ASTM D8421-22
(Test Method)Standard Test Method for Determination of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Matrices by Co-solvation followed by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS)
Standard Test Method for Determination of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Matrices by Co-solvation followed by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS)
SIGNIFICANCE AND USE
5.1 PFAS are widely used in various industrial and commercial products; they are persistent, bio-accumulative, and ubiquitous in the environment. PFAS have been reported to exhibit developmental toxicity, hepatotoxicity, immunotoxicity, and hormone disturbance. PFAS have been detected in soils, sludges, surface, and drinking waters. This is a quick, easy, and robust method to quantitatively determine these compounds at trace levels in water matrices.
5.2 This test method has been validated using reagent water and waters from sites that include landfill leachate, metal finisher, POTW Effluent, Hospital, POTW Influent, Bus washing station, Power Plant and Pulp and paper mill effluent for selected PFAS, refer to the Precision and Bias (Section 17).
SCOPE
1.1 This test method covers the determination of per- and polyfluoroalkyl substances (PFASs) in aqueous matrices using liquid chromatography (LC) and detection with tandem mass spectrometry (MS/MS). These analytes are co-solvated by a 1+1 ratio of sample and methanol then qualitatively and quantitatively determined by this test method. Quantitation is by selected reaction monitoring (SRM) or sometimes referred to as multiple reaction monitoring (MRM).
1.2 The method detection limit (MDL) (see Note 1) and reporting range (see Note 2) for the target analytes are listed in Table 1. The target concentration for the reporting limit for this test method is an integer value that is calculated from the concentration from the lowest standard from the final volume of the prepared sample. This value may be lower than the calculated MDL due to sporadic PFAS hits due to PFAS contamination in consumables/collection tools used during sample collection and preparation. All samples should be taken at a minimal as duplicates in order to compare the precision between the two prepared samples to help ensure the concentration/positive result is reliable.
Note 1: The MDL is determined following the Code of Federal Regulations (CFR), 40 CFR Part 136, Appendix B utilizing dilution and filtration. A detailed process determining the MDL is explained in the reference and is beyond the scope of this test method.
Note 2: Injection volume variations, and sensitivity of the instrument used will change the reporting limit and ranges.
1.2.1 Recognizing continual advancements in the sensitivity of instrumentation, advancements in column chromatography and other processes not recognized here, the reporting limit may be lowered assuming the minimum performance requirements of this test method at the lower concentrations are met.
1.2.2 Depending on data usage, you may modify this test method but limit to modifications that improve performance while still meeting or exceeding the method quality acceptance criteria. Modifications to the solvents, ratio of solvent to sample, or shortening the chromatographic run simply to save time are not allowed. Use Practice E2935 or similar statistical tests to confirm that modifications produce equivalent results on non-interfering samples. In addition, use Guide E2857 or equivalent statistics to re-validate the modified test.
1.2.3 Analyte detections between the method detection limit and the reporting limit are estimated concentrations. The reporting limit is based upon the concentration of the Level 1 calibration standard as shown in Table 5.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on P...
General Information
- Status
- Published
- Publication Date
- 30-Apr-2022
- Technical Committee
- D19 - Water
- Drafting Committee
- D19.06 - Methods for Analysis for Organic Substances in Water
Relations
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-May-2020
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Jan-2018
- Refers
ASTM E2935-17 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2017
- Refers
ASTM E2935-16 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 15-Nov-2016
- Refers
ASTM E2935-15 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2015
- Refers
ASTM E2935-14 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Oct-2014
- Refers
ASTM E2935-13 - Standard Practice for Conducting Equivalence Testing in Laboratory Applications - Effective Date
- 01-Aug-2013
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 01-Jan-2013
- Effective Date
- 15-Jun-2012
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
Overview
ASTM D8421-22 is an international standard that specifies a test method for the determination of per- and polyfluoroalkyl substances (PFAS) in aqueous matrices. Developed by ASTM International, this method utilizes a co-solvation technique followed by detection using liquid chromatography tandem mass spectrometry (LC/MS/MS). PFAS are synthetic chemicals found in a variety of commercial and industrial products. Due to their persistence, bioaccumulation, and potential toxicity, accurate detection and quantitation of PFAS at trace levels in water, wastewater, and environmental samples is essential for environmental monitoring and regulatory compliance.
Key Topics
- Target Analytes: The method covers a comprehensive list of PFAS compounds, including well-known substances such as PFOA, PFOS, GenX, and a variety of perfluoroalkyl carboxylic and sulfonic acids, among others.
- Matrix Scope: Applicable to a wide range of aqueous matrices, such as reagent water, drinking water, surface water, landfill leachate, municipal and industrial wastewater, and effluents from sites like hospitals, power plants, and pulp and paper mills.
- Sample Preparation:
- Samples are co-solvated in a 1:1 methanol:water ratio for efficient extraction.
- The process utilizes selected reaction monitoring (SRM) or multiple reaction monitoring (MRM) for quantitation.
- All samples should be analyzed as duplicates to ensure data reliability and evaluate precision.
- Sensitivity and Reporting:
- The method is robust and can detect trace PFAS concentrations. Method detection limits (MDLs) and reporting ranges are dependent on instrumental sensitivity and sample matrix.
- MDLs are determined following US EPA (40 CFR Part 136, Appendix B) guidelines.
- Modifications to the method are permissible only to improve performance, provided quality criteria are met and validated statistically.
Applications
ASTM D8421-22 offers broad practical value for industries, laboratories, and regulatory bodies that require reliable, validated techniques for PFAS analysis:
- Environmental Monitoring: Ensures accurate PFAS detection in drinking water, surface water, and groundwater, addressing contamination and public health concerns.
- Industrial Discharge and Compliance: Used by industries to monitor PFAS in effluents and comply with discharge permits and environmental regulations.
- Research and Risk Assessment: Supports toxicological and ecological studies by providing reliable quantification of PFAS at low concentrations.
- Site Investigation and Remediation: Essential for sites affected by PFAS contamination, including landfills, treatment plants, and manufacturing facilities.
Laboratories applying this method benefit from its clarity and adaptability, as well as from its validation against a wide variety of real-world matrices. The emphasis on using appropriate sampling containers, high-purity reagents, and thorough instrument calibration minimizes contamination and maximizes reproducibility.
Related Standards
The method aligns with or references several important documents and standards to ensure reliable, quality-assured results:
- ASTM D1193: Specification for Reagent Water
- ASTM D2777, D4841, D5847: Practices and guides for analytical precision, bias, and quality control in water analysis
- ASTM D3856, D8272: Guidelines and frameworks for laboratory systems and analytical method development
- ASTM E694, E2857, E2935: Procedures for glassware, uncertainty analysis, and method validation/equivalence
- US EPA 40 CFR Part 136, Appendix B: Regulatory requirements for MDL determination in environmental analysis
By adopting ASTM D8421-22, organizations ensure their PFAS testing meets international norms for accuracy, reproducibility, and environmental relevance, supporting effective decision-making and regulatory compliance in water quality management.
Buy Documents
ASTM D8421-22 - Standard Test Method for Determination of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Matrices by Co-solvation followed by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS)
REDLINE ASTM D8421-22 - Standard Test Method for Determination of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Matrices by Co-solvation followed by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D8421-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Matrices by Co-solvation followed by Liquid Chromatography Tandem Mass Spectrometry (LC/MS/MS)". This standard covers: SIGNIFICANCE AND USE 5.1 PFAS are widely used in various industrial and commercial products; they are persistent, bio-accumulative, and ubiquitous in the environment. PFAS have been reported to exhibit developmental toxicity, hepatotoxicity, immunotoxicity, and hormone disturbance. PFAS have been detected in soils, sludges, surface, and drinking waters. This is a quick, easy, and robust method to quantitatively determine these compounds at trace levels in water matrices. 5.2 This test method has been validated using reagent water and waters from sites that include landfill leachate, metal finisher, POTW Effluent, Hospital, POTW Influent, Bus washing station, Power Plant and Pulp and paper mill effluent for selected PFAS, refer to the Precision and Bias (Section 17). SCOPE 1.1 This test method covers the determination of per- and polyfluoroalkyl substances (PFASs) in aqueous matrices using liquid chromatography (LC) and detection with tandem mass spectrometry (MS/MS). These analytes are co-solvated by a 1+1 ratio of sample and methanol then qualitatively and quantitatively determined by this test method. Quantitation is by selected reaction monitoring (SRM) or sometimes referred to as multiple reaction monitoring (MRM). 1.2 The method detection limit (MDL) (see Note 1) and reporting range (see Note 2) for the target analytes are listed in Table 1. The target concentration for the reporting limit for this test method is an integer value that is calculated from the concentration from the lowest standard from the final volume of the prepared sample. This value may be lower than the calculated MDL due to sporadic PFAS hits due to PFAS contamination in consumables/collection tools used during sample collection and preparation. All samples should be taken at a minimal as duplicates in order to compare the precision between the two prepared samples to help ensure the concentration/positive result is reliable. Note 1: The MDL is determined following the Code of Federal Regulations (CFR), 40 CFR Part 136, Appendix B utilizing dilution and filtration. A detailed process determining the MDL is explained in the reference and is beyond the scope of this test method. Note 2: Injection volume variations, and sensitivity of the instrument used will change the reporting limit and ranges. 1.2.1 Recognizing continual advancements in the sensitivity of instrumentation, advancements in column chromatography and other processes not recognized here, the reporting limit may be lowered assuming the minimum performance requirements of this test method at the lower concentrations are met. 1.2.2 Depending on data usage, you may modify this test method but limit to modifications that improve performance while still meeting or exceeding the method quality acceptance criteria. Modifications to the solvents, ratio of solvent to sample, or shortening the chromatographic run simply to save time are not allowed. Use Practice E2935 or similar statistical tests to confirm that modifications produce equivalent results on non-interfering samples. In addition, use Guide E2857 or equivalent statistics to re-validate the modified test. 1.2.3 Analyte detections between the method detection limit and the reporting limit are estimated concentrations. The reporting limit is based upon the concentration of the Level 1 calibration standard as shown in Table 5. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on P...
SIGNIFICANCE AND USE 5.1 PFAS are widely used in various industrial and commercial products; they are persistent, bio-accumulative, and ubiquitous in the environment. PFAS have been reported to exhibit developmental toxicity, hepatotoxicity, immunotoxicity, and hormone disturbance. PFAS have been detected in soils, sludges, surface, and drinking waters. This is a quick, easy, and robust method to quantitatively determine these compounds at trace levels in water matrices. 5.2 This test method has been validated using reagent water and waters from sites that include landfill leachate, metal finisher, POTW Effluent, Hospital, POTW Influent, Bus washing station, Power Plant and Pulp and paper mill effluent for selected PFAS, refer to the Precision and Bias (Section 17). SCOPE 1.1 This test method covers the determination of per- and polyfluoroalkyl substances (PFASs) in aqueous matrices using liquid chromatography (LC) and detection with tandem mass spectrometry (MS/MS). These analytes are co-solvated by a 1+1 ratio of sample and methanol then qualitatively and quantitatively determined by this test method. Quantitation is by selected reaction monitoring (SRM) or sometimes referred to as multiple reaction monitoring (MRM). 1.2 The method detection limit (MDL) (see Note 1) and reporting range (see Note 2) for the target analytes are listed in Table 1. The target concentration for the reporting limit for this test method is an integer value that is calculated from the concentration from the lowest standard from the final volume of the prepared sample. This value may be lower than the calculated MDL due to sporadic PFAS hits due to PFAS contamination in consumables/collection tools used during sample collection and preparation. All samples should be taken at a minimal as duplicates in order to compare the precision between the two prepared samples to help ensure the concentration/positive result is reliable. Note 1: The MDL is determined following the Code of Federal Regulations (CFR), 40 CFR Part 136, Appendix B utilizing dilution and filtration. A detailed process determining the MDL is explained in the reference and is beyond the scope of this test method. Note 2: Injection volume variations, and sensitivity of the instrument used will change the reporting limit and ranges. 1.2.1 Recognizing continual advancements in the sensitivity of instrumentation, advancements in column chromatography and other processes not recognized here, the reporting limit may be lowered assuming the minimum performance requirements of this test method at the lower concentrations are met. 1.2.2 Depending on data usage, you may modify this test method but limit to modifications that improve performance while still meeting or exceeding the method quality acceptance criteria. Modifications to the solvents, ratio of solvent to sample, or shortening the chromatographic run simply to save time are not allowed. Use Practice E2935 or similar statistical tests to confirm that modifications produce equivalent results on non-interfering samples. In addition, use Guide E2857 or equivalent statistics to re-validate the modified test. 1.2.3 Analyte detections between the method detection limit and the reporting limit are estimated concentrations. The reporting limit is based upon the concentration of the Level 1 calibration standard as shown in Table 5. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on P...
ASTM D8421-22 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8421-22 has the following relationships with other standards: It is inter standard links to ASTM E694-18(2024), ASTM D1129-13(2020)e2, ASTM E2554-18e1, ASTM E2554-18, ASTM E694-18, ASTM E2935-17, ASTM E2935-16, ASTM E2935-15, ASTM E2935-14, ASTM E2935-13, ASTM E2554-13, ASTM D4841-88(2013), ASTM D4841-88(2013)e1, ASTM D2777-12, ASTM D3856-11. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D8421-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:D8421 −22
Standard Test Method for
Determination of Per- and Polyfluoroalkyl Substances
(PFAS) in Aqueous Matrices by Co-solvation followed by
Liquid Chromatography Tandem Mass Spectrometry (LC/
MS/MS)
This standard is issued under the fixed designation D8421; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.2.2 Depending on data usage, you may modify this test
method but limit to modifications that improve performance
1.1 This test method covers the determination of per- and
whilestillmeetingorexceedingthemethodqualityacceptance
polyfluoroalkyl substances (PFASs) in aqueous matrices using
criteria. Modifications to the solvents, ratio of solvent to
liquid chromatography (LC) and detection with tandem mass
sample, or shortening the chromatographic run simply to save
spectrometry (MS/MS). These analytes are co-solvated by a
time are not allowed. Use Practice E2935 or similar statistical
1+1 ratio of sample and methanol then qualitatively and
tests to confirm that modifications produce equivalent results
quantitatively determined by this test method. Quantitation is
on non-interfering samples. In addition, use Guide E2857 or
by selected reaction monitoring (SRM) or sometimes referred
equivalent statistics to re-validate the modified test.
to as multiple reaction monitoring (MRM).
1.2.3 Analytedetectionsbetweenthemethoddetectionlimit
1.2 The method detection limit (MDL) (see Note 1) and
and the reporting limit are estimated concentrations. The
reportingrange(seeNote2)forthetargetanalytesarelistedin
reporting limit is based upon the concentration of the Level 1
Table1.Thetargetconcentrationforthereportinglimitforthis
calibration standard as shown in Table 5.
test method is an integer value that is calculated from the
1.3 The values stated in SI units are to be regarded as
concentration from the lowest standard from the final volume
standard. No other units of measurement are included in this
of the prepared sample. This value may be lower than the
standard.
calculated MDL due to sporadic PFAS hits due to PFAS
1.4 This standard does not purport to address all of the
contamination in consumables/collection tools used during
safety concerns, if any, associated with its use. It is the
samplecollectionandpreparation.Allsamplesshouldbetaken
responsibility of the user of this standard to establish appro-
at a minimal as duplicates in order to compare the precision
priate safety, health, and environmental practices and deter-
between the two prepared samples to help ensure the
mine the applicability of regulatory limitations prior to use.
concentration/positive result is reliable.
1.5 This international standard was developed in accor-
NOTE 1—The MDL is determined following the Code of Federal
dance with internationally recognized principles on standard-
Regulations (CFR), 40 CFR Part 136, Appendix B utilizing dilution and
ization established in the Decision on Principles for the
filtration. A detailed process determining the MDL is explained in the
Development of International Standards, Guides and Recom-
reference and is beyond the scope of this test method.
mendations issued by the World Trade Organization Technical
NOTE 2—Injection volume variations, and sensitivity of the instrument
used will change the reporting limit and ranges.
Barriers to Trade (TBT) Committee.
1.2.1 Recognizingcontinualadvancementsinthesensitivity
2. Referenced Documents
of instrumentation, advancements in column chromatography
and other processes not recognized here, the reporting limit 2.1 ASTM Standards:
may be lowered assuming the minimum performance require- D1129Terminology Relating to Water
ments of this test method at the lower concentrations are met. D1193Specification for Reagent Water
D2777Practice for Determination of Precision and Bias of
Applicable Test Methods of Committee D19 on Water
This test method is under the jurisdiction ofASTM Committee D19 on Water
andisthedirectresponsibilityofSubcommitteeD19.06onMethodsforAnalysisfor
Organic Substances in Water. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved May 1, 2022. Published June 2022. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2021. Last previous edition approved in 2021 as D8421–21. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D8421-22. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8421−22
D3856Guide for Management Systems in Laboratories 3.2.6 precursor ion, n—ion that reacts to form product ions
Engaged in Analysis of Water or undergoes specified neutral losses.
D4841Practice for Estimation of Holding Time for Water
3.2.7 production,n—ionformedastheproductofareaction
Samples Containing Organic and Inorganic Constituents
involving a precursor ion.
D5847Practice for Writing Quality Control Specifications
3.2.8 single (or selected) reaction monitoring (SRM),
for Standard Test Methods for Water Analysis
n—data acquired from one or more specific product ions
D8272Guide for Development and Optimization of D19
corresponding to m/z selected precursor ions recorded via two
Chemical Analysis Methods Intended for EPA Compli-
or more stages of mass spectrometry.
ance Reporting
3.2.9 tandem mass spectrometer, n—mass spectrometer de-
E694Specification for Laboratory Glass Volumetric Appa-
signed for mass spectrometry/mass spectrometry.
ratus
E2554Practice for Estimating and Monitoring the Uncer- 3.2.10 triple quadrupole mass spectrometer (triple quad or
tainty of Test Results of a Test Method Using Control
QQQ), n—tandem mass spectrometer comprising two trans-
Chart Techniques mission quadrupole mass spectrometers in series, with a
E2857Guide for Validating Analytical Methods (non-selecting) RF-only quadrupole (or other multipole) be-
E2935Practice for Evaluating Equivalence of Two Testing
tween them to act as a collision cell.
Processes
4. Summary of Test Method
2.2 Other Standards:
Code of Federal Regulations 40 CFR Part 136,Appendix B
4.1 The operating conditions presented in this test method
have been validated for use in the determination of PFASs in
3. Terminology
aqueous samples. Alternative instrument operating conditions
3.1 Definitions:
may be used provided data quality objectives are met. Follow
3.1.1 For definitions of terms used in this standard, refer to
the manufacturer’s instructions. The preparation process, as
Terminology D1129.
summarized in 4.2 and described in Section 14 may be
automated, but cannot be modified.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 collision cell, n—chamberintheionpathbetweenm/z
4.2 Samplesareshippedtothelabatatemperaturebetween
separation elements, or between ion source and the first
0°C and 6°C and analyzed within 28 days of collection. A
analyzer,intandemmassspectrometryinspaceconfigurations.
sample (5 mL) is collected and processed in the same collec-
tion tube in order to limit analyte loss; extra samples must be
3.2.2 continuing calibration verification (CCV), n—a mid-
collected for duplicates/triplicates and matrix spikes. All
range calibration standard which checks the continued validity
samples and associated QC samples are spiked with labeled
of the initial calibration of the instrument.
surrogates (QC samples such as laboratory control and matrix
3.2.3 mass spectrometry/mass spectrometry (MS/MS),
spike samples are additionally spiked with target PFASs) and
n—acquisition and study of the spectra of the product ions or
shaken for 2 minutes after adding 5 mL of methanol. The
precursor ions of m/z selected ions, or of precursor ions of a
samples are then filtered through a polypropylene filter.Acetic
selected neutral mass loss.
acid(~10µL)isaddedtoallthesamplestoadjusttopH~4and
3.2.3.1 Discussion—MS/MS can be accomplished using
analyzed by LC/MS/MS. If samples contain more than about
instruments incorporating more than one analyzer (tandem
1.0 g/L suspended or settled solids, (for example, sludge,
mass spectrometry in space) or in trap instruments (tandem
pretreatment, or wastewater influent) adjust to pH ~9 (adding
mass spectrometry in time).
~20 µL of ammonium hydroxide), shake for 2 minutes, filter,
3.2.4 multiple reaction monitoring (MRM), n—application
acidify to pH ~4 (~50 µL acetic acid), and then analyze by
of selected reaction monitoring to multiple product ions from
LC/MS/MS.
one or more precursor ions.
NOTE 3—Sludge in this test method is defined as sewage sample
containing between 0.1 and 2 % solids based upon a sample by weight.
3.2.5 per- and polyfluoroalkyl substances (PFAS),
NOTE 4—Since contact with surfaces may bias data, collect a 5.0-mL
n—synthetic organofluorine chemical compounds with mul-
sample in a graduated 15-mL polypropylene tube in the field so that the
tiple fluorine atoms that includes PFOA, PFOS, GenX, and
whole sample is processed in the lab. Once this 5.0-mL sample is spiked
many other chemicals.
accordingtothistestmethodandmethanolisadded,thesampleisfiltered
3.2.5.1 Discussion—PFAS have a hydrophobic and oleop- into another 15 mL polypropylene tube without analyte loss.
NOTE 5—For accurate volume, the weight of the 15-mLpolypropylene
hobic fluorinated “tail” and a hydrophilic “head” making them
tube may be taken before and after sampling. The density of water is
surfactants. They include the perfluoro sulfonic acids such as
assumed to be 1.0 g/mL unless the exact density of the water sample is
the perfluorooctanesulfonic acid (PFOS) and the perfluoro
known, then that conversion should be used.
carboxylic acids, such as the perfluorooctanoic acid (PFOA).
4.3 Most analytes are identified by comparing the SRM
PFOS and PFOAare persistent organic pollutants. The defini-
transitionanditsconfirmatorySRMtransitioncorrelatedtothe
tion does not include the mass labeled surrogates or internal
known standard SRM transition (Table 3) and quantitated
standards.
utilizing an external calibration. The retention times and ion
ratiosareshowninTable4foreachnativeanalyteandisotope.
Available from National Technical Information Service (NTIS), U.S. Depart-
The surrogates and some analytes only have one SRM transi-
ment of Commerce, 5285 Port Royal Road, Springfield, VA, 22161 or at http://
www.epa.gov/epawaste/hazard/testmethods/index.htm tion due to a less sensitive or non-existent secondary SRM
D8421−22
transition. As an additional quality-control measure, isotopi- qualified,backgroundsubtractionofblankcontaminationisnot
cally labeled surrogate (Table 1, Section 13.3) recoveries are allowed. It has become difficult to ensure consumables are
monitored. With external standard calibrations, there is no PFAS free at the lower concentrations (approximately at less
correction to the data based upon surrogate recoveries. than 30 ng/L) for the entire lot by testing only a very small
Alternatively, extract an isotopically labelled analog of each sub-sample. At a minimum duplicates/triplicates should be
analyte(isotopedilution),ifavailable,andcorrectforrecovery. taken of each sample to evaluate precision between the set.
Only exact isotopes of the native analytes may be used for
6.5 The LC system used should consist, as much as
isotope dilution correction. If a structurally different isotope is
practical, of sample solution or eluent contacting components
used to correct a native analyte this is called surrogate
free of PFAS of interest.
correctionandeithermustbeclearlystatedasperformedinthe
6.6 Polyethylene LC vial caps or any other target analyte
accompanying data report or not allowed. For isotope dilution,
free vial caps should be used.
the analog and the native compound concentrations (areas)
should be within 30% of each other to obtain more accurate
6.7 Polyethylene disposable pipettes or target analyte free
results.The final report issued lists the concentration of PFAS, pipettes should be used. All disposable pipettes should be
ifdetected,orasanon-detectattheRL,ifnotdetected,inng/L checked for release of target analytes of interest.
and the surrogate recoveries.
6.8 DegassersareimportanttocontinuousLCoperationand
NOTE 6—For greater accuracy in the isotope dilution method, add the
most commonly are made of fluorinated polymers. To enable
isotopes at the time sampling or allow the sample and isotope to
use,anisolatorcolumnshouldbeplacedafterthedegasserand
equilibrate for at least 48 hours prior to addition of methanol.
prior to the sample injection valve to separate the PFAS in the
5. Significance and Use
sample from the PFAS in the LC system.
5.1 PFAS are widely used in various industrial and com-
6.9 Electro Spray Ionization (ESI)—ESI should be heated
mercial products; they are persistent, bio-accumulative, and
andoptimizedforrecoveryofcomponentsanalyzedbythistest
ubiquitous in the environment. PFAS have been reported to
method. Using the suggested mobile phase, gradient, and
exhibit developmental toxicity, hepatotoxicity,
adequate column separation minimizes, or eliminates, quench-
immunotoxicity, and hormone disturbance. PFAS have been
ing and enhancing of signal. This method was validated using
detected in soils, sludges, surface, and drinking waters. This is
ESI, however other modes of ionization may be used provided
a quick, easy, and robust method to quantitatively determine
the detection limits and quality control acceptance criteria of
these compounds at trace levels in water matrices.
this method are met.
5.2 This test method has been validated using reagent water
and waters from sites that include landfill leachate, metal 7. Apparatus
finisher, POTW Effluent, Hospital, POTW Influent, Bus wash-
7.1 LC/MS/MS System:
ing station, Power Plant and Pulp and paper mill effluent for
7.1.1 Liquid Chromatography System—Acomplete LC sys-
selected PFAS, refer to the Precision and Bias (Section 17).
tem is required to analyze samples, this includes a sample
injection system, a solvent pumping system capable of mixing
6. Interferences
solvents, a sample compartment capable of maintaining re-
6.1 All glassware is washed in hot water (typically >45ºC)
quired temperature and a temperature-controlled column com-
with detergent and rinsed in hot water followed by distilled
partment. This test method was developed using a ternary
water. The glassware is then dried and heated in an oven
(Table2)pumpingsystem.AbinaryLCsystemmaybeusedby
(typically at 105ºC) for 15 to 30 minutes. All glassware is
adapting the ternary gradient to a binary system.ALC system
subsequently rinsed with methanol or acetonitrile.
that can perform at the flow rates, pressures, controlled
temperatures, sample volumes, and requirements of the stan-
6.2 All reagents and solvents should be pesticide residue
purity or higher to minimize interference. Avoid the use of dard shall be used.
7.1.2 Analytical Column—UHPLC CSH Phenyl-Hexyl, 2.1
PFAS containing caps.
×100mmand1.7µmparticlesizecolumn,oranycolumnthat
6.3 Matrix interferences may be caused by contaminants in
achieves adequate resolution may be used.The retention times
the sample. The extent of matrix interferences varies consid-
andorderofelutionmaychangedependingonthecolumnused
erably depending on variations of the sample matrices. Sepa-
and needs to be monitored.
ration of individual components by the LC is vital in minimi-
7.1.3 Isolator Column—A reverse phase C18 column is
zationofinterferences.Shorteningofruntimessimplytospeed
used to separate the target analytes in the LC system and
analysis should be avoided, unless interferences are known to
solventsfromthetargetanalytesintheanalyticalsample.Place
be absent.
the column between the solvent mixing chamber and the
6.4 Contaminants have been found in reagents, glassware,
injector sample loop.
tubing, glass disposable pipettes, filters, degassers, and other
7.2 Tandem Mass Spectrometer System—A MS/MS system
apparatus and consumables that release PFAS. All these
capable of multiple reaction monitoring (MRM) analysis or
materials and supplies must be routinely demonstrated to be
anysystemthatiscapableofperformingattherequirementsin
free from interferences by analyzing laboratory reagent blanks
this test method.
under the same conditions as the samples. If found, measures
should be taken to remove the contamination or data should be 7.3 Filtration Device:
D8421−22
TABLE 1 Analyte List with Method Detection Limit and Reporting Range
MDL Range
Analyte Name Acronym CAS Number
(ng/L) (ng/L)
Perfluorotetradecanoic acid PFTreA 376-06-7 8.2 10-400
Perfluorotridecanoic acid PFTriA 72629-94-8 17.2 10-400
Perfluorododecanoic acid PFDoA 307-55-1 6.6 10-400
Perfluoroundecanoic acid PFUnA 2058-94-8 3.9 10-400
Perfluorodecanoic acid PFDA 335-76-2 3.4 10-400
Perfluorononanoic acid PFNA 375-95-1 5.2 10-400
Perfluorooctanoic acid PFOA 335-67-1 2.5 10-400
Perfluoroheptanoic acid PFHpA 375-85-9 5.9 10-400
Perfluorohexanoic acid PFHxA 307-24-4 2.1 10-400
Perfluoropentanoic acid PFPeA 2706-90-3 13.0 50-1000
Perfluorobutanoic acid PFBA 375-22-4 17.1 50-1000
Perfluorodecanesulfonic acid PFDS 335-77-3 1.6 10-400
Perfluorononanesulfonic acid PFNS 68259-12-1 1.2 10-400
Perfluorooctanesulfonic acid PFOS 1763-23-1 4.4 10-400
Perfluoroheptanesulfonic acid PFHpS 375-92-8 2.7 10-400
Perfluorohexanesulfonic acid PFHxS 355-46-4 2.3 10-400
Perfluoropentanesulfonic acid PFPeS 2706-91-4 2.7 10-400
Perfluorobutanesulfonic acid PFBS 375-73-5 3.3 10-400
Perfluorooctanesulfonamide PFOSA 754-91-6 2.2 10-400
8:2 Fluorotelomer sulfonic acid 8:2 FTS 39108-34-4 4.5 10-400
6:2 Fluorotelomer sulfonic acid 6:2 FTS 27619-97-2 2.7 10-400
4:2 Fluorotelomer sulfonic acid 4:2 FTS 757124-72-4 3.2 10-400
N-Ethylperfluorooctanesulfonamidoacetic acid NEtFOSAA 2991-50-6 2.6 10-400
N-Methylperfluorooctanesulfonamidoacetic acid NMeFOSAA 2355-31-9 1.3 10-400
Perfluorododecanesulfonic acid PFDoS 79780-39-5 2.2 10-400
N-Methylperfluorooctanesulfonamide NMeFOSA 31506-32-8 2.1 10-400
N-Ethylperfluorooctanesulfonamide NEtFOSA 4151-50-2 1.8 10-400
N-Methylperfluorooctanesulfonamidoethanol NMeFOSE 24448-09-7 3.1 10-400
N-Ethylperfluorooctanesulfonamidoethanol NEtFOSE 1691-99-2 2.7 10-400
Hexafluoropropylene oxide dimer acid HFPO-DA 13252-13-6 3.7 10-400
4,8-dioxa-3H-perfluorononanoic acid ADONA 919005-14-4 2.1 10-400
9-chlorohexadecafluoro-3-oxanonane-1-sulfonic acid 9Cl-PF3ONS 756426-58-1 2.7 10-400
11-chloroeicosafluoro-3-oxaundecane-1-sulfonic acid 11Cl-PF3OUdS 763051-92-9 2.2 10-400
Pentafluorpropanoic acid PFPrA 422-64-0 20.3 50-1000
Perfluoro-3,6-dioxaheptanoic acid NFDHA 151772-58-6 3.7 10-400
Perfluoro(2-ethoxyethane) sulfonic acid PFEESA 113507-82-7 2.2 10-400
Perfluoro-3-methoxypropanoic acid PFMPA 377-73-1 2.6 10-400
Perfluoro-4-methoxybutanoic acid PFMBA 863090-89-5 2.2 10-400
2H,2H,3H,3H-Perfluorohexanoic Acid 3:3 FTCA 356-02-05 3.7 10-400
2H,2H,3H,3H-Perfluorooctanoic Acid 5:3 FTCA 914637-49-3 3.0 10-400
2H,2H,3H,3H-Perfluorodecanoic acid 7:3 FTCA 812-70-4 1.5 10-400
2H-perfluoro-2-octenoic acid FHUEA 70887-88-6 2.5 10-400
2H-perfluoro-2-decenoic acid FOUEA 70887-84-2 2.9 10-400
A
Lithium Bis(trifluoromethane)sulfonimide HQ-115 90076-65-6 9.0 10-400
Surrogates
Perfluoro-n-[ C ]butanoic acid MPFBA NA NA 10-400
Perfluor0-n-[ C ]pentanoic acid M5PFPeA NA NA 10-400
Perfluoro-n-[1,2,3,4,6- C ]hexanoic acid M5PFHxA NA NA 10-400
Perfluoro-n-[1,2,3,4- C ]heptanoic acid M4PFHpA NA NA 10-400
Perfluoro-n-[ C ]octanoic acid M8PFOA NA NA 10-400
Perfluoro-n-[ C ]nonanoic acid M9PFNA NA NA 10-400
Perfluoro-n-[1,2,3,4,5,6- C ]decanoic acid M6PFDA NA NA 10-400
Perfluoro-n-[1,2,3,4,5,6,7- C ]undecanoic acid M7PFUnA NA NA 10-400
Perfluoro-n-[1,2- C ]dodecanoic acid MPFDoA NA NA 10-400
Perfluoro-n-[1,2- C ]tetradecanoic acid M2PFTreA NA NA 10-400
Perfluoro-1-[ C ]octanesulfonamide M8FOSA NA NA 10-400
N-methyl-d -perfluoro-1-octanesulfonamidoacetic acid D3-N-MeFOSAA NA NA 10-400
N-ethyl-d -perfluoro-1-octanesulfonamidoacetic acid D5-N-EtFOSAA NA NA 10-400
N-methyl-d -perfluoro-1-octanesulfanamide d-N-MeFOSA NA NA 10-400
N-ethyl-d -perfluoro-1-octanesulfanamide d-N-EtFOSA NA NA 10-400
2-(N-methyl-d -perfluoro-1-octanesulfonamido)ethan-d4-ol d7-N-MeFOSE NA NA 10-400
2-(N-ethyl-d -perfluoro-1-octanesulfonamido)ethan-d4-ol D9-N-EtFOSE NA NA 10-400
2,3,3,3-Tetrafluoro-2-(1,1,2,2,3,3,3-heptafluoropropoxy- C - MHFPO-DA NA NA 10-400
propanoic acid
1H,1H,2H,2H-perfluoro-1-[1,2- C ]hexane sulfonate M4:2FTS NA NA 10-400
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-octane sulfonate M6:2FTS NA NA 10-400
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-decane sulfonate M8:2FTS NA NA 10-400
Perfluoro-1-[ C ]octanesulfonate M8PFOS NA NA 10-400
Perfluoro-1-[2,3,4- C ]butanesulfonate MPFBS NA NA 10-400
Perfluoro-1-[1,2,3- C ]hexanesulfonate M3PFHxS NA NA 10-400
A
The Lithium is just the counter ion, report only Bis(trifluoromethane)sulfonimide.
D8421−22
TABLE 2 Gradient Conditions for a Ternary Pumping System
95 % Water:
Time Flow 95 % Water: 5 % Acetonitrile,
Acetonitrile %
(min) (mL/min) 5 % Acetonitrile % 400 mM
Ammonium Acetate %
00.3 95 0 5
1 0.3 75 20 5
6 0.3 50 45 5
13 0.3 15 80 5
14 0.4 0 95 5
17 0.4 0 95 5
18 0.4 95 0 5
21 0.4 95 0 5
7.3.1 Hypodermic Syringe—A luer-lock tip glass syringe 8.12 Acetonitrile (CAS #75-05-8).
capable of holding a syringe driven filter unit.
8.13 Methanol (CAS #67-56-1).
7.3.1.1 A 10-mL Lock Tip Glass Syringe size is recom-
8.14 Ammonium acetate (CAS #631-61-8).
mended in this test method.
7.3.2 Filter Unit—Polypropylene syringe-driven filter units 8.15 Acetic acid (CAS #64-19-7).
(0.2 µm) or equivalent, demonstrated contaminant free below
8.16 2-Propanol (isopropyl alcohol, CAS #67-63-0).
1/2 MRL.
8.17 Ammonium hydroxide (CAS #1336-21-6).
8. Reagents and Materials
8.18 PFASs Standards —Refer to Table 1 for the complete
8.1 Purity of Reagents—High Performance Liquid Chroma- analyte list and CAS numbers. These may be purchased from
tography (HPLC) pesticide residue analysis and spectropho- a commercial supplier individually or some as a mixture.
tometry grade chemicals shall be used in all tests. Unless
9. Hazards
indicated otherwise, it is intended that all reagents shall
conform to the Committee on Analytical Reagents of the
9.1 Precaution—The toxicity or carcinogenicity of chemi-
American Chemical Society. Other reagent grades may be
cals used in this test method has not been precisely defined;
used provided they are first determined to be of sufficiently
each chemical should be treated as a potential health hazard,
highpuritytopermittheirusewithoutaffectingtheaccuracyof
and exposure to these chemicals should be minimized. Each
the measurements.
laboratory is responsible for maintaining awareness of OSHA
regulations regarding safe handling of chemicals used in this
8.2 Purity of Water—Unless otherwise indicated, references
test method.
towatershallbeunderstoodtomeanreagentwaterconforming
toType 1 of Specification D1193. It shall be demonstrated that
9.2 Warning—The compound analytes in this test method
this water does not contain contaminants at concentrations
have been classified as known or suspected human or mam-
sufficient to interfere with the analysis.
malian carcinogens. Pure standards and stock solutions should
be handled in a hood or glovebox.
8.3 Gases—Ultrapure nitrogen and argon.
8.4 Vials—Greater than 1.0 mLAmber glass or polypropyl-
10. Sampling
ene autosampler vials.
10.1 Sampling and Preservation—Avoid sample containers
8.5 Polyethylene autosampler vial caps, or equivalent.
and contact with surfaces of fluorinated polymers or PFAS
contaminateditems.Collectfieldblanksthatareexposedtothe
8.6 Syringe—10 or 25-mL filter-adaptable glass syringe
same field conditions as samples and analyze according to this
with luer lock.
test method to assess the potential for field contamination.
8.7 Polypropylene Tubes—15 and 50 mL conical with cali-
Collect 5 6 0.5 mL samples, duplicates/triplicates, matrix
bration lines.
spikes and field blanks in graduated 15 mL polypropylene
8.8 pH paper (pH range 1–14).
tubes. For greater accuracy, the tubes should be pre-weighed
andweighedaftersamplinginordertoachieveanexactweight
8.9 Class A Volumetric Glassware.
of the sample. This weight is then used to calculate volume
8.10 Pipette tips—Polypropylene pipette tips free of release
with the assumed density of the water sample as 1.0 g/mL.
agents or low retention coating of various sizes.
Conventional sampling practices should be followed with the
8.11 Polyethylene Disposable Pipettes.
caution that PFASs containing products may be present in
sampling equipment. All sampling equipment and supplies
shall be PFAS free to prevent contamination of the samples.
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, PFASsstandardsmaybedifficulttofind,somesourcesofPFASsstandardsthat
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma- havebeenfoundsuitableforusewerefromAldrichChemicalCompany,Wellington
copeial Convention, Inc. (USPC), Rockville, MD. LaboratoriesInc.,andWakoLaboratory.Standardsfromothervendorsmaybeused.
D8421−22
EPA Publication SW-846, Guide D3856, and Practices E694 second source and samples shall be in a 50:50 methanol:water
may be used as guides. Ship samples on ice with a trip blank. solution containing 0.1 % acetic acid. In the case of extreme
The temperature of the samples upon receipt at the laboratory
concentration differences amongst samples, it is wise to ana-
shouldbelessthan6°C.Ifthereceivingtemperatureisgreater
lyze a blank after a concentrated sample and before a dilute
than6°C,thesampletemperatureisnotedinthecasenarrative
sample to eliminate carry-over of analytes from sample injec-
accompanying the data. Samples should be stored refrigerated
tiontosampleinjection.Ifaflowthroughneedledesignisused
between 0°C and 6°C from the time of collection until
carry-over should not be a problem. The gradient conditions
analysis. Analyze the sample within 28 days of collection.
for LC are shown in Table 2. To ensure chromatographic
Holdingtimemayvarydependingonthematrixandindividual
separation between the targeted analytes and any unknown
laboratoriesshoulddeterminetheholdingtimeintheirmatrix.
non-targeted potentially interfering compounds, avoid shorten-
ingtheanalysistimesimplytospeedtheanalysis.RefertoFig.
11. Preparation of LC/MS/MS
1 as an example chromatogram of 24 surrogates showing
11.1 LC Chromatograph Operating Conditions:
resolution with limited coelution.
11.1.1 Injections of all standards and samples are made at a
10–30-µL volume. Other injection volumes may be used to 11.2 LC Sample Manager Conditions:
optimize conditions. Calibration Standards, reagent blanks,
11.2.1 Needle Wash Solvent—60 % acetonitrile/40 %
2-propanol. Eight second wash time before and after injection.
Instrumentmanufacturer’sspecificationsshouldbefollowedin
Guides to help determine holding times can be found at: http://www.epa.gov/
order to eliminate sample carry-over.
esd/cmb/research/bs_033cmb06.pdf (2014) and Practice D4841.
FIG. 1Example Chromatogram of 24 Surrogates, at the Level 1 Calibration Concentration, Overlayed Showing Resolution with Limited
Coelution
D8421−22
11.2.2 Temperatures—Column, 35 °C; Sample 12.2.2 Aliquots of SolutionA(Calibration Level 9) are then
compartment, 15°C. dilutedwith50:50methanol:watercontaining0.1%aceticacid
11.2.3 Seal Wash—Solvent: 50% water⁄50 % methanol;
to prepare the desired calibration levels (Table 5) in polypro-
Time: 5 minutes. pylene LC vials. For best results, use the calibration standards
within 24 hours of preparation. Prepare the end CCV at a
11.3 Mass Spectrometer Parameters:
mid-level concentration in a separate LC vial. All calibration
11.3.1 To acquire the maximum number of data points per
standards should be used only once because the analyte
SRM channel while maintaining adequate sensitivity, optimize
concentration in the vial may change after the vial cap is
the tune parameters according to instrument manufacturer
pierced.Changingthecapsimmediatelyaftertheinjectionmay
instructions. Each peak requires a minimum of 10 scans per
alleviate this problem, however, this should be verified in each
peak for adequate quantitation. This test method containing
laboratory. Calibration standards do not need to be filtered.
surrogates, which are select isotopically labeled PFAS, and the
12.2.3 Incorporate a second source standard, if available.
targeted PFAS may be split into multiple MRM acquisition
The second source standard should be analyzed near the
functions to optimize sensitivity. Retention times, and primary
midpoint of the calibration range to verify that the standards
and confirmatory transitions are shown in Table 3. Retention
used are within 630 % of the expected concentration.
times will vary between columns and gradient used. Each
manufacturer may have different terminology to represent Currently, a second source from a different vendor may not be
various mass spectrometer settings, and different set values readily available for all target analytes. In this case, a second
depending on the manufacturer and instrument model. Please lot number from the same vendor may be used.
refer to the manufacturer’s instructions in optimizing detector
12.2.4 Inject each standard and obtain its chromatogram.
settings, including collision energies and cone voltages. Data
The instrument software collects the primary and confirmatory
for this method was collected using electrospray ionization
SRM transitions of each analyte at the specified retention
(ESI) operated in negative mode. In recognition of the ad-
times. Calibration software conducts the quantitation of the
vancement of LCMSMS instrumentation, other MS operating
target analytes and surrogates using the primary SRM transi-
conditions, including ionization techniques may be used pro-
tion. The ratios of the primary/confirmatory MRM transition
vided the quality control criteria of the method is met.
area counts will vary depending on the individual tuning
conditions. Refer to Table 4 for retentionTimes and Ion ratios.
12. Calibration and Standardization
For confirmation of analyte identity, the primary/confirmatory
12.1 The mass spectrometer is calibrated as in accordance
ratio shall be within 30 % of the individual ratios established
withmanufacturer’sspecificationspriortoanalysis.Prepareall
duringtheinitialcalibration.Theaverageionratioiscalculated
calibration solutions using Class A volumetric glassware
for each batch from the initial calibration levels.
(E694).
12.2.5 Depending on sensitivity and sample dependent ma-
trixinterference,theconfirmatorySRMtransitionmaybeused
12.2 Calibration and Standardization—Analyze up to nine
calibration standards containing the PFAS and surrogates prior astheprimarySRMtransitionforquantitationduringanalysis.
to analysis as shown in Table 5.The calibration stock standard
12.2.6 The calibration software manual or the instrument
solution is prepared from the target and surrogate spike
manufacturer should be consulted to ensure correct software
solutions. Stock standard SolutionAcontaining the PFAS and
use. The quantitation method is set using the peak areas in ppt
surrogatesispreparedatCalibrationLevel9concentrationand
(ng/L) units. Concentrations may be calculated using the data
aliquots of that solution are diluted to prepare Calibration
system software to generate linear regression or quadratic
Levels1through8.Thefollowingstepswillproducestandards
calibration curves. Forcing the calibration curve through the
with the concentration values shown in Table 5. The analyst is
origin (X = 0, Y = 0) is not recommended.
responsible for recording initial component weights carefully
12.2.7 Either of two procedures may be used to determine
when working with pure materials and correctly carrying the
calibration function acceptability for linear and non-linear
weights through the dilution calculations. At a minimum, five
curves. These include refitting the calibration data back to the
calibration levels are required when using a linear calibration
model. Both % Error and Relative Standard Error (RSE)
and six calibration levels are required when using a quadratic
evaluate the difference between the measured and the true
calibration curve.An initial nine-points may be used to enable
amounts or concentrations used to create the model.
dropping the lower calibration points if the instrument cannot
12.2.7.1 Calculation of % Error is shown as Eq 1. Percent
achieve low detection limits on certain PFAS. This will allow
error between the calculated and expected amounts should be
at least a five or six-point calibration curve per analyte to be
≤ 30% for all standards.
obtained.
'
x 2 x
12.2.1 Calibration Stock Standard Solution A (Calibration
i i
%Error 5 3100 (1)
Level 9, Table 5) is prepared from the target and surrogate x
i
spike solutions. Transfer 500 µL of the surrogate spike (20
where:
µg/L), 500 µL of PFAS Target Spike I and 500 µL of PFAS
’
x = measured amount of analyte at calibration level i,in
i
Target Spike II (refer to Table 7) to a 50-mL volumetric flask
mass or concentration units, and
and dilute to 50-mL volume with 50:50 methanol:water con-
x = true amount of analyte at calibration level i, in mass or
i
taining 0.1 % acetic acid. Ensure that the analytes are solubi-
concentration units.
lized in the Level 9 standard.
D8421−22
TABLE 3 Transitions for Target Analytes and Surrogates
Analyte Name Acronym CAS Number Primary Ion Transition Confirmation Ion Transition
Perfluorotetradecanoic acid PFTreA 376-06-7 712.9→ 668.9 712.9→ 168.9
Perfluorotridecanoic acid PFTriA 72629-94-8 662.9→ 618.9 662.9→ 168.9
Perfluorododecanoic acid PFDoA 307-55-1 612.9→ 568.9 612.9→ 168.9
Perfluoroundecanoic acid PFUnA 2058-94-8 562.9→ 519 562.9→ 269
Perfluorodecanoic acid PFDA 335-76-2 512.9→ 469 512.9→ 218.9
Perfluorononanoic acid PFNA 375-95-1 462.9→ 419 462.9→ 218.9
Perfluorooctanoic acid PFOA 335-67-1 412.9→ 369 412.9→ 168.9
Perfluoroheptanoic acid PFHpA 375-85-9 362.9→ 318.9 362.9→ 168.9
Perfluorohexanoic acid PFHxA 307-24-4 312.9→ 269 312.9→ 118.9
Perfluoropentanoic acid PFPeA 2706-90-3 262.9→ 218.9 NA
Perfluorobutanoic acid PFBA 375-22-4 212.9→168.9 NA
Perfluorodecanesulfonic acid PFDS 335-77-3 598.9→ 79.9 598.9→ 98.9
Perfluorononanesulfonic acid PFNS 68259-12-1 548.9→ 79.9 548.9→ 98.9
Perfluorooctanesulfonic acid PFOS 1763-23-1 498.9→ 79.9 498.9→ 98.9
Perfluoroheptanesulfonic acid PFHpS 375-92-8 448.9→ 79.9 448.9→ 98.9
Perfluorohexanesulfonic acid PFHxS 355-46-4 398.9→ 79.9 398.9→ 98.9
Perfluoropentanesulfonic acid PFPeS 2706-91-4 348.9→ 79.9 348.9→ 98.9
Perfluorobutanesulfonic acid PFBS 375-73-5 298.9→ 79.9 298.9→ 98.9
Perfluorooctanesulfonamide PFOSA 754-91-6 497.9→ 77.9 NA
8:2 Fluorotelomer sulfonic acid 8:2 FTS 39108-34-4 526.9→ 506.9 526.9→ 80.9
6:2 Fluorotelomer sulfonic acid 6:2 FTS 27619-97-2 427→ 407 427→ 80.9
4:2 Fluorotelomer sulfonic acid 4:2 FTS 757124-72-4 326.9→ 306.9 326.9→ 80.9
N-Ethylperfluorooctanesulfonamidoacetic acid NEtFOSAA 2991-50-6 584→ 419 584→ 482.9
N-Methylperfluorooctanesulfonamidoacetic NMeFOSAA 2355-31-9 569.9→ 419 569.9→ 482.9
acid
Perfluorododecanesulfonic acid PFDoS 79780-39-5 698.9→ 79.9 698.9→ 98.9
N-Methylperfluorooctanesulfonamide NMeFOSA 31506-32-8 511.9→ 168.9 511.9→ 218.9
N-Ethylperfluorooctanesulfonamide NEtFOSA 4151-50-2 525.9→ 168.9 525.9→ 218.9
N-Methylperfluorooctanesulfonamidoethanol NMeFOSE 24448-09-7 616→ 58.9 NA
N-Ethylperfluorooctanesulfonamidoethanol NEtFOSE 1691-99-2 630→ 58.9 NA
Hexafluoropropylene oxide dimer acid HFPO-DA 13252-13-6 285→ 168.9 285→ 184.9
4,8-dioxa-3H-perfluorononanoic acid ADONA 919005-14-4 376.9→ 251 376.9→ 84.9
9-chlorohexadecafluoro-3-oxanonane-1- 9Cl-PF3ONS 756426-58-1 530.9→ 350.9 532.9→ 352.9
sulfonic acid
11-chloroeicosafluoro-3-oxaundecane-1- 11Cl-PF3OUdS 763051-92-9 630.8→ 450.9 632.8→ 452.9
sulfonic acid
Pentafluorpropanoic acid PFPrA 422-64-0 162.9→ 118.9 NA
Perfluoro-3,6-dioxaheptanoic acid NFDHA 151772-58-6 295→ 200.9 295→ 84.9
Perfluoro(2-ethoxyethane)sulfonic acid PFEESA 113507-82-7 314.9→ 134.9 314.9→ 82.9
Perfluoro-3-methoxypropanoic acid PFMPA 377-73-1 228.9→ 84.9 NA
Perfluoro-4-methoxybutanoic acid PFMBA 863090-89-5 278.9→ 84.9 NA
2H,2H,3H,3H-Perfluorohexanoic Acid 3:3 FTCA 356-02-05 241→ 176.9 241→ 116.9
2H,2H,3H,3H-Perfluorooctanoic Acid 5:3 FTCA 914637-49-3 340.9→ 216.9 340.9→237
2H,2H,3H,3H-Perfluorodecanoic acid 7:3 FTCA 812-70-4 440.9→ 337 440.9→ 316.9
2H-perfluoro-2-octenoic acid FHUEA 70887-88-6 356.9→ 292.9 NA
2H-perfluoro-2-decenoic acid FOUEA 70887-70-4 456.9→ 393 NA
Lithium Bis(trifluoromethane)sulfonimide HQ-115 90076-65-6 279.9→146.9 279.9→210.9
Surrogates
Perfluoro-n-[ C ]butanoic acid MPFBA NA 216.9→ 171.9 NA
Perfluoro-n-[ C ]pentanoic acid M5PFPeA NA 267.9→ 222.9 NA
Perfluoro-n-[1,2,3,4,6- C ]hexanoic acid M5PFHxA NA 317.9→ 272.9 NA
Perfluoro-n-[1,2,3,4- C ]heptanoic acid M4PFHpA NA 366.9→ 321.9 NA
Perfluoro-n-[ C ]octanoic acid M8PFOA NA 421→ 376 NA
Perfluoro-n-[ C ]nonanoic acid M9PFNA NA 471.9→ 426.9 NA
Perfluoro-n-[1,2,3,4,5,6- C ]decanoic acid M6PFDA NA 518.9→ 473.9 NA
Perfluoro-n-[1,2,3,4,5,6,7- C7]undecanoic M7PFUnA NA 569.9→ 524.9 NA
acid
Perfluoro-n-[1,2- C ]dodecanoic acid MPFDoA NA 614.9→ 569.9 NA
Perfluoro-n-[1,2- C ]tetradecanoic acid M2PFTreA NA 714.9→ 669.9 NA
Perfluoro-1-[ C ]octanesulfonamide M8FOSA NA 505.9→ 77.9 NA
N-methyl-d -perfluoro-1- D3-N-MeFOSAA NA 572.9→ 418.9 NA
octanesulfonamidoacetic acid
N-ethyl-d -perfluoro-1- D5-N-EtFOSAA NA 589→ 418.9 NA
octanesulfonamidoacetic acid
N-methyl-d -perfluoro-1-octanesulfanamide d-N-MeFOSA NA 514.9→ 168.9 NA
N-ethyl-d -perfluoro-1-octanesulfanamide d-N-EtFOSA NA 531→168.9 NA
2-(N-ethyl-d -perfluoro-1- d7-N-MeFOSE NA 623→ 58.9 NA
octanesulfonamido)ethan-d4-ol
2-(N-methyl-d -perfluoro-1- D9-N-EtFOSE NA 639→ 58.9 NA
octanesulfonamido)ethan-d4-ol
2,3,3,3-Tetrafluoro-2-(1,1,2,2,3,3,3- MHFPO-DA NA 287→ 168.9 NA
heptafluoropropoxy- C -propanoic acid
1H,1H,2H,2H-perfluoro-1-[1,2- C ]hexane M4:2FTS NA 328.9→ 308.9 NA
A
sulfonate 328.9→ 80.9
D8421−22
TABLE3 Continued
Analyte Name Acronym CAS Number Primary Ion Transition Confirmation Ion Transition
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-octane M6:2FTS NA 428.9→ 408.9 NA
A
sulfonate 428.9→ 80.9
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-decane M8:2FTS NA 528.9→ 508.9 NA
A
sulfonate 528.9→ 80.9
Perfluoro-1-[ C ]octanesulfonate M8PFOS NA 506.9→ 79.9 NA
Perfluoro-1-[2,3,4- C ]butanesulfonate M3PFBS NA 301.9→ 79.9 NA
Perfluoro-1-[1,2,3- C ]hexanesulfonate M3PFHxS NA 401.9→ 79.9 NA
A
If high concentrations of the native FTS interfere with Isotope of the FTSs, this transition should be used. It is not as sensitive, but the interference/high bias is removed.
12.2.7.2 Calculation of Relative Standard Error (RSE – 13.2 If a laboratory has not performed the test before or if
expressed as %) is shown in Eq 2. The RSE acceptance limit there has been a major change in the measurement system, for
criterionforthecalibrationmodelisthesameastheRSDlimit. example, new analyst, new instrument, etc., an instrument
qualification study including method detection limit (MDL),
n
' 2
x 2 x
i i
calibration range determination and precision and bias deter-
RSE 5100 3Œ ⁄ ~n 2 p! (2)
F G
(
x
i51
i
mination shall be performed to demonstrate laboratory capa-
bility.
where:
13.2.1 Analyze at least four replicates of a spiked water
x = true amount of analyte in calibration level i, in mass or
i
sample containing the analytes and surrogates at a prepared
concentration units,
’
sample concentration in the range of Calibration Levels 4–7.
x = measured amount of analyte in calibration level i,in
i
Calibration Level 6 was used to establish the QC acceptance
mass or concentration units,
criteria in this test method. Take each replicate through the
p = number of terms in the fitting equation (average = 1,
complete analytical test method including any sample manipu-
linear = 2, quadratic = 3, cubic = 4), and
lation and pretreatment steps.
n = number of calibration points.
13.2.2 Calculate the mean (average) percent recovery and
12.2.8 The retention time window of an unknown shall be
relative standard deviation (RSD) of the four values and
within5%ofthe retention time of the analyte in a midpoint
comparetotheacceptablerangesoftheQCacceptancecriteria
calibration standard. If this is not the case, re-analyze the
for the Initial Demonstration of Performance in Table 6.
calibration curve to determine if there was a shift in retention
13.2.3 Repeat until the single operator precision and mean
time during the analysis. If the retention time of the known
recovery are within the limits in Table 6. If a concentration
standard is correct, and the retention time of the peak in the
other than the recommended concentration is used, refer to
sampleisstillincorrectinthesample,refertotheanalyteasan
PracticeD5847forinformationonapplyingtheFtestandttest
unknown.
in evaluating the acceptability of the mean and standard
12.2.9 Analyze a CCV at the end of each batch of 20
deviation.
samples, 20 samples does not include QC samples. This
13.2.3.1 The QC acceptance criteria for the Initial Demon-
interval may be tightened according to the laboratory’s QA
stration of Performance in Table 6 were generated from the
programoraccreditationrequirements.ThisendCCV,inanew
combined single-laboratory data from reagent water as shown
never pierced sealed vial, should come from the same stock
inSection17.Laboratoriesshouldgeneratetheirownin-house
calibration standard solution that was used to generate the
QCacceptancecriteriawhichmeetorexceedthecriteriainthis
initial calibration curve.The concentration of each analyte and
test method. References on how to generate QC acceptance
surrogate in the end CCV standard shall be within 30 % of the
criteria are Practices D2777, D5847, and E2554.
expectedconcentration.Iftheconcentrationisnotwithin30%,
corrective action is performed and either all samples in the
13.3 Surrogate Spiking Solution:
batcharere-analyzedagainstanewcalibrationcurveorqualify
13.3.1 A surrogate spiking solution containing each isoto-
affected samples. If the analyst inspects the vial containing the
pically labeled PFAS are added to all samples; including
end CCV and notices a probable cause for the failure, a new
method blanks, duplicates, laboratory control samples, matrix
end CCVmay be prepared and analyzed. If this new end CCV
spikes, and reporting limit checks. A stock surrogate spiking
is within 30 % from the expected concentration for the target
solutionispreparedat20µg⁄Lin95%MeOH:5%water.Add
analytes and surrogates, the results do not need to be qualified.
40 µL of this spiking solution into a 5-mL water sample for a
concentration of 160 ng/L of the surrogate in the sample. The
13. Quality Control
results obtained for the surrogate recoveries shall fall within
13.1 Quality control (QC) requirements include the initial the limits of Table 6. If the limits are not met, the affected
resultsshallbequalifiedwithanindicationthattheydonotfall
demonstration of laboratory capability followed by routine
analysesoflaboratoryreagentblanks,fieldreagentblanks,and within the performance criteria of the test method.
laboratory fortified blanks, and matrix spikes. The laboratory 13.3.1.1 Preparethesurrogatespikingsolutionmixcontain-
must maintain records to document the quality of the data ing all twenty-four surrogates shown in Table 1 with 95 %
generated.The criteria in this section were used for, or derived MeOH: 5 % water. It should be replaced every year if not
from, the method validation. previously discarded for quality-control failure.
D8421−22
TABLE 4 Retention Times and Ion Ratios for Target An
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D8421 − 21 D8421 − 22
Standard Test Method for
Determination of Per- and Polyfluoroalkyl Substances
(PFAS) in Aqueous Matrices by Co-solvation followed by
Liquid Chromatography Tandem Mass Spectrometry (LC/
MS/MS)
This standard is issued under the fixed designation D8421; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the determination of per- and polyfluoroalkyl substances (PFASs) in aqueous matrices using liquid
chromatography (LC) and detection with tandem mass spectrometry (MS/MS). These analytes are co-solvated by a 1+1 ratio of
sample and methanol then qualitatively and quantitatively determined by this test method. Quantitation is by selected reaction
monitoring (SRM) or sometimes referred to as multiple reaction monitoring (MRM).
1.2 The method detection limit (MDL) (see Note 1) and reporting range (see Note 2) for the target analytes are listed in Table 1.
The target concentration for the reporting limit for this test method is an integer value that is calculated from the concentration
from the lowest standard from the final volume of the prepared sample. This value may be lower than the calculated MDL due
to sporadic PFAS hits due to PFAS contamination in consumables/collection tools used during sample collection and preparation.
All samples should be taken at a minimal as duplicates in order to compare the precision between the two prepared samples to help
ensure the concentration/positive result is reliable.
NOTE 1—The MDL is determined following the Code of Federal Regulations (CFR), 40 CFR Part 136, Appendix B utilizing dilution and filtration. A
detailed process determining the MDL is explained in the reference and is beyond the scope of this test method.
NOTE 2—Injection volume variations, and sensitivity of the instrument used will change the reporting limit and ranges.
1.2.1 Recognizing continual advancements in the sensitivity of instrumentation, advancements in column chromatography and
other processes not recognized here, the reporting limit may be lowered assuming the minimum performance requirements of this
test method at the lower concentrations are met.
1.2.2 Depending on data usage, you may modify this test method but limit to modifications that improve performance while still
meeting or exceeding the method quality acceptance criteria. Modifications to the solvents, ratio of solvent to sample, or shortening
the chromatographic run simply to save time are not allowed. Use Practice E2935 or similar statistical tests to confirm that
modifications produce equivalent results on non-interfering samples. In addition, use Guide E2857 or equivalent statistics to
re-validate the modified test.
1.2.3 Analyte detections between the method detection limit and the reporting limit are estimated concentrations. The reporting
limit is based upon the concentration of the Level 1 calibration standard as shown in Table 5.
This test method is under the jurisdiction of ASTM Committee D19 on Water and is the direct responsibility of Subcommittee D19.06 on Methods for Analysis for
Organic Substances in Water.
Current edition approved Nov. 1, 2021May 1, 2022. Published December 2021June 2022. Originally approved in 2021. Last previous edition approved in 2021 as
D8421 – 21. DOI: 10.1520/D8421-21.10.1520/D8421-22.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8421 − 22
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1129 Terminology Relating to Water
D1193 Specification for Reagent Water
D2777 Practice for Determination of Precision and Bias of Applicable Test Methods of Committee D19 on Water
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
D4841 Practice for Estimation of Holding Time for Water Samples Containing Organic and Inorganic Constituents
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
D8272 Guide for Development and Optimization of D19 Chemical Analysis Methods Intended for EPA Compliance Reporting
E694 Specification for Laboratory Glass Volumetric Apparatus
E2554 Practice for Estimating and Monitoring the Uncertainty of Test Results of a Test Method Using Control Chart Techniques
E2857 Guide for Validating Analytical Methods
E2935 Practice for Evaluating Equivalence of Two Testing Processes
2.2 Other Standards:
Code of Federal Regulations 40 CFR Part 136, Appendix B
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 collision cell, n—chamber in the ion path between m/z separation elements, or between ion source and the first analyzer, in
tandem mass spectrometry in space configurations.
3.2.2 continuing calibration verification (CCV), n—a mid-range calibration standard which checks the continued validity of the
initial calibration of the instrument.
3.2.3 mass spectrometry/mass spectrometry (MS/MS), n—acquisition and study of the spectra of the product ions or precursor ions
of m/z selected ions, or of precursor ions of a selected neutral mass loss.
3.2.3.1 Discussion—
MS/MS can be accomplished using instruments incorporating more than one analyzer (tandem mass spectrometry in space) or in
trap instruments (tandem mass spectrometry in time).
3.2.4 multiple reaction monitoring (MRM), n—application of selected reaction monitoring to multiple product ions from one or
more precursor ions.
3.2.5 per- and polyfluoroalkyl substances (PFAS), n—synthetic organofluorine chemical compounds with multiple fluorine atoms
that includes PFOA, PFOS, GenX, and many other chemicals.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from National Technical Information Service (NTIS), U.S. Department of Commerce, 5285 Port Royal Road, Springfield, VA, 22161 or at http://www.epa.gov/
epawaste/hazard/testmethods/index.htm
D8421 − 22
3.2.5.1 Discussion—
PFAS have a hydrophobic and oleophobic fluorinated “tail” and a hydrophilic “head” making them surfactants. They include the
perfluoro sulfonic acids such as the perfluorooctanesulfonic acid (PFOS) and the perfluoro carboxylic acids, such as the
perfluorooctanoic acid (PFOA). PFOS and PFOA are persistent organic pollutants. The definition does not include the mass labeled
surrogates or internal standards.
3.2.6 precursor ion, n—ion that reacts to form product ions or undergoes specified neutral losses.
3.2.7 product ion, n—ion formed as the product of a reaction involving a precursor ion.
3.2.8 single (or selected) reaction monitoring (SRM), n—data acquired from one or more specific product ions corresponding to
m/z selected precursor ions recorded via two or more stages of mass spectrometry.
3.2.9 tandem mass spectrometer, n—mass spectrometer designed for mass spectrometry/mass spectrometry.
3.2.10 triple quadrupole mass spectrometer (triple quad or QQQ), n—tandem mass spectrometer comprising two transmission
quadrupole mass spectrometers in series, with a (non-selecting) RF-only quadrupole (or other multipole) between them to act as
a collision cell.
4. Summary of Test Method
4.1 The operating conditions presented in this test method have been validated for use in the determination of PFASs in aqueous
samples. Alternative instrument operating conditions may be used provided data quality objectives are met. Follow the
manufacturer’s instructions. The preparation process, as summarized in 4.2 and described in Section 14 may be automated, but
cannot be modified.
4.2 Samples are shipped to the lab at a temperature between 0°C0 °C and 6°C6 °C and analyzed within 28 days of collection. A
sample (5 mL) is collected and processed in the same collection tube in order to limit analyte loss; extra samples must be collected
for duplicates/triplicates and matrix spikes. All samples and associated QC samples are spiked with labeled surrogates (QC samples
such as laboratory control and matrix spike samples are additionally spiked with target PFASs) and shaken for 2 minutes after
adding 5 mL of methanol. The samples are then filtered through a polypropylene filter. Acetic acid (~10 μL) is added to all the
samples to adjust to pH ~4 and analyzed by LC/MS/MS. If samples contain more than about 1.0 g/L suspended or settled solids,
(for example, sludge, pretreatment, or wastewater influent) adjust to pH ~9 (adding ~20 μL of ammonium hydroxide), shake for
2 minutes, filter, acidify to pH ~4 (~50 μL acetic acid), and then analyze by LC/MS/MS.
NOTE 3—Sludge in this test method is defined as sewage sample containing between 0.1 and 2 % solids based upon a sample by weight.
NOTE 4—Since contact with surfaces may bias data, collect a 5.0-mL sample in a graduated 15-mL polypropylene tube in the field so that the whole sample
is processed in the lab. Once this 5.0-mL sample is spiked according to this test method and methanol is added, the sample is filtered into another 15
mlmL polypropylene tube without analyte loss.
NOTE 5—For accurate volume, the weight of the 15-mL polypropylene tube may be taken before and after sampling. The density of water is assumed
to be 1.0 g/mL unless the exact density of the water sample is known, then that conversion should be used.
4.3 Most analytes are identified by comparing the SRM transition and its confirmatory SRM transition correlated to the known
standard SRM transition (Table 3) and quantitated utilizing an external calibration. The retention times and ion ratios are shown
in Table 4 for each native analyte and isotope. The surrogates and some analytes only have one SRM transition due to a less
sensitive or non-existent secondary SRM transition. As an additional quality-control measure, isotopically labeled surrogate (Table
1, Section 13.3) recoveries are monitored. With external standard calibrations, there is no correction to the data based upon
surrogate recoveries. Alternatively, extract an isotopically labelled analog of each analyte (isotope dilution), if available, and
correct for recovery. Only exact isotopes of the native analytes may be used for isotope dilution correction. If a structurally different
isotope is used to correct a native analyte this is called surrogate correction and either must be clearly stated as performed in the
accompanying data report or not allowed. For isotope dilution, the analog and the native compound concentrations (areas) should
be within 30%30 % of each other to obtain more accurate results. The final report issued lists the concentration of PFAS, if
detected, or as a non-detect at the RL, if not detected, in ng/L and the surrogate recoveries.
NOTE 6—For greater accuracy in the isotope dilution method, add the isotopes at the time sampling or allow the sample and isotope to equilibrate for
at least 48 hours prior to addition of methanol.
D8421 − 22
TABLE 1 Analyte List with Method Detection Limit and Reporting Range
MDL Range
Analyte Name Acronym CAS Number
(ng/L) (ng/L)
Perfluorotetradecanoic acid PFTreA 376-06-7 8.2 10-400
Perfluorotridecanoic acid PFTriA 72629-94-8 17.2 10-400
Perfluorododecanoic acid PFDoA 307-55-1 6.6 10-400
Perfluoroundecanoic acid PFUnA 2058-94-8 3.9 10-400
Perfluorodecanoic acid PFDA 335-76-2 3.4 10-400
Perfluorononanoic acid PFNA 375-95-1 5.2 10-400
Perfluorooctanoic acid PFOA 335-67-1 2.5 10-400
Perfluoroheptanoic acid PFHpA 375-85-9 5.9 10-400
Perfluorohexanoic acid PFHxA 307-24-4 2.1 10-400
Perfluoropentanoic acid PFPeA 2706-90-3 13.0 50-1000
Perfluorobutanoic acid PFBA 375-22-4 17.1 50-1000
Perfluorodecanesulfonic acid PFDS 335-77-3 1.6 10-400
Perfluorononanesulfonic acid PFNS 68259-12-1 1.2 10-400
Perfluorooctanesulfonic acid PFOS 1763-23-1 4.4 10-400
Perfluoroheptanesulfonic acid PFHpS 375-92-8 2.7 10-400
Perfluorohexanesulfonic acid PFHxS 355-46-4 2.3 10-400
Perfluoropentanesulfonic acid PFPeS 2706-91-4 2.7 10-400
Perfluorobutanesulfonic acid PFBS 375-73-5 3.3 10-400
Perfluorooctanesulfonamide PFOSA 754-91-6 2.2 10-400
8:2 Fluorotelomer sulfonic acid 8:2 FTS 39108-34-4 4.5 10-400
6:2 Fluorotelomer sulfonic acid 6:2 FTS 27619-97-2 2.7 10-400
4:2 Fluorotelomer sulfonic acid 4:2 FTS 757124-72-4 3.2 10-400
N-Ethylperfluorooctanesulfonamidoacetic acid NEtFOSAA 2991-50-6 2.6 10-400
N-Methylperfluorooctanesulfonamidoacetic acid NMeFOSAA 2355-31-9 1.3 10-400
Perfluorododecanesulfonic acid PFDoS 79780-39-5 2.2 10-400
N-Methylperfluorooctanesulfonamide NMeFOSA 31506-32-8 2.1 10-400
N-Ethylperfluorooctanesulfonamide NEtFOSA 4151-50-2 1.8 10-400
N-Methylperfluorooctanesulfonamidoethanol NMeFOSE 24448-09-7 3.1 10-400
N-Ethylperfluorooctanesulfonamidoethanol NEtFOSE 1691-99-2 2.7 10-400
Hexafluoropropylene oxide dimer acid HFPO-DA 13252-13-6 3.7 10-400
4,8-dioxa-3H-perfluorononanoic acid ADONA 919005-14-4 2.1 10-400
9-chlorohexadecafluoro-3-oxanonane-1-sulfonic acid 9Cl-PF3ONS 756426-58-1 2.7 10-400
11-chloroeicosafluoro-3-oxaundecane-1-sulfonic acid 11Cl-PF3OUdS 763051-92-9 2.2 10-400
Pentafluorpropanoic acid PFPrA 422-64-0 20.3 50-1000
Perfluoro-3,6-dioxaheptanoic acid NFDHA 151772-58-6 3.7 10-400
Perfluoro(2-ethoxyethane) sulfonic acid PFEESA 113507-82-7 2.2 10-400
Perfluoro-3-methoxypropanoic acid PFMPA 377-73-1 2.6 10-400
Perfluoro-4-methoxybutanoic acid PFMBA 863090-89-5 2.2 10-400
2H,2H,3H,3H-Perfluorohexanoic Acid 3:3 FTCA 356-02-05 3.7 10-400
2H,2H,3H,3H-Perfluorooctanoic Acid 5:3 FTCA 914637-49-3 3.0 10-400
2H,2H,3H,3H-Perfluorodecanoic acid 7:3 FTCA 812-70-4 1.5 10-400
2H-perfluoro-2-octenoic acid FHUEA 70887-88-6 2.5 10-400
2H-perfluoro-2-decenoic acid FOUEA 70887-84-2 2.9 10-400
A
Lithium Bis(trifluoromethane)sulfonimide HQ-115 90076-65-6 9.0 10-400
Surrogates
Perfluoro-n-[ C ]butanoic acid MPFBA NA NA 10-400
Perfluor0-n-[ C ]pentanoic acid M5PFPeA NA NA 10-400
Perfluoro-n-[1,2,3,4,6- C ]hexanoic acid M5PFHxA NA NA 10-400
Perfluoro-n-[1,2,3,4- C ]heptanoic acid M4PFHpA NA NA 10-400
Perfluoro-n-[ C ]octanoic acid M8PFOA NA NA 10-400
Perfluoro-n-[ C ]nonanoic acid M9PFNA NA NA 10-400
Perfluoro-n-[1,2,3,4,5,6- C ]decanoic acid M6PFDA NA NA 10-400
Perfluoro-n-[1,2,3,4,5,6,7- C ]undecanoic acid M7PFUnA NA NA 10-400
Perfluoro-n-[1,2- C ]dodecanoic acid MPFDoA NA NA 10-400
Perfluoro-n-[1,2- C ]tetradecanoic acid M2PFTreA NA NA 10-400
Perfluoro-1-[ C ]octanesulfonamide M8FOSA NA NA 10-400
N-methyl-d -perfluoro-1-octanesulfonamidoacetic acid D3-N-MeFOSAA NA NA 10-400
N-ethyl-d -perfluoro-1-octanesulfonamidoacetic acid D5-N-EtFOSAA NA NA 10-400
N-methyl-d -perfluoro-1-octanesulfanamide d-N-MeFOSA NA NA 10-400
N-ethyl-d -perfluoro-1-octanesulfanamide d-N-EtFOSA NA NA 10-400
2-(N-methyl-d -perfluoro-1-octanesulfonamido)ethan-d4-ol d7-N-MeFOSE NA NA 10-400
2-(N-ethyl-d -perfluoro-1-octanesulfonamido)ethan-d4-ol D9-N-EtFOSE NA NA 10-400
2,3,3,3-Tetrafluoro-2-(1,1,2,2,3,3,3-heptafluoropropoxy- C - MHFPO-DA NA NA 10-400
propanoic acid
1H,1H,2H,2H-perfluoro-1-[1,2- C ]hexane sulfonate M4:2FTS NA NA 10-400
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-octane sulfonate M6:2FTS NA NA 10-400
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-decane sulfonate M8:2FTS NA NA 10-400
Perfluoro-1-[ C ]octanesulfonate M8PFOS NA NA 10-400
Perfluoro-1-[2,3,4- C ]butanesulfonate MPFBS NA NA 10-400
Perfluoro-1-[1,2,3- C ]hexanesulfonate M3PFHxS NA NA 10-400
A
The Lithium is just the counter ion, report only Bis(trifluoromethane)sulfonimide.
D8421 − 22
5. Significance and Use
5.1 PFAS are widely used in various industrial and commercial products; they are persistent, bio-accumulative, and ubiquitous in
the environment. PFAS have been reported to exhibit developmental toxicity, hepatotoxicity, immunotoxicity, and hormone
disturbance. PFAS have been detected in soils, sludges, surface, and drinking waters. This is a quick, easy, and robust method to
quantitatively determine these compounds at trace levels in water matrices.
5.2 This test method has been validated using reagent water and waters from sites that include landfill leachate, metal finisher,
POTW Effluent, Hospital, POTW Influent, Bus washing station, Power Plant and Pulp and paper mill effluent for selected PFAS,
refer to the Precision and Bias (Section 17).
6. Interferences
6.1 All glassware is washed in hot water (typically >45ºC)>45 ºC) with detergent and rinsed in hot water followed by distilled
water. The glassware is then dried and heated in an oven (typically at 105ºC)105 ºC) for 15 to 30 minutes. All glassware is
subsequently rinsed with methanol or acetonitrile.
6.2 All reagents and solvents should be pesticide residue purity or higher to minimize interference. Avoid the use of PFAS
containing caps.
6.3 Matrix interferences may be caused by contaminants in the sample. The extent of matrix interferences varies considerably
depending on variations of the sample matrices. Separation of individual components by the LC is vital in minimization of
interferences. Shortening of run times simply to speed analysis should be avoided, unless interferences are known to be absent.
6.4 Contaminants have been found in reagents, glassware, tubing, glass disposable pipettes, filters, degassers, and other apparatus
and consumables that release PFAS. All these materials and supplies must be routinely demonstrated to be free from interferences
by analyzing laboratory reagent blanks under the same conditions as the samples. If found, measures should be taken to remove
the contamination or data should be qualified, background subtraction of blank contamination is not allowed. It has become
difficult to ensure consumables are PFAS free at the lower concentrations (approximately at less than 30 ng/L) for the entire lot
by testing only a very small sub-sample. At a minimum duplicates/triplicates should be taken of each sample to evaluate precision
between the set.
6.5 The LC system used should consist, as much as practical, of sample solution or eluent contacting components free of PFAS
of interest.
6.6 Polyethylene LC vial caps or any other target analyte free vial caps should be used.
6.7 Polyethylene disposable pipettes or target analyte free pipettes should be used. All disposable pipettes should be checked for
release of target analytes of interest.
6.8 Degassers are important to continuous LC operation and most commonly are made of fluorinated polymers. To enable use, an
isolator column should be placed after the degasser and prior to the sample injection valve to separate the PFAS in the sample from
the PFAS in the LC system.
6.9 Electro Spray Ionization (ESI)—ESI should be heated and optimized for recovery of components analyzed by this test method.
Using the suggested mobile phase, gradient, and adequate column separation minimizes, or eliminates, quenching and enhancing
of signal. This method was validated using ESI, however other modes of ionization may be used provided the detection limits and
quality control acceptance criteria of this method are met.
7. Apparatus
7.1 LC/MS/MS System:
7.1.1 Liquid Chromatography System—A complete LC system is required to analyze samples, this includes a sample injection
system, a solvent pumping system capable of mixing solvents, a sample compartment capable of maintaining required temperature
D8421 − 22
and a temperature-controlled column compartment. This test method was developed using a ternary (Table 2) pumping system. A
binary LC system may be used by adapting the ternary gradient to a binary system. A LC system that can perform at the flow rates,
pressures, controlled temperatures, sample volumes, and requirements of the standard shall be used.
7.1.2 Analytical Column—UHPLC CSH Phenyl-Hexyl, 2.1 × 100 mm and 1.7 μm particle size column, or any column that
achieves adequate resolution may be used. The retention times and order of elution may change depending on the column used
and needs to be monitored.
7.1.3 Isolator Column—A reverse phase C18 column is used to separate the target analytes in the LC system and solvents from
the target analytes in the analytical sample. Place the column between the solvent mixing chamber and the injector sample loop.
7.2 Tandem Mass Spectrometer System—A MS/MS system capable of multiple reaction monitoring (MRM) analysis or any system
that is capable of performing at the requirements in this test method.
7.3 Filtration Device:
7.3.1 Hypodermic Syringe—A luer-lock tip glass syringe capable of holding a syringe driven filter unit.
7.3.1.1 A 10-mL Lock Tip Glass Syringe size is recommended in this test method.
7.3.2 Filter Unit—Polypropylene syringe-driven filter units (0.2 μm) or equivalent, demonstrated contaminant free below 1/2
MRL.
8. Reagents and Materials
8.1 Purity of Reagents—High Performance Liquid Chromatography (HPLC) pesticide residue analysis and spectrophotometry
grade chemicals shall be used in all tests. Unless indicated otherwise, it is intended that all reagents shall conform to the Committee
on Analytical Reagents of the American Chemical Society. Other reagent grades may be used provided they are first determined
to be of sufficiently high purity to permit their use without affecting the accuracy of the measurements.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water conforming to
Type 1 of Specification D1193. It shall be demonstrated that this water does not contain contaminants at concentrations sufficient
to interfere with the analysis.
8.3 Gases—Ultrapure nitrogen and argon.
8.4 Vials—Greater than 1.0 mL Amber glass or polypropylene autosampler vials.
8.5 Polyethylene autosampler vial caps, or equivalent.
TABLE 2 Gradient Conditions for a Ternary Pumping System
95 % Water:
Time Flow 95 % Water: 5 % Acetonitrile,
Acetonitrile %
(min) (mL/min) 5 % Acetonitrile % 400 mM
Ammonium Acetate %
0 0.3 95 0 5
1 0.3 75 20 5
6 0.3 50 45 5
13 0.3 15 80 5
14 0.4 0 95 5
17 0.4 0 95 5
18 0.4 95 0 5
21 0.4 95 0 5
ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference Materials, American Chemical Society, Washington, DC. For
suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and
the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D8421 − 22
8.6 Syringe—10 or 25-mL filter-adaptable glass syringe with luer lock.
8.7 Polypropylene Tubes—15 and 50 mL conical with calibration lines.
8.8 pH paper (pH range 1–14).
8.9 Class A Volumetric Glassware.
8.10 Pipette tips—Polypropylene pipette tips free of release agents or low retention coating of various sizes.
8.11 Polyethylene Disposable Pipettes.
8.12 Acetonitrile (CAS #75-05-8).
8.13 Methanol (CAS #67-56-1).
8.14 Ammonium acetate (CAS #631-61-8).
8.15 Acetic acid (CAS #64-19-7).
8.16 2-Propanol (isopropyl alcohol, CAS #67-63-0).
8.17 Ammonium hydroxide (CAS #1336-21-6).
8.18 PFASs Standards —Refer to Table 1 for the complete analyte list and CAS numbers. These may be purchased from a
commercial supplier individually or some as a mixture.
9. Hazards
9.1 Precaution—The toxicity or carcinogenicity of chemicals used in this test method has not been precisely defined; each
chemical should be treated as a potential health hazard, and exposure to these chemicals should be minimized. Each laboratory
is responsible for maintaining awareness of OSHA regulations regarding safe handling of chemicals used in this test method.
9.2 Warning—The compound analytes in this test method have been classified as known or suspected human or mammalian
carcinogens. Pure standards and stock solutions should be handled in a hood or glovebox.
10. Sampling
10.1 Sampling and Preservation—Avoid sample containers and contact with surfaces of fluorinated polymers or PFAS
contaminated items. Collect field blanks that are exposed to the same field conditions as samples and analyze according to this test
method to assess the potential for field contamination. Collect 5 6 0.5 mlmL samples, duplicates/triplicates, matrix spikes and field
blanks in graduated 15 mlmL polypropylene tubes. For greater accuracy, the tubes should be pre-weighed and weighed after
sampling in order to achieve an exact weight of the sample. This weight is then used to calculate volume with the assumed density
of the water sample as 1.0 g/mL. Conventional sampling practices should be followed with the caution that PFASs containing
products may be present in sampling equipment. All sampling equipment and supplies shall be PFAS free to prevent contamination
of the samples. EPA Publication SW-846, Guide D3856, and Practices E694 may be used as guides. Ship samples on ice with a
trip blank. The temperature of the samples upon receipt at the laboratory should be less than 6°C.6 °C. If the receiving temperature
is greater than 6°C,6 °C, the sample temperature is noted in the case narrative accompanying the data. Samples should be stored
PFASs standards may be difficult to find, some sources of PFASs standards that have been found suitable for use were from Aldrich Chemical Company, Wellington
Laboratories Inc., and Wako Laboratory. Standards from other vendors may be used.
D8421 − 22
refrigerated between 0°C0 °C and 6°C6 °C from the time of collection until analysis. Analyze the sample within 28 days of
collection. Holding time may vary depending on the matrix and individual laboratories should determine the holding time in their
matrix.
11. Preparation of LC/MS/MS
11.1 LC Chromatograph Operating Conditions:
11.1.1 Injections of all standards and samples are made at a 10–30-μL volume. Other injection volumes may be used to optimize
conditions. Calibration Standards, reagent blanks, second source and samples shall be in a 50:50 methanol:water solution
containing 0.1 % acetic acid. In the case of extreme concentration differences amongst samples, it is wise to analyze a blank after
a concentrated sample and before a dilute sample to eliminate carry-over of analytes from sample injection to sample injection.
If a flow through needle design is used carry-over should not be a problem. The gradient conditions for LC are shown in Table
2. To ensure chromatographic separation between the targeted analytes and any unknown non-targeted potentially interfering
compounds, avoid shortening the analysis time simply to speed the analysis. Refer to Fig. 1 as an example chromatogram of 24
surrogates showing resolution with limited coelution.
FIG. 1 Example Chromatogram of 24 Surrogates, at the Level 1 Calibration Concentration, Overlayed Showing Resolution with Limited
Coelution
Guides to help determine holding times can be found at: http://www.epa.gov/esd/cmb/research/bs_033cmb06.pdf (2014) and Practice D4841.
D8421 − 22
11.2 LC Sample Manager Conditions:
11.2.1 Needle Wash Solvent—60 % acetonitrile/40 % 2-propanol. Eight second wash time before and after injection. Instrument
manufacturer’s specifications should be followed in order to eliminate sample carry-over.
11.2.2 Temperatures—Column, 35°C;35 °C; Sample compartment, 15°C.15 °C.
11.2.3 Seal Wash—Solvent: 50% water/5050 % water ⁄50 % methanol; Time: 5 minutes.
11.3 Mass Spectrometer Parameters:
11.3.1 To acquire the maximum number of data points per SRM channel while maintaining adequate sensitivity, optimize the tune
parameters according to instrument manufacturer instructions. Each peak requires a minimum of 10 scans per peak for adequate
quantitation. This test method containing surrogates, which are select isotopically labeled PFAS, and the targeted PFAS may be
split into multiple MRM acquisition functions to optimize sensitivity. Retention times, and primary and confirmatory transitions
are shown in Table 3. Retention times will vary between columns and gradient used. Each manufacturer may have different
terminology to represent various mass spectrometer settings, and different set values depending on the manufacturer and instrument
model. Please refer to the manufacturersmanufacturer’s instructions in optimizing detector settings, including collision energies
and cone voltages. Data for this method was collected using electrospray ionization (ESI) operated in negative mode. In
recognition of the advancement of LCMSMS instrumentation, other MS operating conditions, including ionization techniques may
be used provided the quality control criteria of the method is met.
12. Calibration and Standardization
12.1 The mass spectrometer is calibrated as in accordance with manufacturer’s specifications prior to analysis. Prepare all
calibration solutions using Class A volumetric glassware (E694).
12.2 Calibration and Standardization—Analyze up to nine calibration standards containing the PFAS and surrogates prior to
analysis as shown in Table 5. The calibration stock standard solution is prepared from the target and surrogate spike solutions.
Stock standard Solution A containing the PFAS and surrogates is prepared at Calibration Level 9 concentration and aliquots of that
solution are diluted to prepare Calibration Levels 1 through 8. The following steps will produce standards with the concentration
values shown in Table 5. The analyst is responsible for recording initial component weights carefully when working with pure
materials and correctly carrying the weights through the dilution calculations. At a minimum, five calibration levels are required
when using a linear calibration and six calibration levels are required when using a quadratic calibration curve. An initial
nine-points may be used to enable dropping the lower calibration points if the instrument cannot achieve low detection limits on
certain PFAS. This will allow at least a five or six-point calibration curve per analyte to be obtained.
12.2.1 Calibration Stock Standard Solution A (Calibration Level 9, Table 5) is prepared from the target and surrogate spike
solutions. Transfer 500 μL of the surrogate spike (20 μg/L), 500 μL of PFAS Target Spike I and 500 μL of PFAS Target Spike II
(refer to Table 7) to a 50-mL volumetric flask and dilute to 50-mL volume with 50:50 methanol:water containing 0.1 % acetic acid.
Ensure that the analytes are solubilized in the Level 9 standard.
12.2.2 Aliquots of Solution A (Calibration Level 9) are then diluted with 50:50 methanol:water containing 0.1 % acetic acid to
prepare the desired calibration levels (Table 5) in polypropylene LC vials. For best results, use the calibration standards within 24
hours of preparation. Prepare the end CCV at a mid-level concentration in a separate LC vial. All calibration standards should be
used only once because the analyte concentration in the vial may change after the vial cap is pierced. Changing the caps
immediately after the injection may alleviate this problem, however, this should be verified in each laboratory. Calibration
standards do not need to be filtered.
12.2.3 Incorporate a second source standard, if available. The second source standard should be analyzed near the midpoint of the
calibration range to verify that the standards used are within 630 % of the expected concentration. Currently, a second source from
a different vendor may not be readily available for all target analytes. In this case, a second lot number from the same vendor may
be used.
12.2.4 Inject each standard and obtain its chromatogram. The instrument software collects the primary and confirmatory SRM
transitions of each analyte at the specified retention times. Calibration software conducts the quantitation of the target analytes and
D8421 − 22
TABLE 3 Transitions for Target Analytes and Surrogates
Analyte Name Acronym CAS Number Primary Ion Transition Confirmation Ion Transition
Perfluorotetradecanoic acid PFTreA 376-06-7 712.9 → 668.9 712.9 → 168.9
Perfluorotridecanoic acid PFTriA 72629-94-8 662.9 → 618.9 662.9 → 168.9
Perfluorododecanoic acid PFDoA 307-55-1 612.9 → 568.9 612.9 → 168.9
Perfluoroundecanoic acid PFUnA 2058-94-8 562.9 → 519 562.9 → 269
Perfluorodecanoic acid PFDA 335-76-2 512.9 → 469 512.9 → 218.9
Perfluorononanoic acid PFNA 375-95-1 462.9 → 419 462.9 → 218.9
Perfluorooctanoic acid PFOA 335-67-1 412.9 → 369 412.9 → 168.9
Perfluoroheptanoic acid PFHpA 375-85-9 362.9 → 318.9 362.9 → 168.9
Perfluorohexanoic acid PFHxA 307-24-4 312.9 → 269 312.9 → 118.9
Perfluoropentanoic acid PFPeA 2706-90-3 262.9 → 218.9 NA
Perfluorobutanoic acid PFBA 375-22-4 212.9 →168.9 NA
Perfluorodecanesulfonic acid PFDS 335-77-3 598.9 → 79.9 598.9 → 98.9
Perfluorononanesulfonic acid PFNS 68259-12-1 548.9 → 79.9 548.9 → 98.9
Perfluorooctanesulfonic acid PFOS 1763-23-1 498.9 → 79.9 498.9 → 98.9
Perfluoroheptanesulfonic acid PFHpS 375-92-8 448.9 → 79.9 448.9 → 98.9
Perfluorohexanesulfonic acid PFHxS 355-46-4 398.9 → 79.9 398.9 → 98.9
Perfluoropentanesulfonic acid PFPeS 2706-91-4 348.9 → 79.9 348.9 → 98.9
Perfluorobutanesulfonic acid PFBS 375-73-5 298.9 → 79.9 298.9 → 98.9
Perfluorooctanesulfonamide PFOSA 754-91-6 497.9 → 77.9 NA
8:2 Fluorotelomer sulfonic acid 8:2 FTS 39108-34-4 526.9 → 506.9 526.9 → 80.9
6:2 Fluorotelomer sulfonic acid 6:2 FTS 27619-97-2 427 → 407 427 → 80.9
4:2 Fluorotelomer sulfonic acid 4:2 FTS 757124-72-4 326.9 → 306.9 326.9 → 80.9
N-Ethylperfluorooctanesulfonamidoacetic acid NEtFOSAA 2991-50-6 584 → 419 584 → 482.9
N-Methylperfluorooctanesulfonamidoacetic NMeFOSAA 2355-31-9 569.9 → 419 569.9 → 482.9
acid
Perfluorododecanesulfonic acid PFDoS 79780-39-5 698.9 → 79.9 698.9 → 98.9
N-Methylperfluorooctanesulfonamide NMeFOSA 31506-32-8 511.9 → 168.9 511.9 → 218.9
N-Ethylperfluorooctanesulfonamide NEtFOSA 4151-50-2 525.9 → 168.9 525.9 → 218.9
N-Methylperfluorooctanesulfonamidoethanol NMeFOSE 24448-09-7 616 → 58.9 NA
N-Ethylperfluorooctanesulfonamidoethanol NEtFOSE 1691-99-2 630 → 58.9 NA
Hexafluoropropylene oxide dimer acid HFPO-DA 13252-13-6 285 → 168.9 285 → 184.9
4,8-dioxa-3H-perfluorononanoic acid ADONA 919005-14-4 376.9 → 251 376.9 → 84.9
9-chlorohexadecafluoro-3-oxanonane-1- 9Cl-PF3ONS 756426-58-1 530.9 → 350.9 532.9 → 352.9
sulfonic acid
11-chloroeicosafluoro-3-oxaundecane-1- 11Cl-PF3OUdS 763051-92-9 630.8 → 450.9 632.8 → 452.9
sulfonic acid
Pentafluorpropanoic acid PFPrA 422-64-0 162.9 → 118.9 NA
Perfluoro-3,6-dioxaheptanoic acid NFDHA 151772-58-6 295 → 200.9 295 → 84.9
Perfluoro(2-ethoxyethane)sulfonic acid PFEESA 113507-82-7 314.9 → 134.9 314.9→ 82.9
Perfluoro-3-methoxypropanoic acid PFMPA 377-73-1 228.9 → 84.9 NA
Perfluoro-4-methoxybutanoic acid PFMBA 863090-89-5 278.9 → 84.9 NA
2H,2H,3H,3H-Perfluorohexanoic Acid 3:3 FTCA 356-02-05 241 → 176.9 241→ 116.9
2H,2H,3H,3H-Perfluorooctanoic Acid 5:3 FTCA 914637-49-3 340.9 → 216.9 340.9 →237
2H,2H,3H,3H-Perfluorodecanoic acid 7:3 FTCA 812-70-4 440.9 → 337 440.9 → 316.9
2H-perfluoro-2-octenoic acid FHUEA 70887-88-6 356.9 → 292.9 NA
2H-perfluoro-2-decenoic acid FOUEA 70887-70-4 456.9 → 393 NA
Lithium Bis(trifluoromethane)sulfonimide HQ-115 90076-65-6 279.9 →146.9 279.9 →210.9
Surrogates
Perfluoro-n-[ C ]butanoic acid MPFBA NA 216.9 → 171.9 NA
Perfluoro-n-[ C ]pentanoic acid M5PFPeA NA 267.9 → 222.9 NA
Perfluoro-n-[1,2,3,4,6- C ]hexanoic acid M5PFHxA NA 317.9 → 272.9 NA
Perfluoro-n-[1,2,3,4- C ]heptanoic acid M4PFHpA NA 366.9 → 321.9 NA
Perfluoro-n-[ C ]octanoic acid M8PFOA NA 421 → 376 NA
Perfluoro-n-[ C ]nonanoic acid M9PFNA NA 471.9 → 426.9 NA
Perfluoro-n-[1,2,3,4,5,6- C ]decanoic acid M6PFDA NA 518.9 → 473.9 NA
Perfluoro-n-[1,2,3,4,5,6,7- C7]undecanoic M7PFUnA NA 569.9 → 524.9 NA
acid
Perfluoro-n-[1,2- C ]dodecanoic acid MPFDoA NA 614.9 → 569.9 NA
Perfluoro-n-[1,2- C ]tetradecanoic acid M2PFTreA NA 714.9 → 669.9 NA
Perfluoro-1-[ C ]octanesulfonamide M8FOSA NA 505.9 → 77.9 NA
N-methyl-d -perfluoro-1- D3-N-MeFOSAA NA 572.9 → 418.9 NA
octanesulfonamidoacetic acid
N-ethyl-d -perfluoro-1- D5-N-EtFOSAA NA 589 → 418.9 NA
octanesulfonamidoacetic acid
N-methyl-d -perfluoro-1-octanesulfanamide d-N-MeFOSA NA 514.9 → 168.9 NA
N-ethyl-d -perfluoro-1-octanesulfanamide d-N-EtFOSA NA 531 →168.9 NA
2-(N-ethyl-d -perfluoro-1- d7-N-MeFOSE NA 623 → 58.9 NA
octanesulfonamido)ethan-d4-ol
2-(N-methyl-d -perfluoro-1- D9-N-EtFOSE NA 639 → 58.9 NA
octanesulfonamido)ethan-d4-ol
2,3,3,3-Tetrafluoro-2-(1,1,2,2,3,3,3- MHFPO-DA NA 287 → 168.9 NA
heptafluoropropoxy- C -propanoic acid
1H,1H,2H,2H-perfluoro-1-[1,2- C ]hexane M4:2FTS NA 328.9 → 308.9 NA
A
sulfonate 328.9 → 80.9
D8421 − 22
TABLE 3 Continued
Analyte Name Acronym CAS Number Primary Ion Transition Confirmation Ion Transition
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-octane M6:2FTS NA 428.9 → 408.9 NA
A
sulfonate 428.9 → 80.9
1H,1H,2H,2H-perfluoro-1-[1,2- C ]-decane M8:2FTS NA 528.9 → 508.9 NA
A
sulfonate 528.9 → 80.9
Perfluoro-1-[ C ]octanesulfonate M8PFOS NA 506.9 → 79.9 NA
Perfluoro-1-[2,3,4- C ]butanesulfonate M3PFBS NA 301.9 → 79.9 NA
Perfluoro-1-[1,2,3- C ]hexanesulfonate M3PFHxS NA 401.9 → 79.9 NA
A
If high concentrations of the native FTS interfere with Isotope of the FTSs, this transition should be used. It is not as sensitive, but the interference/high bias is removed.
surrogates using the primary SRM transition. The ratios of the primary/confirmatory MRM transition area counts will vary
depending on the individual tuning conditions. Refer to Table 4 for retention Times and Ion ratios. For confirmation of analyte
identity, the primary/confirmatory ratio shall be within 30 % of the individual ratios established during the initial calibration. The
average ion ratio is calculated for each batch from the initial calibration levels.
12.2.5 Depending on sensitivity and sample dependent matrix interference, the confirmatory SRM transition may be used as the
primary SRM transition for quantitation during analysis.
12.2.6 The calibration software manual or the instrument manufacturer should be consulted to ensure correct software use. The
quantitation method is set using the peak areas in ppt (ng/L) units. Concentrations may be calculated using the data system software
to generate linear regression or quadratic calibration curves. Forcing the calibration curve through the origin (X = 0, Y = 0) is not
recommended.
12.2.7 Either of two procedures may be used to determine calibration function acceptability for linear and non-linear curves. These
include refitting the calibration data back to the model. Both % Error and Relative Standard Error (RSE) evaluate the difference
between the measured and the true amounts or concentrations used to create the model.
12.2.7.1 Calculation of % Error is shown as Eq 1. Percent error between the calculated and expected amounts should be ≤
30%30 % for all standards.
'
x 2 x
i i
%Error 5 3100 (1)
x
i
'
x 2 x
i i
%Error 5 3100 (1)
x
i
where:
’
x = measured amount of analyte at calibration level i, in mass or concentration units, and
i
x = true amount of analyte at calibration level i, in mass or concentration units.
i
12.2.7.2 Calculation of Relative Standard Error (RSE – expressed as %) is shown in Eq 2. The RSE acceptance limit criterion for
the calibration model is the same as the RSD limit.
n
' 2
x 2 x
i i
RSE 5 100 3 ⁄ n 2 p (2)
ΠF G ~ !
(
x
i51 i
where:
x = true amount of analyte in calibration level i, in mass or concentration units,
i
’
x = measured amount of analyte in calibration level i, in mass or concentration units,
i
p = number of terms in the fitting equation (average = 1, linear = 2, quadratic = 3, cubic = 4), and
n = number of calibration points.
12.2.8 The retention time window of an unknown shall be within 5 % of the retention time of the analyte in a midpoint calibration
standard. If this is not the case, re-analyze the calib ration calibration curve to determine if there was a shift in retention time during
the analysis. If the retention time of the known standard is correct, and the retention time of the peak in the sample is still incorrect
in the sample, refer to the analyte as an unknown.
12.2.9 Analyze a CCV at the end of each batch of 20 samples, 20 samples does not include QC samples. This interval may be
D8421 − 22
TABLE 4 Retention Times and Ion Ratios for Target Analytes and Surrogates
Analyte Name Acronym Retention Time Primary/Confirmatory Ion Ratio
Perfluorotetradecanoic acid PFTreA 10.40 4.7
Perfluorotridecanoic acid PFTriA 9.94 4.2
Perfluorododecanoic acid PFDoA 9.43 5.2
Perfluoroundecanoic acid PFUnA 8.90 5.0
Perfluorodecanoic acid PFDA 8.33 5.4
Perfluorononanoic acid PFNA 7.70 4.5
Perfluorooctanoic acid PFOA 7.03 2.4
Perfluoroheptanoic acid PFHpA 6.28 3.7
Perfluorohexanoic acid PFHxA 5.45 20
Perfluoropentanoic acid PFPeA 4.58 NA
Perfluorobutanoic acid PFBA 3.67 NA
Perfluorodecanesulfonic acid PFDS 9.76 1.0
Perfluorononanesulfonic acid PFNS 9.24 1.0
Perfluorooctanesulfonic acid PFOS 8.64 1.2
Perfluoroheptanesulfonic acid PFHpS 8.00 1.0
Perfluorohexanesulfonic acid PFHxS 7.29 1.1
Perfluoropentanesulfonic acid PFPeS 6.49 1.2
Perfluorobutanesulfonic acid PFBS 5.56 1.5
Perfluorooctanesulfonamide PFOSA 10.08 NA
8:2 Fluorotelomer sulfonic acid 8:2 FTS 8.06 3.1
6:2 Fluorotelomer sulfonic acid 6:2 FTS 6.76 3.1
4:2 Fluorotelomer sulfonic acid 4:2 FTS 5.22 3.3
N-Ethylperfluorooctanesulfonamidoacetic acid NEtFOSAA 8.73 1.8
N-Methylperfluorooctanesulfonamidoacetic acid NMeFOSAA 8.47 1.8
Perfluorododecanesulfonic acid PFDoS 10.74 1.0
N-Methylperfluorooctanesulfonamide NMeFOSA 12.10 1.6
N-Ethylperfluorooctanesulfonamide NEtFOSA 12.60 1.6
N-Methylperfluorooctanesulfonamidoethanol NMeFOSE 11.65 NA
N-Ethylperfluorooctanesulfonamidoethanol NEtFOSE 12.16 NA
Hexafluoropropylene oxide dimer acid HFPO-DA 5.75 2.0
4,8-dioxa-3H-perfluorononanoic acid ADONA 6.61 2.2
9-chlorohexadecafluoro-3-oxanonane-1-sulfonic acid 9Cl-PF3ONS 9.21 3.1
11-chloroeicosafluoro-3-oxaundecane-1-sulfonic acid 11Cl-PF3OUdS 10.29 3.1
Pentafluorpropanoic acid PFPrA 1.79 NA
Nonafluoro-3,6-dioxaheptanoic acid NFDHA 5.33 3.1
Perfluoro(2-ethoxyethane)sulfonic acid PFEESA 5.96 15.5
Perfluoro-3-methoxypropanoic acid PFMPA 4.05 NA
Perfluoro-4-methoxybutanoic acid PFMBA 4.84 NA
2H,2H,3H,3H-Perfluorohexanoic Acid 3:3 FTCA 4.27 3.9
2H,2H,3H,3H-Perfluorooctanoic Acid 5:3 FTCA 6.01 1.0
2H,2H,3H,3H-Perfluorodecanoic acid 7:3 FTCA 7.59 1.0
2H-perfluoro-2-octenoic acid FHUEA 6.00 NA
2H-perfluoro-2-decenoic acid FOUEA 7.45 NA
Lithium Bis(trifluoromethane)sulfonimide HQ-115 6.8 6.3
Surrogates
Perfluoro-n-[ C ]butanoic acid MPFBA 3.67 NA
Perfluor0-n-[ C ]pentanoic acid M5PFPeA 4.71 NA
Perfluoro-n-[1,2,3,4,6- C ]hexanoic acid M5PFHxA 5.45 NA
Perfluoro-n-[1,2,3,4- C ]heptanoic acid M4PFHpA 6.28 NA
Perfluoro-n-[ C ]octanoic acid M8PFOA 7.03 NA
Perfluoro-n-[ C ]nonanoic acid M9PFNA 7.70 NA
Perfluoro-n-[1,2,3,4,5,6- C ]decanoic acid M6PFDA 8.34 NA
Perfluoro-n-[1,2,3,4,5,6,7- C ]undecanoic acid M7PFUnA 8.9 NA
Perfluoro-n-[1,2- C ]dodecanoic acid MPFDoA 9.43 NA
Perfluoro-n-[1,2- C ]tetradecanoic acid M2PFTreA 10.4 NA
Perfluoro-1-[ C ]octanesulfonamide M8FOSA 10.09 NA
...







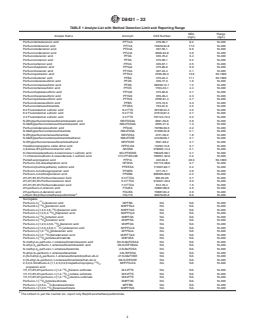
Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...