ASTM D4641-17
(Practice)Standard Practice for Calculation of Pore Size Distributions of Catalysts and Catalyst Carriers from Nitrogen Desorption Isotherms
Standard Practice for Calculation of Pore Size Distributions of Catalysts and Catalyst Carriers from Nitrogen Desorption Isotherms
SIGNIFICANCE AND USE
5.1 Pore volume distribution curves obtained from nitrogen sorption isotherms provide one of the best means of characterizing the pore structure in porous catalysts, provided that the limitations of the method are kept in mind. Used in conjunction with the BET treatment for surface area determination (5), these methods provide an indispensable means for studying the structure associated with pores usually important in catalysts. This practice is particularly useful in studying changes in a series of closely related samples caused by treatments, such as heat, compression, or extrusion often used in catalyst manufacturing. Pore volume distribution curves can often provide valuable information during mechanistic studies dealing with catalyst deactivation.
SCOPE
1.1 This practice covers the calculation of pore size distributions for catalysts and catalyst carriers from nitrogen desorption isotherms. The computational procedure is particularly useful for determining how the pore volume is distributed in catalyst samples containing pores whose sizes range from approximately 1.5 to 100 nm (15 to 1000 Å) in radius. It should be used with caution when applied to isotherms for samples containing pores both within this size range and pores larger than 100 nm (1000 Å) in radius. In such instances the isotherms rise steeply near P/Po = 1 and the total pore volume cannot be well defined. The calculations should begin at a point on the isotherm near saturation preferably in a region near P/Po = 0.99, establishing an upper limit on the pore size distribution range to be studied. Simplifications are necessary regarding pore shape. A cylindrical pore model is assumed, and the method treats the pores as non-intersecting, open-ended capillaries which are assumed to function independently of each other during the adsorption or desorption of nitrogen.
Note 1: This practice is designed primarily for manual computation and a few simplifications have been made for this purpose. For computer computation, the simplified expressions may be replaced by exact expressions.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
General Information
- Status
- Published
- Publication Date
- 31-Jan-2017
- Technical Committee
- D32 - Catalysts
- Drafting Committee
- D32.01 - Physical-Chemical Properties
Relations
- Effective Date
- 01-Feb-2017
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Jan-2024
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Mar-2013
- Effective Date
- 01-Apr-2008
- Effective Date
- 01-Apr-2008
- Effective Date
- 15-Nov-2007
- Effective Date
- 01-Oct-2003
- Effective Date
- 10-Nov-2002
- Effective Date
- 28-Nov-1986
- Effective Date
- 01-Feb-2017
Overview
ASTM D4641-17 is the internationally recognized standard practice for the calculation of pore size distributions in catalysts and catalyst carriers using nitrogen desorption isotherms. Published by ASTM International, this standard is instrumental in the characterization of porous materials, specifically catalysts, through the evaluation of their pore structures and distributions. The method focuses on analyzing how pore volume is distributed among pores in the size range of approximately 1.5 to 100 nanometers in radius (15 to 1000 Å), making it especially relevant for mesoporous solids.
The standard provides methodologies for generating pore volume distribution curves, which are critical for understanding catalyst performance, manufacturing variations, and deactivation mechanisms. By combining these calculations with surface area determination using BET methods, ASTM D4641-17 offers a robust framework for comprehensive catalyst characterization.
Key Topics
- Pore Size Distribution Calculation: The core of ASTM D4641-17 details the computational procedures for determining how pore volume is distributed in catalyst samples from nitrogen desorption data. This involves working with experimentally measured isotherms, often using the desorption branch, and assuming a cylindrical pore model.
- Manual and Computer-Based Methods: While the standard is designed with manual calculations in mind, it also allows for adaptation to computer-assisted computations for greater accuracy.
- Applicability and Cautions: The standard is best applied to samples where pore sizes fall within 1.5-100 nm. Materials with larger pores should be analyzed with caution as isotherms may not provide well-defined total pore volumes.
- Limitations: The method assumes non-intersecting, open-ended capillaries with independent functioning during nitrogen adsorption or desorption. It is important to note these assumptions and the impact on precision and bias in results.
- Data Presentation: ASTM D4641-17 provides guidance on reporting, emphasizing both cumulative and differential pore volume distribution plots for detailed material assessment.
Applications
ASTM D4641-17 is widely used in both industrial and research settings where accurate assessment of catalyst pore structures is essential:
- Catalyst Development: The standard enables manufacturers and researchers to evaluate the effects of various treatments-such as heating, compression, or extrusion-on pore distributions. This helps in optimizing catalyst performance for chemical processes.
- Quality Assurance: Routine quality control of catalyst batches ensures that pore structures remain consistent, supporting reliable process outcomes.
- Deactivation and Mechanistic Studies: Understanding changes in pore size distribution helps pinpoint causes of catalyst deactivation and supports the design of more durable materials.
- Materials Comparison: By applying standardized pore size distribution calculations, different batches or types of catalyst carriers can be objectively compared.
Related Standards
Several ASTM standards are referenced or complement ASTM D4641-17:
- ASTM D3766: Terminology Relating to Catalysts and Catalysis
- ASTM D4222: Test Method for Determination of Nitrogen Adsorption and Desorption Isotherms of Catalysts and Catalyst Carriers by Static Volumetric Measurements
Additionally, the practice draws upon well-established approaches such as the BET method for surface area measurement and the Barrett–Joyner–Halenda (BJH) method for traditional pore size distribution analyses.
Practical Value
ASTM D4641-17 remains a fundamental reference for the catalyst and catalyst carrier industries, providing a reliable and reproducible approach for calculating and presenting pore size distributions based on nitrogen desorption isotherms. Its methods deliver crucial insights that support catalyst optimization, performance analysis, and quality control-ultimately facilitating advances in industrial catalysis and porous material science.
Keywords: ASTM D4641-17, pore size distribution, nitrogen desorption isotherms, catalyst carriers, mesoporous materials, surface area, catalyst characterization, BET, BJH, quality control.
Buy Documents
ASTM D4641-17 - Standard Practice for Calculation of Pore Size Distributions of Catalysts and Catalyst Carriers from Nitrogen Desorption Isotherms
REDLINE ASTM D4641-17 - Standard Practice for Calculation of Pore Size Distributions of Catalysts and Catalyst Carriers from Nitrogen Desorption Isotherms
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D4641-17 is a standard published by ASTM International. Its full title is "Standard Practice for Calculation of Pore Size Distributions of Catalysts and Catalyst Carriers from Nitrogen Desorption Isotherms". This standard covers: SIGNIFICANCE AND USE 5.1 Pore volume distribution curves obtained from nitrogen sorption isotherms provide one of the best means of characterizing the pore structure in porous catalysts, provided that the limitations of the method are kept in mind. Used in conjunction with the BET treatment for surface area determination (5), these methods provide an indispensable means for studying the structure associated with pores usually important in catalysts. This practice is particularly useful in studying changes in a series of closely related samples caused by treatments, such as heat, compression, or extrusion often used in catalyst manufacturing. Pore volume distribution curves can often provide valuable information during mechanistic studies dealing with catalyst deactivation. SCOPE 1.1 This practice covers the calculation of pore size distributions for catalysts and catalyst carriers from nitrogen desorption isotherms. The computational procedure is particularly useful for determining how the pore volume is distributed in catalyst samples containing pores whose sizes range from approximately 1.5 to 100 nm (15 to 1000 Å) in radius. It should be used with caution when applied to isotherms for samples containing pores both within this size range and pores larger than 100 nm (1000 Å) in radius. In such instances the isotherms rise steeply near P/Po = 1 and the total pore volume cannot be well defined. The calculations should begin at a point on the isotherm near saturation preferably in a region near P/Po = 0.99, establishing an upper limit on the pore size distribution range to be studied. Simplifications are necessary regarding pore shape. A cylindrical pore model is assumed, and the method treats the pores as non-intersecting, open-ended capillaries which are assumed to function independently of each other during the adsorption or desorption of nitrogen. Note 1: This practice is designed primarily for manual computation and a few simplifications have been made for this purpose. For computer computation, the simplified expressions may be replaced by exact expressions. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
SIGNIFICANCE AND USE 5.1 Pore volume distribution curves obtained from nitrogen sorption isotherms provide one of the best means of characterizing the pore structure in porous catalysts, provided that the limitations of the method are kept in mind. Used in conjunction with the BET treatment for surface area determination (5), these methods provide an indispensable means for studying the structure associated with pores usually important in catalysts. This practice is particularly useful in studying changes in a series of closely related samples caused by treatments, such as heat, compression, or extrusion often used in catalyst manufacturing. Pore volume distribution curves can often provide valuable information during mechanistic studies dealing with catalyst deactivation. SCOPE 1.1 This practice covers the calculation of pore size distributions for catalysts and catalyst carriers from nitrogen desorption isotherms. The computational procedure is particularly useful for determining how the pore volume is distributed in catalyst samples containing pores whose sizes range from approximately 1.5 to 100 nm (15 to 1000 Å) in radius. It should be used with caution when applied to isotherms for samples containing pores both within this size range and pores larger than 100 nm (1000 Å) in radius. In such instances the isotherms rise steeply near P/Po = 1 and the total pore volume cannot be well defined. The calculations should begin at a point on the isotherm near saturation preferably in a region near P/Po = 0.99, establishing an upper limit on the pore size distribution range to be studied. Simplifications are necessary regarding pore shape. A cylindrical pore model is assumed, and the method treats the pores as non-intersecting, open-ended capillaries which are assumed to function independently of each other during the adsorption or desorption of nitrogen. Note 1: This practice is designed primarily for manual computation and a few simplifications have been made for this purpose. For computer computation, the simplified expressions may be replaced by exact expressions. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
ASTM D4641-17 is classified under the following ICS (International Classification for Standards) categories: 71.040.30 - Chemical reagents. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D4641-17 has the following relationships with other standards: It is inter standard links to ASTM D4641-12, ASTM D3766-24a, ASTM D3766-24, ASTM D3766-08(2018), ASTM D3766-08(2013), ASTM D3766-08, ASTM D4222-03(2008), ASTM D3766-86(2007), ASTM D4222-03, ASTM D3766-86(2002), ASTM D3766-86(1997), ASTM D8325-20. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D4641-17 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D4641 − 17
Standard Practice for
Calculation of Pore Size Distributions of Catalysts and
Catalyst Carriers from Nitrogen Desorption Isotherms
This standard is issued under the fixed designation D4641; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2. Referenced Documents
1.1 This practice covers the calculation of pore size distri- 2.1 ASTM Standards:
butions for catalysts and catalyst carriers from nitrogen des- D3766 Terminology Relating to Catalysts and Catalysis
orption isotherms. The computational procedure is particularly D4222 Test Method for Determination of Nitrogen Adsorp-
useful for determining how the pore volume is distributed in tion and Desorption Isotherms of Catalysts and Catalyst
catalyst samples containing pores whose sizes range from Carriers by Static Volumetric Measurements
approximately1.5to100nm(15to1000Å)inradius.Itshould
3. Terminology
be used with caution when applied to isotherms for samples
containing pores both within this size range and pores larger 3.1 Definitions—Consult Terminology D3766.
than 100 nm (1000 Å) in radius. In such instances the
3.2 Symbols:
isotherms rise steeply near P/P = 1 and the total pore volume
o
cannotbewelldefined.Thecalculationsshouldbeginatapoint
ontheisothermnearsaturationpreferablyinaregionnear P/P
i = numerical index representing each successive
o
= 0.99,establishinganupperlimitontheporesizedistribution
data point, with i=1,2… n.
range to be studied. Simplifications are necessary regarding P (i) = pressure after equilibration during desorption,
pore shape. A cylindrical pore model is assumed, and the torr.
P (i) = liquid nitrogen vapor pressure, torr.
method treats the pores as non-intersecting, open-ended capil-
V = Quantity of gas desorbed (cm3 STP/g); see
laries which are assumed to function independently of each
de
12.4.10 and 12.5 in Test Method D4222.
other during the adsorption or desorption of nitrogen.
r (i) = radius of inner core calculated from Kelvin
k
NOTE 1—This practice is designed primarily for manual computation
equation, Å.
and a few simplifications have been made for this purpose. For computer
T = boiling point of nitrogen, K.
computation, the simplified expressions may be replaced by exact expres-
V = liquid nitrogen molar volume at T,cm /mole.
sions. L
γ = liquid nitrogen surface tension at T, mN/m.
1.2 The values stated in SI units are to be regarded as
t(i) = average thickness of the nitrogen film adsorbed
standard. No other units of measurement are included in this
on the pore walls, Å.
standard.
r (i) = radius of cylindrical pore given by r (i)+ t(i), Å.
p k
1.3 This standard does not purport to address all of the Q = volume correction factor defined as (r¯ /r¯ ) .
p k
∆V (i) = decrease in the amount of nitrogen adsorbed
safety concerns, if any, associated with its use. It is the
T
caused by a lowering in relative pressure, mm /g.
responsibility of the user of this standard to establish appro-
∆V(i) = volume of liquid nitrogen desorbed from pore
priate safety and health practices and determine the applica- f
walls during thinning of the film, mm /g.
bility of regulatory limitations prior to use.
∆V (i) = liquid volume of the inner core in which capillary
k
condensation of the nitrogen occurs, mm /g.
This practice is under the jurisdiction of ASTM Committee D32 on Catalysts
and is the direct responsibility of Subcommittee D32.01 on Physical-Chemical
Properties. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Feb. 1, 2017. Published February 2017. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1987. Last previous edition approved in 2012 as D4641–12. DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/D4641-17. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4641 − 17
This practice is particularly useful in studying changes in a
∆V (i) = liquid volume contained in a group of pores
p
series of closely related samples caused by treatments, such as
having mean radius r¯ ,mm /g.
p
heat, compression, or extrusion often used in catalyst manu-
∑∆v = cumulative pore volume, mm /g.
p
∆S ( i ) = areaoftheporewallsofacylinderhavingvolume facturing. Pore volume distribution curves can often provide
p
valuable information during mechanistic studies dealing with
∆ V,m /g.
p
catalyst deactivation.
4. Summary of Practice
6. Computational Procedure
4.1 The pore size distribution is determined by analyzing
6.1 This procedure requires the use of a series of experi-
the desorption data of the nitrogen isotherm. The nitrogen
mentally measured relative pressures [P (i)/P (i)] and the
4 0
uptake is caused by the multilayer adsorption of a film of
corresponding quantities of nitrogen gas adsorbed [V ] ex-
de
nitrogen on the pore walls and by capillary condensation of the
pressed in units of cm STP/g. The experimental data required
nitrogen in the “inner core” regions of the pores. The relative
in the use of this procedure can be measured by following the
pressureatwhichfillingofthecoreoccursforagivenporesize
steps outlined in Test Method D4222. Inspect the nitrogen
by capillary condensation is predicted from the Kelvin equa-
3 sorption isotherm in the region above P/P = 0.95. If the solid
o
tion (1). During desorption, thinning of the multilayer film
contains no pores larger than 100 nm (1000 Å) radius, the
adsorbed on the pore walls occurs in pores which have
isotherm remains nearly horizontal over a range of P/P
o
previously lost their capillary condensate. Corrections for film
approaching unity and it is a simple matter to select a starting
thinning are determined by a procedure involving the surface
relative pressure within this region, establishing an upper limit
area and radius of the film which becomes exposed as
ontheporesizerangetobestudied.Ifporeslargerthan100nm
desorption proceeds. In principle, the computational procedure
(1000 Å) are present, however, the isotherm rises rapidly near
can be applied to either the adsorption branch or desorption
P/P = 1 and the total pore volume cannot be well defined.
O
branch of the nitrogen isotherm. Unless the presence of
This limiting adsorption can then be identified reliably only if
ink-bottleshapedporesissuggestedbyanabruptclosureofthe
the temperature is very carefully controlled and there are no
desorption branch on the adsorption branch, the distribution
“cold spots” in the apparatus (which lead to bulk condensation
curve derived from the desorption data is preferred, and is
of the gas and a false measure of the adsorption in the
described in this procedure. The computational method is
volumetric method). Selecting the starting relative pressure for
essentially the procedure developed by Barrett, Joyner, and
the computational procedure is then made more difficult. In
Halenda (2), except for the incorporation of a few simplifica-
most cases a starting relative pressure of 0.99 will be suitable,
tions.
which corresponds to an upper limit on pore size of 100 nm
NOTE 2—In cases where it has been established that the adsorption
(1000Å)inradius.Ifnecessary,interpolatethevaluesof V to
de
branch of the nitrogen isotherm is to be analyzed, the procedure proposed
determine the quantity of nitrogen gas adsorbed at the chosen
by Cranston and Inkley (3) can be employed.
starting relative pressure.
NOTE 3—Thanks to major advances in adsorption science and technol-
ogy over the past two decades, it is now widely recognized (see recent
6.2 The procedure requires numerous arithmetical steps
IUPAC recommendations (4)) that modern statistical mechanics methods
which can best be carried out with the aid of a work sheet.An
based on Density Functional Theory or Monte Carlo simulations provide
example (5) of a form found useful in the calculations is
significantly more accurate pore size distributions than classical proce-
provided in Table 1. List in descending order the experimen-
dures based on the Kelvin equation, such as the Barrett–Joyner–Halenda
(BJH) or Cranston-Inkley methods (2, 3). Moreover, the choice of tally determined relative pressures [P (i)/P (I)] in Column 1,
4 0
desorption (equilibrium) vs. adsorption (metastable) branches for reliable
beginning with the value chosen as the starting relative
pore size analysis must take into consideration the potential influence of
pressure. Generally, values below a relative pressure of 0.25
pore connectivity, tensile strength and cavitation effects. In addition,
will not be required in the calculations. Convert the uptake
although nitrogen sorption at 77 K is widely used, its quadrupole
values into a liquid volume (mm /g) by multiplying the value
interactions with polar surfaces can influence isotherm shapes and their
interpretation;therefore,argonadsorptionat87Kisconsideredtobemore
of V in cm STP/g with the conversion factor 1.5468 derived
de
reliable and is now recommended, particularly for samples containing
from V = 34.67cm /mole.ListinColumn9thecorresponding
L
micropores (4). In spite of these advances, the traditional Kelvin-based
quantities of nitrogen adsorbed.
approaches described in this Standard Practice are still deemed to be
useful for routine work (such as industrial process control). 6.3 For each relative pressure, calculate a value for the
radius of the core, r , by means of the Kelvin equation,
k
5. Significance and Use
2γV
L
RTln~P /P !52 (1)
5.1 Pore volume distribution curves obtained from nitrogen
4 0
r
k
sorption isotherms provide one of the best means of character-
given in the form
izing the pore structure in porous catalysts, provided that the
limitationsofthemethodarekeptinmind.Usedinconjunction
9.574
˚
r ~A!52 (2)
k
with the BET treatment for surface area determination (5), ln P /P
~ !
4 0
these methods provide an indispensable means for studying the
with T = 77.35K; γ = 8.88mN/m;and V = 34.67cm /mole.
L
structure associated with pores usually important in catalysts.
List the values in Column 2. For each successive decrement in
relative pressure, calculate r¯ , the mean of the values of r for
k k
the present and previous pressures, and list these mean values
The boldface numbers in parentheses refer to a list of references at the end of
this standard. in Column 3.
D4641 − 17
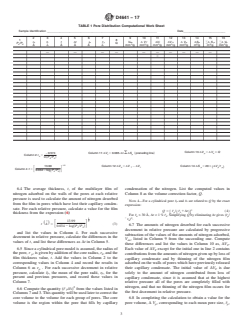
TABLE 1 Pore Distribution Computational Work Sheet
Sample Identification ____________________________________________________________________ Date__________________________
2 3 4 5 6 7 9 10 11 12 13 14 15 16
1 8
r r¯ t ∆t r r¯ V ∆ V ∆V ∆V ∆ V ∆S ^∆ S ^∆ V
k k p p de T f k p p p p
P /P Q
4 0 3 3 3 3 3 2 2 3
Å Å Å Å Å Å mm /g mm /g mm /g mm /g mm /g m /g m /g mm /g
— — — — — —————
C
Column 13:∆V 5 ∆V 3 Q
Column 11:∆V 5 0.085·∆t· ∆S preceding line
29.574 s d p k
f o p
Column 2:r 5
k
lnsP /P d
4 0
1/2
13.99 Column 12:∆V 5 ∆V 2 ∆V Column 14:∆S 5 203 ∆V /r¯
s d
k T f p p p
Column 4:t5
F G
0.0342 logsP /P d
4 0
6.4 The average thickness, t, of the multilayer film of condensation of the nitrogen. List the computed values in
nitrogen adsorbed on the walls of the pores at each relative Column 8 as the volume correction factor, Q.
pressure is used to calculate the amount of nitrogen desorbed
NOTE 4—For a cylindrical pore r¯ and r¯ are related to Q by the exact
p k
from the film in pores which have lost their capillary conden-
expression:
sate. For each relative pressure, calculate a value for the film
Q 5 r¯ / r¯ 1∆t (4)
@ ~ !#
p k
thickness from the expression (6)
For r¯ >30Å, ∆t<1% r¯ . Simplifying Q by eliminating ∆t gives (r¯ /
k k p
¯r ) .
k
13.99 2
˚
~ !
t A 5 (3)
F G
6.7 The amounts of nitrogen desorbed for each successive
0.034 2 log P /P
~ !
4 0
decrement in relative pressure are calculated by progressive
and list the values in Column 4. For each successive
subtraction of the values of the amounts of nitrogen adsorbed,
decrement in relative pressure, calculate the differences in the
V , listed in Column 9 from the succeeding one. Compute
de
values of t, and list these differences as ∆t in Column 5.
these differences and list the values in Column 10 as, ∆V .
T
6.5 Since a cylindrical pore model is assumed, the radius of
Each value of ∆V except for the initial one in line 2 contains
T
the pore, r , is given by addition of the core radius, r , and the
contributions from the amounts of nitrogen given up by loss of
p k
film thickness value, t. Add the values in Column 2 to the
capillary condensate and by thinning of the nitrogen film
corresponding values in Column 4 and record the results in
adsorbed on the walls of pores which have previously released
Column 6 as r . For each successive decrement in relative
their capillary condensate. The initial value of ∆V is due
p
T
pressure, calculate r¯ , the mean of
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D4641 − 12 D4641 − 17
Standard Practice for
Calculation of Pore Size Distributions of Catalysts and
Catalyst Carriers from Nitrogen Desorption Isotherms
This standard is issued under the fixed designation D4641; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This practice covers the calculation of pore size distributions for catalysts and catalyst carriers from nitrogen desorption
isotherms. The computational procedure is particularly useful for determining how the pore volume is distributed in catalyst
samples containing pores whose sizes range from approximately 1.5 to 100 nm (15 to 1000 Å) in radius. It should be used with
caution when applied to isotherms for samples containing pores both within this size range and pores larger than 100 nm (1000
Å) in radius. In such instances the isotherms rise steeply near P/P = 1 and the total pore volume cannot be well defined. The
o
calculations should be begun begin at a point on the isotherm near saturation preferably in a region near P/P = 0.99, establishing
o
an upper limit on the pore size distribution range to be studied. Simplifications are necessary regarding pore shape. A cylindrical
pore model is assumed, and the method treats the pores as non-intersecting, open-ended capillaries which are assumed to function
independently of each other during the adsorption or desorption of nitrogen.
NOTE 1—This practice is designed primarily for manual computation and a few simplifications have been made for this purpose. For computer
computation, the simplified expressions may be replaced by exact expressions.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory
limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:
D3766 Terminology Relating to Catalysts and Catalysis
D4222 Test Method for Determination of Nitrogen Adsorption and Desorption Isotherms of Catalysts and Catalyst Carriers by
Static Volumetric Measurements
3. Terminology
3.1 Definitions—Consult Terminology D3766.
3.2 Symbols:
i = numerical index representing each successive data point, with i = 1, 2… n.
P (i) = pressure after equilibration during desorption, torr.
P (i) = liquid nitrogen vapor pressure, torr.
V = Quantity of gas desorbed (cm3 STP/g); see 12.4.10 and 12.5 in Test Method D4222.
de
r (i) = radius of inner core calculated from Kelvin equation, Å.
k
T = boiling point of nitrogen, K.
V = liquid nitrogen molar volume at T, cm /mole.
L
γ = liquid nitrogen surface tension at T, mN/m.
T(i) = average thickness of the nitrogen film adsorbed on the pore walls, Å.
This practice is under the jurisdiction of ASTM Committee D32 on Catalysts and is the direct responsibility of Subcommittee D32.01 on Physical-Chemical Properties.
Current edition approved May 1, 2012Feb. 1, 2017. Published July 2012February 2017. Originally approved in 1987. Last previous edition approved in 20062012 as
D4641–94(2006).D4641–12. DOI: 10.1520/D4641-12.10.1520/D4641-17.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D4641 − 17
t(i) = average thickness of the nitrogen film adsorbed on the pore walls, Å.
r (i) = radius of cylindrical pore given by r (i) + t(i), Å.
p k
Q = volume correction factor defined as (r¯ /r¯ ) .
p k
ΔV (i) = decrease in the amount of nitrogen adsorbed caused by a lowering in relative pressure, mm /g.
T
ΔV (i) = volume of liquid nitrogen desorbed from pore walls during thinning of the film, mm /g.
f
ΔV (i) = liquid volume of the inner core in which capillary condensation of the nitrogen occurs, mm /g.
k
ΔV (i) = liquid volume contained in a group of pores having mean radius r¯ , mm /g.
p p
∑Δv = cumulative pore volume, mm /g.
p
ΔS (i ) = area of the pore walls of a cylinder having volume Δ V , m /g.
p p
4. Summary of Practice
4.1 The pore size distribution is determined by analyzing the desorption data of the nitrogen isotherm. The nitrogen uptake is
caused by the multilayer adsorption of a film of nitrogen on the pore walls and by capillary condensation of the nitrogen in the
“inner core” regions of the pores. The relative pressure at which filling of the core occurs for a given pore size by capillary
condensation is predicted from the Kelvin equation (1). During desorption, thinning of the multilayer film adsorbed on the pore
walls occurs in pores which have previously lost their capillary condensate. Corrections for film thinning are determined by a
procedure involving the surface area and radius of the film which becomes exposed as desorption proceeds. In principle, the
computational procedure can be applied to either the adsorption branch or desorption branch of the nitrogen isotherm. Unless the
presence of ink-bottle shaped pores is suggested by an abrupt closure of the desorption branch on the adsorption branch, the
distribution curve derived from the desorption data is preferred, and is described in this procedure. The computational method is
essentially the procedure developed by Barrett, Joyner, and Halenda (2)), except for the incorporation of a few simplifications.
NOTE 2—In cases where it has been established that the adsorption branch of the nitrogen isotherm is to be analyzed, the procedure proposed by
Cranston and Inkley (3) is recommended. can be employed.
NOTE 3—Thanks to major advances in adsorption science and technology over the past two decades, it is now widely recognized (see recent IUPAC
recommendations (4)) that modern statistical mechanics methods based on Density Functional Theory or Monte Carlo simulations provide significantly
more accurate pore size distributions than classical procedures based on the Kelvin equation, such as the Barrett–Joyner–Halenda (BJH) or
Cranston-Inkley methods (2, 3). Moreover, the choice of desorption (equilibrium) vs. adsorption (metastable) branches for reliable pore size analysis must
take into consideration the potential influence of pore connectivity, tensile strength and cavitation effects. In addition, although nitrogen sorption at 77
K is widely used, its quadrupole interactions with polar surfaces can influence isotherm shapes and their interpretation; therefore, argon adsorption at 87
K is considered to be more reliable and is now recommended, particularly for samples containing micropores (4). In spite of these advances, the traditional
Kelvin-based approaches described in this Standard Practice are still deemed to be useful for routine work (such as industrial process control).
5. Significance and Use
5.1 Pore volume distribution curves obtained from nitrogen sorption isotherms provide one of the best means of characterizing
the pore structure in porous catalysts, provided that the limitations of the method are kept in mind. Used in conjunction with the
BET treatment for surface area determination (45), these methods provide an indispensable means for studying the structure
associated with pores usually important in catalysts. This practice is particularly useful in studying changes in a series of closely
related samples caused by treatments, such as heat, compression, or extrusion often used in catalyst manufacturing. Pore volume
distribution curves can often provide valuable information during mechanistic studies dealing with catalyst deactivation.
6. Computational Procedure
6.1 This procedure requires the use of a series of experimentally measured relative pressures [P (i)/P (i)] and the corresponding
4 0
quantities of nitrogen gas adsorbed [V ] expressed in units of cm STP/g. The experimental data required in the use of this
de
procedure can be measured by following the steps outlined in Test Method D4222. Inspect the nitrogen sorption isotherm in the
region above P/P = 0.95. If the solid contains no pores larger than 100 nm (1000 Å) radius, the isotherm remains nearly horizontal
o
over a range of P/P approaching unity and it is a simple matter to select a starting relative pressure within this region, establishing
o
an upper limit on the pore size range to be studied. If pores larger than 100 nm (1000 Å) are present, however, the isotherm rises
rapidly near P/P = 1 and the total pore volume cannot be well defined. This limiting adsorption can then be identified reliably
O
only if the temperature is very carefully controlled and there are no “cold spots” in the apparatus (which lead to bulk condensation
of the gas and a false measure of the adsorption in the volumetric method). Selecting the starting relative pressure for the
computational procedure is then made more difficult. In most cases a starting relative pressure of 0.99 will be suitable, which
corresponds to an upper limit on pore size of 100 nm (1000 Å) in radius. If necessary, interpolate the values of V to determine
de
the quantity of nitrogen gas adsorbed at the chosen starting relative pressure.
6.2 The procedure requires numerous arithmetical steps which can best be carried out with the aid of a work sheet. An example
(45) of a form found useful in the calculations is provided in Table 1. List in descending order the experimentally determined
relative pressures [P (i)/P (iI)] in Column 1, beginning with the value chosen as the starting relative pressure. Generally, values
4 0
below a relative pressure of 0.25 will not be required in the calculations. Convert the uptake values into a liquid volume (mm /g)
The boldface numbers in parentheses refer to a list of references at the end of this standard.
D4641 − 17
TABLE 1 Pore Distribution Computational Work Sheet
Sample Identification ____________________________________________________________________ Date__________________________
2 3 4 5 6 7 9 10 11 12 13 14 15 16
1 8
r r¯ t Δt r r¯ V Δ V ΔV ΔV Δ V ΔS ^Δ S ^Δ V
k k p p de T f k p p p p
P /P Q
4 0 3 3 3 3 3 2 2 3
Å Å Å Å Å Å mm /g mm /g mm /g mm /g mm /g m /g m /g mm /g
— — — — — — — — — —
C
Column 13:ΔV 5ΔV 3Q
29.574 Column 11:ΔV 50.085·Δt· ΔS spreceding lined p k
f o p
Column 2:r 5
k
lnsP /P d
4 0
1/2
13.99 Column 12:ΔV 5ΔV 2ΔV Column 14:ΔS 520 3 ΔV /r¯
k T f p s p pd
Column 4:t 5
F G
0.034 2log P /P
s d
4 0
3 3
by multiplying the value of V in cm STP/g with the conversion factor 1.5468 derived from V = 34.67 cm /mole. List in Column
de L
9 the corresponding quantities of nitrogen adsorbed.
6.3 For each relative pressure, calculate a value for the radius of the core, r , by means of the Kelvin equation,
k
2γV
L
RTln P /P 52 (1)
~ !
4 0
r
k
given in the form
9.574
˚
r ~A! 52 (2)
k
ln~P /P !
4 0
with T = 77.35 K; γ = 8.88 mN/m; and V = 34.67 cm /mole. List the values in Column 2. For each successive decrement in
L
relative pressure, calculate r¯ , the mean of the values of r for the present and previous pressures, and list these mean values in
k k
Column 3.
6.4 The average thickness, t, of the multilayer film of nitrogen adsorbed on the walls of the pores at each relative pressure is
used to calculate the amount of nitrogen desorbed from the film in pores which have lost their capillary condensate. For each
relative pressure, calculate a value for the film thickness from the expression (56)
13.99 2
˚
t ~A! 5 (3)
F G
0.034 2 log~P /P !
4 0
and list the values in Column 4. For each successive decrement in relative pressure, calculate the differences in the values of
t, and list these differences as Δt in Column 5.
6.5 Since a cylindrical pore model is assumed, the radius of the pore, r , is given by addition of the core radius, r , and the
p k
film thickness value, t. Add the values in Column 2 to the corresponding values in Column 4 and record the results in Column 6
D4641 − 17
as r . For each successive decrement in relative pressure, calculate r¯ , the mean of the pore radii, r , for the present and previous
p p p
pressures, and record these values in Column 7.
6.6 Compute the quantity (r¯ /r¯ ) from the values listed in Columns 7 and 3. This quantity will be used later to correct the
p k
core volume to the volume for each group of pores. The core volume is the region within the pore that fills by capillary
condensation of the nitrogen. List the computed values in Column 8 as the volume correction factor, Q.
NOTE 4—For a cylindrical pore r¯ and r¯ are related to Q by the exact expression:
p k
Q 5 @ r¯ /~ r¯ 1Δt!# (4)
p k
For r¯ > 30 Å, Δt < 1 % r¯ . Simplifying Q by eliminating Δt gives (r¯ / ¯ r ) .
k k p k
6.7 The amounts of nitrogen desorbed for each successive decrement in rel
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...