ASTM E394-22
(Test Method)Standard Test Method for Iron in Trace Quantities Using the 1,10-Phenanthroline Method
Standard Test Method for Iron in Trace Quantities Using the 1,10-Phenanthroline Method
SIGNIFICANCE AND USE
4.1 This test method is suitable for determining trace concentrations of iron in a wide variety of products, provided that appropriate sample preparation has rendered the iron and sample matrix soluble in water or other suitable solvent (see 10.1 and Note 4).
4.2 This test method assumes that the amount of color developed is proportional to the amount of iron in the test solution. The calibration curve is linear over the specified range. Possible interferences are described in Section 5.
SCOPE
1.1 This test method covers the determination of trace concentrations of iron in the range from 1 to 100 μg/g in a wide variety of products.
1.2 In determining the conformance of the test results using this method to applicable specifications; results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 Consult current OSHA regulations, suppliers’ Safety Data Sheets, and local regulations for all materials used in this specification.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Aug-2022
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.04 - Instrumental Analysis
Relations
- Effective Date
- 01-Nov-2016
- Refers
ASTM E275-08 - Describing and Measuring Performance of Ultraviolet and Visible Spectrophotometers - Effective Date
- 15-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Jun-2007
- Effective Date
- 15-Nov-2006
- Effective Date
- 15-Sep-2006
- Effective Date
- 01-May-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Dec-2004
- Effective Date
- 01-May-2004
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-May-2002
- Effective Date
- 10-Feb-2001
- Effective Date
- 10-Feb-2001
- Effective Date
- 10-May-1999
Overview
ASTM E394-22 is an internationally recognized standard test method for the determination of iron in trace quantities using the 1,10-phenanthroline photometric method. Developed by ASTM International, this standard provides laboratories with a reliable, precise, and repeatable procedure to measure iron concentrations ranging from 1 to 100 μg/g in a variety of products, provided the iron and sample matrix are rendered water-soluble. This spectrophotometric technique utilizes the coloring reaction between iron(II) ions and 1,10-phenanthroline, allowing for accurate quantification based on the intensity of the developed color.
Key Topics
- Trace Iron Determination: The method is suitable for detecting extremely low levels of iron, enhancing quality control where iron contamination must be minimized.
- Photometric/Spectrophotometric Analysis: The standard employs measurement of absorbance at approximately 510 nm after color development with 1,10-phenanthroline.
- Calibration and Linearity: Results assume a linear calibration curve over the specified range, with linear regression recommended for quantification.
- Sample Requirements: Applies to any sample soluble in water or approved solvents with pH less than 2; sample preparation is critical for accuracy.
- Interference Management: Outlines cationic and anionic interferences, particularly from species affecting the reducing agent or interaction with phenanthroline.
- Precision and Reproducibility: Evaluates repeatability, within-laboratory precision, and multilaboratory reproducibility to ensure robust analytical performance.
- Quality Control: Suggests ongoing verification of equipment and sample handling per established QA/QC practices, including interlaboratory testing programs.
Applications
The ASTM E394-22 standard is applicable across a broad range of industries where precise measurement of iron at trace levels is essential:
- Water and Wastewater Testing: Ensures compliance with regulatory specifications by verifying low iron concentrations in potable or industrial water.
- Chemical Manufacturing: Supports process control and contamination detection in aromatic, industrial, and specialty chemicals.
- Metals and Ores: Provides a method for confirming trace iron levels in metal finishing or ore analysis.
- Pharmaceuticals and Food Products: Addresses the need to monitor iron content to meet product safety and quality guidelines.
- Environmental Monitoring: Facilitates environmental impact assessments by quantifying trace iron in natural or industrial samples.
Related Standards
The following ASTM standards offer useful complementary guidance or are referenced within ASTM E394-22:
- ASTM D1193: Specification for Reagent Water
- ASTM D6809: Guide for Quality Control and Quality Assurance for Aromatic Hydrocarbons and Related Materials
- ASTM E29: Practice for Using Significant Digits in Test Data
- ASTM E60: Practice for Analysis of Metals, Ores, and Related Materials by Spectrophotometry
- ASTM E180: Practice for Determining the Precision of ASTM Methods
- ASTM E200: Practice for the Preparation and Storage of Standard Solutions
- ASTM E275: Practice for Measuring Performance of Ultraviolet and Visible Spectrophotometers
- ASTM E300: Practice for Sampling Industrial Chemicals
Practical Value
Adopting ASTM E394-22 ensures consistency, accuracy, and comparability of trace iron analyses in laboratories worldwide. By following this standard, organizations can:
- Enhance product quality with sensitive detection of iron impurities
- Meet global regulatory and customer requirements for trace metal analysis
- Maintain robust internal quality assurance and participate confidently in proficiency testing programs
This standard supports laboratories in achieving high-precision iron determinations, minimizing measurement uncertainty, and strengthening compliance with international best practices.
Keywords: ASTM E394-22, trace iron analysis, 1,10-phenanthroline method, spectrophotometric determination of iron, iron quantification, laboratory quality control, precision measurement, industrial chemical analysis.
Buy Documents
ASTM E394-22 - Standard Test Method for Iron in Trace Quantities Using the 1,10-Phenanthroline Method
REDLINE ASTM E394-22 - Standard Test Method for Iron in Trace Quantities Using the 1,10-Phenanthroline Method
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM E394-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Iron in Trace Quantities Using the 1,10-Phenanthroline Method". This standard covers: SIGNIFICANCE AND USE 4.1 This test method is suitable for determining trace concentrations of iron in a wide variety of products, provided that appropriate sample preparation has rendered the iron and sample matrix soluble in water or other suitable solvent (see 10.1 and Note 4). 4.2 This test method assumes that the amount of color developed is proportional to the amount of iron in the test solution. The calibration curve is linear over the specified range. Possible interferences are described in Section 5. SCOPE 1.1 This test method covers the determination of trace concentrations of iron in the range from 1 to 100 μg/g in a wide variety of products. 1.2 In determining the conformance of the test results using this method to applicable specifications; results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 Consult current OSHA regulations, suppliers’ Safety Data Sheets, and local regulations for all materials used in this specification. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 This test method is suitable for determining trace concentrations of iron in a wide variety of products, provided that appropriate sample preparation has rendered the iron and sample matrix soluble in water or other suitable solvent (see 10.1 and Note 4). 4.2 This test method assumes that the amount of color developed is proportional to the amount of iron in the test solution. The calibration curve is linear over the specified range. Possible interferences are described in Section 5. SCOPE 1.1 This test method covers the determination of trace concentrations of iron in the range from 1 to 100 μg/g in a wide variety of products. 1.2 In determining the conformance of the test results using this method to applicable specifications; results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 Consult current OSHA regulations, suppliers’ Safety Data Sheets, and local regulations for all materials used in this specification. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E394-22 is classified under the following ICS (International Classification for Standards) categories: 71.040.40 - Chemical analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E394-22 has the following relationships with other standards: It is inter standard links to ASTM D6809-02(2016), ASTM E275-08, ASTM E29-08, ASTM D6809-02(2007), ASTM E29-06b, ASTM E29-06a, ASTM E29-06, ASTM D1193-06, ASTM E29-04, ASTM E60-98(2004), ASTM D6809-02, ASTM E29-02e1, ASTM E275-01, ASTM E275-93, ASTM E29-93a(1999). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E394-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E394 − 22
Standard Test Method for
Iron in Trace Quantities Using the 1,10-Phenanthroline
Method
This standard is issued under the fixed designation E394; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* E180 Practice for Determining the Precision of ASTM
Methods for Analysis and Testing of Industrial and Spe-
1.1 This test method covers the determination of trace
cialty Chemicals (Withdrawn 2009)
concentrationsofironintherangefrom1to100µg/ginawide
E275 Practice for Describing and Measuring Performance of
variety of products.
Ultraviolet and Visible Spectrophotometers
1.2 In determining the conformance of the test results using
E300 Practice for Sampling Industrial Chemicals
this method to applicable specifications; results shall be
rounded off in accordance with the rounding-off method of
3. Summary of Test Method
Practice E29.
3.1 This test method is based upon a photometric determi-
1.3 The values stated in SI units are to be regarded as
nation of the 1,10-phenanthroline complex with the iron(II)
standard. No other units of measurement are included in this
ion. The sample is dissolved in a suitable solvent and the iron
standard.
is reduced to the divalent state by the addition of hydroxylam-
ine hydrochloride.The color is then developed, by the addition
1.4 Consult current OSHA regulations, suppliers’ Safety
of 1,10-phenanthroline. After a short reaction period, the
Data Sheets, and local regulations for all materials used in this
absorbance of the solution is measured at approximately
specification.
510 nm using a suitable photometer. The absorbance of the
1.5 This international standard was developed in accor-
solution, once the color is developed, is stable for at least
dance with internationally recognized principles on standard-
several months.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom-
4. Significance and Use
mendations issued by the World Trade Organization Technical
4.1 This test method is suitable for determining trace
Barriers to Trade (TBT) Committee.
concentrations of iron in a wide variety of products, provided
that appropriate sample preparation has rendered the iron and
2. Referenced Documents
sample matrix soluble in water or other suitable solvent (see
2.1 ASTM Standards:
10.1 and Note 4).
D1193 Specification for Reagent Water
4.2 This test method assumes that the amount of color
D6809 Guide for Quality Control and Quality Assurance
developed is proportional to the amount of iron in the test
Procedures for Aromatic Hydrocarbons and Related Ma-
solution. The calibration curve is linear over the specified
terials
range. Possible interferences are described in Section 5.
E29 Practice for Using Significant Digits in Test Data to
Determine Conformance with Specifications
5. Interferences
E60 Practice for Analysis of Metals, Ores, and Related
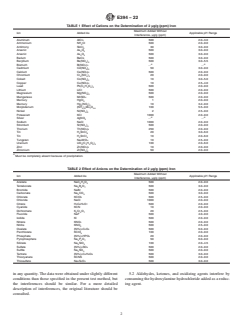
5.1 Fortune and Mellon have made a comprehensive study
Materials by Spectrophotometry
of the interferences of various inorganic ions in this determi-
nation. Table 1 and Table 2, taken from their report, show the
This test method is under the jurisdiction of ASTM Committee D16 on effects of various cations and anions on the determination of
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi-
2.0 µg/g (ppm) iron. If the maximum level of 500 µg/g (ppm)
bility of Subcommittee D16.04 on Instrumental Analysis.
does not interfere, it is very likely that the ion will not interfere
Current edition approved Sept. 1, 2022. Published September 2022. Originally
approved in 1970. Last previous edition approved in 2015 as E394 – 15. DOI:
10.1520/E0394-22.
2 3
For referenced ASTM standards, visit the ASTM website, www.astm.org, or The last approved version of this historical standard is referenced on
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM www.astm.org.
Standards volume information, refer to the standard’s Document Summary page on Fortune, W. B., and Mellon, M. G., Industrial and Engineering Chemistry,
the ASTM website. Analytical Edition, IENAA Vol 10, 1938, pp. 60–64.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E394 − 22
TABLE 1 Effect of Cations on the Determination of 2 µg/g (ppm) Iron
Maximum Added Without
Ion Added As Applicable pH Range
Interference, µg/g (ppm)
Aluminum AlCl 500 2.0–3.0
Ammonium NH Cl 500 2.0–9.0
Antimony SbCl 30 3.0–9.0
Arsenic As O 500 3.0–9.0
2 5
Arsenic As O 500 3.0–9.0
2 3
Barium BaCl 500 3.0–9.0
Beryllium Be(NO ) 500 3.0–5.5
3 2
A A
Bismuth Bi(NO ) . .
3 3
Cadmium Cd(NO ) 50 3.0–9.0
3 2
Calcium Ca(NO ) 500 2.0–9.0
3 2
Chromium Cr (SO ) 20 2.0–9.0
2 4 3
Cobalt Co(NO ) 10 3.0–5.0
3 2
Copper Cu(NO ) 10 2.5–4.0
3 2
Lead Pb(C H O ) 500 2.0–9.0
2 3 2 2
Lithium LiCl 500 2.0–9.0
Magnesium Mg(NO ) 500 2.0–9.0
3 2
Manganese MnSO 500 2.0–9.0
Mercury HgCl 1 2.0–9.0
Mercury Hg (NO ) 10 3.2–9.0
2 3 2
Molybdenum (NH ) Mo O 100 5.5–9.0
4 6 7 24
Nickel Ni(NO ) 2 2.5–9.0
3 2
Potassium KCl 1000 2.0–9.0
A A
Silver AgNO . .
Sodium NaCl 1000 2.0–9.0
Strontium Sr(NO ) 500 2.0–9.0
3 2
Thorium Th(NO ) 250 2.0–9.0
3 4
Tin H SnCl 20 3.0–6.0
2 6
Tin H SnCl 10 2.0–6.0
2 4
Tungsten Na WO 10 2.5–9.0
2 4
Uranium UO (C H O ) 100 2.0–6.0
2 2 3 2 2
Zinc Zn(NO ) 10 2.0–9.0
3 2
Zirconium Zr(NO ) 50 2.0–9.0
3 4
A
Must be completely absent because of precipitation.
TABLE 2 Effect of Anions on the Determination of 2 µg/g (ppm) Iron
Maximum Added Without
Ion Added As Applicable pH Range
Interference, µg/g (ppm)
Acetate NaC H O 500 2.0–9.0
2 3 2
Tetraborate Na B O 500 3.0–9.0
2 4 7
Bromide NaBr 500 2.0–9.0
Carbonate Na CO 500 3.0–9.0
2 3
Chlorate KClO 500 2.5–9.0
Chloride NaCl 1000 2.0–9.0
Citrate H C H O 500 2.0–9.0
3 6 5 7
Cyanide KCN 10 2.0–9.0
Dichromate K Cr O 20 2.5–9.0
2 2 7
Fluoride NaF 500 4.0–9.0
Iodide KI 500 2.0–9.0
Nitrate KNO 500 2.0–9.0
Nitrite KNO 500 2.5–9.0
Oxalate (NH ) C O 500 6.0–9.0
4 2 2 4
Perchlorate KClO 100 2.0–9.0
Phosphate (NH ) HPO 20 2.0–9.0
4 2 4
Pyrophosphate Na P O 50 6.0–9.0
4 2 7
Silicate Na SiO 100 2.0–4.5
2 3
Sulfate (NH ) SO 500 2.0–9.0
4 2 4
Sulfite Na SO 500 2.0–9.0
2 3
Tartrate (NH ) C H O 500 3.0–9.0
4 2 4 9 6
Thiocyanate KCNS 500 2.0–9.0
Thiosulfate Na S O 500 3.0–9.0
2 2 3
in any quantity.The data were obtained under slightly different 5.2 Aldehydes, ketones, and oxidizing agents interfere by
conditions than those specified in the present test method, but
consuming the hydroxylamine hydrochloride added as a reduc-
the interferences should be similar. For a more detailed
ing agent.
description of interferences, the original literature should be
consulted.
E394 − 22
6. Apparatus 7.7 Sulfuric Acid, ACS trace metals grade.
6.1 Photometer, capable of measuring light absorption at
8. Sampling
510 nm and holding a 5-cm or 1-cm cell. Check the perfor-
8.1 Collect the sample in accordance with Practice E300.
mance of the photometer at regular intervals according to the
guidelines given in Practice E275 and the manufacturer’s
8.2 Since this procedure is intended for trace levels of iron,
manual.
care must be taken to ensure that the sample container and
sampling point do not contribute rust (iron) to the sample.
NOTE 1—If a filter photometer is used, a narrow band filter having its
maximum transmission at 480 to 520 nm should be used.Adiscussion of
9. Calibration
photometers and photometric practice is given in Practice E60.
9.1 By means of suitable pipets or a buret, transfer 0
6.2 Absorption Cells, 5-cm or 1-cm light path.
(reagent blank), 2, 4, 6, 8, and 10 mL, respectively, of the
7. Reagents and Materials standard iron solution to each of six 100-mL, glass-stoppered
volumetric flasks. These flasks contain 0, 20, 40, 60, 80, and
7.1 Purity of Reagents—Unless otherwise indicated, it is
100µgofiron,respectively.Dilutethecontentsofeachflaskto
intended that all reagents shall conform to the specifications of
80 mL with water. Develop the color and measure the
theCommitteeonAnalyticalReagentsoftheAmericanChemi-
5 absorbance of each calibration standard as described in 10.3
cal Society, where such specifications are available. Other
and 10.4.
grades may be used, provided it is first ascertained that the
reagent is of sufficiently high purity to permit its use without 9.2 Plot the results in an X-Ygraph, with the micrograms of
irononthex-axisandtherespectiveabsorbancesonthey-axis.
lessening the accuracy of the determination.
Visually evaluate the calibration graph obtained for linearity
7.2 Purity of Water—Unless otherwise indicated, references
and for the absence of obvious outlying values. If so, proceed
to water shall be understood to meanType I or II reagent water
to the next step. If not, investigate for an assignable cause.
as defined in Specification D1193.
9.2.1 Establish a linear regression function from the cali-
7.3 Hydroxylamine Hydrochloride Solution (100 g/L)—
bration data using the statistical method of least squares, for
Dissolve 10 g of hydroxylamine hydrochloride (HONH · HCl)
example,withtheaidofaspreadsheet.Theformulaforalinear
in approximately 60 mL of water, filter, and dilute to 100 mL.
calibration function is:
7.4 Iron, Standard Solution (1 mL = 0.01 mg Fe) may be
y 5 a1bx (1)
prepared from one of the following options:
where:
7.4.1 Option 1—Dissolve 0.1000 g of iron wire in 10 mLof
b = slope of calibration line, and
hydrochloric acid (HCl, 1 + 1) and 1 mL of bromine water
a = intercept.
(which is bromine saturated water at 20 °C). Boil until the
excess bromine is removed. Add 200 mL of HCl, cool, and
9.2.2 Evaluate the linearity of the calibration function by
dilute to 1 L in a volumetric flask. Dilute 100 mL of this
calculating the correlation coefficient r. A typical proper value
solution to 1 L.
is r ≥ 60.9900.
7.4.2 Option 2—Weigh 0.70 6 0.01 g of iron (II) ammo-
NOTE 2—If the photometer readings are percent transmittance, they
nium sulfate hexahydrate (FeSO ·(NH ) SO ·6H O, minimum
4 4 2 4 2
may be converted to absorbance as follows:
purity, 99.5 %) in 500 mL of water containing 20 mL of
sulfuricacid(H SO ,spgr1.84)anddilutingto1Lwithwater.
2 4
A 5 log (2)
S D
T
Dilute 100 mL of this solution to 1 L.
7.4.3 Option 3—Purchased certified iron standard aqueous
where:
solution may also be utilized if its
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: E394 − 15 E394 − 22
Standard Test Method for
Iron in Trace Quantities Using the 1,10-Phenanthroline
Method
This standard is issued under the fixed designation E394; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of trace concentrations of iron in the range from 1 to 100 μg.μg/g in a wide variety
of products.
1.2 This test method is intended to be general for the final steps in the determination of iron and does not include procedures for
sample preparation.
1.2 This test method is applicable to samples whose solutions have a pH less than 2. It is assumed that the pH is adjusted toIn
determining the conformance of the test results using this method to applicable specifications; results shall be rounded off in
accordance with the rounding-off method of Practice E29within this range in the sample preparation.
1.4 Review the current Safety Data Sheets (SDS) for detailed information concerning toxicity, first-aid procedures, handling, and
safety precautions.
1.3 The values givenstated in SI units are the standard. Values in parentheses are for information only.to be regarded as standard.
No other units of measurement are included in this standard.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.4 Consult current OSHA regulations, suppliers’ Safety Data Sheets, and local regulations for all materials used in this
specification.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsibility of
Subcommittee D16.04 on Instrumental Analysis.
Current edition approved Nov. 1, 2015Sept. 1, 2022. Published January 2016September 2022. Originally approved in 1970. Last previous edition approved in 20092015
as E394 – 09.E394 – 15. DOI: 10.1520/E0394-15.10.1520/E0394-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E394 − 22
D1193 Specification for Reagent Water
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E60 Practice for Analysis of Metals, Ores, and Related Materials by Spectrophotometry
E180 Practice for Determining the Precision of ASTM Methods for Analysis and Testing of Industrial and Specialty Chemicals
(Withdrawn 2009)
E200 Practice for Preparation, Standardization, and Storage of Standard and Reagent Solutions for Chemical Analysis
E275 Practice for Describing and Measuring Performance of Ultraviolet and Visible Spectrophotometers
E300 Practice for Sampling Industrial Chemicals
3. Summary of Test Method
3.1 This test method is based upon a photometric determination of the 1,10-phenanthroline complex with the iron(II) ion. The
sample is dissolved in a suitable solvent and the iron is reduced to the divalent state by the addition of hydroxylamine
hydrochloride. The color is then developed, by the addition of 1,10-phenanthroline. After a short reaction period, the absorbance
of the solution is measured at approximately 510 nm 510 nm using a suitable photometer. The absorbance of the solution, once
the color is developed, is stable for at least several months.
4. Significance and Use
4.1 This test method is suitable for determining trace concentrations of iron in a wide variety of products, provided that appropriate
sample preparation has rendered the iron and sample matrix soluble in water or other suitable solvent (see 10.1 and Note 54).
4.2 This test method assumes that the amount of color developed is proportional to the amount of iron in the test solution. The
calibration curve is linear over the specified range. Possible interferences are described in Section 5.
5. Interferences
5.1 Fortune and Mellon have made a comprehensive study of the interferences of various inorganic ions in this determination.
Table 1 and Table 2, taken from their report, show the effects of various cations and anions on the determination of 2.0 μg/g (ppm)
iron. If the maximum level of 500 μg/g (ppm) does not interfere, it is very likely that the ion will not interfere in any quantity.
The data were obtained under slightly different conditions than those specified in the present test method, but the interferences
should be similar. For a more detailed description of interferences, the original literature should be consulted.
5.2 Aldehydes, ketones, and oxidizing agents interfere by consuming the hydroxylamine hydrochloride added as a reducing agent.
6. Apparatus
6.1 Photometer, capable of measuring light absorption at 510 nm and holding a 5-cm or 1-cm cell. Check the performance of the
photometer at regular intervals according to the guidelines given in Practice E275 and the manufacturer’s manual.
NOTE 1—If a filter photometer is used, a narrow band filter having its maximum transmission at 480 to 520 nm should be used. A discussion of
photometers and photometric practice is given in Practice E60.
6.2 Absorption Cells, 5-cm or 1-cm light path.
7. Reagents and Materials
7.1 Purity of Reagents—Unless otherwise indicated, it is intended that all reagents shall conform to the specifications of the
Committee on Analytical Reagents of the American Chemical Society, where such specifications are available. Other grades may
be used, provided it is first ascertained that the reagent is of sufficiently high purity to permit its use without lessening the accuracy
of the determination.
The last approved version of this historical standard is referenced on www.astm.org.
Fortune, W. B., and Mellon, M. G., Industrial and Engineering Chemistry, Analytical Edition, IENAA Vol 10, 1938, pp. 60–64.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society, see Analar Standards for
Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial Convention, Inc. (USP),(USPC),
Rockville, MD.
E394 − 22
TABLE 1 Effect of Cations on the Determination of 2 μg/g (ppm) Iron
Maximum Added Without
Ion Added As Applicable pH Range
Interference, μg/g (ppm)
Aluminum AlCl 500 2.0–3.0
Ammonium NH Cl 500 2.0–9.0
Antimony SbCl 30 3.0–9.0
Arsenic As O 500 3.0–9.0
2 5
Arsenic As O 500 3.0–9.0
2 3
Barium BaCl 500 3.0–9.0
Beryllium Be(NO ) 500 3.0–5.5
3 2
A A
Bismuth Bi(NO ) . .
3 3
Cadmium Cd(NO ) 50 3.0–9.0
3 2
Calcium Ca(NO ) 500 2.0–9.0
3 2
Chromium Cr (SO ) 20 2.0–9.0
2 4 3
Cobalt Co(NO ) 10 3.0–5.0
3 2
Copper Cu(NO ) 10 2.5–4.0
3 2
Lead Pb(C H O ) 500 2.0–9.0
2 3 2 2
Lithium LiCl 500 2.0–9.0
Magnesium Mg(NO ) 500 2.0–9.0
3 2
Manganese MnSO 500 2.0–9.0
Mercury HgCl 1 2.0–9.0
Mercury Hg (NO ) 10 3.2–9.0
2 3 2
Molybdenum (NH ) Mo O 100 5.5–9.0
4 6 7 24
Nickel Ni(NO ) 2 2.5–9.0
3 2
Potassium KCl 1000 2.0–9.0
A A
Silver AgNO . .
Sodium NaCl 1000 2.0–9.0
Strontium Sr(NO ) 500 2.0–9.0
3 2
Thorium Th(NO ) 250 2.0–9.0
3 4
Tin H SnCl 20 3.0–6.0
2 6
Tin H SnCl 10 2.0–6.0
2 4
Tungsten Na WO 10 2.5–9.0
2 4
Uranium UO (C H O ) 100 2.0–6.0
2 2 3 2 2
Zinc Zn(NO ) 10 2.0–9.0
3 2
Zirconium Zr(NO ) 50 2.0–9.0
3 4
A
Must be completely absent because of precipitation.
TABLE 2 Effect of Anions on the Determination of 2 μg/g (ppm) Iron
Maximum Added Without
Ion Added As Applicable pH Range
Interference, μg/g (ppm)
Acetate NaC H O 500 2.0–9.0
2 3 2
Tetraborate Na B O 500 3.0–9.0
2 4 7
Bromide NaBr 500 2.0–9.0
Carbonate Na CO 500 3.0–9.0
2 3
Chlorate KClO 500 2.5–9.0
Chloride NaCl 1000 2.0–9.0
Citrate H C H O 500 2.0–9.0
3 6 5 7
Cyanide KCN 10 2.0–9.0
Dichromate K Cr O 20 2.5–9.0
2 2 7
Fluoride NaF 500 4.0–9.0
Iodide KI 500 2.0–9.0
Nitrate KNO 500 2.0–9.0
Nitrite KNO 500 2.5–9.0
Oxalate (NH ) C O 500 6.0–9.0
4 2 2 4
Perchlorate KClO 100 2.0–9.0
Phosphate (NH ) HPO 20 2.0–9.0
4 2 4
Pyrophosphate Na P O 50 6.0–9.0
4 2 7
Silicate Na SiO 100 2.0–4.5
2 3
Sulfate (NH ) SO 500 2.0–9.0
4 2 4
Sulfite Na SO 500 2.0–9.0
2 3
Tartrate (NH ) C H O 500 3.0–9.0
4 2 4 9 6
Thiocyanate KCNS 500 2.0–9.0
Thiosulfate Na S O 500 3.0–9.0
2 2 3
7.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean Type I or II reagent water as
defined in Specification D1193.
7.3 Hydroxylamine Hydrochloride Solution (100 g/L)—Dissolve 10 g of hydroxylamine hydrochloride (HONH · HCl) in
approximately 60 mL of water, filter, and dilute to 100 mL.
E394 − 22
7.4 Iron, Standard Solution (1 mL = 0.01 mg Fe) (Note 2)—Dissolve 0.1000 g of iron wire in 10 mL of hydrochloric acid (HCl,
1 + 1) and 1 mL of bromine water. Boil until the excess bromine is removed. Add 200 mL of HCl, cool, and dilute to 1 L in a
volumetric flask. Dilute 100 mL of this solution to 1 L.may be prepared from one of the following options:
NOTE 2—As an alternative, the standard iron solution may be prepared by weighing exactly 0.7022 g of iron (II) ammonium sulfate hexahydrate
(FeSO ·(NH ) SO ·6H O, minimum purity, 99.5 %) in 500 mL of water containing 20 mL of sulfuric acid (H So , sp gr 1.84) and diluting to 1 L with
4 4 2 4 2 2 4
water. Dilute 100 mL of this solution to 1 L.
7.4.1 Option 1—Dissolve 0.1000 g of iron wire in 10 mL of hydrochloric acid (HCl, 1 + 1) and 1 mL of bromine water (which
is bromine saturated water at 20 °C). Boil until the excess bromine is removed. Add 200 mL of HCl, cool, and dilute to 1 L in
a volumetric flask. Dilute 100 mL of this solution to 1 L.
7.4.2 Option 2—Weigh 0.70 6 0.01 g of iron (II) ammonium sulfate hexahydrate (FeSO ·(NH ) SO ·6H O, minimum purity,
4 4 2 4 2
99.5 %) in 500 mL of water containing 20 mL of sulfuric acid (H SO , sp gr 1.84) and diluting to 1 L with water. Dilute 100 mL
2 4
of this solution to 1 L.
7.4.3 Option 3—Purchased certified iron standard aqueous solution may also be utilized if its pH is less than 2.
7.5 1,10-Phenanthroline Solution (3 g/L)—Dissolve 0.9 g of 1,10-phenanthroline monohydrate in 30 mL of methanol and dilute
,7
to 300 mL with water.
7.6 Ammonium Acetate—Acetic Acid Solution—Dissolve 100 g of ammonium acetate (CH COONH ) in about 600 mL of water,
3 4
filter, add 200 mL of glacial acetic acid to the filtrate, and dilute to 1 L with water.
7.7 Sulfuric Acid, ACS trace metals grade.
8. Sampling
8.1 Collect the sample in accordance with Practice E300.
8.2 BecauseSince this is a general test method for the final steps in determining iron, specific procedures for sample preparation
are not included (seeprocedure is intended for trace levels of iron, care must be taken to ensure that the sample container and
sampling point do not contribute rust 1.3,(iron) 4
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...