ASTM D8440-22
(Specification)Standard Specification for Food Safety and Quality of Hempseed Products Intended for Human Consumption
Standard Specification for Food Safety and Quality of Hempseed Products Intended for Human Consumption
SCOPE
1.1 In this specification, the maximum allowable e.coli, salmonella spp, yeast and mold, foreign material, and total delta-9 tetrahydrocannabinol (THC) content is established for major hempseed products intended for human consumption. It also establishes acceptable ranges for moisture, peroxide value, and free fatty acid content, which help maintain product shelf life.
1.2 This specification establishes specifications for hempseed protein products to facilitate commerce, but is not a specification for consumer-facing labels, which are determined by authorities having jurisdiction and individual companies. It does not include devitalized hempseed.
1.3 This specification applies to hemp food processors, food companies, wholesalers, regulatory bodies, and analytical laboratories.
1.4 In this specification, the minimum information required in a Certificate of Analysis is described.
1.5 Where aspects of this specification differ from regional regulatory jurisdictional requirements, the jurisdictional authority’s directive shall take precedence.
1.6 Units—The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard.
1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2022
- Technical Committee
- D37 - Cannabis
- Drafting Committee
- D37.07 - Industrial Hemp
Overview
ASTM D8440-22: Standard Specification for Food Safety and Quality of Hempseed Products Intended for Human Consumption establishes internationally recognized criteria for the safety and quality of hempseed products. Developed by ASTM International, this standard outlines maximum allowable limits for contaminants, including E. coli, Salmonella spp, yeast and mold, foreign material, and total delta-9 tetrahydrocannabinol (THC) content. It also provides guidelines for moisture, peroxide value, and free fatty acid content to support product shelf life. The specification is designed for hemp food processors, producers, regulators, wholesalers, and analytical laboratories involved in the manufacture and trade of edible hempseed products such as dehulled hempseed, toasted hempseed, hemp seed oil, and hempseed protein products.
Key Topics
- Microbiological Safety: Defines maximum permissible levels for E. coli, Salmonella spp, yeast, mold, total coliforms, Enterobacteriaceae, and standard plate counts for various hempseed products to mitigate food safety risks.
- Chemical Quality Parameters: Specifies allowable limits for peroxide value, free fatty acids, and moisture content to prevent spoilage and maintain shelf life.
- Physical Contaminants: Establishes acceptable thresholds for foreign materials (such as hemp shells and extraneous matter) through visual assessment, ensuring product integrity.
- Delta-9 THC Content: Sets a strict upper limit for total delta-9 THC (less than 20 ppm) to comply with regulatory requirements and consumer safety.
- Certificate of Analysis: Describes the minimum data required to verify compliance, essential for business-to-business transactions and regulatory reporting.
- Jurisdictional Compliance: Clarifies that regional regulatory directives take precedence if there are differences with this ASTM standard.
Applications
This standard serves multiple practical purposes throughout the hemp food supply chain:
- Quality Control for Producers: Offers hemp food manufacturers and processors a comprehensive framework to monitor safety and quality, ensuring that products meet industry and international expectations before market release.
- Facilitating Trade: By providing common metrics and definitions, ASTM D8440-22 promotes efficient business-to-business transactions and marketability of hempseed foods in global commerce.
- Regulatory Alignment: Regulatory authorities can use this specification to assess compliance, support public health, and harmonize cross-border standards.
- Analytical Laboratories: Lists recognized analytical methods for testing microbial and chemical contaminants, supporting consistent laboratory practices.
- Product Repurposing: Identifies parameters for recalling or repurposing lots that do not meet food-grade criteria, such as for animal feed or further processing, minimizing waste.
- Risk Management: Assists industry stakeholders in implementing effective food safety and quality management systems to reduce liability and maintain consumer confidence.
Related Standards
To fully implement ASTM D8440-22, industry participants may reference several related standards and methods, including:
- ASTM D8270: Terminology Relating to Cannabis
- AOAC Official Methods:
- 935.29: Moisture in Malt
- 955.04D: Protein in Vegetable Protein Products
- 2018.11: Quantitation of Cannabinoids
- AOCS Methods:
- Ca 5a-40: Free Fatty Acids in Fats and Oils
- Cd 8b-90: Peroxide Value
- ISO Standards:
- 6579: Detection of Salmonella
- 16649-1/2: Enumeration of E. coli
- 21528-2: Enumeration of Enterobacteriaceae
- MFHPB Methods: Canadian protocols for microbial analysis of foods
- European and International Norms: DGF and Ph. Eur. methods for peroxide value and other quality metrics
By aligning practices with ASTM D8440-22 and its referenced methods, the hemp food industry can assure product safety, legal compliance, and global market access.
Keywords: hempseed food safety, hempseed product quality, delta-9 THC in hemp foods, food-grade hempseed, ASTM D8440-22, hemp seed oil, hemp protein, microbiological contaminants, food safety certification, hemp food standards.
Buy Documents
ASTM D8440-22 - Standard Specification for Food Safety and Quality of Hempseed Products Intended for Human Consumption
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

Bureau Veritas
Bureau Veritas is a world leader in laboratory testing, inspection and certification services.

DNV
DNV is an independent assurance and risk management provider.
Sponsored listings
Frequently Asked Questions
ASTM D8440-22 is a technical specification published by ASTM International. Its full title is "Standard Specification for Food Safety and Quality of Hempseed Products Intended for Human Consumption". This standard covers: SCOPE 1.1 In this specification, the maximum allowable e.coli, salmonella spp, yeast and mold, foreign material, and total delta-9 tetrahydrocannabinol (THC) content is established for major hempseed products intended for human consumption. It also establishes acceptable ranges for moisture, peroxide value, and free fatty acid content, which help maintain product shelf life. 1.2 This specification establishes specifications for hempseed protein products to facilitate commerce, but is not a specification for consumer-facing labels, which are determined by authorities having jurisdiction and individual companies. It does not include devitalized hempseed. 1.3 This specification applies to hemp food processors, food companies, wholesalers, regulatory bodies, and analytical laboratories. 1.4 In this specification, the minimum information required in a Certificate of Analysis is described. 1.5 Where aspects of this specification differ from regional regulatory jurisdictional requirements, the jurisdictional authority’s directive shall take precedence. 1.6 Units—The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SCOPE 1.1 In this specification, the maximum allowable e.coli, salmonella spp, yeast and mold, foreign material, and total delta-9 tetrahydrocannabinol (THC) content is established for major hempseed products intended for human consumption. It also establishes acceptable ranges for moisture, peroxide value, and free fatty acid content, which help maintain product shelf life. 1.2 This specification establishes specifications for hempseed protein products to facilitate commerce, but is not a specification for consumer-facing labels, which are determined by authorities having jurisdiction and individual companies. It does not include devitalized hempseed. 1.3 This specification applies to hemp food processors, food companies, wholesalers, regulatory bodies, and analytical laboratories. 1.4 In this specification, the minimum information required in a Certificate of Analysis is described. 1.5 Where aspects of this specification differ from regional regulatory jurisdictional requirements, the jurisdictional authority’s directive shall take precedence. 1.6 Units—The values stated in SI units are to be regarded as the standard. No other units of measurement are included in this standard. 1.7 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.8 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D8440-22 is classified under the following ICS (International Classification for Standards) categories: 67.040 - Food products in general; 67.050 - General methods of tests and analysis for food products. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D8440-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation:D8440 −22
Standard Specification for
Food Safety and Quality of Hempseed Products Intended for
Human Consumption
This standard is issued under the fixed designation D8440; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
INTRODUCTION
The processing of hempseed results in several food products such as toasted hempseed, dehulled
hempseed, hemp seed oil, and hempseed protein products. These products in which hempseed is the
main or sole ingredient may be consumed with no microbial pathogen reduction step to manage food
safety risk. Specifications relating to food safety and shelf-life attributes are defined in this
specification to protect buyers and consumers from food safety risk, and where possible, address shelf
life as it relates to potential spoilage. Meeting these specifications will assist product consistency and
business-to-business (B2B) transactions.
1. Scope 1.7 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 In this specification, the maximum allowable e.coli,
responsibility of the user of this standard to establish appro-
salmonella spp, yeast and mold, foreign material, and total
priate safety, health, and environmental practices and deter-
delta-9 tetrahydrocannabinol (THC) content is established for
mine the applicability of regulatory limitations prior to use.
major hempseed products intended for human consumption. It
1.8 This international standard was developed in accor-
alsoestablishesacceptablerangesformoisture,peroxidevalue,
dance with internationally recognized principles on standard-
and free fatty acid content, which help maintain product shelf
ization established in the Decision on Principles for the
life.
Development of International Standards, Guides and Recom-
1.2 This specification establishes specifications for hemp-
mendations issued by the World Trade Organization Technical
seed protein products to facilitate commerce, but is not a
Barriers to Trade (TBT) Committee.
specification for consumer-facing labels, which are determined
2. Referenced Documents
by authorities having jurisdiction and individual companies. It
does not include devitalized hempseed.
2.1 ASTM Standards:
D8270 Terminology Relating to Cannabis
1.3 This specification applies to hemp food processors, food
2.2 AOAC International Standards:
companies,wholesalers,regulatorybodies,andanalyticallabo-
AOAC 935.29 Moisture in Malt
ratories.
AOAC 955.04D Protein in Vegetable Protein Products
1.4 In this specification, the minimum information required
AOAC 2018.11 Quantitation of Cannabinoids in Cannabis
in a Certificate of Analysis is described.
Dried Plant Materials, Concentrates and Oils
1.5 Where aspects of this specification differ from regional
2.3 AOCS Standards:
regulatory jurisdictional requirements, the jurisdictional au- AOCS Ca 5a-40 Free FattyAcids in Crude and Refined Fats
thority’s directive shall take precedence.
and Oils
AOCS Cd 8b-90 Peroxide Value, Acetic Acid, Isooctane
1.6 Units—The values stated in SI units are to be regarded
Method
as the standard. No other units of measurement are included in
this standard.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on
This specification is under the jurisdiction of ASTM Committee D37 on the ASTM website.
Cannabis and is the direct responsibility of Subcommittee D37.07 on Industrial Available from AOAC International (AOAC), Official Methods of Analysis,
Hemp. 2275 Research Blvd, Ste 300, Rockville, MD 20850, http://www.aoac.org.
Current edition approved April 1, 2022. Published May 2022. DOI: 10.1520/ Available from American Oil Chemists’ Society (AOCS), 2710 S. Boulder,
D8440-22. Urbana, IL 61802-6996, http://www.aocs.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D8440−22
2.4 DGHM Standard (German Society for Hygiene and 3.2.4 toasted hempseed, n—hempseed that has been dry
Microbiology): roasted to modify its taste and texture.
DGF C-V 6a Peroxide Value Method according to Wheeler
3.2.4.1 Discussion—This seed is not similar to devitalized
2.5 ISO Standards:
hempseed.
ISO 6579 Horizontal Methods for the Detection, Enumera-
tion and Serotyping of Salmonella
4. General Requirements
ISO 16649-1 Horizontal method for the enumeration of
4.1 Consistent specifications for hempseed products using a
beta-glucuronidase-positive Escherichia coli, Part 1
combination of attributes representing both the presence and
ISO 16649-2 Horizontal method for the enumeration of
absence of certain characteristics will provide a credible means
beta-glucuronidase-positive Escherichia coli, Part 2
for buyers and sellers to target product for appropriate use(s).
ISO 21528-2 Horizontal method for the enumeration of
Enterobacteriaceae, Part 2 4.2 Hemp food processors and laboratories can verify mini-
2.6 MFHPB Standards: mum food safety and quality aspects for hempseed products
intended for human consumption.
MFHPB-20 Methods for the Isolation and Identification of
Salmonellafrom Foods and Environmental Samples
4.3 Hempseed products that may be discarded on visual
MFHPB-22 Enumeration of Yeasts and Moulds in Food
review or in the absence of clearly defined quality metrics can
MFHPB-33 Enumeration of Total Aerobic Bacteria in Food
be used by processors or redirected to other uses.
Products and Food Ingredients Using 3M™ Petrifilm™
4.4 Hempseed products that are assessed as unacceptable
Aerobic Count Plates
for human use may be repurposed for other byproducts or
MFHPB-34 Enumeration of Escherichia coli and Coliforms
further processing.
in Food Products and Food Ingredients Using 3M™
Petrifilm™ E. coli Count Plates
4.5 This specification can be used within buyer-seller trans-
2.7 Ph.Eur. Standard:
actions or for marketing or quality control purposes.
Ph.Eur. 9.0, 2.5.5, Peroxide Value
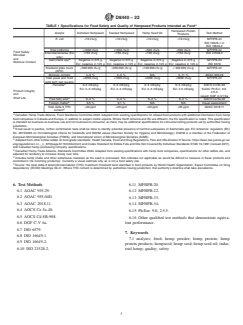
5. Specification for Food Safety and Quality
3. Terminology
5.1 Specifications in Table 1 indicate: (1) the minimum
3.1 Definitions—See Terminology D8270.
number of tests/analytes that shall be represented on a certifi-
3.2 Definitions of Terms Specific to This Standard:
cate of analysis and (2) the groupings of tests that shall
3.2.1 devitalized hempseed, n—sterilized intact achene
determine if a lot/batch of hempseed product is acceptable for
(fruit) produced from a hemp plant that is incapable of
human consumption or repurposed.
germination.
5.2 Maximum allowable amounts of key microbials and
3.2.1.1 Discussion—This is whole seed that has been ster-
acceptable moisture range levels are identified in Table 1.If
ilizedthroughhighheatorothermethodsasperrequirementby
any one result is out of specification, the lot is no longer a
countries that do not allow importation of viable hemp seed. It
food-grade product unless otherwise treat
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...