ASTM E981-19
(Test Method)Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals
Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals
SIGNIFICANCE AND USE
3.1 This test method was developed to meet the following criteria:
3.1.1 It provides positive recognition of sensory irritants of widely varying potencies.
3.1.2 It is sufficiently simple to permit the testing of large numbers of materials.
3.1.3 This test method is capable of generating concentration-response curves for purposes of compound comparison.
3.1.4 This test method has good reproducibility.
3.2 This test method can be used for a variety of divergent purposes, including the assessment of comparative irritancy of compounds or formulations and setting interim exposure levels for the workplace (1, 2).2
3.3 It has been shown that for a wide variety of chemicals and mixtures, a perfect rank order correlation exists between the decreases in respiratory rate in mice and subjective reports of sensory irritation in man (1, 3, 4, 5).
3.4 A quantitative estimate of the sensory irritancy of a wide variety of materials can be obtained from concentration-response curves developed using this method (1, 3, 4, 6, 7, 8, 9).
3.5 Although this test method is intended to measure sensory irritation of the nasal mucosa, the cornea is innervated by the same nerve. This animal model will, therefore, allow an estimate of the irritant potential of cosmetic ingredients or other household products to the eye, assuming that they can be aerosolized (10).
3.6 This test method is recommended for setting interim guidelines for exposure of humans to chemicals in the workplace, to assess acute sensory irritation resulting from inadvertent spills of household products, and to assess the comparative irritancy of formulations or materials intended for a variety of uses (see Appendix X2).
FIG. 1 Typical Tracing of Normal Mouse Respiration (Top), and of a“ Moderate” Sensory Irritant Response (Bottom)
Note 1: Taken from Ref. (3).
FIG. 2 Typical Tracing of Normal Mouse Respiration (Top), a Moderate Pulmonary Irritant Response (Center), and an Extreme Pulmonary Irr...
SCOPE
1.1 This laboratory test method provides a rapid means of determining sensory irritant potential of airborne chemicals or mixtures. It may also be used to estimate threshold limit values (TLV) for man. However, it cannot be used to evaluate the relative obnoxiousness of odors.
1.2 This test method is intended as a supplement to, not a replacement for, chronic inhalation studies used to establish allowable human tolerance levels.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard information is given in Section 6.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2019
- Technical Committee
- E50 - Environmental Assessment, Risk Management and Corrective Action
- Drafting Committee
- E50.47 - Biological Effects and Environmental Fate
Relations
- Replaces
ASTM E981-04(2012) - Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals - Effective Date
- 01-Feb-2019
- Effective Date
- 01-Feb-2019
Overview
ASTM E981-19: Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals provides a reliable laboratory procedure to evaluate the sensory irritant potential of airborne chemicals or chemical mixtures. Established by ASTM International, this standard is widely recognized for its application in estimating sensory irritation and predicting acceptable exposure levels for humans, particularly in workplace and industrial environments.
The method measures the reduction in respiratory rate in mice upon exposure to potential sensory irritants in airborne form. This approach enables efficient identification and comparison of chemical irritancy across a range of potencies, supporting the establishment of interim exposure guidelines and safety assessments for a variety of substances. The standard serves as an essential supplement to chronic inhalation studies but is not intended as a replacement for long-term toxicological evaluations.
Key Topics
- Sensory Irritancy Evaluation: The standard supports positive identification of sensory irritants in chemicals and formulations, utilizing a reproducible method with broad applicability.
- Concentration-Response Analysis: By enabling the creation of concentration-response curves, ASTM E981-19 facilitates robust compound comparisons and quantitative irritancy assessment.
- Reproducibility and Simplicity: The outlined test method is designed to be straightforward, enabling high-throughput testing of multiple chemicals or mixtures.
- Comparative and Regulatory Utility: This method underpins setting interim permissible exposure limits and assists in safety decisions for workplace and consumer environments.
- Animal Correlation: The direct correlation between reflex inhibition of respiration in mice and human sensory irritation reports provides a scientific foundation for extrapolation.
- Acute vs. Chronic Assessment: ASTM E981-19 addresses acute sensory irritancy but supports, rather than replaces, comprehensive long-term inhalation studies.
Applications
ASTM E981-19 is applied in various practical scenarios where assessing airborne chemical irritancy is critical to human health and safety:
- Workplace Safety: Establishing interim exposure levels for chemicals in occupational environments, minimizing the risk of sensory irritation in workers.
- Industrial Hygiene: Comparing the sensory irritation potential of new or existing industrial chemicals and formulations to ensure compliance with regulatory guidelines.
- Product Development: Evaluating the irritant potential of cosmetics, household products, and industrial materials, helping manufacturers ensure product safety before market release.
- Emergency Response: Assessing acute sensory irritation risks in cases of accidental chemical spills or releases, enabling quick decisions about evacuation or remediation actions.
- Research and Regulatory Compliance: Supporting toxicological studies and regulatory submissions with quantitative data on the irritancy potential of airborne substances.
Related Standards
For a comprehensive approach to chemical safety, the following standards are often used in conjunction with ASTM E981-19:
- ASTM E1370 – Standard Guide for Air Sampling and Analysis of Volatile Organic Compounds
- ASTM D6332 – Standard Practice for Testing Biological Responses to Municipal Solid Waste Incinerator Ash
- ISO 13344 – Determination of Acute Inhalation Toxicity of Fire Effluents
- OSHA and NIOSH Guidelines – Exposure limits and chemical hazard communication standards
These related standards, along with ASTM E981-19, help organizations build robust occupational safety, industrial hygiene, and chemical risk management programs.
Keywords: ASTM E981-19, sensory irritancy, airborne chemicals, inhalation, workplace exposure, chemical safety, irritant testing, industrial hygiene, concentration-response, animal bioassay, threshold limit values (TLVs), chemical risk assessment.
Buy Documents
ASTM E981-19 - Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals
REDLINE ASTM E981-19 - Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals
Get Certified
Connect with accredited certification bodies for this standard

NSF International
Global independent organization facilitating standards development and certification.

CIS Institut d.o.o.
Personal Protective Equipment (PPE) certification body. Notified Body NB-2890 for EU Regulation 2016/425 PPE.

Kiwa BDA Testing
Building and construction product certification.
Sponsored listings
Frequently Asked Questions
ASTM E981-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Estimating Sensory Irritancy of Airborne Chemicals". This standard covers: SIGNIFICANCE AND USE 3.1 This test method was developed to meet the following criteria: 3.1.1 It provides positive recognition of sensory irritants of widely varying potencies. 3.1.2 It is sufficiently simple to permit the testing of large numbers of materials. 3.1.3 This test method is capable of generating concentration-response curves for purposes of compound comparison. 3.1.4 This test method has good reproducibility. 3.2 This test method can be used for a variety of divergent purposes, including the assessment of comparative irritancy of compounds or formulations and setting interim exposure levels for the workplace (1, 2).2 3.3 It has been shown that for a wide variety of chemicals and mixtures, a perfect rank order correlation exists between the decreases in respiratory rate in mice and subjective reports of sensory irritation in man (1, 3, 4, 5). 3.4 A quantitative estimate of the sensory irritancy of a wide variety of materials can be obtained from concentration-response curves developed using this method (1, 3, 4, 6, 7, 8, 9). 3.5 Although this test method is intended to measure sensory irritation of the nasal mucosa, the cornea is innervated by the same nerve. This animal model will, therefore, allow an estimate of the irritant potential of cosmetic ingredients or other household products to the eye, assuming that they can be aerosolized (10). 3.6 This test method is recommended for setting interim guidelines for exposure of humans to chemicals in the workplace, to assess acute sensory irritation resulting from inadvertent spills of household products, and to assess the comparative irritancy of formulations or materials intended for a variety of uses (see Appendix X2). FIG. 1 Typical Tracing of Normal Mouse Respiration (Top), and of a“ Moderate” Sensory Irritant Response (Bottom) Note 1: Taken from Ref. (3). FIG. 2 Typical Tracing of Normal Mouse Respiration (Top), a Moderate Pulmonary Irritant Response (Center), and an Extreme Pulmonary Irr... SCOPE 1.1 This laboratory test method provides a rapid means of determining sensory irritant potential of airborne chemicals or mixtures. It may also be used to estimate threshold limit values (TLV) for man. However, it cannot be used to evaluate the relative obnoxiousness of odors. 1.2 This test method is intended as a supplement to, not a replacement for, chronic inhalation studies used to establish allowable human tolerance levels. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard information is given in Section 6. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 This test method was developed to meet the following criteria: 3.1.1 It provides positive recognition of sensory irritants of widely varying potencies. 3.1.2 It is sufficiently simple to permit the testing of large numbers of materials. 3.1.3 This test method is capable of generating concentration-response curves for purposes of compound comparison. 3.1.4 This test method has good reproducibility. 3.2 This test method can be used for a variety of divergent purposes, including the assessment of comparative irritancy of compounds or formulations and setting interim exposure levels for the workplace (1, 2).2 3.3 It has been shown that for a wide variety of chemicals and mixtures, a perfect rank order correlation exists between the decreases in respiratory rate in mice and subjective reports of sensory irritation in man (1, 3, 4, 5). 3.4 A quantitative estimate of the sensory irritancy of a wide variety of materials can be obtained from concentration-response curves developed using this method (1, 3, 4, 6, 7, 8, 9). 3.5 Although this test method is intended to measure sensory irritation of the nasal mucosa, the cornea is innervated by the same nerve. This animal model will, therefore, allow an estimate of the irritant potential of cosmetic ingredients or other household products to the eye, assuming that they can be aerosolized (10). 3.6 This test method is recommended for setting interim guidelines for exposure of humans to chemicals in the workplace, to assess acute sensory irritation resulting from inadvertent spills of household products, and to assess the comparative irritancy of formulations or materials intended for a variety of uses (see Appendix X2). FIG. 1 Typical Tracing of Normal Mouse Respiration (Top), and of a“ Moderate” Sensory Irritant Response (Bottom) Note 1: Taken from Ref. (3). FIG. 2 Typical Tracing of Normal Mouse Respiration (Top), a Moderate Pulmonary Irritant Response (Center), and an Extreme Pulmonary Irr... SCOPE 1.1 This laboratory test method provides a rapid means of determining sensory irritant potential of airborne chemicals or mixtures. It may also be used to estimate threshold limit values (TLV) for man. However, it cannot be used to evaluate the relative obnoxiousness of odors. 1.2 This test method is intended as a supplement to, not a replacement for, chronic inhalation studies used to establish allowable human tolerance levels. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Specific hazard information is given in Section 6. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E981-19 is classified under the following ICS (International Classification for Standards) categories: 13.040.01 - Air quality in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E981-19 has the following relationships with other standards: It is inter standard links to ASTM E981-04(2012), ASTM E1302-23. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E981-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E981 − 19
Standard Test Method for
Estimating Sensory Irritancy of Airborne Chemicals
This standard is issued under the fixed designation E981; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope transducers, which sense changes created by inspiration and
expiration.Theamplifiedsignalsaretransmittedtoapolygraph
1.1 This laboratory test method provides a rapid means of
recorder.
determining sensory irritant potential of airborne chemicals or
mixtures.Itmayalsobeusedtoestimatethresholdlimitvalues 2.4 The concentration of airborne irritant that produces a
(TLV) for man. However, it cannot be used to evaluate the 50% decrease in respiratory rate (RD50) is determined from
relative obnoxiousness of odors. the concentration-response curve constructed from the various
data points obtained with a series of concentrations.
1.2 This test method is intended as a supplement to, not a
replacement for, chronic inhalation studies used to establish
3. Significance and Use
allowable human tolerance levels.
3.1 This test method was developed to meet the following
1.3 This standard does not purport to address all of the
criteria:
safety concerns, if any, associated with its use. It is the
3.1.1 It provides positive recognition of sensory irritants of
responsibility of the user of this standard to establish appro-
widely varying potencies.
priate safety, health, and environmental practices and deter-
3.1.2 It is sufficiently simple to permit the testing of large
mine the applicability of regulatory limitations prior to use.
numbers of materials.
Specific hazard information is given in Section 6.
3.1.3 This test method is capable of generating
1.4 This international standard was developed in accor-
concentration-responsecurvesforpurposesofcompoundcom-
dance with internationally recognized principles on standard-
parison.
ization established in the Decision on Principles for the
3.1.4 This test method has good reproducibility.
Development of International Standards, Guides and Recom-
3.2 This test method can be used for a variety of divergent
mendations issued by the World Trade Organization Technical
purposes, including the assessment of comparative irritancy of
Barriers to Trade (TBT) Committee.
compoundsorformulationsandsettinginterimexposurelevels
for the workplace (1, 2).
2. Summary of Test Method
2.1 This test method quantitatively measures irritancy as 3.3 It has been shown that for a wide variety of chemicals
and mixtures, a perfect rank order correlation exists between
indicatedbythereflexinhibitionofrespirationinmiceexposed
the decreases in respiratory rate in mice and subjective reports
to sensory irritants.
of sensory irritation in man (1, 3, 4, 5).
2.2 Four mice are simultaneously exposed to the airborne
3.4 Aquantitativeestimateofthesensoryirritancyofawide
chemical. Usually a sufficient number of groups of animals are
exposed to a geometric series of concentrations so that a variety of materials can be obtained from concentration-
response curves developed using this method (1, 3, 4, 6, 7, 8,
concentration-response curve can be constructed. For simple
preliminary comparisons, however, a single group of four 9).
animals at one concentration will suffice.
3.5 Although this test method is intended to measure sen-
2.3 The mice are placed in a body plethysmograph attached sory irritation of the nasal mucosa, the cornea is innervated by
the same nerve. This animal model will, therefore, allow an
to an exposure chamber so that only the head is exposed to the
test material. The plethysmographs are connected to pressure estimate of the irritant potential of cosmetic ingredients or
other household products to the eye, assuming that they can be
aerosolized (10).
This test method is under the jurisdiction of ASTM Committee E50 on
3.6 This test method is recommended for setting interim
Environmental Assessment, Risk Management and Corrective Action and is the
direct responsibility of Subcommittee E50.47 on Biological Effects and Environ- guidelines for exposure of humans to chemicals in the
mental Fate.
Current edition approved Feb. 1, 2019. Published March 2019. Originally
approved 1984. Last previous edition approved in 2012 as E981–04(2012). DOI: Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
10.1520/E0981-19. this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E981 − 19
workplace, to assess acute sensory irritation resulting from
inadvertent spills of household products, and to assess the
comparativeirritancyofformulationsormaterialsintendedfor
a variety of uses (see Appendix X2).
3.7 This test method will detect irritating effects at concen-
trations far below those at which pathological changes are
observed (9).
NOTE 1—A good overview of the toxicological evaluation of irritant
compounds is given in Ref (8).
4. Apparatus
4.1 The apparatus required to perform this test is listed
below. The basic components for testing any type of material
arethesame.Alistofsuitableapparatusandsuppliersisfound
in Appendix X1.
4.2 Plethysmograph Tubes.
4.3 Exposure Chamber, constructed entirely of glass, with a
NOTE 1—Taken from Ref. (8).
volume of 2.3 L.
FIG. 2 Typical Tracing of Normal Mouse Respiration (Top), a Mod-
4.4 S.T.103/60GroundGlassJoint,thatallowsaccesstothe erate Pulmonary Irritant Response (Center), and an Extreme Pul-
monary Irritant Response (Bottom)
inside of the exposure chamber.
4.5 Perforated Rubber Dental Dam, reinforced with electri-
cal tape.
4.14 Frequency-to-Voltage Converter, operating in the aver-
4.6 Rubber Stoppers.
aging mode instead of the pulse mode. See Appendix X1.7.
4.7 “T”Tube,withatube6cmlongandthe“T”12cmlong.
4.15 VoltageAdditionandDivisionEquipment,toobtainthe
4.8 Vacuum Pump. signal average for four mice.
4.9 Flowmeter. 4.16 Signal Averages.
4.10 Absolute Filter. 4.17 Oscillograph.
4.11 Sodium Carbonate-Activated Charcoal Filter. 4.18 Aerosol Generator.
4.12 Pressure Transducer. 4.19 Timer.
4.13 Polygraph Recorders. 4.20 Control Valve.
NOTE 1—Taken from Ref. (3).
FIG. 1 Typical Tracing of Normal Mouse Respiration (Top), and of a“ Moderate” Sensory Irritant Response (Bottom)
E981 − 19
5. Reagents
5.1 Technical reagents may be used in all tests where
solvents other than water are required.
5.2 Solutions containing 1 to 3% of the test material are
used for comparative studies.
6. Hazards
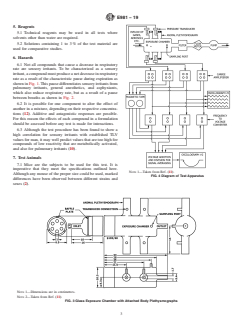
6.1 Not all compounds that cause a decrease in respiratory
rate are sensory irritants. To be characterized as a sensory
irritant,acompoundmustproduceanetdecreaseinrespiratory
rate as a result of the characteristic pause during expiration as
showninFig.1.Thispausedifferentiatessensoryirritantsfrom
pulmonary irritants, general anesthetics, and asphyxiants,
which also reduce respiratory rate, but as a result of a pause
between breaths as shown in Fig. 2.
6.2 It is possible for one component to alter the effect of
another in a mixture, depending on their respective concentra-
tions (12). Additive and antagonistic responses are possible.
For this reason the effects of each compound in a formulation
should be assessed before any test is made for interactions.
6.3 Although the test procedure has been found to show a
high correlation for sensory irritants with established TLV
values for man, it may well predict values that are too high for
compounds of low reactivity that are metabolically activated,
and also for pulmonary irritants (10).
7. Test Animals
7.1 Mice are the subjects to be used for this test. It is
imperative that they meet the specifications outlined here.
NOTE 1—Taken from Ref. (11).
Although any mouse of the proper size could be used, marked
FIG. 4 Diagram of Test Apparatus
differences have been observed between different strains and
sexes (2).
NOTE 1—Dimensions are in centimetres.
NOTE 2—Taken from Ref. (11).
FIG. 3 Glass Exposure Chamber with Attached Body Plethysmographs
E981 − 19
7.1.1 Male Swiss-Webster mice shall be used as the test
subjects.
7.1.2 Only animals weighing between 22 and 28 g may be
used. Smaller mice might be able to crawl into the exposure
chamber,whilelargeronesmaynotbeabletobreathenormally
in the apparatus.
7.1.3 The same system can be used with guinea pigs or rats
with an airflow of 2 L/min when using head dome (9).
8. Preparation of Apparatus
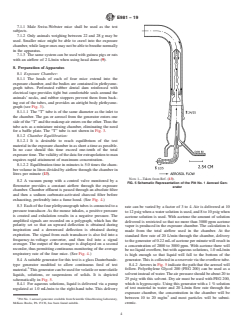
8.1 Exposure Chamber:
8.1.1 The heads of each of four mice extend into the
exposure chamber, and the bodies are contained in plethysmo-
graph tubes. Perforated rubber dental dam reinforced with
electrical tape provides tight but comfortable seals around the
animals’ necks, and rubber stoppers prevent them from back-
ing out of the tubes, and provides an airtight body plethysmo-
graph (see Fig. 3).
8.1.1.1 The “T” tube is of the same diameter as the inlet to
the chamber. The gas or aerosol from the generator enters one
sideofthe“T”andthemakeupairentersontheother.Thusthe
tube acts as a miniature mixing chamber, eliminating the need
for a baffle plate. The “T” tube is not shown in Fig. 3.
8.1.2 Chamber Equilibration:
8.1.2.1 It is desirable to reach equilibrium of the test
materialintheexposurechamberinasshortatimeaspossible.
In no case should this time exceed one-tenth of the total
exposuretime.Thevalidityofthedataforextrapolationtoman
requires rapid attainment of maximum concentration.
8.1.2.2 Equilibration time in minutes is 5.0 times the cham-
ber volume in litres divided by airflow through the chamber in
litres per minute (13).
NOTE 1—Taken from Ref. (13).
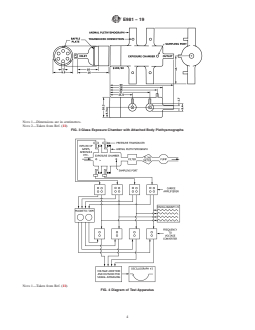
8.2 A vacuum pump with a control valve monitored by a
FIG. 5 Schematic Representation of the Pitt No. 1 Aerosol Gen-
flowmeter provides a constant airflow through the exposure
erator
chamber. Chamber effluent is passed through an absolute filter
and then a sodium carbonate-activated charcoal filter before
exhausting, preferably into a fume hood. (See Fig. 4.)
8.3 Eachofthefourplethysmographtubesisconnectedtoa
rate can be varied by a factor of 3 to 4. Air is delivered at 10
pressure transducer. As the mouse inhales, a positive pressure
to12psigwhenawatersolutionisused,and8to10psigwhen
is created and exhalation results in a negative pressure. The
acetone solution is used. With acetone the amount of solution
amplified signals are recorded on a polygraph, which has the
delivered is restricted so that no more than 3000 ppm acetone
polarity set so that an upward deflection is obtained during
vapor is produced in the exposure chamber. The calculation is
inspiration and a downward deflection is obtained during
made from the total airflow used in the chamber. At the
expiration. The signal from each transducer is also fed into a
standard flow rate of 20 L/min through the chamber, delivery
frequency-to-voltage converter, and then fed into a signal
tothegeneratorof0.22mLofacetoneperminutewillresultin
averager. The output of the averager is displayed on a second
a concentration of 2800 to 3000 ppm. With acetone there will
recorder, thus permitting continuous monitoring of the average
be no liquid overflow, but with aqueous solutions, 1.0 mL/min
respiratory rate of the four mice. (See Fig. 4.)
is high enough so that liquid will fall to the bottom of the
generator.Thisiscollectedinareservoirviatheoverflowtube.
8.4 Asuitable generator for this test is a glass Dautrebande-
8.4.2 Arrows in Fig. 5 indicate the path that the aerosol will
type generator modified to allow continuous feed of test
follow. Polyethylene Glycol 200 (PEG 200) can be used as a
material. Thisgeneratorcanbeusedforvolatileornonvolatile
solventinsteadofwater.Theairpressureshouldbeabout20to
liquids, solutions, or suspensions of solids. It is depicted
25 psig with this solvent. Dry air must be used with PEG 200,
schematically in Fig. 5.
whichishygroscopic.Usingthisgeneratorwitha1%solution
8.4.1 For aqueous solutions, liquid is delivered via a pump
of test material in water and 20 L/min flow rate through the
regulated at 1.0 mL/min to the right-hand tube. This delivery
exposure chamber, the concentration in the chamber will be
between 10 to 20 mg/m and most particles will be submi-
PittNo.1aerosolgeneratoravailablefromScientificGlassblowingLaboratory,
McKees Rocks, PA 15136, has been found suitable. cronic.
E981 − 19
NOTE 1—Taken from Ref. (11).
FIG. 6 Typical Tracing Obtained from a Single Animal Prior to and During Exposure to a Sensory Irritant (Top). Average
Respiratory Rate of Four Mice During Course of Exposure (Bottom)
8.4.3 The Dautrebande-type generator can also be used to analysis is adequate, or an infrared analyzer or gas chromato-
vaporize liquids for exposure of animals to vapors. For this graphic analysis can be used.
purpose, the liquid is delivered at a known rate by a regulated
8.7 Gases are delivered directly into the exposure chamber
pump and airflow is set at 10 to 20 psig. For liquids of lower
via an appropriate flowmeter.
vapor pressure, heating tape can be used around the generator
8.8 With the exception of the exposure chamber which is
to increase vaporization efficiency. For aerosols or vapors
essentially a unique piece of apparatus, other parts can be
likely to oxidize rapidly in air, dry nitrogen should be used
substituted by similar equipment.Also, minicomputers can be
instead of air. When this is done, pure oxygen is added to the
used to replace the frequency-to-voltage converter and signal-
chamber airflow to maintain 18 to 20% O in the exposure
averaging device. The magnetic tape is not required, and a
chamber. When suspensions are to be tested, the suspended
four-trace oscilloscope with storage capability can replace
material must be very fine to prevent clogging of the tip on the
oscillograph No. 1.
generator. Although larger tips can be used if required, a
degradation of aerosolizing performance will result from their
9. Sample Preparation
use.
9.1 Because of the large variety of chemicals and formula-
8.5 Tostartandstoptestmaterialgeneration,atimerandan
tions that can be tested by this procedure, and the tremendous
associated control valve are needed in conjunction with the differences in irritant potential between them, no specific
aerosol generator.
stipulation for sample preparation can be made. The only
requirement for concentration is that the levels to be tested are
8.6 When using water or acetone a “dry” particle will be
spaced at even logarithmic intervals to allow good
produced, since both solvents will evaporate. However, PEG
concentration-response curves to be generated from the data
200 will not evaporate and a liquid droplet is obtained. Mass
obtained. The information provided in the succeeding para-
concentration in the chamber should be obtained by sampling
graphsofthissectionisthereforeintendedforgeneralguidance
on filters and weighing on an appropriate balance. A better
only.
method, but one not required in a screening experiment, is
appropriate chemical analysis. When acetone is used, its 9.2 For solids and nonvolatile liquids, solutions are pre-
concentrationinthechambershouldbeverified.Indicatortube pared in an appropriate solvent.Water and polyethylene glycol
E981 − 19
NOTE1—Decreasesinrespiratoryrateof12to20%aregradedasslight
responses.
NOTE 2—Taken from Ref. (1).
FIG. 7 Typical Tracings with Intensity of the Reaction Graded
as Slight
200 (PEG 200) are the most commonly used for this purpose, 10.2.1 To assure that a generator is performing correctly,
although 0.1 N HCl, 0.1 N NaOH, and acetone can also be test solutions of 1% NaCl in water and of undiluted PEG 200
used. In the case of acetone, which is a mild irritant, the should be tested. Start the generator at the pressure recom-
concentration in the chamber should be kept below 3000 ppm mended for the particular solvent, and shine a light beam
to avoid irritation from the solvent. acrossthegeneratoroutlet.Aconstantflowofparticlesmustbe
visible. Water without solute will evaporate too quickly to be
9.3 As an indication of concentrations to be expected, 1%
observed, and therefore should not be used for this test.
aqueousbasic,oracidicsolutionsproduceconcentrationsof10
to20mg/m atanairflowof20L/minintheexposurechamber.
10.3 Plethysmograph—The plethysmograph chambers re-
Polyethyleneglycol200solutionswillproduceaconcentration
quire minimal calibration to assure equivalence of response
of 40 to 50 mg/m of the solute under similar circumstances.
from all four chambers. All that is required is that a signal of
sufficientamplitudebedisplayedontherecordingpolygraphto
9.4 Gases shall be mixed with room air to produce the
discern the respiratory pattern of each animal. The amplitude
desired concentrations.
should be about the same for each animal, but this is not
10. Calibration
critical.
10.1 Inthistestmethod,threepartsoftheequipmentrequire
10.4 Flowmeter—The flowmeter must be calibrated so that
calibration.Oncethesecalibrationshavebeenmade,recalibra-
desired flow rates are uniformly maintained. These rates are
tion is not necessary for the conditions previously used unless
easily determined for various readings on the flowmeter, and
the apparatus is disassembled.
will remain constant as long as the air supply is constant.
Oil-washed air from a compressed gas cylinder in conjunction
10.2 Generator—Determine the particle size of the aerosol
withacalibratedgagefromareputablemanufacturershouldbe
droplets emitted by each generator for each type of solution or
used as a source of air for the generators.
suspension to assure the validity of the tests. A 1% aqueous
solution under 10 to 12 psig will produce particles of aerody-
11. Pretest Conditioning
namic equivalent diameter of 0.6 to 0.8 µm, with a geometric
standarddeviationof2.0to2.5.WithPEG200atapressureof
11.1 Itisessentialthathealthyanimalsareusedforthistest.
20 to 25 psig, the particle size will be 1.0 to 2.0 µm, with a
In order to assure that this is so, it is necessary to hold and to
similargeometricdeviation.Particlesizeanalysismaybemade
observe them for 7 days prior to use.
using an Anderson mini-impactor or other appropriate tech-
nique. 11.2 The mice may be gang-housed if desired.
E981 − 19
NOTE 1—Decreases in respiratory rate of 20 to 50% are graded as moderate responses.
NOTE 2—Taken from Ref. (1).
FIG. 8 Typical Tracings with Intensity of the Reaction Graded as Moderate
11.2.1 Thoroughly clean and sanitize the cages prior to use, 12.1.3 Standard airflow rate through the exposure chamber
and provide ground corncob or similar bedding. shall be 20 L/min.
12.1.4 Test compound shall be generated at a level of 100
11.3 Individually identify each animal.
mg/m of air.
11.4 Take weights of the mice at the time the mice are
12.1.5 Exposure time shall be 30 min. This lengthy expo-
caged, and again just prior to the test to assure reasonable
sure time has been found necessary to detect slow-acting
weightgain.Itisalsoadvisabletonotefoodconsumptionasan
irritants such as isocyanates, particularly at low concentrations
additional check on animal health.
(15).
11.5 Maintain the laboratory animal housing environment
12.1.6 A 10-minute recovery period shall be recorded after
accordingtoacceptableanimalcareaccreditationrequirements
the aerosol exposure is completed.
(14). Significant deviations therefrom must be noted and
12.2 Almost any desired change can be made in these
reported.
parameters to serve a given need. The limitations on such
12. Selection of Test Parameters changes are noted below:
12.2.1 The mice used must be as stipulated.
12.1 For the purpose of comparing a variety of sensory
12.2.2 No change can be made in acclimation time.
irritants, the test parameters listed below have been found
12.2.3 Airflowratescanbevariedfrom16to100L/minfor
desirable:
aerosols, and 2 to 100 L/min for gases.
12.1.1 Male, SwissWebster mice weighing between 22 and
28 g shall be used as test subjects. 12.2.4 Exposure time may vary from 3 to 180 min. In order
12.1.2 A ten-minute acclimation period, in which the mice to assure that a maximum response has been obtained, a
are in the plethysmograph tubes, but breathing room air, is to plateau of response for at least 1 min must be obtained. The
be used. minimum decrease in respiratory rate considered significant is
E981
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E981 − 04 (Reapproved 2012) E981 − 19
Standard Test Method for
Estimating Sensory Irritancy of Airborne Chemicals
This standard is issued under the fixed designation E981; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This laboratory test method provides a rapid means of determining sensory irritant potential of airborne chemicals or
mixtures. It may also be used to estimate threshold limit values (TLV) for man. However, it cannot be used to evaluate the relative
obnoxiousness of odors.
1.2 This test method is intended as a supplement to, not a replacement for, chronic inhalation studies used to establish allowable
human tolerance levels.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. Specific hazard information is given in Section 6.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Summary of Test Method
2.1 This test method quantitatively measures irritancy as indicated by the reflex inhibition of respiration in mice exposed to
sensory irritants.
2.2 Four mice are simultaneously exposed to the airborne chemical. Usually a sufficient number of groups of animals are
exposed to a geometric series of concentrations so that a concentration-response curve can be constructed. For simple preliminary
comparisons, however, a single group of four animals at one concentration will suffice.
2.3 The mice are placed in a body plethysmograph attached to an exposure chamber so that only the head is exposed to the test
material. The plethysmographs are connected to pressure transducers, which sense changes created by inspiration and expiration.
The amplified signals are transmitted to a polygraph recorder.
2.4 The concentration of airborne irritant that produces a 50 % decrease in respiratory rate (RD50) is determined from the
concentration-response curve constructed from the various data points obtained with a series of concentrations.
3. Significance and Use
3.1 This test method was developed to meet the following criteria:
3.1.1 It provides positive recognition of sensory irritants of widely varying potencies.
3.1.2 It is sufficiently simple to permit the testing of large numbers of materials.
3.1.3 This test method is capable of generating concentration-response curves for purposes of compound comparison.
3.1.4 This test method has good reproducibility.
3.2 This test method can be used for a variety of divergent purposes, including the assessment of comparative irritancy of
compounds or formulations and setting interim exposure levels for the workplace (1, 2).
3.3 It has been shown that for a wide variety of chemicals and mixtures, a perfect rank order correlation exists between the
decreases in respiratory rate in mice and subjective reports of sensory irritation in man (1, 3, 4, 5).
This test method is under the jurisdiction of ASTM Committee E50 on Environmental Assessment, Risk Management and Corrective Action and is the direct
responsibility of Subcommittee E50.47 on Biological Effects and Environmental Fate.
Current edition approved Dec. 1, 2012Feb. 1, 2019. Published December 2012March 2019. Originally approved 1984. Last previous edition approved in 20042012 as
E981 – 04.E981 – 04(2012). DOI: 10.1520/E0981-04R12.10.1520/E0981-19.
The boldface numbers in parentheses refer to the list of references at the end of this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E981 − 19
3.4 A quantitative estimate of the sensory irritancy of a wide variety of materials can be obtained from concentration-response
curves developed using this method (1, 3, 4, 6, 7, 8, 9).
3.5 Although this test method is intended to measure sensory irritation of the nasal mucosa, the cornea is innervated by the same
nerve. This animal model will, therefore, allow an estimate of the irritant potential of cosmetic ingredients or other household
products to the eye, assuming that they can be aerosolized (10).
3.6 This test method is recommended for setting interim guidelines for exposure of humans to chemicals in the workplace, to
assess acute sensory irritation resulting from inadvertent spills of household products, and to assess the comparative irritancy of
formulations or materials intended for a variety of uses (see Appendix X2).
3.7 This test method will detect irritating effects at concentrations far below those at which pathological changes are observed
(9).
NOTE 1—A good overview of the toxicological evaluation of irritant compounds is given in Ref (8).
4. Apparatus
4.1 The apparatus required to perform this test is listed below. The basic components for testing any type of material are the
same. A list of suitable apparatus and suppliers is found in Appendix X1.
4.2 Plethysmograph Tubes.
4.3 Exposure Chamber, constructed entirely of glass, with a volume of 2.3 L.
4.4 S.T.103/60 Ground Glass Joint, that allows access to the inside of the exposure chamber.
4.5 Perforated Rubber Dental Dam, reinforced with electrical tape.
4.6 Rubber Stoppers.
4.7 “T” Tube, with a tube 6 cm long and the “T” 12 cm long.
4.8 Vacuum Pump.
4.9 Flowmeter.
4.10 Absolute Filter.
4.11 Sodium Carbonate-Activated Charcoal Filter.
4.12 Pressure Transducer.
4.13 Polygraph Recorders.
4.14 Frequency-to-Voltage Converter, operating in the averaging mode instead of the pulse mode. See Appendix X1.7.
4.15 Voltage Addition and Division Equipment, to obtain the signal average for four mice.
NOTE 1—Taken from Ref. (3).
FIG. 1 Typical Tracing of Normal Mouse Respiration (Top), and of a“ Moderate” Sensory Irritant Response (Bottom)
E981 − 19
NOTE 1—Taken from Ref. (8).
FIG. 2 Typical Tracing of Normal Mouse Respiration (Top), a Moderate Pulmonary Irritant Response (Center), and an Extreme Pulmo-
nary Irritant Response (Bottom)
4.16 Signal Averages.
4.17 Oscillograph.
4.18 Aerosol Generator.
4.19 Timer.
4.20 Control Valve.
5. Reagents
5.1 Technical reagents may be used in all tests where solvents other than water are required.
5.2 Solutions containing 1 to 3 % of the test material are used for comparative studies.
6. Hazards
6.1 Not all compounds that cause a decrease in respiratory rate are sensory irritants. To be characterized as a sensory irritant,
a compound must produce a net decrease in respiratory rate as a result of the characteristic pause during expiration as shown in
Fig. 1. This pause differentiates sensory irritants from pulmonary irritants, general anesthetics, and asphyxiants, which also reduce
respiratory rate, but as a result of a pause between breaths as shown in Fig. 2.
6.2 It is possible for one component to alter the effect of another in a mixture, depending on their respective concentrations (12).
Additive and antagonistic responses are possible. For this reason the effects of each compound in a formulation should be assessed
before any test is made for interactions.
6.3 Although the test procedure has been found to show a high correlation for sensory irritants with established TLV values for
man, it may well predict values that are too high for compounds of low reactivity that are metabolically activated, and also for
pulmonary irritants (10).
7. Test Animals
7.1 Mice are the subjects to be used for this test. It is imperative that they meet the specifications outlined here. Although any
mouse of the proper size could be used, marked differences have been observed between different strains and sexes (2).
7.1.1 Male Swiss-Webster mice shall be used as the test subjects.
7.1.2 Only animals weighing between 22 and 28 g may be used. Smaller mice might be able to crawl into the exposure chamber,
while larger ones may not be able to breathe normally in the apparatus.
7.1.3 The same system can be used with guinea pigs or rats with an airflow of 2 L/min when using head dome (9).
8. Preparation of Apparatus
8.1 Exposure Chamber:
E981 − 19
NOTE 1—Dimensions are in centimetres.
NOTE 2—Taken from Ref. (11).
FIG. 3 Glass Exposure Chamber with Attached Body Plethysmographs
NOTE 1—Taken from Ref. (11).
FIG. 4 Diagram of Test Apparatus
E981 − 19
8.1.1 The heads of each of four mice extend into the exposure chamber, and the bodies are contained in plethysmograph tubes.
Perforated rubber dental dam reinforced with electrical tape provides tight but comfortable seals around the animals’ necks, and
rubber stoppers prevent them from backing out of the tubes, and provides an airtight body plethysmograph (see Fig. 3).
8.1.1.1 The “T” tube is of the same diameter as the inlet to the chamber. The gas or aerosol from the generator enters one side
of the “T” and the makeup air enters on the other. Thus the tube acts as a miniature mixing chamber, eliminating the need for a
baffle plate. The “T” tube is not shown in Fig. 3.
8.1.2 Chamber Equilibration:
8.1.2.1 It is desirable to reach equilibrium of the test material in the exposure chamber in as short a time as possible. In no case
should this time exceed one-tenth of the total exposure time. The validity of the data for extrapolation to man requires rapid
attainment of maximum concentration.
8.1.2.2 Equilibration time in minutes is 5.0 times the chamber volume in litres divided by airflow through the chamber in litres
per minute (13).
8.2 A vacuum pump with a control valve monitored by a flowmeter provides a constant airflow through the exposure chamber.
Chamber effluent is passed through an absolute filter and then a sodium carbonate-activated charcoal filter before exhausting,
preferably into a fume hood. (See Fig. 4.)
8.3 Each of the four plethysmograph tubes is connected to a pressure transducer. As the mouse inhales, a positive pressure is
created and exhalation results in a negative pressure. The amplified signals are recorded on a polygraph, which has the polarity
set so that an upward deflection is obtained during inspiration and a downward deflection is obtained during expiration. The signal
from each transducer is also fed into a frequency-to-voltage converter, and then fed into a signal averager. The output of the
averager is displayed on a second recorder, thus permitting continuous monitoring of the average respiratory rate of the four mice.
(See Fig. 4.)
8.4 A suitable generator for this test is a glass Dautrebande-type generator modified to allow continuous feed of test material.
This generator can be used for volatile or nonvolatile liquids, solutions, or suspensions of solids. It is depicted schematically in
Fig. 5.
8.4.1 For aqueous solutions, liquid is delivered via a pump regulated at 1.0 mL/min to the right-hand tube. This delivery rate
can be varied by a factor of 3 to 4. Air is delivered at 10 to 12 psig when a water solution is used, and 8 to 10 psig when acetone
solution is used. With acetone the amount of solution delivered is restricted so that no more than 3000 ppm acetone vapor is
produced in the exposure chamber. The calculation is made from the total airflow used in the chamber. At the standard flow rate
of 20 L/min through the chamber, delivery to the generator of 0.22 mL of acetone per minute will result in a concentration of 2800
to 3000 ppm. With acetone there will be no liquid overflow, but with aqueous solutions, 1.0 mL/min is high enough so that liquid
will fall to the bottom of the generator. This is collected in a reservoir via the overflow tube.
8.4.2 Arrows in Fig. 5 indicate the path that the aerosol will follow. Polyethylene Glycol 200 (PEG 200) can be used as a solvent
instead of water. The air pressure should be about 20 to 25 psig with this solvent. Dry air must be used with PEG 200, which is
hygroscopic. Using this generator with a 1 % solution of test material in water and 20 L/min flow rate through the exposure
chamber, the concentration in the chamber will be between 10 to 20 mg/m and most particles will be submicronic.
8.4.3 The Dautrebande-type generator can also be used to vaporize liquids for exposure of animals to vapors. For this purpose,
the liquid is delivered at a known rate by a regulated pump and airflow is set at 10 to 20 psig. For liquids of lower vapor pressure,
heating tape can be used around the generator to increase vaporization efficiency. For aerosols or vapors likely to oxidize rapidly
in air, dry nitrogen should be used instead of air. When this is done, pure oxygen is added to the chamber airflow to maintain 18
to 20 % O in the exposure chamber. When suspensions are to be tested, the suspended material must be very fine to prevent
clogging of the tip on the generator. Although larger tips can be used if required, a degradation of aerosolizing performance will
result from their use.
8.5 To start and stop test material generation, a timer and an associated control valve are needed in conjunction with the aerosol
generator.
8.6 When using water or acetone a “dry” particle will be produced, since both solvents will evaporate. However, PEG 200 will
not evaporate and a liquid droplet is obtained. Mass concentration in the chamber should be obtained by sampling on filters and
weighing on an appropriate balance. A better method, but one not required in a screening experiment, is appropriate chemical
analysis. When acetone is used, its concentration in the chamber should be verified. Indicator tube analysis is adequate, or an
infrared analyzer or gas chromatographic analysis can be used.
8.7 Gases are delivered directly into the exposure chamber via an appropriate flowmeter.
8.8 With the exception of the exposure chamber which is essentially a unique piece of apparatus, other parts can be substituted
by similar equipment. Also, minicomputers can be used to replace the frequency-to-voltage converter and signal-averaging device.
The magnetic tape is not required, and a four-trace oscilloscope with storage capability can replace oscillograph No. 1.
Pitt No. 1 aerosol generator available from Scientific Glassblowing Laboratory, McKees Rocks, PA 15136, has been found suitable.
E981 − 19
NOTE 1—Taken from Ref. (13).
FIG. 5 Schematic Representation of the Pitt No. 1 Aerosol Generator
9. Sample Preparation
9.1 Because of the large variety of chemicals and formulations that can be tested by this procedure, and the tremendous
differences in irritant potential between them, no specific stipulation for sample preparation can be made. The only requirement
for concentration is that the levels to be tested are spaced at even logarithmic intervals to allow good concentration-response curves
to be generated from the data obtained. The information provided in the succeeding paragraphs of this section is therefore intended
for general guidance only.
9.2 For solids and nonvolatile liquids, solutions are prepared in an appropriate solvent. Water and polyethylene glycol 200 (PEG
200) are the most commonly used for this purpose, although 0.1 N HCl, 0.1 N NaOH, and acetone can also be used. In the case
of acetone, which is a mild irritant, the concentration in the chamber should be kept below 3000 ppm to avoid irritation from the
solvent.
9.3 As an indication of concentrations to be expected, 1 % aqueous basic, or acidic solutions produce concentrations of 10 to
20 mg/m at an airflow of 20 L/min in the exposure chamber. Polyethylene glycol 200 solutions will produce a concentration of
40 to 50 mg/m of the solute under similar circumstances.
9.4 Gases shall be mixed with room air to produce the desired concentrations.
10. Calibration
10.1 In this test method, three parts of the equipment require calibration. Once these calibrations have been made, recalibration
is not necessary for the conditions previously used unless the apparatus is disassembled.
10.2 Generator—Determine the particle size of the aerosol droplets emitted by each generator for each type of solution or
suspension to assure the validity of the tests. A 1 % aqueous solution under 10 to 12 psig will produce particles of aerodynamic
equivalent diameter of 0.6 to 0.8 μm, with a geometric standard deviation of 2.0 to 2.5. With PEG 200 at a pressure of 20 to 25
E981 − 19
NOTE 1—Taken from Ref. (11).
FIG. 6 Typical Tracing Obtained from a Single Animal Prior to and During Exposure to a Sensory Irritant (Top). Average
Respiratory Rate of Four Mice During Course of Exposure (Bottom)
psig, the particle size will be 1.0 to 2.0 μm, with a similar geometric deviation. Particle size analysis may be made using an
Anderson mini-impactor or other appropriate technique.
10.2.1 To assure that a generator is performing correctly, test solutions of 1 % NaCl in water and of undiluted PEG 200 should
be tested. Start the generator at the pressure recommended for the particular solvent, and shine a light beam across the generator
outlet. A constant flow of particles must be visible. Water without solute will evaporate too quickly to be observed, and therefore
should not be used for this test.
10.3 Plethysmograph—The plethysmograph chambers require minimal calibration to assure equivalence of response from all
four chambers. All that is required is that a signal of sufficient amplitude be displayed on the recording polygraph to discern the
respiratory pattern of each animal. The amplitude should be about the same for each animal, but this is not critical.
10.4 Flowmeter—The flowmeter must be calibrated so that desired flow rates are uniformly maintained. These rates are easily
determined for various readings on the flowmeter, and will remain constant as long as the air supply is constant. Oil-washed air
from a compressed gas cylinder in conjunction with a calibrated gage from a reputable manufacturer should be used as a source
of air for the generators.
11. Pretest Conditioning
11.1 It is essential that healthy animals are used for this test. In order to assure that this is so, it is necessary to hold and to
observe them for 7 days prior to use.
11.2 The mice may be gang-housed if desired.
11.2.1 Thoroughly clean and sanitize the cages prior to use, and provide ground corncob or similar bedding.
11.3 Individually identify each animal.
11.4 Take weights of the mice at the time the mice are caged, and again just prior to the test to assure reasonable weight gain.
It is also advisable to note food consumption as an additional check on animal health.
11.5 Maintain the laboratory animal housing environment according to acceptable animal care accreditation requirements (14).
Significant deviations therefrom must be noted and reported.
E981 − 19
NOTE 1—Decreases in respiratory rate of 12 to 20 % are graded as slight
responses.
NOTE 2—Taken from Ref. (1).
FIG. 7 Typical Tracings with Intensity of the Reaction Graded
as Slight
12. Selection of Test Parameters
12.1 For the purpose of comparing a variety of sensory irritants, the test parameters listed below have been found desirable:
12.1.1 Male, Swiss Webster mice weighing between 22 and 28 g shall be used as test subjects.
12.1.2 A ten-minute acclimation period, in which the mice are in the plethysmograph tubes, but breathing room air, is to be used.
12.1.3 Standard airflow rate through the exposure chamber shall be 20 L/min.
12.1.4 Test compound shall be generated at a level of 100 mg/m of air.
12.1.5 Exposure time shall be 30 min. This lengthy exposure time has been found necessary to detect slow-acting irritants such
as isocyanates, particularly at low concentrations (15).
12.1.6 A 10-minute recovery period shall be recorded after the aerosol exposure is completed.
12.2 Almost any desired change can be made in these parameters to serve a given need. The limitations on such changes are
noted below:
12.2.1 The mice used must be as stipulated.
12.2.2 No change can be made in acclimation time.
12.2.3 Airflow rates can be varied from 16 to 100 L/min for aerosols, and 2 to 100 L/min for gases.
12.2.4 Exposure time may vary from 3 to 180 min. In order to assure that a maximum response has been obtained, a plateau
of response for at least 1 min must be obtained. The minimum decrease in respiratory rate considered significant is 12 %, provided
that it is either sustained for 3 min or reproducible in three groups of animals for at least 1 min. In no case will the decrease in
respiratory rate exceed 80
...





Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...