ASTM D6503-24
(Test Method)Standard Test Method for Enterococci in Water Using Enterolert
Standard Test Method for Enterococci in Water Using Enterolert
SIGNIFICANCE AND USE
5.1 This test provides an easy and reliable method for the detection of enterococci in water within 24 h. For recreational water (fresh and marine) testing is performed to insure areas are safe for swimming. Enterolert also can be used for testing bottled water, wastewater, ground water, and drinking water.
SCOPE
1.1 This test method covers a simple procedure for the detection of enterococci in water and wastewater. It is based on IDEXX’s patented Defined Substrate Technology (DST).2 This product, Enterolert, utilizes a nutrient indicator that fluoresces when metabolized. It can detect these bacteria at one most probable number (MPN)/100 mL within 24 h. The presence of this microorganism in water is an indication of fecal contamination and the possible presence of enteric pathogens.
1.2 This test method can be used successfully with drinking water, source water, recreational (fresh and marine) water, wastewater, and bottled water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2024
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.24 - Water Microbiology
Relations
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Apr-2024
- Effective Date
- 01-Apr-2024
Overview
ASTM D6503-24 - Standard Test Method for Enterococci in Water Using Enterolert provides a practical, standardized approach for the detection and quantification of enterococci bacteria in various water samples. Developed by ASTM International, this test method employs IDEXX’s patented Defined Substrate Technology (DST) with the Enterolert reagent, which fluoresces in the presence of enterococci. Suitable for both presence-absence and quantitative analysis, this standard ensures efficient and reliable monitoring of water quality for public safety and regulatory compliance.
Key Topics
- Purpose and Utility: The method is designed for fast (within 24 hours), easy, and accurate detection of enterococci – an important indicator of fecal contamination and potential pathogenic organisms in water.
- Applicable Water Types: The standard is suitable for testing:
- Drinking water
- Source water
- Recreational water (fresh and marine)
- Bottled water
- Wastewater
- Groundwater
- Methodology:
- Uses Enterolert reagent in combination with nutrient indicators that emit fluorescence under UV light when metabolized by enterococci.
- Offers Minimum Detection Limit of 1 most probable number (MPN)/100 mL.
- Results can be reported as “presence-absence” or as MPN using Quanti-Tray systems for enumeration.
- Safety and Quality Control:
- Recommends standard laboratory practices and detailed quality control measures for sample handling and test accuracy.
- Users are responsible for validating this method in matrices not previously tested.
Applications
ASTM D6503-24 has broad applications across multiple sectors with emphasis on protecting public health and supporting regulatory frameworks:
- Recreational Water Quality: Ensures swimming areas (lakes, rivers, beaches, pools) are safe from microbiological contamination.
- Drinking and Bottled Water Testing: Rapidly confirms the microbiological safety of potable water and bottled water supplies.
- Wastewater and Source Water Monitoring: Assists in compliance with environmental regulations by providing data on effluent quality and watershed health.
- Emergency Response: Offers a fast turn-around (24 hours) for the detection of fecal contamination during natural disasters or infrastructure failures.
- Industrial Testing: Supports industries needing to monitor outgoing water to meet environmental discharge standards.
Related Standards
For comprehensive water testing and laboratory practices, consider the following related ASTM standards:
- ASTM D1129: Terminology Relating to Water
- ASTM D1193: Specification for Reagent Water
- ASTM D2777: Practice for Determination of Precision and Bias of Applicable Test Methods
- ASTM D3370: Practices for Sampling Water from Flowing Process Streams
Practical Value
Adhering to ASTM D6503-24 allows water testing laboratories, utilities, and regulatory agencies to:
- Achieve consistent and repeatable results in enterococci detection
- Satisfy public health and regulatory requirements efficiently
- Make timely decisions about the safety of water resources
- Leverage standardized, internationally recognized methodologies
Keywords
bottled water, drinking water, enterococci, Enterolert, most probable number, presence-absence, Quanti-Tray, recreational water, source water, wastewater, water quality testing, ASTM D6503-24
Buy Documents
ASTM D6503-24 - Standard Test Method for Enterococci in Water Using Enterolert
REDLINE ASTM D6503-24 - Standard Test Method for Enterococci in Water Using Enterolert
Frequently Asked Questions
ASTM D6503-24 is a standard published by ASTM International. Its full title is "Standard Test Method for Enterococci in Water Using Enterolert". This standard covers: SIGNIFICANCE AND USE 5.1 This test provides an easy and reliable method for the detection of enterococci in water within 24 h. For recreational water (fresh and marine) testing is performed to insure areas are safe for swimming. Enterolert also can be used for testing bottled water, wastewater, ground water, and drinking water. SCOPE 1.1 This test method covers a simple procedure for the detection of enterococci in water and wastewater. It is based on IDEXX’s patented Defined Substrate Technology (DST).2 This product, Enterolert, utilizes a nutrient indicator that fluoresces when metabolized. It can detect these bacteria at one most probable number (MPN)/100 mL within 24 h. The presence of this microorganism in water is an indication of fecal contamination and the possible presence of enteric pathogens. 1.2 This test method can be used successfully with drinking water, source water, recreational (fresh and marine) water, wastewater, and bottled water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test provides an easy and reliable method for the detection of enterococci in water within 24 h. For recreational water (fresh and marine) testing is performed to insure areas are safe for swimming. Enterolert also can be used for testing bottled water, wastewater, ground water, and drinking water. SCOPE 1.1 This test method covers a simple procedure for the detection of enterococci in water and wastewater. It is based on IDEXX’s patented Defined Substrate Technology (DST).2 This product, Enterolert, utilizes a nutrient indicator that fluoresces when metabolized. It can detect these bacteria at one most probable number (MPN)/100 mL within 24 h. The presence of this microorganism in water is an indication of fecal contamination and the possible presence of enteric pathogens. 1.2 This test method can be used successfully with drinking water, source water, recreational (fresh and marine) water, wastewater, and bottled water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6503-24 is classified under the following ICS (International Classification for Standards) categories: 07.100.20 - Microbiology of water. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6503-24 has the following relationships with other standards: It is inter standard links to ASTM D6503-19, ASTM D8429-21, ASTM D4412-19. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6503-24 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6503 − 24
Standard Test Method for
1,2
Enterococci in Water Using Enterolert
This standard is issued under the fixed designation D6503; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This test method covers a simple procedure for the 2.1 ASTM Standards:
detection of enterococci in water and wastewater. It is based on D1129 Terminology Relating to Water
IDEXX’s patented Defined Substrate Technology (DST). This D1193 Specification for Reagent Water
product, Enterolert, utilizes a nutrient indicator that fluoresces D2777 Practice for Determination of Precision and Bias of
when metabolized. It can detect these bacteria at one most Applicable Test Methods of Committee D19 on Water
probable number (MPN)/100 mL within 24 h. The presence of D3370 Practices for Sampling Water from Flowing Process
this microorganism in water is an indication of fecal contami- Streams
nation and the possible presence of enteric pathogens.
3. Terminology
1.2 This test method can be used successfully with drinking
3.1 Definitions—For definitions of terms used in this test
water, source water, recreational (fresh and marine) water,
method, refer to Terminology D1129.
wastewater, and bottled water. It is the user’s responsibility to
ensure the validity of this test method for waters of untested 3.2 Definitions of Terms Specific to This Standard:
matrices.
3.2.1 enterococci, n—a gram positive bacteria possessing
the enzyme β-D-glucosidase, which cleaves the nutrient indi-
1.3 The values stated in SI units are to be regarded as
cator and produces fluorescence under a long wave length
standard. No other units of measurement are included in this
(365 nm to 366 nm) ultraviolet (UV) light.
standard.
3.2.2 most probable number (MPN), n—a statistical method
1.4 This standard does not purport to address all of the
for determining bacterial density based on the Poisson distri-
safety concerns, if any, associated with its use. It is the
bution.
responsibility of the user of this standard to establish appro-
priate safety, health, and environmental practices and deter- 3.2.3 presence-absence, n—a term used to indicate if en-
terococci are present or absent in a water sample.
mine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accor- 3.2.3.1 Discussion—It is a qualitative value, “yes” or “no”
for reporting results.
dance with internationally recognized principles on standard-
ization established in the Decision on Principles for the
3.2.4 Quanti-Tray, n—a system for the quantification of
Development of International Standards, Guides and Recom-
enterococci.
mendations issued by the World Trade Organization Technical
3.2.4.1 Discussion—It consists of a sealer and trays which
Barriers to Trade (TBT) Committee.
have multi-wells and can enumerate up to 2419 MPN/100 mL
without dilution using the Quanti-Tray 2000 tray or it can
detect up to 200.5 MPN/100 mL using the 51-well Quanti-
Tray.
This test method is under the jurisdiction of ASTM Committee D19 on Water
and is the direct responsibility of Subcommittee D19.24 on Water Microbiology.
3.2.5 snap pack, n—a package containing Enterolert reagent
Current edition approved April 1, 2024. Published April 2024. Originally
for testing 100 mL sample either in the P/A format or quanti-
approved in 1999. Last previous edition approved in 2019 as D6503 – 19. DOI:
10.1520/D6503-24. tatively.
Enterolert and Quanti-Tray are trademarks of IDEXX Laboratories, Inc., in
Westbrook, ME. IDEXX Laboratories, Inc. is the sole source of supply of Enterolert
and Quanti-Tray. If you are aware of alternative suppliers, please provide this For referenced ASTM standards, visit the ASTM website, www.astm.org, or
information to ASTM International Headquarters. Your comments will receive contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
careful consideration at a meeting of the responsible technical committee, which Standards volume information, refer to the standard’s Document Summary page on
you may attend. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6503 − 24
4. Summary of Test Method 10.2 Sample Storage Temperature and Handling
Conditions—Ice or refrigerate water samples at a temperature
4.1 This test method is used for the detection of enterococci,
of 2 °C to 8 °C during transit to the laboratory. Use insulated
such as E. faecium, E. faecalis in drinking water, source water,
containers to ensure proper maintenance of storage tempera-
recreational waters (marine water and fresh), wastewaters, and
tures. Take care that sample bottles are not totally immersed in
bottled water. When the reagent is added to the sample and
water during transit or storage.
incubated at 41 °C 6 0.5 °C for 24 h and up to 28 h, Enterolert
can detect these bacteria at 1 MPN/100 mL. Fluorescence is 10.3 Holding Time Limitations—Examine samples, as soon
produced when enterococci metabolizes the nutrient indicator. as possible, after collection. Do not hold samples longer than 8
Enterolert can be used as a presence-absence test or for h between collection and incubation of samples.
quantification (5-tube, 10-tube MPN, 15-tube serial dilution or
11. Quality Control Check
the Quanti-Tray system).
11.1 Check and record temperatures in incubators twice
5. Significance and Use
daily (separated by at least 4 h) when in use to ensure
temperature is within stated limits.
5.1 This test provides an easy and reliable method for the
detection of enterococci in water within 24 h. For recreational
11.2 Quality control should be conducted on each new lot of
water (fresh and marine) testing is performed to insure areas
Enterolert. See package insert for the recommended quality
are safe for swimming. Enterolert also can be used for testing
control procedure, using American Type Culture Collection
bottled water, wastewater, ground water, and drinking water.
(ATCC) bacteria strains or equivalent. The following protocol
is an example using the ATCC bacterial strains:
6. Interferences
11.2.1 For each type of the ATCC bacterial strain listed
below, streak the culture onto labeled TSA or blood agar plates
6.1 The presence of Bacillus spp. can interfere with the
and incubate at 35 °C for 18 h to 24 h.
testing of marine water samples. To eliminate interference, a
11.2.2 For each bacterial strain, touch a 1 μL loop to a
1:10 dilution is required with sterile water (deionized or
colony and use it to inoculate a labeled test tube containing
distilled).
5 mL of sterile deionized water. Close cap and shake thor-
oughly.
7. Apparatus
11.2.3 For each bacterial strain, take a 1 μL loop from the
7.1 Ultraviolet Lamp, 6 watt long wavelength or equivalent
test tube (11.2.2) and use it to inoculate a labeled vessel
(365 nm to 366 nm).
containing 100 mL of sterile deionized water.
7.2 41 °C 6 0.5 °C Incubator, air or water bath.
11.2.4 Follow the Enterolert presence/absence or MPN
steps listed in Section 12, Procedure, to test these controls.
7.3 Vessels, sterile, non-fluorescent.
Compare the test results to the following expected results:
7.4 Quanti-Tray Sealer.
Control ATTC No. Expected Result
7.5 Quanti-Tray 51-well or Quanti-Tray 2000.
Enterococcus faecium 335667 Fluorescence
Serratia marcescens (g, –) 43862 No fluorescence
Aerococcus viridians (g, +) 10400 No fluorescence
8. Reagents and Materials
12. Procedure
8.1 Purity of Water—Unless otherwise indicated, references
NOTE 1—A 1:10 dilution is required for marine water samples or
to water shall be understood to mean reagent water conforming
samples containing the presence of salt.
to Specification D1193, Type IV. Sterilize the water by either
12.1 Presence/Absence—See package insert.
autoclaving or by sterile filtration (0.22 micron-filtered water).
12.1.1 Samples should be brought to room temperature
8.2 Enterolert Test Kit.
(18 °C to 30 °C).
12.1.2 Carefully separate one snap pack from the strip.
9. Precautions
12.1.3 Tap the snap pack to insure that all of the powder is
9.1 The analyst must observe the normal good laboratory
towards the bottom of the pack.
practices and safety procedures required in a microbiology
12.1.4 Open the pack by snapping back the top of the score
laboratory while preparing, using, and disposing of cultures,
line. Do not touch the opening of pack.
reagents and materials and while operating sterilization equip-
12.1.5 Add the reagent to a 100 mL water sample, which is
ment and other equipment.
in a sterile, transparent, non-fluorescent vessel.
12.1.6 Aseptically cap and seal the vessel.
10. Sampling
12.1.7 Shake until dissolved.
12.1.8 Incubate Enterolert for 24 h and up to 28 h at 41 °C
10.1 Collect the sample as described in detail in the USEPA
microbiological methods manual and in accordance with 6 0.5 °C,
12.1.9 Read results at 24 h and up to 28 h. If the sample is
Practices D3370.
inadvertently incubated over 28 h without observation, the
following guidelines apply: Lack of fluorescence after 28 h is
a valid negative test. Fluorescence after 28 h is an invalid
Bordner, R.H., Winter, J.A., and Scarpino, P.V., Eds., Microbiological Methods
for Monitoring the Environment, Water, and Wastes, EPA-600/8-78-017. result.
D6503 − 24
12.1.10 Check for fluorescence by placing a 6 watt or Refer to the MPN table (see Table 1) provided with the
equivalent 365 nm to 366 nm UV light within 5 in. of the Quanti-Tray to determine the MPN/100 mL.
sample in a dark environment. Be sure the light is facing away
12.3 MPN—5-tube × 20 mL, 10-tube × 10 mL and 15-tube
from your eyes and towards the vessel. If fluorescence is
serial dilution.
observed, the presence of enterococci is confirmed.
12.3.1 Follow 12.1.1 – 12.1.7.
12.2 MPN—Quanti-Tray enumeration test procedure for 12.3.2 Use sterile nonfluorescent tubes or transfer 20 mL of
100 mL sample (see package insert). the reagent sample into five sterile non-fluorescent tubes or
12.2.1 Follow steps 12.1.1 – 12.1.7. transfer 10 mL of the reagent sample into five sterile non-
12.2.2 Pour the reagent sample into the Quanti-Tray avoid- fluorescent tubes.
ing contact with the foil tab and seal the tray according to the 12.3.3 For 15 tube serial dilution.
Quanti-Tray package insert. 12.3.4 Incubate for 24 h and up to 28 h at 41 °C 6 0.5 °C.
12.2.3 Incubate for 24 h and up to 28 h at 41 °C 6 0.5 °C. 12.3.5 Follow 12.1.9 and 12.1.10 for interpretation.
12.2.4 Follow the same interpretation instructions from 12.3.6 Refer to the MPN tables (see Tables 2-5) to deter-
12.1.9 through 12.1.10, and count the number of positive wells. mine the MPN/100 mL.
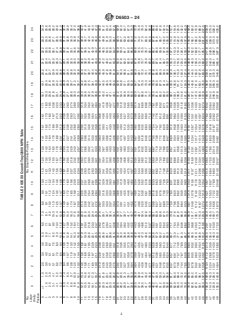
TABLE 1 51-Well Quanti-Tray MPN Table
95 % Confidence Limits
No. of Wells Giving Positive Reaction MPN/100-mL Sample
Lower Upper
0 <1 0.0 3.7
1 1.0 0.3 5.6
2 2.0 0.6 7.3
3 3.1 1.1 9.0
4 4.2 1.7 10.7
5 5.3 2.3 12.3
6 6.4 3.0 13.9
7 7.5 3.7 15.5
8 8.7 4.5 17.1
9 9.9 5.3 18.8
10 11.1 6.1 20.5
11 12.4 7.0 22.1
12 13.7 7.9 23.9
13 15.0 8.8 25.7
14 16.4 9.8 27.5
15 17.8 10.8 29.4
16 19.2 11.9 31.3
17 20.7 13.0 33.3
18 22.2 14.1 35.2
19 23.8 15.3 37.3
20 25.4 16.5 39.4
21 27.1 17.7 41.6
22 28.8 19.0 43.9
23 30.6 20.4 46.3
24 32.4 21.8 48.7
25 34.4 23.3 51.2
26 36.4 24.7 53.9
27 38.4 26.4 56.6
28 40.6 28.0 59.5
29 42.9 29.7 62.5
30 45.3 31.5 65.6
31 47.8 33.4 69.0
32 50.4 35.4 72.5
33 53.1 37.5 76.2
34 56.0 39.7 80.1
35 59.1 42.0 84.4
36 62.4 44.6 88.8
37 65.9 47.2 93.7
38 69.7 50.0 99.0
39 73.8 53.1 104.8
40 78.2 56.4 111.2
41 83.1 59.9 118.3
42 88.5 63.9 126.2
43 94.5 68.2 135.4
44 101.3 73.1 146.0
45 109.1 78.6 158.7
46 118.4 85.0 174.5
47 129.8 92.7 195.0
48 144.5 102.3 224.1
49 165.2 115.2 272.2
50 200.5 135.8 387.6
51 >200.5 146.1 infinite
D6503 − 24
TABLE 2 IDEXX Quanti-Tray/2000 MPN Table
No. No. Small Wells Positive
Large
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24
Wells
Positive
0 <1 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 11.0 12.0 13.0 14.1 15.1 16.1 17.1 18.1 19.1 20.2 21.2 22.2 23.2 24.3
1 1.0 2.0 3.0 4.0 5.0 6.0 7.1 8.1 9.1 10.1 11.1 12.1 13.2 14.2 15.2 16.2 17.3 18.3 19.3 20.4 21.4 22.4 23.5 24.5 25.6
2 2.0 3.0 4.1 5.1 6.1 7.1 8.1 9.2 10.2 11.2 12.2 13.3 14.3 15.4 16.4 17.4 18.5 19.5 20.6 21.6 22.7 23.7 24.8 25.8 26.9
3 3.1 4.1 5.1 6.1 7.2 8.2 9.2 10.3 11.3 12.4 13.4 14.5 15.5 16.5 17.6 18.6 19.7 20.8 21.8 22.9 23.9 25.0 26.1 27.1 28.2
4 4.1 5.2 6.2 7.2 8.3 9.3 10.4 11.4 12.5 13.5 14.6 15.6 16.7 17.8 18.8 19.9 21.0 22.0 23.1 24.2 25.3 26.3 27.4 28.5 29.6
5 5.2 6.3 7.3 8.4 9.4 10.5 11.5 12.6 13.7 14.7 15.8 16.9 17.9 19.0 20.1 21.2 22.2 23.3 24.4 25.5 26.6 27.7 28.8 29.9 31.0
6 6.3 7.4 8.4 9.5 10.6 11.6 12.7 13.8 14.9 16.0 17.0 18.1 19.2 20.3 21.4 22.5 23.6 24.7 25.8 26.9 28.0 29.1 30.2 31.3 32.4
7 7.4 8.5 9.6 10.7 11.8 12.8 13.9 15.0 16.1 17.2 18.3 19.4 20.5 21.6 22.7 23.8 24.9 26.0 27.1 28.3 29.4 30.5 31.6 32.8 33.9
8 8.6 9.7 10.8 11.9 13.0 14.1 15.2 16.3 17.4 18.5 19.6 20.7 21.8 22.9 24.1 25.2 26.3 27.4 28.6 29.7 30.8 32.0 33.1 34.3 35.4
9 9.8 10.9 12.0 13.1 14.2 15.3 16.4 17.6 18.7 19.8 20.9 22.0 23.2 24.3 25.4 26.6 27.7 28.9 30.0 31.2 32.3 33.5 34.6 35.8 37.0
10 11.0 12.1 13.2 14.4 15.5 16.6 17.7 18.9 20.0 21.1 22.3 23.4 24.6 25.7 26.9 28.0 29.2 30.3 31.5 32.7 33.8 35.0 36.2 37.4 38.6
11 12.2 13.4 14.5 15.6 16.8 17.9 19.1 20.2 21.4 22.5 23.7 24.8 26.0 27.2 28.3 29.5 30.7 31.9 33.0 34.2 35.4 36.6 37.8 39.0 40.2
12 13.5 14.6 15.8 16.9 18.1 19.3 20.4 21.6 22.8 23.9 25.1 26.3 27.5 28.6 29.8 31.0 32.2 33.4 34.6 35.8 37.0 38.2 39.5 40.7 41.9
13 14.8 16.0 17.1 18.3 19.5 20.6 21.8 23.0 24.2 25.4 26.6 27.8 29.0 30.2 31.4 32.6 33.8 35.0 36.2 37.5 38.7 39.9 41.2 42.4 43.6
14 16.1 17.3 18.5 19.7 20.9 22.1 23.3 24.4 25.7 26.9 28.1 29.3 30.5 31.7 33.0 34.2 35.4 36.7 37.9 39.1 40.4 41.6 42.9 44.2 45.4
15 17.5 18.7 19.9 21.1 22.3 23.5 24.7 25.9 27.2 28.4 29.6 30.9 32.1 33.3 34.6 35.8 37.1 38.4 39.6 40.9 42.2 43.4 44.7 46.0 47.3
16 18.9 20.1 21.3 22.6 23.8 25.0 26.2 27.5 28.7 30.0 31.2 32.5 33.7 35.0 36.3 37.5 38.8 40.1 41.4 42.7 44.0 45.3 46.6 47.9 49.2
17 20.3 21.6 22.8 24.1 25.3 26.6 27.8 29.1 30.3 31.6 32.9 34.1 35.4 36.7 38.0 39.3 40.6 41.9 43.2 44.5 45.9 47.2 48.5 49.8 51.2
18 21.8 23.1 24.3 25.6 26.9 28.1 29.4 30.7 32.0 33.3 34.6 35.9 37.2 38.5 39.8 41.1 42.4 43.8 45.1 46.5 47.8 49.2 50.5 51.9 53.2
19 23.3 24.6 25.9 27.2 28.5 29.8 31.1 32.4 33.7 35.0 36.3 37.6 39.0 40.3 41.6 43.0 44.3 45.7 47.1 48.4 49.8 51.2 52.6 54.0 55.4
20 24.9 26.2 27.5 28.8 30.1 31.5 32.8 34.1 35.4 36.8 38.1 39.5 40.8 42.2 43.6 44.9 46.3 47.7 49.1 50.5 51.9 53.3 54.7 56.1 57.6
21 26.5 27.8 29.2 30.5 31.8 33.2 34.5 35.9 37.3 38.6 40.0 41.4 42.8 44.1 45.5 46.9 48.4 49.8 51.2 52.6 54.1 55.5 56.9 58.4 59.9
22 28.2 29.5 30.9 32.3 33.6 35.0 36.4 37.7 39.1 40.5 41.9 43.3 44.8 46.2 47.6 49.0 50.5 51.9 53.4 54.8 56.3 57.8 59.3 60.7 62.3
23 29.9 31.3 32.7 34.1 35.5 36.8 38.2 39.7 41.1 42.5 43.9 45.4 46.8 48.3 49.7 51.2 52.7 54.2 55.6 57.1 58.6 60.2 61.7 63.2 64.7
24 31.7 33.1 34.5 35.9 37.3 38.8 40.2 41.7 43.1 44.6 46.0 47.5 49.0 50.5 52.0 53.5 55.0 56.5 58.0 59.5 61.1 62.6 64.2 65.8 67.3
25 33.6 35.0 36.4 37.9 39.3 40.8 42.2 43.7 45.2 46.7 48.2 49.7 51.2 52.7 54.3 55.8 57.3 58.9 60.5 62.0 63.6 65.2 66.8 68.4 70.0
26 35.5 36.9 38.4 39.9 41.4 42.8 44.3 45.9 47.4 48.9 50.4 52.0 53.5 55.1 56.7 58.2 59.8 61.4 63.0 64.7 66.3 67.9 69.6 71.2 72.9
27 37.4 38.9 40.4 42.0 43.5 45.0 46.5 48.1 49.6 51.2 52.8 54.4 56.0 57.6 59.2 60.8 62.4 64.1 65.7 67.4 69.1 70.8 72.5 74.2 75.9
28 39.5 41.0 42.6 44.1 45.7 47.3 48.8 50.4 52.0 53.6 55.2 56.9 58.5 60.2 61.8 63.5 65.2 66.9 68.6 70.3 72.
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6503 − 19 D6503 − 24
Standard Test Method for
1,2
Enterococci in Water Using Enterolert
This standard is issued under the fixed designation D6503; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers a simple procedure for the detection of enterococci in water and wastewater. It is based on IDEXX’s
patented Defined Substrate Technology (DST). This product, Enterolert, utilizes a nutrient indicator that fluoresces when
metabolized. It can detect these bacteria at one most probable number (MPN)/100 mL within 24 h. The presence of this
microorganism in water is an indication of fecal contamination and the possible presence of enteric pathogens.
1.2 This test method can be used successfully with drinking water, source water, recreational (fresh and marine) water, wastewater,
and bottled water. It is the user’s responsibility to ensure the validity of this test method for waters of untested matrices.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3370 Practices for Sampling Water from Flowing Process Streams
3. Terminology
3.1 Definitions—For definitions of terms used in this test method, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.24 on Water Microbiology.
Current edition approved April 1, 2019April 1, 2024. Published May 2019April 2024. Originally approved in 1999. Last previous edition approved in 20142019 as
D6503 – 14.D6503 – 19. DOI: 10.1520/D6503-19.10.1520/D6503-24.
Enterolert and Quanti-Tray are trademarks of IDEXX Laboratories, Inc., in Westbrook, ME. IDEXX Laboratories, Inc. is the sole source of supply of Enterolert and
Quanti-Tray. If you are aware of alternative suppliers, please provide this information to ASTM International Headquarters. Your comments will receive careful consideration
at a meeting of the responsible technical committee, which you may attend.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6503 − 24
3.2.1 enterococci, n—a gram positive bacteria possessing the enzyme β-D-glucosidase, which cleaves the nutrient indicator and
produces fluorescence under a long wave length (365–366 (365 nm to 366 nm) ultraviolet (UV) light.
3.2.2 most probable number (MPN), n—a statistical method for determining bacterial density based on the Poisson distribution.
3.2.3 presence-absence, n—a term used to indicate if enterococci are present or absent in a water sample.
3.2.3.1 Discussion—
It is a qualitative value, “yes” or “no” for reporting results.
3.2.4 Quanti-Tray, n—a system for the quantification of enterococci.
3.2.4.1 Discussion—
It consists of a sealer and trays which have multi-wells and can enumerate up to 2419 MPN/100 mL without dilution using the
Quanti-Tray 2000 tray or it can detect up to 200.5 MPN/100 mL using the 51-well Quanti-Tray.
3.2.5 snap pack, n—a package containing Enterolert reagent for testing 100-mL100 mL sample either in the P/A format or
quantitatively, with the Quanti-Tray system.quantitatively.
4. Summary of Test Method
4.1 This test method is used for the detection of enterococci, such as E. faecium, E. faecalis in drinking water, source water,
recreational waters (marine water and fresh), wastewaters, and bottled water. When the reagent is added to the sample and
incubated at 41 6 0.5°C41 °C 6 0.5 °C for 24 h and up to 28 h, Enterolert can detect these bacteria at 1 MPN/100 mL.
Fluorescence is produced when enterococci metabolizes the nutrient indicator. Enterolert can be used as a presence-absence test
or for quantification (5-tube, 10-tube MPN, 15-tube serial dilution or the Quanti-Tray system).
5. Significance and Use
5.1 This test provides an easy and reliable method for the detection of enterococci in water within 24 h. For recreational water
(fresh and marine) testing is performed to insure areas are safe for swimming. Enterolert also can be used for testing bottled water,
wastewater, ground water, and drinking water.
6. Interferences
6.1 The presence of Bacillus spp. can interfere with the testing of marine water samples. To eliminate interference, a 1:10 dilution
is required with sterile water (deionized or distilled).
7. Apparatus
7.1 Ultraviolet Lamp, 6-watt long wavelength (365–366 6 watt long wavelength or equivalent (365 nm to 366 nm).
7.2 4160.5°C 41 °C 6 0.5 °C Incubator, air or water bath.
7.3 Vessels, sterile, non-fluorescent.
7.4 Quanti-Tray Sealer.
7.5 Quanti-Tray 51-well or Quanti-Tray 2000.
8. Reagents and Materials
8.1 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Specification D1193, Type IV. Sterilize the water by either autoclaving or by sterile filtration (0.22 micron-filtered water).
8.2 Enterolert Test Kit.
D6503 − 24
9. Precautions
9.1 The analyst must observe the normal good laboratory practices and safety procedures required in a microbiology laboratory
while preparing, using, and disposing of cultures, reagents and materials and while operating sterilization equipment and other
equipment.
10. Sampling
10.1 Collect the sample as described in detail in the USEPA microbiological methods manual and in accordance with Practices
D3370.
10.2 Sample Storage Temperature and Handling Conditions—Ice or refrigerate water samples at a temperature of 2 to 8°C2 °C
to 8 °C during transit to the laboratory. Use insulated containers to ensure proper maintenance of storage temperatures. Take care
that sample bottles are not totally immersed in water during transit or storage.
10.3 Holding Time Limitations—Examine samples, as soon as possible, after collection. Do not hold samples longer than 8 h
between collection and incubation of samples.
11. Quality Control Check
11.1 Check and record temperatures in incubators twice daily (separated by at least 4 hours)h) when in use to ensure temperature
is within stated limits.
11.2 Quality control should be conducted on each new lot of Enterolert. See package insert for the recommended quality control
procedure, using American Type Culture Collection (ATCC) bacteria strains or equivalent. The following protocol is an example
using the ATCC bacterial strains:
11.2.1 For each type of the ATCC bacterial strain listed below, streak the culture onto labeled TSA or blood agar plates and
incubate at 35°C for 1835 °C for 18 h to 24 h.
11.2.2 For each bacterial strain, touch a 1-μl1 μL loop to a colony and use it to inoculate a labeled test tube containing 5 mL of
sterile deionized water. Close cap and shake thoroughly.
11.2.3 For each bacterial strain, take a 1-μl1 μL loop from the test tube (11.2.2) and use it to inoculate a labeled vessel containing
100 mL of sterile deionized water.
11.2.4 Follow the Enterolert presence/absence or MPN steps listed in Section 12, Procedure, to test these controls. Compare the
test results to the following expected results:
Control ATTC No. Expected Result
Enterococcus faecium 335667 Fluorescence
Serratia marcescens (g, –) 43862 No fluorescence
Aerococcus viridians (g, +) 10400 No fluorescence
12. Procedure
NOTE 1—A 1:10 dilution is required for marine water samples or samples containing the presence of salt.
12.1 Presence/Absence—See package insert.
12.1.1 Samples should be brought to room temperature (18 to 30°C).(18 °C to 30 °C).
12.1.2 Carefully separate one snap pack from the strip.
12.1.3 Tap the snap pack to insure that all of the powder is towards the bottom of the pack.
Bordner, R.H., Winter, J.A., and Scarpino, P.V., Eds., Microbiological Methods for Monitoring the Environment, Water, and Wastes, EPA-600/8-78-017.
D6503 − 24
12.1.4 Open the pack by snapping back the top of the score line. Do not touch the opening of pack.
12.1.5 Add the reagent to a 100-mL100 mL water sample, which is in a sterile, transparent, non-fluorescent vessel.
12.1.6 Aseptically cap and seal the vessel.
12.1.7 Shake until dissolved.
12.1.8 Incubate Enterolert for 24 h and up to 28 h at 41 6 0.5°C,41 °C 6 0.5 °C,
12.1.9 Read results at 24 h and up to 28 h. If the sample is inadvertently incubated over 28 h without observation, the following
guidelines apply: Lack of fluorescence after 28 h is a valid negative test. Fluorescence after 28 h is an invalid result.
12.1.10 Check for fluorescence by placing a 6-W 365–366-nm 6 watt or equivalent 365 nm to 366 nm UV light within 5 in. of
the sample in a dark environment. Be sure the light is facing away from your eyes and towards the vessel. If fluorescence is
observed, the presence of enterococci is confirmed.
12.2 MPN—Quanti-Tray enumeration test procedure for 100-mL100 mL sample (see package insert).
12.2.1 Follow steps 12.1.1 – 12.1.7.
12.2.2 Pour the reagent sample into the Quanti-Tray avoiding contact with the foil tab and seal the tray according to the
Quanti-Tray package insert.
12.2.3 Incubate for 24 h and up to 28 h at 41 6 0.5°C.41 °C 6 0.5 °C.
12.2.4 Follow the same interpretation instructions from 12.1.9 through 12.1.10, and count the number of positive wells. Refer to
the MPN table (see Table 1) provided with the Quanti-Tray to determine the MPN/100 mL.
12.3 MPN—5-tube × 20 mL, 10-tube × 10 mL and 15-tube serial dilution.
12.3.1 Follow 12.1.1 – 12.1.7.
12.3.2 Use sterile nonfluorescent tubes or transfer 20 mL of the reagent sample into five sterile non-fluorescent tubes or transfer
10 mL of the reagent sample into five sterile non-fluorescent tubes.
12.3.3 For 15 tube serial dilution.
12.3.4 Incubate for 24 h and up to 28 h at 41 6 0.5°C.41 °C 6 0.5 °C.
12.3.5 Follow 12.1.9 and 12.1.10 for interpretation.
12.3.6 Refer to the MPN tables (see Tables 2-45) to determine the MPN/100 mL.
13. Calculation
13.1 For P/A, there are no calculations. For quantification, refer to Quanti-Tray MPN tables and for the 5, 10, and 15 tube test
results refer to the respective MPN tables.
14. Report
14.1 Report as positive or negative for presence/absence testing.
14.2 Reporting of results is based on calculation of enterococci density determined from the appropriate MPN tables.tables and
dilution used, if any.
Standard Methods for the Examination of Water and Waste Water, 19th Edition.
D6503 − 24
TABLE 1 51-Well Quanti-Tray MPN Table
95 % Confidence Limits
No. of Wells Giving Positive Reaction MPN/100-mL Sample
Lower Upper
0 <1 0.0 3.7
1 1.0 0.3 5.6
2 2.0 0.6 7.3
3 3.1 1.1 9.0
4 4.2 1.7 10.7
5 5.3 2.3 12.3
6 6.4 3.0 13.9
7 7.5 3.7 15.5
8 8.7 4.5 17.1
9 9.9 5.3 18.8
10 11.1 6.1 20.5
11 12.4 7.0 22.1
12 13.7 7.9 23.9
13 15.0 8.8 25.7
14 16.4 9.8 27.5
15 17.8 10.8 29.4
16 19.2 11.9 31.3
17 20.7 13.0 33.3
18 22.2 14.1 35.2
19 23.8 15.3 37.3
20 25.4 16.5 39.4
21 27.1 17.7 41.6
22 28.8 19.0 43.9
23 30.6 20.4 46.3
24 32.4 21.8 48.7
25 34.4 23.3 51.2
26 36.4 24.7 53.9
27 38.4 26.4 56.6
28 40.6 28.0 59.5
29 42.9 29.7 62.5
30 45.3 31.5 65.6
31 47.8 33.4 69.0
32 50.4 35.4 72.5
33 53.1 37.5 76.2
34 56.0 39.7 80.1
35 59.1 42.0 84.4
36 62.4 44.6 88.8
37 65.9 47.2 93.7
38 69.7 50.0 99.0
39 73.8 53.1 104.8
40 78.2 56.4 111.2
41 83.1 59.9 118.3
42 88.5 63.9 126.2
43 94.5 68.2 135.4
44 101.3 73.1 146.0
45 109.1 78.6 158.7
46 118.4 85.0 174.5
47 129.8 92.7 195.0
48 144.5 102.3 224.1
49 165.2 115.2 272.2
50 200.5 135.8 387.6
51 >200.5 146.1 infinite
15. Precision and Bias
15.1 Precision—A limited collaborative study was conducted. Nine technicians from three laboratories tested three different
matrixes at three levels following Practice D2777. Outliers were rejected in accordance with the statistical tests outlined in Practice
D2777. All data from one technician was rejected for recreational water-marine and single values were rejected for both
recreational water-fresh at the low level and for recreational water-marine at the low level. The mean count, the overall standard
deviation (St), and the single operator standard deviation (So), are indicated in Table 56.
15.2 Bias—The mean value obtained for the samples (drinking water, recreational water fresh and marine) from the nine
technicians for the low-, mid- and high-spiked samples all fall within the 95 % confidence interval (poisson distribution) of the
actual values obtained from plating on blood agar.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D19-1167. Contact ASTM Customer
Service at service@astm.org.
D6503 − 24
TABLE 2 IDEXX Quanti-Tray/2000 MPN Table
No. No. Small Wells Positive
Large
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24
Wells
Positive
0 <1 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 11.0 12.0 13.0 14.1 15.1 16.1 17.1 18.1 19.1 20.2 21.2 22.2 23.2 24.3
1 1.0 2.0 3.0 4.0 5.0 6.0 7.1 8.1 9.1 10.1 11.1 12.1 13.2 14.2 15.2 16.2 17.3 18.3 19.3 20.4 21.4 22.4 23.5 24.5 25.6
2 2.0 3.0 4.1 5.1 6.1 7.1 8.1 9.2 10.2 11.2 12.2 13.3 14.3 15.4 16.4 17.4 18.5 19.5 20.6 21.6 22.7 23.7 24.8 25.8 26.9
3 3.1 4.1 5.1 6.1 7.2 8.2 9.2 10.3 11.3 12.4 13.4 14.5 15.5 16.5 17.6 18.6 19.7 20.8 21.8 22.9 23.9 25.0 26.1 27.1 28.2
4 4.1 5.2 6.2 7.2 8.3 9.3 10.4 11.4 12.5 13.5 14.6 15.6 16.7 17.8 18.8 19.9 21.0 22.0 23.1 24.2 25.3 26.3 27.4 28.5 29.6
5 5.2 6.3 7.3 8.4 9.4 10.5 11.5 12.6 13.7 14.7 15.8 16.9 17.9 19.0 20.1 21.2 22.2 23.3 24.4 25.5 26.6 27.7 28.8 29.9 31.0
6 6.3 7.4 8.4 9.5 10.6 11.6 12.7 13.8 14.9 16.0 17.0 18.1 19.2 20.3 21.4 22.5 23.6 24.7 25.8 26.9 28.0 29.1 30.2 31.3 32.4
7 7.4 8.5 9.6 10.7 11.8 12.8 13.9 15.0 16.1 17.2 18.3 19.4 20.5 21.6 22.7 23.8 24.9 26.0 27.1 28.3 29.4 30.5 31.6 32.8 33.9
8 8.6 9.7 10.8 11.9 13.0 14.1 15.2 16.3 17.4 18.5 19.6 20.7 21.8 22.9 24.1 25.2 26.3 27.4 28.6 29.7 30.8 32.0 33.1 34.3 35.4
9 9.8 10.9 12.0 13.1 14.2 15.3 16.4 17.6 18.7 19.8 20.9 22.0 23.2 24.3 25.4 26.6 27.7 28.9 30.0 31.2 32.3 33.5 34.6 35.8 37.0
10 11.0 12.1 13.2 14.4 15.5 16.6 17.7 18.9 20.0 21.1 22.3 23.4 24.6 25.7 26.9 28.0 29.2 30.3 31.5 32.7 33.8 35.0 36.2 37.4 38.6
11 12.2 13.4 14.5 15.6 16.8 17.9 19.1 20.2 21.4 22.5 23.7 24.8 26.0 27.2 28.3 29.5 30.7 31.9 33.0 34.2 35.4 36.6 37.8 39.0 40.2
12 13.5 14.6 15.8 16.9 18.1 19.3 20.4 21.6 22.8 23.9 25.1 26.3 27.5 28.6 29.8 31.0 32.2 33.4 34.6 35.8 37.0 38.2 39.5 40.7 41.9
13 14.8 16.0 17.1 18.3 19.5 20.6 21.8 23.0 24.2 25.4 26.6 27.8 29.0 30.2 31.4 32.6 33.8 35.0 36.2 37.5 38.7 39.9 41.2 42.4 43.6
14 16.1 17.3 18.5 19.7 20.9 22.1 23.3 24.4 25.7 26.9 28.1 29.3 30.5 31.7 3
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...