ASTM D6142-21
(Test Method)Standard Test Method for Analysis of Phenol by Capillary Gas Chromatography
Standard Test Method for Analysis of Phenol by Capillary Gas Chromatography
SIGNIFICANCE AND USE
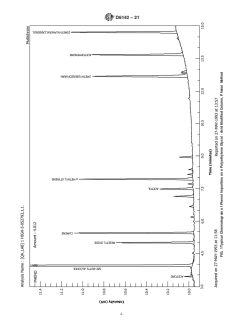
5.1 This test method is suitable for setting specifications on phenol and for use as an internal quality control tool where phenol is produced or is used in a manufacturing process. It may also be used in development or research work involving phenol. It is generally applied to determining those commonly occurring impurities such as mesityl oxide, cumene, hydroxyacetone, acetone, α-methylstyrene, 2-methylbenzofuran, and acetophenone.
5.2 Purity is commonly reported by subtracting the determined expected impurities from 100.00. However, a gas chromatographic analysis cannot determine absolute purity if water is present or unknown components are contained within the material being examined.
SCOPE
1.1 This test method covers the determination of known impurities in phenol by gas chromatography (GC). It is generally meant for the analysis of phenol of 99.9 % or greater purity.
1.2 This test method has been found applicable over impurity concentrations up to 70 mg/kg. Users of this method believe it is linear over a wider range. The limit of detection (LOD) is 1.7 mg/kg and the limit of quantitation (LOQ) is 5.8 mg/kg.
Note 1: LOD is defined in 7.1 as part of the method setup. The values above were calculated based on the ILS data for acetone, acetophenone, α-methylstyrene, and 2-methylbenzofuran in Table 3.
1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2021
- Technical Committee
- D16 - Aromatic, Industrial, Specialty and Related Chemicals
- Drafting Committee
- D16.02 - Oxygenated Aromatics
Relations
- Refers
ASTM D3852-20 - Standard Practice for Sampling and Handling Phenol, Cresols, and Cresylic Acid - Effective Date
- 01-Jun-2020
- Effective Date
- 01-Nov-2016
- Refers
ASTM D3852-16 - Standard Practice for Sampling and Handling Phenol, Cresols, and Cresylic Acid - Effective Date
- 01-Jun-2016
- Effective Date
- 01-Oct-2015
- Effective Date
- 01-Jul-2014
- Effective Date
- 01-May-2013
- Refers
ASTM D3852-07(2012) - Standard Practice for Sampling and Handling Phenol, Cresols, and Cresylic Acid - Effective Date
- 01-Jun-2012
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Feb-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
- Effective Date
- 15-Jul-2007
- Effective Date
- 01-Jun-2007
- Effective Date
- 01-Mar-2007
Overview
ASTM D6142-21 is the Standard Test Method for Analysis of Phenol by Capillary Gas Chromatography, developed by ASTM International. This test method provides a reliable and precise approach to determining the concentrations of known impurities in high-purity phenol (99.9% or greater) using capillary gas chromatography (GC). The standard is widely used in quality control, product specification, and research applications where phenol is manufactured or utilized. It addresses key analytical challenges and sets procedures for achieving accurate results and comparability between laboratories.
Key Topics
- Gas Chromatography for Impurity Analysis: Employs GC with a capillary column and flame ionization detector (FID) to identify and quantify impurities such as mesityl oxide, cumene, hydroxyacetone, acetone, α-methylstyrene, 2-methylbenzofuran, and acetophenone.
- Internal Standard Technique: Utilizes an internal standard (such as sec-butyl alcohol) to enhance quantitation accuracy, ensuring consistent analytical results.
- Sensitivity and Range: Suitable for impurity concentrations from 15 up to 70 mg/kg, with a limit of detection (LOD) of 1.7 mg/kg and a limit of quantitation (LOQ) of 5.8 mg/kg.
- Reporting and Units: Results are to be reported in SI units and rounded in accordance with ASTM Practice E29.
- Quality Control and Interlaboratory Consistency: Emphasizes quality assurance protocols, recommending regular analysis of control samples and participation in interlaboratory studies for result validation.
- Specification and Regulatory Compliance: Assists users in meeting product specifications and complying with safety, health, and environmental regulations.
Applications
The ASTM D6142-21 standard is essential for:
- Manufacturers of Phenol: Used as an internal quality control tool and for setting material specifications, ensuring the product meets high purity requirements before market release or internal use.
- Chemical Processing and Production: Supports the monitoring and control of impurity levels in phenol when employed as a feedstock or intermediate, helping identify contamination sources or process drift.
- Research and Development: Offers a robust method for analyzing phenol in experimental contexts or during the development of new phenol-based products.
- Regulatory Testing and Auditing: Provides documented, reproducible procedures for compliance with industry standards and regulatory audits.
- Analytical Laboratories: Serves as a reference protocol for determining phenol purity and impurity profiles in contract or quality control laboratories.
Related Standards
For comprehensive implementation and greater context, users are encouraged to consult the following related ASTM standards:
- ASTM D3852: Practice for Sampling and Handling Phenol, Cresols, and Cresylic Acid
- ASTM D4307: Practice for Preparation of Liquid Blends for Use as Analytical Standards
- ASTM D4790: Terminology of Aromatic Hydrocarbons and Related Chemicals
- ASTM D6809: Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
- ASTM E29: Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
- ASTM E355: Practice for Gas Chromatography Terms and Relationships
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
- ASTM E1510: Practice for Installing Fused Silica Open Tubular Capillary Columns in Gas Chromatographs
By referencing these standards in conjunction with ASTM D6142-21, laboratories and manufacturers can ensure best practices for the analysis of phenol by gas chromatography and maintain high standards of product purity and reliability in their operations.
Keywords: phenol analysis, gas chromatography, ASTM D6142-21, impurities in phenol, quality control, analytical chemistry, laboratory standards, capillary GC, chemical purity, industrial chemicals
Buy Documents
ASTM D6142-21 - Standard Test Method for Analysis of Phenol by Capillary Gas Chromatography
REDLINE ASTM D6142-21 - Standard Test Method for Analysis of Phenol by Capillary Gas Chromatography
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D6142-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Analysis of Phenol by Capillary Gas Chromatography". This standard covers: SIGNIFICANCE AND USE 5.1 This test method is suitable for setting specifications on phenol and for use as an internal quality control tool where phenol is produced or is used in a manufacturing process. It may also be used in development or research work involving phenol. It is generally applied to determining those commonly occurring impurities such as mesityl oxide, cumene, hydroxyacetone, acetone, α-methylstyrene, 2-methylbenzofuran, and acetophenone. 5.2 Purity is commonly reported by subtracting the determined expected impurities from 100.00. However, a gas chromatographic analysis cannot determine absolute purity if water is present or unknown components are contained within the material being examined. SCOPE 1.1 This test method covers the determination of known impurities in phenol by gas chromatography (GC). It is generally meant for the analysis of phenol of 99.9 % or greater purity. 1.2 This test method has been found applicable over impurity concentrations up to 70 mg/kg. Users of this method believe it is linear over a wider range. The limit of detection (LOD) is 1.7 mg/kg and the limit of quantitation (LOQ) is 5.8 mg/kg. Note 1: LOD is defined in 7.1 as part of the method setup. The values above were calculated based on the ILS data for acetone, acetophenone, α-methylstyrene, and 2-methylbenzofuran in Table 3. 1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 This test method is suitable for setting specifications on phenol and for use as an internal quality control tool where phenol is produced or is used in a manufacturing process. It may also be used in development or research work involving phenol. It is generally applied to determining those commonly occurring impurities such as mesityl oxide, cumene, hydroxyacetone, acetone, α-methylstyrene, 2-methylbenzofuran, and acetophenone. 5.2 Purity is commonly reported by subtracting the determined expected impurities from 100.00. However, a gas chromatographic analysis cannot determine absolute purity if water is present or unknown components are contained within the material being examined. SCOPE 1.1 This test method covers the determination of known impurities in phenol by gas chromatography (GC). It is generally meant for the analysis of phenol of 99.9 % or greater purity. 1.2 This test method has been found applicable over impurity concentrations up to 70 mg/kg. Users of this method believe it is linear over a wider range. The limit of detection (LOD) is 1.7 mg/kg and the limit of quantitation (LOQ) is 5.8 mg/kg. Note 1: LOD is defined in 7.1 as part of the method setup. The values above were calculated based on the ILS data for acetone, acetophenone, α-methylstyrene, and 2-methylbenzofuran in Table 3. 1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off in accordance with the rounding-off method of Practice E29. 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D6142-21 is classified under the following ICS (International Classification for Standards) categories: 71.040.50 - Physicochemical methods of analysis. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D6142-21 has the following relationships with other standards: It is inter standard links to ASTM D3852-20, ASTM D6809-02(2016), ASTM D3852-16, ASTM D4307-99(2015), ASTM D4790-14, ASTM E691-13, ASTM D3852-07(2012), ASTM E691-11, ASTM D4790-11, ASTM D4307-99(2010), ASTM E29-08, ASTM E691-08, ASTM D4790-07, ASTM D6809-02(2007), ASTM E355-96(2007). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D6142-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D6142 − 21
Standard Test Method for
Analysis of Phenol by Capillary Gas Chromatography
This standard is issued under the fixed designation D6142; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope* 2. Referenced Documents
1.1 This test method covers the determination of known 2.1 ASTM Standards:
impurities in phenol by gas chromatography (GC). It is D3852 Practice for Sampling and Handling Phenol, Cresols,
generally meant for the analysis of phenol of 99.9 % or greater and Cresylic Acid
purity. D4307 Practice for Preparation of Liquid Blends for Use as
Analytical Standards
1.2 This test method has been found applicable over impu-
D4790 Terminology ofAromatic Hydrocarbons and Related
rity concentrations up to 70 mg/kg. Users of this method
Chemicals
believe it is linear over a wider range. The limit of detection
D6809 Guide for Quality Control and Quality Assurance
(LOD) is 1.7 mg/kg and the limit of quantitation (LOQ) is
Procedures for Aromatic Hydrocarbons and Related Ma-
5.8 mg⁄kg.
terials
NOTE 1—LOD is defined in 7.1 as part of the method setup. The values
E29 Practice for Using Significant Digits in Test Data to
above were calculated based on the ILS data for acetone, acetophenone,
Determine Conformance with Specifications
α-methylstyrene, and 2-methylbenzofuran in Table 3.
E355 Practice for Gas ChromatographyTerms and Relation-
1.3 In determining the conformance of the test results using
ships
this method to applicable specifications, results shall be
E691 Practice for Conducting an Interlaboratory Study to
rounded off in accordance with the rounding-off method of
Determine the Precision of a Test Method
Practice E29.
E1510 Practice for Installing Fused Silica Open Tubular
1.4 The values stated in SI units are to be regarded as Capillary Columns in Gas Chromatographs
standard. No other units of measurement are included in this 2.2 Other Document:
standard. OSHA Regulations, 29 CFR paragraphs 1910.1000 and
1910.1200
1.5 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
3. Terminology
responsibility of the user of this standard to establish appro-
3.1 See Terminology D4790 for definitions of terms used in
priate safety, health, and environmental practices and deter-
this test method.
mine the applicability of regulatory limitations prior to use.
For specific hazard statements, see Section 9.
4. Summary of Test Method
1.6 This international standard was developed in accor-
dance with internationally recognized principles on standard- 4.1 A known amount of an internal standard is added to a
ization established in the Decision on Principles for the sample of phenol. The prepared sample is mixed and analyzed
Development of International Standards, Guides and Recom- by a gas chromatograph equipped with a flame ionization
mendations issued by the World Trade Organization Technical detector (FID).The peak area of each impurity and the internal
Barriers to Trade (TBT) Committee.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This test method is under the jurisdiction of ASTM Committee D16 on contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Aromatic, Industrial, Specialty and Related Chemicals and is the direct responsi- Standards volume information, refer to the standard’s Document Summary page on
bility of Subcommittee D16.02 on Oxygenated Aromatics. the ASTM website.
Current edition approved Nov. 1, 2021. Published November 2021. Originally AvailablefromU.S.GovernmentPrintingOfficeSuperintendentofDocuments,
approved in 1997. Last previous edition approved in 2016 as D6142 – 16. DOI: 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
10.1520/D6142-21. www.access.gpo.gov.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6142 − 21
standard is measured. The amount of each impurity is calcu- 8. Reagents
lated from the ratio of the peak area of the internal standard
8.1 Purity of Reagent—Reagent grade chemicals shall be
versus the peak area of the impurity. Results are reported in
used in all tests. Unless otherwise indicated, it is intended that
milligrams per kilogram.
all reagents shall conform to the specifications of the Commit-
tee onAnalytical Reagents of theAmerican Chemical Society,
5. Significance and Use
where such specifications are available. Other grades may be
5.1 This test method is suitable for setting specifications on
used, provided it is first ascertained that the reagent is of
phenol and for use as an internal quality control tool where
sufficiently high purity to permit its use without lessening the
phenol is produced or is used in a manufacturing process. It
accuracy of the determination.
may also be used in development or research work involving
8.2 High Purity Phenol (99.99 % or greater purity).
phenol. It is generally applied to determining those commonly
8.3 Carrier Gas, Makeup, and Detector Gases—Helium,
occurring impurities such as mesityl oxide, cumene,
hydroxyacetone, acetone, α-methylstyrene, hydrogen,nitrogen,orothercarrier,makeupanddetectorgases
99.999 % minimum purity. Oxygen in carrier gas less than
2-methylbenzofuran, and acetophenone.
1 ppm, less than 0.5 ppm is preferred. Purify carrier, makeup,
5.2 Purity is commonly reported by subtracting the deter-
anddetectorgasestoremoveoxygen,water,andhydrocarbons.
mined expected impurities from 100.00. However, a gas
Helium was the carrier used for the conditions in Table 1.
chromatographic analysis cannot determine absolute purity if
8.4 Compressed Air—Purify air to remove water and hydro-
water is present or unknown components are contained within
carbons.Air for an FID should contain less than 0.1 ppmTHC.
the material being examined.
8.5 Equipment Setup Check Sample:
6. Interferences
8.5.1 For GC standards, a setup check sample should be
6.1 The internal standard chosen must be sufficiently re-
included to:
solved from any impurity and the phenol peak.
8.5.1.1 Determine retention times for the components mea-
sured in GC standards.
6.2 Anysolventusedmustalsobesufficientlyresolvedfrom
8.5.1.2 Verify there is adequate resolution to measure the
any impurity, the internal standard, and the phenol peak.
components of interest in GC standards.
7. Apparatus
8.5.1.3 Determine that the equipment has the sensitivity
specified in the scope of the standard.
7.1 Gas Chromatograph—Any chromatograph having a
8.5.2 For GC standards and standards that determine trace
flame ionization detector that can be operated at the conditions
levels, the equipment setup check sample should contain a
given in Table 1. Chromatographic data systems are preferred
component with a concentration that is approximately two
but electronic integration may be used if the user can demon-
times the LOD stated in the scope of the standard. When the
strate that the results are consistent with the precision state-
equipmentsetupchecksampleisanalyzed,anacceptableresult
ment.
for the trace component is 650 % of the expected concentra-
7.2 Columns—The choice of column is based on resolution
tion.
requirements. Any column may be used that is capable of
8.5.2.1 ForGCstandardswheretheprimarymaterialcannot
resolving all significant impurities from the major component.
be purified so that no impurities are detected, the following is
The column and conditions described in Table 1 have been
suggested:
used successfully and shall be used as referee in cases of
(1) Add an impurity that is not present in the primary
dispute.
material. Determine that the impurity has the following prop-
erties:
(a) The impurity is essentially inert and unreactive in the
TABLE 1 Recommended Operating Conditions
primary material.
Column: To use for identification of all components
Tubing fused silica (b) The retention time is sufficiently separated from other
Stationary phase polyethylene glycol-acid modified
impurities so that there will be no mistake in identification.
Solid support crosslinked
(c) The impurity is completely vaporized in the injection
Film thickness, µ 0.5
Length, m 50
port.
Inside diameter, mm 0.32
(d) The impurity is well behaved on the column, that is,
Carrier Gas Helium. Nitrogen or hydrogen are permitted. Nitrogen
no fronting or tailing.
and hydrogen carrier gas require different conditions.
The user must conduct the necessary evaluation to
(e) The response factor is known and not significantly
determine that equivalent results are obtained.
different from the components of interest.
Flow rate mL/min 1.3
Temperature, °C
Injector 180
Detector 230
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Oven
Standard-Grade Reference Materials, American Chemical Society, Washington,
Initial, °C 110 for 6 min
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
Rate, °C 12 per min
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
Final, °C 210 for 90 min
Internal Standard sec-butyl alcohol U.K., and the Unit
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D6142 − 16 D6142 − 21
Standard Test Method for
Analysis of Phenol by Capillary Gas Chromatography
This standard is issued under the fixed designation D6142; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope*
1.1 This test method covers the determination of known impurities in phenol by gas chromatography (GC). It is generally meant
for the analysis of phenol of 99.9 % or greater purity.
1.2 This test method has been found applicable over impurity concentrations of 15 up to 70 mg/kg. Users of this method believe
it is linear over a wider range. The limit of detection (LOD) is 1.7 mg/kg and the limit of quantitation (LOQ) is 5.8 mg ⁄kg.
NOTE 1—LOD is defined in 7.1 as part of the method setup. The values above were calculated based on the ILS data for acetone, acetophenone,
α-methylstyrene, and 2-methylbenzofuran in Table 3.
1.3 In determining the conformance of the test results using this method to applicable specifications, results shall be rounded off
in accordance with the rounding-off method of Practice E29.
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. For specific hazard statements, see Section 9.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D3852 Practice for Sampling and Handling Phenol, Cresols, and Cresylic Acid
D4307 Practice for Preparation of Liquid Blends for Use as Analytical Standards
D4790 Terminology of Aromatic Hydrocarbons and Related Chemicals
D6809 Guide for Quality Control and Quality Assurance Procedures for Aromatic Hydrocarbons and Related Materials
E29 Practice for Using Significant Digits in Test Data to Determine Conformance with Specifications
E355 Practice for Gas Chromatography Terms and Relationships
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
This test method is under the jurisdiction of ASTM Committee D16 on Aromatic Hydrocarbons Aromatic, Industrial, Specialty and Related Chemicals and is the direct
responsibility of Subcommittee D16.02 on Oxygenated Aromatics.
Current edition approved June 1, 2016Nov. 1, 2021. Published June 2016November 2021. Originally approved in 1997. Last previous edition approved in 20122016 as
D6142D6142 – 16. – 12. DOI: 10.1520/D6142-16.10.1520/D6142-21.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D6142 − 21
E1510 Practice for Installing Fused Silica Open Tubular Capillary Columns in Gas Chromatographs
2.2 Other Document:
OSHA Regulations, 29 CFR paragraphs 1910.1000 and 1910.1200
3. Terminology
3.1 See Terminology D4790 for definitiondefinitions of terms used in this test method.
4. Summary of Test Method
4.1 A known amount of an internal standard is added to a sample of phenol. The prepared sample is mixed and analyzed by a gas
chromatograph equipped with a flame ionization detector (FID). The peak area of each impurity and the internal standard is
measured. The amount of each impurity is calculated from the ratio of the peak area of the internal standard versus the peak area
of the impurity. Results are reported in milligrams per kilogram.
5. Significance and Use
5.1 This test method is suitable for setting specifications on phenol and for use as an internal quality control tool where phenol
is produced or is used in a manufacturing process. It may also be used in development or research work involving phenol. It is
generally applied to determining those commonly occurring impurities such as mesityl oxide, cumene, hydroxyacetone, acetone,
alpha-methylstyrene,α-methylstyrene, 2-methylbenzofuran, and acetophenone.
5.2 Purity is commonly reported by subtracting the determined expected impurities from 100.00. However, a gas chromatographic
analysis cannot determine absolute purity if water is present or unknown components are contained within the material being
examined.
6. Interferences
6.1 The internal standard chosen must be sufficiently resolved from any impurity and the phenol peak.
6.2 Any solvent used must also be sufficiently resolved from any impurity, the internal standard, and the phenol peak.
7. Apparatus
7.1 Gas Chromatograph—Any chromatograph having a flame ionization detector that can be operated at the conditions given in
TABLE 1 Recommended Operating Conditions
Column: To use for identification of all components
Tubing fused silica
Stationary phase polyethylene glycol-acid modified
Solid support crosslinked
Film thickness, μ 0.5
Length, m 50
Inside diameter, m 0.32
Inside diameter, mm 0.32
Carrier Gas Helium. Nitrogen or hydrogen are permitted. Nitrogen
and hydrogen carrier gas require different conditions.
The user must conduct the necessary evaluation to
determine that equivalent results are obtained.
Flow rate mL/min 1.3
Temperature, °C
Injector 180
Detector 230
Oven
Initial, °C 110 for 6 min
Rate, °C 12 per min
Final, °C 210 for 90 min
Internal Standard sec-butyl alcohol
Available from U.S. Government Printing Office Superintendent of Documents, 732 N. Capitol St., NW, Mail Stop: SDE, Washington, DC 20401, http://
www.access.gpo.gov.
D6142 − 21
Table 1. The system should have sufficient sensitivity to obtain a minimum peak height response for a 2 mg/kg impurity twice the
height of the signal background noise.Chromatographic data systems are preferred but electronic integration may be used if the
user can demonstrate that the results are consistent with the precision statement.
7.2 Columns—The choice of column is based on resolution requirements. Any column may be used that is capable of resolving
all significant impurities from the major component. The column and conditions described in Table 1 have been used successfully
and shall be used as referee in cases of dispute.
7.3 Recorder—Chromatographic data systems are preferred but electronic integration may be used if the user can demonstrate that
the results are consistent with the precision statement. Recorders are not considered adequate for meeting the precision
requirements of this standard.
8. Reagents
8.1 Purity of Reagent—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available. Other grades may be used, provided it is first ascertained that the reagent is of sufficiently high
purity to permit its use without lessening the accuracy of the determination.
8.2 High Purity Phenol—Phenol (99.99 % or greater purity).
8.3 Carrier Gas, Makeup, and Detector Gases—Helium, hydrogen, nitrogen, or other carrier, makeup and detector gases 99.999 %
minimum purity. Oxygen in carrier gas less than 1 ppm, 1 ppm, less than 0.5 ppm is preferred. Purify carrier, makeup, and detector
gases to remove oxygen, water, and hydrocarbons. Helium was the carrier used for the conditions in Table 1.
8.4 Compressed Air—Purify air to remove water and hydrocarbons. Air for an FID should contain less than 0.1 ppm THC.
8.5 Pure Compounds for Calibration,Equipment Setup Check Sample: shall include mesityl oxide, cumene, hydroxyacetone,
acetone, alpha-methylstyrene, 2-methylbenzofuran, and acetophenone. The purity of all reagents should be 99.9 % or greater. If
the purity is less than 99 %, the concentration and identification of impurities must be known so that the composition of the
standard can be adjusted for the presence of the impurities.
8.5.1 For GC standards, a setup check sample should be included to:
8.5.1.1 Determine retention times for the components measured in GC standards.
8.5.1.2 Verify there is adequate resolution to measure the components of interest in GC standards.
8.5.1.3 Determine that the equipment has the sensitivity specified in the scope of the standard.
8.5.2 For GC standards and standards that determine trace levels, the equipment setup check sample should contain a component
with a concentration that is approximately two times the LOD stated in the scope of the standard. When the equipment setup check
sample is analyzed, an acceptable result for the trace component is 650 % of the expected concentration.
8.5.2.1 For GC standards where the primary material cannot be purified so that no impurities are detected, the following is
suggested:
(1) Add an impurity that is not present in the primary material. Determine that the impurity has the following properties:
(a) The impurity is essentially inert and unreactive in the primary material.
(b) The retention time is sufficiently separated from other impurities so that there will be no mistake in identification.
(c) The impurity is completely vaporized in the injection port.
(d) The impurity is well behaved on the column, that is, no fronting or tailing.
(e) The response factor is known and not significantly different from the components of interest.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade R
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...