ASTM D5197-21
(Test Method)Standard Test Method for Determination of Formaldehyde and Other Carbonyl Compounds in Air (Active Sampler Methodology)
Standard Test Method for Determination of Formaldehyde and Other Carbonyl Compounds in Air (Active Sampler Methodology)
SIGNIFICANCE AND USE
5.1 This test method provides an analytical procedure for measuring formaldehyde and other carbonyl compounds in indoor, workplace, ambient air or for emission testing.
SCOPE
1.1 This test method presents a procedure for the determination of formaldehyde (HCHO) and other carbonyl compounds (aldehydes and ketones) in air. Other carbonyl compounds that have been successfully quantified by this method include acetaldehyde, acetone, propanal (propionaldehyde), 2-butanone (methyl ethyl ketone), butyraldehyde, benzaldehyde, isovaleraldehyde, valeraldehyde, o-tolualdehyde, m-tolualdehyde, p-tolualdehyde, hexanal, and 2,5-dimethylbenzaldehyde.
1.2 This test method involves drawing air through a cartridge containing silica gel coated with 2,4-dinitrophenylhydrazine (DNPH) reagent. Carbonyl compounds readily form stable derivatives with the acidified DNPH reagent. The DNPH derivatives are analyzed for parent aldehydes and ketones using high performance liquid chromatography (HPLC) or ultra-high performance liquid chromatography (UHPLC). UHPLC systems use higher pressures and smaller particle sizes in columns compared to HPLC systems. The sampling procedure is a modification of U.S. EPA Method TO-11A (see 2.2).
1.3 This test method is based on the reaction of carbonyl compounds with DNPH in the presence of an acid to form stable derivatives according to the reaction shown in Fig. 1, (where: both R and R1 are alkyl or aromatic groups (ketones), or either, or both R or R1 is a hydrogen atom (aldehydes)). The determination of formaldehyde and other carbonyl compounds, as DNPH derivatives, is similar to that of U.S. EPA Method TO-11A in that it uses HPLC or UHPLC for separation of carbonyl compounds followed by UV adsorption or photodiode array detection. This test method exceeds the stated applicability of TO-11A to include other carbonyl compounds that can be determined as stated in 10.2.4. This test method is suitable for determination of formaldehyde and other carbonyl compounds in the airborne concentration range from approximately 10 ppbv/v (12 μg/m3), requires sampling for 1 h at 1 L/min) to 1 ppmv/v (1.2 mg/m3). Lower concentrations in air may be determined using higher sampling volume and with control of contamination, appropriate selection of flow rate and sampling duration.
FIG. 1 Reaction of Carbonyl Compounds
1.4 The sampling method gives a time-weighted average (TWA) sample. It can be used for long-term (1 to 24 h) or short-term (5 to 60 min) sampling of air for formaldehyde. Shorter sampling times or low flow rates will result in higher detection limits and may result in greater variation in co-located sampler results. Tests should be performed over a duration and a flow rate that allows the data quality objective of the project to be achieved. Sample times for other carbonyls, such as acetaldehyde, may be limited to short term (1).2 The data provides total concentrations of carbonyl compounds from which time weighted average concentrations can be calculated.
1.5 This test method instructs the user on how to prepare sampling cartridges from commercially available chromatographic grade silica gel cartridges3 by the application of acidified DNPH to each cartridge.
1.6 The sampling flow rate, as described in this test method, has been validated for sampling rates up to 1.5 L/min for formaldehyde. This flow rate limitation is principally due to the high pressure drop (>8 kPa at 1.0 L/min) across user prepared silica gel cartridges which have a particle size of 55 to 105 µm. These cartridges are not generally compatible with battery-powered pumps used in personal sampling equipment (for example, those used by industrial hygienists).
1.7 Alternatively, pre-coated DNPH silica gel cartridges are commercially available and may be substituted provided they can be demonstrated to meet blank and analyte trapping acceptance criteria (2). Some of these use silica gel of a larger parti...
General Information
- Status
- Published
- Publication Date
- 31-Aug-2021
- Technical Committee
- D22 - Air Quality
- Drafting Committee
- D22.05 - Indoor Air
Relations
- Effective Date
- 01-Sep-2020
- Effective Date
- 15-Mar-2020
- Effective Date
- 01-Mar-2020
- Effective Date
- 01-Sep-2019
- Effective Date
- 01-Mar-2017
- Effective Date
- 15-Oct-2015
- Effective Date
- 01-Jul-2015
- Effective Date
- 01-Dec-2014
- Effective Date
- 01-May-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-Apr-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Apr-2010
- Effective Date
- 15-Dec-2008
Overview
ASTM D5197-21 is an internationally recognized standard test method that defines procedures for the determination of formaldehyde and other carbonyl compounds in air using active sampling methodology. Developed by ASTM International, this method enables the reliable measurement of airborne formaldehyde, aldehydes, and ketones in various environments, including indoor, workplace, ambient, and emission-testing contexts. The technique employs solid-phase extraction cartridges coated with 2,4-dinitrophenylhydrazine (DNPH), followed by analysis via high performance liquid chromatography (HPLC) or ultra-high performance liquid chromatography (UHPLC).
Key Topics
- Formaldehyde and Carbonyl Compounds Detection: The standard covers the identification and quantification of formaldehyde and a wide range of other carbonyls, such as acetaldehyde, acetone, benzaldehyde, and several tolualdehydes, using DNPH derivatization and chromatographic analysis.
- Sampling Methodology: Air samples are actively drawn through DNPH-coated silica gel cartridges, where carbonyl compounds react to form stable hydrazones, which can be reliably analyzed by HPLC/UHPLC.
- Applicability: The method is suitable for detecting airborne carbonyls in concentrations ranging from approximately 10 ppbv/v (12 μg/m³) up to 1 ppmv/v (1.2 mg/m³), with potential for even lower detection by increasing sample volume or adjusting sampling duration.
- Adaptability: The procedure is a modification of the U.S. EPA Method TO-11A, extending its applicability to a broader range of carbonyl species.
- Time-Weighted Average (TWA) Sampling: The methodology provides a TWA sample, suitable for both short-term (as little as 5 minutes) and long-term (up to 24 hours) monitoring.
- Quality Assurance: Guidance is included for preparing DNPH-coated cartridges, controlling sampling flow rates, and addressing analytical interferences.
Applications
ASTM D5197-21 is utilized in a variety of air quality and occupational hygiene monitoring programs, including:
- Indoor Air Quality Monitoring: Ensures compliance with regulations for indoor pollutants, such as formaldehyde, in residential, commercial, or public spaces.
- Workplace Exposure Assessment: Used by industrial hygienists to monitor employee exposure to hazardous carbonyl compounds in manufacturing or processing facilities.
- Ambient and Emission Testing: Supports environmental impact studies by quantifying emissions of carbonyls from industrial stacks, vehicles, and other sources.
- Research and Development: Facilitates air pollution and toxicology studies in academic and commercial laboratory settings.
The method's detailed instructions on cartridge preparation, sampling equipment, and interferences make it highly adaptable for field and laboratory use.
Related Standards
For a comprehensive approach to air sampling and analysis, the following standards and methods are frequently used in conjunction with ASTM D5197-21:
- ASTM D1193 - Specification for Reagent Water
- ASTM D1356 - Terminology Relating to Sampling and Analysis of Atmospheres
- ASTM D3195 - Practice for Rotameter Calibration
- ASTM D3686 - Practice for Sampling Atmospheres to Collect Organic Compound Vapors
- EPA Method TO-11A - Compendium of Methods for the Determination of Toxic Organic Compounds in Ambient Air
- ASTM E682 - Practice for Liquid Chromatography Terms and Relationships
Practical Value
Adopting ASTM D5197-21 provides a scientifically validated framework for accurate detection and quantification of hazardous carbonyls in air. Laboratories, environmental consultants, industrial operators, and regulators benefit from:
- Consistent, reproducible measurements across different environments
- Improved compliance with occupational and environmental regulatory requirements
- Reliable support for health risk assessments related to formaldehyde and related compounds
- Compatibility with a range of commercially available and laboratory-prepared DNPH sampling cartridges
Utilizing this standard ensures high-quality air monitoring data, critical for protecting human health and the environment.
Buy Documents
ASTM D5197-21 - Standard Test Method for Determination of Formaldehyde and Other Carbonyl Compounds in Air (Active Sampler Methodology)
REDLINE ASTM D5197-21 - Standard Test Method for Determination of Formaldehyde and Other Carbonyl Compounds in Air (Active Sampler Methodology)
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D5197-21 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of Formaldehyde and Other Carbonyl Compounds in Air (Active Sampler Methodology)". This standard covers: SIGNIFICANCE AND USE 5.1 This test method provides an analytical procedure for measuring formaldehyde and other carbonyl compounds in indoor, workplace, ambient air or for emission testing. SCOPE 1.1 This test method presents a procedure for the determination of formaldehyde (HCHO) and other carbonyl compounds (aldehydes and ketones) in air. Other carbonyl compounds that have been successfully quantified by this method include acetaldehyde, acetone, propanal (propionaldehyde), 2-butanone (methyl ethyl ketone), butyraldehyde, benzaldehyde, isovaleraldehyde, valeraldehyde, o-tolualdehyde, m-tolualdehyde, p-tolualdehyde, hexanal, and 2,5-dimethylbenzaldehyde. 1.2 This test method involves drawing air through a cartridge containing silica gel coated with 2,4-dinitrophenylhydrazine (DNPH) reagent. Carbonyl compounds readily form stable derivatives with the acidified DNPH reagent. The DNPH derivatives are analyzed for parent aldehydes and ketones using high performance liquid chromatography (HPLC) or ultra-high performance liquid chromatography (UHPLC). UHPLC systems use higher pressures and smaller particle sizes in columns compared to HPLC systems. The sampling procedure is a modification of U.S. EPA Method TO-11A (see 2.2). 1.3 This test method is based on the reaction of carbonyl compounds with DNPH in the presence of an acid to form stable derivatives according to the reaction shown in Fig. 1, (where: both R and R1 are alkyl or aromatic groups (ketones), or either, or both R or R1 is a hydrogen atom (aldehydes)). The determination of formaldehyde and other carbonyl compounds, as DNPH derivatives, is similar to that of U.S. EPA Method TO-11A in that it uses HPLC or UHPLC for separation of carbonyl compounds followed by UV adsorption or photodiode array detection. This test method exceeds the stated applicability of TO-11A to include other carbonyl compounds that can be determined as stated in 10.2.4. This test method is suitable for determination of formaldehyde and other carbonyl compounds in the airborne concentration range from approximately 10 ppbv/v (12 μg/m3), requires sampling for 1 h at 1 L/min) to 1 ppmv/v (1.2 mg/m3). Lower concentrations in air may be determined using higher sampling volume and with control of contamination, appropriate selection of flow rate and sampling duration. FIG. 1 Reaction of Carbonyl Compounds 1.4 The sampling method gives a time-weighted average (TWA) sample. It can be used for long-term (1 to 24 h) or short-term (5 to 60 min) sampling of air for formaldehyde. Shorter sampling times or low flow rates will result in higher detection limits and may result in greater variation in co-located sampler results. Tests should be performed over a duration and a flow rate that allows the data quality objective of the project to be achieved. Sample times for other carbonyls, such as acetaldehyde, may be limited to short term (1).2 The data provides total concentrations of carbonyl compounds from which time weighted average concentrations can be calculated. 1.5 This test method instructs the user on how to prepare sampling cartridges from commercially available chromatographic grade silica gel cartridges3 by the application of acidified DNPH to each cartridge. 1.6 The sampling flow rate, as described in this test method, has been validated for sampling rates up to 1.5 L/min for formaldehyde. This flow rate limitation is principally due to the high pressure drop (>8 kPa at 1.0 L/min) across user prepared silica gel cartridges which have a particle size of 55 to 105 µm. These cartridges are not generally compatible with battery-powered pumps used in personal sampling equipment (for example, those used by industrial hygienists). 1.7 Alternatively, pre-coated DNPH silica gel cartridges are commercially available and may be substituted provided they can be demonstrated to meet blank and analyte trapping acceptance criteria (2). Some of these use silica gel of a larger parti...
SIGNIFICANCE AND USE 5.1 This test method provides an analytical procedure for measuring formaldehyde and other carbonyl compounds in indoor, workplace, ambient air or for emission testing. SCOPE 1.1 This test method presents a procedure for the determination of formaldehyde (HCHO) and other carbonyl compounds (aldehydes and ketones) in air. Other carbonyl compounds that have been successfully quantified by this method include acetaldehyde, acetone, propanal (propionaldehyde), 2-butanone (methyl ethyl ketone), butyraldehyde, benzaldehyde, isovaleraldehyde, valeraldehyde, o-tolualdehyde, m-tolualdehyde, p-tolualdehyde, hexanal, and 2,5-dimethylbenzaldehyde. 1.2 This test method involves drawing air through a cartridge containing silica gel coated with 2,4-dinitrophenylhydrazine (DNPH) reagent. Carbonyl compounds readily form stable derivatives with the acidified DNPH reagent. The DNPH derivatives are analyzed for parent aldehydes and ketones using high performance liquid chromatography (HPLC) or ultra-high performance liquid chromatography (UHPLC). UHPLC systems use higher pressures and smaller particle sizes in columns compared to HPLC systems. The sampling procedure is a modification of U.S. EPA Method TO-11A (see 2.2). 1.3 This test method is based on the reaction of carbonyl compounds with DNPH in the presence of an acid to form stable derivatives according to the reaction shown in Fig. 1, (where: both R and R1 are alkyl or aromatic groups (ketones), or either, or both R or R1 is a hydrogen atom (aldehydes)). The determination of formaldehyde and other carbonyl compounds, as DNPH derivatives, is similar to that of U.S. EPA Method TO-11A in that it uses HPLC or UHPLC for separation of carbonyl compounds followed by UV adsorption or photodiode array detection. This test method exceeds the stated applicability of TO-11A to include other carbonyl compounds that can be determined as stated in 10.2.4. This test method is suitable for determination of formaldehyde and other carbonyl compounds in the airborne concentration range from approximately 10 ppbv/v (12 μg/m3), requires sampling for 1 h at 1 L/min) to 1 ppmv/v (1.2 mg/m3). Lower concentrations in air may be determined using higher sampling volume and with control of contamination, appropriate selection of flow rate and sampling duration. FIG. 1 Reaction of Carbonyl Compounds 1.4 The sampling method gives a time-weighted average (TWA) sample. It can be used for long-term (1 to 24 h) or short-term (5 to 60 min) sampling of air for formaldehyde. Shorter sampling times or low flow rates will result in higher detection limits and may result in greater variation in co-located sampler results. Tests should be performed over a duration and a flow rate that allows the data quality objective of the project to be achieved. Sample times for other carbonyls, such as acetaldehyde, may be limited to short term (1).2 The data provides total concentrations of carbonyl compounds from which time weighted average concentrations can be calculated. 1.5 This test method instructs the user on how to prepare sampling cartridges from commercially available chromatographic grade silica gel cartridges3 by the application of acidified DNPH to each cartridge. 1.6 The sampling flow rate, as described in this test method, has been validated for sampling rates up to 1.5 L/min for formaldehyde. This flow rate limitation is principally due to the high pressure drop (>8 kPa at 1.0 L/min) across user prepared silica gel cartridges which have a particle size of 55 to 105 µm. These cartridges are not generally compatible with battery-powered pumps used in personal sampling equipment (for example, those used by industrial hygienists). 1.7 Alternatively, pre-coated DNPH silica gel cartridges are commercially available and may be substituted provided they can be demonstrated to meet blank and analyte trapping acceptance criteria (2). Some of these use silica gel of a larger parti...
ASTM D5197-21 is classified under the following ICS (International Classification for Standards) categories: 71.080.80 - Aldehydes and ketones. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D5197-21 has the following relationships with other standards: It is inter standard links to ASTM D1356-20a, ASTM D1356-20, ASTM D3686-20, ASTM E682-92(2019), ASTM D3631-99(2017), ASTM D1356-15a, ASTM D1356-15, ASTM D1356-14b, ASTM D1356-14a, ASTM D1356-14, ASTM D3686-13, ASTM E682-92(2011), ASTM D3631-99(2011), ASTM D1356-05(2010), ASTM D3686-08. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D5197-21 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D5197 − 21
Standard Test Method for
Determination of Formaldehyde and Other Carbonyl
Compounds in Air (Active Sampler Methodology)
This standard is issued under the fixed designation D5197; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1ppm (1.2mg⁄m ). Lower concentrations in air may be
v/v
determined using higher sampling volume and with control of
1.1 This test method presents a procedure for the determi-
contamination, appropriate selection of flow rate and sampling
nation of formaldehyde (HCHO) and other carbonyl com-
duration.
pounds (aldehydes and ketones) in air. Other carbonyl com-
pounds that have been successfully quantified by this method 1.4 The sampling method gives a time-weighted average
include acetaldehyde, acetone, propanal (propionaldehyde), (TWA) sample. It can be used for long-term (1 to 24 h) or
2-butanone (methyl ethyl ketone), butyraldehyde, short-term (5 to 60 min) sampling of air for formaldehyde.
benzaldehyde, isovaleraldehyde, valeraldehyde, Shorter sampling times or low flow rates will result in higher
o-tolualdehyde, m-tolualdehyde, p-tolualdehyde, hexanal, and detection limits and may result in greater variation in co-
2,5-dimethylbenzaldehyde. located sampler results. Tests should be performed over a
duration and a flow rate that allows the data quality objective
1.2 This test method involves drawing air through a car-
oftheprojecttobeachieved.Sampletimesforothercarbonyls,
tridge containing silica gel coated with 2,4-
such as acetaldehyde, may be limited to short term (1). The
dinitrophenylhydrazine(DNPH)reagent.Carbonylcompounds
dataprovidestotalconcentrationsofcarbonylcompoundsfrom
readily form stable derivatives with the acidified DNPH
whichtimeweightedaverageconcentrationscanbecalculated.
reagent. The DNPH derivatives are analyzed for parent alde-
hydes and ketones using high performance liquid chromatog- 1.5 This test method instructs the user on how to prepare
raphy (HPLC) or ultra-high performance liquid chromatogra- sampling cartridges from commercially available chromato-
phy (UHPLC). UHPLC systems use higher pressures and graphic grade silica gel cartridges by the application of
smaller particle sizes in columns compared to HPLC systems. acidified DNPH to each cartridge.
The sampling procedure is a modification of U.S. EPAMethod
1.6 Thesamplingflowrate,asdescribedinthistestmethod,
TO-11A (see 2.2).
has been validated for sampling rates up to 1.5 L/min for
1.3 This test method is based on the reaction of carbonyl formaldehyde.Thisflowratelimitationisprincipallyduetothe
compounds with DNPH in the presence of an acid to form
high pressure drop (>8 kPa at 1.0 L/min) across user prepared
stable derivatives according to the reaction shown in Fig. 1, silicagelcartridgeswhichhaveaparticlesizeof55to105µm.
(where: both R and R are alkyl or aromatic groups (ketones),
These cartridges are not generally compatible with battery-
or either, or both R or R is a hydrogen atom (aldehydes)).The powered pumps used in personal sampling equipment (for
determinationofformaldehydeandothercarbonylcompounds,
example, those used by industrial hygienists).
as DNPH derivatives, is similar to that of U.S. EPA Method
1.7 Alternatively, pre-coated DNPH silica gel cartridges are
TO-11A in that it uses HPLC or UHPLC for separation of
commercially available and may be substituted provided they
carbonylcompoundsfollowedbyUVadsorptionorphotodiode
can be demonstrated to meet blank and analyte trapping
array detection. This test method exceeds the stated applica-
acceptance criteria (2). Some of these use silica gel of a larger
bilityofTO-11Atoincludeothercarbonylcompoundsthatcan
particle size that results in a lower pressure drop across the
be determined as stated in 10.2.4. This test method is suitable
cartridge. These low pressure drop cartridges may be more
for determination of formaldehyde and other carbonyl com-
suitable for sampling air using battery-powered personal sam-
poundsintheairborneconcentrationrangefromapproximately
pling pumps.
10ppb (12 µg/m ), requires sampling for 1h at 1L⁄min) to
v/v
1 2
This test method is under the jurisdiction of ASTM Committee D22 on Air The boldface numbers in parentheses refer to a list of references at the end of
Quality and is the direct responsibility of Subcommittee D22.05 on Indoor Air. this standard.
Current edition approved Sept. 1, 2021. Published May 2022. Originally The cartridge used in the development and performance evaluation of this test
approved in 1991. Last previous edition approved in 2016 as D5197–16. DOI: method was the Sep-Pak Plus Silica cartridge. Other manufactures make similar
10.1520/D5197-22. products.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5197 − 21
2.3 Other Documents:
40 CFR Part 136 Appendix B, the MURDefinition and
Procedure for the Determination of the Method Detection
Limit – Revision 2
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this test method, refer
to Terminology D1356 and Practice E682.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 All other pertinent abbreviations and symbols are
defined when first cited in this test method.
4. Summary of Test Method
4.1 A known volume of air is drawn through a prepacked
FIG. 1 Reaction of Carbonyl Compounds silica gel cartridge coated with acidified DNPH, at a sampling
rateof0.5to1.5L/minforanappropriateperiodoftimebased
upon the intended use of the measurement. Both sampling rate
1.8 The values stated in SI units are to be regarded as
and time are dependent upon carbonyl concentrations in the
standard. No other units of measurement are included in this
test atmosphere.
standard.
4.2 After sampling, the sample cartridges are individually
1.9 This standard does not purport to address all of the
capped and placed in individual bottles or other sealable
safety concerns, if any, associated with its use. It is the
containers. Sample identifying tags or labels are attached and
responsibility of the user of this standard to establish appro-
the individual sample containers which are then placed in a
priate safety, health, and environmental practices and deter-
friction-top can or other suitable sealable secondary container
mine the applicability of regulatory limitations prior to use.
with a pouch of charcoal for transport to the laboratory for
1.10 This international standard was developed in accor-
analysis. Charcoal may only be useful if sampling chemicals
dance with internationally recognized principles on standard-
other than formaldehyde and acetaldehyde. The cartridges are
ization established in the Decision on Principles for the
stored at <4 °C protected from light until analysis.
Development of International Standards, Guides and Recom-
Alternatively, the cartridges may be desorbed, diluted to a
mendations issued by the World Trade Organization Technical
known volume, and refrigerated at <4°C until analysis.
Barriers to Trade (TBT) Committee.
NOTE1—Are-sealablefoil-linedplasticpouchofthetypeincludedwith
some commercial pre-coated DNPH cartridges may be used for storing a
2. Referenced Documents
DNPH-coated cartridge after sampling, if appropriate.
2.1 ASTM Standards:
4.3 The DNPH-carbonyl derivatives are determined using a
D1193Specification for Reagent Water
gradient HPLC or UHPLC system, equipped with a C18
D1356Terminology Relating to Sampling and Analysis of
reverse phase column and an ultraviolet (UV) absorption or
Atmospheres
photodiode array detector operated at 360 nm.
D3195Practice for Rotameter Calibration
4.4 A blank cartridge is likewise desorbed and analyzed in
D3631Test Methods for Measuring Surface Atmospheric
accordance with 4.3.
Pressure
D3686Practice for Sampling Atmospheres to Collect Or-
4.5 Formaldehyde and other carbonyl compounds in the
ganic Compound Vapors (Activated Charcoal Tube Ad-
sample are identified and quantified by comparison of their
sorption Method)
retention times and peak heights or peak areas of their
E682Practice for Liquid Chromatography Terms and Rela-
corresponding DNPH derivatives with those of standard solu-
tionships
tions.
2.2 EPA Methods:
5. Significance and Use
Method TO-11A EPA-625/R-96/010b, Compendium of
Methods for the Determination of Toxic Organic Com-
5.1 This test method provides an analytical procedure for
pounds in Ambient Air, U.S. Environmental Protection
measuring formaldehyde and other carbonyl compounds in
Agency, Research Triangle Park, NC, January 1999
indoor, workplace, ambient air or for emission testing.
6. Interferences
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
6.1 There are a number of known interferences and factors
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
potentially impacting sampling and quantification of carbonyl
Standards volume information, refer to the standard’s Document Summary page on
the ASTM website.
AvailablefromUnitedStatesEnvironmentalProtectionAgency(EPA),William
Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460, Available from U.S. Government Publishing Office (GPO), 732 N. Capitol St.,
http://www.epa.gov. NW, Washington, DC 20401, http://www.gpo.gov.
D5197 − 21
compounds using acidified DNPH impregnated cartridges. the devices described in 6.2.2 and 6.2.4 before the sample
These interferences and other factors are summarized in Table reaches the cartridge (4).
1.
6.2.1 The presence of ozone in the sample stream is readily
inferredfromtheappearanceofnewcompoundswithretention
6.2 Ozone (~50 ppbv and above) has been shown to
times shorter than that of the hydrazone of formaldehyde. Fig.
interfere negatively by reacting with both the DNPH and its
2 shows chromatograms of samples of a formaldehyde-spiked
carbonyl derivatives (hydrazones) in the cartridge (4-7). The
air stream with and without ozone.
extent of interference depends on the temporal variations of
both ozone and the carbonyl compounds and the duration of 6.2.2 Themostdirectsolutiontotheozoneinterferenceisto
sampling. Significant (~45%) negative interference from remove the ozone before the sample stream reaches the
ozone was observed even at concentrations of formaldehyde cartridge. This process entails constructing an ozone denuder
and ozone typical of clean ambient air (2 ppbv and 40 ppbv, or scrubber and placing it in front of the cartridge. Typically,
respectively) when air was sampled for three hours at 1 L/min. denuders and scrubbers use potassium iodide (KI). Manganese
It is highly recommended that ozone be removed by means of oxide scrubbers have also been used (8). At least some air
TABLE 1 Interferences and Other Factors Impacting Sampling and Analysis of Carbonyls Using DNPH Impregnated Cartridges
(Adapted From et al. (3))
Section Agent or Parameter Influenced Species Interferences Remedy
6.2 Ozone All Carbonyls Positive and negative artifacts Sample with upstream ozone
on carbonyl derivatives; scrubber
reaction with carbonyl
compounds; baseline and
retention time shifts
6.3 Nitrogen Dioxide and Nitric Formaldehyde and Nitrogen dioxide and nitric Better chromatographic
Oxide Acetaldehyde oxide react with DNPH forming separation
side products which may co-
elute with formaldehyde and
acetaldehyde derivative peaks
6.4 Relative Humidity (RH) Ketones, Carbonyls at Poor ketone collection Use alternative derivation agent
extremes efficiencies at nominal for ketones
sampling flow rates, leading to
large underestimation of ketone
concentrations; relative
humidity below 10 % and
above 75 % can result in low
carbonyl collection efficiencies
6.5 Polymerization Unsaturated Carbonyls Derivatives undergo Use alternate quantification
polymerization method for acrolein,
methacrolein, and
crotonaldehyde
6.6 DNPH Reagent Contamination Formaldehyde and Other Formaldehyde and other Purify DNPH by
Carbonyls carbonyls present in DNPH recrystallization
reagent
6.7 Co-elution All Carbonyls Isomeric aldehydes or ketones Better chromatographic
may co-elute with DNPH separation
derivatives of carbonyls in
sample
6.8 Sunlight All Carbonyls Artifacts may be created Store cartridges in opaque
containers. Shield outdoor
samples
6.9 Temperature All Carbonyls High temperatures can cause Store cartridges at <4 °C
disassociation of carbonyl-
DNPH derivatives with loss of
the carbonyl
6.10 Particles All Carbonyls Particulates collected on the Filter any acetonitrile extract
surface of the cartridge packing with visible particles prior to
may cause cartridge clogging analysis to prevent clogging of
and baseline disturbance HPLC
6.11 Sample Duration Acetaldehyde Low collection efficiencies can Only report acetaldehyde
occur at sample durations concentrations for sampling
greater than two hours times less than two hours
D5197 − 21
6.3 Nitrogen dioxide and nitric oxide can react with DNPH
forming side products which may chromatographically co-
elute or overlap with the formaldehyde and acetaldehyde
derivative peaks (11, 12). Better chromatographic separation
can be achieved by altering the separation conditions, for
example, by using alternative HPLC columns or mobile phase
compositions.
6.4 Low collection efficiencies may occur for formaldehyde
and other carbonyls in both very dry air (<10% RH) (13) and
very moist air (>75% RH) (10). Ketones are less reactive than
aldehydes and are more readily impacted by the sampling
conditions. Collection efficiencies of acetone and 2-butanone
in atmospheres with relative humidity above 50 % can be as
low as 20 % (3). Air temperature also may impact collection
efficiency. If the ambient air temperature during sampling is
below 15°C, a heated inlet probe is recommended.
6.5 Acrolein, methacrolein and crotonaldehyde should not
be quantified using the analytical procedure described in 10.2
duetotheformationofmultiplederivativepeaks (14-16).Inan
acidic environment in the presence of excess DNPH, the
DNPH derivatives of acrolein, methacrolein and crotonalde-
FIG. 2 Cartridge Samples of Formaldehyde in an Air Stream with
(A) and without (B) Ozone
hyde have been shown to partially transform into several
compounds that have UV spectra suggesting the presence of
the DNPH chromophore. The sequential conversion of the
moisture (relative humidity >10 % at 25°C) is required for
carbonyl-DNP-hydrazone (monomer) to carbonyl-DNP-
effective ozone removal when using KI (9).Adenuder may be
hydrazone-DNPH (dimer), and finally 2(carbonyl-DNP-
constructed by filling a 1-m section of 0.64-cm outside
hydrazone)-DNPH (trimer) has been demonstrated (14). The
diameter by 0.46-cm inside diameter copper tubing with a
chromatic response areas of the dimers and trimers have been
saturatedsolutionofKIinwater,allowingthesolutiontostand
summed in the past to estimate the concentration of acrolein.
for a few minutes (~5), draining the solution and drying the
However, this process does not account for the variations in
tubing with a stream of clean air or nitrogen for about 1 h.The
carbonyl in the trimer, the varying response factors for the
capacity of the ozone denuder as described is about 100
dimer and trimer, and the potential for co-elution of other
ppmv-hour of ozone. Test aldehydes (formaldehyde,
hydrazine products (for example, from crotonaldehyde) that
acetaldehyde, propionaldehyde, benzaldehyde, and
complicate quantification. Hence, the summing of the dimer
p-tolualdehyde) that were dynamically spiked into an ambient
and trimers to estimate acrolein concentration is not a reliable
sample air stream passed through the denuder. Scrubbers may
quantitative procedure (15).
be constructed by impregnating 37-mm cellulose fiber filters
6.6 Contamination of DNPH reagent with formaldehyde
with 0.6M KI solution.
and other carbonyls such as acetone is frequently encountered.
6.2.3 Ozone scrubbers (cartridges filled with granular KI)
The DNPH must be purified by multiple recrystallizations in
are also commercially available from suppliers of pre-coated
UV-grade acetonitrile. Recrystallization is accomplished, at 40
DNPH cartridges. However, in high humidity environments
to 60°C, by slow evaporation of the solvent to maximize
these scrubbers can become saturated with water, reducing the
crystal size. The purified DNPH crystals are stored under
sample flow through the cartridge. To overcome the moisture
UV-grade acetonitrile until use. Impurity levels of carbonyl
issue in high humidity environments, the ozone scrubbers
compounds in the DNPH and in commercial coated DNPH
should be maintained at a temperature of 90°C and consistent
cartridges are determined prior to use by HPLC or UHPLC
sample flow should be verified at the end of the sampling
and, at a minimum, should be less than 0.15 µg per cartridge.
period.
Acceptable blanks are dictated by the application, that is, the
6.2.4 UsingKIdenudersandscrubbersunderhighhumidity
compounds that are being measured, their expected concentra-
conditionscancauseinterferences.MoistKIcantrapcarbonyls
tions and the desired detection level.
prior to the DNPH cartridge. Wet KI can form iodine and the
hydroxylradicalwhichcanmigratetotheDNPHcartridgeand 6.7 The solid sorbent sampling procedure is for the sam-
degrade the DNPH and the carbonyl-DNP-hydrazone deriva- pling and analysis of specific carbonyl compounds that are
tives (6). This reaction can be avoided by keeping the ozone identified based on their chromatographic retention times.
scrubbers at 90°C. Alternatively, the hydroxyl radical can be Certainisomericaldehydesorketonesmaybechromatographi-
neutralized by placing an acid permeated filter between the cally unresolved by the HPLC system and may co-elute with
ozonedenuder/scrubberandtheDNPHcartridge,thus,increas- DNPH derivatives of the target carbonyl compounds in the
ing the collection efficiency in the presence of ozone and sample. Organic compounds that are retained by the sample
elevated relative humidity (10). and that have UV absorbance at 360 nm may also cause
D5197 − 21
interferences. Such interferences can often be identified and applications, it may be desirable or required that the sampling
overcome by altering the chromatographic separation condi- system flow is certified on a periodic basis against a NIST or
tions. Alternatively, UHPLC systems can improve chromato- equivalent reference standard.
graphic resolution and overcome interferences and some coe-
NOTE 2—An example of a sampling system for ambient air consisting
lution issues.
of a heated manifold/sample inlet, a denuder/cartridge assembly, a flow
meter, a vacuum gage/pump, a timer and a power supply is shown in Fig.
6.8 Exposure of the DNPH-coated sampling cartridges to
3. In operation, ambient air is drawn through the denuder/cartridge
directsunlightmayproduceartifactsandshouldbeavoidedby
assembly with a vacuum pump at a fixed flow rate between 0.5 to 1.5
storing cartridges in opaque containers such as foil-lined
L/min.
pouches (17). When sampling outdoors, samplers should be
NOTE3—Apressuredropthroughtheuser-preparedsamplecartridgeof
shielded from direct exposure to sunlight.
about 19 kPa at a sampling rate of 1.5 L/min has been observed. Some
commercially available pre-coated cartridges may exhibit lower pressure
6.9 Hightemperaturescancausedisassociationofcarbonyl-
drops, which will permit the use of battery-operated personal sampling
DNPH derivatives with loss of the carbonyl as the carbonyl-
pumps.
DNPH reaction is an equilibrium reaction. Formaldehyde-
7.2 HPLC or UHPLC System, an example HPLC system
DNPH derivatives are particularly sensitive to temperature.
used for this analysis consists of two or more mobile phase
Cartridgesshouldbechilledat<4°Cpriortosampling,assoon
reservoirs; a single or a dual high-pressure pump system
aspossibleaftersampling,andextractsshouldalsobestoredat
equipped with a mobile phase gradient programmer, an injec-
<4°Cpriortoanalysis (18).Note,excessivelylowtemperature
tion valve (automatic sampler with a fixed-volume sampling
(typically <10°C) storage of commercially produced car-
loop (for example, 10 L, 20 L)); a C18 reverse phase (RP)
tridges should be avoided to prevent mounting rings from
column(forexample,25-cmby4.6-mminsidediameter);aUV
falling off or cartridge bodies from cracking.
detector operating at 360 nm; and a data system. A typical
6.10 Particulates collected on the surface of the cartridge
gradient HPLC system configuration is shown in Fig. 4.
packingmaycausecartridgecloggingandcreateback-pressure
during analysis. If these particulates are insoluble in acetoni- NOTE 4—Most commercial HPLC analytical systems will be adequate
for this application.
trile (for example: α-pinene aerosol) they may create signifi-
cant baseline disturbance during analysis. To prevent clogging
7.3 Stopwatch.
of HPLC components, remove insoluble acetonitrile particles
7.4 Friction-Top Metal Can (for example, 4-LPaint Can) or
by filtration prior to analysis (19).
Other Suitable Container, with polyethylene air bubble pack-
6.11 Sample collection efficiency was shown to be between
ingorothersuitablepadding,toholdandcushionsamplevials.
1–62 % for 24-hour sampling of acetaldehyde at a sample
7.5 Thermometer, to record temperature.
collection rate of 0.15L⁄min. Collection efficiencies of 100 %
were reported for sampling of less than 2 hours (1).Itis
7.6 Barometer (refer to Test Methods D3631).
recommended that reporting of acetaldehyde concentrations
should be from samples of 2 hours or less. 7.7 Suction Filtration Apparatus,forfilteringHPLCmobile
phase (optional).
7. Apparatus
7.8 Volumetric Flasks, various sizes, 5 to 2000 mL.
7.1 Sampling System, capable of accurately and precisely
sampling 0.5 to 1.50 L/min to the nearest 0.01 L/min. In some 7.9 Pipets, various sizes, 1 to 50 mL.
FIG. 3 A Dual-Cartridge Sampling System with Heated Manifold for Carbonyl Compounds in Ambient Air
D5197 − 21
FIG. 4 A Typical Gradient HPLC System Configuration for Determination of Carbonyl Compounds Collected on DNPH Cartridges
7.10 Helium Purge Line, for degassing HPLC mobile phase
(optional).
7.11 Erlenmeyer Flask, 1 L, for preparing HPLC mobile
phase.
7.12 Graduated Cylinder, 1 L, for preparing HPLC mobile
phase.
7.13 Syringes, for HPLC injection, with capacity at least
four times the loop volume (see 7.2) (optional).
7.14 Sample Vials.
7.15 Melting Point Apparatus, (optional).
7.16 Rotameters (refer to Practice D3195), Soap Bubble
Meter,or Wet Test Meter.
7.17 Graduated Syringes.
7.18 Mass Flowmeters, Mass Flow Controllers, or Other
Suitable Device for metering/setting air flow rate of 0.5 to 1.5
L/min through sample cartridge.
7.19 Positive Displacement, Repetitive Dispensing Pipets,0
to 10-mL range.
7.20 Cartridge Drying Manifold, with multiple standard
male syringe connectors (see Fig. 5).
7.21 LiquidSyringes(polypropylenesyringesareadequate),
10 mL, used to prepare acidified DNPH-coated cartridges.
FIG. 5 Syringe Rack for Coating and Drying Sample Cartridges
7.22 Syringe Rack, made from an aluminum plate or other
suitablematerial(0.16by36by53-cm)withadjustablelegson
7.23 Syringe Fittings/Plugs, to connect cartridges to the
four corners. A matrix (5 by 9) of circular holes of diameter
sampling system and to cap prepared cartridges.
slightly larger than the diameter of the 10-mL syringes,
symmetrically drilled from the center of the plate, to enable 7.24 Hot Plates, Beakers, Flasks, Measuring and Dispos-
batch processing of 45 cartridges for cleaning, coating, or able Pipets, Volumetric Flasks, and so forth, used in the
sample elution, or combination thereof (see Fig. 5). purification of DNPH.
D5197 − 21
7.25 BorosilicateGlassCultureTubes,(20by125mm)with concentrations/longer exposures (see the Safety Data Sheet
polypropylene screw caps or other suitable container to trans- (SDS) for more details).
port coated cartridges. 9.1.1 Prepare a supersaturated solution of DNPH by boiling
excess DNPH in 200 mLof acetonitrile for approximately 1 h.
7.26 Heated Probe, necessary for when the temperature of
9.1.2 After 1 h, remove and transfer the supernatant to a
sampled air is below 15°C.
covered beaker on a hot plate and allow gradual cooling to 40
7.27 Cartridge Sampler, prepacked with silica gel and
to 60°C.
coated with DNPH in accordance with Section 9,oras
9.1.3 Maintain the solution at this temperature (40°C) until
commercially available.
95% of solvent has evaporated.
7.28 Polyethylene Gloves, used to handle silica gel car- 9.1.4 Decant the solution to waste, and rinse the remaining
tridges. crystals twice with three times their apparent volume of
acetonitrile.
8. Reagents and Materials
9.1.5 Transfer the crystals to another clean beaker, add
200mL of acetonitrile, heat to boiling, and again let crystals
8.1 Purity of Reagents—Reagent grade chemicals shall be
grow slowly at 40 to 60°C until 95% of the solvent has
used in all tests. Unless otherwise indicated, it is intended that
evaporated.
all reagents conform to the specifications of the Committee on
9.1.6 Repeat rinsing process as described in 9.1.4.
Analytical Reagents of theAmerican Chemical Society where
such specifications are available. 9.1.7 Take an aliquot of the second rinse, dilute ten times
with acetonitrile, acidify with 1 mL of 3.8 M perchloric acid
8.2 Purity of Water—Unless otherwise indicated, references
per 100 mL of DNPH solution, and analyze by HPLC, in
to water shall be understood to mean reagent water as defined
accordance with 10.2.4.
by Type II of Specification D1193.
NOTE 6—An acid is necessary to catalyze the reaction of the carbonyls
8.3 2,4-Dinitrophenylhydrazine (DNPH), recrystallized at
with DNPH. Most strong inorganic acids such as hydrochloric, sulfuric,
least twice with UV-grade acetonitrile before use.
phosphoric or perchloric acids will perform satisfactorily. Perchloric acid
was the preferred catalyst for impinger sampling when using acetonitrile
8.4 Acetonitrile, UV-grade.
solutionofDNPHastheabsorbingsolution.TheDNPHderivativesdonot
8.5 Perchloric Acid, 60%, specific gravity 1.51.
precipitate from solution as readily as when hydrochloric acid is used as
thecatalyst.ThisisanidealsituationforanHPLCanalyticalfinishasthis
8.6 Hydrochloric Acid, 36.5–38%, specific gravity 1.19.
minimizes sample handling. For most ambient air sampling, precipitation
is not a problem because the carbonyl concentration is generally in the
8.7 Formaldehyde, 37% solution (w/w).
parts per billion range.
8.8 Aldehydes and Ketones, used for preparation of DNPH
9.1.8 An acceptable impurity level in 9.1.7 is typically
derivative standards (optional).
<0.025 µg/mL of formaldehyde DNPH reagent derivative. If
8.9 Ethanol or Methanol.
the impurity level is not acceptable for intended sampling
8.10 Silica Gel Solid-Phase Extraction Cartridges. application, repeat recrystallization.
9.1.9 Transfer the purified crystals to an all-glass reagent
8.11 Nitrogen, high-purity grade (best source).
bottle,add200mLofacetonitrile,stopper,shakegently,andlet
8.12 Charcoal, granular (best source).
stand overnight.Analyze the supernatant as in 9.1.7 by HPLC
8.13 Helium, high-purity grade (best source). in accordance with 10.2.3.
9.1.10 If the impurity level is not satisfactory, pipet the
9. Preparation of Reagents and Cartridges
solutiontowaste,thenadd25mLofacetonitriletothepurified
NOTE 5—This section is intended for users who desire to prepare their
crystals. Repeat rinsing with 20-mL portions of acetonitrile
own sampling cartridges by coating prepacked silica gel cartridges with
until a satisfactorily low impurity level in the supernatant is
acidified DNPH. Users who intend to purchase DNPH-coated cartridges
confirmed by HPLC analysis.
andDNPHderivativestandardsfromcommercialsourcesmayskipanyor
all portions of this section. Users are cautioned to check that the carbonyl 9.1.11 If the impurity level is satisfactory, add another
background of the purchased cartridges meet the quality control and
25mL of acetonitrile, stopper, and shake the reagent bottle,
accuracy required for their intended applications.
then set aside. The saturated solution above the purified
9.1 Purification of 2,4-Dinitrophenylhydrazine (DNPH):
crystals is the stock DNPH reagent.
Warning—This procedure should be performed under a prop-
9.1.12 Maintain only a minimum volume of saturated solu-
erly ventilated hood and behind a protective shield, as there is
tion adequate for day-to-day operation. This will minimize
an explosion potential from perchloric acid and inhalation of
waste of purified reagent, should it be necessary to re-rinse the
acetonitrile can result in nose and throat irritation (brief
crystals to decrease the level of impurity for applications
exposure at 500 ppm) or more serious effects at higher
requiring more stringent purity specifications.
9.1.13 Use clean pipets when removing saturated DNPH
stock solution for any analytical applications. Do not pour the
ACS Reagent Chemicals, Specifications and Procedures for Reagents and
stock solution from the reagent bottle.
Standard-Grade Reference Materials, American Chemical Society, Washington,
DC. For suggestions on the testing of reagents not listed by theAmerican Chemical
9.2 Preparation of DNPH-Formaldehyde Derivative:
Society, see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset,
9.2.1 To a portion of the recrystallized DNPH add sufficient
U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharma-
copeial Convention, Inc. (USPC), Rockville, MD. 2 N HCl to obtain an approximately saturated solution.Add to
D5197 − 21
thissolutionformaldehydeinmolarexcessoftheDNPH.Filter rack for coating and drying the sample cartridges is illustrated
theDNPH-formaldehydeprecipitate,washitwith2 NHCland in Fig. 5(a) and Fig. 5(b).
water, and allow it to dry in air. 9.4.2.2 Using a positive displacement, repetitive pipet, add
9.2.2 Check the purity of the DNPH-formaldehyde deriva- 10 mL of acetonitrile to each of the syringes.
9.4.2.3 Let liquid drain to waste by gravity.
tivebymeltingpoint(166°C)determinationorHPLC/UHPLC
analysis. If the impurity level is not acceptable, recrystallize
NOTE 10—Remove any air bubbles that may be trapped between the
the derivative in ethanol. Repeat the purity check and recrys-
syringe and the silica cartridge by displacing them with the acetonitrile in
tallization as necessary until an acceptable level of purity (for
the syringe.
example, 99%) is achieved.
9.4.2.4 Set the repetitive dispenser containing the acidified
9.2.3 The DNPH derivatives of formaldehyde and other
DNPH coating solution to dispense 7 mL into the cartridges.
carbonylcompoundssuitableforuseasstandardsarecommer-
9.4.2.5 Once the effluent flow at the outlet of the cartridge
cially available both in the form of pure crystals and as
has stopped, dispense 7 mLof the coating reagent into each of
individual or mixed stock solutions in acetonitrile.
the syringes.
9.3 Preparation of DNPH-Formaldehyde Standards: 9.4.2.6 Let the coating reagent drain by gravity through the
cartridge until flow at the other end of the cartridge stops.
9.3.1 Prepare a standard stock solution of the DNPH form-
9.4.2.7 Wipe the excess liquid at the outlet of each of the
aldehyde derivative by dissolving accurately weighed amounts
cartridges with clean tissue paper.
in acetonitrile.
9.4.2.8 Assemble a drying manifold as shown in Fig. 5(b).
9.3.2 Prepare a working calibration standard mix from the
Thiscontainsapreviouslyprepared,DNPH-coatedcartridgeat
standard stock solution. The concentration of the DNPH
each of the exit ports (for example, these scrubber or “guard
formaldehyde derivative in the standard mix solutions should
cartridges” can be prepared by drying a few of the newly
be adjusted to reflect the range of concentrations expected in
coated cartridges in accordance with 9.4.2.9 – 9.4.2.15 and
real samples.
“sacrificing” these few to ensure the purity of the rest). The
NOTE 7—Individual stock solutions of approximately 100 mg/L are
“guardcartridges”servetoremovetracesofformaldehydethat
prepared by dissolving 10 mg of the solid derivative in 100 mL of
may be present in the nitrogen gas supply.
acetonitrile. The individual solution is used to prepare calibration stan-
9.4.2.9 Insert cartridge connectors (flared at both ends, 0.64
dards containing the derivative of interest at concentrations of 0.5 to
20µg⁄mL that spans the concentration of interest.
by2.5-cmoutsidediameterTFE-fluorocarbonFEPtubingwith
insidediameterslightlysmallerthantheoutsidediameterofthe
9.3.3 Store all standard solutions in tightly capped contain-
cartridge port) onto the long end of the scrubber cartridges.
ers at <4°C in a refrigerator.They should be stable for several
9.4.2.10 Remove the cartridges from the syringes and con-
months.
nect the short ends of the cartridges to the open end of the
9.4 Preparation of DNPH-Coated Cartridges:
cartridge connectors already attached to the scrubber car-
NOTE 8—This procedure must be performed in an atmosphere with a tridges.
very low aldehyde background. All glassware and plastic ware must be
9.4.2.11 Pass nitrogen through each of the cartridges at
scrupulously cleaned and rinsed with deionized water and aldehyde-free
about 300 to 400 mL/min.
acetonitrile. Contact of reagents with laboratory air must be minimized.
9.4.2.12 Rinse the exterior surfaces and outlet end of the
Polyethylene gloves must be worn when handling the cartridges.
cartridges with acetonitrile using a Pasteur pipet.
9.4.1 DNPH Coating Solution:
9.4.2.13 After 15 min, stop the flow of nitrogen, wipe the
9.4.1.1 Pipet 30 mL of saturated DNPH stock solution into
cartridge exterior free of rinse acetonitrile, and remove the
a 1000-mL volumetric flask, then add 500 mL acetonitrile.
dried cartridges.
9.4.1.2 Acidify with 1.0 mL of concentrated HCl.
9.4.2.14 Plug both ends of the coated cartridge with stan-
NOTE9—Theatmosphereabovetheacidifiedsolutionshouldpreferably dard polypropylene male syringe plugs and place the plugged
be filtered through a DNPH-coated silica gel cartridge, to minimize
cartridge in a borosilicate glass culture tube with polypropyl-
contamination from laboratory air. Shake the solution, then make up to
ene screw caps.
volumewithacetonitrile.Stoppertheflask,invert,andshakeseveraltimes
9.4.2.15 Put a serial number and a lot number label on each
until the solution is homogeneous. Transfer the acidified solution to a
of the individual cartridge glass storage containers and refrig-
reagent bottle equipped witha0to 10-mL range positive displacement
dispenser. erate the prepared lot until use.
9.4.1.3 Prime the dispenser and slowly dispense 10 to
10. Procedure
20mL to waste.
9.4.1.4 Dispense an aliquot solution to a sample vial, and
10.1 Sample Collection:
check the impurity level of the acidified solution by HPLC/
10.1.1 Assemble the sampling system, and ensure that the
UHPLC in accordance with 9.1.
pump is capable of a constant flow rate throughout the
9.4.1.5 The impurity level should be <0.025 µg/mLformal-
sampling period. The coated cartridges can be used as direct
dehyde as the DNPH derivative, similar to that in the DNPH
probes and traps for sampling air when the temperature is
stock solution.
above15°C(see7.26).Addanozonedenuderorscrubber(see
9.4.2 Coating of Silica Gel Cartridges: 6.2) if required.
9.4.2.1 Openthecartridgepackage,connecttheshortendto 10.1.2 Beforesamplecollection,checkthesystemforleaks.
a 10-mL syringe, and place it in the syringe rack. The syringe Plugtheinlet(shortend)ofthecartridgesonoflowisindicated
D5197 − 21
at the outlet end of the pump. The mass flowmeter should not 10.1.7 Turnthesampleronandadjusttheflowtothedesired
indicate any air flow through the sampling apparatus. As rate.Atypical flow rate is 1.0 L/min through one cartridge and
0.8 L/min for two cartridges in series.
required or desired the flow can be qualified against a NISTor
similarly traceable flow standard. 10.1.8 Operate the sampler for the desired period, with
periodic recording of the sampling variables.
NOTE 11—The silica gel is held in the cartridge between two fine
10.1.9 If the ambient air temperature during sampling is
porosity filter frits. Air flow during sampling could change as airborne
below15°C,aheatedinletprobeisrecommendedtominimize
particulates deposit on the front frit.The flow change could be significant
the liquid water interferences in the DNPH cartridge and to
whensamplingparticulate-ladenatmospheres.Forunattendedorextended
increase reaction rates.
samplingperiods,amassflowcontrolleror,asappropriate,acompensated
personal sampling pump is highly recommended to maintain constant
10.1.10 At the end of the sampling period, stop the flow.
flow. The mass flow controller should be set at least 20% below its
The flow rate must be checked just before stopping the flow. If
maximum airflow rate.
the flow rates at the beginning and end of the sampling period
10.1.3 Install the entire assembly, an example is shown in differ by more than 15%, the sample should be marked as
Fig. 3, (including a “dummy” sampling cartridge) and check suspect.
10.1.11 Immediately after sampling, remove the cartridge
the flow rate at a value near the desired rate. In general, flow
(using polyethylene gloves) from the sampling system, cap
rates of 0.5 to 1.5 L/min should be employed. The total moles
with the original end plugs, and place it back in the original
ofcarbonylinthevolumeofairsampledshouldnotexceedthat
labeled glass culture tube. Cap the culture tube, seal it with
of the DNPH (0.005 to 0.01 mmol/cartridge for commercially
TFE-fluorocarbon tape. If appropriate, a re-sealable foil-lined
available pre-coated cartridges). In general, a safe estimate of
plastic pouch may be used instead of the glass culture tube for
the sample size should be approximately 75% of the DNPH
storing the exposed cartridge. Refrigerate the culture tube or
loading of the cartridge (100 to 200 g as HCHO). Generally,
pouch containing the exposed sample cartridge at <4 °C until
flowcalibrationisaccomplishedusingasoapbubbleflowmeter
analysis. The refrigeration period prior to analysis must not
or calibrated wet test meter connected to the flow exit,
exceed 14 days.
assuming the system is sealed. If the system is not sealed a
sacrificial cartridge can be temporarily replace the sample
NOTE 13—If samples are to be shipped to a central laboratory for
analysis, the duration of the non-refrigerated period should be kept to a
cartridge allowing the flow calibration to be measured at the
minimum, preferably less than two days.
inlet to the sacrificial cartridge.
10.1.12 The average sample flow rate is calculated from the
NOTE 12—Test Method D3686 describes an appropriate calibration
following equation:
scheme that does not require a sealed flow system downstream of the
Q 1Q 1.1Q
pump.
1 2 n
Q 5 (1)
A
n
10.1.4 The operator must measure and record the sampling
flow rate at the beginning and end of the sampling period to where:
determine sample volume. If the sampling period exceeds 2 h,
Q = average flow rate, mL/min,
A
the flow rate should be measured at intermediate points during Q,Q,.Q = flow rates determined at beginning, end,
1 2 n
the sampling period. Include a rotameter to allow observation
and intermediate points during sampling,
of the flow rate without interruption of the sampling process. and
n = number of points averaged.
Alternatively, a sampling pump which directly measures and
continuously records the flow can be used. If the sampler was
10.1.13 The total flow volume is then calculated using the
qualified against a flow standard, this should also be recorded.
following equation:
10.1.5 Before sampling, remove the glass culture tube from
T 2 T 3Q
~ !
2 1 A
V 5 (2)
the friction-top metal can or other suitable container. Let the
m
cartridge warm to room temperature in the glass tube before
where:
connectingittothesampletrain.Withacommercialpre-coated
DNPH cartridge, let the cartridge warm to room temperature V = totalvolume,L,sampledatthemeasuredtempera-
m
ture and pressure,
before connecting to the sampling train.
T = stop time, min,
10.1.6 Using polyethylene gloves, remove the coated car-
T = start time, min,
tridge from the glass tube, remove the syringe plugs, and
T –T = total sampling time, min, and
2 1
connect the cartridge to the sampling system with a syringe
Q = average flow rate, mL/min.
A
adapter fitting. Seal the glass tube for later use and connect the
10.1.14 Thetotalvolume(V )atstandardconditions,25°C
m
cartridge to the sampling train so that the short end becomes
and 101.3 kPa, is calculated from the following equation:
the sample inlet. With commercial pre-coated cartridges, fol-
low the manufacturer’s instructions. Some cartridges may be P 298
A
V 5 V 3 3 (3)
s m
constructed from sealed-glass tubes. For these, break the ends 101.3 2731t
A
of the cartridge with a tube breaker. Connect the cartridge by
where:
inserting the end with the smaller quantity of sorbent to the
V = total sample volume, L, at 25°C and 101.3-kPa
s
samplingtrainsothatthelargerquantityofsorbentisattheair
pressure,
inlet end. Use care when handling the broken ends.
D5197 − 21
10.2.3.2 Operate the instrument in the isocratic mode if the
V = total sample volume, L, at measured temperature and
m
analytesofinterestarelimitedtothederivativesformaldehyde,
pressure,
acetaldehyde,acetoneandpropionaldehyde;otherwise,operate
P = average pressure, kPa, and
A
t = average temperature, °C. the system in the gradient mode. For isocratic separation of
A
formaldehyde,theparametersinTable2canbeusedforHPLC
10.2 Sample Analysis:
based analyses.
10.2.1 Sample Storage—Samples are returned to the labo-
Before each analysis, check the detector baseline to ensure
ratory in an insulated container at <4°C with appropriate
stable conditions.
padding and stored in a refrigerator at <4°C until analysis.
10.2.3.3 The operating parameters found adequate for the
Alternatively, the samples may also be stored alone in a
separation of the 17 carbonyls within the scope of this test
refrigerator at <4°C in their individual glass containers or
method are shown in Table 3.
pouches. The time between sampling and analysis must not
10.2.3.4 The gradient program in 10.2.3.3 adequately sepa-
exceed 14 days.
rates DNPH derivatives of formaldehyde and acetaldehyde;
10.2.2 Sample Desorption:
acrolein and its principal transformation product from the
NOTE 14—Sample desorption can be automated provided quantitative
derivatives of acetone and propionaldehyde; the derivative of
recovery during extraction is demonstrated, for example, by performance
crotonaldehyde from its principal transformation product; the
testing.
derivative of methacrolein from the derivatives of 2-butanone
10.2.2.1 Connect the sample cartridge (inlet or short end
and butyraldehyde; the derivatives of isovaleraldehyde,
during sampling) to a clean syringe.
valeraldehyde, hexanal, 2,5-dimethylbenzaldehyde; and the
isomers of the tolualdehyde derivatives. Due to transforma-
NOTE 15—The liquid flow during desorption should be in the same
tions (6.5) acrolein, methacrolein and crotonaldehyde should
direction as the air flow during sampling to prevent insoluble particulates
from getting into the eluate. Reverse desorption may be performed if the not be quantified with this method.
eluate is filtered prior to HPLC or UHPLC analysis. A filtered blank
10.2.3.5 Chromatographic parameters that have been used
extract must be analyzed to confirm that no contamination is being
in round robin analysis of the carbonyl derivatives participated
introduced by the LC filter.
in by several laboratories have been described in the literature
10.2.2.2 Place the cartridge/syringe in the syringe rack.
(20).
10.2.2.3 Extract the DNPH derivatives of the carbonyls and
NOTE 18—Column manufacturers as well as suppliers of pre-coated
the unreacted DNPH from the cartridge (gravity feed) by
DNPH cartridges usually recommend optimal conditions for the sepa
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D5197 − 16 D5197 − 21
Standard Test Method for
Determination of Formaldehyde and Other Carbonyl
Compounds in Air (Active Sampler Methodology)
This standard is issued under the fixed designation D5197; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method presents a procedure for the determination of formaldehyde (HCHO) and other carbonyl compounds
(aldehydes and ketones) in air. Other carbonyl compounds that have been successfully quantified by this method include
acetaldehyde, acetone, propanal (propionaldehyde), 2-butanone (methyl ethyl ketone), butyraldehyde, benzaldehyde,
isovaleraldehyde, valeraldehyde, o-tolualdehyde, m-tolualdehyde, p-tolualdehyde, hexanal, and 2,5-dimethylbenzaldehyde.
1.2 This test method involves drawing air through a cartridge containing silica gel coated with 2,4-dinitrophenylhydrazine
(DNPH) reagent. Carbonyl compounds readily form stable derivatives with the acidified DNPH reagent. The DNPH derivatives
are analyzed for parent aldehydes and ketones utilizingusing high performance liquid chromatography (HPLC). (HPLC) or
ultra-high performance liquid chromatography (UHPLC). UHPLC systems use higher pressures and smaller particle sizes in
columns compared to HPLC systems. The sampling procedure is a modification of U.S. EPA Method TO-11A (see 2.2).
1.3 This test method is based on the specific reaction of carbonyl compounds with DNPH in the presence of an acid to form stable
derivatives according to the reaction shown in Fig. 1, (where: both R and R are alkyl or aromatic groups (ketones), or either, or
both R or R is a hydrogen atom (aldehydes)). The determination of formaldehyde and other carbonyl compounds, as DNPH
derivatives, is similar to that of U.S. EPA Method TO-11A in that it utilizes HPLC with UV detection as the analytical finish. The
applicability of this test method is extended beyond uses HPLC or UHPLC for separation of carbonyl compounds followed by UV
adsorption or photodiode array detection. This test method exceeds the stated applicability of TO-11A to include other carbonyl
compounds that can be determined as stated in 10.2.4. This test method is suitable for determination of formaldehyde and other
carbonyl compounds in the airborne concentration range from approximately 1010 ppb ppb (12 μg/m ), requires sampling for
v/v
1 h at 1 L ⁄min) to 11 ppm ppm(1.2 mg ⁄m (v/v). ). Lower concentrations in air may be determined with careful using higher
v/v
sampling volume and with control of contamination, appropriate selection of flow rate and sampling duration.
1.4 The sampling method gives a time-weighted average (TWA) sample. It can be used for long-term (1 to 24 h) or short-term
(5 to 60 min) sampling of air for formaldehyde. Shorter sampling times or low flow rates will result in higher detection limits and
may result in greater variation in co-located sampler results. Tests should be performed over a duration and a flow rate that allows
the data quality objective of the project to be achieved. Sample times for other carbonyls, such as acetaldehyde, may be limited
to short term (1). The data provides total concentrations of carbonyl compounds from which time weighted average concentrations
can be calculated.
This test method is under the jurisdiction of ASTM Committee D22 on Air Quality and is the direct responsibility of Subcommittee D22.05 on Indoor Air.
Current edition approved Oct. 1, 2016Sept. 1, 2021. Published November 2016May 2022. Originally approved in 1991. Last previous edition approved in 20092016 as
ɛ1
D5197 – 09D5197 – 16. . DOI: 10.1520/D5197-16.10.1520/D5197-22.
The boldface numbers in parentheses refer to a list of references at the end of this standard.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D5197 − 21
FIG. 1 Reaction of Carbonyl Compounds
1.5 This test method instructs the user on how to prepare sampling cartridges from commercially available chromatographic grade
silica gel cartridges by the application of acidified DNPH to each cartridge.
1.6 The sampling flow rate, as described in this test method, has been validated for sampling rates up to 1.5 L/min for
formaldehyde. This flow rate limitation is principally due to the high pressure drop (>8 kPa at 1.0 L/min) across the user prepared
silica gel cartridges which have a particle size of 55 to 105 μm. These cartridges are not generally compatible with battery-powered
pumps used in personal sampling equipment (for example, those used by industrial hygienists).
1.7 Alternatively, pre-coated DNPH silica gel cartridges are also commercially available and may be substituted provided they can
be demonstrated to perform equivalently meet blank and analyte trapping acceptance criteria (2). Some of these use silica gel of
a larger particle size that results in a lower pressure drop across the cartridge. These low pressure drop cartridges may be more
suitable for sampling air using battery-powered personal sampling pumps.
1.8 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.9 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.10 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D1193 Specification for Reagent Water
D1356 Terminology Relating to Sampling and Analysis of Atmospheres
D3195 Practice for Rotameter Calibration
D3631 Test Methods for Measuring Surface Atmospheric Pressure
D3686 Practice for Sampling Atmospheres to Collect Organic Compound Vapors (Activated Charcoal Tube Adsorption Method)
E682 Practice for Liquid Chromatography Terms and Relationships
2.2 EPA Methods:
Method TO-11A EPA-625/R-96/010b, Compendium of Methods for the Determination of Toxic Organic Compounds in Ambient
Air, U.S. Environmental Protection Agency, Research Triangle Park, NC, January 1999
The cartridge used in the development and performance evaluation of this test method was the Sep-Pak Plus Silica cartridge. Other manufactures make similar products.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from United States Environmental Protection Agency (EPA), William Jefferson Clinton Bldg., 1200 Pennsylvania Ave., NW, Washington, DC 20460,
http://www.epa.gov.
D5197 − 21
2.3 Other Documents:
40 CFR Part 136 Appendix B, the MUR Definition and Procedure for the Determination of the Method Detection Limit –
Revision 2
3. Terminology
3.1 Definitions:
3.1.1 For definitions of terms used in this test method, refer to Terminology D1356 and Practice E682.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 All other pertinent abbreviations and symbols are defined when first cited in this test method.
4. Summary of Test Method
4.1 A known volume of air is drawn through a prepacked silica gel cartridge coated with acidified DNPH, at a sampling rate of
0.5 to 1.5 L/min for an appropriate period of time. time based upon the intended use of the measurement. Both sampling rate and
time are dependent upon carbonyl concentrations in the test atmosphere.
4.2 After sampling, the sample cartridges are individually capped and placed in individual bottles or other sealable containers.
Sample identifying tags or labels are attached and the individual sample containers which are then placed in a friction-top can or
other suitable sealable secondary container with a pouch of charcoal for transport to the laboratory for analysis. Charcoal may only
be useful if sampling chemicals other than formaldehyde and acetaldehyde. The cartridges are stored at <4°C <4 °C protected from
light until analysis. Alternatively, the cartridges may be desorbed, diluted to a known volume, and refrigerated at <4°C<4 °C until
analysis.
NOTE 1—A re-sealable foil-lined plastic pouch of the type included with some commercial pre-coated DNPH cartridges may be used for storing a
DNPH-coated cartridge after sampling, if appropriate.
4.3 The DNPH-carbonyl derivatives are determined using a gradient HPLC or UHPLC system, equipped with a C18 reverse phase
column and an ultraviolet (UV) absorption or photodiode array detector operated at 360 nm.
4.4 A blank cartridge is likewise desorbed and analyzed in accordance with 4.3.
4.5 Formaldehyde and other carbonyl compounds in the sample are identified and quantified by comparison of their retention times
and peak heights or peak areas of their corresponding DNPH derivatives with those of standard solutions.
5. Significance and Use
5.1 This test method provides an analytical procedure for measuring formaldehyde and other carbonyl compounds in indoor,
workplace, outdoorambient air or for emission testing.
6. Interferences
6.1 There are a number of known interferences and factors potentially impacting sampling and quantification of carbonyl
compounds using acidified DNPH impregnated cartridges. These interferences and other factors are summarized in Table 1.
6.2 Ozone (~50 ppbv and above) has been shown to interfere negatively by reacting with both the DNPH and its carbonyl
derivatives (hydrazones) in the cartridge (4-7). The extent of interference depends on the temporal variations of both ozone and
the carbonyl compounds and the duration of sampling. Significant (~45 %) negative interference from ozone was observed even
at concentrations of formaldehyde and ozone typical of clean ambient air (2 ppbv and 40 ppbv, respectively) when air was sampled
for three hours at 1 L/min. It is highly recommended that ozone be removed by means of the devices described in 6.2.2 and 6.2.4
before the sample reaches the cartridge (4).
Available from U.S. Government Publishing Office (GPO), 732 N. Capitol St., NW, Washington, DC 20401, http://www.gpo.gov.
D5197 − 21
TABLE 1 Interferences and Other Factors Impacting Sampling and Analysis of Carbonyls Using DNPH Impregnated Cartridges
(Adapted From et al. (3))
Section Agent or Parameter Influenced Species Interferences Remedy
6.2 Ozone All Carbonyls Positive and negative artifacts Sample with upstream ozone
on carbonyl derivatives; scrubber
reaction with carbonyl
compounds; baseline and
retention time shifts
6.3 Nitrogen Dioxide and Nitric Formaldehyde and Nitrogen dioxide and nitric Better chromatographic
Oxide Acetaldehyde oxide react with DNPH forming separation
side products which may co-
elute with formaldehyde and
acetaldehyde derivative peaks
6.4 Relative Humidity (RH) Ketones, Carbonyls at Poor ketone collection Use alternative derivation agent
extremes efficiencies at nominal for ketones
sampling flow rates, leading to
large underestimation of ketone
concentrations; relative
humidity below 10 % and
above 75 % can result in low
carbonyl collection efficiencies
6.5 Polymerization Unsaturated Carbonyls Derivatives undergo Use alternate quantification
polymerization method for acrolein,
methacrolein, and
crotonaldehyde
6.6 DNPH Reagent Contamination Formaldehyde and Other Formaldehyde and other Purify DNPH by
Carbonyls carbonyls present in DNPH recrystallization
reagent
6.7 Co-elution All Carbonyls Isomeric aldehydes or ketones Better chromatographic
may co-elute with DNPH separation
derivatives of carbonyls in
sample
6.8 Sunlight All Carbonyls Artifacts may be created Store cartridges in opaque
containers. Shield outdoor
samples
6.9 Temperature All Carbonyls High temperatures can cause Store cartridges at <4°C
disassociation of carbonyl-
DNPH derivatives with loss of
the carbonyl
6.9 Temperature All Carbonyls High temperatures can cause Store cartridges at <4 °C
disassociation of carbonyl-
DNPH derivatives with loss of
the carbonyl
6.10 Particles All Carbonyls Particulates collected on the Filter any acetonitrile extract
surface of the cartridge packing with visible particles prior to
may cause cartridge clogging analysis to prevent clogging of
and baseline disturbance HPLC
6.11 Sample Duration Acetaldehyde Low collection efficiencies can Only report acetaldehyde
occur at sample durations concentrations for sampling
greater than two hours times less than two hours
6.2.1 The presence of ozone in the sample stream is readily inferred from the appearance of new compounds with retention times
shorter than that of the hydrazone of formaldehyde. Fig. 2 shows chromatograms of samples of a formaldehyde-spiked air stream
with and without ozone.
6.2.2 The most direct solution to the ozone interference is to remove the ozone before the sample stream reaches the cartridge.
This process entails constructing an ozone denuder or scrubber and placing it in front of the cartridge. Typically, denuders and
scrubbers utilizeuse potassium iodide (KI). Manganese oxide scrubbers have also been used (8). At least some air moisture (relative
humidity >10 % at 25°C)25 °C) is required for effective ozone removal when using KI (9). A denuder may be constructed by filling
a 1-m section of 0.64-cm outside diameter by 0.46-cm inside diameter copper tubing with a saturated solution of KI in water,
allowing the solution to stand for a few minutes (~5), draining the solution and drying the tubing with a stream of clean air or
D5197 − 21
FIG. 2 Cartridge Samples of Formaldehyde in an Air Stream with (A) and without (B) Ozone
nitrogen for about 1 h. The capacity of the ozone denuder as described is about 100 ppmv-hour of ozone. Test aldehydes
(formaldehyde, acetaldehyde, propionaldehyde, benzaldehyde, and p-tolualdehyde) that were dynamically spiked into an ambient
sample air stream passed through the denuder. Scrubbers may be constructed by impregnating 37-mm cellulose fiber filters with
0.6M KI solution.
6.2.3 Ozone scrubbers (cartridges filled with granular KI) are also commercially available from suppliers of pre-coated DNPH
cartridges. However, in high humidity environments these scrubbers can become saturated with water, reducing the sample flow
through the cartridge. To overcome the moisture issue in high humidity environments, the ozone scrubbers should be maintained
at a temperature of 90°C90 °C and consistent sample flow should be verified at the end of the sampling period.
6.2.4 Using KI denuders and scrubbers under high humidity conditions can cause interferences. Moist KI can trap carbonyls prior
to the DNPH cartridge. Wet KI can form iodine and the hydroxyl radical which can migrate to the DNPH cartridge and degrade
the DNPH and the carbonyl-DNP-hydrazone derivatives (6). This reaction can be avoided by keeping the ozone scrubbers at a
temperature of 90°C. 90 °C. Alternatively, the hydroxyl radical can be neutralized by placing an acid permeated filter between the
ozone denuder/scrubber and the DNPH cartridge, thus, increasing the collection efficiency in the presence of ozone and elevated
relative humidity (10).
6.3 Nitrogen dioxide and nitric oxide can react with DNPH forming side products which may chromatographically co-elute or
overlap with the formaldehyde and acetaldehyde derivative peaks (11, 12). Better chromatographic separation can be achieved by
altering the separation conditions, for example, by using alternative HPLC columns or mobile phase compositions.
6.4 Low collection efficiencies may occur for formaldehyde and other carbonyls in both very dry air (<10 % RH) (13) and very
moist air (>75%(>75 % RH) (10). Ketones are less reactive than aldehydes and are more readily impacted by the sampling
conditions. Collection efficiencies of acetone and 2-butanone in atmospheres with relative humidity above 50 % can be as low as
20 % (3). Air temperature also may impact collection efficiency. If the ambient air temperature during sampling is below
15°C,15 °C, a heated inlet probe is recommended.
6.5 Acrolein, methacrolein and crotonaldehyde should not be quantified using the analytical procedure described in 10.210.2 due
to the formation of multiple derivative peaks (14-16). In an acidic environment in the presence of excess DNPH, the DNPH
derivatives of acrolein, methacrolein and crotonaldehyde have been shown to partially transform into several compounds that have
UV spectra suggesting the presence of the DNPH chromophore. The sequential conversion of the carbonyl-DNP-hydrazone
(monomer) to carbonyl-DNP-hydrazone-DNPH (dimer), and finally 2(carbonyl-DNP-hydrazone)-DNPH (trimer) has been
demonstrated (14). The chromatic response areas of the dimers and trimers have been summed in the past to estimate the
concentration of acrolein. However, this process does not account for the variations in carbonyl in the trimer, the varying response
D5197 − 21
factors for the dimer and trimer, and the potential for co-elution of other hydrazine products (for example, from crotonaldehyde)
that complicate quantification. Hence, the summing of the dimer and trimers to estimate acrolein concentration is not a reliable
quantitative procedure (15).
6.6 Contamination of DNPH reagent with formaldehyde and other carbonyls such as acetone is a frequently encountered problem.
encountered. The DNPH must be purified by multiple recrystallizations in UV-grade acetonitrile. Recrystallization is
accomplished, at 40 to 60°C,60 °C, by slow evaporation of the solvent to maximize crystal size. The purified DNPH crystals are
stored under UV-grade acetonitrile until use. Impurity levels of carbonyl compounds in the DNPH and in commercial coated
DNPH cartridges are determined prior to use by HPLC or UHPLC and, at a minimum, should be less than 0.15 μg per cartridge.
Acceptable blanks are dictated by the application, that is, the compounds that are being measured, their expected concentrations
and the desired detection level.
6.7 The solid sorbent sampling procedure is for the sampling and analysis of specific carbonyl compounds that are identified based
on their chromatographic retention times. Certain isomeric aldehydes or ketones may be chromatographically unresolved by the
HPLC system and may co-elute with DNPH derivatives of the target carbonyl compounds in the sample. Organic compounds that
are retained by the sample and that have UV absorbance at 360 nm may also cause interferences. Such interferences can often be
identified and overcome by altering the chromatographic separation conditions. Alternatively, UHPLC systems can improve
chromatographic resolution and overcome interferences and some coelution issues.
6.8 Exposure of the DNPH-coated sampling cartridges to direct sunlight may produce artifacts and should be avoided by storing
cartridges in opaque containers such as foil-lined pouches (17). When sampling outdoors, samplers should be shielded from direct
exposure to sunlight.
6.9 High temperatures can cause disassociation of carbonyl-DNPH derivatives with loss of the carbonyl as the carbonyl-DNPH
reaction is an equilibrium reaction. Formaldehyde-DNPH derivatives are particularly sensitive to temperature. Cartridges should
be chilled at <4°C<4 °C prior to sampling, as soon as possible after sampling, and extracts should also be stored at <4°C<4 °C
prior to analysis (18). Note, excessively low temperature (typically <10 °C) storage of commercially produced cartridges should
be avoided to prevent mounting rings from falling off or cartridge bodies from cracking.
6.10 Particulates collected on the surface of the cartridge packing may cause cartridge clogging and create back-pressure during
analysis. If these particulates are insoluble in acetonitrile (for example: α-pinene aerosol) they may create significant baseline
disturbance during analysis. To prevent clogging of HPLC components, remove insoluble acetonitrile particles by filtration prior
to analysis (19).
6.11 Sample collection efficiency was shown to be between 1–62 % for 24-hour sampling of acetaldehyde. acetaldehyde at a
sample collection rate of 0.15 L ⁄min. Collection efficiencies of 100 % were reported for samplessampling of less than 2 hours (1).
It is recommended that reporting of acetaldehyde concentrations should be from samples of 2 hours or less.
7. Apparatus
7.1 Sampling System, capable of accurately and precisely sampling 0.5 to 1.50 L/min to the nearest 0.01 L/min. In some
applications, it may be desirable or required that the sampling system flow is certified on a periodic basis against a NIST or
equivalent reference standard.
NOTE 2—An example of a sampling system for ambient air consisting of a heated manifold/sample inlet, a denuder/cartridge assembly, a flow meter, a
vacuum gage/pump, a timer and a power supply is shown in Fig. 3. In operation, ambient air is drawn through the denuder/cartridge assembly with a
vacuum pump at a fixed flow rate between 0.5 to 1.5 L/min.
NOTE 3—A pressure drop through the user-prepared sample cartridge of about 19 kPa at a sampling rate of 1.5 L/min has been observed. Some
commercially available pre-coated cartridges may exhibit lower pressure drops, which will permit the use of battery-operated personal sampling pumps.
7.2 HPLC or UHPLC System, an example HPLC system used for this analysis consists of two or more mobile phase reservoirs;
a single or a dual high-pressure pump system equipped with a mobile phase gradient programmer, an injection valve (automatic
sampler with a fixed-volume sampling loop (for example, 10 L, 20 L)); a C18 reverse phase (RP) column (for example, 25-cm
by 4.6-mm inside diameter); a UV detector operating at 360 nm; and a data system. A typical gradient HPLC system configuration
is shown in Fig. 4.
D5197 − 21
FIG. 3 A Dual-Cartridge Sampling System with Heated Manifold for Carbonyl Compounds in Ambient Air
FIG. 4 A Typical Gradient HPLC System Configuration for Determination of Carbonyl Compounds Collected on DNPH Cartridges
NOTE 4—Most commercial HPLC analytical systems will be adequate for this application.
7.3 Stopwatch.
7.4 Friction-Top Metal Can (for example, 4-L Paint Can) or Other Suitable Container, with polyethylene air bubble packing or
other suitable padding, to hold and cushion sample vials.
7.5 Thermometer, to record temperature.
7.6 Barometer (refer to Test Methods D3631).
7.7 Suction Filtration Apparatus, for filtering HPLC mobile phase (optional).
D5197 − 21
7.8 Volumetric Flasks, various sizes, 5 to 2000 mL.
7.9 Pipets, various sizes, 1 to 50 mL.
7.10 Helium Purge Line, for degassing HPLC mobile phase (optional).
7.11 Erlenmeyer Flask, 1 L, for preparing HPLC mobile phase.
7.12 Graduated Cylinder, 1 L, for preparing HPLC mobile phase.
7.13 Syringes, for HPLC injection, with capacity at least four times the loop volume (see 7.2) (optional).
7.14 Sample Vials.
7.15 Melting Point Apparatus, (optional).
7.16 Rotameters (refer to Practice D3195), Soap Bubble Meter, or Wet Test Meter.
7.17 Graduated Syringes.
7.18 Mass Flowmeters, Mass Flow Controllers, or Other Suitable Device for metering/setting air flow rate of 0.5 to 1.5 L/min
through sample cartridge.
7.19 Positive Displacement, Repetitive Dispensing Pipets, 0 to 10-mL range.
7.20 Cartridge Drying Manifold, with multiple standard male syringe connectors (see Fig. 5).
7.21 Liquid Syringes (polypropylene syringes are adequate), 10 mL, used to prepare acidified DNPH-coated cartridges.
7.22 Syringe Rack, made from an aluminum plate or other suitable material (0.16 by 36 by 53-cm) with adjustable legs on four
corners. A matrix (5 by 9) of circular holes of diameter slightly larger than the diameter of the 10-mL syringes, symmetrically
drilled from the center of the plate, to enable batch processing of 45 cartridges for cleaning, coating, or sample elution, or
combination thereof (see Fig. 5).
7.23 Syringe Fittings/Plugs, to connect cartridges to the sampling system and to cap prepared cartridges.
7.24 Hot Plates, Beakers, Flasks, Measuring and Disposable Pipets, Volumetric Flasks, and so forth, used in the purification of
DNPH.
7.25 Borosilicate Glass Culture Tubes, (20 by 125 mm) with polypropylene screw caps or other suitable container to transport
coated cartridges.
7.26 Heated Probe, necessary for when the temperature of sampled air is below 15°C.15 °C.
7.27 Cartridge Sampler, prepacked with silica gel and coated with DNPH in accordance with Section 9, or as commercially
available.
7.28 Polyethylene Gloves, used to handle silica gel cartridges.
D5197 − 21
FIG. 5 Syringe Rack for Coating and Drying Sample Cartridges
8. Reagents and Materials
8.1 Purity of Reagents—Reagent grade chemicals shall be used in all tests. Unless otherwise indicated, it is intended that all
reagents conform to the specifications of the Committee on Analytical Reagents of the American Chemical Society where such
specifications are available.
8.2 Purity of Water—Unless otherwise indicated, references to water shall be understood to mean reagent water as defined by Type
II of Specification D1193.
8.3 2,4-Dinitrophenylhydrazine (DNPH), recrystallized at least twice with UV-grade acetonitrile before use.
8.4 Acetonitrile, UV-grade.
8.5 Perchloric Acid, 60 %, specific gravity 1.51.
8.6 Hydrochloric Acid, 36.5–38 %, specific gravity 1.19.
8.7 Formaldehyde, 37 % solution (w/w).
8.8 Aldehydes and Ketones, used for preparation of DNPH derivative standards (optional).
8.9 Ethanol or Methanol.
Reagent Chemicals, American Chemical Society Specifications, American Chemical ACS Reagent Chemicals, Specifications and Procedures for Reagents and
Standard-Grade Reference Materials, American Chemical Society, Washington, DC. For suggestions on the testing of reagents not listed by the American Chemical Society,
see Analar Standards for Laboratory Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia and National Formulary, U.S. Pharmacopeial
Convention, Inc. (USPC), Rockville, MD.
D5197 − 21
8.10 Silica Gel Solid-Phase Extraction Cartridges.
8.11 Nitrogen, high-purity grade (best source).
8.12 Charcoal, granular (best source).
8.13 Helium, high-purity grade (best source).
9. Preparation of Reagents and Cartridges
NOTE 5—This section is intended for users who desire to prepare their own sampling cartridges by coating prepacked silica gel cartridges with acidified
DNPH. Users who intend to purchase DNPH-coated cartridges and DNPH derivative standards from commercial sources may skip any or all portions
of this section. Users are cautioned to check that the carbonyl background of the purchased cartridges meet the quality control and accuracy required for
their intended applications.
9.1 Purification of 2,4-Dinitrophenylhydrazine (DNPH): Warning—This procedure should be performed under a properly
ventilated hood and behind a protective shield, as there is an explosion potential from perchloric acid and inhalation of acetonitrile
can result in nose and throat irritation (brief exposure at 500 ppm) or more serious effects at higher concentrations/longer exposures
(see the Safety Data Sheet (SDS) for more details).
9.1.1 Prepare a supersaturated solution of DNPH by boiling excess DNPH in 200 mL of acetonitrile for approximately 1 h.
9.1.2 After 1 h, remove and transfer the supernatant to a covered beaker on a hot plate and allow gradual cooling to 40 to
60°C.60 °C.
9.1.3 Maintain the solution at this temperature (40°C)(40 °C) until 95 % of solvent has evaporated.
9.1.4 Decant the solution to waste, and rinse the remaining crystals twice with three times their apparent volume of acetonitrile.
9.1.5 Transfer the crystals to another clean beaker, add 200 mL 200 mL of acetonitrile, heat to boiling, and again let crystals grow
slowly at 40 to 60°C60 °C until 95 % of the solvent has evaporated.
9.1.6 Repeat rinsing process as described in 9.1.4.
9.1.7 Take an aliquot of the second rinse, dilute ten times with acetonitrile, acidify with 1 mL of 3.8 M perchloric acid per 100
mL of DNPH solution, and analyze by HPLC, in accordance with 10.2.4.
NOTE 6—An acid is necessary to catalyze the reaction of the carbonyls with DNPH. Most strong inorganic acids such as hydrochloric, sulfuric, phosphoric
or perchloric acids will perform satisfactorily. Perchloric acid was the preferred catalyst for impinger sampling when using acetonitrile solution of DNPH
as the absorbing solution. The DNPH derivatives do not precipitate from solution as readily as when hydrochloric acid is used as the catalyst. This is
an ideal situation for an HPLC analytical finish as this minimizes sample handling. For most ambient air sampling, precipitation is not a problem because
the carbonyl concentration is generally in the parts per billion range.
9.1.8 An acceptable impurity level in 9.1.7 is typically <0.025 μg/mL of formaldehyde DNPH reagent derivative. If the impurity
level is not acceptable for intended sampling application, repeat recrystallization.
9.1.9 Transfer the purified crystals to an all-glass reagent bottle, add 200 mL of acetonitrile, stopper, shake gently, and let stand
overnight. Analyze the supernatant as in 9.1.7 by HPLC in accordance with 10.2.3.
9.1.10 If the impurity level is not satisfactory, pipet the solution to waste, then add 25 mL of acetonitrile to the purified crystals.
Repeat rinsing with 20-mL portions of acetonitrile until a satisfactorily low impurity level in the supernatant is confirmed by HPLC
analysis.
9.1.11 If the impurity level is satisfactory, add another 25 mL 25 mL of acetonitrile, stopper, and shake the reagent bottle, then
set aside. The saturated solution above the purified crystals is the stock DNPH reagent.
D5197 − 21
9.1.12 Maintain only a minimum volume of saturated solution adequate for day-to-day operation. This will minimize waste of
purified reagent, should it be necessary to rerinsere-rinse the crystals to decrease the level of impurity for applications requiring
more stringent purity specifications.
9.1.13 Use clean pipets when removing saturated DNPH stock solution for any analytical applications. Do not pour the stock
solution from the reagent bottle.
9.2 Preparation of DNPH-Formaldehyde Derivative:
9.2.1 To a portion of the recrystallized DNPH add sufficient 2 N HCl to obtain an approximately saturated solution. Add to this
solution formaldehyde in molar excess of the DNPH. Filter the DNPH-formaldehyde precipitate, wash it with 2 N HCl and water,
and allow it to dry in air.
9.2.2 Check the purity of the DNPH-formaldehyde derivative by melting point (166°C)(166 °C) determination or HPLCHPLC/
UHPLC analysis. If the impurity level is not acceptable, recrystallize the derivative in ethanol. Repeat the purity check and
recrystallization as necessary until an acceptable level of purity (for example, 99 %) is achieved.
9.2.3 The DNPH derivatives of formaldehyde and other carbonyl compounds suitable for use as standards are commercially
available both in the form of pure crystals and as individual or mixed stock solutions in acetonitrile.
9.3 Preparation of DNPH-Formaldehyde Standards:
9.3.1 Prepare a standard stock solution of the DNPH formaldehyde derivative by dissolving accurately weighed amounts in
acetonitrile.
9.3.2 Prepare a working calibration standard mix from the standard stock solution. The concentration of the DNPH formaldehyde
derivative in the standard mix solutions should be adjusted to reflect the range of concentrations expected in real samples.
NOTE 7—Individual stock solutions of approximately 100 mg/L are prepared by dissolving 10 mg of the solid derivative in 100 mL of acetonitrile. The
individual solution is used to prepare calibration standards containing the derivative of interest at concentrations of 0.5 to 2020 μg μg/mL ⁄mL that spans
the concentration of interest.
9.3.3 Store all standard solutions in tightly capped containers at <4°C<4 °C in a refrigerator. They should be stable for several
months.
9.4 Preparation of DNPH-Coated Cartridges:
NOTE 8—This procedure must be performed in an atmosphere with a very low aldehyde background. All glassware and plastic ware must be scrupulously
cleaned and rinsed with deionized water and aldehyde-free acetonitrile. Contact of reagents with laboratory air must be minimized. Polyethylene gloves
must be worn when handling the cartridges.
9.4.1 DNPH Coating Solution:
9.4.1.1 Pipet 30 mL of saturated DNPH stock solution into a 1000-mL volumetric flask, then add 500 mL acetonitrile.
9.4.1.2 Acidify with 1.0 mL of concentrated HCl.
NOTE 9—The atmosphere above the acidified solution should preferably be filtered through a DNPH-coated silica gel cartridge, to minimize contamination
from laboratory air. Shake the solution, then make up to volume with acetonitrile. Stopper the flask, invert, and shake several times until the solution is
homogeneous. Transfer the acidified solution to a reagent bottle equipped with a 0 to 10-mL range positive displacement dispenser.
9.4.1.3 Prime the dispenser and slowly dispense 10 to 20 mL 20 mL to waste.
9.4.1.4 Dispense an aliquot solution to a sample vial, and check the impurity level of the acidified solution by HPLCHPLC/
UHPLC in accordance with 9.1.
D5197 − 21
9.4.1.5 The impurity level should be <0.025 μg/mL formaldehyde as the DNPH derivative, similar to that in the DNPH stock
solution.
9.4.2 Coating of Silica Gel Cartridges:
9.4.2.1 Open the cartridge package, connect the short end to a 10-mL syringe, and place it in the syringe rack. The syringe rack
for coating and drying the sample cartridges is illustrated in Fig. 5(a) and Fig. 5(b).
9.4.2.2 Using a positive displacement, repetitive pipet, add 10 mL of acetonitrile to each of the syringes.
9.4.2.3 Let liquid drain to waste by gravity.
NOTE 10—Remove any air bubbles that may be trapped between the syringe and the silica cartridge by displacing them with the acetonitrile in the syringe.
9.4.2.4 Set the repetitive dispenser containing the acidified DNPH coating solution to dispense 7 mL into the cartridges.
9.4.2.5 Once the effluent flow at the outlet of the cartridge has stopped, dispense 7 mL of the coating reagent into each of the
syringes.
9.4.2.6 Let the coating reagent drain by gravity through the cartridge until flow at the other end of the cartridge stops.
9.4.2.7 Wipe the excess liquid at the outlet of each of the cartridges with clean tissue paper.
9.4.2.8 Assemble a drying manifold as shown in Fig. 5(b). This contains a previously prepared, DNPH-coated cartridge at each
of the exit ports (for example, these scrubber or “guard cartridges” can be prepared by drying a few of the newly coated cartridges
in accordance with 9.4.2.9 – 9.4.2.15 and “sacrificing” these few to ensure the purity of the rest). The “guard cartridges” serve to
remove traces of formaldehyde that may be present in the nitrogen gas supply.
9.4.2.9 Insert cartridge connectors (flared at both ends, 0.64 by 2.5-cm outside diameter TFE-fluorocarbon FEP tubing with inside
diameter slightly smaller than the outside diameter of the cartridge port) onto the long end of the scrubber cartridges.
9.4.2.10 Remove the cartridges from the syringes and connect the short ends of the cartridges to the open end of the cartridge
connectors already attached to the scrubber cartridges.
9.4.2.11 Pass nitrogen through each of the cartridges at about 300 to 400 mL/min.
9.4.2.12 Rinse the exterior surfaces and outlet end of the cartridges with acetonitrile using a Pasteur pipet.
9.4.2.13 After 15 min, stop the flow of nitrogen, wipe the cartridge exterior free of rinse acetonitrile, and remove the dried
cartridges.
9.4.2.14 Plug both ends of the coated cartridge with standard polypropylene male syringe plugs and place the plugged cartridge
in a borosilicate glass culture tube with polypropylene screw caps.
9.4.2.15 Put a serial number and a lot number label on each of the individual cartridge glass storage containers and refrigerate the
prepared lot until use.
10. Procedure
10.1 Sample Collection:
10.1.1 Assemble the sampling system, and ensure that the pump is capable of a constant flow rate throughout the sampling period.
The coated cartridges can be used as direct probes and traps for sampling air when the temperature is above 15°C15 °C (see 7.26).
Add an ozone denuder or scrubber (see 6.2) if required.
10.1.2 Before sample collection, check the system for leaks. Plug the inlet (short end) of the cartridge so no flow is indicated at
D5197 − 21
the outlet end of the pump. The mass flowmeter should not indicate any air flow through the sampling apparatus. As required or
desired the flow can be qualified against a NIST or similarly traceable flow standard.
NOTE 11—The silica gel is held in the cartridge between two fine porosity filter frits. Air flow during sampling could change as airborne particulates
deposit on the front frit. The flow change could be significant when sampling particulate-laden atmospheres. For unattended or extended sampling periods,
a mass flow controller or, as appropriate, a compensated personal sampling pump is highly recommended to maintain constant flow. The mass flow
controller should be set at least 20 % below its maximum airflow rate.
10.1.3 Install the entire assembly, an example is shown in Fig. 3, (including a “dummy” sampling cartridge) and check the flow
rate at a value near the desired rate. In general, flow rates of 0.5 to 1.5 L/min should be employed. The total moles of carbonyl
in the volume of air sampled should not exceed that of the DNPH (0.005 to 0.01 mmol/cartridge for commercially available
pre-coated cartridges). In general, a safe estimate of the sample size should be approximately 75 % of the DNPH loading of the
cartridge (100 to 200 g as HCHO). Generally, flow calibration is accomplished using a soap bubble flowmeter or calibrated wet
test meter connected to the flow exit, assuming the system is sealed. If the system is not sealed a sacrificial cartridge can be
temporarily replace the sample cartridge allowing the flow calibration to be measured at the inlet to the sacrificial cartridge.
NOTE 12—Test Method D3686 describes an appropriate calibration scheme that does not require a sealed flow system downstream of the pump.
10.1.4 The operator must measure and record the sampling flow rate at the beginning and end of the sampling period to determine
sample volume. If the sampling period exceeds 2 h, the flow rate should be measured at intermediate points during the sampling
period. Include a rotameter to allow observation of the flow rate without interruption of the sampling process. Alternatively, a
sampling pump which directly measures and continuously records the flow can be used. If the sampler was qualified against a flow
standard, this should also be recorded.
10.1.5 Before sampling, remove the glass culture tube from the friction-top metal can or other suitable container. Let the cartridge
warm to room temperature in the glass tube before connecting it to the sample train. With a commercial pre-coated DNPH
cartridge, let the cartridge warm to room temperature before connecting to the sampling train.
10.1.6 Using polyethylene gloves, remove the coated cartridge from the glass tube, remove the syringe plugs, and connect the
cartridge to the sampling system with a syringe adapter fitting. Seal the glass tube for later use,use and connect the cartridge to
the sampling train so that the short end becomes the sample inlet. With commercial pre-coated cartridges, follow the
manufacturer’s instructions. Some cartridges may be constructed from sealed-glass tubes. For these, break the ends of the cartridge
with a tube breaker. Connect the cartridge by inserting the end with the smaller quantity of sorbent to the sampling train so that
the larger quantity of sorbent is at the air inlet end. Use care when handling the broken ends.
10.1.7 Turn the sampler on and adjust the flow to the desired rate. A typical flow rate is 1.0 L/min through one cartridge and 0.8
L/min for two cartridges in series.
10.1.8 Operate the sampler for the desired period, with periodic recording of the sampling variables.
10.1.9 If the ambient air temperature during sampling is below 15°C,15 °C, a heated inlet probe is recommended to minimize the
liquid water interferences in the DNPH cartridge and to increase reaction rates.
10.1.10 At the end of the sampling period, stop the flow. The flow rate must be checked just before stopping the flow. If the flow
rates at the beginning and end of the sampling period differ by more than 15 %, the sample should be marked as suspect.
10.1.11 Immediately after sampling, remove the cartridge (using polyethylene gloves) from the sampling system, cap with the
original end plugs, and place it back in the original labeled glass culture tube. Cap the culture tube, seal it with TFE-fluorocarbon
tape. If appropriate, a re-sealable foil-lined plastic pouch may be used instead of the glass culture tube for storing the exposed
cartridge. Refrigerate the culture tube or pouch containing the exposed sample cartridge at < 4 °C <4 °C until analysis. The
refrigeration period prior to analysis shouldmust not exceed 14 days.
NOTE 13—If samples are to be shipped to a central laboratory for analysis, the duration of the non-refrigerated period should be kept to a minimum,
preferably less than two days.
10.1.12 The average sample flow rate is calculated from the following equation:
D5197 − 21
Q 1Q 1.1Q
1 2 n
Q 5 (1)
A
n
where:
Q = average flow rate, mL/min,
A
Q , Q , . . . Q = flow rates determined at beginning, end, and intermediate points during sampling, and
1 2 n
n = number of points averaged.
10.1.13 The total flow volume is then calculated using the following equation:
~T 2 T !3Q
2 1 A
V 5 (2)
m
where:
V = total volume, L, sampled at the measured temperature and pressure,
m
T = stop time, min,
T = start time, min,
T – T = total sampling time, min, and
2 1
Q = average flow rate, mL/min.
A
10.1.14 The total volume (V ) at standard conditions, 25°C25 °C and 101.3 kPa, is calculated from the following equation:
m
P 298
A
V 5 V 3 3 (3)
s m
101.3 2731t
A
where:
V = total sample volume, L, at 25°C and 101.3-kPa pressure,
s
V = total sample volume, L, at 25 °C and 101.3-kPa pressure,
s
V = total sample volume, L, at measured temperature and pressure,
m
P = average pressure, kPa, and
A
t = average temperature, °C.
A
10.2 Sample Analysis:
10.2.1 Sample Storage—The samples Samples are returned to the laboratory in an insulated box container at <4 °C with
appropriate padding and stored in a refrigerator at <4°C<4 °C until analysis. Alternatively, the samples may also be stored alone
in a refrigerator at <4°C<4 °C in their individual glass containers or pouches. The time between sampling and analysis shouldmust
not exceed 14 days.
10.2.2 Sample Desorption:
NOTE 14—Sample desorption can be automated provided quantitative recovery during extraction is demonstrated, for example, by performance testing.
10.2.2.1 Connect the sample cartridge (inlet or short end during sampling) to a clean syringe.
NOTE 15—The liquid flow during desorption should be in the same direction as the air flow during sampling to prevent insoluble particulates from getting
into the eluate. Reverse desorption may be performed if the eluate is filtered prior to HPL
...


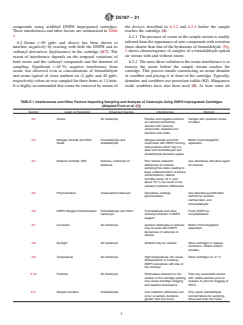
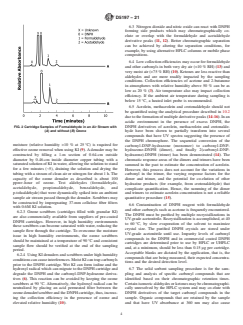



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...