ASTM D4547-20
(Guide)Standard Guide for Sampling Waste and Soils for Volatile Organic Compounds
Standard Guide for Sampling Waste and Soils for Volatile Organic Compounds
SIGNIFICANCE AND USE
5.1 This guide describes sample collection and handling procedures designed to minimize losses of VOCs. The principal mechanisms for the loss of VOCs from materials during collection, handling, and storage are volatilization and biodegradation. Susceptibility of various VOCs to these two loss mechanisms is both compound and matrix specific. In general, compounds with higher vapor pressures are more susceptible to volatilization than compounds with lower vapor pressures. Also, aerobically degradable compounds are generally more susceptible to biodegradation than anaerobically degradable compounds. In some cases, the formation of other compounds not originally present in the material can occur. Loss or gain of VOCs leads to analytical results that are unrepresentative of field conditions.
5.2 Ancillary information concerning sample collection, handling, and storage for VOC analysis is provided in Appendix X1 – Appendix X3. These appendixes and cited references are recommended reading for those unfamiliar with the many challenges presented during the collection, handling, and storage of samples for VOC analysis.
SCOPE
1.1 This guide describes recommended procedures for the collection, handling, and preparation of solid waste, soil, and sediment samples for subsequent determination of volatile organic compounds (VOCs). This class of compounds includes low molecular weight aromatics, hydrocarbons, halogenated hydrocarbons, ketones, acetates, nitriles, acrylates, ethers, and sulfides with boiling points below 200° Celsius (C) that are insoluble or slightly soluble in water.
1.2 Methods of sample collection, handling, storage, and preparation for analysis are described.
1.3 This guide does not cover the details of sampling design, laboratory preparation of containers, and the analysis of the samples.
1.4 It is recommended that this guide be used in conjunction with Guide D4687.
1.5 This guide offers an organized collection of information or a series of options and does not recommend a specific course of action. This document cannot replace education or experience and should be used in conjunction with professional judgment. Not all aspects of this guide may be applicable in all circumstances. This ASTM standard is not intended to represent or replace the standard of care by which the adequacy of a given professional service must be judged, nor should this document be applied without consideration of a project’s many unique aspects. The word “standard” in the title of this document means only that the document has been approved through the ASTM consensus process.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. Reporting of test results in units other than SI shall not be regarded as nonconformance with this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2020
- Technical Committee
- D34 - Waste Management
- Drafting Committee
- D34.01.02 - Sampling Techniques
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Feb-2020
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Sep-2017
- Effective Date
- 01-Apr-2017
- Effective Date
- 01-Nov-2016
- Effective Date
- 01-Feb-2016
- Effective Date
- 01-Sep-2015
- Effective Date
- 01-Feb-2015
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Jan-2012
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-May-2010
- Effective Date
- 01-Jul-2009
Overview
ASTM D4547-20 is an international standard guide developed by ASTM International for the sampling of waste, soils, and sediments to determine the presence and concentration of volatile organic compounds (VOCs). The guide is designed to promote best practices for the collection, handling, storage, and preparation of samples to ensure the integrity of VOC analysis. This standard is critical for environmental investigations and waste management, where accurate VOC measurement is essential for risk assessment, regulatory compliance, and remediation planning.
Key Topics
- VOC Sample Integrity: The guide emphasizes minimizing VOC loss during sample collection, handling, and storage. The primary mechanisms for VOC loss are volatilization and biodegradation, which can be influenced by the type of compound and sample matrix.
- Sample Collection Procedures: It recommends using nonreactive tools and airtight containers, such as glass or stainless steel, to avoid contamination and prevent VOC evaporation or adsorption.
- Sample Preparation: Two main methods are outlined for preparing samples for VOC analysis:
- Methanol Extraction: Offers efficient extraction and inhibits biodegradation but may require special handling due to methanol’s flammability.
- Vapor Partitioning: Includes techniques such as purge-and-trap and headspace analysis, which typically provide lower detection limits but may have limitations on sample size and efficiency for certain materials.
- Preservation and Storage: Guidelines for physical (low temperature) and chemical (methanol or acidification) preservation are provided to extend storage life and maintain VOC stability for up to 14 days, provided specific conditions and QA objectives are met.
- Practical Considerations: The guide highlights the importance of professional judgment and context-specific adaptation, recognizing that not all procedures may be applicable in every scenario.
Applications
- Soil and Solid Waste Management: The standard is widely used in environmental site assessments, landfill monitoring, and contaminated land studies to assess the presence of hazardous VOCs.
- Hazardous Waste Site Investigations: Essential for Superfund and similar remediation projects where regulatory compliance and risk assessments depend on reliable VOC data.
- Sample Custody & Laboratory Analysis: Laboratories and field teams rely on D4547-20 for consistent, accurate sample handling procedures, ensuring data quality objectives are met.
- Consulting and Regulatory Reporting: Environmental professionals use the guide to document procedures in compliance with local, national, and international regulations regarding VOC monitoring in waste and soils.
Related Standards
- ASTM D4687: Guide for General Planning of Waste Sampling, recommended for use alongside D4547-20 for comprehensive waste sampling strategies.
- ASTM D6282: Guide for Direct Push Soil Sampling for Environmental Site Characterizations, relevant for subsurface sample collection.
- ASTM D4700: Guide for Soil Sampling from the Vadose Zone.
- ASTM D6418 & D8170: Practices and guides for using disposable samplers for VOC testing in soils.
- ASTM D5792: Practice for Generation of Environmental Data Related to Waste Management Activities, provides guidance on establishing data quality objectives.
- US DOT 49 CFR Part 172: Transportation regulations for hazardous materials, including methanol-preserved samples.
Practical Value
ASTM D4547-20 ensures that environmental assessments and waste monitoring practices yield reliable and representative results for volatile organic compound analysis. By following this guide, professionals can effectively minimize VOC losses, improve analytical accuracy, and support informed decision-making in environmental and waste management sectors. The guide is also instrumental in maintaining regulatory and industry standards in environmental sampling.
Keywords: ASTM D4547-20, VOC sampling, waste sampling, soil sampling, volatile organic compounds, sample integrity, methanol extraction, vapor partitioning, environmental site assessment, waste management standards.
Buy Documents
ASTM D4547-20 - Standard Guide for Sampling Waste and Soils for Volatile Organic Compounds
REDLINE ASTM D4547-20 - Standard Guide for Sampling Waste and Soils for Volatile Organic Compounds
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM D4547-20 is a guide published by ASTM International. Its full title is "Standard Guide for Sampling Waste and Soils for Volatile Organic Compounds". This standard covers: SIGNIFICANCE AND USE 5.1 This guide describes sample collection and handling procedures designed to minimize losses of VOCs. The principal mechanisms for the loss of VOCs from materials during collection, handling, and storage are volatilization and biodegradation. Susceptibility of various VOCs to these two loss mechanisms is both compound and matrix specific. In general, compounds with higher vapor pressures are more susceptible to volatilization than compounds with lower vapor pressures. Also, aerobically degradable compounds are generally more susceptible to biodegradation than anaerobically degradable compounds. In some cases, the formation of other compounds not originally present in the material can occur. Loss or gain of VOCs leads to analytical results that are unrepresentative of field conditions. 5.2 Ancillary information concerning sample collection, handling, and storage for VOC analysis is provided in Appendix X1 – Appendix X3. These appendixes and cited references are recommended reading for those unfamiliar with the many challenges presented during the collection, handling, and storage of samples for VOC analysis. SCOPE 1.1 This guide describes recommended procedures for the collection, handling, and preparation of solid waste, soil, and sediment samples for subsequent determination of volatile organic compounds (VOCs). This class of compounds includes low molecular weight aromatics, hydrocarbons, halogenated hydrocarbons, ketones, acetates, nitriles, acrylates, ethers, and sulfides with boiling points below 200° Celsius (C) that are insoluble or slightly soluble in water. 1.2 Methods of sample collection, handling, storage, and preparation for analysis are described. 1.3 This guide does not cover the details of sampling design, laboratory preparation of containers, and the analysis of the samples. 1.4 It is recommended that this guide be used in conjunction with Guide D4687. 1.5 This guide offers an organized collection of information or a series of options and does not recommend a specific course of action. This document cannot replace education or experience and should be used in conjunction with professional judgment. Not all aspects of this guide may be applicable in all circumstances. This ASTM standard is not intended to represent or replace the standard of care by which the adequacy of a given professional service must be judged, nor should this document be applied without consideration of a project’s many unique aspects. The word “standard” in the title of this document means only that the document has been approved through the ASTM consensus process. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. Reporting of test results in units other than SI shall not be regarded as nonconformance with this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This guide describes sample collection and handling procedures designed to minimize losses of VOCs. The principal mechanisms for the loss of VOCs from materials during collection, handling, and storage are volatilization and biodegradation. Susceptibility of various VOCs to these two loss mechanisms is both compound and matrix specific. In general, compounds with higher vapor pressures are more susceptible to volatilization than compounds with lower vapor pressures. Also, aerobically degradable compounds are generally more susceptible to biodegradation than anaerobically degradable compounds. In some cases, the formation of other compounds not originally present in the material can occur. Loss or gain of VOCs leads to analytical results that are unrepresentative of field conditions. 5.2 Ancillary information concerning sample collection, handling, and storage for VOC analysis is provided in Appendix X1 – Appendix X3. These appendixes and cited references are recommended reading for those unfamiliar with the many challenges presented during the collection, handling, and storage of samples for VOC analysis. SCOPE 1.1 This guide describes recommended procedures for the collection, handling, and preparation of solid waste, soil, and sediment samples for subsequent determination of volatile organic compounds (VOCs). This class of compounds includes low molecular weight aromatics, hydrocarbons, halogenated hydrocarbons, ketones, acetates, nitriles, acrylates, ethers, and sulfides with boiling points below 200° Celsius (C) that are insoluble or slightly soluble in water. 1.2 Methods of sample collection, handling, storage, and preparation for analysis are described. 1.3 This guide does not cover the details of sampling design, laboratory preparation of containers, and the analysis of the samples. 1.4 It is recommended that this guide be used in conjunction with Guide D4687. 1.5 This guide offers an organized collection of information or a series of options and does not recommend a specific course of action. This document cannot replace education or experience and should be used in conjunction with professional judgment. Not all aspects of this guide may be applicable in all circumstances. This ASTM standard is not intended to represent or replace the standard of care by which the adequacy of a given professional service must be judged, nor should this document be applied without consideration of a project’s many unique aspects. The word “standard” in the title of this document means only that the document has been approved through the ASTM consensus process. 1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. Reporting of test results in units other than SI shall not be regarded as nonconformance with this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4547-20 is classified under the following ICS (International Classification for Standards) categories: 13.080.05 - Examination of soils in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4547-20 has the following relationships with other standards: It is inter standard links to ASTM D5792-10(2023), ASTM D5681-23, ASTM D5058-12(2020), ASTM D5681-18, ASTM D5681-17, ASTM D3550/D3550M-17, ASTM D5681-16a, ASTM D5681-16, ASTM D5792-10(2015), ASTM D4700-15, ASTM D5681-13, ASTM D5058-12, ASTM D5792-10, ASTM D6640-01(2010), ASTM D5681-09. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4547-20 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4547 − 20
Standard Guide for
Sampling Waste and Soils for Volatile Organic Compounds
This standard is issued under the fixed designation D4547; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
1.1 This guide describes recommended procedures for the
1.8 This international standard was developed in accor-
collection, handling, and preparation of solid waste, soil, and
dance with internationally recognized principles on standard-
sediment samples for subsequent determination of volatile
ization established in the Decision on Principles for the
organic compounds (VOCs).This class of compounds includes
Development of International Standards, Guides and Recom-
low molecular weight aromatics, hydrocarbons, halogenated
mendations issued by the World Trade Organization Technical
hydrocarbons, ketones, acetates, nitriles, acrylates, ethers, and
Barriers to Trade (TBT) Committee.
sulfides with boiling points below 200° Celsius (C) that are
insoluble or slightly soluble in water.
2. Referenced Documents
1.2 Methods of sample collection, handling, storage, and 2
2.1 ASTM Standards:
preparation for analysis are described.
D1193 Specification for Reagent Water
1.3 Thisguidedoesnotcoverthedetailsofsamplingdesign, D1586/D1586M Test Method for Standard Penetration Test
laboratory preparation of containers, and the analysis of the (SPT) and Split-Barrel Sampling of Soils
samples. D3550/D3550M Practice for Thick Wall, Ring-Lined, Split
Barrel, Drive Sampling of Soils
1.4 Itisrecommendedthatthisguidebeusedinconjunction
D4687 Guide for General Planning of Waste Sampling
with Guide D4687.
D4700 Guide for Soil Sampling from the Vadose Zone
1.5 This guide offers an organized collection of information
D5058 Practices for Compatibility of Screening Analysis of
oraseriesofoptionsanddoesnotrecommendaspecificcourse
Waste
of action. This document cannot replace education or experi-
D5681 Terminology for Waste and Waste Management
ence and should be used in conjunction with professional
D5792 Practice for Generation of Environmental Data Re-
judgment. Not all aspects of this guide may be applicable in all
lated to Waste Management Activities: Development of
circumstances. This ASTM standard is not intended to repre-
Data Quality Objectives
sent or replace the standard of care by which the adequacy of
D6051 Guide for Composite Sampling and Field Subsam-
a given professional service must be judged, nor should this
pling for Environmental Waste Management Activities
document be applied without consideration of a project’s many
D6232 Guide for Selection of Sampling Equipment for
unique aspects. The word “standard” in the title of this
WasteandContaminatedMediaDataCollectionActivities
document means only that the document has been approved
D6282/D6282M Guide for Direct Push Soil Sampling for
through the ASTM consensus process.
Environmental Site Characterizations
1.6 The values stated in SI units are to be regarded as D6418 Practice for Using the Disposable En Core Sampler
for Sampling and Storing Soil for Volatile OrganicAnaly-
standard. No other units of measurement are included in this
standard. Reporting of test results in units other than SI shall sis (Withdrawn 2018)
D6640 Practice for Collection and Handling of Soils Ob-
not be regarded as nonconformance with this standard.
tained in Core Barrel Samplers for Environmental Inves-
1.7 This standard does not purport to address all of the
tigations
safety concerns, if any, associated with its use. It is the
D8170 Guide for Using Disposable Handheld Soil Core
responsibility of the user of this standard to establish appro-
Samplers for the Collection and Storage of Soil for
1 2
This guide is under the jurisdiction of ASTM Committee D34 on Waste For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Management and is the direct responsibility of Subcommittee D34.01.02 on contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Sampling Techniques. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved Sept. 1, 2020. Published September 2020. Originally the ASTM website.
approved in 1991. Last previous edition approved in 2015 as D4547 – 15. DOI: The last approved version of this historical standard is referenced on
10.1520/D4547-20. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4547 − 20
Volatile Organic Analysis 6. Selection of Sample Preparation Method for VOC
4 Analysis
2.2 Federal Standard:
6.1 Introduction:
Code of Federal Regulations (CFR), Title 49, Part
6.1.1 Sample collection, handling, and preservation meth-
172 Transportation, List of Hazardous Substances and
ods should be compatible with the method used to prepare the
Reportable Quantities
sample for VOC analysis, and meet the project’s data quality
objectives (see Practice D5792). Preparation of a sample for
3. Terminology
instrumental analysis can be initiated either in the field or
3.1 Definitions:
laboratory. In either case, prior to analysis, the sample should
3.1.1 sample, n—a portion of material taken from a larger
be placed into a tared volatile organic analysis (VOA) vial or
quantity for the purpose of estimating properties or composi-
bottle meeting the specifications given in 7.3. When working
tion of the larger quantity. (D5681)
with an uncharacterized solid waste, it is advisable to perform
3.1.2 subsample, n—a portion of a sample taken for the
compatibility tests (see Practices D5058) between the sample
purpose of estimating properties or composition of the whole
material and the solution (see 6.2 and 6.3) into which it will be
sample. (D6051)
transferred in preparation for analysis. For instance, when
3.1.2.1 Discussion—A subsample, by definition, is also a collecting highly contaminated soils or waste of unknown
sample.
composition, it is strongly recommended that preliminary
testing be performed to adequately characterize the waste
4. Summary of Guide materials so that when the user applies the procedures cited in
this guide, there will be no chemical reaction which may
4.1 This guide addresses the use of tools for sample collec-
jeopardize the user’s safety.
tion and transfer, conditions for sample storage, sample
6.1.2 Figs. 1 and 2 are flow diagrams showing some
preservation, and two common means of sample preparation
different options for combining sample collection, handling,
foranalysis.Specialattentionisgiventoeachstepfromsample
and preparation methods for instrumental analysis.
collectiontoanalysistolimitthelossofVOCsbyvolatilization
6.2 Methanol Extraction:
and biodegradation. The sample collected and analyzed should
6.2.1 This method involves the extraction of VOCs from a
be representative of the matrix material sampled. The two
samplewithmethanolandthesubsequenttransferofanaliquot
methods cited for the preparation of samples for VOC analysis
of the extract to water for either purge-and-trap, headspace
are methanol extraction and vapor partitioning (that is, purge-
analysis, or vacuum distillation.
and-trap, headspace, and vacuum distillation). The method of
6.2.2 Advantages of methanol extraction are: (1) large
sample preparation forVOC analysis should be consistent with
the data quality objectives (see Practice D5792). samples or composite samples, or both, can be collected to
enhancerepresentatives(seeGuideD6051),(2)biodegradation
isinhibited,(3)anefficientextractionofVOCsfromthematrix
5. Significance and Use
materials can be achieved with methanol due to its strong
5.1 This guide describes sample collection and handling
affinity for these compounds and favorable wetting properties,
procedures designed to minimize losses of VOCs. The princi-
(4) a subsample can be analyzed several times, and (5) sample
pal mechanisms for the loss of VOCs from materials during
extracts can be archived, if verified that VOC losses have not
collection, handling, and storage are volatilization and biodeg-
occurred (see 10.1.1).
radation. Susceptibility of various VOCs to these two loss
6.2.3 Theprimarydisadvantagesofmethanolextractionare:
mechanisms is both compound and matrix specific. In general,
(1) samples may have to be shipped as a flammable liquid
compoundswithhighervaporpressuresaremoresusceptibleto
depending on the amount of methanol present (for example,
volatilization than compounds with lower vapor pressures.
U.S. DOT Reg. 49 CFR§172.101), (2) hazards to personnel
Also, aerobically degradable compounds are generally more
due to methanol’s toxicity and flammability, (3) detection
susceptible to biodegradation than anaerobically degradable
limits are elevated due to analyte dilution, (4) possible inter-
compounds. In some cases, the formation of other compounds
ference of the methanol peak with VOCs of interest, (5)
not originally present in the material can occur. Loss or gain of
potential adverse impact of methanol on the performance of
VOCs leads to analytical results that are unrepresentative of
certain gas chromatograph/detector systems, and (6) samples
field conditions.
extracted with methanol must be disposed of as a regulated
5.2 Ancillary information concerning sample collection, waste.
handling, and storage for VOC analysis is provided in Appen-
6.2.4 Logistical challenges of performing these tasks in the
dix X1 – Appendix X3. These appendixes and cited references field can be overcome by extracting samples with methanol
are recommended reading for those unfamiliar with the many
once they have been received in a laboratory, provided that the
challenges presented during the collection, handling, and samples are transported in an airtight container (see 7.3.2, 9.1,
storage of samples for VOC analysis. and 9.2). Furthermore, if VOC levels are unknown, a replicate
sample can be obtained and screened to determine if methanol
extraction is appropriate for the expected contaminant concen-
trations.
Available from Superintendent of Documents, U.S. Government Printing
Office, Washington, DC 20402.
D4547 − 20
FIG. 1 Sample Handling Options for Cohesive Materials
D4547 − 20
FIG. 2 Sample Handling Options for Noncohesive and Cementitious Materials
D4547 − 20
6.3 Vapor Partitioning: 7.2 Tools—There are often several steps to sampling, par-
ticularly if it involves obtaining bulk material from the
6.3.1 Vapor partitioning involves the direct analysis of a
subsurface.Mostoftheequipmentusedtoobtainsamplesfrom
sample by either purge-and-trap, headspace, or vacuum distil-
the subsurface was originally developed for the geotechnical
lation. In each case, the sample is placed into a tared volatile
industry; however, several devices have been developed spe-
analysis (VOA) vial or flask (for vacuum distillation) contain-
ing water or a preservative solution (for example, acidified cifically for environmental sampling by direct push methods
water) from which the vapor is removed for analysis without (Guide D6282/D6282M). The subsurface bulk sampling sys-
the container being opened. tems are designed to obtain intact cylindrical cores of material,
ranginganywherefrom2.5to10.2cmindiameterand30.5cm
6.3.2 The principal advantages of this method are: (1)itcan
or more in length. Two geotechnical tools that have been used
offer lower detection limits than methanol extraction because
for subsurface sample collection are the split-spoon sampler
no dilution is involved, (2) there are no organic solvent
(Test Method D1586/D1586M), which opens to expose the
interferences, and (3) there is no use of regulated organic
solvents, which may require special shipment, disposal, and entirelengthofthematerialobtainedforsubsampling,andcore
field handling practices. barrel liners (ring-lined barrel sampling, see Practice D3550/
D3550M) that are typically subsampled through open ends.
6.3.3 The disadvantages associated with vapor partitioning
Core barrel liners should not be used for storage of samples
are: (1) the VOA vial (VOA vials are different sizes for
intended for VOC analysis because they do not have airtight
automated purge-and-trap and headspace instrumentation) or
seals; see Appendix X1. Single-tube and dual-tube sampling
adapter used in conjunction with aVOAvial, or both, often are
devices have been developed for environmental applications
instrument specific, (2) sample size is limited (<10 g) by
automated systems, (3) a matrix-appropriate method of pres- (Guide D6282/D6282M) and are usually operated by direct
ervation may be necessary (see Appendix X2), (4) vapor pushmethods.Samplelinersusedinthebulksamplingsystems
partitioning is less efficient at recovering VOCs from some
come in a variety of lengths and materials (stainless steel,
materials than methanol extraction, and (5) when using purge-
brass, PTFE, rigid plastics, etc.).Additional information on the
and-trap or vacuum distillation, only a single analysis of the
design and application of different types of subsurface collec-
same sample can be made; similarly only a single analysis may
tion systems that are available can be found in Guides D4700,
be possible with headspace analysis unless concentrations
D6232, and D6282/D6282M. Subsurface materials retrieved
allow for the use of a small injection volume.
for VOC characterization should be obtained (sampling tubes
6.3.4 Limitations imposed by vapor phase partitioning filled and brought to the surface) as quickly as possible and
methods with regard to number of analyses that can be remain intact and undisturbed until they are subsampled (see
performed on a single sample can be addressed by taking Practice D6640). Subsampling a bulk sample should occur as
replicate samples.
quickly as possible after it is brought to the surface. It is
important for subsampling to occur quickly because for bulk
6.3.5 When employing vapor phase partitioning methods,
sampleretrievalsystemswherethematerialisexposeddirectly
the logistical challenges of performing sample preparation in
to the atmosphere for a period of time during subsampling,
the field (see 7.3.3) can be avoided by performing the prepa-
there is an opportunity for VOC loss to occur. Suggested liner
ration step in the laboratory, so long as the sample is trans-
ported to the laboratory in an airtight container (see 7.2.1). If configurations and examples of sampling techniques for bulk
VOC levels are unknown, a replicate sample can be obtained soil samples collected by single-tube direct push methods that
and screened to determine if it is appropriate to use a vapor provide limited exposure of the soil being sampled to the
partitioning method of sample preparation. atmosphere are described in Appendix X3 (see 8.1 – 8.3 for
additional sampling guidance).
7. Sampling Tools and Containers
7.2.1 In addition to the coring devices which retrieve bulk
quantities of material, there are smaller hand-operated coring
7.1 All sample handling devices and vessels used to collect
tools for obtaining samples of the appropriate size (for
and store samples for analysis should be constructed of
example, coring devices capable of collecting 3- and 15-cm
nonreactive materials that will not sorb, leach, or diffuse
volumes, for collection of approximately 5- and 25-g samples,
constituents of interest. Examples of materials that meet these
respectively) for analysis (see Fig. 3 and Guide D8170). If one
criteriaareglass,stainlesssteel,steel,andbrass.Materialssuch
of these smaller coring devices is used to store the sample (see
as polytetrafluoroethylene (PTFE) and many rigid plastics also
9.1.1), the main body shall be constructed of materials that are
can be used; however, it should be recognized that they may
nonreactive and have airtight seals that show limited sorption
havesomelimitedadsorptivepropertiesorallowslowdiffusive
and penetration of VOCs. Hand-operated coring devices that
passage of some VOCs. Materials which show limited reactiv-
are used to store samples shall be evaluated using the proce-
ity can be used when they have a very short period of contact
dure given in Appendix X4 to ensure that the device meets
with the sample or when they are necessary for making airtight
these criteria.Any alternate evaluation technique shall produce
(hermetic) seals. Collection tools and storage containers made
equalorbetterrecoveriesofthecompoundsstatedinAppendix
ofmaterialsotherthanthosecitedinthissectionshouldonlybe
used after they have demonstrated equivalency (see 7.2.1).All X4.When the coring device is only used to rapidly transfer the
sample to a VOA vial, corer material requirements are less
collection tools and storage containers should be cleaned in a
manner consistent with their intended use. stringent.
D4547 − 20
FIG. 3 Generic Disposable Handheld Soil Core Sampler
7.3 Containers—VOAvials and bottles used for storage and sample to the container. A predetermined volume of sample
preparation of samples for analysis should be made of glass that corresponds to a weight in grams (g) that is equivalent to
and have airtight seals. To achieve an airtight seal, these or less than the volume (mL) of methanol can then be
containers should have a thick septum cushion between the introduced. The ratio (typically 1:1 to 10:1, methanol to
sealing material (PTFE) and cap (rigid plastic screw cap or material) between the two constituents should allow for for-
aluminum crimp top). PTFE-lined caps that do not have mation of a clear layer of methanol over the sample after
flexible septum backing often fail to achieve a liquid or airtight thorough mixing.The difference in weight of the container and
seal. Furthermore, the thickness of the PTFE used for a lined cap, measured before and after the sample is introduced, is
septum should be at least 0.254 mm. usedtoestablishthesample’swetweight.Becauseofthewater
7.3.1 Preparation of VOA Vial or Bottle—Record the tared (moisture) present in most samples, calculation of the recovery
weight of the VOAvial or bottle and cap prior to the sampling ofsampleanalyteconcentrationsshouldaccountforthissource
event. Moreover, depending on the method of sample prepa- of dilution.
ration and analysis, the vessel may also contain a solvent or 7.3.3 Preparation of Containers for Vapor Partitioning—
VOC-free water and, if required, a PTFE or glass-coated stir Container preparation applies to both field and laboratory
bar at the time of sample collection (see Section 9). sample preparation. This method allows for the direct analysis
7.3.2 Preparation of Containers for Methanol Extraction— of a sample by either purge-and-trap or headspace techniques.
Container preparation applies to both field and laboratory In both cases, the sample is placed into aVOAvial from which
sample preparation. The appropriate volume of analytical- the vapor (only a portion for headspace analysis) is removed
grade methanol (high-performance liquid chromatography, for analysis without the container being opened. Moreover,
spectrographic, or purge-and-trap) is added to the organic-free water that contains no detectable levels of VOCs and, if
container by the laboratory that supplies the container, by the required, a PTFE or glass-coated stir bar should be present in
sample collector, or by a third party. The party that adds the the VOA vial prior to introducing the sample. Current auto-
methanol to the container should also be responsible for mated equipment for purge-and-trap and headspace systems
providingtripblanks(seeGuideD4687).Ifmethanolispresent typically use 40 and 22-mL VOA vials, respectively. The
in the sample container in the field, this container should be volume of water used for these two different systems is
opened only to add the sample(s). The tared weight of the typically 10 mL or less. Furthermore, the tared weight of the
container with methanol should be recorded prior to adding the container should be recorded prior to adding the sample. The
D4547 − 20
differenceinweightofthecontainer,measuredbeforeandafter sample masses or composite samples may be preferred as the
the sample is added, is used to determine the sample’s wet heterogeneity of the material increases.
weight.
8.1.1 Sampling of Cohesive But Uncemented Materials
7.3.4 Preparation of Samples Transported and Stored in Using Devices Designed to Obtain a Sample Appropriate for
Analysis—Collect appropriate size sample for analysis using a
Solventless VOA Vials or Bottles—When samples are held in a
metal or rigid plastic coring tool (see Figs. 3 and 4). For
VOAvial or bottle, methanol can be added to the container by
example, coring tools for the purpose of transferring a sample
piercing the septum. In the case of a 5-g sample contained in a
can be made from disposable plastic syringes by cutting off the
40-mL VOA vial, 5.0 mL of methanol can be added without
tapered front end and removing the rubber cap from the
overpressurization. When larger samples and bottles, or both,
plunger. Pre-fabricated plastic syringes/coring tools are avail-
are used, experimental trials should be performed to establish
able commercially. This type of coring device is not appropri-
what volume of solution can be added. If performed manually,
ate for sample storage. These smaller coring devices help
a 0.635 mm nominal outer diameter and 0.318 nominal inner
maintain the sample structure during collection and transfer to
diameter or smaller needle (see Note 1) should be used to add
the VOA vial or a larger bottle. When inserting a clean coring
the methanol.After the methanol is introduced, the soil sample
toolintoafreshsurfaceforsamplecollection,airshouldnotbe
should be dispersed. If the sample is held beyond the
trapped behind the sample. If air is trapped, it could either pass
laboratory-prescribed extraction and analysis period (typically
through the sampled material causingVOCs to be lost or cause
24 h or less), it is recommended that either the pierced septum
the sample to be pushed prematurely from the coring tool. For
be replaced with one that is intact or that an aliquot of the
greater ease in pushing coring tools into the solid matrix, their
methanol be transferred to an appropriate vessel for storage. If
front edge can be sharpened.
the pierced septum is replaced, it should be replaced with one
8.1.2 Obtain an undisturbed sample by pushing the barrel of
thatisintactbyreplacingthecapofthevialorbottlewithacap
the coring tool into a freshly exposed surface and removing the
having an intact septum. When the laboratory-prescribed
corer once filled. Clean the exterior of the barrel by wiping
extraction method uses elevated temperatures, or sonication, or
withacleandisposabletowel.Ifthecoringtoolsamplerisused
both, it is recommended that the pierced septum be replaced
as a storage container, cap the open end after ensuring that the
prior to this treatment. To limit the loss of VOCs when
sealing surfaces are cleaned (see 9.1.1). If the device is solely
replacing the cap or transferring an aliquot of methanol, the
used for collection and not storage, immediately extrude the
container should be cooled to 4 6 2 °C, and these functions
sample into a tared VOA vial or bottle by gently pushing the
should be performed as quickly as possible. Cooling the
plunger. During transfer of the sample into the container, care
samplelowersthevaporpressureoftheanalytesofinterestand
should be taken to prevent the sample from contacting the
reduces the pressure created by adding the methanol. By
sealingsurfaceswhichcancomprisethesesurfacesandprevent
having methanol present in the collection vessel prior to
an airtight seal. The volume of material collected should not
introducing the sample, the additional precautions associated
cause excessive stress on the coring tool during intrusion into
with a pierced septum can be avoided. In addition, having
the material, or be so large that the sample easily falls apart
methanolpresentinthecollectionvesselreducesthepossibility
during extrusion. Obtaining and transferring a sample should
of VOCs being lost during the transfer step (that is, extrusion
be done rapidly (<10 s) to reduce volatilization losses. If the
of a plug of soil from the sampling tool into the empty vessel).
vial or bottle contains methanol or another liquid, it should be
NOTE 1—This is a 23-gage needle.
held at an angle when extruding the sample into the container
to minimize splashing. Just before capping, a visual inspection
8. Sample Collection
of the lip and threads of the sample vessel should be made, and
any foreign debris should be removed with a clean towel,
8.1 General Sampling Guidance—Using an appropriate
allowing an airtight seal to form.
sized coring tool (see below), collect samples from freshly
exposedsurfacesofthesoilorwastewithminimaldisturbance.
8.2 Devices That Can Be Used for Sampling a Cemented
The soil or waste to be sampled may be from an intact pit face
Material—Samples of hard or cementitious material may be
or the relatively undisturbed contents in a split-barrel sampler,
obtained by fragmenting a larger portion of the material using
acorebarrelliner,oralinerfromasingleordouble-tubedirect
a clean chisel to generate aggregate(s) of a size that can be
push sampling system. Before sampling, remove several cen-
placed into a tared VOA vial or bottle. When transferring the
timeters of material to expose a fresh surface. Obtain a sample
aggregate(s), precautions must be taken to prevent compromis-
bypressing(orhammeringinafewcases)theendofthecoring
ing the sealing surfaces and threads of the container. Losses of
tool into the pit face or into the end or side of a larger core
VOCs by using this procedure are dependent on the location of
sample. Remove the coring tool. The optimum diameter of the
the contaminant relative to the surface of the material being
coring tool depends on the following: size of the opening on sampled. Therefore, caution should be taken in the interpreta-
the collection vial or bottle (tool should fit inside mouth),
tionofthedataobtainedfrommaterialsthatfitthisdescription.
particle size of the solid materials (for example, gravel-size As a last resort when this task can not be performed on-site, a
particleswouldrequirelargersamplers),andvolumeofsample
large sample can be collected in a vapor-tight container and
required for analysis. For example when a 5-g sample of soil is transportedtothelaboratoryforfurtherprocessing(see9.1and
specified, only a single 3-cm
volume would need to be 9.2). Collecting, fragmenting, and adding the sample to a
collected (assuming the soil has density of 1.7 g/cm ). Larger container should be accomplished as quickly as possible.
D4547 − 20
FIG. 4 A Coring Tool Made by Cutting the Tip Off a Plastic Syringe
8.3 Devices That Can Be Used for Sampling a Noncohesive from the same stratum and place in a suitable container for the
Material—When sampling gravel or a mixture of gravel and determination of percent moisture content of the waste or soil.
fines that cannot be easily obtained or transferred using coring This sample should be collected in a container suitable for the
tools, as a last resort, a sample can be quickly transferred using retention of soil moisture. The location adjacent to where the
a spatula or scoop (see discussion concerning the use of a sample for VOC analysis was collected should be inspected
spatula or scoop to collect samples for VOC analysis, in visually and its characteristics logged. This adjacent material
Appendix X1). If the tared collection vial or bottle contains can also be retained for determining other relevant properties,
methanoloranaqueoussolution,transferthesampletothevial such as general appearance, color, presence of oils, other
or bottle with minimal splashing and without the spatula or visible signs of contamination, grain-size distribution, organic
scoop contacting the liquid contents. For some solids, a carbon content, etc. Collection of these ancillary samples
wide-bottom funnel or similar channeling device may be should be performed after the collection of samples for VOC
necessary to facilitate transfer to the container and prevent analysis.
compromising of the sealing surfaces of the container. Caution
9. Sample Storage, Transportation, and Preservation
should be taken in the interpretation of the data obtained from
materials that fit this description. Losses of VOCs are likely 9.1 Sample Storage and Transportation:
because of the nature of the sampling method and the nonco-
9.1.1 General Considerations—During an initial 48 h stor-
hesive nature of the material exposes more surface area to the age and transportation period, all samples for VOC analysis
atmosphere than for other types of samples. Another potential
should be held in airtight containers and cooled to at least 4 6
source of error during the sampling process is the separation of 2 °C. For storage periods beyond 48 h, these airtight contain-
coarser materials from fines, which can bias the concentration
ers should contain a chemical preservative or be held under
data if the different particle sizes, which have different surface conditions that ensure retention of the analytes of concern (see
areas, are not properly represented in the sample.
9.2).
8.4 Sampling Location Physical Characteristics—Collect a
separate co-located sample within a couple of centimeters and SW-846, Method 5035 A (July 2002), 3rd Update.
D4547 − 20
9.2 Sample Preservation Beyond 48 Hours: low pH condition (see Appendix X2 for other potential
9.2.1 Currently, it is recommended that samples stored in an problems).Acidified sample storage (total time period starting
airtight container should be held for no longer than 48 h at 4 6 at collection) can be extended for up to 14 days at 4 6 2 °C.
2 °C prior to analysis or preservation. Longer storage times at Longer periods of acidified sample storage can be used
4 62 °CcanbeappliedifitcanbedemonstratedthattheVOC provided it can be demonstrated that the VOC concentrations
concentrations are not affected or that the data generated at the arenotaffectedorthatthedatageneratedatthetimeofanalysis
time of analysis meets the data quality objectives. Extended meets the data quality objectives.
sample storage can be achieved by using either physical or
chemical methods of preservation. Sample preservation can be 10. Sample Preparation
initiated at the time of sample collection or after arrival in a
10.1 Methanol Extraction—Completely disperse samples in
laboratory.
methanol, if possible, by shaking, vortexing, or by sonication.
9.2.2 Physical Preservation—Samples contained in a VOA
Allow suspended particulates to settle so that an aliquot of
vial, a bottle, or coring device are physically preserved by
clear supernatant can be removed for analysis. Some materials
low-temperature storage (for example, preferably in a freezer
will show a slow release of VOCs to methanol; therefore, the
set for –12 6 5 °C, or by using a temporary refrigerant, for
dissolved VOC concentration may increase with increasing
example, a mixture of salt and ice or dry ice; see Appendix
extraction time (see Appendix X1). This increased extraction
X2). Under this condition, sample storage (total time period
of VOCs over time may be particularly pronounced with
starting at collection) can be extended up to 14 days. Longer
cementitious materials or materials that strongly adsorbVOCs.
periods of low-temperature storage can be used provided it can
Transfer of an aliquot for analysis should be performed using
be demonstrated that the VOC concentrations are not affected
a clean, liquid-tight syringe (composed of glass and metal) to
or that the data generated at the time of analysis meets the data
withdraw the aliquot through the septum of the sample
quality objective. When low-temperature storage is used for
container. If necessary, the cap of the sample container can be
samples intended for analysis by vapor partitioning, VOC-free
removed, and an aliquot can be collected with either a clean
water and, if required, a PTFE or glass-coated stir bar should
syringe or pipette. However, the time that the cap is off the
be present prior to adding the sample in the field (see 7.3.3).
sample container should be minimized as much as possible to
Specialprecautionsmaybenecessary,forexample,positioning
limit volatilization losses of solvent andVOCs. For purge-and-
the sample vessel on its side, when using a low-temperature
trapanalysis,methanolaliquotvolumesoflessthan0.2mLare
condition with VOA vials smaller than 40 mL, or when more
typically transferred to a vessel containing 5 mL of organic-
than 5 mL of water is present, because of the potential for
free water. For headspace analysis, methanol aliquot volumes
breakage caused by the formation of ice. When the sample is
as large as 1.0 mL, depending on the detector and analytes of
intended to be prepared by methanol extraction, this solvent
concern, can be transferred to VOAvials containing 10 mL of
can be introduced through the septa of the storage vessel after
organic-free water. When the re-analysis of the sample extract
low-temperature storage (see 7.3.4). In either case, immedi-
is a possibility, an aliquot of the extract should be archived by
ately following low-temperature storage of samples in VOA
transferring it to an airtight vial (for example, 2 mL) with a
vials or bottles, the caps should be checked and tightened, if
PTFE-lined cap.
necessary. For coring tools used as storage containers, the
10.1.1 Periodic weighing of the sample container can be
sample is extruded into a prepared VOA vial after low-
usedtodetermineifahermeticsealisbeingmaintained,thatis,
temperature storage (see Guide D8170).
no weight loss of methanol.
9.2.3 Chemical Preservation—Samples immersed in metha-
10.2 Vapor Partitioning—Completely disperse samples in
nol are chemically preserved. Under this condition, storage
water, if possible, by shaking, vortexing, or by sonication.This
(total time period starting at collection) at 4 6 2 °C can be
mixing of the solid material with the aqueous solution not only
extended for up to 14 days. Longer periods of methanol
helps prevent the plugging of the sparging needle used by
preservation can be used provided if it can be demonstrated
purge-and-trap systems, but also assists in attaining an equi-
that the VOC concentrations are not affected or that the data
librium state by completely exposing the sample to the
generated at the time of analysis meets the data quality
partitioning solution. To enhance partitioning into the vapor
objectives. A chemical preservation method for samples in-
phase, automated purge-and-trap and headspace analysis sys-
tended to be analyzed by vapor partitioning is acidification to
tems warm the sample to a selected temperature (typically
a pH of 2 or less with either sodium bisulfate or hydrochloric
40 °C for purge-and-trap, this temperature or higher for head-
acid.However,VOC-contaminatedsamplescontainingcarbon-
space) prior to removing vapors for analysis.
ates should not be added to an acidic solution because of
effervescence (for guidance on testing for carbonates and the
11. Keywords
use of these acids to preserve samples, see Appendix X5).
Precautions must also be taken when preserving by acidifica- 11.1 field sampling; methanol extraction; sample collection;
tion because certain compounds within the following classes,
sample handling; soil; solid wastes; vapor phase partitioning;
olefins, ketones, esters, ethers, and sulfides, will react under a volatile organic compounds
D4547 − 20
APPENDIXES
(Nonmandatory Information)
X1. ADDITIONAL INFORMATION CONCERNING SAMPLE COLLECTION AND HANDLING FOR VOC ANALYSIS
X1.1 Volatile organic compounds (VOCs) are among the bottle to capacity, the sealing surfaces often become compro-
most frequently identified soil contaminants at Superfund and mised (dirty), preventing a vapor-tight seal during storage. For
other hazardous waste sites. Because some VOCs are poten- these reasons, this procedure has been shown to result in
tially mutagenic, carcinogenic, and teratogenic, they often are concentrations that are less than 10 % of the in-situ contami-
key factors in the risk assessment process at these sites. These nation (6-9).
risk assessments and other remedial decisions depend upon an
X1.6 Another procedure that often performs poorly is the
accurate understanding of the levels and extent of VOC
useofcorebarrellinerscoveredwitheitherPTFEoraluminum
contamination in soils and other materials.
foil for sample transportation and storage, because these
X1.2 In most materials, VOCs coexist as gaseous, liquid,
barriers fail to serve as airtight seals (3, 11). Sheets of PTFE
and solid (sorbed) phases. The VOC equilibrium that exists
that are only 0.05 or 0.08 mm thick are easily penetrated by
among these phases is controlled by physiochemical
VOCvapors.ElasticformulationsofPTFE(forexample,white
properties, material properties, and environmental variables (1,
plumber’s tape) show much greater rates of VOC penetration
2). Unaccountedlossofanalytesfromanyphasemayresultin
thannon-elastic,semi-translucentformulations.Aluminumfoil
rendering the sample unrepresentative of the material from
fails because an airtight seal is often not achieved when this
which it was taken. For this reason, sample collection,
material is folded over the end of a core barrel liner and it
handling,andanalysismustbeperformedunderconditionsthat
sometimes corrodes, allowing small holes to form. The addi-
maintain the accountability of all phases present (3).
tion of plastic caps and sealing tape does not improve the
performance of either PTFE or aluminum foil, since pliable
X1.3 In general, uncontrolled losses of VOCs from materi-
plastics sorb VOCs and sealing tapes often contain VOCs in
als occur through two mechanisms: volatilization and biodeg-
the adhesive, which serves as a potential artifact source.
radation. Volatilization losses occur whenever gaseous
molecules, which have diffusion coefficients up to four orders
X1.7 The procedures discussed in this guide are designed to
of magnitude greater than liquid diffusion coefficients, are
limit VOC losses by volatilization and biodegradation. This is
allowed to move freely. Therefore, whenever a new surface is
accomplished by stressing that (1) samples be collected only
exposed, VOC losses are incurred. The extent to which VOCs
from freshly exposes surfaces, (2) collection and transfer of a
are lost depends on the vapor phase concentration (analyte
sample be performed quickly and with minimal disruption to
vapor pressure), surface area exposed, duration of exposure,
its physical state, (3) samples be held under conditions that
porosity of matrix, and perhaps meteorological conditions (4).
ensure retention of the analytes of interest, and (4) in the case
of samples collected for vapor partitioning methods of analysis
X1.4 Biological degradation of VOCs in samples is usually
(purge-and-trap or headspace), the VOA vial’s airtight seal
dominated by aerobic processes because many conventional
never be broken prior to analysis.
intrusive collection methods expose the sample to the atmo-
sphere. The rate of this biological degradation is dependent on
X1.8 AnadditionalsourceofinaccuracyinVOCanalysesis
several factors, including the indigenous microbiological
their incomplete extraction from the sampled material (12-17).
population, chemical properties of the VOC, and temperature.
This source of error can either be due to differences between
Provided that sufficient quantities of electron acceptors,
sample preparation methods (that is, methanol extraction
nutrients, and moisture are present, indigenous microbes con-
versus vapor partitioning) or the kinetics of the desorption
tinue to aerobically degrade compounds even when stored at
processes. In general, as the indigenous total organic carbon
4°C (5). Non-halogenated aromatic compounds are quite
content increases in a matrix, recoveries using vapor partition-
susceptible to this loss mechanism. To inhibit biodegradation
ing methods would be expected to decrease compared to
of these compounds, the sample can be immersed in methanol,
methanol extraction, particularly for the more hydrophobic
frozen (see Appendix X2), or acidified to a pH of 2 or less.
analytes (17). Heat or sonication, or both, have been shown to
X1.5 One sampling procedure that fails to achieve account-
speed up desorption processes (that is, mass transfer of analyte
ability of all VOC phases is the collection of a bulk sample by
from the sampled matrix to the vapor phase or methanol);
using a spatula-type device to completely fill a bottle for
therefore, these more aggressive extraction steps should be
sample storage and transportation (4, 6-10). Samples collected
considered when a totalVOC concentration is needed. In some
andtransferredwithspatula-typedevicesfailtocontrolsurface
instances, incomplete extraction due to either the method used
areaexposure.Moreover,intheprocessoffillingabulksample
or the kinetics associated with the release of VOCs from a
givenmatrixcanresultinanunderestimation(perhapsasmuch
6 as an order of magnitude) of the total VOC concentration
The boldface numbers in parentheses refer to a list of references at the end of
this standard. present in a sample (12, 14, 16, 17).
D4547 − 20
X2. INFORMATION ON THE PRESERVATION OF SAMPLES BY STORAGE UNDER LOW-TEMPERATURE CONDITIONS
X2.1 Losses of VOCs due to biological degradation can be is filled with bags containing a 1:3.6 weight ratio of NaCl to
abated by storing samples at low temperatures, for example, in ice; initially establish a temperature of –14 6 3 °C, and keep
a freezer set at –12 6 5 °C (Practice D6418) (18, 19). This
below –5 °C for up to 20 h (18). However, a shorter period of
method of preservation offers several advantages over the
refrigeration below –5 °C would most likely have resulted if
recommended in-field chemical preservation option: no prior
the cooler had been opened frequently for adding or removing
knowledge of the VOC concentrations is necessary, fewer
samples.Ifdryiceisused,itshouldbeplacedbelowandabove
Department of Transportation (DOT) regulatory requirements
thesamplecontainerswhilenotcomingintodirectcontactwith
mustbemet,andfieldpersonneldon’thavetohandlechemical
them. This can be accomplished by placing the sample con-
solutions or weigh samples. Moreover, freezing, rather than
tainers inside a cardboard box that is then placed inside the
acidification, can be used to preserve VOCs in carbonaceous
cooler with dry ice packed around the sample box. This
soils. An additional concern with sample acidification is that
precaution is necessary because when dry ice comes into direct
with some soil matrices, the formation of acetone, a regulated
contact with some plastic materials it can cause them to crack,
compound itself, has been observed.
or may cause the seal on a bottle to fail because materials (that
is, glass and plastic) have different contractile properties. The
X2.2 Low temperatures that will freeze a soil sample can be
temperature inside a cooler packed with dry ice usually cannot
achieved with a freezer, by mixing table salt and ice together,
be measured with conventional thermometers, since it is often
or by using dry ice. It is recommended that a freezer be used in
below –20 °C. This extreme low temperature may affect the
preference to these other temporary refrigerants, whenever
integrity of the seals of the sample container. There are also
possible. These other methods of achieving low temperatures
some special shipping concerns with the use of these two
that will freeze soil are intended for short-term use (one to
temporary refrigerants. For the salt and ice mixtur
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4547 − 15 D4547 − 20
Standard Guide for
Sampling Waste and Soils for Volatile Organic Compounds
This standard is issued under the fixed designation D4547; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide describes recommended procedures for the collection, handling, and preparation of solid waste, soil, and sediment
samples for subsequent determination of volatile organic compounds (VOCs). This class of compounds includes low molecular
weight aromatics, hydrocarbons, halogenated hydrocarbons, ketones, acetates, nitriles, acrylates, ethers, and sulfides with boiling
points below 200° Celsius (C) that are insoluble or slightly soluble in water.
1.2 Methods of sample collection, handling, storage, and preparation for analysis are described.
1.3 This guide does not cover the details of sampling design, laboratory preparation of containers, and the analysis of the samples.
1.4 It is recommended that this guide be used in conjunction with Guide D4687.
1.5 This guide offers an organized collection of information or a series of options and does not recommend a specific course of
action. This document cannot replace education or experience and should be used in conjunction with professional judgment. Not
all aspects of this guide may be applicable in all circumstances. This ASTM standard is not intended to represent or replace the
standard of care by which the adequacy of a given professional service must be judged, nor should this document be applied
without consideration of a project’s many unique aspects. The word “standard” in the title of this document means only that the
document has been approved through the ASTM consensus process.
1.6 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
Reporting of test results in units other than SI shall not be regarded as nonconformance with this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
This guide is under the jurisdiction of ASTM Committee D34 on Waste Management and is the direct responsibility of Subcommittee D34.01.02 on Sampling Techniques.
Current edition approved Sept. 1, 2015Sept. 1, 2020. Published October 2015September 2020. Originally approved in 1991. Last previous edition approved in 20092015
as D4547 - 09.D4547 – 15. DOI: 10.1520/D4547-15.10.1520/D4547-20.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4547 − 20
D1193 Specification for Reagent Water
D1586D1586/D1586M Test Method for Standard Penetration Test (SPT) and Split-Barrel Sampling of Soils
D3550D3550/D3550M Practice for Thick Wall, Ring-Lined, Split Barrel, Drive Sampling of Soils
D4687 Guide for General Planning of Waste Sampling
D4700 Guide for Soil Sampling from the Vadose Zone
D5058 Practices for Compatibility of Screening Analysis of Waste
D5681 Terminology for Waste and Waste Management
D5792 Practice for Generation of Environmental Data Related to Waste Management Activities: Development of Data Quality
Objectives
D6051 Guide for Composite Sampling and Field Subsampling for Environmental Waste Management Activities
D6232 Guide for Selection of Sampling Equipment for Waste and Contaminated Media Data Collection Activities
D6282D6282/D6282M Guide for Direct Push Soil Sampling for Environmental Site Characterizations
D6418 Practice for Using the Disposable En Core Sampler for Sampling and Storing Soil for Volatile Organic Analysis
(Withdrawn 2018)
D6640 Practice for Collection and Handling of Soils Obtained in Core Barrel Samplers for Environmental Investigations
D8170 Guide for Using Disposable Handheld Soil Core Samplers for the Collection and Storage of Soil for Volatile Organic
Analysis
2.2 Federal Standard:
Title 49Code of Federal Regulations (CFR), Title 49, Part 172 Transportation, Code of Federal Regulations (CFR), Part 172, List
of Hazardous Substances and Reportable Quantities
3. Terminology
3.1 sample, n—a portion of material taken from a larger quantity for the purpose of estimating properties or composition of the
larger quantity. (D5681)
3.1 Definitions:
3.1.1 sample, n—a portion of material taken from a larger quantity for the purpose of estimating properties or composition of the
larger quantity. (D5681)
3.1.2 subsample, n—a portion of a sample taken for the purpose of estimating properties or composition of the whole sample.
(D6051)
3.1.2.1 Discussion—
A subsample, by definition, is also a sample.
3.2 subsample, n—a portion of a sample taken for the purpose of estimating properties or composition of the whole sample.
(D6051)
3.2.1 Discussion—
A subsample, by definition, is also a sample.
4. Summary of Guide
4.1 This guide addresses the use of tools for sample collection and transfer, conditions for sample storage, sample preservation,
and two common means of sample preparation for analysis. Special attention is given to each step from sample collection to
analysis to limit the loss of VOCs by volatilization and biodegradation. The sample collected and analyzed should be representative
of the matrix material sampled. The two methods cited for the preparation of samples for VOC analysis are methanol extraction
and vapor partitioning (that is, purge-and-trap, headspace, and vacuum distillation). The method of sample preparation for VOC
analysis should be consistent with the data quality objectives (see Practice D5792).
5. Significance and Use
5.1 This guide describes sample collection and handling procedures designed to minimize losses of VOCs. The principal
mechanisms for the loss of VOCs from materials during collection, handling, and storage are volatilization and biodegradation.
Susceptibility of various VOCs to these two loss mechanisms is both compound and matrix specific. In general, compounds with
The last approved version of this historical standard is referenced on www.astm.org.
Available from Superintendent of Documents, U.S. Government Printing Office, Washington, DC 20402.
D4547 − 20
higher vapor pressures are more susceptible to volatilization than compounds with lower vapor pressures. Also, aerobically
degradable compounds are generally more susceptible to biodegradation than anaerobically degradable compounds. In some cases,
the formation of other compounds not originally present in the material can occur. Loss or gain of VOCs leads to analytical results
that are unrepresentative of field conditions.
5.2 Ancillary information concerning sample collection, handling, and storage for VOC analysis is provided in Appendix
X1Appendix X1 – Appendix X3, Appendix X2, and Appendix X3. These appendixes and cited references are recommended
reading for those unfamiliar with the many challenges presented during the collection, handling, and storage of samples for VOC
analysis.
6. Selection of Sample Preparation Method for VOC Analysis
6.1 Introduction:
6.1.1 Sample collection, handling, and preservation methods should be compatible with the method used to prepare the sample
for VOC analysis, and meet the project’s data quality objectives (see Practice D5792). Preparation of a sample for instrumental
analysis can be initiated either in the field or laboratory. In either case, prior to analysis, the sample should be placed into a tared
volatile organic analysis (VOA) vial or bottle meeting the specifications given in 7.3. When working with an uncharacterized solid
waste, it is advisable to perform compatibility tests (see Test Methods Practices D5058) between the sample material and the
solution (see 6.2 and 6.3) into which it will be transferred in preparation for analysis. For instance, when collecting highly
contaminated soils or waste of unknown composition, it is strongly recommended that preliminary testing be performed to
adequately characterize the waste materials so that when the user applies the procedures cited in this guide, there will be no
chemical reaction which may jeopardize the user’s safety.
6.1.2 Figs. 1 and 2 are flow diagrams showing some different options for combining sample collection, handling, and preparation
methods for instrumental analysis.
6.2 Methanol Extraction:
6.2.1 This method involves the extraction of VOCs from a sample with methanol and the subsequent transfer of an aliquot of the
extract to water for either purge-and-trap, headspace analysis, or vacuum distillation.
6.2.2 Advantages of methanol extraction areare: (1) large samples or composite samples, or both, can be collected to enhance
representatives (see Guide D6051), (2) biodegradation is inhibited, (3) an efficient extraction of VOCs from the matrix materials
can be achieved with methanol due to its strong affinity for these compounds and favorable wetting properties, (4) a subsample
can be analyzed several times, and (5) sample extracts can be archived, if verified that VOC losses have not occurred (see 10.1.1).
6.2.3 The primary disadvantages of methanol extraction areare: (1) samples may have to be shipped as a flammable liquid
depending on the amount of methanol present (for example, U.S. DOT reg. 49CFR§172.101), Reg. 49 CFR§172.101), (2) hazards
to personnel due to methanol’s toxicity and flammability, (3) detection limits are elevated due to analyte dilution, (4) possible
interference of the methanol peak with VOCs of interest, (5) potential adverse impact of methanol on the performance of certain
gas chromatograph/detector systems, and (6) samples extracted with methanol must be disposed of as a regulated waste.
6.2.4 Logistical challenges of performing these tasks in the field can be overcome by extracting samples with methanol once they
have been received in a laboratory, provided that the samples are transported in an airtight container (see 7.3.2, 9.1, and 9.2).
Furthermore, if VOC levels are unknown, a replicate sample can be obtained and screened to determine if methanol extraction is
appropriate for the expected contaminant concentrations.
D4547 − 20
FIG. 1 Sample Handling Options for Cohesive Materials
D4547 − 20
FIG. 2 Sample Handling Options for Non-CohesiveNoncohesive and Cementitious Materials
D4547 − 20
6.3 Vapor Partitioning:
6.3.1 Vapor partitioning involves the direct analysis of a sample by either purge-and-trap, headspace, or vacuum distillation. In
each case, the sample is placed into a tared volatile analysis (VOA) vial or flask (for vacuum distillation) containing water or a
preservative solution (for example, acidified water) from which the vapor is removed for analysis without the container being
opened.
6.3.2 The principal advantages of this method areare: (1) it can offer lower detection limits than methanol extraction because no
dilution is involved, (2) there are no organic solvent interferences, and (3) there is no use of regulated organic solvents, which may
require special shipment, disposal, and field handling practices.
6.3.3 The disadvantages associated with vapor partitioning areare: (1) the VOA vial (VOA vials are different sizes for automated
purge-and-trap and headspace instrumentation) or adapter used in conjunction with a VOA vial, or both, often are instrument
specific, (2) sample size is limited (<10 g) by automated systems, (3) a matrix-appropriate method of preservation may be
necessary (see Appendix X2), (4) vapor partitioning is less efficient at recovering VOCs from some materials than methanol
extraction, and (5) when using purge-and-trap or vacuum distillation, only a single analysis of the same sample can be made;
similarly only a single analysis may be possible with headspace analysis unless concentrations allow for the use of a small injection
volume.
6.3.4 Limitations imposed by vapor phase partitioning methods with regard to number of analyses that can be performed on a
single sample can be addressed by taking replicate samples.
6.3.5 When employing vapor phase partitioning methods, the logistical challenges of performing sample preparation in the field
(see 7.3.3) can be avoided by performing the preparation step in the laboratory, so long as the sample is transported to the
laboratory in an airtight container (see 7.2.1). If VOC levels are unknown, a replicate sample can be obtained and screened to
determine if it is appropriate to use a vapor partitioning method of sample preparation.
7. Sampling Tools and Containers
7.1 All sample handling devices and vessels used to collect and store samples for analysis should be constructed of nonreactive
materials that will not sorb, leach, or diffuse constituents of interest. Examples of materials that meet these criteria are glass,
stainless steel, steel, and brass. Materials,Materials such as polytetrafluoroethylene (PTFE) and many rigid plastics also can be
used; however, it should be recognized that they may have some limited adsorptive properties or allow slow diffusive passage of
some VOCs. Materials which show limited reactivity can be used when they have a very short period of contact with the sample
or when they are necessary for making airtight (hermetic) seals. Collection tools and storage containers made of materials other
than those cited in this section should only be used after they have demonstrated equivalency (see 7.2.1). All collection tools and
storage containers should be cleaned in a manner consistent with their intended use.
7.2 Tools—There are often several steps to sampling, particularly if it involves obtaining bulk material from the subsurface. Most
of the equipment used to obtain samples from the subsurface was originally developed for the geotechnical industry; however,
several devices have been developed specifically for environmental sampling by direct push methods (Guide D6282D6282/
D6282M). The subsurface bulk sampling systems are designed to obtain intact cylindrical cores of material, ranging anywhere
from 2.5 to 10.2 cm in diameter and 30.5 cm or more in length. Two geotechnical tools that have been used for subsurface sample
collection are the split-spoon sampler (Test Method D1586D1586/D1586M), which opens to expose the entire length of the
material obtained for subsampling, and core barrel liners (ring-lined barrel sampling, see Practice D3550D3550/D3550M) that are
typically subsampled through open ends. Core barrel liners should not be used for storage of samples intended for VOC analysis
because they do not have airtight seals,seals; see Appendix X1. Single tube and dual tube Single-tube and dual-tube sampling
devices have been developed for environmental applications (Guide D6282D6282/D6282M) and are usually operated by direct
push methods. Sample liners used in the bulk sampling systems come in a variety of lengths and materials (stainless steel, brass,
PTFE, rigid plastics, etc.). Additional information on the design and application of different types of subsurface collection systems
that are available can be found in GuideGuides D4700, D6232, and Guide D6282D6282/D6282M. Subsurface materials retrieved
for VOC characterization should be obtained (sampling tubes filled and brought to the surface) as quickly as possible and remain
intact and undisturbed until they are subsampled (see Practice D6640). Subsampling a bulk sample should occur as quickly as
possible after it is brought to the surface. It is important for subsampling to occur quickly because for bulk sample retrieval systems
where the material is exposed directly to the atmosphere for a period of time during subsampling, there is an opportunity for VOC
loss to occur. Suggested liner configurations and examples of sampling techniques for bulk soil samples collected by single tube
D4547 − 20
single-tube direct push methods that provide limited exposure of the soil being sampled to the atmosphere,atmosphere are
described in Appendix X3 (see 8.18.1 – 8.3 through 8.3for additional sampling guidance).
7.2.1 In addition to the coring devices which retrieve bulk quantities of material, there are smaller hand-operated coring tools for
obtaining samples of the appropriate size (for example, coring devices capable of collecting 3- and 15-cm volumes, for collection
of approximately 5- and 25-g samples, respectively) for analysis (see Fig. 3 and Guide D8170). If one of these smaller coring
devices is used to store the sample (see 9.1.1), the main body shall be constructed of materials that are nonreactive and have airtight
seals that show limited sorption and penetration of VOCs. Hand-operated coring devices that are used to store samples shall be
evaluated using the procedure given in Appendix X4 to ensure that the device meets these criteria. Any alternate evaluation
technique shall produce equal or better recoveries of the compounds stated in Appendix X4. When the coring device is only used
to rapidly transfer the sample to a VOA vial, corer material requirements are less stringent.
7.3 Containers—VOA vials and bottles used for storage and preparation of samples for analysis should be made of glass and have
airtight seals. To achieve an airtight seal, these containers should have a thick septum cushion between the sealing material (PTFE)
and cap (rigid plastic screw cap or aluminum crimp top). PTFE-lined caps that do not have flexible septum backing often fail to
achieve a liquid or airtight seal. Furthermore, the thickness of the PTFE used for a lined septum should be at least 0.254 mm.
7.3.1 Preparation of VOA Vial or Bottle—Record the tared weight of the VOA vial or bottle and cap prior to the sampling event.
Moreover, depending on the method of sample preparation and analysis, the vessel may also contain a solvent, or VOC free water,
and if requiredsolvent or VOC-free water and, if required, a PTFE or glass coated glass-coated stir bar at the time of sample
collection (see Section 9).
7.3.2 Preparation of Containers for Methanol Extraction—Container preparation applies to both field and laboratory sample
preparation. The appropriate volume of analytical-grade methanol (high performance (high-performance liquid chromatography,
spectrographic or purge and trap) spectrographic, or purge-and-trap) is added to the organic-free container by the laboratory that
supplies the container, by the sample collector, or by a third party. The party that adds the methanol to the container should also
be responsible for providing trip blanks (see Guide D4687). If methanol is present in the sample container in the field, this
FIG. 3 Miniature Generic Disposable Handheld Soil Core Sampler
D4547 − 20
container should be opened only to add the sample(s). The tared weight of the container with methanol should be recorded prior
to adding the sample to the container. A predetermined volume of sample that corresponds to a weight in grams (g) that is
equivalent to or less than the volume (mL) of methanol can then be introduced. The ratio (typically 1:1 to 10:1, methanol to
material) between the two constituents should allow for formation of a clear layer of methanol over the sample after thorough
mixing. The difference in weight of the container and cap, measured before and after the sample is introduced, is used to establish
the sample’s wet weight. Because of the water (moisture) present in most samples, calculation of the recovery of sample analyte
concentrations should account for this source of dilution.
7.3.3 Preparation of Containers for Vapor Partitioning—Container preparation applies to both field and laboratory sample
preparation. This method allows for the direct analysis of a sample by either purge-and-trap or headspace techniques. In both cases,
the sample is placed into a VOA vial from which the vapor (only a portion for headspace analysis) is removed for analysis without
the container being opened. Moreover, water that contains no detectable levels of VOCs and, if required, a PTFE or glass coated
glass-coated stir bar should be present in the VOA vial prior to introducing the sample. Current automated equipment for
purge-and-trap and headspace systems typically use 40 and 22-mL VOA vials, respectively. The volume of water used for these
two different systems is typically 10 mL or less. Furthermore, the tared weight of the container should be recorded prior to adding
the sample. The difference in weight of the container, measured before and after the sample is added, is used to determine the
sample’s wet weight.
7.3.4 Preparation of Samples Transported and Stored in Solventless VOA Vials or Bottles—When samples are held in a VOA vial
or bottle, methanol can be added to the container by piercing the septum. In the case of a 5-g sample contained in a 40-mL VOA
vial, 5.0 mL of methanol can be added without over pressurization. overpressurization. When larger samples and bottles, or both,
are used, experimental trials should be performed to establish what volume of solution can be added. If performed manually, a
0.635 mm nominal outer diameter and 0.318 nominal inner diameter or smaller needle (see NoteNote 11) should be used to add
the methanol. After the methanol is introduced, the soil sample should be dispersed. If the sample is held beyond the
laboratory-prescribed extraction and analysis period (typically 24 h or less), it is recommended that either the pierced septum be
replaced with one that is intact or that an aliquot of the methanol be transferred to an appropriate vessel for storage. If the pierced
septum is replaced, it should be replaced with one that is intact by replacing the cap of the vial or bottle with a cap having an intact
septum. When the laboratory prescribed laboratory-prescribed extraction method uses elevated temperatures, or sonication, or
both, it is recommended that the pierced septum be replaced prior to this treatment. To limit the loss of VOCs when replacing the
cap or transferring an aliquot of methanol, the container should be cooled to 4 6 2°C,2 °C, and these functions should be
performed as quickly as possible. Cooling the sample lowers the vapor pressure of the analytes of interest and reduces the pressure
created by adding the methanol. By having methanol present in the collection vessel prior to introducing the sample, the additional
precautions associated with a pierced septum can be avoided. In addition, having methanol present in the collection vessel reduces
the possibility of VOCs being lost during the transfer step (that is, extrusion of a plug of soil from the sampling tool into the empty
vessel).
NOTE 1—This is a 23-gage needle.
8. Sample Collection
8.1 General Sampling Guidance—Using an appropriate sized coring tool (see below), collect samples from freshly exposed
surfaces of the soil or waste with minimal disturbance. The soil or waste to be sampled may be from an intact pit face or the
relatively undisturbed contents in a split-barrel sampler, a core barrel liner, or a liner from a single or double tube double-tube direct
push sampling system. Before sampling, remove several centimeters of material to expose a fresh surface. Obtain a sample by
pressing (or hammering in a few cases) the end of the coring tool into the pit face or into the end or side of a larger core sample.
Remove the coring tool. The optimum diameter of the coring tool depends on the following: size of the opening on the collection
vial or bottle (tool should fit inside mouth), particle size of the solid materials (for example, gravel-size particles would require
larger samplers), and volume of sample required for analysis. For example when a 5-g sample of soil is specified, only a single
3 3
3-cm volume would need to be collected (assuming the soil has density of 1.7 g/cm ). Larger sample masses or composite samples
may be preferred as the heterogeneity of the material increases.
8.1.1 Sampling of Cohesive butBut Uncemented Materials Using Devices Designed to Obtain a Sample Appropriate for
Analysis—Collect appropriate size sample for analysis using a metal or rigid plastic coring tool (see Figs. 3 and 4). For example,
coring tools for the purpose of transferring a sample can be made from disposable plastic syringes by cutting off the tapered front
end and removing the rubber cap from the plunger. Pre-fabricated plastic syringes/coring tools are available commercially. This
type of coring device is not appropriate for sample storage. These smaller coring devices help maintain the sample structure during
collection and transfer to the VOA vial or a larger bottle. When inserting a clean coring tool into a fresh surface for sample
collection, air should not be trapped behind the sample. If air is trapped, it could either pass through the sampled material causing
D4547 − 20
FIG. 4 A Coring Tool Made by Cutting the Tip Off a Plastic Syringe
VOCs to be lost or cause the sample to be pushed prematurely from the coring tool. For greater ease in pushing coring tools into
the solid matrix, their front edge can be sharpened.
8.1.2 Obtain an undisturbed sample by pushing the barrel of the coring tool into a freshly exposed surface and removing the corer
once filled. Clean the exterior of the barrel by wiping with a clean disposable towel. If the coring tool sampler is used as a storage
container, cap the open end after ensuring that the sealing surfaces are cleaned (see 9.1.1). If the device is solely used for collection
and not storage, immediately extrude the sample into a tared VOA vial or bottle by gently pushing the plunger. During transfer
of the sample into the container, care should be taken to prevent the sample from contacting the sealing surfaces which can
comprise these surfaces and prevent an airtight seal. The volume of material collected should not cause excessive stress on the
coring tool during intrusion into the material, or be so large that the sample easily falls apart during extrusion. Obtaining and
transferring a sample should be done rapidly (<10 s) to reduce volatilization losses. If the vial or bottle contains methanol or
another liquid, it should be held at an angle when extruding the sample into the container to minimize splashing. Just before
capping, a visual inspection of the lip and threads of the sample vessel should be made, and any foreign debris should be removed
with a clean towel, allowing an airtight seal to form.
8.2 Devices That Can beBe Used for Sampling a Cemented Material—Samples of hard or cementitious material may be obtained
by fragmenting a larger portion of the material using a clean chisel to generate aggregate(s) of a size that can be placed into a tared
VOA vial or bottle. When transferring the aggregate(s), precautions must be taken to prevent compromising the sealing surfaces
and threads of the container. Losses of VOCs by using this procedure are dependent on the location of the contaminant relative
to the surface of the material being sampled. Therefore, caution should be taken in the interpretation of the data obtained from
materials that fit this description. As a last resort when this task can not be performed on-site, a large sample can be collected in
a vapor-tight container and transported to the laboratory for further processing (see 9.1 and 9.2). Collecting, fragmenting, and
adding the sample to a container should be accomplished as quickly as possible.
D4547 − 20
8.3 Devices That Can beBe Used for Sampling a Noncohesive Material—When sampling gravel,gravel or a mixture of gravel and
fines,fines that cannot be easily obtained or transferred using coring tools, as a last resort, a sample can be quickly transferred using
a spatula or scoop (see discussion concerning the use of a spatula or scoop to collect samples for VOC analysis, in Appendix X1).
If the tared collection vial or bottle contains methanol or an aqueous solution, transfer the sample to the vial or bottle with minimal
splashing and without the spatula or scoop contacting the liquid contents. For some solids, a wide-bottom funnel or similar
channeling device may be necessary to facilitate transfer to the container and prevent compromising of the sealing surfaces of the
container. Caution should be taken in the interpretation of the data obtained from materials that fit this description. Losses of VOCs
are likely because of the nature of the sampling method and the noncohesive nature of the material exposes more surface area to
the atmosphere than for other types of samples. Another potential source of error during the sampling process,process is the
separation of coarser materials from fines, which can bias the concentration data if the different particle sizes, which have different
surface areas, are not properly represented in the sample.
8.4 Sampling Location Physical Characteristics—Collect a separate co-located sample within a couple of centimeters and from
the same stratum and place in a suitable container for the determination of percent moisture content of the waste or soil. This
sample should be collected in a container suitable for the retention of soil moisture. The location adjacent to where the sample for
VOC analysis was collected should be inspected visually and its characteristics logged. This adjacent material can also be retained
for determining other relevant properties, such as general appearance, color, presence of oils, other visible signs of contamination,
grain-size distribution, organic carbon content, etc. Collection of these ancillary samples should be performed after the collection
of samples for VOC analysis.
9. Sample Storage, Transportation, and Preservation
9.1 Sample Storage and Transportation:
9.1.1 General Considerations—During an initial 48 h storage and transportation period, all samples for VOC analysis should be
held in air-tightairtight containers and cooled to at least 4 6 2°C.2 °C. For storage periods beyond 48 h, these air-tightairtight
containers should contain a chemical preservative or be held under conditions that ensure retention of the analytes of concern (see
9.2).
9.2 Sample Preservation Beyond 48 Hours:
9.2.1 Currently, it is recommended that samples stored in an air-tightairtight container should be held for no longer than 48 h at
4 6 2°C2 °C prior to analysis or preservation. Longer storage times at 4 6 2°C2 °C can be applied if it can be demonstrated that
the VOC concentrations are not affected or that the data generated at the time of analysis meets the data quality objectives.
Extended sample storage can be achieved by using either physical or chemical methods of preservation. Sample preservation can
be initiated at the time of sample collection or after arrival in a laboratory.
9.2.2 Physical Preservation—Samples contained in a VOA vial, a bottle, or coring device are physically preserved by low
temperature low-temperature storage (for example, preferably in a freezer set for -12–12 6 5°C,5 °C, or by using a temporary
refrigerant, for example, a mixture of salt and ice or dry ice,ice; see Appendix X2). Under this condition, sample storage (total time
period starting at collection) can be extended up to 14 days. Longer periods of low temperature low-temperature storage can be
used provided it can be demonstrated that the VOC concentrations are not affected or that the data generated at the time of analysis
meets the data quality objective. When low temperature low-temperature storage is used for samples intended for analysis by vapor
partitioning, VOC free VOC-free water and, if required, a PTFE or glass coated glass-coated stir bar should be present prior to
adding the sample in the field (see 7.3.3). Special precautions may be necessary, for example, positioning the sample vessel on its
side, when using a low temperature low-temperature condition with VOA vials smaller than 40 mL, or when more than 5 mL of
water is present, because of the potential for breakage caused by the formation of ice. When the sample is intended to be prepared
by methanol extraction, this solvent can be introduced through the septa of the storage vessel after low temperature
low-temperature storage (see 7.3.4). In either case, immediately following low temperature low-temperature storage of samples in
VOA vials or bottles, the caps should be checked and tightened, if necessary. For coring tools used as storage containers, the sample
is extruded into a prepared VOA vial after low-temperature storage (see PracticeGuide D6418D8170).
9.2.3 Chemical Preservation—Samples immersed in methanol are chemically preserved. Under this condition, storage (total time
period starting at collection) at 4 6 2°C2 °C can be extended for up to 14 days. Longer periods of methanol preservation can be
SW-846, Method 5035 A (July 2002), 3rd Update.
D4547 − 20
used provided if it can be demonstrated that the VOC concentrations are not affected or that the data generated at the time of
analysis meets the data quality objectives. A chemical preservation method for samples intended to be analyzed by vapor
partitioning,partitioning is acidification to a pH of 2 or less with either sodium bisulfate or hydrochloric acid. However,
VOC-contaminated samples containing carbonates should not be added to an acidic solution because of effervescence (for
guidance on testing for carbonates and the use of these acids to preserve samples, see Appendix X5). Precautions must also be
taken when preserving by acidification because certain compounds within the following classes;classes, olefins, ketones, esters,
ethers, and sulfides, will react under a low pH condition (see Appendix X2 for other potential problems). Acidified sample storage
(total time period starting at collection) can be extended for up to 14 days at 4 6 2°C.2 °C. Longer periods of acidified sample
storage can be used provided it can be demonstrated that the VOC concentrations are not affected or that the data generated at the
time of analysis meets the data quality objectives.
10. Sample Preparation
10.1 Methanol Extraction—Completely disperse samples in methanol, if possible, by shaking, vortexing, or by sonication. Allow
suspended particulates to settle so that an aliquot of clear supernatant can be removed for analysis. Some materials will show a
slow release of VOCs to methanol; therefore, the dissolved VOC concentration may increase with increasing extraction time (see
Appendix X1). This increased extraction of VOCs over time may be particularly pronounced with cementitious materials or
materials that strongly adsorb VOCs. Transfer of an aliquot for analysis should be performed using a clean, liquid-tight syringe
(composed of glass and metal) to withdraw the aliquot through the septum of the sample container. If necessary, the cap of the
sample container can be removed, and an aliquot can be collected with either a clean syringe or pipette. However, the time that
the cap is off the sample container should be minimized as much as possible to limit volatilization losses of solvent and VOCs.
For purge-and-trap analysis, methanol aliquot volumes of less than 0.2 mL are typically transferred to a vessel containing 5 mL
of organic-free water. For headspace analysis, methanol aliquot volumes as large as 1.0 mL, depending on the detector and analytes
of concern, can be transferred to VOA vials containing 10 mL of organic-free water. When the re-analysis of the sample extract
is a possibility, an aliquot of the extract should be archived by transferring it to an airtight vial (for example, 2 mL) with a
PTFE-lined cap.
10.1.1 Periodic weighing of the sample container can be used to determine if a hermetic seal is being maintained, that is, no weight
loss of methanol.
10.2 Vapor Partitioning—Completely disperse samples in water, if possible, by shaking, vortexing, or by sonication. This mixing
of the solid material with the aqueous solution not only helps prevent the plugging of the sparging needle used by purge-and-trap
systems, but also assists in attaining an equilibrium state by completely exposing the sample to the partitioning solution. To
enhance partitioning into the vapor phase, automated purge-and-trap and headspace analysis systems warm the sample to a selected
temperature (typically 40°C40 °C for purge-and-trap, this temperature or higher for headspace) prior to removing vapors for
analysis.
11. Keywords
11.1 field sampling; methanol extraction; sample collection; sample handling; soil; solid wastes; vapor phase partitioning; volatile
organic compounds
APPENDIXES
(Nonmandatory Information)
X1. ADDITIONAL INFORMATION CONCERNING SAMPLE COLLECTION AND HANDLING FOR VOC ANALYSIS
X1.1 Volatile organic compounds (VOCs) are among the most frequently identified soil contaminants at Superfund and other
hazardous waste sites. Because some VOCs are potentially mutagenic, carcinogenic, and teratogenic, they often are key factors in
the risk assessment process at these sites. These risk assessments and other remedial decisions depend upon an accurate
understanding of the levels and extent of VOC contamination in soils and other materials.
D4547 − 20
X1.2 In most materials, VOCs coexist as gaseous, liquid, and solid (sorbed) phases. The VOC equilibrium that exists among these
phases is controlled by physiochemical properties, material properties, and environmental variables ((1, 2).). Unaccounted loss
of analytes from any phase may result in rendering the sample unrepresentative of the material from which it was taken. For this
reason, sample collection, handling, and analysis must be performed under conditions that maintain the accountability of all phases
present ((3).).
X1.3 In general, uncontrolled losses of VOCs from materials occur through two mechanisms: volatilization and biodegradation.
Volatilization losses occur whenever gaseous molecules, which have diffusion coefficients up to four orders of magnitude greater
than liquid diffusion coefficients, are allowed to move freely. Therefore, whenever a new surface is exposed, VOC losses are
incurred. The extent to which VOCs are lost depends on the vapor phase concentration (analyte vapor pressure), surface area
exposed, duration of exposure, porosity of matrix, and perhaps meteorological conditions ((4).).
X1.4 Biological degradation of VOCs in samples is usually dominated by aerobic processes because many conventional intrusive
collection methods expose the sample to the atmosphere. The rate of this biological degradation is dependent on several factors,
including the indigenous microbiological population, chemical properties of the VOC, and temperature. Provided that sufficient
quantities of electron acceptors, nutrients, and moisture are present, indigenous microbes continue to aerobically degrade
compounds even when stored at 4°C (4 °C (5).). Non-halogenated aromatic compounds are quite susceptible to this loss
mechanism. To inhibit biodegradation of these compounds, the sample can be immersed in methanol, frozen (see Appendix X2),
or acidified to a pH of 2 or less.
X1.5 One sampling procedure that fails to achieve accountability of all VOC phases is the collection of a bulk sample by using
a spatula-type device to completely fill a bottle for sample storage and transportation ((4, 6-10).). Samples collected and transferred
with spatula-type devices fail to control surface area exposure. Moreover, in the process of filling a bulk sample bottle to capacity,
the sealing surfaces often become compromised (dirty), preventing a vapor-tight seal during storage. For these reasons, this
procedure has been shown to result in concentrations that are less than 10 % of the in-situ contamination ((6-9).).
X1.6 Another procedure that often performs poorly is the use of core barrel liners covered with either PTFE or aluminum foil for
sample transportation and storage, because these barriers fail to serve as air-tightairtight seals ((3, 11).). Sheets of PTFE that are
only 0.05 or 0.08 mm thick are easily penetrated by VOC vapors. Elastic formulations of PTFE (for example, white plumber’s tape)
show much greater rates of VOC penetration than non-elasticnon-elastic, semi-translucent formulations. Aluminum foil fails
because an airtight seal is often not achieved when this material is folded over the end of a core barrel liner and it sometimes
corrodes, allowing small holes to form. The addition of plastic caps and sealing tape does not improve the performance of either
PTFE or aluminum foil, since pliable plastics sorb VOCs and sealing tapes often contain VOCs in the adhesive, which serves as
a potential artifact source.
X1.7 The procedures discussed in this guide are designed to limit VOC losses by volatilization and biodegradation. This is
accomplished by stressing that (1) samples be collected only from freshly exposes surfaces, (2) collection and transfer of a sample
be performed quickly and with minimal disruption to its physical state, (3) samples be held under conditions that ensure retention
of the analytes of interest, and,and (4) in the case of samples collected for vapor partitioning methods of analysis (purge-and-trap
or headspace), the VOA vial’s airtight seal never be broken prior to analysis.
X1.8 An additional source of inaccuracy in VOC analyses is their incomplete extraction from the sampled material ((12-17).). This
source of error can either be due to differences between sample preparation methods (that is, methanol extraction versus vapor
partitioning) or the kinetics of the desorption processes. In general, as the indigenous total organic carbon content increases in a
matrix, recoveries using vapor partitioning methods would be expected to decrease compared to methanol extraction, particularly
for the more hydrophobic analytes ((17).). Heat or sonication, or both, have been shown to speed up desorption processes (that is,
The boldface numbers in parentheses refer to a list of references at the end of this standard.
D4547 − 20
mass transfer of analyte from the sampled matrix to the vapor phase or methanol); therefore, these more aggressive extraction steps
should be considered when a total VOC concentration is needed. In some instances, incomplete extraction due to either the method
used or the kinetics associated with the release of VOCs from a given matrix can result in an under estimation underestimation
(perhaps as much as an order of magnitude) of the total VOC concentration present in a sample ((12, 14, 16, 17).).
X2. INFORMATION ON THE PRESERVATION OF SAMPLES BY STORAGE UNDER LOW TEMPERATURE LOW-
TEMPERATURE CONDITIONS
X2.1 Losses of VOCs due to biological degradation can be abated by storing samples at low temperatures, for example, in a
freezer set at -12–12 6 5°C (5 °C (Practice D6418,) (18, 19).). This method of preservation offers several advantages over the
recommended in-field chemical preservation option: no prior knowledge of the VOC concentrations is necessary, fewer
Department of Transportation (DOT) regulatory requirements must be met, and fi
...


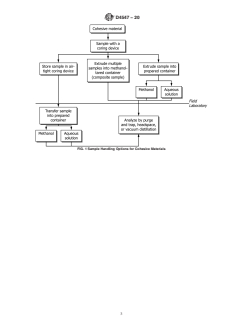
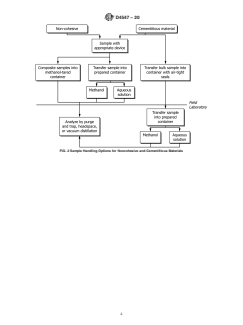



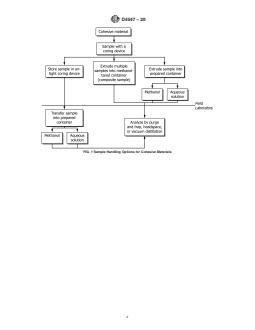
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...