ASTM D3339-21
(Test Method)Standard Test Method for Acid Number of Petroleum Products by Semi-Micro Color Indicator Titration
Standard Test Method for Acid Number of Petroleum Products by Semi-Micro Color Indicator Titration
SIGNIFICANCE AND USE
5.1 This test method measures the acid number of oils obtained from laboratory oxidation tests using smaller amounts of sample than Test Methods D974 or D664. It has specific application in Test Method D943 in which small aliquots of oil are periodically removed for testing by Test Method D3339. This test method, therefore, provides a means of monitoring the relative oxidation of oils, by measuring changes in acid number, at different time intervals and under the various oxidizing test conditions.
SCOPE
1.1 This test method covers the determination of acidic constituents in new or used petroleum products and lubricants soluble or nearly soluble in mixtures of toluene, and isopropyl alcohol. The test method is especially intended for cases in which the amount of sample available to be analyzed is too small to allow accurate analysis by Test Methods D974 or D664. It is applicable for the determination of acids having dissociation constants in water larger than 10−9. Extremely weak acids having dissociation constants smaller than 10−9 do not interfere. Salts titrate if their hydrolysis constants are larger than 10−9.
1.2 This test method can be used to indicate relative changes in acid number that occur in an oil during use under oxidizing conditions. Although the titration is made under definite equilibrium conditions, the method does not measure an absolute acidic property that can be used to predict performance of an oil under service conditions. No general relationship between bearing corrosion and acid number is known.
1.3 Since this test method requires substantially less sample than Test Methods D974 or D664, it provides an advantageous means of monitoring an oxidation test by changes in acid number by (1) minimizing test sample depletion for acid number analyses and thus minimizing the disturbance of the test or (2) allowing additional acid number analyses to be made while maintaining the same test sample depletion and thus providing additional data.
Note 1: Some oils, such as many cutting oils, rust-proofing oils, and similar compounded oils, or excessively dark-colored oils, may be more difficult to analyze by this test method due to obscurity of the color-indicator end point. These oils can be analyzed by Test Method D664 provided sufficient sample is available. However, this situation is much less likely using Test Method D3339 than using Test Method D974 due to the use of a more highly dilute sample during the titration and due to the greater stability of the end point color change. The acid numbers obtained by Test Method D3339 may or may not be numerically the same as those obtained by Test Method D664 but they should be of the same order of magnitude.
Note 2: The results obtained using this method have been found to be numerically the same as those obtained using Test Method D974, within the precision of the two methods, for new or oxidized lubricants of the type primarily intended for hydraulic or steam turbine type service. The oxidized lubricants were obtained using the Test Method D943 oxidation test. This correlation is shown by the correlation coefficient r = 0.989 with slope s = + 1.017 and intercept y = + 0.029, calculated using the acid numbers obtained using both titration methods for the samples used for the precision statement (12.2).2
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 9, A1.1.4, A2.3.1, and A2.10.1.
1.6 This international standard was developed in accordance with internationally recognized principles on sta...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2021
- Technical Committee
- D02 - Petroleum Products, Liquid Fuels, and Lubricants
- Drafting Committee
- D02.06 - Analysis of Liquid Fuels and Lubricants
Relations
- Effective Date
- 15-Jun-2017
- Effective Date
- 01-May-2017
- Effective Date
- 01-Dec-2014
- Effective Date
- 15-Apr-2012
- Refers
ASTM D4057-06(2011) - Standard Practice for Manual Sampling of Petroleum and Petroleum Products - Effective Date
- 01-Jun-2011
- Effective Date
- 15-May-2011
- Effective Date
- 15-May-2011
- Effective Date
- 01-Mar-2009
- Effective Date
- 01-Nov-2007
- Effective Date
- 01-Jul-2007
- Effective Date
- 01-Nov-2006
- Effective Date
- 01-Nov-2006
- Effective Date
- 01-Nov-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Jan-2006
Overview
ASTM D3339-21, titled Standard Test Method for Acid Number of Petroleum Products by Semi-Micro Color Indicator Titration, is a widely recognized method for the precise determination of acidic constituents in new or used petroleum products and lubricants. Developed by ASTM International, this standard provides procedures for testing samples that are soluble or nearly soluble in toluene and isopropyl alcohol mixtures. It is especially useful when only a small sample is available, offering significant advantages over other titration methods such as ASTM D974 or D664.
This test method is primarily intended for monitoring the oxidation of oils during laboratory tests. By measuring the acid number over time, it supports the evaluation of oil degradation and contamination, contributing to effective maintenance and quality control in industrial environments.
Key Topics
- Acid Number Determination: This method quantifies the amount of acidic constituents in petroleum products by titration, expressed as milligrams of potassium hydroxide (KOH) per gram of sample.
- Semi-Micro Color Indicator Titration: The procedure utilizes a color indicator (p-naphtholbenzein) for detecting the endpoint of titration, enabling accurate results using minimal sample sizes.
- Sample Size Efficiency: Requires significantly less sample than traditional methods, making it suitable for studies where preserving the sample is essential.
- Applicability: Suitable for oils with acids having dissociation constants in water greater than 10^-9. Very weak acids and some dark-colored oils may require alternative methods.
- Testing Under Oxidation: Valuable for periodic monitoring of lubricant degradation in controlled oxidation tests, especially those following ASTM D943.
Applications
- Lubricant Condition Monitoring: Enables trend analysis of acid number in hydraulic and turbine oils during use, supporting predictive maintenance and minimizing equipment failure risks.
- R&D and Quality Assurance: Essential for research settings with limited sample volumes, allowing repeated analysis without significant sample depletion or disturbance of ongoing oxidation tests.
- Petroleum Product Evaluation: Used for both new and used oils to indicate changes in acidic content, helping assess oil lifespan and the potential need for replacement.
- Compliance with Industry Standards: Supports manufacturers and laboratories in meeting industry benchmarks for petroleum product analysis.
- Correlation with Other Standards: Results have shown strong correlation with measurements made by Test Method D974, ensuring reliability and consistency.
Related Standards
- ASTM D974: Standard Test Method for Acid and Base Number by Color-Indicator Titration
- ASTM D664: Standard Test Method for Acid Number of Petroleum Products by Potentiometric Titration
- ASTM D943: Test Method for Oxidation Characteristics of Inhibited Mineral Oils
- ASTM D1193: Specification for Reagent Water
- ASTM D4057 and D4177: Practices for Manual and Automatic Sampling of Petroleum and Petroleum Products
By adhering to ASTM D3339-21, laboratories and industries benefit from reliable acid number determination, especially in applications where only small sample volumes are available. This method helps maintain the quality and operational reliability of petroleum-based oils, supporting decision-making in product development, quality control, and maintenance programs. Accurate tracking of acid number trends is critical for optimum equipment performance and oil management, making ASTM D3339-21 a valuable standard in the petroleum industry.
Buy Documents
ASTM D3339-21 - Standard Test Method for Acid Number of Petroleum Products by Semi-Micro Color Indicator Titration
REDLINE ASTM D3339-21 - Standard Test Method for Acid Number of Petroleum Products by Semi-Micro Color Indicator Titration
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D3339-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Acid Number of Petroleum Products by Semi-Micro Color Indicator Titration". This standard covers: SIGNIFICANCE AND USE 5.1 This test method measures the acid number of oils obtained from laboratory oxidation tests using smaller amounts of sample than Test Methods D974 or D664. It has specific application in Test Method D943 in which small aliquots of oil are periodically removed for testing by Test Method D3339. This test method, therefore, provides a means of monitoring the relative oxidation of oils, by measuring changes in acid number, at different time intervals and under the various oxidizing test conditions. SCOPE 1.1 This test method covers the determination of acidic constituents in new or used petroleum products and lubricants soluble or nearly soluble in mixtures of toluene, and isopropyl alcohol. The test method is especially intended for cases in which the amount of sample available to be analyzed is too small to allow accurate analysis by Test Methods D974 or D664. It is applicable for the determination of acids having dissociation constants in water larger than 10−9. Extremely weak acids having dissociation constants smaller than 10−9 do not interfere. Salts titrate if their hydrolysis constants are larger than 10−9. 1.2 This test method can be used to indicate relative changes in acid number that occur in an oil during use under oxidizing conditions. Although the titration is made under definite equilibrium conditions, the method does not measure an absolute acidic property that can be used to predict performance of an oil under service conditions. No general relationship between bearing corrosion and acid number is known. 1.3 Since this test method requires substantially less sample than Test Methods D974 or D664, it provides an advantageous means of monitoring an oxidation test by changes in acid number by (1) minimizing test sample depletion for acid number analyses and thus minimizing the disturbance of the test or (2) allowing additional acid number analyses to be made while maintaining the same test sample depletion and thus providing additional data. Note 1: Some oils, such as many cutting oils, rust-proofing oils, and similar compounded oils, or excessively dark-colored oils, may be more difficult to analyze by this test method due to obscurity of the color-indicator end point. These oils can be analyzed by Test Method D664 provided sufficient sample is available. However, this situation is much less likely using Test Method D3339 than using Test Method D974 due to the use of a more highly dilute sample during the titration and due to the greater stability of the end point color change. The acid numbers obtained by Test Method D3339 may or may not be numerically the same as those obtained by Test Method D664 but they should be of the same order of magnitude. Note 2: The results obtained using this method have been found to be numerically the same as those obtained using Test Method D974, within the precision of the two methods, for new or oxidized lubricants of the type primarily intended for hydraulic or steam turbine type service. The oxidized lubricants were obtained using the Test Method D943 oxidation test. This correlation is shown by the correlation coefficient r = 0.989 with slope s = + 1.017 and intercept y = + 0.029, calculated using the acid numbers obtained using both titration methods for the samples used for the precision statement (12.2).2 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 9, A1.1.4, A2.3.1, and A2.10.1. 1.6 This international standard was developed in accordance with internationally recognized principles on sta...
SIGNIFICANCE AND USE 5.1 This test method measures the acid number of oils obtained from laboratory oxidation tests using smaller amounts of sample than Test Methods D974 or D664. It has specific application in Test Method D943 in which small aliquots of oil are periodically removed for testing by Test Method D3339. This test method, therefore, provides a means of monitoring the relative oxidation of oils, by measuring changes in acid number, at different time intervals and under the various oxidizing test conditions. SCOPE 1.1 This test method covers the determination of acidic constituents in new or used petroleum products and lubricants soluble or nearly soluble in mixtures of toluene, and isopropyl alcohol. The test method is especially intended for cases in which the amount of sample available to be analyzed is too small to allow accurate analysis by Test Methods D974 or D664. It is applicable for the determination of acids having dissociation constants in water larger than 10−9. Extremely weak acids having dissociation constants smaller than 10−9 do not interfere. Salts titrate if their hydrolysis constants are larger than 10−9. 1.2 This test method can be used to indicate relative changes in acid number that occur in an oil during use under oxidizing conditions. Although the titration is made under definite equilibrium conditions, the method does not measure an absolute acidic property that can be used to predict performance of an oil under service conditions. No general relationship between bearing corrosion and acid number is known. 1.3 Since this test method requires substantially less sample than Test Methods D974 or D664, it provides an advantageous means of monitoring an oxidation test by changes in acid number by (1) minimizing test sample depletion for acid number analyses and thus minimizing the disturbance of the test or (2) allowing additional acid number analyses to be made while maintaining the same test sample depletion and thus providing additional data. Note 1: Some oils, such as many cutting oils, rust-proofing oils, and similar compounded oils, or excessively dark-colored oils, may be more difficult to analyze by this test method due to obscurity of the color-indicator end point. These oils can be analyzed by Test Method D664 provided sufficient sample is available. However, this situation is much less likely using Test Method D3339 than using Test Method D974 due to the use of a more highly dilute sample during the titration and due to the greater stability of the end point color change. The acid numbers obtained by Test Method D3339 may or may not be numerically the same as those obtained by Test Method D664 but they should be of the same order of magnitude. Note 2: The results obtained using this method have been found to be numerically the same as those obtained using Test Method D974, within the precision of the two methods, for new or oxidized lubricants of the type primarily intended for hydraulic or steam turbine type service. The oxidized lubricants were obtained using the Test Method D943 oxidation test. This correlation is shown by the correlation coefficient r = 0.989 with slope s = + 1.017 and intercept y = + 0.029, calculated using the acid numbers obtained using both titration methods for the samples used for the precision statement (12.2).2 1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 9, A1.1.4, A2.3.1, and A2.10.1. 1.6 This international standard was developed in accordance with internationally recognized principles on sta...
ASTM D3339-21 is classified under the following ICS (International Classification for Standards) categories: 75.080 - Petroleum products in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3339-21 has the following relationships with other standards: It is inter standard links to ASTM D943-17, ASTM D664-11a(2017), ASTM D974-14e1, ASTM D974-12, ASTM D4057-06(2011), ASTM D664-11, ASTM D974-11, ASTM D664-09, ASTM D974-07, ASTM D664-07, ASTM D664-06a, ASTM D664-06ae1, ASTM D974-06, ASTM D1193-06, ASTM D664-06. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3339-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D3339 − 21
Standard Test Method for
Acid Number of Petroleum Products by Semi-Micro Color
Indicator Titration
This standard is issued under the fixed designation D3339; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
obtained by Test Method D664 but they should be of the same order of
1. Scope*
magnitude.
1.1 This test method covers the determination of acidic
NOTE 2—The results obtained using this method have been found to be
constituents in new or used petroleum products and lubricants numerically the same as those obtained using Test Method D974, within
the precision of the two methods, for new or oxidized lubricants of the
soluble or nearly soluble in mixtures of toluene, and isopropyl
type primarily intended for hydraulic or steam turbine type service. The
alcohol. The test method is especially intended for cases in
oxidized lubricants were obtained using the Test Method D943 oxidation
which the amount of sample available to be analyzed is too
test.Thiscorrelationisshownbythecorrelationcoefficient r=0.989with
small to allow accurate analysis by Test Methods D974 or
slope s=+1.017 and intercept y=+0.029, calculated using the acid
D664. It is applicable for the determination of acids having numbersobtainedusingbothtitrationmethodsforthesamplesusedforthe
−9
precision statement (12.2).
dissociation constants in water larger than 10 . Extremely
−9
weak acids having dissociation constants smaller than 10 do
1.4 The values stated in SI units are to be regarded as
notinterfere.Saltstitrateiftheirhydrolysisconstantsarelarger
standard. No other units of measurement are included in this
−9
than 10 .
standard.
1.2 Thistestmethodcanbeusedtoindicaterelativechanges 1.5 This standard does not purport to address all of the
in acid number that occur in an oil during use under oxidizing safety concerns, if any, associated with its use. It is the
conditions. Although the titration is made under definite responsibility of the user of this standard to establish appro-
equilibrium conditions, the method does not measure an priate safety, health, and environmental practices and deter-
absolute acidic property that can be used to predict perfor- mine the applicability of regulatory limitations prior to use.
mance of an oil under service conditions. No general relation- For specific warning statements, see Sections 7 and 9, A1.1.4,
ship between bearing corrosion and acid number is known. A2.3.1, and A2.10.1.
1.6 This international standard was developed in accor-
1.3 Since this test method requires substantially less sample
dance with internationally recognized principles on standard-
thanTest Methods D974 or D664, it provides an advantageous
ization established in the Decision on Principles for the
means of monitoring an oxidation test by changes in acid
Development of International Standards, Guides and Recom-
number by (1) minimizing test sample depletion for acid
mendations issued by the World Trade Organization Technical
number analyses and thus minimizing the disturbance of the
Barriers to Trade (TBT) Committee.
testor(2)allowingadditionalacidnumberanalysestobemade
while maintaining the same test sample depletion and thus
2. Referenced Documents
providing additional data.
2.1 ASTM Standards:
NOTE 1—Some oils, such as many cutting oils, rust-proofing oils, and
D664Test Method for Acid Number of Petroleum Products
similar compounded oils, or excessively dark-colored oils, may be more
by Potentiometric Titration
difficult to analyze by this test method due to obscurity of the color-
D943TestMethodforOxidationCharacteristicsofInhibited
indicator end point. These oils can be analyzed by Test Method D664
provided sufficient sample is available. However, this situation is much Mineral Oils
less likely usingTest Method D3339 than usingTest Method D974 due to
D974Test Method for Acid and Base Number by Color-
the use of a more highly dilute sample during the titration and due to the
Indicator Titration
greaterstabilityoftheendpointcolorchange.Theacidnumbersobtained
D1193Specification for Reagent Water
by Test Method D3339 may or may not be numerically the same as those
Use of the correlation coefficient is given in Mack, C., Essentials of Statistics
This test method is under the jurisdiction of ASTM Committee D02 on for Scientists and Technologists, Plenum Press, New York, NY, 1967, or other
Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of publications on statistics.
Subcommittee D02.06 on Analysis of Liquid Fuels and Lubricants. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2021. Published November 2021. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1974. Last previous edition approved in 2017 as D3339–12 (2017). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/D3339-21. the ASTM website.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3339 − 21
D4057Practice for Manual Sampling of Petroleum and to remove atmospheric carbon dioxide and water, for example,
Petroleum Products tubes containing 10mesh to 20mesh anhydrous calcium sul-
D4177Practice for Automatic Sampling of Petroleum and fate as desiccant and soda-lime as carbon dioxide absorbent.
Petroleum Products Precautions must be taken to prevent introduction of any
soda-lime into the titrant reservoir or buret.
3. Terminology
6.3 Titration Beaker—100mL capacity tall-form Berzelius
3.1 Definitions:
beaker without pouring spout. Approximate dimensions are
3.1.1 acid number, n—the quantity of a specified base,
47mm in inside diameter and 79mm in height.
expressed in milligrams of potassium hydroxide per gram of
6.4 Titration Beaker Purging Stopper—Astoppertoenclose
sample, required to titrate a sample in a specified solvent to a
the titration beaker. The stopper must be composed of an
specified endpoint using a specified detection system.
elastomeric material, such as neoprene, that is essentially
3.1.1.1 Discussion—In this test method, acids or salts with
−9
unaffected by the titration solvent. Approximate stopper di-
dissociation constants greater than 10 , are titrated to a green
mensions are 53mm top diameter, 45mm bottom diameter,
end point with p-naphtholbenzein indicator.
and 25mm height. The stopper is fitted with a 8mm outside
3.1.2 used oil, n—any oil that has been in a piece of
diameterglassinlettubeextending15mm 62mmbeyondthe
equipment (for example, an engine, gearbox, transformer or
bottom of the stopper and with a 7mm 6 1mm inside
turbine) whether operated or not.
diameter hole. The inlet tube and hole are placed on opposite
3.1.2.1 Discussion—Typically, in this test method, the acid-
sides of the stopper with a center-to-center separation distance
ity of oxidized hydraulic or steam turbine oils is measured.
of 30mm 6 1mm.
4. Summary of Test Method 6.5 Purge Gas Rotameter, capable of indicating a flow rate
of 10L⁄h.
4.1 To determine the acid number a sample of the oil is
dissolved in a solvent consisting of toluene, isopropyl alcohol, 6.6 Stirrer Motor, variable speed, magnetically linked.
and a small amount of water. The resulting single-phase
NOTE 3—Apropeller may also be used instead of a magnetic stirrer to
solution is titrated at room temperature under a nitrogen
mix the samples.
atmosphere with standardized 0.01 M potassium hydroxide
6.7 Stirring Bar, cylindrical, TFE-fluorocarbon encased,
(KOH) in isopropyl alcohol to the stable green color of the
25.4mm long and 7.9mm in diameter.
added p-naphtholbenzein indicator.
6.8 Pipet, capable of transferring 0.100mL 6 0.002mL of
5. Significance and Use
titration indicator solution.
5.1 This test method measures the acid number of oils
6.9 Titration Solvent Buret—A 500mL or larger capacity
obtainedfromlaboratoryoxidationtestsusingsmalleramounts
buretwith5mLsubdivisions.Thetopoftheburetisstoppered
of sample than Test Methods D974 or D664. It has specific
and connected with an absorption tube, as in 6.2, to remove
applicationinTestMethodD943inwhichsmallaliquotsofoil
atmosphericcarbondioxideandwater.Analternativemeansof
are periodically removed for testing by Test Method D3339.
dispensing the titration solvent may be used provided a
Thistestmethod,therefore,providesameansofmonitoringthe
dispensingrepeatabilitywithin 61mLfor40mLisobtainable
relative oxidation of oils, by measuring changes in acid
and the solvent in the dispenser is isolated from atmospheric
number, at different time intervals and under the various
carbon dioxide and water.
oxidizing test conditions.
NOTE 4—An automated photometric device may also be used for
detectingthetitrationendpoint.However,theprecisionestimatesgivenin
6. Apparatus (Refer to Fig. 1)
Section 13 may not apply to this mode of titration.
6.1 Titration Buret—A micro scale, automatic buret with
7. Reagents and Materials
0.01mL subdivisions and at least a 2mL buret capacity.
7.1 Purity of Reagents—Reagent grade chemicals shall be
6.2 Titrant Reservoir—The preferred reservoir is one that is
used in all tests. Unless otherwise indicated, it is intended that
integral with the automatic buret, such as shown in Fig. 1.A
all reagents shall conform to the specifications of the Commit-
titrantreservoirseparatefromtheautomaticburetmaybeused
tee onAnalytical Reagents of theAmerican Chemical Society,
if the line connecting the reservoir with the buret is all glass.
where such specifications are available or to other such
Exposure of the titrant to light should be minimized by use of
recognizedstandardsforreagentchemicals. Othergradesmay
amber glass for the reservoir, by wrapping the reservoir with
be used, provided it is first ascertained that the reagent is of
foil such as aluminum foil, or by other suitable means. Also,
sufficient high purity to permit its use without lessening the
the tube in the reservoir for titrant withdrawal is adjusted so
accuracy of the determination.
that the end of the tube is about 20mm from the bottom of the
reservoir so that any precipitate that may collect on the bottom
of the reservoir will not be disturbed. To further avoid
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington,
disturbing any precipitate in the reservoir, movement of the
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
reservoir must be minimized.
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
6.2.1 With either type of reservoir all entrances and exits to
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
the buret and reservoir must be connected to absorption tubes copeial Convention, Inc. (USPC), Rockville, MD.
D3339 − 21
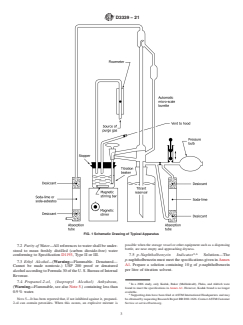
FIG. 1 Schematic Drawing of Typical Apparatus
possible when the storage vessel or other equipment such as a dispensing
7.2 Purity of Water—All references to water shall be under-
bottle, are near empty and approaching dryness.
stood to mean freshly distilled (carbon dioxide-free) water
5,6
conforming to Specification D1193, Type II or III. 7.5 p-Naphtholbenzein Indicator Solution—The
p-naphtholbenzeinmustmeetthespecificationsgiveninAnnex
7.3 Ethyl Alcohol—(Warning—Flammable. Denatured—
A1. Prepare a solution containing 10g of p-naphtholbenzein
Cannot be made nontoxic.) USP 200 proof or denatured
per litre of titration solvent.
alcoholaccordingtoFormula30oftheU.S.BureauofInternal
Revenue.
7.4 Propanol-2-ol, (Isopropyl Alcohol) Anhydrous,
In a 2006 study, only Kodak, Baker (Mallinkrodt), Fluka, and Aldrich were
(Warning—Flammable, see also Note 5.) containing less than
found to meet the specifications in AnnexA1. However, Kodak brand is no longer
available.
0.9% water.
Supporting data have been filed atASTM International Headquarters and may
NOTE 5—It has been reported that, if not inhibited against it, propanol- beobtainedbyrequestingResearchReportRR:D02-1626.ContactASTMCustomer
2-ol can contain peroxides. When this occurs, an explosive mixture is Service at service@astm.org.
D3339 − 21
A
TABLE 1 Size of Sample
7.6 Nitrogen, dry and carbon dioxide-free.
Total Acid Number
7.6.1 In order to obtain repeatable results and a stable end
Sample Weight
Range, mg KOH/g
point color change, it is especially important that the nitrogen Range, g
Sample
purgegasbefreeofcarbondioxide.Prepurifiedgradenitrogen
< 0.01 5.0 to 2.0
has been found to be satisfactory.
0.01<0.1 2.0to1.5
0.1<0.5 1.5to1.0
7.7 Phenolphthalein Indicator Solution—Dissolve 0.10g of
0.5<3.0 1.0to0.2
solid pure phenolphthalein in 50mL of water and 50mL of >3.0 0.2to0.1
A
ethyl alcohol.
Dark colored samples with total acid numbers of 0.1 or less may require use of
smaller size samples to minimize interference of the sample color with the indicator
7.8 Titration Solvent—Consisting of 5.0mL 6 0.1mL of
colors.
water, 495mL 6 1mL of anhydrous propanol-2-ol, and
500mL 6 1mL of toluene.
7.9 Potassium Hydroxide Solution, Standard Alcoholic
8.1.1 Whensamplingusedlubricants,thespecimenshallbe
(0.01 M), (Warning—Corrosive.)
representative of the system sampled and shall be free of
contamination from external sources.
NOTE 6—Commercially available reagents may be used in place of
8.1.2 Agitateusedoilsamplesthoroughlytoensurethatany
laboratory preparations.
sedimentpresentishomogeneouslysuspendedbeforeanalysis,
7.9.1 Preparation, Add 3g of solid KOH to approximately
as the sediment can be acidic or basic or have adsorbed acidic
1Lofanhydrouspropanol-2-ol(isopropylalcohol)(containing
or basic material from the sample. When necessary, samples
less than 0.9% water) in a 2L Erlenmeyer flask. Boil the
are warmed to aid mixing.
mixture gently on a steam bath for 10min to 15min while
stirring to prevent caking of solids on the bottom of the flask.
NOTE 9—As used oils can change appreciably in storage, samples
should be tested as soon as possible after removal from the lubricating
Add about 1g of barium hydroxide and again boil gently for
system and the dates of sampling and testing, if known, should be noted.
about 10min. Cool to room temperature, stopper to prevent
contactwiththeroomatmosphereandallowtostandovernight
9. Procedure
(about 16h). Filter the supernatant liquid through a 10µm
9.1 Sample Titration—Weigh to the nearest 0.1mg the
TFE-fluorocarbonmembranefilterwhileavoidingunnecessary
required amount of sample into a clean, tared titration beaker.
exposure to the atmosphere and then dilute the solution
Guidelines for determining the amount of sample required are
(approximately 0.05 M) with anhydrous isopropyl alcohol to a
giveninTable1.Placethemagneticstirringbarintothebeaker
total volume of about 5L (Note 7). Store the titrant in a
taking care not to splash the sample. Pour 40mL of titration
chemically resistant dispensing bottle out of contact with cork,
solvent into the titration beaker, using part of this volume of
rubber, or saponifiable stopcock lubricant and protected by a
solvent to wash down any sample that may have splashed onto
guard tube containing
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D3339 − 12 (Reapproved 2017) D3339 − 21
Standard Test Method for
Acid Number of Petroleum Products by Semi-Micro Color
Indicator Titration
This standard is issued under the fixed designation D3339; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Scope*
1.1 This test method covers the determination of acidic constituents in new or used petroleum products and lubricants soluble or
nearly soluble in mixtures of toluene, and isopropyl alcohol. The test method is especially intended for cases in which the amount
of sample available to be analyzed is too small to allow accurate analysis by Test Methods D974 or D664. It is applicable for the
−9
determination of acids having dissociation constants in water larger than 10 . Extremely weak acids having dissociation constants
−9 −9
smaller than 10 do not interfere. Salts titrate if their hydrolysis constants are larger than 10 .
1.2 This test method can be used to indicate relative changes in acid number that occur in an oil during use under oxidizing
conditions. Although the titration is made under definite equilibrium conditions, the method does not measure an absolute acidic
property that can be used to predict performance of an oil under service conditions. No general relationship between bearing
corrosion and acid number is known.
1.3 Since this test method requires substantially less sample than Test Methods D974 or D664, it provides an advantageous means
of monitoring an oxidation test by changes in acid number by (1) minimizing test sample depletion for acid number analyses and
thus minimizing the disturbance of the test or (2) allowing additional acid number analyses to be made while maintaining the same
test sample depletion and thus providing additional data.
NOTE 1—Some oils, such as many cutting oils, rust-proofing oils, and similar compounded oils, or excessively dark-colored oils, may be more difficult
to analyze by this test method due to obscurity of the color-indicator end point. These oils can be analyzed by Test Method D664 provided sufficient
sample is available. However, this situation is much less likely using Test Method D3339 than using Test Method D974 due to the use of a more highly
dilute sample during the titration and due to the greater stability of the end point color change. The acid numbers obtained by Test Method D3339 may
or may not be numerically the same as those obtained by Test Method D664 but they should be of the same order of magnitude.
NOTE 2—The results obtained using this method have been found to be numerically the same as those obtained using Test Method D974, within the
precision of the two methods, for new or oxidized lubricants of the type primarily intended for hydraulic or steam turbine type service. The oxidized
lubricants were obtained using the Test Method D943 oxidation test. This correlation is shown by the correlation coefficient r = 0.989 with slope
s = + 1.017 and intercept y = + 0.029, calculated using the acid numbers obtained using both titration methods for the samples used for the precision
statement (12.2).
1.4 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
This test method is under the jurisdiction of ASTM Committee D02 on Petroleum Products, Liquid Fuels, and Lubricants and is the direct responsibility of Subcommittee
D02.06 on Analysis of Liquid Fuels and Lubricants.
Current edition approved May 1, 2017Nov. 1, 2021. Published June 2017November 2021. Originally approved in 1974. Last previous edition approved in 20122017 as
D3339 – 12.D3339 – 12 (2017). DOI: 10.1520/D3339-12R17.10.1520/D3339-21.
Use of the correlation coefficient is given in Mack, C., Essentials of Statistics for Scientists and Technologists, Plenum Press, New York, NY, 1967, or other publications
on statistics.
*A Summary of Changes section appears at the end of this standard
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3339 − 21
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. For specific warning statements, see Sections 7 and 9, A1.1.4, A2.3.1, and
A2.10.1.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D664 Test Method for Acid Number of Petroleum Products by Potentiometric Titration
D943 Test Method for Oxidation Characteristics of Inhibited Mineral Oils
D974 Test Method for Acid and Base Number by Color-Indicator Titration
D1193 Specification for Reagent Water
D4057 Practice for Manual Sampling of Petroleum and Petroleum Products
D4177 Practice for Automatic Sampling of Petroleum and Petroleum Products
3. Terminology
3.1 Definitions:
3.1.1 acid number, n—the quantity of a specified base, expressed in milligrams of potassium hydroxide per gram of sample,
required to titrate a sample in a specified solvent to a specified endpoint using a specified detection system.
3.1.1.1 Discussion—
−9
In this test method, acids or salts with dissociation constants greater than 10 , are titrated to a green end point with
p-naphtholbenzein indicator.
3.1.2 used oil, n—any oil that has been in a piece of equipment (for example, an engine, gearbox, transformer or turbine) whether
operated or not.
3.1.2.1 Discussion—
Typically, in this test method, the acidity of oxidized hydraulic or steam turbine oils is measured.
4. Summary of Test Method
4.1 To determine the acid number a sample of the oil is dissolved in a solvent consisting of toluene, isopropyl alcohol, and a small
amount of water. The resulting single-phase solution is titrated at room temperature under a nitrogen atmosphere with standardized
0.01 M potassium hydroxide (KOH) in isopropyl alcohol to the stable green color of the added p-naphtholbenzein indicator.
5. Significance and Use
5.1 This test method measures the acid number of oils obtained from laboratory oxidation tests using smaller amounts of sample
than Test Methods D974 or D664. It has specific application in Test Method D943 in which small aliquots of oil are periodically
removed for testing by Test Method D3339. This test method, therefore, provides a means of monitoring the relative oxidation of
oils, by measuring changes in acid number, at different time intervals and under the various oxidizing test conditions.
6. Apparatus (Refer to Fig. 1)
6.1 Titration Buret—A micro scale, automatic buret with 0.01 mL subdivisions and at least a 2 mL buret capacity.
6.2 Titrant Reservoir—The preferred reservoir is one that is integral with the automatic buret, such as shown in Fig. 1. A titrant
reservoir separate from the automatic buret may be used if the line connecting the reservoir with the buret is all glass. Exposure
of the titrant to light should be minimized by use of amber glass for the reservoir, by wrapping the reservoir with foil such as
aluminum foil, or by other suitable means. Also, the tube in the reservoir for titrant withdrawal is adjusted so that the end of the
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
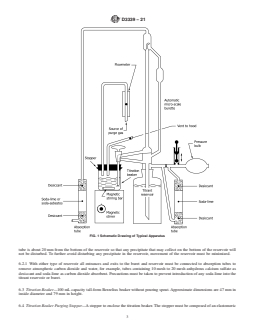
D3339 − 21
FIG. 1 Schematic Drawing of Typical Apparatus
tube is about 20 mm from the bottom of the reservoir so that any precipitate that may collect on the bottom of the reservoir will
not be disturbed. To further avoid disturbing any precipitate in the reservoir, movement of the reservoir must be minimized.
6.2.1 With either type of reservoir all entrances and exits to the buret and reservoir must be connected to absorption tubes to
remove atmospheric carbon dioxide and water, for example, tubes containing 10 mesh to 20 mesh anhydrous calcium sulfate as
desiccant and soda-lime as carbon dioxide absorbent. Precautions must be taken to prevent introduction of any soda-lime into the
titrant reservoir or buret.
6.3 Titration Beaker—100 mL capacity tall-form Berzelius beaker without pouring spout. Approximate dimensions are 47 mm in
inside diameter and 79 mm in height.
6.4 Titration Beaker Purging Stopper—A stopper to enclose the titration beaker. The stopper must be composed of an elastomeric
D3339 − 21
material, such as neoprene, that is essentially unaffected by the titration solvent. Approximate stopper dimensions are 53 mm top
diameter, 45 mm bottom diameter, and 25 mm height. The stopper is fitted with a 8 mm outside diameter glass inlet tube extending
15 mm 6 2 mm beyond the bottom of the stopper and with a 7 mm 6 1 mm inside diameter hole. The inlet tube and hole are
placed on opposite sides of the stopper with a center-to-center separation distance of 30 mm 6 1 mm.
6.5 Purge Gas Rotameter, capable of indicating a flow rate of 10 L ⁄h.
6.6 Stirrer Motor, variable speed, magnetically linked.
NOTE 3—A propeller may also be used instead of a magnetic stirrer to mix the samples.
6.7 Stirring Bar, cylindrical, TFE-fluorocarbon encased, 25.4 mm long and 7.9 mm in diameter.
6.8 Pipet, capable of transferring 0.100 mL 6 0.002 mL of titration indicator solution.
6.9 Titration Solvent Buret—A 500 mL or larger capacity buret with 5 mL subdivisions. The top of the buret is stoppered and
connected with an absorption tube, as in 6.2, to remove atmospheric carbon dioxide and water. An alternative means of dispensing
the titration solvent may be used provided a dispensing repeatability within 61 mL for 40 mL is obtainable and the solvent in the
dispenser is isolated from atmospheric carbon dioxide and water.
NOTE 4—An automated photometric device may also be used for detecting the titration end point. However, the precision estimates given in Section 13
may not apply to this mode of titration.
7. Reagents and Materials
7.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents shall conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society, where
such specifications are available or to other such recognized standards for reagent chemicals. Other grades may be used, provided
it is first ascertained that the reagent is of sufficient high purity to permit its use without lessening the accuracy of the determination.
7.2 Purity of Water—All references to water shall be understood to mean freshly distilled (carbon dioxide-free) water conforming
to Specification D1193, Type II or III.
7.3 Ethyl Alcohol—(Warning—Flammable. Denatured—Cannot be made nontoxic.) USP 200 proof or denatured alcohol
according to Formula 30 of the U. S. Bureau of Internal Revenue.
7.4 Propanol-2-ol, (Isopropyl Alcohol) Anhydrous, (Warning—Flammable, see also Note 5.) containing less than 0.9 % water.
NOTE 5—It has been reported that, if not inhibited against it, propanol-2-ol can contain peroxides. When this occurs, an explosive mixture is possible when
the storage vessel or other equipment such as a dispensing bottle, are near empty and approaching dryness.
5,6
7.5 p-Naphtholbenzein Indicator Solution—The p-naphtholbenzein must meet the specifications given in Annex A1. Prepare a
solution containing 10 g of p-naphtholbenzein per litre of titration solvent.
7.6 Nitrogen, dry and carbon dioxide-free.
7.6.1 In order to obtain repeatable results and a stable end point color change, it is especially important that the nitrogen purge
gas be free of carbon dioxide. Prepurified grade nitrogen has been found to be satisfactory.
Reagent Chemicals, American Chemical Society Specifications,ACS Reagent Chemicals, Specifications and Procedures for Reagents and Standard-Grade Reference
Materials, American Chemical Society, Washington, DC. For Suggestionssuggestions on the testing of reagents not listed by the American Chemical Society, see
AnnualAnalar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
In a 2006 study, only Kodak, Baker (Mallinkrodt), Fluka, and Aldrich were found to meet the specifications in Annex A1. However, Kodak brand is no longer available.
Supporting data have been filed at ASTM International Headquarters and may be obtained by requesting Research Report RR:D02-1626. Contact ASTM Customer
Service at service@astm.org.
D3339 − 21
7.7 Phenolphthalein Indicator Solution—Dissolve 0.10 g of solid pure phenolphthalein in 50 mL of water and 50 mL of ethyl
alcohol.
7.8 Titration Solvent—Consisting of 5.0 mL 6 0.1 mL of water, 495 mL 6 1 mL of anhydrous propanol-2-ol, and 500 mL 6 1 mL
of toluene.
7.9 Potassium Hydroxide Solution, Standard Alcoholic (0.01 M), (Warning—Corrosive.)
NOTE 6—Commercially available reagents may be used in place of laboratory preparations.
7.9.1 Preparation, Add 3 g of solid KOH to approximately 1 L of anhydrous propanol-2-ol (isopropyl alcohol) (containing less
than 0.9 % water) in a 2 L Erlenmeyer flask. Boil the mixture gently on a steam bath for 10 min to 15 min while stirring to prevent
caking of solids on the bottom of the flask. Add about 1 g of barium hydroxide and again boil gently for about 10 min. Cool to
room temperature, stopper to prevent contact with the room atmosphere and allow to stand overnight (about 16 h). Filter the
supernatant liquid through a 10 μm TFE-fluorocarbon membrane filter while avoiding unnecessary exposure to the atmosphere and
then dilute the solution (approximately 0.05 M) with anhydrous isopropyl alcohol to a total volume of about 5 L (Note 7). Store
the titrant in a chemically resistant dispensing bottle out of contact with cork, rubber, or saponifiable stopcock lubricant and
protected by a guard tube containing soda-lime. Minimize exposure of the titrant to light by storing in the dark or in an amber bottle
or by wrapping the bottle with aluminum foil.
NOTE 7—Care should be taken to ensure a final normality of 0.011 6 0.002.
NOTE 8—Because of the relatively large coefficient of cubic expansion of organic liquids, such as propanol-2-ol, the standard alcoholic solutions should
be standardized at temperatures close to those employed in the titrations of samples.
7.9.2 Standardization—The titrant is standardized (Note 8) against dried (at least 1 h at 110 °C), pure potassium hydrogen
phthalate using the method described in 9.1 for the acid number analysis, with the exception that water (40 mL 6 1 mL) is used
as the solvent and 6 drops of phenolphthalein solution is used as the indicator. The blank is obtained in the same manner except
that the potassium hydrogen phthalate is excluded. Standardize the titrant frequently enough to detect changes of 0.0003 M. The
mean molarity determined by at least duplicate analyses is used as the titrant molarity for the acid number calculations.
7.10 Toluene (Warning—Flammable. Vapor harmful.) Nitration-grade, or equivalent.
8. Preparation of Used Oil Samples
8.1 Strict observance of the sampling procedure described in 8.2 is necessary, since the sediment itself is acidic or basic or has
adsorbed acidic or basic material from the sample. Failure to obtain a representative sample causes serious errors.
8.1 Heat the sample (When applicable, refer Note 9) of used oil to 60 °C 6to Practice D4057 5 °C in the original container and
agitate until all sediment is homogeneously suspended in the oil ((Manual Sampling) Note 10). If the original container is of
opaque material, or if itor Practice D4177 is more than three fourths full, transfer the entire sample to a clear glass bottle having
a capacity at least one third greater than the volume of the sample. Transfer all traces of sediment from the original container to
the bottle by violent agitation of portions of the sample in the original container. After complete suspension of all sediment, strain
the sample or a convenient aliquot of the sample through a 100-mesh screen for the removal of large contaminating particles
((Automatic Sampling) for proper sampling techniques.Note 11).
NOTE 9—When samples are visibly free of sediment, the heating procedure may be omitted.
8.1.1 When sampling used lubricants, the specimen shall be representative of the system sampled and shall be free of
contamination from
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...