Application and Analysis of Radiometric Monitors for Reactor Vessel Surveillance
Keywords: fast neutron detector, iron activation, neutron reaction rate, neutron dosimetry, Fe(n,p), ASTM E263, nuclear reactor surveillance, gamma spectrometry, radiation measurement standards.
Buy Documents
ASTM E263-18 - Standard Test Method for Measuring Fast-Neutron Reaction Rates by Radioactivation of Iron
REDLINE ASTM E263-18 - Standard Test Method for Measuring Fast-Neutron Reaction Rates by Radioactivation of Iron
Get Certified
Connect with accredited certification bodies for this standard

DNV
DNV is an independent assurance and risk management provider.

Lloyd's Register
Lloyd's Register is a global professional services organisation specialising in engineering and technology.

DNV Energy Systems
Energy and renewable energy certification.
Sponsored listings
Frequently Asked Questions
ASTM E263-18 is a standard published by ASTM International. Its full title is "Standard Test Method for Measuring Fast-Neutron Reaction Rates by Radioactivation of Iron". This standard covers: SIGNIFICANCE AND USE 5.1 Refer to Guide E844 for guidance on the selection, irradiation, and quality control of neutron dosimeters. 5.2 Refer to Practice E261 for a general discussion of the determination of fast-neutron fluence rate with threshold detectors. 5.3 Pure iron in the form of foil or wire is readily available and easily handled. 5.4 Fig. 1 shows a plot of cross section as a function of neutron energy for the fast-neutron reaction 54Fe(n,p)54Mn (1).3 This figure is for illustrative purposes only to indicate the range of response of the 54Fe(n,p)54Mn reaction. Refer to Guide E1018 for recommended tabulated dosimetry cross sections. FIG. 1 54Fe(n,p)54Mn Cross Section 5.5 54Mn has a half-life of 312.19 (3) days4 (2) and emits a gamma ray with an energy of 834.855 (3) keV (2). 5.6 Interfering activities generated by neutron activation arising from thermal or fast neutron interactions are 2.57878 (46)-h 56Mn, 44.494 (12) days 59Fe, and 5.2711 (8) years 60Co (2,3). (Consult the latest version of Ref (2) for more precise values currently accepted for the half-lives.) Interference from 56Mn can be eliminated by waiting 48 h before counting. Although chemical separation of 54Mn from the irradiated iron is the most effective method for eliminating 59Fe and 60Co, direct counting of iron for 54Mn is possible using high-resolution detector systems or unfolding or stripping techniques, especially if the dosimeter was covered with cadmium or boron during irradiation. Altering the isotopic composition of the iron dosimeter is another useful technique for eliminating interference from extraneous activities when direct sample counting is to be employed. 5.7 The vapor pressures of manganese and iron are such that manganese diffusion losses from iron can become significant at temperatures above about 700°C. Therefore, precautions must be taken to avoid the diffusion loss of 54Mn from iron dosimeters at high temperature. Encapsulating the iron dosimeter in quartz o... SCOPE 1.1 This test method describes procedures for measuring reaction rates by the activation reaction 54Fe(n,p)54Mn. 1.2 This activation reaction is useful for measuring neutrons with energies above approximately 2.2 MeV and for irradiation times up to about three years, provided that the analysis methods described in Practice E261 are followed. If dosimeters are analyzed after irradiation periods longer than three years, the information inferred about the fluence during irradiation periods more than three years before the end of the irradiation should not be relied upon without supporting data from dosimeters withdrawn earlier. 1.3 With suitable techniques, fission-neutron fluence rates above 108 cm−2·s−1 can be determined. However, in the presence of a high thermal-neutron fluence rate (for example, >2 × 1014 cm−2·s −1) 54Mn depletion should be investigated. 1.4 Detailed procedures describing the use of other fast-neutron detectors are referenced in Practice E261. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 Refer to Guide E844 for guidance on the selection, irradiation, and quality control of neutron dosimeters. 5.2 Refer to Practice E261 for a general discussion of the determination of fast-neutron fluence rate with threshold detectors. 5.3 Pure iron in the form of foil or wire is readily available and easily handled. 5.4 Fig. 1 shows a plot of cross section as a function of neutron energy for the fast-neutron reaction 54Fe(n,p)54Mn (1).3 This figure is for illustrative purposes only to indicate the range of response of the 54Fe(n,p)54Mn reaction. Refer to Guide E1018 for recommended tabulated dosimetry cross sections. FIG. 1 54Fe(n,p)54Mn Cross Section 5.5 54Mn has a half-life of 312.19 (3) days4 (2) and emits a gamma ray with an energy of 834.855 (3) keV (2). 5.6 Interfering activities generated by neutron activation arising from thermal or fast neutron interactions are 2.57878 (46)-h 56Mn, 44.494 (12) days 59Fe, and 5.2711 (8) years 60Co (2,3). (Consult the latest version of Ref (2) for more precise values currently accepted for the half-lives.) Interference from 56Mn can be eliminated by waiting 48 h before counting. Although chemical separation of 54Mn from the irradiated iron is the most effective method for eliminating 59Fe and 60Co, direct counting of iron for 54Mn is possible using high-resolution detector systems or unfolding or stripping techniques, especially if the dosimeter was covered with cadmium or boron during irradiation. Altering the isotopic composition of the iron dosimeter is another useful technique for eliminating interference from extraneous activities when direct sample counting is to be employed. 5.7 The vapor pressures of manganese and iron are such that manganese diffusion losses from iron can become significant at temperatures above about 700°C. Therefore, precautions must be taken to avoid the diffusion loss of 54Mn from iron dosimeters at high temperature. Encapsulating the iron dosimeter in quartz o... SCOPE 1.1 This test method describes procedures for measuring reaction rates by the activation reaction 54Fe(n,p)54Mn. 1.2 This activation reaction is useful for measuring neutrons with energies above approximately 2.2 MeV and for irradiation times up to about three years, provided that the analysis methods described in Practice E261 are followed. If dosimeters are analyzed after irradiation periods longer than three years, the information inferred about the fluence during irradiation periods more than three years before the end of the irradiation should not be relied upon without supporting data from dosimeters withdrawn earlier. 1.3 With suitable techniques, fission-neutron fluence rates above 108 cm−2·s−1 can be determined. However, in the presence of a high thermal-neutron fluence rate (for example, >2 × 1014 cm−2·s −1) 54Mn depletion should be investigated. 1.4 Detailed procedures describing the use of other fast-neutron detectors are referenced in Practice E261. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E263-18 is classified under the following ICS (International Classification for Standards) categories: 17.240 - Radiation measurements; 27.120.30 - Fissile materials and nuclear fuel technology. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E263-18 has the following relationships with other standards: It is inter standard links to ASTM E263-13, ASTM E1018-20e1, ASTM E1018-20, ASTM E844-18, ASTM E170-17, ASTM E170-16a, ASTM E170-16, ASTM E170-15a, ASTM E1005-15, ASTM E261-15, ASTM E170-15, ASTM E170-14a, ASTM E170-14, ASTM E844-09(2014)e2, ASTM E844-09(2014)e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E263-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E263 − 18
Standard Test Method for
Measuring Fast-Neutron Reaction Rates by Radioactivation
of Iron
This standard is issued under the fixed designation E263; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope 2. Referenced Documents
1.1 This test method describes procedures for measuring 2.1 ASTM Standards:
54 54
reaction rates by the activation reaction Fe(n,p) Mn. D1193Specification for Reagent Water
E170Terminology Relating to Radiation Measurements and
1.2 Thisactivationreactionisusefulformeasuringneutrons
Dosimetry
withenergiesaboveapproximately2.2MeVandforirradiation
E181Test Methods for Detector Calibration andAnalysis of
times up to about three years, provided that the analysis
Radionuclides
methodsdescribedinPracticeE261arefollowed.Ifdosimeters
E261Practice for Determining Neutron Fluence, Fluence
are analyzed after irradiation periods longer than three years,
Rate, and Spectra by Radioactivation Techniques
the information inferred about the fluence during irradiation
E844Guide for Sensor Set Design and Irradiation for
periods more than three years before the end of the irradiation
Reactor Surveillance
should not be relied upon without supporting data from
E944Guide for Application of Neutron Spectrum Adjust-
dosimeters withdrawn earlier.
ment Methods in Reactor Surveillance
1.3 With suitable techniques, fission-neutron fluence rates
E1005Test Method for Application and Analysis of Radio-
8 −2 −1
above 10 cm ·s can be determined. However, in the pres-
metric Monitors for Reactor Vessel Surveillance
ence of a high thermal-neutron fluence rate (for example, >2 ×
E1018Guide for Application of ASTM Evaluated Cross
14 −2 −1 54
10 cm ·s ) Mn depletion should be investigated.
Section Data File
1.4 Detailed procedures describing the use of other fast-
3. Terminology
neutron detectors are referenced in Practice E261.
3.1 Definitions:
1.5 The values stated in SI units are to be regarded as
3.1.1 Refer to Terminology E170 for definitions of terms
standard. No other units of measurement are included in this
relating to radiation measurements and neutron dosimetry.
standard.
1.6 This standard does not purport to address all of the
4. Summary of Test Method
safety concerns, if any, associated with its use. It is the
4.1 High-purity iron is irradiated in a neutron field, thereby
responsibility of the user of this standard to establish appro-
54 54 54
producing radioactive Mn from the Fe(n,p) Mn activation
priate safety, health, and environmental practices and deter-
reaction.
mine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accor-
4.2 The gamma rays emitted by the radioactive decay of
dance with internationally recognized principles on standard-
Mn are counted in accordance with Test Methods E181. The
ization established in the Decision on Principles for the
reaction rate, as defined by Practice E261, is calculated from
Development of International Standards, Guides and Recom-
the decay rate and irradiation conditions.
mendations issued by the World Trade Organization Technical
4.3 Radioassay of the Mn activity may be accomplished
Barriers to Trade (TBT) Committee.
by directly counting the irradiated iron dosimeter, or by first
chemically separating the Mn activity prior to counting.
ThistestmethodisunderthejurisdictionofASTMCommitteeE10onNuclear
Technology and Applications and is the direct responsibility of Subcommittee
E10.05 on Nuclear Radiation Metrology. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2018. Published January 2019. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1965 as E263–65T. Last previous edition approved in 2013 as Standards volume information, refer to the standard’s Document Summary page on
E263–13. DOI: 10.1520/E0263-18. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E263 − 18
59 60
4.4 The neutron fluence rate above about 2.2 MeVcan then is the most effective method for eliminating Fe and Co,
be calculated from the spectral-weighted neutron activation direct counting of iron for Mn is possible using high-
cross section as defined by Practice E261. resolution detector systems or unfolding or stripping
techniques, especially if the dosimeter was covered with
5. Significance and Use
cadmium or boron during irradiation. Altering the isotopic
5.1 Refer to Guide E844 for guidance on the selection, composition of the iron dosimeter is another useful technique
for eliminating interference from extraneous activities when
irradiation, and quality control of neutron dosimeters.
direct sample counting is to be employed.
5.2 Refer to Practice E261 for a general discussion of the
determination of fast-neutron fluence rate with threshold de-
5.7 Thevaporpressuresofmanganeseandironaresuchthat
tectors.
manganesediffusionlossesfromironcanbecomesignificantat
temperatures above about 700°C. Therefore, precautions must
5.3 Pure iron in the form of foil or wire is readily available
be taken to avoid the diffusion loss of Mn from iron
and easily handled.
dosimeters at high temperature. Encapsulating the iron dosim-
5.4 Fig. 1 shows a plot of cross section as a function of
eter in quartz or vanadium will contain the manganese at
54 54
neutron energy for the fast-neutron reaction Fe(n,p) Mn
temperatures up to about 900°C.
(1). This figure is for illustrative purposes only to indicate the
54 54
5.8 Sections 6, 7 and 8 that follow were specifically written
range of response of the Fe(n,p) Mn reaction. Refer to
to describe the method of chemical separation and subsequent
Guide E1018 for recommended tabulated dosimetry cross
counting of the Mn activity. When one elects to count the
sections.
iron dosimeters directly, those portions of Sections 6, 7 and 8
54 4
5.5 Mn has a half-life of 312.19 (3) days (2) and emits a
that pertain to radiochemical separation should be disregarded.
gamma ray with an energy of 834.855 (3) keV (2).
NOTE1—Thefollowingportionsofthistestmethodapplyalsotodirect
5.6 Interfering activities generated by neutron activation
sample-counting methods: 6.1 – 6.3, 7.4, 7.9, 7.10, 8.1 – 8.5, 8.18, 8.19,
arising from thermal or fast neutron interactions are 2.57878
and9–12.
56 59 60
(46)-h Mn, 44.494 (12) days Fe, and 5.2711 (8) years Co
(2,3). (Consult the latest version of Ref (2) for more precise 6. Apparatus (Note 1)
values currently accepted for the half-lives.) Interference from
6.1 High–Resolution Gamma-Ray Spectrometer, because of
Mn can be eliminated by waiting 48 h before counting.
its high resolution, the germanium detector is useful when
Although chemical separation of Mn from the irradiated iron
contaminant activities are present. SeeTest Methods E181 and
E1005.
Theboldfacenumbersinparenthesesrefertothelistofreferenceslocatedatthe
6.2 Precision Balance, able to achieve the required accu-
end of this test method.
racy.
The un-bolded number in parenthesis after the unit indicates the uncertainty in
theleastsignificantdigits.Forexample,1.89(2)keVwouldindicateavalueof1.89
6.3 Digital Computer, useful for data analysis (optional).
keV 6 0.02 keV.
54 54
FIG. 1 Fe(n,p) Mn Cross Section
E263 − 18
6.4 Chemical Separation Cylinder, borosilicate glass, about 7.5 Hydrochloric Acid (sp gr 1.19, 1190 kg/m )—
25-mL capacity, equipped with stopcock and funnel. This Concentrated hydrochloric acid (HCl).
apparatus is illustrated in Fig. 2.
7.6 Hydrochloric Acid (1 + 3)—Mix 1 volume of concen-
6.5 Beakers,borosilicateglass,50mL; volumetric flasks,25
trated HCl (sp gr 1.19) with 3 volumes of water.
and 50 mL, and volumetric pipets, 1 mL.
7.7 Manganese Carrier Solution (10 mg MnCl /cm ).
7. Reagents and Materials (Note 1)
7.8 Nitric Acid (sp gr 1.42, 1420 kg/m )—Concentrated
7.1 Purity of Reagents—Reagent-grade chemicals shall be
nitric acid (HNO ).
used in all tests. Unless otherwise indicated, it is intended that
7.9 Encapsulating Materials—Brass, stainless steel, copper,
all reagents shall conform to the specifications of the Commit-
aluminum, quartz, or vanadium have been used as primary
tee onAnalytical Reagents of theAmerican Chemical Society,
encapsulating materials. The container should be constructed
where such specifications are available. Other grades may be
in such a manner that it will not create a significant flux
used, provided it is first ascertained that the reagent is of
perturbation and that it may be opened easily, especially if the
sufficiently high purity to permit its use without lessening the
capsule is to be opened remotely. (See Guide E844.)
accuracy of the activity determination.
7.2 PurityofWater—Unlessotherwiseindicated,references 7.10 Thepurityoftheironisimportantinthatnoimpurities
to water shall be understood to mean reagent-grade water should be present which produce long-lived radionuclides that
conforming to Specification D1193. interfere with the Mn radioassay. This condition includes
species that will accompany Mn through the separation
7.3 Anion Exchange Resin, strongly basic type, 100 to 200
scheme and that have gamma rays, of energy 0.6 MeV or
mesh size.
higher.Thepresenceofimpuritiesmaybedeterminedeitherby
7.4 Iron Foil or Wire, h
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E263 − 13 E263 − 18
Standard Test Method for
Measuring Fast-Neutron Reaction Rates by Radioactivation
of Iron
This standard is issued under the fixed designation E263; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope
54 54
1.1 This test method describes procedures for measuring reaction rates by the activation reaction Fe(n,p) Mn.
1.2 This activation reaction is useful for measuring neutrons with energies above approximately 2.2 MeV and for irradiation
times up to about 3 years (for longer irradiations, see three years, provided that the analysis methods described in Practice E261).
are followed. If dosimeters are analyzed after irradiation periods longer than three years, the information inferred about the fluence
during irradiation periods more than three years before the end of the irradiation should not be relied upon without supporting data
from dosimeters withdrawn earlier.
8 −2 −1
1.3 With suitable techniques, fission-neutron fluence rates above 10 cm ·s can be determined. However, in the presence of
14 −2 −1 54
a high thermal-neutron fluence rate (for example, >2 × 10 cm ·s ) Mn depletion should be investigated.
1.4 Detailed procedures describing the use of other fast-neutron detectors are referenced in Practice E261.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
E170 Terminology Relating to Radiation Measurements and Dosimetry
E181 Test Methods for Detector Calibration and Analysis of Radionuclides
E261 Practice for Determining Neutron Fluence, Fluence Rate, and Spectra by Radioactivation Techniques
E844 Guide for Sensor Set Design and Irradiation for Reactor Surveillance
E944 Guide for Application of Neutron Spectrum Adjustment Methods in Reactor Surveillance
E1005 Test Method for Application and Analysis of Radiometric Monitors for Reactor Vessel Surveillance
E1018 Guide for Application of ASTM Evaluated Cross Section Data File
3. Terminology
3.1 Definitions:
3.1.1 Refer to Terminology E170 for definitions of terms relating to radiation measurements and neutron dosimetry.
This test method is under the jurisdiction of ASTM Committee E10 on Nuclear Technology and Applications and is the direct responsibility of Subcommittee E10.05
on Nuclear Radiation Metrology.
Current edition approved June 1, 2013Dec. 1, 2018. Published July 2013January 2019. Originally approved in 1965 as E263 – 65 T. Last previous edition approved in
20092013 as E263 – 09.E263 – 13. DOI: 10.1520/E0263-13.10.1520/E0263-18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E263 − 18
4. Summary of Test Method
54 54 54
4.1 High-purity iron is irradiated in a neutron field, thereby producing radioactive Mn from the Fe(n,p) Mn activation
reaction.
4.2 The gamma rays emitted by the radioactive decay of
Mn are counted in accordance with Test Methods E181. The reaction rate, as defined by Practice E261, is calculated from the
decay rate and irradiation conditions.
4.3 Radioassay of the Mn activity may be accomplished by directly counting the irradiated iron dosimeter, or by first
chemically separating the Mn activity prior to counting.
4.4 The neutron fluence rate above about 2.2 MeV can then be calculated from the spectral-weighted neutron activation cross
section as defined by Practice E261.
5. Significance and Use
5.1 Refer to Guide E844 for guidance on the selection, irradiation, and quality control of neutron dosimeters.
5.2 Refer to Practice E261 for a general discussion of the determination of fast-neutron fluence rate with threshold detectors.
5.3 Pure iron in the form of foil or wire is readily available and easily handled.
54 54 3
5.4 Fig. 1 shows a plot of cross section as a function of neutron energy for the fast-neutron reaction Fe(n,p) Mn (1). This
54 54
figure is for illustrative purposes only to indicate the range of response of the Fe(n,p) Mn reaction. Refer to Guide E1018 for
descriptions of recommended tabulated dosimetry cross sections.
54 4
5.5 Mn has a half-life of 312.13312.19 (3) days (2) and emits a gamma ray with an energy of 834.838 (5)834.855 (3) keV
(2).
5.6 Interfering activities generated by neutron activation arising from thermal or fast neutron interactions are 2.5789
56 59 60
(1)-h2.57878 (46)-h Mn, 44.495 (9) day44.494 (12) days Fe, and 1925.28 (1) day5.2711 (8) years Co (2,3). (Consult the latest
version of Ref (2) for more precise values currently accepted for the half-lives.) Interference from Mn can be eliminated by
waiting 48 h before counting. Although chemical separation of Mn from the irradiated iron is the most effective method for
59 60 54
eliminating Fe and Co, direct counting of iron for Mn is possible using high-resolution detector systems or unfolding or
54 54
FIG. 1 Fe(n,p) Mn Cross Section
The boldface numbers in parentheses refer to the list of references located at the end of this test method.
The un-bolded number in parenthesis after the unit indicates the uncertainty in the least significant digits. For example, 1.89 (2) keV would indicate a value of 1.89 keV
6 0.02 keV.
E263 − 18
stripping techniques, especially if the dosimeter was covered with cadmium or boron during irradiation. Altering the isotopic
composition of the iron dosimeter is another useful technique for eliminating interference from extraneous activities when direct
sample counting is to be employed.
5.7 The vapor pressures of manganese and iron are such that manganese diffusion losses from iron can become significant at
temperatures above about 700°C. Therefore, precautions must be taken to avoid the diffusion loss of Mn from iron dosimeters
at high temperature. Encapsulating the iron dosimeter in quartz or vanadium will contain the manganese at temperatures up to
about 900°C.
5.8 Sections 6, 7 and 8 that follow were specifically written to describe the method of chemical separation and subsequent
counting of the Mn activity. When one elects to count the iron dosimeters directly, those portions of Sections 6, 7 and 8 that
pertain to radiochemical separation should be disregarded.
NOTE 1—The following portions of this test method apply also to direct sample-counting methods: 6.1 – 6.3, 7.4, 7.9, 7.10, 8.1 – 8.5, 8.18, 8.19, and
9 – 12.
6. Apparatus (Note 1)
6.1 High–Resolution Gamma-Ray Spectrometer, because of its high resolution, the germanium detector is useful when
contaminant activities are present. See Test Methods E181 and E1005.
6.2 Precision Balance, able to achieve the required accuracy.
6.3 Digital Computer, useful for data analysis (optional).
6.4 Chemical Separation Cylinder, borosilicate glass, about 25-mL capacity, equipped with stopcock and funnel. This apparatus
is illustrated in Fig. 2.
6.5 Beakers, borosilicate glass, 50 mL; volumetric flasks, 25 and 50 mL, and volumetric pipets, 1 mL.
7. Reagents and Materials (Note 1)
7.1 Purity of Reagents—Reagent-grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
FIG. 2 Ion-Exchange Separation Apparatus
E263 − 18
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the activity determination.
7.2 Purity of Water— Unless otherwise indicated, references to water shall be understood to mean reagent-grade water
conforming to Specification D1193.
7.3 Anion Exchange Resin, strongly basic type, 100 to 200 mesh size.
7.4 Iron Foil or Wire, high purity.
7.5 Hydrochloric Acid (sp gr 1.19, 1190 kg/m )—Concentrated hydrochloric acid (HCl).
7.6 Hydrochloric Acid (1 + 3)—Mix 1 volume of concentrated HCl (sp gr 1.19) with 3 volumes of water.
7.7 Manganese Carrier Solution (10 mg MnCl /cm ).
7.8 Nitric Acid (sp gr 1.42, 1420 kg/m )—Concentrated nitric acid (HNO ).
7.9 Encapsulating Materials—Brass, stainless steel, copper, aluminum, quartz, or vanadium have been used as primary
encapsulating materials. The container should be constructed in such a manner that it will not create a significant flux perturbation
and that it may be opened easily, especially if the capsule is to be opened remotely. (See Guide E844.)
7.10 The purity of the iron is important in that no impurities should be present which produce long-lived radionuclides that
54 54
interfere with the Mn radioassay. This condition includes species that will accompany Mn through the separation scheme and
that have gamma rays, of energy 0.6 MeV or higher. The presence of impurities may be determined either by emission spect
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...