ASTM E806-21
(Test Method)Standard Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure)
Standard Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure)
SIGNIFICANCE AND USE
4.1 CCl4 and CHCl3 may be present in trace amounts in liquid chlorine. The use of chlorine to purify water would then transfer these compounds to the water. Therefore, when the concentrations of the CCl4 and CHCl3 in the liquid chlorine are known, the maximum amounts contributed to the water by the chlorine can be estimated.
SCOPE
1.1 This test method is designed for the determination of carbon tetrachloride (CCl4) and chloroform (CHCl3) in liquid chlorine. The lower limit of detection is dependent on the sample size and the instrument used; five ppm (w/w) is achievable.
1.2 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7 and in 9.2.3.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2021
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.13 - Chlorine
Relations
- Effective Date
- 01-Nov-2016
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Jun-2007
- Effective Date
- 15-Nov-2006
- Effective Date
- 15-Sep-2006
- Effective Date
- 01-May-2006
- Effective Date
- 01-Dec-2004
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-May-2002
- Effective Date
- 10-May-1999
Overview
ASTM E806-21 establishes a standardized test method for the determination of carbon tetrachloride (CCl₄) and chloroform (CHCl₃) in liquid chlorine, using a direct injection gas chromatographic procedure. Developed by ASTM International, this standard supports quality assurance and regulatory compliance for chlorine producers and users, particularly where chlorine is employed for water purification. Trace contaminants such as CCl₄ and CHCl₃ may transfer from chlorine to treated water, highlighting the need for precise quantification as specified in ASTM E806-21.
Key Topics
Scope and Relevance
- Applies to the quantification of carbon tetrachloride and chloroform in liquid chlorine.
- Facilitates detection and reporting at concentrations as low as 5 ppm (w/w), depending on sample size and instrumentation.
- Utilizes only SI units for measurement consistency.
Apparatus & Materials
- Requires a gas chromatograph (GC) equipped with a suitable detector (typically flame ionization) and specific column materials compatible with chlorine.
- Use of autoinjectors is mandatory when implementing an external standard for results calculation.
- Handles sample collection using specialized nickel, Monel, or tantalum sample cylinders to preserve analyte stability.
Health and Safety Considerations
- Chlorine is highly toxic and requires stringent handling protocols including well-ventilated fume hoods, eye protection, and respiratory equipment.
- Safe disposal and environmental management of excess or purged chlorine is emphasized to prevent harmful releases.
Analytical Process
- Samples are injected directly into the gas chromatograph.
- Quantification is achieved through peak area comparison between samples and calibration standards.
- Calibration must account for matrix effects inherent to liquid chlorine.
Quality Assurance
- Laboratories applying this standard are required to maintain robust QA/QC systems.
- Performance of analytical instruments and overall method validation must be periodically checked using standard quality control samples.
Applications
Water Purification
- Ensures that levels of carbon tetrachloride and chloroform contributed by chlorine do not exceed regulatory thresholds in potable water supplies.
- Provides utilities and water treatment facilities with a validated method for monitoring chlorine purity and trace contaminant carryover.
Industrial Chlorine Production
- Serves chlorine manufacturers and distributors by supporting product consistency, customer specification compliance, and safety requirements.
- Assists in internal quality control and third-party laboratory verifications.
Regulatory Compliance
- Supports compliance with occupational and environmental regulations, such as OSHA exposure limits and relevant transportation requirements for hazardous chemicals.
- Standardizes reporting procedures and rounding in accordance with Practice ASTM E29.
Related Standards
- ASTM D6809 – Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials.
- ASTM E29 – Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications.
- OSHA Regulations (29 CFR 1910.1000 and 1910.1200) – Air contaminant exposure limits and hazard communication.
- 49 CFR 173 – US Department of Transportation requirements for shipping liquefied compressed gases, including liquid chlorine.
Summary
ASTM E806-21 offers a reliable and internationally recognized method for the analysis of carbon tetrachloride and chloroform in liquid chlorine. Its adoption ensures accuracy, repeatability, and regulatory compliance in critical applications such as water purification and industrial chlorine production, while also addressing safety, quality assurance, and environmental stewardship. For implementation and further details, refer to the current official version of this ASTM standard.
Buy Documents
ASTM E806-21 - Standard Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure)
REDLINE ASTM E806-21 - Standard Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E806-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Carbon Tetrachloride and Chloroform in Liquid Chlorine by Direct Injection (Gas Chromatographic Procedure)". This standard covers: SIGNIFICANCE AND USE 4.1 CCl4 and CHCl3 may be present in trace amounts in liquid chlorine. The use of chlorine to purify water would then transfer these compounds to the water. Therefore, when the concentrations of the CCl4 and CHCl3 in the liquid chlorine are known, the maximum amounts contributed to the water by the chlorine can be estimated. SCOPE 1.1 This test method is designed for the determination of carbon tetrachloride (CCl4) and chloroform (CHCl3) in liquid chlorine. The lower limit of detection is dependent on the sample size and the instrument used; five ppm (w/w) is achievable. 1.2 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7 and in 9.2.3. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 CCl4 and CHCl3 may be present in trace amounts in liquid chlorine. The use of chlorine to purify water would then transfer these compounds to the water. Therefore, when the concentrations of the CCl4 and CHCl3 in the liquid chlorine are known, the maximum amounts contributed to the water by the chlorine can be estimated. SCOPE 1.1 This test method is designed for the determination of carbon tetrachloride (CCl4) and chloroform (CHCl3) in liquid chlorine. The lower limit of detection is dependent on the sample size and the instrument used; five ppm (w/w) is achievable. 1.2 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazards statements are given in Section 7 and in 9.2.3. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E806-21 is classified under the following ICS (International Classification for Standards) categories: 71.060.10 - Chemical elements. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E806-21 has the following relationships with other standards: It is inter standard links to ASTM D6809-02(2016), ASTM E29-08, ASTM D6809-02(2007), ASTM E29-06b, ASTM E29-06a, ASTM E29-06, ASTM E29-04, ASTM D6809-02, ASTM E29-02e1, ASTM E29-93a(1999). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E806-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E806 − 21
Standard Test Method for
Carbon Tetrachloride and Chloroform in Liquid Chlorine by
Direct Injection (Gas Chromatographic Procedure)
This standard is issued under the fixed designation E806; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* E29 Practice for Using Significant Digits in Test Data to
Determine Conformance with Specifications
1.1 This test method is designed for the determination of
E180 Practice for Determining the Precision of ASTM
carbon tetrachloride (CCl ) and chloroform (CHCl ) in liquid
4 3
Methods for Analysis and Testing of Industrial and Spe-
chlorine. The lower limit of detection is dependent on the
cialty Chemicals (Withdrawn 2009)
sample size and the instrument used; five ppm (w/w) is
2.2 Federal Standard:
achievable.
OSHA Regulations, 29 CFR paragraphs 19100.1000 and
1.2 In determining the conformance of the test results using
1910.1200 Air contaminates – Table of exposure limits
this method to applicable specifications, results shall be
and Hazard Communication
rounded off in accordance with the rounding-off method of
49 CFR 173 Code of Federal Regulations Title 49 Transpor-
Practice E29.
tation;Shippers’GeneralRequirementsforShipmentsand
1.3 The values stated in SI units are to be regarded as
Packagings, including Sections:
standard. No other units of measurement are included in this
173.304 Charging of Cylinders with Liquefied Compressed
standard.
Gas
1.4 This standard does not purport to address all of the 173.314 Requirements for Compressed Gases in Tank Cars
173.315 Compressed Gases in Cargo Tanks and Portable
safety concerns, if any, associated with its use. It is the
Tank Containers
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter-
2.3 Other Document:
mine the applicability of regulatory limitations prior to use.
Chlorine Institute Pamphlet No. 1 Chlorine Basics
Specific hazards statements are given in Section 7 and in 9.2.3.
1.5 This international standard was developed in accor-
3. Summary of Test Method
dance with internationally recognized principles on standard-
3.1 A sample of liquid chlorine is injected into a gas
ization established in the Decision on Principles for the
chromatograph (GC), equipped with a column capable of
Development of International Standards, Guides and Recom-
separating CCl and CHCl from Cl and other impurities,
4 3 2
mendations issued by the World Trade Organization Technical
using a suitable syringe. The amounts of CCl and CHCl in
4 3
Barriers to Trade (TBT) Committee.
the sample are determined by comparison of the areas of the
peaks, obtained with the samples, to areas of peaks obtained
2. Referenced Documents
with suitable calibration standards, under the same conditions.
2.1 ASTM Standards:
D6809 Guide for Quality Control and Quality Assurance
4. Significance and Use
Procedures for Aromatic Hydrocarbons and Related Ma-
4.1 CCl and CHCl may be present in trace amounts in
4 3
terials
liquid chlorine. The use of chlorine to purify water would then
transfer these compounds to the water. Therefore, when the
concentrationsoftheCCl andCHCl intheliquidchlorineare
4 3
This test method is under the jurisdiction of ASTM Committee D16 on
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi-
bility of Subcommittee D16.13 on Chlorine.
Current edition approved Nov. 1, 2021. Published December 2021. Originally The last approved version of this historical standard is referenced on
approved in 1981. Last previous edition approved in 2017 as E806 – 17a. DOI: www.astm.org.
10.1520/E0806-21. Available from U.S. Government Printing Office, Superintendent of
For referenced ASTM standards, visit the ASTM website, www.astm.org, or Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM www.access.gpo.gov.
Standards volume information, refer to the standard’s Document Summary page on Available from The Chlorine Institute Inc., 1300 Wilson Blvd., Suite 525,
the ASTM website. Arlington, VA 22209, https://www.chlorineinstitute.org.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E806 − 21
known, the maximum amounts contributed to the water by the 6.4 Chloroform, reagent grade.
chlorine can be estimated.
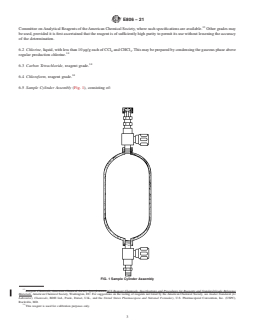
6.5 Sample Cylinder Assembly (Fig. 1), consisting of:
6.5.1 Sample Cylinders; nickel,Monel,ortantalum(Note
5. Apparatus
1), 400-mLcapacity, double-ended, specially cleaned (Note 2).
5.1 Gas Chromatograph, equipped with:
6.5.2 Valves, having a packing resistant to liquid chlorine.
5.1.1 Injection Port, must be lined with glass, Monel, or
6.5.3 Holder for a Septum, that can be easily assembled.
nickel; or column must be installed for on-column injection.
NOTE 1—Carbon or stainless steel cylinders and fittings are not suitable
5.1.2 Septa, from Viton. Silicone septa may produce arti-
as CHCl is unstable in the presence of FeCl and Cl .
3 3 2
facts that may interfere with the analysis.
NOTE 2—A procedure for cleaning cylinders and valves, for use with
5.1.3 Column, Column Materials, and Packing, must be
liquid chlorine, is given in Appendix X2.
compatible with chlorine. Silanized supports and silanized
6.6 Fittings, for transferring chlorine from one cylinder to
glass wool must be avoided. Column must be able to separate
another.
Cl , CCl , and CHCl . Columns that have been found to be
2 4 3
suitable are:
5.1.3.1 Nickel Tubing, 3.05 m by 3.175 mm outside
diameter, packed with 10 % sodium chloride solution on
8 15
If samples are to be shipped outside any plant, cylinders approved by the U.S.
Porasil C (see Appendix X1 for packing preparation). This is
Department of Transportation must be used.
the preferred packing.
Packing made from Teflon, registered trademark of The Chemours Company,
5.1.3.2 Polytetrafluoroethylene Tubing, 3.05mby2mm
Viton, Kel-F, or equivalent have been found suitable for this purpose.
inside diameter, packed with 20 % Kel-F No. 10 oil on 60/80 Swagelok, registered trademark of Swagelok Company, or equivalent fittings
have been found suitable for this purpose.
mesh Chromosorb WAW.
5.1.3.3 Glass Tubing, 3.05 m by 2 mm inside diameter,
packed with 20 % Halocarbon 1025 on 60/80 mesh Chromo-
sorb W AW.
5.1.4 Flame Ionization Detector.
5.1.5 Recorder, compatible with the GC detector output.
5.1.6 Electronic Integrator (optional), compatible with the
GC detector output.
5.2 Balance, capacity 5000 g, reading to 61g.
5.3 Autoinjectors are required for all gas chromatograph
standards using an external standard to calculate results.
6. Reagents and Materials
6.1 Purity of Reagents—Unless otherwise indicated, it is
intended that all reagents shall conform to the specifications of
theCommitteeonAnalyticalReagentsoftheAmericanChemi-
cal Society, where such specifications are available. Other
grades may be used, provided it is first ascertained that the
reagent is of sufficiently high purity to permit its use without
lessening the accuracy of the determination.
6.2 Chlorine,liquid,withlessthan10µg/geachofCCl and
CHCl .Thismaybepreparedbycondensingthegaseousphase
above regular production chlorine.
6.3 Carbon Tetrachloride, reagent grade.
Monel is a registered trademark of Special Metals Corporation.
Viton is a registered trademark of The Chemours Company.
Porasil is a trademark of Waters Associates, Inc.
Kel-F is a registered trademark of 3M Company.
Chromosorb is a registered trademark of Imerys Minerals California, Inc.
Halocarbon is a registered trademark of Halocarbon Products Corporation.
A 400-mL nickel cylinder filled with liquid chlorine weighs about 4000 g.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD.
This reagent is used for calibration purposes only. FIG. 1 Sample Cylinder Assembly
E806 − 21
6.7 Syringe, 10 to 100-µL, capable of holding liquid chlo- 9. Preparation of Standards for Calibration
rine under pressure, with 26-gage disposable needle.
9.1 Prepare standards in liquid chlorine, so that matrix
effects of the chlorine on the gas chromatographic column and
NOTE 3—Disposable needles are recommended because corrosion with
permanent needles may cause problems.
detector are compensated.
9.2 Method of Additions—Add CCl and CHCl to cylinder
4 3
7. Hazards
containing liquid chlorine as follows:
7.1 Chlorine is a corrosive and toxic material. A well-
9.2.1 Obtain a supply cylinder of liquid chlorine that has
ventilated fume hood should be used to house all test
less than 10 ppm each of CCl and CHCl , and that contains at
4 3
equipment, except the gas chromatograph, when this product is
least 5000 g of chlorine. Label this cylinder No. 1.
analyzed in the laboratory.
9.2.2 Obtain a clean, evacuated, sample cylinder equipped
with a septum on one of the valves. Label this cylinder No. 2
7.2 The analysis should be attempted only by persons who
and weigh it to 61g.
are thoroughly familiar with the handling of chlorine, and even
an experienced person should not work alone. The operator 9.2.3 Connect cylinder No. 1 to cylinder No. 2 by means of
mustbeprovidedwithadequateeyeprotectionandarespirator. fittings (6.6) such that the liquid phase of chlorine can flow
Splashes of liquid chlorine destroy clothing and, if such from 1 to 2. Open the valves between the cylinders and cool
clothing is next to the skin, will produce irritations and burns. cylinder No. 2 with ice. Liquid chlorine will be transferred
from cylinder No. 1 to cylinder No. 2. Close the valves when
7.3 When sampling and working with chlorine out of doors,
sufficient chlorine has been transferred. Disconnect the cylin-
peopledownwindfromsuchoperationshouldbewarnedofthe
dersandweighcylinderNo.2to 61gtodeterminetheweight
possible release of chlorine vapors.
of chlorine transferred. (Warning—Do not allow cylinder No.
7.4 It is recommended that means be available for disposal 2 to become liquid full. A good rule is that the weight of
of excess chlorine in an environmentally safe and acceptable chlorine in the cylinder should not be more than 125 % of the
manner. If chlorine cannot be disposed of in a chlorine weight of water that the cylinder could contain.)
consuming process, a chlorine absorption system should be
9.2.4 Retain cylinder No. 1 to prepare further standards.
provided.When the analysis and sampling regimen requires an
9.2.5 Prepare an approximately 50/50 mix of CCl and
initialpurgingofchlorinefromacontainer,thepurgedchlorine
CHCl and record amounts of each added. Calculate the
should be similarly handled. Purging to the atmosphere should
volume of this mixture needed to prepare one level of standard
be avoided.
for calibration, using a calculation similar to that given in 9.3.
9.2.6 Fill the high-pressure syringe (6.7) with approxi-
7.5 In the event chlorine is inhaled, first aid should be
mately the volume of the CCl /CHCl mixture as calculated in
summoned immediately and oxygen administered without
4 3
9.3.3. Weigh the syringe plus liquid to 6 0.1 mg. Transfer the
delay.
liquid mixture through the septum into the vapor space of
7.6 ReviewthecurrentSafetyDataSheet(SDS)fordetailed
cylinder No. 2. Keep a finger tightly over the plunger to
informationconcerningtoxicity,firstaidprocedures,andsafety
preventblowout.Immediatelyremoveandreweighthesyringe
precautions.
to 6 0.1 mg. The difference between the two weights is the
total weight of CCl and CHCl added.
4 3
8. Sampling
9.2.7 Shake cylinder No. 2 to assure complete solution of
8.1 Sampling from tank cars, barges, storage tanks, and the CCl and CHCl in the chlorine.
4 3
large cylinders presents unique problems. Each facility,
9.2.8 Calculate the added concentration of CCl and CHCl
4 3
however, must be capable of delivering a liquid sample (not
in the spiked standard as indicated in 9.4.
gas) for test. See Chlorine Institute Pamphlet No. 1.
9.2.9 Prepare at least three standards containing three dif-
ferentlevelsofCCl andCHCl ,bracketingtheexpectedlevel.
4 3
8.2 Since the location of these larger facilities may not be at
Also, transfer some of the original chlorine into a sample
the immediate site of analysis, sample collection in a suitable
cylinder without adding CCl or CHCl .
4 3
secondary container is recommended to facilitate its safe
9.2.10 The long term stability of the calibration standards
transport to the laboratory for tests (DOT regulations may be
has not been evaluated.
applicable).
9.3 Example of amounts of CCl and CHCl to be added to
8.3 It is recommended that samples be collected from these
4 3
liquid chlorine to produce desired standard:
facilities in small-size cylinders, with cylinders and valves
fabricated of tantalum, Monel, or nickel (carbon or stainless 9.3.1 ProposedmixtureofCCl andCHCl (averagedensity
4 3
steel are unsuitable), and capable of being negotiated in the about 1.5 g/mL, or 1.5 mg/µL):
laboratory fume hood. Proper and safe sampling techniques
CCl 47.55 g
CHCl 44.40 g
must be followed. Do not allow the sample cylinder to become
Total 91.95 g
liquid full.Agood rule is that the weight of the chlorine in the
cylinder should not be more than 125 % of the weight of the 9.3.2 To prepare 500 g of chlorine with spiked levels of 20
water that the cylinder could contain. This rule is stated in ppm each of CCl and CHCl (total of 40 µg/g), the necessary
4 3
accordance with 49 CFR 173. grams (W) of the CCl /CHCl mixture is as follows:
4 3
E806 − 21
W 40 10.2.4 Column Temperature—60°C.
5 (1)
500 10
10.2.5 Injector Temperature—75°C.
or
10.3 Column—Halocarbon on Chromosorb or equivalent:
W 5 0.020 g 5 20 mg (2)
10.3.1 Detector—Flame ionization.
9.3.3 The necessary volume in µL (V) is then:
10.3.2 Detector Temperature—150°C.
10.3.3 Carrier Gas—Nitrogen at 30 mL/min.
W 20
V 5 5 5 13µL (3)
density 1.5 10.3.4 Column Tempera
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E806 − 17a E806 − 21
Standard Test Method for
Carbon Tetrachloride and Chloroform in Liquid Chlorine by
Direct Injection (Gas Chromatographic Procedure)
This standard is issued under the fixed designation E806; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method is designed for the determination of carbon tetrachloride (CCl ) and chloroform (CHCl ) in liquid chlorine.
4 3
The lower limit of detection is dependent on the sample size and the instrument used; five ppm (w/w) is achievable.
1.2 Review the current Safety Data Sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety
precautions.In determining the conformance of the test results using this method to applicable specifications, results shall be
rounded off in accordance with the rounding-off method of Practice E29.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use. Specific hazards statements are given in Section 7 and in 9.2.3.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
2.2 Federal Standard:
OSHA Regulations, 29 CFR paragraphs 19100.1000 and 1910.1200 Air contaminates – Table of exposure limits and Hazard
Communication
49 CFR 173 CFR 173 Code of Federal Regulations Title 49 Transportation; Shippers’ General Requirements for Shipments and
Packagings, including Sections:
173.304 Charging of Cylinders with Liquefied Compressed Gas
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsibility of
Subcommittee D16.13 on Chlorine.
Current edition approved July 1, 2017Nov. 1, 2021. Published July 2017December 2021. Originally approved in 1981. Last previous edition approved in 2017 as
E806 – 17.E806 – 17a. DOI: 10.1520/E0806-17a.10.1520/E0806-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Available from U.S. Government Printing Office, Superintendent of Documents, 732 N. Capitol St., NW, Washington, DC 20401-0001, http://www.access.gpo.gov.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E806 − 21
173.314 Requirements for Compressed Gases in Tank Cars
173.315 Compressed Gases in Cargo Tanks and Portable Tank Containers
2.3 Other Document:
Chlorine Institute Pamphlet No. 1 Chlorine Basics
3. Summary of Test Method
3.1 A sample of liquid chlorine is injected into a gas chromatograph (GC), equipped with a column capable of separating CCl
and CHCl from Cl and other impurities, using a suitable syringe. The amounts of CCl and CHCl in the sample are determined
3 2 4 3
by comparison of the areas of the peaks, obtained with the samples, to areas of peaks obtained with suitable calibration standards,
under the same conditions.
4. Significance and Use
4.1 CCl and CHCl may be present in trace amounts in liquid chlorine. The use of chlorine to purify water would then transfer
4 3
these compounds to the water. Therefore, when the concentrations of the CCl and CHCl in the liquid chlorine are known, the
4 3
maximum amounts contributed to the water by the chlorine can be estimated.
5. Apparatus
5.1 Gas Chromatograph, equipped with:
5.1.1 Injection Port, must be lined with glass, Monel, or nickel; or column must be installed for on-column injection.
5.1.2 Septa, from Viton. Silicone septa may produce artifacts that may interfere with the analysis.
5.1.3 Column, Column Materials, and Packing, must be compatible with chlorine. Silanized supports and silanized glass wool
must be avoided. Column must be able to separate Cl , CCl , and CHCl . Columns that have been found to be suitable are:
2 4 3
5.1.3.1 Nickel Tubing, 3.05 m by 3.175 mm outside diameter, packed with 10 % sodium chloride solution on Porasil C (see
Appendix X1 for packing preparation). This is the preferred packing.
5.1.3.2 Polytetrafluoroethylene Tubing, 3.05 m by 2 mm inside diameter, packed with 20 % Kel-F No. 10 oil on 60/80 mesh
Chromosorb W AW.
5.1.3.3 Glass Tubing, 3.05 m by 2 mm inside diameter, packed with 20 % Halocarbon 1025 on 60/80 mesh Chromosorb W AW.
5.1.4 Flame Ionization Detector.
5.1.5 Recorder, compatible with the GC detector output.
5.1.6 Electronic Integrator (optional), compatible with the GC detector output.
5.2 Balance, capacity 5000 g, reading to 6 1 g.
5.3 Autoinjectors are required for all gas chromatograph standards using an external standard to calculate results.
6. Reagents and Materials
6.1 Purity of Reagents—Unless otherwise indicated, it is intended that all reagents shall conform to the specifications of the
Available from The Chlorine Institute Inc., 1300 Wilson Blvd., Suite 525, Arlington, VA 22209, https://www.chlorineinstitute.org.
Monel is a registered trademark of Special Metals Corporation.
Viton is a registered trademark of The Chemours Company.
Porasil is a trademark of Waters Associates, Inc.
Kel-F is a registered trademark of 3M Company.
Chromosorb is a registered trademark of Imerys Minerals California, Inc.
Halocarbon is a registered trademark of Halocarbon Products Corporation.
A 400-mL nickel cylinder filled with liquid chlorine weighs about 4000 g.
E806 − 21
Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
6.2 Chlorine, liquid, with less than 10 μg/g each of CCl and CHCl . This may be prepared by condensing the gaseous phase above
4 3
regular production chlorine.
6.3 Carbon Tetrachloride, reagent grade.
6.4 Chloroform, reagent grade.
6.5 Sample Cylinder Assembly (Fig. 1), consisting of:
FIG. 1 Sample Cylinder Assembly
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC),
Rockville, MD.
This reagent is used for calibration purposes only.
E806 − 21
6.5.1 Sample Cylinders; nickel, nickel, Monel, or tantalum (Note 1), 400-mL capacity, double-ended, specially cleaned (Note 2).
6.5.2 Valves, having a packing resistant to liquid chlorine.
6.5.3 Holder for a Septum, that can be easily assembled.
NOTE 1—Carbon or stainless steel cylinders and fittings are not suitable as CHCl is unstable in the presence of FeCl and Cl .
3 3 2
NOTE 2—A procedure for cleaning cylinders and valves, for use with liquid chlorine, is given in Appendix X2.
6.6 Fittings, for transferring chlorine from one cylinder to another.
6.7 Syringe, 10 to 100-μL, capable of holding liquid chlorine under pressure, with 26-gage disposable needle.
NOTE 3—Disposable needles are recommended because corrosion with permanent needles may cause problems.
7. Hazards
7.1 Chlorine is a corrosive and toxic material. A well-ventilated fume hood should be used to house all test equipment, except the
gas chromatograph, when this product is analyzed in the laboratory.
7.2 The analysis should be attempted only by persons who are thoroughly familiar with the handling of chlorine, and even an
experienced person should not work alone. The operator must be provided with adequate eye protection and a respirator. Splashes
of liquid chlorine destroy clothing and, if such clothing is next to the skin, will produce irritations and burns.
7.3 When sampling and working with chlorine out of doors, people downwind from such operation should be warned of the
possible release of chlorine vapors.
7.4 It is recommended that means be available for disposal of excess chlorine in an environmentally safe and acceptable manner.
If chlorine cannot be disposed of in a chlorine consuming process, a chlorine absorption system should be provided. When the
analysis and sampling regimen requires an initial purging of chlorine from a container, the purged chlorine should be similarly
handled. Purging to the atmosphere should be avoided.
7.5 In the event chlorine is inhaled, first aid should be summoned immediately and oxygen administered without delay.
7.6 Review the current Safety Data Sheet (SDS) for detailed information concerning toxicity, first aid procedures, and safety
precautions.
8. Sampling
8.1 Sampling from tank cars, barges, storage tanks, and large cylinders presents unique problems. Each facility, however, must be
capable of delivering a liquid sample (not gas) for test. See Chlorine Institute Pamphlet No. 1.
8.2 Since the location of these larger facilities may not be at the immediate site of analysis, sample collection in a suitable
secondary container is recommended to facilitate its safe transport to the laboratory for tests (DOT regulations may be applicable).
8.3 It is recommended that samples be collected from these facilities in small-size cylinders, with cylinders and valves fabricated
of tantalum, Monel, or nickel (carbon or stainless steel are unsuitable), and capable of being negotiated in the laboratory fume
hood. Proper and safe sampling techniques must be followed. Do not allow the sample cylinder to become liquid full. A good rule
If samples are to be shipped outside any plant, cylinders approved by the U.S. Department of Transportation must be used.
Packing made from Teflon, registered trademark of The Chemours Company, Viton, Kel-F, or equivalent have been found suitable for this purpose.
Swagelok, registered trademark of Swagelok Company, or equivalent fittings have been found suitable for this purpose.
E806 − 21
is that the weight of the chlorine in the cylinder should not be more than 125 % of the weight of the water that the cylinder could
contain. This rule is stated in accordance with 49 CFR 173.
9. Preparation of Standards for Calibration
9.1 Prepare standards in liquid chlorine, so that matrix effects of the chlorine on the gas chromatographic column and detector are
compensated.
9.2 Method of Additions—Add CCl and CHCl to cylinder containing liquid chlorine as follows:
4 3
9.2.1 Obtain a supply cylinder of liquid chlorine that has less than 10 ppm each of CCl and CHCl , and that contains at least 5000
4 3
g of chlorine. Label this cylinder No. 1.
9.2.2 Obtain a clean, evacuated, sample cylinder equipped with a septum on one of the valves. Label this cylinder No. 2 and weigh
it to 6 1 g.
9.2.3 Connect cylinder No. 1 to cylinder No. 2 by means of fittings (6.6) such that the liquid phase of chlorine can flow from 1
to 2. Open the valves between the cylinders and cool cylinder No. 2 with ice. Liquid chlorine will be transferred from cylinder
No. 1 to cylinder No. 2. Close the valves when sufficient chlorine has been transferred. Disconnect the cylinders and weigh cylinder
No. 2 to 6 1 g to determine the weight of chlorine transferred. (Warning—Do not allow cylinder No. 2 to become liquid full.
A good rule is that the weight of chlorine in the cylinder should not be more than 125 % of the weight of water that the cylinder
could contain.Warning)—Do not allow cylinder No. 2 to become liquid full. A good rule is that the weight of chlorine in the
cylinder should not be more than 125 % of the weight of water that the cylinder could contain.)
9.2.4 Retain cylinder No. 1 to prepare further standards.
9.2.5 Prepare an approximately 50/50 mix of CCl and CHCl and record amounts of each added. Calculate the volume of this
4 3
mixture needed to prepare one level of standard for calibration, using a calculation similar to that given in 9.3.
9.2.6 Fill the high-pressure syringe (6.7) with approximately the volume of the CCl /CHCl mixture as calculated in 9.3.3. Weigh
4 3
the syringe plus liquid to 6 0.1 mg. Transfer the liquid mixture through the septum into the vapor space of cylinder No. 2. Keep
a finger tightly over the plunger to prevent blow out. Immediately remove and reweigh the syringe to 6 0.1 mg. The difference
between the two weights is the total weight of CCl and CHCl added.
4 3
9.2.7 Shake cylinder No. 2 to assure complete solution of the CCl and CHCl in the chlorine.
4 3
9.2.8 Calculate the added concentration of CCl and CHCl in the spiked standard as indicated in 9.4.
4 3
9.2.9 Prepare at least three standards containing three different levels of CCl and CHCl , bracketing the expected level. Also,
4 3
transfer some of the original chlorine into a sample cylinder without adding CCl or CHCl .
4 3
9.2.10 The long term stability of the calibration standards has not been evaluated.
9.3 Example of amounts of CCl and CHCl to be added to liquid chlorine to produce desired standard:
4 3
9.3.1 Proposed mixture of CCl and CHCl (average density about 1.5 g/mL, or 1.5 mg/μL)mg/μL):
4 3
CCl 47.55 g
CHCl 44.40 g
Total 91.95 g
9.3.2 To prepare 500 g of chlorine with spiked levels of 20 ppm each of CCl and CHCl (total of 40 μg/g), the necessary grams
4 3
(W) of the CCl /CHCl mixture is as follows:
4 3
W 40
5 (1)
500 10
or
W 5 0.020 g5 20 mg (2)
E806 − 21
9.3.3 The necessary volume in μL (V) is then:
W 20
V 5 5 5 13 μL (3)
density 1.5
9.4 Example of calculation of spiked amounts of CCl and CHCl added:
4 3
9.4.1 The weight of mixture added is:
Initial syringe weight with 13 μL 17.6715g
Weight of syringe after transfer 17.6529g
CCl /CHCl added 0.0186g
4 3
9.4.2 The weight of cylinder No. 2:
Weight with chlorine 3575g
Weight empty 3088g
Weight of chlorine 487g
9.4.3 Weight of CCl added:
47.55
~0.0186! 5 0.0096 g (4)
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...