ASTM E649-17a
(Test Method)Standard Test Method for Bromine in Chlorine
Standard Test Method for Bromine in Chlorine
SIGNIFICANCE AND USE
4.1 Low levels of bromine contaminant in chlorine cause problems in some industrial uses. This test method may be used to determine bromine in liquid or gaseous chlorine at levels as low as 4 ug/kg.
SCOPE
1.1 This test method covers the determination of bromine in liquid chlorine and in gaseous chlorine with a lower limit of detection of 4 ug/kg by weight.
1.2 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures, handling, and safety precautions.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 7.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2017
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.13 - Chlorine
Relations
- Effective Date
- 01-Jul-2017
- Effective Date
- 01-Nov-2016
- Effective Date
- 01-Jun-2007
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM E649-17a: Standard Test Method for Bromine in Chlorine establishes a standardized test method for determining trace levels of bromine in both liquid and gaseous chlorine. Developed by ASTM International, this standard offers analytical laboratories, chemical producers, and industries reliant on chlorine a means to reliably detect bromine contamination as low as 4 micrograms per kilogram (µg/kg) by weight. The precise detection of bromine in chlorine is crucial because even low concentrations may negatively impact various industrial processes, affecting product quality or causing operational issues.
Key Topics
- Trace Bromine Detection: Specifies procedures for analyzing very low bromine concentrations in liquid and gaseous chlorine, ensuring reliable detection to the lower threshold of 4 µg/kg.
- Safety and Handling: Emphasizes the importance of reviewing Safety Data Sheets (SDS) before using this method, addressing toxicity, first-aid procedures, handling, and necessary safety precautions, particularly for chlorine and corrosive reagents.
- Sampling Techniques: Details practices for properly sampling both liquid and gaseous chlorine, utilizing specialized apparatus to avoid contamination and ensure user safety.
- Analytical Procedure: Describes stepwise procedures involving absorption of chlorine into sodium hydroxide, chemical reduction and oxidation, and quantitative titration using sodium thiosulfate.
- Quality Control: Outlines expectations for laboratory quality control, encouraging use of quality control samples and adherence to established QA/QC protocols to ensure accuracy and repeatability.
Applications
The ASTM E649-17a test method is suited for:
- Chemical Manufacturing: Chlorine producers and processors use this standard to verify product specification and reduce the risk of undesirable chemical reactions due to bromine impurities.
- Industrial Quality Control: Industrial users of chlorine-such as in water treatment, plastics manufacturing, or pharmaceuticals-apply this test method to safeguard sensitive processes where bromine contamination could degrade end-product quality.
- Laboratory Testing: Analytical and environmental laboratories employ this standard for regulatory compliance, certification, and monitoring of chlorine purity.
- Supply Chain Verification: Ensures that chlorine supplied between manufacturers and end-users meets stringent impurity thresholds, promoting consistent quality in downstream applications.
Using this standard helps reduce equipment corrosion, unintended side reactions, and safety hazards, thereby supporting optimal performance for chlorine-dependent operations.
Related Standards
ASTM E649-17a references and aligns with several other ASTM standards and international guidelines:
- ASTM D1193 - Specification for Reagent Water: Ensures purity of water used in analytical procedures.
- ASTM D6809 - Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials: Provides QA/QC framework for chemical analysis.
- ASTM E200 - Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis: Guidance on managing chemicals used within the procedure.
- International Standardization Principles: Developed in accordance with WTO Technical Barriers to Trade (TBT) principles to support international harmonization.
Summary
By following ASTM E649-17a, organizations can accurately determine low levels of bromine in chlorine, enhancing product quality, regulatory compliance, and overall safety. The standard's rigorous safety, sampling, and analytical protocols, paired with recommendations for quality assurance, make it an essential resource for industries where chlorine purity directly influences operational success.
Keywords: ASTM E649, bromine in chlorine, trace bromine analysis, chlorine purity, chemical analysis, industrial quality control, sampling procedures, reagent water, standard test method.
Buy Documents
ASTM E649-17a - Standard Test Method for Bromine in Chlorine
REDLINE ASTM E649-17a - Standard Test Method for Bromine in Chlorine
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E649-17a is a standard published by ASTM International. Its full title is "Standard Test Method for Bromine in Chlorine". This standard covers: SIGNIFICANCE AND USE 4.1 Low levels of bromine contaminant in chlorine cause problems in some industrial uses. This test method may be used to determine bromine in liquid or gaseous chlorine at levels as low as 4 ug/kg. SCOPE 1.1 This test method covers the determination of bromine in liquid chlorine and in gaseous chlorine with a lower limit of detection of 4 ug/kg by weight. 1.2 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures, handling, and safety precautions. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 7. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Low levels of bromine contaminant in chlorine cause problems in some industrial uses. This test method may be used to determine bromine in liquid or gaseous chlorine at levels as low as 4 ug/kg. SCOPE 1.1 This test method covers the determination of bromine in liquid chlorine and in gaseous chlorine with a lower limit of detection of 4 ug/kg by weight. 1.2 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures, handling, and safety precautions. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use. Specific hazard statements are given in Section 7. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E649-17a is classified under the following ICS (International Classification for Standards) categories: 71.060.10 - Chemical elements. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E649-17a has the following relationships with other standards: It is inter standard links to ASTM E649-17, ASTM D6809-02(2016), ASTM D6809-02(2007), ASTM D1193-06, ASTM D6809-02, ASTM D1193-99e1, ASTM D1193-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E649-17a is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E649 − 17a
Standard Test Method for
Bromine in Chlorine
This standard is issued under the fixed designation E649; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 3. Summary of Test Method
1.1 This test method covers the determination of bromine in 3.1 Chlorine gas is sampled by absorption in aqueous
liquid chlorine and in gaseous chlorine with a lower limit of sodium hydroxide (NaOH). Liquid chlorine is first vaporized,
detection of 4 ug/kg by weight. and the vapor is absorbed in aqueous sodium hydroxide. An
aliquot of the sample solution is reduced with an excess of
1.2 Review the current Safety Data Sheets (SDS) for de-
sulfite ion, acidified, and excess sulfur dioxide (SO ) boiled
tailed information concerning toxicity, first-aid procedures,
out. In a carefully buffered solution, bromide is oxidized to
handling, and safety precautions.
bromate by hypochlorite. Excess hypochlorite is reduced to
1.3 This standard does not purport to address all of the
chloride by formate. In the presence of molybdate catalyst,
safety concerns, if any, associated with its use. It is the
bromate is reduced to bromide by iodide, and the liberated
responsibility of the user of this standard to establish appro-
iodine is titrated with standard sodium thiosulfate solution.
priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
4. Significance and Use
Specific hazard statements are given in Section 7.
4.1 Low levels of bromine contaminant in chlorine cause
1.4 This international standard was developed in accor-
problems in some industrial uses. This test method may be
dance with internationally recognized principles on standard-
used to determine bromine in liquid or gaseous chlorine at
ization established in the Decision on Principles for the
levels as low as 4 ug/kg.
Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
5. Apparatus
Barriers to Trade (TBT) Committee.
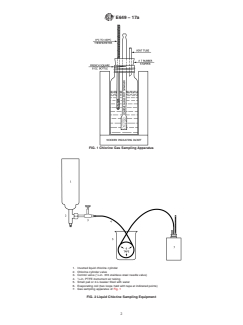
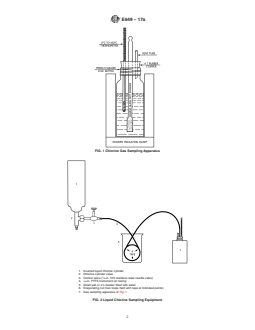
5.1 The construction of the chlorine gas sampling apparatus
and of the assembled sampling equipment is shown in Figs. 1
2. Referenced Documents
and 2. Modification of the equipment to deal with special
2.1 ASTM Standards:
sampling circumstances may be necessary. In Fig. 2, the
D1193 Specification for Reagent Water
control valve is shown with an adaptor for connection to a
D6809 Guide for Quality Control and Quality Assurance
chlorine cylinder valve. Other adaptors will be required when
Procedures for Aromatic Hydrocarbons and Related Ma-
sampling liquid chlorine in liquefaction plant streams or from
terials
large shipping or storage containers.
E180 Practice for Determining the Precision of ASTM
5.2 A 10-mL buret calibrated in 0.05-mL divisions is used
Methods for Analysis and Testing of Industrial and Spe-
when titrating with 0.01 N standard sodium thiosulfate solu-
cialty Chemicals (Withdrawn 2009)
tion.
E200 Practice for Preparation, Standardization, and Storage
of Standard and Reagent Solutions for ChemicalAnalysis
6. Reagents
6.1 Purity of Reagents—Unless otherwise indicated, it is
intended that all reagents should conform to the specifications
This test method is under the jurisdiction of ASTM Committee D16 on
of the Committee on Analytical Reagents of the American
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi-
Chemical Society, where such specifications are available.
bility of Subcommittee D16.13 on Chlorine.
Other grades may be used, provided it is first ascertained that
Current edition approved July 1, 2017. Published July 2017. Originally approved
in 1978. Last previous edition approved in 2017 as E649 – 17. DOI: 10.1520/
E0649-17a.
2 4
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Reagent Chemicals, American Chemical Society Specifications, American
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Chemical Society, Washington, DC. For suggestions on the testing of reagents not
Standards volume information, refer to the standard’s Document Summary page on listed by the American Chemical Society, see Analar Standards for Laboratory
the ASTM website. Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
The last approved version of this historical standard is referenced on and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
www.astm.org. MD.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E649 − 17a
FIG. 1 Chlorine Gas Sampling Apparatus
1. Inverted liquid chlorine cylinder
2. Chlorine cylinder valve
3. Control valve ( ⁄4-in. 316 stainless steel needle valve)
4. ⁄4-in. PTFE instrument air tubing
5. Small pail or 2-L beaker filled with water
6. Evaporating coil (two loops held with tape at indicated points)
7. Gas sampling apparatus of Fig. 1
FIG. 2 Liquid Chlorine Sampling Equipment
E649 − 17a
the reagent is of sufficiently high purity to permit its use ately flush with water for at least 15 min; then get prompt
without lessening the accuracy of the determination. medical attention. Goggles should be worn when handling
these chemicals.
6.2 Purity of Water—Unless otherwise indicated, references
to water shall be understood to mean Type II or Type III 7.2 Chlorine sampling should be performed only by persons
reagent water conforming to Specification D1193. thoroughly familiar with handling this material and with
operation of the sampling system. Personnel should be
6.3 Chloride Solution—Dissolve 200 g of sodium chloride
equipped with a respirator, goggles, faceshield, and gloves. If
(NaCl) in 940 mL of water.
possible, perform sampling in a hood or other well-ventilated
6.4 Formate Solution—Dissolve 50 g of sodium formate in
area. In sampling liquid chlorine, do not leave the space
175 mL of water.
between two closed valves filled with liquid chlorine (see 8.6).
6.5 Hypochlorite Solution—Dissolve 6.2 g of NaOH in 190
7.3 When sampling and working with chlorine out of doors,
mLof water and chlorinate slowly, with stirring, to give a gain
people downwind from such operation should be warned of the
in weight of 5.0 g.
possible release of chlorine vapors.
NOTE 1—If desired, a commercial hypochlorite bleach solution may be
7.4 It is recommended that means be available for disposal
used as the hypochlorite solution. Although such commercial bleach
of excess chlorine in an environmentally safe and acceptable
solutions are not made from reagent grade chemicals, the reagent blank
manner. If chlorine cannot be disposed of in a chlorine-
determination (see 9.4) corrects for the bromine and any chlorate content
consuming process, a chlorine absorption system should be
in such solutions. A proprietary 5.25 % sodium hypochlorite bleach
solution is satisfactory for this use. Because the sodium chloride and provided.When the analysis and sampling regimen requires an
hypochlorite solutions contribute most to the reagent blanks, dispense
initialpurgingofchlorinefromacontainer,thepurgedchlorine
these solutions by pipet to ensure uniformity of blanks and determinations
should be similarly handled. Purging to the atmosphere should
(see 9.1 and 9.2).
be avoided.
6.6 Molybdate Solution (44 g/L)—Dissolve 4.4 g of ammo-
7.5 In the event chlorine is inhaled, first aid should be
nium molybdate (NH ) Mo O ·4H O) in 10 mL of 6 N
4 6 7 24 2
summoned immediately and oxygen administered without
ammonium hydroxide (NH OH) and dilute with 90 mL of
delay.
water.
7.6 Chlorine is a corrosive and toxic material. A well-
6.7 Phenolphthalein Indicator Solution (1 g/L)—Dissolve
ventilated fume hood should be used to house all test equip-
0.1 g of phenolphthalein and 0.1 g sodium carbonate (Na CO )
2 3
ment when this product is analyzed in the laboratory.
in 5 mL of water and dilute to 100 mL.
6.8 Phosphate Solution (100 g/L)—Dissolve 50 g of
8. Sampling
monobasic sodium phosphate (NaH PO ·H O) in 500 mL of
2 4 2
8.1 Record to the nearest 0.1 g the tare of the clean and dry
water.
chlorine gas sampling apparatus of Fig. 1, except for the
6.9 Sodium Hydroxide Solution (10 %)—Dissolve 1 part by
insulatingjacket.Add150mLof10 %NaOHsolutionandseal
weight of sodium hydroxide (NaOH) in 9 parts of water.
the stopper firmly in place. Record the tare of this assembly to
the nearest 0.1 g. Place the apparatus in the insulating jacket
6.10 Sodium Thiosulfate, Standard Solution (0.01 N)—
and allow to stand to reach temperature equilibrium.
Prepare 0.01 N sodium thiosulfate solution fresh daily by
accurate dilution of standard 0.1 N sodium thiosulfate solution.
8.2 Saturated chlorine vapor will contain only about 0.3 as
Prepare, standardize, and restandardize 0.1 N sodium thiosul-
much bromine as the liquid chlorine with which it is in
fate solution in accordance with Practice E200.
equilibrium. Hence, when sampling containers of liquid
chlorine, it is necessary to make sure that only the liquid phase
6.11 Starch Solution (10 g/L)—Dissolve1gof soluble
is sampled. Gaseous chlorine sources may be sampled directly,
starch in 100 mL of boiling water. Make up fresh daily.
using a small valve to control the chlorine flow. Feed the liquid
6.12 Sulfuric Acid (6 N)—Add slowly and cautiously with
chlorine through vaporizing equipment before passing the
constant stirring, 1 volume of concentrated sulfuric acid (sp gr
resulting vapor into the chlorine gas sampling apparatus.There
1.84, H SO ) to 5.5 volumes of water. Warning: Use goggles
2 4
is no need for a vaporizer when sampling a source of gaseous
when preparing this solution. Cool to room temperature before
chlorine.
use.
8.3 A satisfactory vaporizer for sampling liquid chlorine is
6.13 Sulfuric Acid (1+1)—Add slowly with stirring 1
readily prepared from a 1.5 to 2-m length of ⁄4-in. (6.35-mm)
volume of concentrated sulfuric acid (H SO , sp gr 1.84) to 1
2 4
outside diameter polytrafluoroethylene (PTFE) instrument air
volume of water. Warning: Use goggles when preparing
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E649 − 17 E649 − 17a
Standard Test Method for
Bromine in Chlorine
This standard is issued under the fixed designation E649; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of bromine in liquid chlorine and in gaseous chlorine with a lower limit of
detection of 4 ug/kg by weight.
1.2 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures, handling,
and safety precautions.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use. Specific hazard statements are given in Section 7.
1.3 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures, handling,
and safety precautions.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
E200 Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis
3. Summary of Test Method
3.1 Chlorine gas is sampled by absorption in aqueous sodium hydroxide (NaOH). Liquid chlorine is first vaporized, and the
vapor is absorbed in aqueous sodium hydroxide. An aliquot of the sample solution is reduced with an excess of sulfite ion, acidified,
and excess sulfur dioxide (SO ) boiled out. In a carefully buffered solution, bromide is oxidized to bromate by hypochlorite. Excess
hypochlorite is reduced to chloride by formate. In the presence of molybdate catalyst, bromate is reduced to bromide by iodide,
and the liberated iodine is titrated with standard sodium thiosulfate solution.
4. Significance and Use
4.1 Low levels of bromine contaminant in chlorine cause problems in some industrial uses. This test method may be used to
determine bromine in liquid or gaseous chlorine at levels as low as 4 ug/kg.
5. Apparatus
5.1 The construction of the chlorine gas sampling apparatus and of the assembled sampling equipment is shown in Figs. 1 and
2. Modification of the equipment to deal with special sampling circumstances may be necessary. In Fig. 2, the control valve is
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic Hydrocarbons Aromatic, Industrial, Specialty and Related Chemicals and is the direct
responsibility of Subcommittee D16.16 on Industrial and Specialty Product Standards.
Current edition approved March 1, 2017July 1, 2017. Published March 2017July 2017. Originally approved in 1978. Last previous edition approved in 20112017 as
E649 – 00 (2011).E649 – 17. DOI: 10.1520/E0649-17.10.1520/E0649-17a.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E649 − 17a
FIG. 1 Chlorine Gas Sampling Apparatus
1. Inverted liquid chlorine cylinder
2. Chlorine cylinder valve
3. Control valve ( ⁄4-in. 316 stainless steel needle valve)
4. ⁄4-in. PTFE instrument air tubing
5. Small pail or 2-L beaker filled with water
6. Evaporating coil (two loops held with tape at indicated points)
7. Gas sampling apparatus of Fig. 1
FIG. 2 Liquid Chlorine Sampling Equipment
E649 − 17a
shown with an adaptor for connection to a chlorine cylinder valve. Other adaptors will be required when sampling liquid chlorine
in liquefaction plant streams or from large shipping or storage containers.
5.2 A 10-mL buret calibrated in 0.05-mL divisions is used when titrating with 0.01 N standard sodium thiosulfate solution.
6. Reagents
6.1 Purity of Reagents—Unless otherwise indicated, it is intended that all reagents should conform to the specifications of the
Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
6.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean Type II or Type III reagent
water conforming to Specification D1193.
6.3 Chloride Solution—Dissolve 200 g of sodium chloride (NaCl) in 940 mL of water.
6.4 Formate Solution—Dissolve 50 g of sodium formate in 175 mL of water.
6.5 Hypochlorite Solution—Dissolve 6.2 g of NaOH in 190 mL of water and chlorinate slowly, with stirring, to give a gain in
weight of 5.0 g.
NOTE 1—If desired, a commercial hypochlorite bleach solution may be used as the hypochlorite solution. Although such commercial bleach solutions
are not made from reagent grade chemicals, the reagent blank determination (see 9.4) corrects for the bromine and any chlorate content in such solutions.
A proprietary 5.25 % sodium hypochlorite bleach solution is satisfactory for this use. Because the sodium chloride and hypochlorite solutions contribute
most to the reagent blanks, dispense these solutions by pipet to ensure uniformity of blanks and determinations (see 9.1 and 9.2).
6.6 Molybdate Solution (44 g/L)—Dissolve 4.4 g of ammonium molybdate (NH ) Mo O ·4H O) in 10 mL of 6 N ammonium
4 6 7 24 2
hydroxide (NH OH) and dilute with 90 mL of water.
6.7 Phenolphthalein Indicator Solution (1 g/L)—Dissolve 0.1 g of phenolphthalein and 0.1 g sodium carbonate (Na CO ) in 5
2 3
mL of water and dilute to 100 mL.
6.8 Phosphate Solution (100 g/L)—Dissolve 50 g of monobasic sodium phosphate (NaH PO ·H O) in 500 mL of water.
2 4 2
6.9 Sodium Hydroxide Solution (10 %)—Dissolve 1 part by weight of sodium hydroxide (NaOH) in 9 parts of water.
6.10 Sodium Thiosulfate, Standard Solution (0.01 N)—Prepare 0.01 N sodium thiosulfate solution fresh daily by accurate
dilution of standard 0.1 N sodium thiosulfate solution. Prepare, standardize, and restandardize 0.1 N sodium thiosulfate solution
in accordance with Practice E200.
6.11 Starch Solution (10 g/L)—Dissolve 1 g of soluble starch in 100 mL of boiling water. Make up fresh daily.
6.12 Sulfuric Acid (6 N)—Add slowly and cautiously with constant stirring, 1 volume of concentrated sulfuric acid (sp gr 1.84,
H SO ) to 5.5 volumes of water. Warning: Use goggles when preparing this solution. Cool to room temperature before use.
2 4
6.13 Sulfuric Acid (1 + 1)—Add slowly with stirring 1 volume of concentrated sulfuric acid (H SO , sp gr 1.84) to 1 volume
2 4
of water. Warning: Use goggles when preparing this solution.
7. Hazards
7.1 Concentrated sulfuric acid, sodium hydroxide, and sodium hypochlorite solutions are rapidly corrosive to skin and eyes. In
case of accidental contact with skin, immediately flush affected part with large volumes of water. For eyes, immediately flush with
water for at least 15 min; then get prompt medical attention. Goggles should be worn when handling these chemicals.
7.2 Chlorine sampling should be performed only by persons thoroughly familiar with handling this material and with operation
of the sampling system. Personnel should be equipped with a respirator, goggles, faceshield, and gloves. If possible, perform
sampling in a hood or other well-ventilated area. In sampling liquid chlorine, do not leave the space between two closed valves
filled with liquid chlorine (see 8.6).
7.3 When sampling and working with chlorine out of doors, people downwind from such operation should be warned of the
possible release of chlorine vapors.
7.4 It is recommended that means be available for disposal of excess chlorine in an environmentally safe and acceptable manner.
If chlorine cannot be disposed of in a chlorine-consuming process, a chlorine absorption system should be provided. When the
analysis and sampling regimen requires an initial purging of chlorine from a container, the purged chlorine should be similarly
handled. Purging to the atmosphere should be avoided.
7.5 In the event chlorine is inhaled, first aid should be summoned immediately and oxygen administered without delay.
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by
the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
E649 − 17a
7.6 Chlorine is a corrosive and toxic material. A well-ventilated fume hood should be used to house all test equipment when
this product is analyzed in the laboratory.
8. Sampling
8.1 Record to the nearest 0.1 g the tare of the clean and dry chlorine gas sampling apparatus of Fig. 1, except for the insulating
jacket. Add 150 mL of 10 % NaOH solution and seal the stopper firmly in place. Record the tare of this assembly to the nearest
0.1 g. Place the apparatus in the insulating jacket and allow to stand to reach temperature equilibrium.
8.2 Saturated chlorine vapor will contain only about 0.3 as much bromine as the liquid chlorine with which it is in equilibrium.
Hence, when sampling containers of liquid chlorine, it is necessary to make sure that only the liquid phase is sampled. Gaseous
chlorine sources may be samp
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...