ASTM E788-97(2019)
(Specification)Standard Specification for Pipet, Blood Diluting
Standard Specification for Pipet, Blood Diluting
ABSTRACT
This specification covers the requirements for glass reusable blood diluting pipets that are used for performing red and white cell corpuscle determinations. The pipets shall be made of common spirit bore white back tubing or clear glass with a white stripe applied to the outer surface of the tubing. The beads shall be made of glass or ceramic composition and shall be of red, clear, or white coloring. Materials shall be tested and the individual types shall conform to specified values of design, capacity, capacity markings, graduation lines, accuracy, workmanship, identification, and pigmentation.
SCOPE
1.1 This specification covers requirements for glass reusable blood diluting pipets that are used for performing red and white cell corpuscle determinations.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Jun-2019
- Technical Committee
- E41 - Laboratory Apparatus
- Drafting Committee
- E41.01 - Laboratory Ware and Supplies
Relations
- Effective Date
- 01-Jul-2019
- Effective Date
- 01-Feb-2024
- Effective Date
- 01-Jul-2019
- Effective Date
- 01-Jan-2018
- Effective Date
- 01-Nov-2013
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Dec-2011
- Effective Date
- 01-Sep-2010
- Effective Date
- 01-Jul-2010
- Effective Date
- 15-May-2010
- Effective Date
- 01-Nov-2008
- Effective Date
- 01-Nov-2006
- Effective Date
- 01-Oct-2005
- Effective Date
- 01-May-2005
- Effective Date
- 01-Jan-2000
Overview
ASTM E788-97(2019): Standard Specification for Pipet, Blood Diluting establishes the requirements for reusable glass blood diluting pipets specifically designed for red and white blood cell corpuscle determinations. Published by ASTM International, this standard ensures that pipets used in clinical and laboratory applications meet stringent guidelines for material composition, dimensional accuracy, marking, and overall quality. Blood diluting pipets compliant with ASTM E788 are essential tools in medical laboratories for precise blood sample analysis, contributing to reliable hematological diagnostics.
Key Topics
- Material Specifications: Pipets must be manufactured from spirit bore white back glass tubing or clear glass with a white stripe for visibility and identification. Sealed beads used for marking are made from glass or ceramic in red, clear, or white colors to differentiate between red and white cell pipets.
- Design and Construction: The standard covers both single-piece and three-piece fused pipet designs, with precise dimensional tolerances for length, bore size, and tip diameter to ensure consistent sample handling.
- Graduation and Capacity Markings: Pipets must feature clearly defined graduation lines, accurately calibrated for both bulb and stem capacities. These markings are fused in dark amber or black colors for legibility and longevity.
- Accuracy and Workmanship: Strict accuracy requirements are outlined, including capacity tolerances and volumetric ratios for both red and white cell pipets. Rigorous workmanship standards require pipets to be free of imperfections and strain, with permanent, durable markings that withstand cleaning and use.
- Identification: Each pipet must display the manufacturer’s name or registered trademark and include a color-coded bead (red for red cell pipets, white or clear for white cell pipets) for easy identification in the laboratory setting.
- Testing and Packaging: The document defines testing procedures for accuracy, strain-free construction, and color durability. Packaging must comply with relevant ASTM specifications for laboratory glassware.
Applications
ASTM E788-compliant blood diluting pipets are critical in the following areas:
- Hematology Laboratories: Used for red and white blood cell counts in manual microscopy and diagnostic procedures.
- Clinical Diagnostics: Essential for preparing diluted blood samples for accurate cell corpuscle determination in medical testing.
- Educational and Research Institutions: Support laboratory training, method validation, and scientific studies requiring consistent, high-precision blood sampling.
- Quality Control: Ensures consistency and accuracy in sample preparation processes, reducing variability and supporting regulatory compliance.
By adhering to ASTM E788, laboratories ensure that their manual blood dilution instruments offer traceable performance, safety, and compatibility with standardized operating procedures.
Related Standards
- ASTM E694: Specification for Laboratory Glass Volumetric Apparatus
- ASTM E920: Specification for Commercially Packaged Laboratory Apparatus
- ASTM E921: Specification for Export Packaged Laboratory Apparatus
- ASTM E1133: Practice for Performance Testing of Packaged Laboratory Apparatus
- ASTM E1157: Specification for Sampling and Testing of Reusable Laboratory Glassware
These related ASTM standards provide additional guidance on laboratory glassware materials, packaging, testing, and performance – supporting integrated quality assurance in laboratory environments.
Keywords: ASTM E788, blood diluting pipet, glass pipets, laboratory apparatus, red cell pipet, white cell pipet, hematology, reusable pipet standard, laboratory glassware standards, medical diagnostics
Buy Documents
ASTM E788-97(2019) - Standard Specification for Pipet, Blood Diluting
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM E788-97(2019) is a technical specification published by ASTM International. Its full title is "Standard Specification for Pipet, Blood Diluting". This standard covers: ABSTRACT This specification covers the requirements for glass reusable blood diluting pipets that are used for performing red and white cell corpuscle determinations. The pipets shall be made of common spirit bore white back tubing or clear glass with a white stripe applied to the outer surface of the tubing. The beads shall be made of glass or ceramic composition and shall be of red, clear, or white coloring. Materials shall be tested and the individual types shall conform to specified values of design, capacity, capacity markings, graduation lines, accuracy, workmanship, identification, and pigmentation. SCOPE 1.1 This specification covers requirements for glass reusable blood diluting pipets that are used for performing red and white cell corpuscle determinations. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ABSTRACT This specification covers the requirements for glass reusable blood diluting pipets that are used for performing red and white cell corpuscle determinations. The pipets shall be made of common spirit bore white back tubing or clear glass with a white stripe applied to the outer surface of the tubing. The beads shall be made of glass or ceramic composition and shall be of red, clear, or white coloring. Materials shall be tested and the individual types shall conform to specified values of design, capacity, capacity markings, graduation lines, accuracy, workmanship, identification, and pigmentation. SCOPE 1.1 This specification covers requirements for glass reusable blood diluting pipets that are used for performing red and white cell corpuscle determinations. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM E788-97(2019) is classified under the following ICS (International Classification for Standards) categories: 11.100 - Laboratory medicine. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM E788-97(2019) has the following relationships with other standards: It is inter standard links to ASTM E788-97(2013), ASTM E694-18(2024), ASTM E920-97(2019), ASTM E694-18, ASTM E920-97(2013), ASTM E1157-87(2018)e1, ASTM E1157-87(2011), ASTM E1133-86(2010), ASTM E694-99(2010), ASTM E921-97(2010), ASTM E920-97(2008), ASTM E1157-87(2006), ASTM E1133-86(2005), ASTM E694-99(2005), ASTM E1133-86(1996)e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM E788-97(2019) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: E788 −97 (Reapproved 2019)
Standard Specification for
Pipet, Blood Diluting
This standard is issued under the fixed designation E788; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
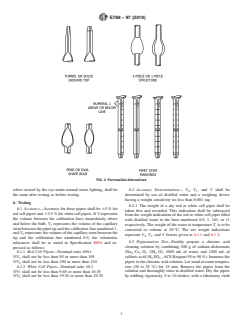
1. Scope 4. Materials
1.1 Thisspecificationcoversrequirementsforglassreusable 4.1 The pipets shall be made of common spirit bore white
blooddilutingpipetsthatareusedforperformingredandwhite back tubing or clear glass with a white stripe applied to the
cell corpuscle determinations. outer surface of the tubing.
1.2 The values stated in SI units are to be regarded as 4.2 The beads shall be made of glass or ceramic composi-
standard. No other units of measurement are included in this tion and shall be of red, clear, or white coloring.
standard.
5. Dimensions and Permissible Variations
1.3 This international standard was developed in accor-
5.1 Design—The red and white cell blood dilution pipets
dance with internationally recognized principles on standard-
shall consist of a small uniform bore glass tube which shall
ization established in the Decision on Principles for the
have a bulb of proper size near the proximal end (see Fig. 1).
Development of International Standards, Guides and Recom-
As an alternative, the pipets may be constructed of three pieces
mendations issued by the World Trade Organization Technical
of glass that are fused together to form a one-piece pipet (see
Barriers to Trade (TBT) Committee.
Fig. 2). The bulb shall contain a nonspherical glass or ceramic
bead sufficiently large enough to prevent its being impacted in
2. Referenced Documents
the constriction portion of the bulb. The cross section of the
2.1 ASTM Standards:
pipet at any point shall be circular. The distal end of the white
E694 Specification for Laboratory Glass Volumetric Appa-
cell pipet shall be pulled to a point and then ground and
ratus
polished to a tapered tip. The distal end of the red cell pipet
E920 Specification for Commercially Packaged Laboratory
shall be ground and polished to a tapered tip. The inside
Apparatus
diameter (ID) of the pipet bore at the tip end of the white cell
E921 Specification for Export Packaged Laboratory Appa-
pipet shall be 0.2 to 0.5 mm. The external diameter of the
ratus
ground and polished tip shall not exceed 2.0 mm. The overall
E1133 Practice for Performance Testing of Packaged Labo-
lengthofbothpipetsshallbe104to121mm.Theproximalend
ratory Apparatus for United States Government Procure-
may be of funnel design, with an exterior taper, or ground and
ments
polishedwithataperofsufficientangletopermitapplicationof
E1157 Specification for Sampling and Testing of Reusable
a rubber suction tube or other suction device (see Fig. 2).
Laboratory Glassware
5.2 Capacity—The capacity of the red cell pipet bulb shall
3. Classification be 0.8 to 1.2 cm and the capacity of the white cell bulb shall be
0.2 to 0.4 cm . The capacity of the red cell pipet stem (bulb to
3.1 This specification covers two different pipet designs and
distal end) shall be 0.008 to 0.012 cm and the capacity of the
permissible alternatives.
white cell pipet stem shall be 0.02 to 0.04 cm .
3.1.1 Red and white cell blood diluting pipets (see Fig. 1).
3.1.2 Permissible alternative designs (see Fig. 2). 5.3 Capacity Markings:
5.3.1 Pipet Bulb Markings—The red and white cell pipet
bulb capacity shall be confined to and indicated by calibration
This specification is under the jurisdiction of ASTM Committee E41 on
lines on the pipet. These lines shall be located on both sides of
Laboratory Apparatus and is the direct responsibility of Subcommittee E41.01 on
the bulb and shall be within 2 mm to 6 mm above the bulb
Laboratory Ware and Supplies.
(proximalend)andwithin3mmto6mmbelowthebulb(distal
Current edition approved July 1, 2019. Published August 2019. Originally
end). The exact manner for measuring these line placements is
approvedin1981.Lastpreviouseditionapprovedin2013asE788 – 97(2013).DOI:
10.1520/E0788-97R19.
specified in Fig. 1.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
5.3.2 Pipet Stem Markings—The graduation lines on the
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
stem of the red and white cell pipets shall be equally divided
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website. into two or ten divisions. If there are two divisions, the two
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
E788 − 97 (2019)
FIG. 1 Pipet Dimensions
calibration lines on the stem will be numbered 0.5 and 1. If 5.6.2 Construction shall be such that the mechanical
there are ten divisions, the ten calibration lines will be strength is provided to withstand the rigors of normal use. The
numbered 0.5 on the fifth line and 1 on the tenth line pipets shall be free of strain when tested in accordance with
respectively. The top line (above bulb at the proximal end)
6.4.
shall be numbered 101 on the red cell pipet and 11 on the white
5.7 Identification—Each pipet shall have the name or regis-
cell pi
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...