ASTM D5542-16
(Test Method)Standard Test Methods for Trace Anions in High Purity Water by Ion Chromatography
Standard Test Methods for Trace Anions in High Purity Water by Ion Chromatography
SIGNIFICANCE AND USE
4.1 The anions fluoride, chloride, and sulfate have been identified as important contributors to corrosion of high pressure boilers, electric power turbines and their associated heat exchangers. Many electric power utilities attempt to reduce these contaminants in their boiler feed water to less than 1 μg/L.
4.2 In the semiconductor manufacturing process these ions, among others, have been identified as a cause of low product yield and, thus, must be monitored and controlled to levels similar to those required by the electric power industry.
4.3 Low molecular weight organic acids, such as acetate and formate, have been found in many steam generator feed waters and condensates. They are believed to come from the high temperature breakdown of organic matter found in boiler make up water. It is felt that these organic acids promote corrosion by lowering the pH of boiler waters and may even be corrosive themselves.
4.4 Such low molecular weight organics may also be produced when ultraviolet light is used to produce bacteria-free water for semiconductor processing. Such polar organic contaminants are suspected of causing reduced semiconductor yields.
4.5 Phosphates are commonly added to drum boilers in the low mg/L level to precipitate calcium and magnesium and thereby prevent scale formation. Ion chromatography can be used to monitor the concentration of such chemicals in boiler water, as well as detect unwanted carry-over into the steam.
SCOPE
1.1 These test methods cover the determination of trace (μg/L) levels of fluoride, acetate, formate, chloride, phosphate, and sulfate in high purity water using ion chromatography in combination with sample preconcentration. Other anions, such as bromide, nitrite, nitrate, sulfite, and iodide can be determined by this method. However, since they are rarely present in significant concentrations in high purity water, they are not included in this test method. Two test methods are presented and their ranges of application, as determined by a collaborative study, are as follows:
Range Tested
(μg/L Added)
Limit of DetectionA
(Single Operator)
(μg/L)
Sections
Test Method A:
7–16
Chloride
0–24
0.8
Phosphate
0–39
B
Sulfate
0–55
1.8
Test Method B:
17–24
Fluoride
0–14
0.7
Acetate
0–414
6.8
Formate
0–346
5.6
(A) Limit of detection is lowest measurable concentration not reportable as zero at 99 % level of confidence as per EPRI study as cited in Sections 16 and 24.(B) Insufficient data to calculate limit of detection.
1.2 It is the user's responsibility to ensure the validity of these test methods for waters of untested matrices.
1.3 The common practical range of Test Method A is as follows: chloride, 1 to 100 μg/L, phosphate, 3 to 100 μg/L, and sulfate, 2 to 100 μg/L.
1.4 The common practical range of Test Method B is as follows: fluoride, 1 to 100 μg/L, acetate, 10 to 200 μg/L, and formate, 5 to 200 μg/L.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-May-2016
- Technical Committee
- D19 - Water
Relations
- Effective Date
- 01-May-2020
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Feb-2017
- Effective Date
- 15-Feb-2016
- Refers
ASTM D3856-11 - Standard Guide for Management Systems in Laboratories Engaged in Analysis of Water - Effective Date
- 15-Nov-2011
- Effective Date
- 15-Jun-2011
- Effective Date
- 01-May-2011
- Effective Date
- 01-Feb-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Mar-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Dec-2007
- Effective Date
- 15-Dec-2006
- Effective Date
- 01-Sep-2006
Overview
ASTM D5542-16 is a standard developed by ASTM International that details test methods for the determination of trace anions in high purity water using ion chromatography. The standard specifically focuses on measuring extremely low levels (in the micrograms per liter range) of fluoride, chloride, sulfate, phosphate, acetate, and formate, ensuring water quality in industrial applications where even minimal contamination can cause significant operational issues. This standard also provides guidelines for precision, calibration, sampling, and quality control for laboratories analyzing high purity water.
Key Topics
- Trace Anion Detection: Methods outlined in ASTM D5542-16 allow for precise quantification of trace anions such as fluoride, chloride, sulfate, phosphate, acetate, and formate.
- Ion Chromatography: The test methods rely on ion chromatography with sample preconcentration, providing high sensitivity and selectivity for trace-level detection.
- Quality Control Procedures: The standard emphasizes rigorous quality control, including calibration verification standards, laboratory control samples, reagent blanks, matrix spikes, and duplicate analyses.
- Interference and Contamination Control: Special attention is given to preventing contamination at ultra-low concentrations and addressing potential analytical interferences.
- Application Ranges: Two test methods are described (A and B) for specific anion groups, each with defined practical concentration ranges suitable for ultrapure water analysis.
Applications
ASTM D5542-16 is critical for industries and laboratories that require stringent control over trace anion contamination in high purity water systems:
- Power Generation: Trace levels of anions such as fluoride, chloride, and sulfate are recognized contributors to corrosion in high pressure boilers, turbines, and heat exchangers. Monitoring and reduction of these contaminants to levels below 1 μg/L are essential for minimizing corrosion and extending equipment lifespan.
- Semiconductor Manufacturing: The presence of anions and low molecular weight organic acids can negatively affect semiconductor yield and process reliability. ASTM D5542-16 supports monitoring these ions to minimize product defects and improve quality.
- Steam and Boiler Water Management: The test methods help in detecting and controlling low molecular weight organic acids formed from the breakdown of organic materials or UV sterilization. These acids can lower pH and increase corrosion risk.
- Water Treatment Chemicals: The standard can be used to verify the dosing and carryover of treatment chemicals like phosphate, which is often added to control hardness and prevent scaling.
Related Standards
ASTM D5542-16 references several other key ASTM standards relevant to sampling and analysis of water and steam:
- ASTM D1066 - Practice for Sampling Steam
- ASTM D1129 - Terminology Relating to Water
- ASTM D1193 - Specification for Reagent Water
- ASTM D3370 - Practices for Sampling Water from Closed Conduits
- ASTM D3856 - Guide for Management Systems in Laboratories Analyzing Water
- ASTM D4210, D4453, D5810, D5847 - Quality control and handling practices for water sample analysis
Practical Value
Adopting ASTM D5542-16 helps laboratories and industries:
- Ensure ultra-trace anion detection for stringent process requirements
- Maintain regulatory and product quality standards
- Minimize risks of corrosion, scaling, and manufacturing defects
- Implement robust quality control practices in water analysis
This standard is an authoritative reference for managing contamination in high purity water systems critical to high-performance industrial operations, particularly in the energy and electronics sectors.
Keywords: ASTM D5542-16, trace anions, ion chromatography, high purity water, boiler corrosion, semiconductor water quality, sulfate detection, chloride analysis, water sampling standards, anion monitoring, laboratory quality control.
Buy Documents
ASTM D5542-16 - Standard Test Methods for Trace Anions in High Purity Water by Ion Chromatography
REDLINE ASTM D5542-16 - Standard Test Methods for Trace Anions in High Purity Water by Ion Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5542-16 is a standard published by ASTM International. Its full title is "Standard Test Methods for Trace Anions in High Purity Water by Ion Chromatography". This standard covers: SIGNIFICANCE AND USE 4.1 The anions fluoride, chloride, and sulfate have been identified as important contributors to corrosion of high pressure boilers, electric power turbines and their associated heat exchangers. Many electric power utilities attempt to reduce these contaminants in their boiler feed water to less than 1 μg/L. 4.2 In the semiconductor manufacturing process these ions, among others, have been identified as a cause of low product yield and, thus, must be monitored and controlled to levels similar to those required by the electric power industry. 4.3 Low molecular weight organic acids, such as acetate and formate, have been found in many steam generator feed waters and condensates. They are believed to come from the high temperature breakdown of organic matter found in boiler make up water. It is felt that these organic acids promote corrosion by lowering the pH of boiler waters and may even be corrosive themselves. 4.4 Such low molecular weight organics may also be produced when ultraviolet light is used to produce bacteria-free water for semiconductor processing. Such polar organic contaminants are suspected of causing reduced semiconductor yields. 4.5 Phosphates are commonly added to drum boilers in the low mg/L level to precipitate calcium and magnesium and thereby prevent scale formation. Ion chromatography can be used to monitor the concentration of such chemicals in boiler water, as well as detect unwanted carry-over into the steam. SCOPE 1.1 These test methods cover the determination of trace (μg/L) levels of fluoride, acetate, formate, chloride, phosphate, and sulfate in high purity water using ion chromatography in combination with sample preconcentration. Other anions, such as bromide, nitrite, nitrate, sulfite, and iodide can be determined by this method. However, since they are rarely present in significant concentrations in high purity water, they are not included in this test method. Two test methods are presented and their ranges of application, as determined by a collaborative study, are as follows: Range Tested (μg/L Added) Limit of DetectionA (Single Operator) (μg/L) Sections Test Method A: 7–16 Chloride 0–24 0.8 Phosphate 0–39 B Sulfate 0–55 1.8 Test Method B: 17–24 Fluoride 0–14 0.7 Acetate 0–414 6.8 Formate 0–346 5.6 (A) Limit of detection is lowest measurable concentration not reportable as zero at 99 % level of confidence as per EPRI study as cited in Sections 16 and 24.(B) Insufficient data to calculate limit of detection. 1.2 It is the user's responsibility to ensure the validity of these test methods for waters of untested matrices. 1.3 The common practical range of Test Method A is as follows: chloride, 1 to 100 μg/L, phosphate, 3 to 100 μg/L, and sulfate, 2 to 100 μg/L. 1.4 The common practical range of Test Method B is as follows: fluoride, 1 to 100 μg/L, acetate, 10 to 200 μg/L, and formate, 5 to 200 μg/L. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 4.1 The anions fluoride, chloride, and sulfate have been identified as important contributors to corrosion of high pressure boilers, electric power turbines and their associated heat exchangers. Many electric power utilities attempt to reduce these contaminants in their boiler feed water to less than 1 μg/L. 4.2 In the semiconductor manufacturing process these ions, among others, have been identified as a cause of low product yield and, thus, must be monitored and controlled to levels similar to those required by the electric power industry. 4.3 Low molecular weight organic acids, such as acetate and formate, have been found in many steam generator feed waters and condensates. They are believed to come from the high temperature breakdown of organic matter found in boiler make up water. It is felt that these organic acids promote corrosion by lowering the pH of boiler waters and may even be corrosive themselves. 4.4 Such low molecular weight organics may also be produced when ultraviolet light is used to produce bacteria-free water for semiconductor processing. Such polar organic contaminants are suspected of causing reduced semiconductor yields. 4.5 Phosphates are commonly added to drum boilers in the low mg/L level to precipitate calcium and magnesium and thereby prevent scale formation. Ion chromatography can be used to monitor the concentration of such chemicals in boiler water, as well as detect unwanted carry-over into the steam. SCOPE 1.1 These test methods cover the determination of trace (μg/L) levels of fluoride, acetate, formate, chloride, phosphate, and sulfate in high purity water using ion chromatography in combination with sample preconcentration. Other anions, such as bromide, nitrite, nitrate, sulfite, and iodide can be determined by this method. However, since they are rarely present in significant concentrations in high purity water, they are not included in this test method. Two test methods are presented and their ranges of application, as determined by a collaborative study, are as follows: Range Tested (μg/L Added) Limit of DetectionA (Single Operator) (μg/L) Sections Test Method A: 7–16 Chloride 0–24 0.8 Phosphate 0–39 B Sulfate 0–55 1.8 Test Method B: 17–24 Fluoride 0–14 0.7 Acetate 0–414 6.8 Formate 0–346 5.6 (A) Limit of detection is lowest measurable concentration not reportable as zero at 99 % level of confidence as per EPRI study as cited in Sections 16 and 24.(B) Insufficient data to calculate limit of detection. 1.2 It is the user's responsibility to ensure the validity of these test methods for waters of untested matrices. 1.3 The common practical range of Test Method A is as follows: chloride, 1 to 100 μg/L, phosphate, 3 to 100 μg/L, and sulfate, 2 to 100 μg/L. 1.4 The common practical range of Test Method B is as follows: fluoride, 1 to 100 μg/L, acetate, 10 to 200 μg/L, and formate, 5 to 200 μg/L. 1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM D5542-16 is classified under the following ICS (International Classification for Standards) categories: 71.040.30 - Chemical reagents. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5542-16 has the following relationships with other standards: It is inter standard links to ASTM D1129-13(2020)e2, ASTM D1066-18e1, ASTM D1066-18, ASTM D4453-17, ASTM D4453-16, ASTM D3856-11, ASTM D1066-11, ASTM D5810-96(2011), ASTM D4453-11, ASTM D3370-10, ASTM D1129-10, ASTM D3370-08, ASTM D3370-07, ASTM D1066-06, ASTM D1129-06ae1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5542-16 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5542 − 16
Standard Test Methods for
Trace Anions in High Purity Water by Ion Chromatography
This standard is issued under the fixed designation D5542; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope responsibility of the user of this standard to establish appro-
priate safety and health practices and determine the applica-
1.1 These test methods cover the determination of trace
bility of regulatory limitations prior to use.
(µg/L) levels of fluoride, acetate, formate, chloride, phosphate,
and sulfate in high purity water using ion chromatography in
2. Referenced Documents
combination with sample preconcentration. Other anions, such
as bromide, nitrite, nitrate, sulfite, and iodide can be deter-
2.1 ASTM Standards:
mined by this method. However, since they are rarely present D1066 Practice for Sampling Steam
in significant concentrations in high purity water, they are not
D1129 Terminology Relating to Water
included in this test method. Two test methods are presented D1192 Guide for Equipment for Sampling Water and Steam
and their ranges of application, as determined by a collabora-
in Closed Conduits (Withdrawn 2003)
tive study, are as follows: D1193 Specification for Reagent Water
A
D3370 Practices for Sampling Water from Closed Conduits
Limit of Detection
Range Tested
(Single Operator) Sections
D3856 Guide for Management Systems in Laboratories
(µg/L Added)
(µg/L)
Engaged in Analysis of Water
Test Method A: 7–16
Chloride 0–24 0.8
D4210 Practice for Intralaboratory Quality Control Proce-
B
Phosphate 0–39
dures and a Discussion on Reporting Low-Level Data
Sulfate 0–55 1.8
(Withdrawn 2002)
Test Method B: 17–24
Fluoride 0–14 0.7
D4453 Practice for Handling of High Purity Water Samples
Acetate 0–414 6.8
D5810 Guide for Spiking into Aqueous Samples
Formate 0–346 5.6
D5847 Practice for Writing Quality Control Specifications
A
Limit of detection is lowest measurable concentration not reportable as zero at
for Standard Test Methods for Water Analysis
99 % level of confidence as per EPRI study as cited in Sections 16 and 24.
B
Insufficient data to calculate limit of detection.
3. Terminology
1.2 It is the user’s responsibility to ensure the validity of
these test methods for waters of untested matrices.
3.1 Definitions:
3.1.1 For definitions of terms used in this standard, refer to
1.3 The common practical range of Test Method A is as
Terminology D1129.
follows: chloride, 1 to 100 µg/L, phosphate, 3 to 100 µg/L, and
3.2 Definitions of Terms Specific to This Standard:
sulfate, 2 to 100 µg/L.
3.2.1 analytical columns, n—a combination of one or more
1.4 The common practical range of Test Method B is as
guard columns followed by one or more separator columns
follows: fluoride, 1 to 100 µg/L, acetate, 10 to 200 µg/L, and
used to separate the ions of interest.
formate, 5 to 200 µg/L.
3.2.1.1 Discussion—It should be remembered that all of the
1.5 The values stated in SI units are to be regarded as columns in series contribute to the overall capacity of the
standard. No other units of measurement are included in this analytical column set.
standard.
3.2.2 breakthrough volume, n—the maximum sample vol-
1.6 This standard does not purport to address all of the ume that can be passed through a concentrator column before
the least tightly bound ion of interest is eluted.
safety concerns, if any, associated with its use. It is the
These test methods are under the jurisdiction of ASTM Committee D19 on
Water and are the direct responsibility of Subcommittee D19.03 on Sampling Water For referenced ASTM standards, visit the ASTM website, www.astm.org, or
and Water-Formed Deposits, Analysis of Water for Power Generation and Process contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Use, On-Line Water Analysis, and Surveillance of Water. Standards volume information, refer to the standard’s Document Summary page on
Current edition approved June 1, 2016. Published June 2016. Originally the ASTM website.
approved in 1994. Last previous edition approved in 2009 as D5542 – 04 (2009). The last approved version of this historical standard is referenced on
DOI: 10.1520/D5542-16. www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5542 − 16
detector background and at the same time enhance detector
response to the ions of interest.
4. Significance and Use
4.1 The anions fluoride, chloride, and sulfate have been
identified as important contributors to corrosion of high pres-
sure boilers, electric power turbines and their associated heat
exchangers. Many electric power utilities attempt to reduce
these contaminants in their boiler feed water to less than 1
µg/L.
4.2 In the semiconductor manufacturing process these ions,
among others, have been identified as a cause of low product
yield and, thus, must be monitored and controlled to levels
similar to those required by the electric power industry.
4.3 Lowmolecularweightorganicacids,suchasacetateand
formate, have been found in many steam generator feed waters
and condensates. They are believed to come from the high
temperature breakdown of organic matter found in boiler make
upwater.Itisfeltthattheseorganicacidspromotecorrosionby
lowering the pH of boiler waters and may even be corrosive
themselves.
4.4 Such low molecular weight organics may also be
producedwhenultravioletlightisusedtoproducebacteria-free
water for semiconductor processing. Such polar organic con-
taminants are suspected of causing reduced semiconductor
yields.
4.5 Phosphates are commonly added to drum boilers in the
low mg/L level to precipitate calcium and magnesium and
thereby prevent scale formation. Ion chromatography can be
used to monitor the concentration of such chemicals in boiler
water, as well as detect unwanted carry-over into the steam.
5. Reagents
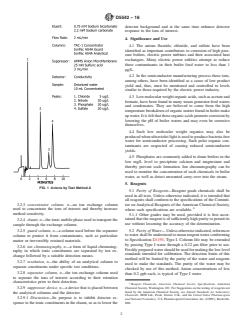
FIG. 1 Anions by Test Method A
5.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
all reagents shall conform to the specifications of the Commit-
3.2.3 concentrator column, n—an ion exchange column
tee onAnalytical Reagents of theAmerican Chemical Society,
used to concentrate the ions of interest and thereby increase
where such specifications are available.
method sensitivity.
5.1.1 Other grades may be used, provided it is first ascer-
tained that the reagent is of sufficiently high purity to permit its
3.2.4 eluant, n—the ionic mobile phase used to transport the
use without lessening the accuracy of the determination.
sample through the exchange column.
5.2 Purity of Water—Unlessotherwiseindicated,references
3.2.5 guard column, n—a column used before the separator
towatershallbeunderstoodtomeanreagentwaterconforming
column to protect it from contaminants, such as particulate
to Specification D1193, Type I. Column life may be extended
matter or irreversibly retained materials.
by passing Type I water through a 0.22 µm filter prior to use.
3.2.6 ion chromatography, n—a form of liquid chromatog-
Freshlypreparedwatershouldbeusedformakingthelowlevel
raphy in which ionic constituents are separated by ion ex-
standards intended for calibration. The detection limits of this
change followed by a suitable detection means.
method will be limited by the purity of the water and reagents
3.2.7 resolution, n—the ability of an analytical column to
used to make the standards. The purity of the water may be
separate constituents under specific test conditions.
checked by use of this method. Anion concentrations of less
3.2.8 separator column, n—the ion exchange column used
than 0.2 ppb each, is typical of Type I water.
to separate the ions of interest according to their retention
characteristics prior to their detection.
Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For Suggestions on the testing of reagents not
3.2.9 suppressor device, n—a device that is placed between
listed by the American Chemical Society, see Annual Standards for Laboratory
the analytical columns and the detector.
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
3.2.9.1 Discussion—Its purpose is to inhibit detector re-
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
sponse to the ionic constituents in the eluant, so as to lower the MD.
D5542 − 16
6. Sampling acceptable instrument performance.This verification should be
performed on each analysis day or whenever fresh eluent has
6.1 Collect samples in accordance with Practice D1066,
been prepared. The CVS is a solution of method analytes of
Guide D1192, Practices D3370, and Practice D4453, as appli-
known concentration (mid-calibration range) used to fortify
cable.
reagent water. The CVS must be prepared from a different
6.2 Collect samples in polystyrene bottles that should be
source than the calibration standards. If the determined CVS
filled to overflow and capped, so as to exclude air. Glass
concentrations are not within 615 % of the known values, the
sample bottles should not be used, as they can contribute ionic
analyst shall reanalyze the CVS. If the values still fall outside
contamination.
acceptable limits, a new calibration curve is required which
6.3 Samples should be analyzed within 48 h of sampling. mustbeconfirmedbyasuccessfulCVSbeforecontinuingwith
on-going analyses.
When acetate, formate or phosphate data are required, refrig-
erate at 4°C upon sampling.
8.4 OnecontinuingCVSshallbeanalyzedwitheachsample
batch (maximum of 20 samples) to verify the previously
6.4 To prevent added ionic contamination, no preservation
established calibration curves. If the determined analyte con-
or filtration of the sample shall be done.
centrations fall outside acceptable limits (615 %) that analyte
is judged out of control, and the source of the problem must be
TEST METHOD A—CHLORIDE, PHOSPHATE, AND
identified before continuing with on-going analyses. All
SULFATE
samples following the last acceptable CVS should be reana-
lyzed.
7. Scope
8.5 One Laboratory Control Sample (LCS) shall be ana-
7.1 This test method is optimized for the quantitative
lyzed with each sample batch (maximum of 20 samples) to
determination of trace levels of chloride, phosphate, and
ensure the test method is in control. The LCS is a solution of
sulfate. Anions such as fluoride, acetate, and formate can be
the test method analytes spiked at concentration levels of the
detected by this method, but are not reliably resolved from
IDP solution added to a matrix that sufficiently challenges the
each other. See Fig. 1 for a typical chromatogram.
test method. The LCS must be taken through all of the steps of
7.2 Using a concentrated sample volume of 20 mL, the test
this analytical method including sample preservation and
method is applicable in the range outlined in Section 1. The
pretreatment. The analyte recoveries for the LCS must fall
range of this test method may be extended by concentrating a
within the control limits listed below:
smaller or a larger sample volume. Be sure not to exceed
Upper Control Limit 5 x13S (1)
concentrator column breakthrough volume (see annex).
Lower Control Limit 5 x 2 3S (2)
8. Quality Control
where:
8.1 Before this test is applied to analyzing unknown
x = percent mean recovery, and
samples, the analyst shall establish quality control procedures
S = standard deviation of the mean recovery, as determined
as recommended in Practices D4210 and D5847, and Guide
from historical values for the equivalent concentration
D3856. In order to be certain that analytical values obtained by
and matrix.
this test method are valid and accurate within the confidence
8.5.1 If the results do not fall within these limits, analysis of
limits of the tests, the QC procedures described in this section
samples is halted until the problem is corrected. Either all
must be followed.
samples in the batch must be reanalyzed so as to pass these
8.2 The laboratory using this test shall perform an initial
performance criteria, or the results must be qualified with an
demonstration of laboratory capability. Analyze seven repli-
indication that they do not fall within the performance criteria
cates of an Initial Demonstration of Performance (IDP) solu-
of the test method.
tion. The IDP solution contains method analytes of known
8.6 A reagent blank shall be analyzed as part of the initial
concentration, prepared from a different source to the calibra-
generation of calibration curves. A reagent blank shall also be
tion standards, used to fortify reagent water. Ideally, the IPD
analyzed with each sample batch (maximum of 20 samples) to
solution should be prepared by an independent source from
check for contamination introduced by the laboratory or use of
reference materials.
the test method.
8.2.1 The mean and standard deviation of seven values for
each test method analyte shall then be calculated and
8.7 One matrix spike (MS) shall be analyzed with each
compared, according to Practice D5847, to the single operator
sample batch (maximum of 20 samples) to test method
precision and recovery established for this test method.
recovery. Spike a portion of one sample from each batch with
8.2.2 If the values obtained for the IDP precision and
aknownconcentrationofthemethodanalytes.TheMSshallbe
recovery do not meet the criteria described above, initial
prepared in accordance with that outlined in Guide D5810 and
demonstration of performance must be repeated until the
section 11.11 of Guide D3856. The % recovery of the spike
results fall within these criteria.
must fall within % recovery 6 analyst % RSD for an
equivalent spike concentration and matrix.
8.3 When beginning use of this method, a Calibration
Verification Standard (CVS) containing each test method 8.8 One matrix duplicate (MD) shall be analyzed with each
analyteshallbeanalyzedtoverifythecalibrationstandardsand sample batch (maximum of 20 samples) to test method
D5542 − 16
9.1.1 This also converts the anions to their acid form, thus
enhancing their conductivity. The eluant stream then passes
throughanelectricalconductivitydetector,wheretheseparated
anions are detected. A strip chart recorder and/or a chromato-
graphic integrator is used for data presentation.
9.2 The anions are identified based on their retention times,
when compared to known standards. Quantitation is accom-
plished by measuring the peak height or area and comparing it
to a calibration curve generated from known standards.
10. Interferences
10.1 When working at microgram per litre concentrations
and lower, contamination can be a very serious problem.
Extremecaremustbeexercisedinallphasesofthetestmethod
(samplecollection,storage,andanalysis)toeliminatecontami-
nation.
10.2 As with other types of chromatography, if one of the
sample components is present at very high concentration
levels, it may interfere by causing a very large peak on the
chromatogram that could mask other peaks present. This type
FIG. 2 Schematic of an Ion Chromatograph
of interference may normally be minimized by dilution of the
sample, depending on the concentration of other anions.
10.3 When loading concentrator columns, high concentra-
precision. If non-detects are expected in all the samples to be
tionsofcertainanionsmaycauselowbreakthroughvolumesof
analyzed, a Matrix Spike Duplicate (MSD) shall be analyzed
other anions. These certain anions may act as eluants and
instead of a MD. Prepare the MSD as outlined in Guides
displace other anions from the concentrator column. See annex
D5810 and D3856. The percent recovery of the spike must fall
to determine breakthrough volume. Do not attempt to concen-
within % recovery 6 analyst % RSD for an equivalent spike
trateavolumeofsamplegreaterthan80 %ofthebreakthrough
concentration and matrix. Calculate the standard deviation and
volume.
use the F-test (see Practice D5847, section 6.3.1.1) to compare
with the single operator precision given in Tables 3–8 for the
10.4 Samples containing high (mg/L) concentrations of
equivalent analyte concentration and matrix type. Evaluate
ammonia, morpholine, or other additives which raise the
performance according to Practice D5847.
hydroxide concentration (pH) of the sample may cause low
breakthroughvolumes.Thisproblemmaybeavoidedbytaking
8.9 An independent reference material (IRM) shall be ana-
such samples after the cation resin of a cation conductivity
lyzedatleastonceperquarterinordertoverifythequantitative
detector.
valuesproducedbythetestmethod.Theanalyteconcentrations
ofthereferencematerialshouldbeinappropriaterangeascited
11. Apparatus
in 1.1 of these test methods. The recovery values obtained for
each test method analyte must fall within the control limits
11.1 Ion Chromatograph—The ion chromatograph should
specified by the supplier of the IRM.
have the following components assembled, as shown in Fig. 2.
8.10 The laboratory may perform additional quality control 11.1.1 Eluant and Regenerant Containers.
as desired or as required for regulatory compliance.
11.1.2 Eluant Pump, capable of delivering 2 to 5 mL/min of
eluant at a pressure of up to 2000 psig. Wetted parts of the
9. Summary of Test Method
pump should be nonmetallic, so as not to contaminate the
concentrator or analytical columns with metals, or both.
9.1 A flow diagram of an ion chromatograph is shown in
11.1.3 Sample Pump, capable of delivering up to 5 mL/min
Fig.2.Withthesamplingvalveintheloadposition,thedesired
of sample at a pressure of at least 200 psig. Wetted parts of the
volume of sample (for example, 20 mL) is pumped through a
pump should be nonmetallic, so as not to contaminate the
concentrator column where the anions of interest are trapped.
concentrator and/or analytical columns with metals.
The sampling valve is then switched to the inject position and
11.1.4 Concentrator Column—Anion exchange column
the pumped eluant, containing sodium carbonate and
with sufficient capacity to concentrate at least 20 mLof sample
bicarbonate, sweeps these anions through the analytical col-
before reaching chloride breakthrough.
umns where they are separated according to their retention
characteristics relative to the anions in the eluant. The eluant 11.1.5 Guard Column— Anion exchange column, typically
stream next passes through a suppressor where all cations are of the same anion exchange material used in the separator
exchanged for hydrogen ions. This converts the carbonate and column. The purpose of this column is to protect the separator
bicarbonate in the eluant to the poorly ionized carbonic acid, column from particulate matter and irreversibly retained ma-
thus reducing the background conductivity. terials.
D5542 − 16
11.1.6 Separator Column—Anionexchangecolumncapable stock solution to 1 L. These solutions should be prepared fresh
of separating chloride from the injection void volume, as well weekly and should be stored in polypropylene or polystyrene
as resolving the anions chloride, phosphate, and sulfate. bottles.
11.1.7 Suppressor Column—A cation exchanger which is
12.5 Working Standard Solutions—Prepare a blank and at
continuously regenerated by a flow of dilute sulfuric acid.
least three different working standards containing the anions of
11.1.8 Detector—A low-volume, flow-through,
interest. The combination anion solutions should be prepared
temperature-compensated electrical conductivity cell equipped
in volumetric flasks and then transferred to polystyrene bottles.
with a meter capable of reading from 0 to 15 000 uS/cm on a
These standards must be prepared fresh daily. The concentra-
linear scale.
tion range for the three standards will be dependent on the
11.1.9 Recorder, compatible with the detector output with a
levels expected in the samples. If desired, a standard may be
full-scale response time of2sor less.
prepared that contains all six anions.Atypical range would be
11.1.10 Integrator—Anelectronicintegrator,suchasisused
5, 10, and 25 µg/L of each anion per standard. This would be
with gas and liquid chromatographs, may be used to quantitate
prepared by taking 5, 10, and 25 mL of the standard stock
peak area, as well as peak height. The peak area data can be
solution and diluting to 1 L with water for each standard. The
used in the same way peak height is used to quantitate results.
blank standard is a portion of the same water used to prepare
11.1.11 Sample Bottles—Polystyrene culture bottles with a the working standard solutions.
total capacity of approximately 270 mL have been found
12.6 Some investigators prefer to work with standard solu-
satisfactory.
tions that are prepared by diluting microlitre qualities of stock
11.1.12 The following is a summary of the columns and
standards (or low level standards) using push-button microlitre
suppressor components used in the collaborative study.
pipettes. These have been found to be adequate for many
Concentrator column: AG-4A
purposes, but their precision may be limited.
Guard column: AG-4A
Separator column: AS-4A
A
13. Calibration
Suppressor device: Anion MicroMembrane Suppressor
A
Anion MicroMembrane Suppressor is a registered trademark of Dionex Corp.
13.1 Determine the retention time for each anion by analyz-
ingastandardsolutioncontainingonlytheanionofinterestand
12. Reagents
noting the time required for a peak to appear on the chromato-
gram.
12.1 Eluant—Dissolve 0.25 g of sodium bicarbonate (0.75
millimolar) and 0.93 g of sodium carbonate (2.2 millimolar) in
13.2 Analyze the blank and each of the working standard
water and dilute to 4 L with water. Other eluants may also
solutions described in 12.5, according to the following:
prove to be acceptable, provided they give the proper resolu-
13.2.1 Analyze the blank (known volume of water loaded
tion between the component peaks.
onto the concentrator column).
13.2.2 Analyze the standard solutions (known volume of
12.2 Suppressor Regenerant—Cautiously add 3 mL of con-
standard solution loaded onto the concentrator column).
centrated sulfuric acid to 4 L of water.
13.2.3 If a measurable peak is noted on the blank chromato-
12.3 Stock Solutions:
gram that coincides in retention time with a peak of interest,
12.3.1 Fluoride Solution, Stock (1.00 mL = 1.00 mg F)—
determine the concentration of each of the ions in the blank by
Dissolve 2.210 g of sodium fluoride (NaF) in water and dilute
plotting the standard concentration on the abcissa and the peak
to 1 L with water.
height or area on the ordinate of linear graph paper.
12.3.2 Acetate Solution Stock (1.00 mL = 1 mg acetate)—
13.2.4 Extrapolate the line through the abscissa. The point
Dissolve 1.389 g of sodium acetate in water and dilute to 1 L
at which the line intercepts the abscissa represents the concen-
with water.
tration of the anion in the blank.
12.3.3 Formate Solution Stock (1.00 mL = 1 mg formate)—
13.2.5 Addtheconcentrationoftheanionintheblanktothe
Dissolve 1.511 g sodium formate in water and dilute to 1 L
nominal concentration of the prepared standard solution.
with water.
13.2.5.1 Example—A 10-µg/L prepared chloride standard
12.3.4 Chloride Solution Stock (1.00 mL = 1.00 mg
solution with a blank correction of 0.2 µg/L would be plotted
chloride)— Dry sodium chloride (NaCl) for1hat 100°C and
as 10.2 µg/L chloride.
cool in a desiccator. Dissolve 1.648 g of the dry salt in water
13.3 Reconstructacalibrationcurvebyplottingtheadjusted
and dilute to 1 L with water.
concentration of the standards versus peak area or peak height
12.3.5 Phosphate Solution Stock (1.00 mL = 1.00 mg
or adjust the concentration of the standard values entered into
PO )—Dissolve 1.433 g of potassium dihydrogen phosphate
a computer or computing integrator for direct read-out in
(KH PO ) in water and dilute to 1 L with water.
2 4
concentration units.
12.3.6 Sulfate Solution Stock (1.00 mL = 1.0
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5542 − 04 (Reapproved 2009) D5542 − 16
Standard Test Methods for
Trace Anions in High Purity Water by Ion Chromatography
This standard is issued under the fixed designation D5542; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 These test methods cover the determination of trace (μg/L) levels of fluoride, acetate, formate, chloride, phosphate, and
sulfate in high purity water using ion chromatography in combination with sample preconcentration. Other anions, such as
bromide, nitrite, nitrate, sulfite, and iodide can be determined by this method. However, since they are rarely present in significant
concentrations in high purity water, they are not included in this test method. Two test methods are presented and their ranges of
application, as determined by a collaborative study, are as follows:
A
Limit of Detection
Range Tested
(Single Operator) Sections
(μg/L Added)
(μg/L)
Test Method A: 7–15
Test Method A: 7–16
Chloride 0–24 0.8
B
Phosphate 0–39
Sulfate 0–55 1.8
Test Method B: 16–23
Test Method B: 17–24
Fluoride 0–14 0.7
Acetate 0–414 6.8
Formate 0–346 5.6
A
Limit of detection is lowest measurable concentration not reportable as zero at 99 % level of confidence as per EPRI study as cited in Sections 1516 and 2324.
B
Insufficient data to calculate limit of detection.
1.2 It is the user’s responsibility to ensure the validity of these test methods for waters of untested matrices.
1.3 The common practical range of Test Method A is as follows: chloride, 1 to 100 μg/L, phosphate, 3 to 100 μg/L, and sulfate,
2 to 100 μg/L.
1.4 The common practical range of Test Method B is as follows: fluoride, 1 to 100 μg/L, acetate, 10 to 200 μg/L, and formate,
5 to 200 μg/L.
1.5 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
D1066 Practice for Sampling Steam
D1129 Terminology Relating to Water
D1192 Guide for Equipment for Sampling Water and Steam in Closed Conduits (Withdrawn 2003)
D1193 Specification for Reagent Water
D3370 Practices for Sampling Water from Closed Conduits
D3856 Guide for Management Systems in Laboratories Engaged in Analysis of Water
These test methods are under the jurisdiction of ASTM Committee D19 on Water and are the direct responsibility of Subcommittee D19.03 on Sampling Water and
Water-Formed Deposits, Analysis of Water for Power Generation and Process Use, On-Line Water Analysis, and Surveillance of Water.
Current edition approved May 1, 2009June 1, 2016. Published June 2009June 2016. Originally approved in 1994. Last previous edition approved in 20042009 as
D5542–04. DOI: 10.1520/D5542-04R09. – 04 (2009). DOI: 10.1520/D5542-16.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
The last approved version of this historical standard is referenced on www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5542 − 16
D4210 Practice for Intralaboratory Quality Control Procedures and a Discussion on Reporting Low-Level Data (Withdrawn
2002)
D4453 Practice for Handling of High Purity Water Samples
D5810 Guide for Spiking into Aqueous Samples
D5847 Practice for Writing Quality Control Specifications for Standard Test Methods for Water Analysis
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in these test methods this standard, refer to Terminology D1129.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 analytical columns—columns, n—a combination of one or more guard columns followed by one or more separator
columns used to separate the ions of interest. It should be remembered that all of the columns in series contribute to the overall
capacity of the analytical column set.
3.2.1.1 Discussion—
It should be remembered that all of the columns in series contribute to the overall capacity of the analytical column set.
3.2.2 breakthrough volume—volume, n—the maximum sample volume that can be passed through a concentrator column before
the least tightly bound ion of interest is eluted.
3.2.3 concentrator column—column, n—an ion exchange column used to concentrate the ions of interest and thereby increase
method sensitivity.
3.2.4 eluant—eluant, n—the ionic mobile phase used to transport the sample through the exchange column.
3.2.5 guard column—column, n—a column used before the separator column to protect it from contaminants, such as particulate
matter or irreversibly retained materials.
3.2.6 ion chromatography—chromatography, n—a form of liquid chromatography in which ionic constituents are separated by
ion exchange followed by a suitable detection means.
3.2.7 resolution—resolution, n—the ability of an analytical column to separate constituents under specific test conditions.
3.2.8 separator column—column, n—the ion exchange column used to separate the ions of interest according to their retention
characteristics prior to their detection.
3.2.9 suppressor device—device, n—a device that is placed between the analytical columns and the detector. Its purpose is to
inhibit detector response to the ionic constituents in the eluant, so as to lower the detector background and at the same time enhance
detector response to the ions of interest.
3.2.9.1 Discussion—
D5542 − 16
FIG. 1 Anions by Test Method A
Its purpose is to inhibit detector response to the ionic constituents in the eluant, so as to lower the detector background and at the
same time enhance detector response to the ions of interest.
4. Significance and Use
4.1 The anions fluoride, chloride, and sulfate have been identified as important contributors to corrosion of high pressure boilers,
electric power turbines and their associated heat exchangers. Many electric power utilities attempt to reduce these contaminants
in their boiler feed water to less than 1 μg/L.
4.2 In the semiconductor manufacturing process these ions, among others, have been identified as a cause of low product yield
and, thus, must be monitored and controlled to levels similar to those required by the electric power industry.
4.3 Low molecular weight organic acids, such as acetate and formate, have been found in many steam generator feed waters
and condensates. They are believed to come from the high temperature breakdown of organic matter found in boiler make up water.
It is felt that these organic acids promote corrosion by lowering the pH of boiler waters and may even be corrosive themselves.
4.4 Such low molecular weight organics may also be produced when ultraviolet light is used to produce bacteria-free water for
semiconductor processing. Such polar organic contaminants are suspected of causing reduced semiconductor yields.
4.5 Phosphates are commonly added to drum boilers in the low mg/L level to precipitate calcium and magnesium and thereby
prevent scale formation. Ion chromatography can be used to monitor the concentration of such chemicals in boiler water, as well
as detect unwanted carry-over into the steam.
5. Reagents
5.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available.
5.1.1 Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use
without lessening the accuracy of the determination.
5.2 Purity of Water— Unless otherwise indicated, references to water shall be understood to mean reagent water conforming
to Specification D1193, Type I. Column life may be extended by passing Type I water through a 0.22 μm filter prior to use. Freshly
prepared water should be used for making the low level standards intended for calibration. The detection limits of this method will
be limited by the purity of the water and reagents used to make the standards. The purity of the water may be checked by use of
this method. Anion concentrations of less than 0.2 ppb each, is typical of Type I water.
6. Sampling
6.1 Collect samples in accordance with Practice D1066, SpecificationGuide D1192, PracticePractices D3370, and Practice
D4453, as applicable.
6.2 Collect samples in polystyrene bottles that should be filled to overflow and capped, so as to exclude air. Glass sample bottles
should not be used, as they can contribute ionic contamination.
6.3 Samples should be analyzed within 48 h of sampling. When acetate, formate or phosphate data are required, refrigerate at
4°C upon sampling.
6.4 To prevent added ionic contamination, no preservation or filtration of the sample shall be done.
TEST METHOD A—CHLORIDE, PHOSPHATE, AND SULFATE
7. Scope
7.1 This test method is optimized for the quantitative determination of trace levels of chloride, phosphate, and sulfate. Anions
such as fluoride, acetate, and formate can be detected by this method, but are not reliably resolved from each other. See Fig. 1 for
a typical chromatogram.
7.2 Using a concentrated sample volume of 20 mL, the test method is applicable in the range outlined in Section 1. The range
of this test method may be extended by concentrating a smaller or a larger sample volume. Be sure not to exceed concentrator
column breakthrough volume (see annex).
Reagent Chemicals, American Chemical Society Specifications, American Chemical Society, Washington, DC. For Suggestions on the testing of reagents not listed by
the American Chemical Society, see Annual Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National
Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville, MD.
D5542 − 16
8. Quality Control
8.1 Before this test is applied to analyzing unknown samples, the analyst shall establish quality control procedures as
recommended in Practices D4210 and D5847, and Guide D3856. In order to be certain that analytical values obtained by this test
method are valid and accurate within the confidence limits of the tests, the QC procedures described in this section must be
followed.
8.2 The laboratory using this test shall perform an initial demonstration of laboratory capability. Analyze seven replicates of an
Initial Demonstration of Performance (IDP) solution. The IDP solution contains method analytes of known concentration, prepared
from a different source to the calibration standards, used to fortify reagent water. Ideally, the IPD solution should be prepared by
an independent source from reference materials.
8.2.1 The mean and standard deviation of seven values for each test method analyte shall then be calculated and compared,
according to Practice D5847, to the single operator precision and recovery established for this test method.
8.2.2 If the values obtained for the IDP precision and recovery do not meet the criteria described above, initial demonstration
of performance must be repeated until the results fall within these criteria.
8.3 When beginning use of this method, a Calibration Verification Standard (CVS) containing each test method analyte shall
be analyzed to verify the calibration standards and acceptable instrument performance. This verification should be performed on
each analysis day or whenever fresh eluent has been prepared. The CVS is a solution of method analytes of known concentration
(mid-calibration range) used to fortify reagent water. The CVS must be prepared from a different source than the calibration
standards. If the determined CVS concentrations are not within 615 % of the known values, the analyst shall reanalyze the CVS.
If the values still fall outside acceptable limits, a new calibration curve is required which must be confirmed by a successful CVS
before continuing with on-going analyses.
8.4 One continuing CVS shall be analyzed with each sample batch (maximum of 20 samples) to verify the previously
established calibration curves. If the determined analyte concentrations fall outside acceptable limits (615 %) that analyte is
judged out of control, and the source of the problem must be identified before continuing with on-going analyses. All samples
following the last acceptable CVS should be reanalyzed.
8.5 One Laboratory Control Sample (LCS) shall be analyzed with each sample batch (maximum of 20 samples) to ensure the
test method is in control. The LCS is a solution of the test method analytes spiked at concentration levels of the IDP solution added
to a matrix that sufficiently challenges the test method. The LCS must be taken through all of the steps of this analytical method
including sample preservation and pretreatment. The analyte recoveries for the LCS must fall within the control limits listed below:
Upper Control Limit 5 x13S (1)
Lower Control Limit 5 x 2 3S (2)
where:
x = percent mean recovery, and
S = standard deviation of the mean recovery, as determined from historical values for the equivalent concentration and matrix.
8.5.1 If the results do not fall within these limits, analysis of samples is halted until the problem is corrected. Either all samples
in the batch must be reanalyzed so as to pass these performance criteria, or the results must be qualified with an indication that
they do not fall within the performance criteria of the test method.
8.6 A reagent blank shall be analyzed as part of the initial generation of calibration curves. A reagent blank shall also be
analyzed with each sample batch (maximum of 20 samples) to check for contamination introduced by the laboratory or use of the
test method.
8.7 One matrix spike (MS) shall be analyzed with each sample batch (maximum of 20 samples) to test method recovery. Spike
a portion of one sample from each batch with a known concentration of the method analytes. The MS shall be prepared in
accordance with that outlined in Guide D5810 and section 11.11 of Guide D3856. The % recovery of the spike must fall within
% recovery 6 analyst % RSD for an equivalent spike concentration and matrix.
8.8 One matrix duplicate (MD) shall be analyzed with each sample batch (maximum of 20 samples) to test method precision.
If non-detects are expected in all the samples to be analyzed, a Matrix Spike Duplicate (MSD) shall be analyzed instead of a MD.
Prepare the MSD as outlined in Guides D5810 and D3856. The percent recovery of the spike must fall within % recovery 6 analyst
% RSD for an equivalent spike concentration and matrix. Calculate the standard deviation and use the F-test (see Practice D5847,
section 6.3.1.1) to compare with the single operator precision given in Tables 3–8 for the equivalent analyte concentration and
matrix type. Evaluate performance according to Practice D5847.
8.9 An independent reference material (IRM) shall be analyzed at least once per quarter in order to verify the quantitative values
produced by the test method. The analyte concentrations of the reference material should be in appropriate range as cited in 1.1
of these test methods. The recovery values obtained for each test method analyte must fall within the control limits specified by
the supplier of the IRM.
D5542 − 16
FIG. 2 Schematic of an Ion Chromatograph
8.10 The laboratory may perform additional quality control as desired or as required for regulatory compliance.
9. Summary of Test Method
9.1 A flow diagram of an ion chromatograph is shown in Fig. 2. With the sampling valve in the load position, the desired volume
of sample (for example, 20 mL) is pumped through a concentrator column where the anions of interest are trapped. The sampling
valve is then switched to the inject position and the pumped eluant, containing sodium carbonate and bicarbonate, sweeps these
anions through the analytical columns where they are separated according to their retention characteristics relative to the anions
in the eluant. The eluant stream next passes through a suppressor where all cations are exchanged for hydrogen ions. This converts
the carbonate and bicarbonate in the eluant to the poorly ionized carbonic acid, thus reducing the background conductivity.
9.1.1 This also converts the anions to their acid form, thus enhancing their conductivity. The eluant stream then passes through
an electrical conductivity detector, where the separated anions are detected. A strip chart recorder and/or a chromatographic
integrator is used for data presentation.
9.2 The anions are identified based on their retention times, when compared to known standards. Quantitation is accomplished
by measuring the peak height or area and comparing it to a calibration curve generated from known standards.
10. Interferences
10.1 When working at microgram per litre concentrations and lower, contamination can be a very serious problem. Extreme care
must be exercised in all phases of the test method (sample collection, storage, and analysis) to eliminate contamination.
10.2 As with other types of chromatography, if one of the sample components is present at very high concentration levels, it
may interfere by causing a very large peak on the chromatogram that could mask other peaks present. This type of interference
may normally be minimized by dilution of the sample, depending on the concentration of other anions.
10.3 When loading concentrator columns, high concentrations of certain anions may cause low breakthrough volumes of other
anions. These certain anions may act as eluants and displace other anions from the concentrator column. See annex to determine
breakthrough volume. Do not attempt to concentrate a volume of sample greater than 80 % of the breakthrough volume.
10.4 Samples containing high (mg/L) concentrations of ammonia, morpholine, or other additives which raise the hydroxide
concentration (pH) of the sample may cause low breakthrough volumes. This problem may be avoided by taking such samples after
the cation resin of a cation conductivity detector.
11. Apparatus
11.1 Ion Chromatograph—The ion chromatograph should have the following components assembled, as shown in Fig. 2.
11.1.1 Eluant and Regenerant Containers.
11.1.2 Eluant Pump, capable of delivering 2 to 5 mL/min of eluant at a pressure of up to 2000 psig. Wetted parts of the pump
should be nonmetallic, so as not to contaminate the concentrator or analytical columns with metals, or both.
D5542 − 16
11.1.3 Sample Pump, capable of delivering up to 5 mL/min of sample at a pressure of at least 200 psig. Wetted parts of the pump
should be nonmetallic, so as not to contaminate the concentrator and/or analytical columns with metals.
11.1.4 Concentrator Column—Anion exchange column with sufficient capacity to concentrate at least 20 mL of sample before
reaching chloride breakthrough.
11.1.5 Guard Column— Anion exchange column, typically of the same anion exchange material used in the separator column.
The purpose of this column is to protect the separator column from particulate matter and irreversibly retained materials.
11.1.6 Separator Column—Anion exchange column capable of separating chloride from the injection void volume, as well as
resolving the anions chloride, phosphate, and sulfate.
11.1.7 Suppressor Column—A membrane based cation exchanger which is continuously regenerated by a flow of dilute sulfuric
acid.
11.1.8 Detector—A low-volume, flow-through, temperature-compensated electrical conductivity cell equipped with a meter
capable of reading from 0 to 1000 15 000 uS/cm on a linear scale.
11.1.9 Recorder, compatible with the detector output with a full-scale response time of 2 s or less.
11.1.10 Integrator—An electronic integrator, such as is used with gas and liquid chromatographs, may be used to quantitate
peak area, as well as peak height. The peak area data can be used in the same way peak height is used to quantitate results.
11.1.11 Sample Bottles—Polystyrene culture bottles with a total capacity of approximately 270 mL have been found satisfactory.
11.1.12 The following is a summary of the columns and suppressor components used in the collaborative study.
Concentrator column: AG-4A
Guard column: AG-4A
Separator column: AS-4A
A
Suppressor device: Anion MicroMembrane Suppressor
A
Anion MicroMembrane Suppressor is a registered trademark of Dionex Corp.
12. Reagents
12.1 Eluant—Dissolve 0.25 g of sodium bicarbonate (0.75 millimolar) and 0.93 g of sodium carbonate (2.2 millimolar) in water
and dilute to 4 L with water. Other eluants may also prove to be acceptable, provided they give the proper resolution between the
component peaks.
12.2 Suppressor Regenerant—Cautiously add 3 mL of concentrated sulfuric acid to 4 L of water.
12.3 Stock Solutions:
12.3.1 Fluoride Solution, Stock (1.00 mL = 1.00 mg F)—Dissolve 2.210 g of sodium fluoride (NaF) in water and dilute to 1 L
with water.
12.3.2 Acetate Solution Stock (1.00 mL = 1 mg acetate)—Dissolve 1.389 g of sodium acetate in water and dilute to 1 L with
water.
12.3.3 Formate Solution Stock (1.00 mL = 1 mg formate)—Dissolve 1.511 g sodium formate in water and dilute to 1 L with
water.
12.3.4 Chloride Solution Stock (1.00 mL = 1.00 mg chloride)— Dry sodium chloride (NaCl) for 1 h at 100°C and cool in a
desiccator. Dissolve 1.648 g of the dry salt in water and dilute to 1 L with water.
12.3.5 Phosphate Solution Stock (1.00 mL = 1.00 mg PO )—Dissolve 1.433 g of potassium dihydrogen phosphate (KH PO )
4 2 4
in water and dilute to 1 L with water.
12.3.6 Sulfate Solution Stock (1.00 mL = 1.00 mg SO )—Dry sodium sulfate for 1 h at 105°C and cool in a desiccator. Dissolve
1.479 g of the dried salt in water and dilute to 1 L with water.
12.4 Intermediate Standard Solutions—Prepare a 1000 μg/L standard of each anion by diluting 1.00 mL of each standard stock
solution to 1 L. These solutions should be prepared fresh weekly and should be stored in polypropylene or polystyrene bottles.
12.5 Working Standard Solutions —Solutions—Prepare a blank and at least three different working standards containing the
anions of interest. The combination anion solutions should be prepared in volumetric flasks and then transferred to polystyrene
bottles. These standards must be prepared fresh daily. The concentration range for the three standards will be dependent on the
levels expected in the samples. If desired, a standard may be prepared that contains all six anions. A typical range would be 5, 10,
and 25 μg/L of each anion per standard. This would be prepared by taking 5, 10, and 25 mL of the standard stock solution and
diluting to 1 L with water for each standard. The blank standard is a portion of the same water used to prepare the working standard
solutions.
12.6 Some investigators prefer to work with standard solutions that are prepared by diluting microlitre qualities of stock
standards (or low level standards) using push-button microlitre pipettes. These have been found to be adequate for many purposes,
but their precision may be limited.
13. Calibration
13.1 Determine the retention time for each anion by analyzing a standard solution containing only the anion of interest and
noting the time required for a peak to appear on the chromatogram.
D5542 − 16
13.2 Analyze the blank and each of the working standard solutions described in 11.512.5, according to the following:
13.2.1 Analyze the blank (known volume of water loaded onto the concentrator column).
13.2.2 Analyze the standard solutions (known volume of standard solution loaded onto the concentrator column).
13.2.3 If a measurable peak is noted on the blank chromatogram that coincides in retention time with a peak of interest,
determine the concentration of each of the ions in the blank by plotting the standard concentration on the abcissa and the peak
height or area on the ordinate of linear graph paper.
13.2.4 Extrapolate the line through the abscissa. The point at which the line intercepts the abscissa represents the concentration
of the anion in the blank.
13.2.5 Add the concentration of the anion in the blank to the nominal concentration of the prepared standard solution.
13.2.5.1 Example—A 10-μg/L prepared chloride standard solution with a blank correction of 0.2 μg/L would be plotted as 10.2
μg/L chloride.
13.3 Reconstruct a calibration curve by plotting the adjusted concentration of the st
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...