ASTM D4742-23
(Test Method)Standard Test Method for Oxidation Stability of Gasoline Automotive Engine Oils by Thin-Film Oxygen Uptake (TFOUT)
Standard Test Method for Oxidation Stability of Gasoline Automotive Engine Oils by Thin-Film Oxygen Uptake (TFOUT)
SIGNIFICANCE AND USE
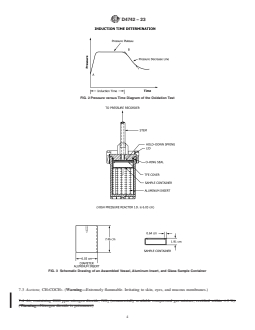
5.1 This test method is used to evaluate oxidation stability of lubricating base oils with additives in the presence of chemistries similar to those found in gasoline engine service. Test results on some ASTM reference oils have been found to correlate with sequence IIID engine test results in hours for a 375 % viscosity increase.5 The test does not constitute a substitute for engine testing, which measures wear, oxidation stability, volatility, and deposit control characteristics of lubricants. Properly interpreted, the test may provide input on the oxidation stability of lubricants under simulated engine chemistry.
5.2 This test method is intended to be used as a bench screening test and quality control tool for lubricating base oil manufacturing, especially for re-refined lubricating base oils. This test method is useful for quality control of oxidation stability of re-refined oils from batch to batch.
5.3 This test method is useful for screening formulated oils prior to engine tests. Within similar additive chemistry and base oil types, the ranking of oils in this test appears to be predictive of ranking in engine tests. When oils having completely different additive chemistry or base oil type are compared, oxidation stability results may not reflect the actual engine test result.
5.4 Other oxidation stability test methods have demonstrated that soluble metal catalyst supplies are very inconsistent and they have significant effects on the test results. Thus, for test comparisons, the same source and same batch of metal naphthenates shall be used.
Note 2: It is also recommended as a good research practice not to use different batches of the fuel component in test comparisons.
SCOPE
1.1 This test method evaluates the oxidation stability of engine oils for gasoline automotive engines. This test, run at 160 °C, utilizes a high pressure reactor pressurized with oxygen along with a metal catalyst package, a fuel catalyst, and water in a partial simulation of the conditions to which an oil may be subjected in a gasoline combustion engine. This test method can be used for engine oils with viscosity in the range from 4 mm2/s (cSt) to 21 mm2/s (cSt) at 100 °C, including re-refined oils.
1.2 This test method is not a substitute for the engine testing of an engine oil in established engine tests, such as Sequence IIID.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Exception—Pressure units are provided in psig, and dimensions are provided in inches in Annex A1, because these are the industry accepted standard and the apparatus is built according to the figures shown.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 8.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2023
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.09.0G - Oxidation Testing of Engine Oils
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Feb-2019
- Effective Date
- 01-May-2017
- Effective Date
- 15-Jul-2015
- Effective Date
- 01-Jul-2014
- Effective Date
- 01-Oct-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 15-Jan-2012
- Effective Date
- 15-Jan-2012
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
- Effective Date
- 15-May-2011
- Effective Date
- 15-Jun-2009
- Effective Date
- 01-Mar-2009
- Effective Date
- 01-Oct-2008
Overview
ASTM D4742-23 is the standard test method developed by ASTM International for evaluating the oxidation stability of gasoline automotive engine oils using the Thin-Film Oxygen Uptake Test (TFOUT). This test simulates the oxidative environment of a gasoline engine by subjecting oil samples to elevated temperatures, pressurized oxygen, and metal catalysts, thereby providing critical data on the oil's resistance to oxidation. The method serves as a reliable bench test for screening lubricating base oils, including re-refined oils, and allows for quality control and preliminary assessment before full engine testing.
This standard is applicable to engine oils with kinematic viscosities between 4 mm²/s and 21 mm²/s at 100°C, reflecting a wide range of gasoline automotive engine oils. Although TFOUT provides valuable information on oxidation stability, it does not substitute for comprehensive engine testing, which also measures wear, volatility, and deposit control characteristics.
Key Topics
- Oxidation Stability Evaluation: Measures the time until rapid oxidation occurs, known as the "oxidation induction time," helping predict how an oil may perform under engine-like oxidative stress.
- TFOUT Procedure: Involves subjecting an oil sample-combined with an oxidized fuel component, metal catalyst mixture, and water-to 160°C in a rotating high-pressure reactor, simulating real-world engine conditions.
- Quality Control for Re-refined Oils: Particularly effective in assuring consistency across batches of re-refined base oils, making it an important tool for manufacturers utilizing recycled materials.
- Bench Screening Tool: Offers a rapid and cost-effective method to compare formulated oils prior to more expensive and time-consuming engine testing, especially within similar base oil types and additive chemistries.
- Importance of Consistent Reagents: Ensures test reliability by using the same batches of metal catalysts and fuel components throughout comparative studies, minimizing variability in results.
Applications
ASTM D4742-23 is widely utilized in the petroleum and lubricants industry, particularly for:
- Manufacturing Quality Assurance: Allows lubrication manufacturers to monitor batch-to-batch consistency in oxidation stability, especially when using re-refined base stocks.
- Research and Development: Supports R&D teams in screening new additive formulations or base oil types, enabling early identification of oxidation performance before engine trials.
- Formulation Benchmarking: Provides valuable comparative data to rank the oxidation stability of various oil formulations, aiding in product development and differentiation.
- Regulatory and Compliance: Helps ensure that lubricants meet industry guidelines for oxidation resistance, contributing to prolonged engine life and reduced maintenance costs.
- Predictive Assessment: While not a substitute for engine testing (Sequence IIID and others), TFOUT can indicate performance trends and potential issues, guiding further testing and validation.
Related Standards
Several other ASTM and industry standards complement and reference ASTM D4742-23:
- ASTM D2272: Rotating Pressure Vessel Oxidation Test (RPVOT) for steam turbine oils, which also evaluates oxidation stability.
- ASTM D664: Measurement of acid number in petroleum products by potentiometric titration, often used in conjunction with oxidation testing.
- ASTM D4057: Manual sampling method for petroleum and petroleum products.
- ASTM D1193: Specifications for reagent water used in laboratory testing.
- Engine Sequence Tests: Such as Sequence IIID, which provide comprehensive evaluations of oil performance in real engines.
By utilizing ASTM D4742-23, oil manufacturers, formulators, and quality control laboratories gain a robust, repeatable method for assessing the oxidation resistance of gasoline engine oils, supporting consistent product performance and efficient benchmarking in the lubricants market.
Buy Documents
ASTM D4742-23 - Standard Test Method for Oxidation Stability of Gasoline Automotive Engine Oils by Thin-Film Oxygen Uptake (TFOUT)
REDLINE ASTM D4742-23 - Standard Test Method for Oxidation Stability of Gasoline Automotive Engine Oils by Thin-Film Oxygen Uptake (TFOUT)
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D4742-23 is a standard published by ASTM International. Its full title is "Standard Test Method for Oxidation Stability of Gasoline Automotive Engine Oils by Thin-Film Oxygen Uptake (TFOUT)". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is used to evaluate oxidation stability of lubricating base oils with additives in the presence of chemistries similar to those found in gasoline engine service. Test results on some ASTM reference oils have been found to correlate with sequence IIID engine test results in hours for a 375 % viscosity increase.5 The test does not constitute a substitute for engine testing, which measures wear, oxidation stability, volatility, and deposit control characteristics of lubricants. Properly interpreted, the test may provide input on the oxidation stability of lubricants under simulated engine chemistry. 5.2 This test method is intended to be used as a bench screening test and quality control tool for lubricating base oil manufacturing, especially for re-refined lubricating base oils. This test method is useful for quality control of oxidation stability of re-refined oils from batch to batch. 5.3 This test method is useful for screening formulated oils prior to engine tests. Within similar additive chemistry and base oil types, the ranking of oils in this test appears to be predictive of ranking in engine tests. When oils having completely different additive chemistry or base oil type are compared, oxidation stability results may not reflect the actual engine test result. 5.4 Other oxidation stability test methods have demonstrated that soluble metal catalyst supplies are very inconsistent and they have significant effects on the test results. Thus, for test comparisons, the same source and same batch of metal naphthenates shall be used. Note 2: It is also recommended as a good research practice not to use different batches of the fuel component in test comparisons. SCOPE 1.1 This test method evaluates the oxidation stability of engine oils for gasoline automotive engines. This test, run at 160 °C, utilizes a high pressure reactor pressurized with oxygen along with a metal catalyst package, a fuel catalyst, and water in a partial simulation of the conditions to which an oil may be subjected in a gasoline combustion engine. This test method can be used for engine oils with viscosity in the range from 4 mm2/s (cSt) to 21 mm2/s (cSt) at 100 °C, including re-refined oils. 1.2 This test method is not a substitute for the engine testing of an engine oil in established engine tests, such as Sequence IIID. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Exception—Pressure units are provided in psig, and dimensions are provided in inches in Annex A1, because these are the industry accepted standard and the apparatus is built according to the figures shown. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 8. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is used to evaluate oxidation stability of lubricating base oils with additives in the presence of chemistries similar to those found in gasoline engine service. Test results on some ASTM reference oils have been found to correlate with sequence IIID engine test results in hours for a 375 % viscosity increase.5 The test does not constitute a substitute for engine testing, which measures wear, oxidation stability, volatility, and deposit control characteristics of lubricants. Properly interpreted, the test may provide input on the oxidation stability of lubricants under simulated engine chemistry. 5.2 This test method is intended to be used as a bench screening test and quality control tool for lubricating base oil manufacturing, especially for re-refined lubricating base oils. This test method is useful for quality control of oxidation stability of re-refined oils from batch to batch. 5.3 This test method is useful for screening formulated oils prior to engine tests. Within similar additive chemistry and base oil types, the ranking of oils in this test appears to be predictive of ranking in engine tests. When oils having completely different additive chemistry or base oil type are compared, oxidation stability results may not reflect the actual engine test result. 5.4 Other oxidation stability test methods have demonstrated that soluble metal catalyst supplies are very inconsistent and they have significant effects on the test results. Thus, for test comparisons, the same source and same batch of metal naphthenates shall be used. Note 2: It is also recommended as a good research practice not to use different batches of the fuel component in test comparisons. SCOPE 1.1 This test method evaluates the oxidation stability of engine oils for gasoline automotive engines. This test, run at 160 °C, utilizes a high pressure reactor pressurized with oxygen along with a metal catalyst package, a fuel catalyst, and water in a partial simulation of the conditions to which an oil may be subjected in a gasoline combustion engine. This test method can be used for engine oils with viscosity in the range from 4 mm2/s (cSt) to 21 mm2/s (cSt) at 100 °C, including re-refined oils. 1.2 This test method is not a substitute for the engine testing of an engine oil in established engine tests, such as Sequence IIID. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Exception—Pressure units are provided in psig, and dimensions are provided in inches in Annex A1, because these are the industry accepted standard and the apparatus is built according to the figures shown. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 8. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D4742-23 is classified under the following ICS (International Classification for Standards) categories: 75.100 - Lubricants, industrial oils and related products. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4742-23 has the following relationships with other standards: It is inter standard links to ASTM A314-23, ASTM A314-19, ASTM D664-11a(2017), ASTM A314-15, ASTM D2272-14, ASTM A314-13a, ASTM A314-13, ASTM E1-13, ASTM B211-12, ASTM B211-12e1, ASTM D4057-06(2011), ASTM D664-11, ASTM D2272-09, ASTM D664-09, ASTM A314-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4742-23 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4742 − 23
Standard Test Method for
Oxidation Stability of Gasoline Automotive Engine Oils by
Thin-Film Oxygen Uptake (TFOUT)
This standard is issued under the fixed designation D4742; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
2.1 ASTM Standards:
1.1 This test method evaluates the oxidation stability of
A314 Specification for Stainless Steel Billets and Bars for
engine oils for gasoline automotive engines. This test, run at
Forging
160 °C, utilizes a high pressure reactor pressurized with
B211 Specification for Aluminum and Aluminum-Alloy
oxygen along with a metal catalyst package, a fuel catalyst, and
Rolled or Cold-Finished Bar, Rod, and Wire (Metric)
water in a partial simulation of the conditions to which an oil
B0211_B0211M
may be subjected in a gasoline combustion engine. This test
D664 Test Method for Acid Number of Petroleum Products
method can be used for engine oils with viscosity in the range
by Potentiometric Titration
2 2
from 4 mm /s (cSt) to 21 mm /s (cSt) at 100 °C, including
D1193 Specification for Reagent Water
re-refined oils.
D2272 Test Method for Oxidation Stability of Steam Tur-
bine Oils by Rotating Pressure Vessel
1.2 This test method is not a substitute for the engine testing
D4057 Practice for Manual Sampling of Petroleum and
of an engine oil in established engine tests, such as Sequence
Petroleum Products
IIID.
E1 Specification for ASTM Liquid-in-Glass Thermometers
1.3 The values stated in SI units are to be regarded as
standard. No other units of measurement are included in this 3. Terminology
standard.
3.1 Definitions of Terms Specific to This Standard:
1.3.1 Exception—Pressure units are provided in psig, and
3.1.1 break point, n—the precise point of time at which
dimensions are provided in inches in Annex A1, because these
rapid oxidation of the oil begins.
are the industry accepted standard and the apparatus is built
3.1.2 oxidation induction time, n—the time until the oil
according to the figures shown.
begins to oxidize at a relatively rapid rate as indicated by the
decrease of oxygen pressure.
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the 3.1.3 oxygen uptake, n—oxygen absorbed by oil as a result
responsibility of the user of this standard to establish appro- of oil oxidation.
priate safety, health, and environmental practices and deter-
4. Summary of Test Method
mine the applicability of regulatory limitations prior to use.
For specific warning statements, see Sections 7 and 8.
4.1 The test oil is mixed in a glass container with three other
liquids that are used to simulate engine conditions: (1) an
1.5 This international standard was developed in accor-
oxidized/nitrated fuel component (Annex A2), (2) a mixture of
dance with internationally recognized principles on standard-
soluble metal naphthenates (lead, copper, iron, manganese, and
ization established in the Decision on Principles for the
stannous octoate (Annex A3)), and (3) Type I reagent water.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical 4.2 The glass container holding the oil mixture is placed in
Barriers to Trade (TBT) Committee.
a high pressure reactor equipped with a pressure gauge. The
high pressure reactor is sealed, charged with oxygen to a
pressure of 620 kPa (90 psig), and placed in an oil or dry
This test method is under the jurisdiction of ASTM Committee D02 on
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of
Subcommittee D02.09.0G on Oxidation Testing of Engine Oils. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved July 1, 2023. Published August 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1988. Last previous edition approved in 2017 as D4742 – 17. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D4742-23. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4742 − 23
FIG. 1 Schematic Drawing of Oxidation Test Apparatus
3,4
bath at 160 °C at an angle of 30° from the horizontal. The Test results on some ASTM reference oils have been found to
high pressure reactor is rotated axially at a speed of 100 r ⁄min correlate with sequence IIID engine test results in hours for a
forming a thin film of oil within the glass container resulting in 375 % viscosity increase. The test does not constitute a
a relatively large oil-oxygen contact area. substitute for engine testing, which measures wear, oxidation
stability, volatility, and deposit control characteristics of lubri-
NOTE 1—A pressure sensing device can be used in place of a pressure
cants. Properly interpreted, the test may provide input on the
gauge.
oxidation stability of lubricants under simulated engine chem-
4.3 The pressure of the high pressure reactor is recorded
istry.
continuously from the beginning of the test and the test is
5.2 This test method is intended to be used as a bench
terminated when a rapid decrease of the high pressure reactor
screening test and quality control tool for lubricating base oil
pressure is observed (Point B, Fig. A1.2). The period of time
manufacturing, especially for re-refined lubricating base oils.
that elapses between the time when the high pressure reactor is
This test method is useful for quality control of oxidation
placed in the oil or dry bath and the time at which the pressure
stability of re-refined oils from batch to batch.
begins to decrease rapidly is called the oxidation induction
time and is used as a measure of the relative oil oxidation
5.3 This test method is useful for screening formulated oils
stability.
prior to engine tests. Within similar additive chemistry and
base oil types, the ranking of oils in this test appears to be
5. Significance and Use
predictive of ranking in engine tests. When oils having
5.1 This test method is used to evaluate oxidation stability
completely different additive chemistry or base oil type are
of lubricating base oils with additives in the presence of
compared, oxidation stability results may not reflect the actual
chemistries similar to those found in gasoline engine service.
engine test result.
5.4 Other oxidation stability test methods have demon-
The sole source of supply of the catalyst and dry bath known to the committee strated that soluble metal catalyst supplies are very inconsistent
at this time is Tannas Co., 4800 James Savage Rd., Midland, MI 48642.
If you are aware of alternative suppliers, please provide this information to
ASTM International Headquarters. Your comments will receive careful consider- Ku, C. S. and Hsu, S. M., “A Thin Film Uptake Test for the Evaluation of
ation at a meeting of the responsible technical committee, which you may attend. Automotive Lubricants,” Lubrication Engineering, 40, 2, 1984, pp. 75–83.
D4742 − 23
FIG. 2 Pressure versus Time Diagram of the Oxidation Test
and they have significant effects on the test results. Thus, for
test comparisons, the same source and same batch of metal
naphthenates shall be used.
NOTE 2—It is also recommended as a good research practice not to use
different batches of the fuel component in test comparisons.
6. Apparatus
6.1 High Pressure Reactor, glass sample container, alumi-
num insert, pressure gauge, thermometer, test bath and acces-
sories are shown in Fig. 2 and Fig. 3, and described in Annex
A1.
NOTE 3—It is reported in literature that the oxidation high pressure
reactor can be modified from the Test Method D2272 oxidation high
pressure reactor by insertion of an aluminum cylinder.
6.2 Precision Pressure Gauge—A certified precision pres-
sure gauge is used to accurately control the oxygen feed to the
FIG. 3 Schematic Drawing of an Assembled Vessel, Aluminum
high pressure reactor. The gauge has a sufficient range to Insert, and Glass Sample Container
encompass 0 kPa to 650 kPa or more (;90 psig) required by
the test method with division 2.0 kPa (;0.5 psig) or better to
7.4 Hexane, C H , Practical Grade or other suitable hydro-
enable readings to be made to 2.0 kPa (;0.25 psig).
6 14
carbon solvent. (Warning—Extremely flammable. Harmful if
inhaled. May produce nerve cell damage. Skin irritant on
7. Reagents
repeated contact. Aspiration hazard.)
7.1 Purity of Reagents—Reagent grade chemicals shall be
7.5 Isopropyl Alcohol, CH CH(CH )OH. (Warning—
used in all tests. Unless otherwise indicated, it is intended that
3 3
Flammable. Eye irritant. Vapors narcotic.)
all reagents shall conform to the specifications of the Commit-
tee on Analytical Reagents of the American Chemical Society.
7.6 Oxygen. (Warning—Oxygen vigorously accelerates
combustion.)
7.2 Purity of Water—Unless otherwise indicated, references
to water shall be understood to mean distilled water meeting
7.7 Potassium Hydroxide, Alcohol Solution (1.5 %)—
requirements of reagent water as defined by Type I of Speci-
Dissolve 12 g of potassium hydroxide (KOH) pellets in 1 L of
fication D1193.
99 % isopropyl alcohol. (Warning—Potassium hydroxide is
corrosive.)
7.3 Acetone, CH COCH . (Warning—Extremely flam-
3 3
mable. Irritating to skin, eyes, and mucous membranes.)
8. Materials
8.1 Fuel Component—The fuel component is intended to
simulate an oxidized nitrated gasoline fraction. This compo-
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
nent may be prepared in accordance with the procedures
DC. For suggestions on the testing of reagents not listed by the American Chemical
described in Annex A2. (See 8.2 and Note 4.) (Warning—Fuel
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
component is extremely flammable. Vapors harmful if inhaled.
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. Skin irritant on repeated contact. Aspiration hazard.)
D4742 − 23
3,4
8.2 Soluble Metal Catalyst Mixture—This catalyst is a 10.1.1 Weigh the clean glass sample container to the nearest
mixture of soluble metal catalysts (lead, copper, iron, mg.
manganese, and tin). The catalyst may be prepared according
10.1.2 Weigh 1.500 g 6 0.001 g of oil sample into the
to the procedures described in Annex A3. (See 8.2 and Note 4.) container.
(Warning—Lead and manganese naphthenates are known to
10.1.3 Add 0.050 g 6 0.001 g each of both the fuel com-
be poisonous, but all naphthenates should be handled with care. ponent and the soluble metal catalyst mixture.
Naphthenates are also flammable.)
10.1.4 Add 0.030 g 6 0.001 g of distilled water (Type I) to
the glass sample container.
NOTE 4—With some catalyst chemicals, reactivity can be a problem.
10.1.5 Just prior to inserting the glass sample container into
This can be curtailed by storing in a refrigerator at approximately 5 °C.
The catalyst chemicals remain effective up to six months after the septum the high pressure reactor, thoroughly mix the catalyst compo-
is punctured if they are stored as noted above.
nents within the sample container by hand-rotation (approxi-
NOTE 5—It is recommended that each new batch of fuel or metal
mately five rotations) and proceed immediately to 10.2. Delay
catalyst be correlated with a standard reference oil, in accordance with
may result in variation of results.
good laboratory practices.
10.2 High Pressure Reactor Charging and Assembly—
8.3 Liquid Detergent (Industrial Strength).
Immediately and rapidly assemble the high pressure reactor as
8.4 Silicone Stopcock Grease.
shown in Fig. 1 and Fig. 2. Refer to manufacturer’s manual for
details.
9. Preparation of Apparatus
10.2.1 Put the aluminum insert into the high pressure
9.1 Glass Sample Container—Rinse the glass sample con-
reactor followed by the glass sample container and the TFE
tainer with hexane or other suitable hydrocarbon solvent. Clean
(tetra-fluoroethylene) plastic cover disk.
the glass container with hot detergent solution and water. Rinse
10.2.2 Place the stainless hold-down spring on top of the
the container with acetone. Blow the container dry with clean
TFE disk and the glass sample container.
compressed air.
NOTE 9—The stainless steel hold-down spring not only holds down the
NOTE 6—A clean glass sample container is important for obtaining
TFE cover disk but, more importantly, prevents the glass sample container
repeatable results. Thorough cleaning of the glass sample can be accom-
from slipping inside the high pressure reactor with consequent poor
plished by (1) hexane rinse, (2) acetone soak for 15 min, (3) hot detergent
results.
solution and water soak for 4 h, (4) acetone rinse, and (5) hexane rinse. A
clean glass sample container is important for obtaining repeatable results. 10.2.3 Apply a thin coating of silicone stopcock grease to
NOTE 7—A segmented glass sample container has been found suitable
the O-ring high pressure reactor seal located in the gasket
to prevent premature mixing of the catalyst components.
groove of the high pressure reactor lid to provide lubrication.
9.2 Cleaning of High Pressure Reactor—Wash the inside of
10.2.4 Insert the lid into the high pressure reactor.
the high pressure reactor, aluminum insert, lid, and inside with
10.2.5 Place and tighten the cap on the high pressure reactor
hot detergent solution and water. Rinse the inside of the stem
to lock down the lid in the high pressure reactor.
with isopropyl alcohol and blow dry with clean compressed air.
10.2.6 Attach the high pressure oxygen hose and purge the
If the high pressure reactor, insert, lid, or inside of the stem
high pressure reactor twice at about 620 kPa (90 psig) of
emits acidic odor after simple cleaning, wash with a solution of
oxygen to remove the air originally present in the high pressure
alcoholic potassium hydroxide (KOH) of 1.5 %, and repeat the
reactor.
cleaning procedures. For dry baths, refer to manufacturer’s
NOTE 10—It is advisable to avoid releasing the oxygen too rapidly by
manuals for details.
decreasing the pressure to atmospheric in no less than one minute to avoid
9.3 Cleaning of High Pressure Reactor Stem—It is recom- possible foaming and overflow of the sample from the glass sample
container.
mended to periodically disassemble, inspect, and clean the high
pressure reactor stem. Rinse the inside of the stem with
10.2.7 Charge the high pressure reactor to 620 kPa (90 psig)
isopropyl alcohol and blow dry with clean compressed air.
oxygen at 21 °C using an in-line precision pressure gauge to
After cleansing, it is recommended to insert a dry pipe cleaner
monitor the high pressure reactor pressure with 1 kPa
into the transducer line opening for removal of potential
(0.12 psig). (For ambient temperatures other than 21 °C, in-
residue buildup.
crease (or decrease) the initial pressure by 2.5 kPa (0.4 psig)
for each 1 °C above (or below) 21 °C.)
NOTE 8—It is recommended to replace the O-rings when reassembling
10.2.8 Fill the high pressure reactor to the required pressure
the pressure transducers.
and close the inlet valve. Test the high pressure reactor for
9.4 To obtain a representative sample of stocks of uniform
leaks by immersing in water or by using soap solution.
petroleum products, follow Practice D4057.
10.3 Oxidation—Before starting the test, bring the heating
9.5 Cleaning of Catalyst Syringes—Individual catalyst sy-
bath to the test temperature at 160 °C while the stirrer is in
ringes are to be used for each catalyst component. They are to
operation. (This can be done during preparation of the sample
be thoroughly cleaned and dried prior to each use. (See Annex
and the high pressure reactor.)
A4 for recommended procedure.)
10.3.1 Switch off the stirrer and insert the high pressure
10. Procedure
reactor into the carriages. Note the time, and restart the stirrer.
10.1 Weighing and Mixing Sample and Catalyst Compo-
NOTE 11—The time for the bath to reach the operating temperature after
nents: insertion of the high pressure reactor may differ for different apparatus
D4742 − 23
assemblies and should be observed for each unit (a unit may carry one,
12. Precision and Bias
two, three, or four high pressure reactors). The objective is to find a set of
12.1 The precision of this test method, as determined by
conditions which does not permit a drop of more than 2 °C after insertion
statistical examination of interlaboratory results on break point
of the high pressure reactor(s) and allows the high pressure reactor
pressure to reach plateau within 15 min.
time, is as follows:
12.1.1 Repeatability—The difference between successive
10.3.2 If an auxiliary heater is used, keep it on for the first
results, obtained by the same operator with the same apparatus
5 min of the run and then turn it off (Note 11).
under constant operating conditions on identical test material,
10.3.3 Allow the bath temperature to level out at the test
would in the long run, in the normal and co
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4742 − 17 D4742 − 23
Standard Test Method for
Oxidation Stability of Gasoline Automotive Engine Oils by
Thin-Film Oxygen Uptake (TFOUT)
This standard is issued under the fixed designation D4742; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method evaluates the oxidation stability of engine oils for gasoline automotive engines. This test, run at 160 °C,
utilizes a high pressure reactor pressurized with oxygen along with a metal catalyst package, a fuel catalyst, and water in a partial
simulation of the conditions to which an oil may be subjected in a gasoline combustion engine. This test method can be used for
2 2
engine oils with viscosity in the range from 4 mm /s (cSt) to 21 mm /s (cSt) at 100 °C, including re-refined oils.
1.2 This test method is not a substitute for the engine testing of an engine oil in established engine tests, such as Sequence IIID.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Exception—Pressure units are provided in psig, and dimensions are provided in inches in Annex A1, because these are the
industry accepted standard and the apparatus is built according to the figures shown.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and healthsafety, health, and environmental practices and determine
the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 8.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
A314 Specification for Stainless Steel Billets and Bars for Forging
B211 Specification for Aluminum and Aluminum-Alloy Rolled or Cold-Finished Bar, Rod, and Wire (Metric) B0211_B0211M
D664 Test Method for Acid Number of Petroleum Products by Potentiometric Titration
D1193 Specification for Reagent Water
D2272 Test Method for Oxidation Stability of Steam Turbine Oils by Rotating Pressure Vessel
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
E1 Specification for ASTM Liquid-in-Glass Thermometers
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.09.0G on Oxidation Testing of Engine Oils.
Current edition approved July 1, 2017July 1, 2023. Published July 2017August 2023. Originally approved in 1988. Last previous edition approved in 20162017 as
D4742 – 16.D4742 – 17. DOI: 10.1520/D4742-17.10.1520/D4742-23.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4742 − 23
3. Terminology
3.1 Definitions of Terms Specific to This Standard:
3.1.1 break point—point, n—the precise point of time at which rapid oxidation of the oil begins.
3.1.2 oxidation induction time—time, n—the time until the oil begins to oxidize at a relatively rapid rate as indicated by the
decrease of oxygen pressure.
3.1.3 oxygen uptake—uptake, n—oxygen absorbed by oil as a result of oil oxidation.
4. Summary of Test Method
4.1 The test oil is mixed in a glass container with three other liquids that are used to simulate engine conditions: (1) an
oxidized/nitrated fuel component (Annex A2), (2) a mixture of soluble metal naphthenates (lead, copper, iron, manganese, and tin
naphthenatesstannous octoate (Annex A3)), and (3) Type I reagent water.
4.2 The glass container holding the oil mixture is placed in a high pressure reactor equipped with a pressure gauge. The high
3,4
pressure reactor is sealed, charged with oxygen to a pressure of 620 kPa (90 psig), and placed in an oil or dry bath at 160 °C
at an angle of 30° from the horizontal. The high pressure reactor is rotated axially at a speed of 100 r ⁄min forming a thin film of
oil within the glass container resulting in a relatively large oil-oxygen contact area.
NOTE 1—A pressure sensing device can be used in place of a pressure gauge.
4.3 The pressure of the high pressure reactor is recorded continuously from the beginning of the test and the test is terminated
when a rapid decrease of the high pressure reactor pressure is observed (Point B, Fig. A1.2). The period of time that elapses
between the time when the high pressure reactor is placed in the oil or dry bath and the time at which the pressure begins to
decrease rapidly is called the oxidation induction time and is used as a measure of the relative oil oxidation stability.
5. Significance and Use
5.1 This test method is used to evaluate oxidation stability of lubricating base oils with additives in the presence of chemistries
similar to those found in gasoline engine service. Test results on some ASTM reference oils have been found to correlate with
sequence IIID engine test results in hours for a 375 % viscosity increase. The test does not constitute a substitute for engine
testing, which measures wear, oxidation stability, volatility, and deposit control characteristics of lubricants. Properly interpreted,
the test may provide input on the oxidation stability of lubricants under simulated engine chemistry.
5.2 This test method is intended to be used as a bench screening test and quality control tool for lubricating base oil manufacturing,
especially for re-refined lubricating base oils. This test method is useful for quality control of oxidation stability of re-refined oils
from batch to batch.
5.3 This test method is useful for screening formulated oils prior to engine tests. Within similar additive chemistry and base oil
types, the ranking of oils in this test appears to be predictive of ranking in engine tests. When oils having completely different
additive chemistry or base oil type are compared, oxidation stability results may not reflect the actual engine test result.
5.4 Other oxidation stability test methods have demonstrated that soluble metal catalyst supplies are very inconsistent and they
have significant effects on the test results. Thus, for test comparisons, the same source and same batch of metal naphthenates shall
be used.
NOTE 2—It is also recommended as a good research practice not to use different batches of the fuel component in test comparisons.
The sole source of supply of the catalyst and dry bath known to the committee at this time is Tannas Co., 4800 James Savage Rd., Midland, MI 48642.
If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration at a
meeting of the responsible technical committee, which you may attend.
Ku, C. S. and Hsu, S. M., “A Thin Film Uptake Test for the Evaluation of Automotive Lubricants,” Lubrication Engineering, 40, 2, 1984, pp. 75–83.
D4742 − 23
FIG. 1 Schematic Drawing of Oxidation Test Apparatus
6. Apparatus
6.1 High Pressure Reactor, glass sample container, aluminum insert, pressure gauge, thermometer, test bath and accessories are
shown in Fig. 2 and Fig. 3, and described in Annex A1.
NOTE 3—It is reported in literature that the oxidation high pressure reactor can be modified from the Test Method D2272 oxidation high pressure reactor
by insertion of an aluminum cylinder.
6.2 Precision Pressure Gauge—A certified precision pressure gauge is used to accurately control the oxygen feed to the high
pressure reactor. The gauge has a sufficient range to encompass 0 kPa to 650 kPa or more (;90 psig) required by the test method
with division 2.0 kPa (;0.5 psig) or better to enable readings to be made to 2.0 kPa (;0.25 psig).
7. Reagents
7.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society.
7.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean distilled water meeting
requirements of reagent water as defined by Type I of Specification D1193.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D4742 − 23
FIG. 2 Pressure versus Time Diagram of the Oxidation Test
FIG. 3 Schematic Drawing of an Assembled Vessel, Aluminum Insert, and Glass Sample Container
7.3 Acetone, CH COCH . (Warning—Extremely flammable. Irritating to skin, eyes, and mucous membranes.)
3 3
7.4 Air, containing 2000 ppm nitrogen dioxide, NO (commercially available compressed gas mixture, certified within 65 %).
(Warning—Nitrogen dioxide is poisonous.)
D4742 − 23
7.4 Hexane, C H , Practical Grade or other suitable hydrocarbon solvent. (Warning—Extremely flammable. Harmful if inhaled.
6 14
May produce nerve cell damage. Skin irritant on repeated contact. Aspiration hazard.)
7.5 Isopropyl Alcohol, CH CH(CH )OH. (Warning—Flammable. Eye irritant. Vapors narcotic.)
3 3
7.6 Oxygen. (Warning—Oxygen vigorously accelerates combustion.)
7.7 Potassium Hydroxide, Alcohol Solution (1.5 %)—Dissolve 12 g of potassium hydroxide (KOH) pellets in 1 L of 99 %
isopropyl alcohol. (Warning—Potassium hydroxide is corrosive.)
8. Materials
8.1 Fuel Component—The fuel component is intended to simulate an oxidized nitrated gasoline fraction. This component may be
prepared in accordance with the procedures described in Annex A2. (See 8.2 and Note 4.) (Warning—Fuel component is
extremely flammable. Vapors harmful if inhaled. Skin irritant on repeated contact. Aspiration hazard.)
3,4
8.2 Soluble Metal Catalyst Mixture—This catalyst is a mixture of soluble metal catalysts (lead, copper, iron, manganese, and
tin). The catalyst may be prepared according to the procedures described in Annex A3. (See 8.2 and Note 4.) (Warning—Lead
and manganese naphthenates are known to be poisonous, but all naphthenates should be handled with care. Naphthenates are also
flammable.)
NOTE 4—With some catalyst chemicals, reactivity can be a problem. This can be curtailed by storing in a refrigerator at approximately 5 °C. The catalyst
chemicals remain effective up to six months after the septum is punctured if they are stored as noted above.
NOTE 5—It is recommended that each new batch of fuel or metal catalyst be correlated with a standard reference oil, in accordance with good laboratory
practices.
8.3 Liquid Detergent (Industrial Strength).
8.4 Silicone Stopcock Grease.
9. Preparation of Apparatus
9.1 Glass Sample Container—Rinse the glass sample container with hexane or other suitable hydrocarbon solvent. Clean the glass
container with hot detergent solution and water. Rinse the container with acetone. Blow the container dry with clean compressed
air.
NOTE 6—A clean glass sample container is important for obtaining repeatable results. Thorough cleaning of the glass sample can be accomplished by (1)
hexane rinse, (2) acetone soak for 15 min, (3) hot detergent solution and water soak for 4 h, (4) acetone rinse, and (5) hexane rinse. A clean glass sample
container is important for obtaining repeatable results.
NOTE 7—A segmented glass sample container has been found suitable to prevent premature mixing of the catalyst components.
9.2 Cleaning of High Pressure Reactor—Wash the inside of the high pressure reactor, aluminum insert, lid, and inside with hot
detergent solution and water. Rinse the inside of the stem with isopropyl alcohol and blow dry with clean compressed air. If the
high pressure reactor, insert, lid, or inside of the stem emits acidic odor after simple cleaning, wash with a solution of alcoholic
potassium hydroxide (KOH) of 1.5 %, and repeat the cleaning procedures. For dry baths, refer to manufacturer’s manuals for
details.
9.3 Cleaning of High Pressure Reactor Stem—It is recommended to periodically disassemble, inspect, and clean the high pressure
reactor stem. Rinse the inside of the stem with isopropyl alcohol and blow dry with clean compressed air. After cleansing, it is
recommended to insert a dry pipe cleaner into the transducer line opening for removal of potential residue buildup.
NOTE 8—It is recommended to replace the O-rings when reassembling the pressure transducers.
D4742 − 23
9.4 To obtain a representative sample of stocks of uniform petroleum products, follow Practice D4057.
9.5 Cleaning of Catalyst Syringes—Individual catalyst syringes are to be used for each catalyst component. They are to be
thoroughly cleaned and dried prior to each use. (See Annex A4 for recommended procedure.)
10. Procedure
10.1 Weighing and Mixing Sample and Catalyst Components:
10.1.1 Weigh the clean glass sample container to the nearest mg.
10.1.2 Weigh 1.500 g 6 0.001 g of oil sample into the container.
10.1.3 Add 0.060 g0.050 g 6 0.001 g each of both the fuel component and the soluble metal catalyst mixture.
10.1.4 Add 0.030 g 6 0.001 g of distilled water (Type I) to the glass sample container.
10.1.5 Just prior to inserting the glass sample container into the high pressure reactor, thoroughly mix the catalyst components
within the sample container by hand-rotation (approximately five rotations) and proceed immediately to 10.2. Delay may result
in variation of results.
10.2 High Pressure Reactor Charging and Assembly—Immediately and rapidly assemble the high pressure reactor as shown in
Fig. 1 and Fig. 2. Refer to manufacturer’s manual for details.
10.2.1 Put the aluminum insert into the high pressure reactor followed by the glass sample container and the TFE
(tetra-fluoroethylene) plastic cover disk.
10.2.2 Place the stainless hold-down spring on top of the TFE disk and the glass sample container.
NOTE 9—The stainless steel hold-down spring not only holds down the TFE cover disk but, more importantly, prevents the glass sample container from
slipping inside the high pressure reactor with consequent poor results.
10.2.3 Apply a thin coating of silicone stopcock grease to the O-ring high pressure reactor seal located in the gasket groove of
the high pressure reactor lid to provide lubrication.
10.2.4 Insert the lid into the high pressure reactor.
10.2.5 Place and tighten the cap on the high pressure reactor to lock down the lid in the high pressure reactor.
10.2.6 Attach the high pressure oxygen hose and purge the high pressure reactor twice at about 620 kPa (90 psig) of oxygen to
remove the air originally present in the high pressure reactor.
NOTE 10—It is advisable to avoid releasing the oxygen too rapidly by decreasing the pressure to atmospheric in no less than one minute to avoid possible
foaming and overflow of the sample from the glass sample container.
10.2.7 Charge the high pressure reactor to 620 kPa (90 psig) oxygen at 21 °C using an in-line precision pressure gauge to monitor
the high pressure reactor pressure with 1 kPa (0.12 psig). (For ambient temperatures other than 21 °C, increase (or decrease) the
initial pressure by 2.5 kPa (0.4 psig) for each 1 °C above (or below) 21 °C.)
10.2.8 Fill the high pressure reactor to the required pressure and close the inlet valve. Test the high pressure reactor for leaks by
immersing in water or by using soap solution.
10.3 Oxidation—Before starting the test, bring the heating bath to the test temperature at 160 °C while the stirrer is in operation.
(This can be done during preparation of the sample and the high pressure reactor.)
D4742 − 23
10.3.1 Switch off the stirrer and insert the high pressure reactor into the carriages. Note the time, and restart the stirrer.
NOTE 11—The time for the bath to reach the operating temperature after insertion of the high pressure reactor may differ for different apparatus assemblies
and should be observed for each unit (a unit may carry one, two, three, or four high pressure reactors). The objective is to find a set of conditions which
does not permit a drop of more than 2 °C after insertion of the high pressure reactor(s) and allows the high pressure reactor pressure to reach plateau within
15 min.
10.3.2 If an auxiliary heater is used, keep it on for the first 5 min of the run and then turn it off (Note 11).
10.3.3 Allow the bath temperature to level out at the test temperature which must occur within 15 min after insertion of the high
pressure vessel. Maintaining the test temperature within the specified limits of 160 °C 6 0.3 °C during the entire test run is the
most important single factor assuring both repeatability and reproducibility of test results. If the test temperature cannot be
maintained as specified, the test results should not be considered valid.
10.4 Keep the high pressure reactor completely submerged and maintain continuous and uniform rotation throughout the test. A
standard rotational speed of 100 r ⁄min 6 5 r ⁄min is required; any appreciable variations in this speed could cause erratic results.
10.5 Monitor the pressure of the high pressure reactor. If a dial pressure gauge is used, make readings at least every 5 min. (The
maximum pressure must be reached within 15 min.) After a test period (the induction time), the pressure decreases because of
oxygen absorption by oil (the break point).
10.5.1 When the oil reaches the break point, the pressure decreases rapidly. The test can be terminated as soon as sufficient
information has been collected to form a tangent to the decreasing pressure trace (see 10.6) or, if desired, continued until pressure
decreases to some further level.
NOTE 12—The pressure within the high pressure reactor increases at the beginning because of gas expansion accompanying the temperature increase of
the high pressure reactor. Following this rise, the pressure reaches a plateau as shown in Fig. A1.2. This pressure may gradually drop slightly during the
test. A gradual decrease of the pressure is not unusual and does not invalidate the test.
NOTE 13—If a break in pressure does not occur within 300 min to 500 min, the operator may elect to terminate the test. A slow decrease in pressure may
result from a leak in the reactor or a very gradual oxidation process.
10.6 Record the time at
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...