ASTM D7493-22
(Test Method)Standard Test Method for Online Measurement of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatograph and Electrochemical Detection
Standard Test Method for Online Measurement of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatograph and Electrochemical Detection
SIGNIFICANCE AND USE
5.1 Gaseous fuels, such as natural gas, petroleum gases and bio-gases, contain sulfur compounds that are naturally occurring or that are added as odorants for safety purposes. These sulfur compounds are odorous, toxic, corrosive to equipment, and can inhibit or destroy catalysts employed in gas processing and other end uses. Their accurate continuous measurement is important to gas processing, operation and use, and is frequently of regulatory interest.
5.2 Small amounts (typically, total of 4 to 6 ppm(v)) of sulfur odorants are added to natural gas and other fuel gases for safety purposes. Some sulfur odorants are reactive and may be oxidized to form more stable sulfur compounds having higher odor thresholds which adversely impact the potential safety of the gas delivery systems and gas users. Gaseous fuels are analyzed for sulfur compounds and odorant levels to assist in pipeline integrity surveillance and to ensure appropriate odorant levels for public safety.
5.3 This method offers an on-line method to continuously identify and quantify individual target sulfur species in gaseous fuel with automatic calibration and validation.
SCOPE
1.1 This test method is for on-line measurement of gas phase sulfur-containing compounds in gaseous fuels by gas chromatography (GC) and electrochemical (EC) detection. This test method is applicable to hydrogen sulfide, C1 to C4 mercaptans, sulfides, and tetrahydrothiophene (THT).
1.1.1 Carbonyl sulfide (COS) is not measured according to this test method.
1.1.2 The detection range for sulfur compounds is approximately from 0.1 to 100 ppm(v) (mL/m3) or 0.1 to 100 mg/m3 at 25 °C, 101.3 kPa. The detection range will vary depending on the sample injection volume, chromatographic peak separation, and the sensitivity of the specific EC detector.
1.2 This test method describes a GC-EC method using capillary GC columns and a specific detector for natural gas and other gaseous fuels composed of mainly light (C4 and smaller) hydrocarbons. Alternative GC columns including packed columns, detector designs, and instrument parameters may be used, provided that chromatographic separation, quality control, and measurement objectives needed to comply with user or regulator needs, or both, are achieved.
1.3 This test method does not intend to identify and measure all individual sulfur species and is mainly employed for monitoring naturally occurring reduced sulfur compounds commonly found in natural gas and fuel gases or employed as an odorant in these gases.
1.4 This test method is typically employed in repetitive or continuous on-line monitoring of sulfur components in natural gas and fuel gases using a single sulfur calibration standard. Guidance for producing calibration curves specific to particular analytes or enhanced quality control procedures can be found in Test Methods D5504, D5623, D6228, D6968, ISO 19739, or GPA 2199.
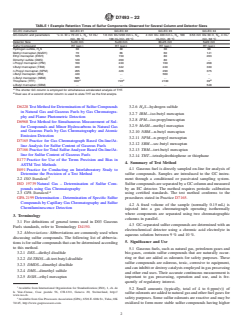
1.5 The test method can be used for measuring sulfur compounds listed in Table 1 in air or other gaseous matrices, provided that compounds that can interfere with the GC separation and electrochemical detection are not present.
1.6 This test method is written as a companion to Practices D5287, D7165 and D7166.
1.7 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Tec...
General Information
- Status
- Published
- Publication Date
- 31-Oct-2022
- Technical Committee
- D03 - Gaseous Fuels
- Drafting Committee
- D03.12 - On-Line/At-Line Analysis of Gaseous Fuels
Relations
- Effective Date
- 01-Mar-2024
- Effective Date
- 01-Oct-2023
- Effective Date
- 15-Dec-2019
- Refers
ASTM D4626-95(2019) - Standard Practice for Calculation of Gas Chromatographic Response Factors - Effective Date
- 01-Dec-2019
- Effective Date
- 01-Jul-2016
- Refers
ASTM D5287-08(2015) - Standard Practice for Automatic Sampling of Gaseous Fuels (Withdrawn 2024) - Effective Date
- 01-Jun-2015
- Effective Date
- 01-May-2014
- Effective Date
- 15-Jan-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Dec-2010
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-Apr-2010
- Effective Date
- 01-Jan-2010
Overview
ASTM D7493-22 is a globally recognized standard test method developed by ASTM International for the online measurement of sulfur compounds in natural gas and gaseous fuels. Using gas chromatography (GC) combined with electrochemical detection (EC), this method enables real-time identification and quantification of key sulfur-containing species, including hydrogen sulfide, mercaptans (C1–C4), sulfides, and tetrahydrothiophene (THT). Accurate detection of sulfur compounds is crucial for gas processing, system integrity, equipment protection, regulatory compliance, and public safety, particularly because sulfur compounds can be odorous, toxic, corrosive, and catalyst inhibitors.
Key Topics
- Continuous and Online Monitoring: The test method describes procedures for continuous, online measurement, ideal for pipeline monitoring and process control where real-time data are required.
- Sulfur Compound Scope: Applicable to most commonly found sulfur compounds in fuel gases, including:
- Hydrogen sulfide (H₂S)
- Methyl, ethyl, propyl, butyl mercaptans
- Dimethyl sulfide, tetrahydrothiophene (THT)
- Detection Range: Sulfur compounds can be reliably measured in the range of 0.1 to 100 ppm(v) (or mg/m³), depending on equipment and sample size.
- Calibration and Validation: The standard includes guidance for regular calibration using certified sulfur standards and addresses quality control protocols to ensure measurement accuracy.
- Technical Flexibility: While recommending capillary GC columns and EC detectors, the standard permits alternative columns, detector designs, or parameters, provided required separation, quality assurance, and regulatory objectives are met.
Applications
ASTM D7493-22 provides significant value across several sectors:
- Pipeline Integrity and Safety: Monitoring sulfur and odorant levels helps prevent corrosion, ensures proper odorization, and detects leaks, thereby supporting public safety and compliance with legal requirements.
- Gas Processing Facilities: By detecting sulfur compounds, operators can protect sensitive equipment and catalysts from contamination or deactivation.
- Regulatory Compliance: The method supports adherence to environmental and safety regulations that limit sulfur emissions and mandate specific odorant concentrations.
- Quality Assurance: Continuous, online analysis offers reliable data for product certification and transactional transparency in natural gas supply.
- Research and Development: Served as a reference for developing new analytical instrumentation or improving existing gas analysis workflows, especially where sulfur control is critical.
Related Standards
ASTM D7493-22 is designed to work in conjunction with, or as an alternative to, other international and ASTM standards addressing sulfur compound analysis in gaseous fuels:
- ASTM D5504: Sulfur compounds measurement by gas chromatography and chemiluminescence.
- ASTM D5623: Sulfur compounds in light petroleum liquids by GC with sulfur-selective detection.
- ASTM D6228: Sulfur compounds in gaseous fuels by GC and flame photometric detection.
- ASTM D6968: Simultaneous measurement of sulfur compounds and minor hydrocarbons in natural gas by GC and atomic emission detection.
- ASTM D5287, D7165, D7166: Practices for sampling and online analysis of gaseous fuels.
- ISO 19739: Determination of sulfur compounds in natural gas by gas chromatography.
- GPA 2199: For specific sulfur compounds by capillary GC and chemiluminescence.
By adhering to ASTM D7493-22, gas industry professionals can ensure robust, reproducible analysis of sulfur levels in natural gas and fuel gases, supporting both operational efficiency and regulatory compliance through state-of-the-art online measurement capabilities.
Buy Documents
ASTM D7493-22 - Standard Test Method for Online Measurement of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatograph and Electrochemical Detection
REDLINE ASTM D7493-22 - Standard Test Method for Online Measurement of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatograph and Electrochemical Detection
Get Certified
Connect with accredited certification bodies for this standard

ABS Quality Evaluations Inc.
American Bureau of Shipping quality certification.

Element Materials Technology
Materials testing and product certification.

ABS Group Brazil
ABS Group certification services in Brazil.
Sponsored listings
Frequently Asked Questions
ASTM D7493-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Online Measurement of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatograph and Electrochemical Detection". This standard covers: SIGNIFICANCE AND USE 5.1 Gaseous fuels, such as natural gas, petroleum gases and bio-gases, contain sulfur compounds that are naturally occurring or that are added as odorants for safety purposes. These sulfur compounds are odorous, toxic, corrosive to equipment, and can inhibit or destroy catalysts employed in gas processing and other end uses. Their accurate continuous measurement is important to gas processing, operation and use, and is frequently of regulatory interest. 5.2 Small amounts (typically, total of 4 to 6 ppm(v)) of sulfur odorants are added to natural gas and other fuel gases for safety purposes. Some sulfur odorants are reactive and may be oxidized to form more stable sulfur compounds having higher odor thresholds which adversely impact the potential safety of the gas delivery systems and gas users. Gaseous fuels are analyzed for sulfur compounds and odorant levels to assist in pipeline integrity surveillance and to ensure appropriate odorant levels for public safety. 5.3 This method offers an on-line method to continuously identify and quantify individual target sulfur species in gaseous fuel with automatic calibration and validation. SCOPE 1.1 This test method is for on-line measurement of gas phase sulfur-containing compounds in gaseous fuels by gas chromatography (GC) and electrochemical (EC) detection. This test method is applicable to hydrogen sulfide, C1 to C4 mercaptans, sulfides, and tetrahydrothiophene (THT). 1.1.1 Carbonyl sulfide (COS) is not measured according to this test method. 1.1.2 The detection range for sulfur compounds is approximately from 0.1 to 100 ppm(v) (mL/m3) or 0.1 to 100 mg/m3 at 25 °C, 101.3 kPa. The detection range will vary depending on the sample injection volume, chromatographic peak separation, and the sensitivity of the specific EC detector. 1.2 This test method describes a GC-EC method using capillary GC columns and a specific detector for natural gas and other gaseous fuels composed of mainly light (C4 and smaller) hydrocarbons. Alternative GC columns including packed columns, detector designs, and instrument parameters may be used, provided that chromatographic separation, quality control, and measurement objectives needed to comply with user or regulator needs, or both, are achieved. 1.3 This test method does not intend to identify and measure all individual sulfur species and is mainly employed for monitoring naturally occurring reduced sulfur compounds commonly found in natural gas and fuel gases or employed as an odorant in these gases. 1.4 This test method is typically employed in repetitive or continuous on-line monitoring of sulfur components in natural gas and fuel gases using a single sulfur calibration standard. Guidance for producing calibration curves specific to particular analytes or enhanced quality control procedures can be found in Test Methods D5504, D5623, D6228, D6968, ISO 19739, or GPA 2199. 1.5 The test method can be used for measuring sulfur compounds listed in Table 1 in air or other gaseous matrices, provided that compounds that can interfere with the GC separation and electrochemical detection are not present. 1.6 This test method is written as a companion to Practices D5287, D7165 and D7166. 1.7 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Tec...
SIGNIFICANCE AND USE 5.1 Gaseous fuels, such as natural gas, petroleum gases and bio-gases, contain sulfur compounds that are naturally occurring or that are added as odorants for safety purposes. These sulfur compounds are odorous, toxic, corrosive to equipment, and can inhibit or destroy catalysts employed in gas processing and other end uses. Their accurate continuous measurement is important to gas processing, operation and use, and is frequently of regulatory interest. 5.2 Small amounts (typically, total of 4 to 6 ppm(v)) of sulfur odorants are added to natural gas and other fuel gases for safety purposes. Some sulfur odorants are reactive and may be oxidized to form more stable sulfur compounds having higher odor thresholds which adversely impact the potential safety of the gas delivery systems and gas users. Gaseous fuels are analyzed for sulfur compounds and odorant levels to assist in pipeline integrity surveillance and to ensure appropriate odorant levels for public safety. 5.3 This method offers an on-line method to continuously identify and quantify individual target sulfur species in gaseous fuel with automatic calibration and validation. SCOPE 1.1 This test method is for on-line measurement of gas phase sulfur-containing compounds in gaseous fuels by gas chromatography (GC) and electrochemical (EC) detection. This test method is applicable to hydrogen sulfide, C1 to C4 mercaptans, sulfides, and tetrahydrothiophene (THT). 1.1.1 Carbonyl sulfide (COS) is not measured according to this test method. 1.1.2 The detection range for sulfur compounds is approximately from 0.1 to 100 ppm(v) (mL/m3) or 0.1 to 100 mg/m3 at 25 °C, 101.3 kPa. The detection range will vary depending on the sample injection volume, chromatographic peak separation, and the sensitivity of the specific EC detector. 1.2 This test method describes a GC-EC method using capillary GC columns and a specific detector for natural gas and other gaseous fuels composed of mainly light (C4 and smaller) hydrocarbons. Alternative GC columns including packed columns, detector designs, and instrument parameters may be used, provided that chromatographic separation, quality control, and measurement objectives needed to comply with user or regulator needs, or both, are achieved. 1.3 This test method does not intend to identify and measure all individual sulfur species and is mainly employed for monitoring naturally occurring reduced sulfur compounds commonly found in natural gas and fuel gases or employed as an odorant in these gases. 1.4 This test method is typically employed in repetitive or continuous on-line monitoring of sulfur components in natural gas and fuel gases using a single sulfur calibration standard. Guidance for producing calibration curves specific to particular analytes or enhanced quality control procedures can be found in Test Methods D5504, D5623, D6228, D6968, ISO 19739, or GPA 2199. 1.5 The test method can be used for measuring sulfur compounds listed in Table 1 in air or other gaseous matrices, provided that compounds that can interfere with the GC separation and electrochemical detection are not present. 1.6 This test method is written as a companion to Practices D5287, D7165 and D7166. 1.7 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.9 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Tec...
ASTM D7493-22 is classified under the following ICS (International Classification for Standards) categories: 75.160.30 - Gaseous fuels. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D7493-22 has the following relationships with other standards: It is inter standard links to ASTM D5623-24, ASTM D4626-23, ASTM D4150-19, ASTM D4626-95(2019), ASTM D4150-08(2016), ASTM D5287-08(2015), ASTM E177-14, ASTM D5623-94(2014), ASTM E691-13, ASTM E177-13, ASTM E691-11, ASTM D6228-10, ASTM E177-10, ASTM D3609-00(2010), ASTM D7166-10. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D7493-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D7493 − 22
Standard Test Method for
Online Measurement of Sulfur Compounds in Natural Gas
and Gaseous Fuels by Gas Chromatograph and
Electrochemical Detection
This standard is issued under the fixed designation D7493; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.5 The test method can be used for measuring sulfur
compounds listed in Table 1 in air or other gaseous matrices,
1.1 This test method is for on-line measurement of gas
provided that compounds that can interfere with the GC
phase sulfur-containing compounds in gaseous fuels by gas
separation and electrochemical detection are not present.
chromatography (GC) and electrochemical (EC) detection.
This test method is applicable to hydrogen sulfide, C1 to C4 1.6 This test method is written as a companion to Practices
mercaptans, sulfides, and tetrahydrothiophene (THT). D5287, D7165 and D7166.
1.1.1 Carbonyl sulfide (COS) is not measured according to
1.7 Units—The values stated in SI units are to be regarded
this test method.
as standard. No other units of measurement are included in this
1.1.2 The detection range for sulfur compounds is approxi-
standard.
3 3
mately from 0.1 to 100 ppm(v) (mL/m ) or 0.1 to 100 mg/m
1.8 This standard does not purport to address all of the
at 25 °C, 101.3 kPa. The detection range will vary depending
safety concerns, if any, associated with its use. It is the
on the sample injection volume, chromatographic peak
responsibility of the user of this standard to establish appro-
separation, and the sensitivity of the specific EC detector.
priate safety, health, and environmental practices and deter-
1.2 This test method describes a GC-EC method using
mine the applicability of regulatory limitations prior to use.
capillary GC columns and a specific detector for natural gas
1.9 This international standard was developed in accor-
and other gaseous fuels composed of mainly light (C4 and
dance with internationally recognized principles on standard-
smaller) hydrocarbons. Alternative GC columns including
ization established in the Decision on Principles for the
packed columns, detector designs, and instrument parameters
Development of International Standards, Guides and Recom-
may be used, provided that chromatographic separation, qual-
mendations issued by the World Trade Organization Technical
ity control, and measurement objectives needed to comply with
Barriers to Trade (TBT) Committee.
user or regulator needs, or both, are achieved.
2. Referenced Documents
1.3 This test method does not intend to identify and measure
all individual sulfur species and is mainly employed for
2.1 ASTM Standards:
monitoring naturally occurring reduced sulfur compounds
D3609 Practice for Calibration Techniques Using Perme-
commonly found in natural gas and fuel gases or employed as
ation Tubes
an odorant in these gases.
D4150 Terminology Relating to Gaseous Fuels
D4626 Practice for Calculation of Gas Chromatographic
1.4 This test method is typically employed in repetitive or
Response Factors
continuous on-line monitoring of sulfur components in natural
D5287 Practice for Automatic Sampling of Gaseous Fuels
gas and fuel gases using a single sulfur calibration standard.
D5504 Test Method for Determination of Sulfur Compounds
Guidance for producing calibration curves specific to particular
in Natural Gas and Gaseous Fuels by Gas Chromatogra-
analytes or enhanced quality control procedures can be found
phy and Chemiluminescence
in Test Methods D5504, D5623, D6228, D6968, ISO 19739, or
D5623 Test Method for Sulfur Compounds in Light Petro-
GPA 2199.
leum Liquids by Gas Chromatography and Sulfur Selec-
tive Detection
This test method is under the jurisdiction of ASTM Committee D03 on Gaseous
Fuels and is the direct responsibility of Subcommittee D03.12 on On-Line/At-Line
Analysis of Gaseous Fuels. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Nov. 1, 2022. Published April 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 2008. Last previous edition approved in 2018 as D7493 – 14 (2018). Standards volume information, refer to the standard’s Document Summary page on
DOI: 10.1520/D7493-22. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7493 − 22
TABLE 1 Example Retention Times of Sulfur Components Observed for Several Column and Detector Sizes
GC-EC instrument GC-EC #1 GC-EC #2 GC-EC #3 GC-EC #4
GC-Column and parameters ⁄8 in. ID × 70 cm L, N , 12 mL/ 1.6 mm ID×1200 mm L, N , 4 mm ID× 400 mm L, N , 100 0.53 mm IDx 30 m, N , 4 mL/
2 2 2 2
min, 65 °C 100 mL/min, 20 °C mL/min, 20 °C min, 58 °C
Detector Size 5×20 mm 5×20 mm 30×25 mm 5x20 mm
Sulfur Compound RT (sec.) RT (sec.) RT (sec.) RT (sec.)
Hydrogen sulfide, H S 30 30 30 98
Methyl mercaptan (MeSH) 70 66 60 141
Ethyl mercaptan (EtSH) 105 150 80 203
Dimethyl sulfide (DMS) 120 200 80 .
i-Propyl mercaptan (IPM) 160 240 160 268
t-Butyl mercaptan (TBM) 220 342 240 330
n-Propyl mercaptan (NPM) 265 426 290 375
i-Butyl mercaptan (IBM) 440 . 560 .
n-Butyl mercaptan (NBM) 585 . . .
A A B
Thiophane (THT) 900 720 2100 39
s-Butyl mercaptan (SBM) . . . 548
A
The shorter GC column is employed for simultaneous accelerated analysis of THT.
B
Dual use of a second shorter column is used to elute THT as the first analyte.
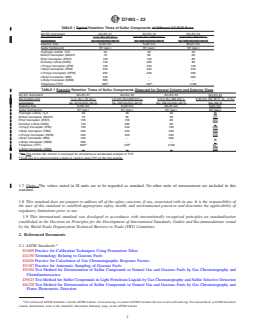
D6228 Test Method for Determination of Sulfur Compounds 3.2.6 H S—hydrogen sulfide
in Natural Gas and Gaseous Fuels by Gas Chromatogra-
3.2.7 IBM—iso-butyl mercaptan
phy and Flame Photometric Detection
3.2.8 IPM—iso-propylmercaptan
D6968 Test Method for Simultaneous Measurement of Sul-
3.2.9 MeSH—methyl mercaptan
fur Compounds and Minor Hydrocarbons in Natural Gas
and Gaseous Fuels by Gas Chromatography and Atomic 3.2.10 NBM—n-butyl mercaptan
Emission Detection
3.2.11 NPM—n-propyl mercaptan
D7165 Practice for Gas Chromatograph Based On-line/At-
3.2.12 SBM—sec-butyl mercaptan
line Analysis for Sulfur Content of Gaseous Fuels
3.2.13 TBM—tert-butyl mercaptan
D7166 Practice for Total Sulfur Analyzer Based On-line/At-
line for Sulfur Content of Gaseous Fuels
3.2.14 THT—tetrahydrothiophene or thiophane
E177 Practice for Use of the Terms Precision and Bias in
4. Summary of Test Method
ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to 4.1 Gaseous fuel is directly sampled on-line for analysis of
Determine the Precision of a Test Method sulfur compounds. Samples are introduced to the GC instru-
2.2 ISO Standard: ment through a conditioned or passivated sampling system.
ISO 19739 Natural Gas – Determination of Sulfur Com- Sulfur compounds are separated by a GC column and measured
pounds using Gas Chromatography by an EC detector. The method requires periodic calibration
2.3 GPA Standard: using certified standards. The test method conforms to the
procedures stated in Practice D7165.
GPA 2199 Determination - Determination of Specific Sulfur
Compounds by Capillary Gas Chromatography and Sulfur
4.2 A fixed volume of the sample (normally 0.15 mL) is
Chemiluminescence Detection
injected into a gas chromatograph operating isothermally
where components are separated using two chromatographic
3. Terminology
columns in parallel.
3.1 For definitions of general terms used in D03 Gaseous
4.3 GC-separated sulfur compounds are determined with an
Fuels standards, refer to Terminology D4150.
electrochemical detector using a chromic acid electrolyte in
3.2 Abbreviations: Abbreviations are commonly used when
aqueous solution between 9 % and 10 %.
discussing sulfur compounds. The following list of abbrevia-
5. Significance and Use
tions is for sulfur compounds that can be determined according
to this method.
5.1 Gaseous fuels, such as natural gas, petroleum gases and
3.2.1 DES—diethyl disulfide
bio-gases, contain sulfur compounds that are naturally occur-
ring or that are added as odorants for safety purposes. These
3.2.2 DI-TBDS—di-tert-butyl-disulfide
sulfur compounds are odorous, toxic, corrosive to equipment,
3.2.3 DMDS—dimethyl disulfide
and can inhibit or destroy catalysts employed in gas processing
3.2.4 DMS—dimethyl sulfide
and other end uses. Their accurate continuous measurement is
3.2.5 EtSH—ethyl mercaptan
important to gas processing, operation and use, and is fre-
quently of regulatory interest.
Available from International Organization for Standardization (ISO), 1, ch. de
5.2 Small amounts (typically, total of 4 to 6 ppm(v)) of
la Voie-Creuse, Case postale 56, CH-1211, Geneva 20, Switzerland, http://
sulfur odorants are added to natural gas and other fuel gases for
www.iso.ch.
safety purposes. Some sulfur odorants are reactive and may be
Available from Gas Processors Association (GPA), 6526 E. 60th St., Tulsa, OK
74145, http://www.gasprocessors.com. oxidized to form more stable sulfur compounds having higher
D7493 − 22
odor thresholds which adversely impact the potential safety of volume sample loops (0.05 to 0.15 mL) may be used to target
the gas delivery systems and gas users. Gaseous fuels are multiple concentration ranges for components in a gas, pro-
analyzed for sulfur compounds and odorant levels to assist in
vided chromatographic separation and quality control objec-
pipeline integrity surveillance and to ensure appropriate odor-
tives are obtained. The same non-reactive materials are used
ant levels for public safety.
for the sample loop to minimize decomposition or absorption
of reactive species. The sampling and GC inlet system must be
5.3 This method offers an on-line method to continuously
well conditioned or passivated and evaluated frequently for
identify and quantify individual target sulfur species in gaseous
compatibility with trace quantities of reactive sulfur
fuel with automatic calibration and validation.
compounds, such as tert-butyl mercaptan (TBM). A program-
6. Apparatus
mable and computer-controlled multi-stream sample selector
can be used to sample fuel gases and calibration gases.
6.1 Chromatograph—Industrial gas chromatograph with an
isothermal oven, automatic injection valve, and software nec-
6.1.2 Column Temperature—The GC must be capable of
essary for interfacing to a chromic acid electrochemical detec-
maintaining an isothermal temperature, normally at 58 °C, with
tor and designed for the intended application. The GC system
temperature variation not exceeding 60.5 °C.
must be inert, well-conditioned and passivated with a gas
6.1.3 Carrier and Detector Gas Control—Constant flow
containing the sulfur compounds of interest to ensure reliable
control of carrier and detector gases is necessary for optimal
results.
and consistent analytical performance. Control is best provided
6.1.1 Sample Inlet System—The gas sample is introduced to
by use of pressure regulators like Piezo valves and fixed flow
the GC by sample loop injection. An automated non-reactive
restrictors. Piezo valves allow for remote adjustments when
gas sampling valve is employed for a fixed sample loop
needed. The gas flow is measured using a gas flow meter either
injection. The sample injection port must be heated continu-
volumetrically or based upon mass flow rates. Mass flow
ously at a temperature significantly (~10 °C) above the tem-
controllers, capable of maintaining gas flow constant to 61 %
perature at which the gas is sampled to avoid sample conden-
at the required flow rates are used. The supply pressure of the
sation and discrimination. Inert tubing made of non-permeable,
gas delivered to the gas chromatograph must be at least 69 kPa
non-sorbing and non-reactive materials, as short as possible
greater than the regulated gas at the instrument to compensate
and heat traced at the same temperature, is employed for
for system back pressure.
transferring the sample from a sample source to the gas
sampling valve and to the GC inlet system. Silica-coated 316 6.1.4 Detector—An EC detector, whose operation is based
upon the reduction/oxidation reaction between reduced sulfur
stainless steel (s.s.) and non-permeable polytetrafluoroethylene
(PTFE) type tubing are often employed. Different size fixed- compounds and a solution of chromic acid (Fig. 1), is used in
FIG. 1 Typical Electrochemical Detection Cell
D7493 − 22
this method. The detector is set according to the manufacturer’s 6.3.1.1 Graphic presentation of the chromatogram.
specifications for this application.
6.3.1.2 Digital display of chromatographic peak areas.
6.1.4.1 The detector consists of an electrochemical system 6.3.1.3 Identification of peaks by retention time or relative
made of glass and a container made of glass or methyl retention time, or both.
polymethacrylate. The electrodes, two pieces of platinum 6.3.1.4 Calculation and use of response factors.
gauze grids, are arranged vertically in parallel, and are welded
6.3.1.5 External standard calculation and data presentation.
in a borosilicate glass tube. These grids are isolated from each
6.3.1.6 Instrument control for electrochemical detector
other and other conductive materials and connected to an
operation, such as gas pressure and flow control.
amplifier for data acquisition.
6.1.4.2 The electrolyte, a solution of chromium (VI) oxide
7. Reagents and Materials
in distilled or deionized water (100 g ⁄L or 0.66 mole ⁄L), is
7.1 High-pressure Cylinder Reference Gas Standards—Gas
contained in an acid-resistant vessel. A tube fitted with the
standards of high purity with certified stability and accuracy
electrodes is dipped into the solution such that the liquid is
are used.
retained by capillary action within the tube at a level approxi-
7.1.1 Sulfur Gas Standards—Single or multiple sulfur com-
mately midway between two grids.
pounds in a compressed gas of high purity nitrogen, helium, or
6.1.4.3 The gas flow from the GC column is discharged
methane base gas may be used. Care must be exercised in the
through a narrow glass or PTFE tube (0.7 mm ID) immediately
use of compressed gas standards since they can introduce
above the upper grid center (normally 5 mm). Each sulfur
errors in measurement due to lack of uniformity in their
compound sequentially elutes and reacts with chromic acid.
manufacture or analyte instability. The non-mandatory proto-
Possible reaction mechanisms are illustrated as Eq 1 and Eq 2.
col for compressed gas standards cited in Appendix X1 of Test
The redox reaction occurs on the electrode surface, creating a
Method D5504 can be used to ensure the quality of standards
potential difference between the two electrodes, thus causing a
and to establish traceability to a National Institute of Standards
current to be measured (using a low resistance measuring
and Technology (NIST), national metrology institute (NMI), or
circuit). For example, TBM is oxidized to t-butyl sulfoxide and
other standard reference material (SRM).
chromium oxide (Eq 2).
7.1.2 Multiple sulfur gas standard mixes should be used as
2 CrO 12 R 2 SH→2 RS 5 O1Cr O (1)
3 2 3 recommended by a compressed gas standard manufacturer to
assure the long-term stability of sulfur compounds. The stan-
where:
dard should by re-certified as per manufacturers’ recommen-
R = organic moieties, such as CxHy.
dations or as needed for regulatory compliance.
2 CrO 12 C H 2 SH→2 C H 2 SO1Cr O 1H O (2)
7.1.3 Compressed Gas Standard Delivery System—Pressure
3 4 9 4 9 2 3 2
regulators, gas lines, and fittings must be appropriate for the
6.2 Column—30 m of 0.53 mm ID metallic capillary col-
delivery of sulfur gases and well passivated or inert.
umn with 3 μm film thickness of 624 or equivalent stationary
phase has been successfully used in performance of this test
7.2 Sulfur Permeation Standards—Gaseous standards gen-
method. However, other columns including packed columns
erated from individual or a combination of certified permeation
that provide adequate retention and resolution characteristics
tubes and devices at a constant temperature (60.1 °C) and a
under the experimental conditions as described in 8.1 can be
constant flow rate can be used for calibrations. The standard
used. A second GC column of the same ID and phase, but of a
concentration is calculated by mass loss at a fixed temperature
shorter length, can be employed for faster measurement of
and dilution gas flow rate. Permeation devices should be
late-eluting sulfur compounds such as THT. In this case, two
calibrated gravimetrically to the nearest 0.01 mg. Impurities
columns are connected to the GC injection system using a
permeated from each device must be detected, measured, and
10-port valve to direct sample flow through the appropriate
accounted for in the mass loss if they are present above a level
column and then onto the EC detector; thus, allowing measure-
of 0.1 % of the permeated sulfur species. Permeation devices
ment of low molecular weight sulfur gases and high molecular
shall be discarded when the liquid content is reduced to less
weight sulfur gases such as THT from a single sample
than 10 % of the initial volume. See Practice D3609 for further
injection, THT may exit first in such mounting of columns if a
information on the proper use of permeation devices.
dual column (column commutation) is used. The elution of
7.2.1 Warning—Sulfur compounds may be flammable and
high molecular weight sulfur gases such as THT may also be
may be harmful if ingested or inhaled.
accelerated by sped up carrier gas flow rate after the elution of
7.3 Carrier Gas—Helium or nitrogen with a minimum
TBM. When samples may contain high boiling or instrument
purity of 99.999 % with maximum concentrations of 1 ppm(v)
damaging substances, a backflush column may be employed to
oxygen, 1 ppm(v) water, and 0.5 ppm(v) total hydrocarbons.
remove these materials before they reach the chromatographic
Use of air as a carrier gas is not recommended since many
column and EC dete
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D7493 − 14 (Reapproved 2018) D7493 − 22
Standard Test Method for
Online Measurement of Sulfur Compounds in Natural Gas
and Gaseous Fuels by Gas Chromatograph and
Electrochemical Detection
This standard is issued under the fixed designation D7493; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method is for on-line measurement of volatile gas phase sulfur-containing compounds in gaseous fuels by gas
chromatography (GC) and electrochemical (EC) detection. This test method is applicable to hydrogen sulfide, C1 to C4
mercaptans, sulfides, and tetrahydrothiophene (THT).
1.1.1 Carbonyl sulfide (COS) is not covered in measured according to this test method.
3 3
1.1.2 The detection range for sulfur compounds is approximately from 0.1 to 100 ppmvppm(v) (mL/m ) or 0.1 to 100 mg/m . at
25 °C, 101.3 kPa. The detection range maywill vary depending on the sample injection volume, chromatographic peak separation,
and the sensitivity toof the specific EC detector.
1.2 This test method describes a GC-EC method employing packedusing capillary GC columns and a specific detector for natural
gas and other gaseous fuelfuels composed of mainly light (C4 and smaller) hydrocarbons. Alternative GC columns including
packed columns, detector designs, and instrument parameters may be used, provided that chromatographic separation, quality
control, and measurement objectives needed to comply with user,user or regulator needs, or both, are achieved.
1.3 This test method does not intend to identify and measure all individual sulfur species,species and is mainly employed for
monitoring naturally occurring reduced sulfur compounds commonly found in natural gas and fuel gases or employed as an odorant
in these gases.
1.4 TheThis test method is typically employed in repetitive or continuous on-line monitoring of sulfur components in natural gas
and fuel gases using a single sulfur calibration standard. Need for a multipoint calibration curve or Guidance for producing
calibration curves specific to particular analytes or enhanced quality control procedures can be satisfied by making use of
procedures delineated found in Test Methods D5504, D5623, D6228, D6968, ISO 19739, or GPA 2199.
1.5 The test method can be used for measurement of all measuring sulfur compounds listed in Table 1 in air or other gaseous
matrices, provided that no compounds that can interfere with the GC separation and electrochemical detection are not present.
1.6 This test method is written as a companion to Practices D5287, D7165 and D7166.
This test method is under the jurisdiction of ASTM Committee D03 on Gaseous Fuels and is the direct responsibility of Subcommittee D03.12 on On-Line/At-Line
Analysis of Gaseous Fuels.
Current edition approved July 1, 2018Nov. 1, 2022. Published July 2018April 2023. Originally approved in 2008. Last previous edition approved in 20142018 as
D7493D7493 – 14 (2018).-14. DOI: 10.1520/D7493-14R18.10.1520/D7493-22.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D7493 − 22
TABLE 1 Typical Retention Times of Sulfur Components of Different GC-ECD Runs
GC-EC instrument GC-EC #1 GC-EC #2 GC-EC #3
GC-Column and ⁄8 in. ID× 70 cm L, 1.6 mm ID× 1200 mm L, 4 mm ID× 400 mm L,
parameters N , 12 mL/min, 65 °C N , 100 mL/min, 20 °C N , 100 mL/min, 20 °C
2 2 2
Detector Size 5×20 mm 5×20 mm 30×25 mm
Sulfur Compound RT (sec.) RT (sec.) RT (sec.)
Hydrogen sulfide, H S 30 30 30
Methyl mercaptan (MeSH) 70 66 60
Ethyl mercaptan (EtSH) 105 150 80
Dimethyl sulfide (DMS) 120 200 80
i-Propyl mercaptan (IPM) 160 240 160
t-Butyl mercaptan (TBM) 220 342 240
n-Propyl mercaptan (NPM) 265 426 290
i-Butyl mercaptan (IBM) 440 . 560
n-Butyl mercaptan (NBM) 585 . .
A A
Thiophane (THT) 900 720 2100
TABLE 1 Example Retention Times of Sulfur Components Observed for Several Column and Detector Sizes
GC-EC instrument GC-EC #1 GC-EC #2 GC-EC #3 GC-EC #4
GC-Column and ⁄8 in. ID × 70 cm L, 1.6 mm ID×1200 mm L, 4 mm ID× 400 mm L, 0.53 mm IDx 30 m, N , 4 mL/
parameters N , 12 mL/min, 65 °C N , 100 mL/min, 20 °C N , 100 mL/min, 20 °C min, 58 °C
2 2 2
Detector Size 5×20 mm 5×20 mm 30×25 mm 5x20 mm
Sulfur Compound RT (sec.) RT (sec.) RT (sec.) RT (sec.)
Hydrogen sulfide, H S 30 30 30 98
Methyl mercaptan (MeSH) 70 66 60 141
Ethyl mercaptan (EtSH) 105 150 80 203
Dimethyl sulfide (DMS) 120 200 80 .
i-Propyl mercaptan (IPM) 160 240 160 268
t-Butyl mercaptan (TBM) 220 342 240 330
n-Propyl mercaptan (NPM) 265 426 290 375
i-Butyl mercaptan (IBM) 440 . 560 .
n-Butyl mercaptan (NBM) 585 . . .
A A B
Thiophane (THT) 900 720 2100 39
s-Butyl mercaptan (SBM) . . . 548
A
The The shorter GC column is employed for simultaneous accelerated analysis of THT.
B
Dual use of a second shorter column is used to elute THT as the first analyte.
1.7 Units—The values stated in SI units are to be regarded as standard. No other units of measurement are included in this
standard.
1.8 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of
the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.9 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
D3609 Practice for Calibration Techniques Using Permeation Tubes
D4150 Terminology Relating to Gaseous Fuels
D4626 Practice for Calculation of Gas Chromatographic Response Factors
D5287 Practice for Automatic Sampling of Gaseous Fuels
D5504 Test Method for Determination of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatography and
Chemiluminescence
D5623 Test Method for Sulfur Compounds in Light Petroleum Liquids by Gas Chromatography and Sulfur Selective Detection
D6228 Test Method for Determination of Sulfur Compounds in Natural Gas and Gaseous Fuels by Gas Chromatography and
Flame Photometric Detection
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
D7493 − 22
D6968 Test Method for Simultaneous Measurement of Sulfur Compounds and Minor Hydrocarbons in Natural Gas and Gaseous
Fuels by Gas Chromatography and Atomic Emission Detection
D7165 Practice for Gas Chromatograph Based On-line/At-line Analysis for Sulfur Content of Gaseous Fuels
D7166 Practice for Total Sulfur Analyzer Based On-line/At-line for Sulfur Content of Gaseous Fuels
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
2.2 ISO Standards:Standard:
ISO 19739 Natural gasGas – Determination of sulfur compounds by gas chromatographySulfur Compounds using Gas
Chromatography
2.3 GPA StandardStandard:
GPA 2199 Determination - Determination of Specific Sulfur Compounds by Capillary Gas Chromatography and Sulfur
Chemiluminescence Detection
3. Terminology
3.1 Common terminology used in this method are cited in For definitions of general terms used in D03 Gaseous Fuels standards,
refer to Terminology D4150. Sulfur compounds are commonly referred by their initials (chemical or formula), for example,
3.2 Abbreviations:
hydrogen sulfide = H S
methyl mercaptan = MeSH (MM)
ethyl mercaptan = EtSH (EM)
dimethyl sulfide = DMS
i-Propyl mercaptan = IPM
n-Propyl mercaptan = NPM
t-Butyl mercaptan = TBM
tetrahydrothiophene = THT or Thiophane
3.2 Abbreviations: Abbreviations are commonly used when discussing sulfur compounds. The following list of abbreviations
is for sulfur compounds that can be determined according to this method.
3.2.1 DES—diethyl disulfide
3.2.2 DI-TBDS—di-tert-butyl-disulfide
3.2.3 DMDS—dimethyl disulfide
3.2.4 DMS—dimethyl sulfide
3.2.5 EtSH—ethyl mercaptan
3.2.6 H S—hydrogen sulfide
3.2.7 IBM—iso-butyl mercaptan
3.2.8 IPM—iso-propylmercaptan
3.2.9 MeSH—methyl mercaptan
3.2.10 NBM—n-butyl mercaptan
Available from International Organization for Standardization (ISO), 1, ch. de la Voie-Creuse, Case postale 56, CH-1211, Geneva 20, Switzerland, http://www.iso.ch.
Available from Gas Processors Association (GPA), 6526 E. 60th St., Tulsa, OK 74145, http://www.gasprocessors.com.
D7493 − 22
3.2.11 NPM—n-propyl mercaptan
3.2.12 SBM—sec-butyl mercaptan
3.2.13 TBM—tert-butyl mercaptan
3.2.14 THT—tetrahydrothiophene or thiophane
4. Summary of Test Method
4.1 Gaseous fuel is directly sampled on-line for analysis of sulfur compounds. Samples are introduced to the GC instrument
through a conditioned or passivated sampling system. Sulfur compounds are separated by a GC column and measured by an EC
detector. The method requires periodic calibration using certified standards. The test method conforms to the practicesprocedures
stated in Practice D7165.
4.2 A fixed volume of the sample (normally 0.25 mL) 0.15 mL) is injected into a gas chromatograph operating isothermally where
components are separated using two chromatographic columns.columns in parallel.
4.3 GC-separated sulfur compounds are determined usingwith an electrochemical detector utilizingusing a chromic acid
electrolyte.electrolyte in aqueous solution between 9 % and 10 %.
5. Significance and Use
5.1 Gaseous fuels, such as natural gas, petroleum gases and bio-gases, contain sulfur compounds that are naturally occurring or
that are added as odorants for safety purposes. These sulfur compounds are odorous, toxic, corrosive to equipment, and can inhibit
or destroy catalysts employed in gas processing and other end uses. Their accurate continuous measurement is important to gas
processing, operation and utilization,use, and is frequently of regulatory interest.
5.2 Small amounts (typically, total of 4 to 6 ppmv) 6 ppm(v)) of sulfur odorants are added to natural gas and other fuel gases for
safety purposes. Some sulfur odorants are reactive,reactive and may be oxidized to form more stable sulfur compounds having
lowerhigher odor thresholds which adversely impact the potential safety of the gas delivery systems and gas users. Gaseous fuels
are analyzed for sulfur compounds and odorant levels to assist in pipeline integrity surveillance and to ensure appropriate odorant
levels for public safety.
5.3 This method offers an on-line techniquemethod to continuously identify and quantify individual target sulfur species in
gaseous fuel with automatic calibration and validation.
6. Apparatus
6.1 Chromatograph—Industrial gas chromatograph with an isothermal oven, automatic injection valve, and software necessary for
interfacing to a chromic acid electrochemical detector and designed for the intended application. The GC system must be inert,
well-conditioned and passivated with a gas containing the sulfur compounds of interest to ensure reliable results.
6.1.1 Sample Inlet System—The gas sample is introduced to the GC by sample loop injection. An automated non-reactive gas
sampling valve is employed for a fixed sample loop injection. The sample injection port must be heated continuously at a
temperature significantly (~10 °C) above the temperature at which the gas wasis sampled to avoid sample condensation and
discrimination. Inert tubing made of non-permeable, non-sorbing and non-reactive materials, as short as possible and heat traced
at the same temperature, should be is employed for transferring the sample from a sample source to the gas sampling valve and
to the GC inlet system. Silica-coated 316 stainless steel (s.s.) and non-permeable polytetrafluoroethylene (PTFE) type tubing are
often employed. Different size fixed-volume sample loops (0.25 to 10.0 mL) (0.05 to 0.15 mL) may be used to target multiple
concentration ranges for components in a gas, provided chromatographic separation and quality control objectives are obtained.
The same non-reactive materials are used for the sample loop to avoid possible minimize decomposition or absorption of reactive
species. The sampling and GC inlet system must be well conditioned or passivated and evaluated frequently for compatibility with
trace quantities of reactive sulfur compounds, such as tert-butyl mercaptan. mercaptan (TBM). A programmable and
computer-controlled multi-stream sample selector can be used to sample fuel gases and calibration gases.
D7493 − 22
6.1.2 Column Temperature—The gas chromatograph GC must be capable of maintaining an isothermal temperature, normally at
65 °C,58 °C, with temperature variation not exceeding 60.5 °C.
6.1.3 Carrier and Detector Gas Control—Constant flow control of carrier and detector gases is necessary for optimumoptimal and
consistent analytical performance. Control is best provided by the use of pressure regulators like Piezo valves and fixed flow
restrictors. Piezo valves allow for remote adjustments when needed. The gas flow rate is measured using a gas flow meter either
volumetrically or based upon mass flow rates. Mass flow controllers, capable of maintaining gas flow constant to 61 % 61 % at
the required flow rates should be are used. The supply pressure of the gas delivered to the gas chromatograph must be at least 69
kPa (10 psi) 69 kPa greater than the regulated gas at the instrument to compensate for system back pressure.
6.1.4 Detector—An EC detector, whose operation is based upon the reduction/oxidation reaction between reduced sulfur
compounds and a solution of chromic acid (Fig. 1), is used in this method. The detector is set according to the manufacturer’s
specifications for this particular application. One EC detector is normally employed for measurement. A second column is
employed for detection of late-eluting sulfur compounds, such as THT. application.
6.1.4.1 The detector consists of a an electrochemical system made of glass and a container made of glass or methyl
polymethacrylate container. polymethacrylate. The electrodes, two pieces of platinum gauze grids, are arranged vertically in
parallel, and are welded in a borosilicate glass tube. These grids are isolated from each other and other conductive materials and
connected to an amplifier for data acquisition.
6.1.4.2 The electrolyte, a solution of chromium (VI) oxide in distilled or deionized water (100(100 g g/L or 0.66⁄L or 0.66 mole
mole/L), ⁄L), is contained in an acid-resistant vessel. A tube fitted with the electrodes is dipped into the solution such that the liquid
is retained by capillary action within the tube at a level approximately midway between two grids.
6.1.4.3 The gas flow from the GC column is discharged through a narrow glass or PTFE tube (2 mm (0.7 mm ID) immediately
above the upper grid center (normally 5 mm). 5 mm). Each sulfur compound sequentially elutes and reacts with chromic acid.
Possible reaction mechanisms are illustrated as Eq 1 and Eq 2. The redox reaction occurs on the electrode surface, creating a
potential difference between the two electrodes, thus causing a current to be measured (using a low resistance measuring circuit).
For example, t-butyl mercaptan TBM is oxidized to t-butyl sulfoxide and chromium oxide (Eq 2).
FIG. 1 Typical Electrochemical Detection Cell
D7493 − 22
2 CrO 12 R 2 SH→2 RS 5 O1Cr O (1)
3 2 3
where:
R = organic moieties, such as CxHy
R = organic moieties, such as CxHy.
2 CrO 12 C H 2 SH→2 C H 2 SO1Cr O 1H O (2)
3 4 9 4 9 2 3 2
6.2 Column—1200 mm of 1.6 mm ID glass or PTFE tubing packed with 150 to 180 um (80 to 100 mesh) Chromosorb W support
30 m of 0.53 mm ID metallic capillary column with 3 μm film thickness of 624 or equivalent stationary phase has been successfully
used in performance of this test method. However, other columns including packed columns that provide adequate retention and
resolution characteristics under the experimental conditions as described in 8.1 can be used. A second GC column of the same ID
and phase, but of a shorter length, can be employed for faster measurement of late-eluting sulfur compounds such as THT. In this
case, two columns are connected to the GC injection system using a 10-port valve to direct sample flow through the appropriate
column and then onto the EC detector; thus, allowing measurement of low molecular weight sulfur gases and high molecular
weight sulfur gases such as THT from a single sample injection. injection, THT may exit first in such mounting of columns if a
dual column (column commutation) is used. The elution of high molecular weight sulfur gases such as THT may also be
accelerated by increased sped up carrier gas flow rate after the elution of TBM. When samples may contain high boiling or
instrument damaging substances, a backflush column may be employed to remove these materials before they reach the
chromatographic column and EC detector. The performance of GC columns shall giveprovide adequate separation of target sulfur
compounds for the particular application.
6.3 Data Acquisition
6.3.1 The device and software must have the following capabilities:
6.3.1.1 Graphic presentation of the chromatogram.
6.3.1.2 Digital display of chromatographic peak areas.
6.3.1.3 Identification of peaks by retention time or relative retention time, or both.
6.3.1.4 Calculation and use of response factors.
6.3.1.5 External standard calculation and data presentation.
6.3.1.6 Instrument control for electrochemical detector operation, such as gas pressure and flow control.
7. Reagents and Materials
7.1 High-pressure Cylinder Reference Gas Standards—Gas standards of high purity with certified stability and accuracy are used.
7.1.1 Sulfur Gas Standards—Single or multiple sulfur compounds in a compressed gas of high purity nitrogen, helium, or methane
base gas may be used. Care must be exercised in the use of compressed gas standards since they can introduce errors in
measurement due to lack of uniformity in their manufacture or instability in their storage and use. analyte instability. The
non-mandatory protocol for compressed gas standards cited in Appendix X1 of Test Method D5504 can be used to ensure the
quality of standards and to establish traceability to a National Institute of Standards and Technology (NIST) or other (NIST),
national metrology institute (NMI) (NMI), or other standard reference material.material (SRM).
7.1.2 Multiple sulfur gas standard mixes should be used as recommended by a compressed gas standard manufacturer to assure
the long term long-term stability of sulfur components.compounds. The standard should by re-certified as per manufacturers’
recommendations or as needed for regulatory compliance.
7.1.3 Compressed Gas Standard Delivery System—Pressure regulators, gas lines, and fittings must be inert, appropriate for the
delivery of sulfur gases and well passivated.passivated or inert.
7.2 Sulfur Permeation Standards—Gaseous standards generated from individual or a combination of certified permeation tubes
D7493 − 22
and devices at a constant temperature (60.1 °C) and a constant flow rate can be used for calibrations. The standard concentration
is calculated by mass loss at a fixed temperature and dilution gas flow rate. Permeation devices should be calibrated gravimetrically
to the nearest 0.01 mg. 0.01 mg. Impurities permeated from each device must be detected, measured, and accounted for in the mass
loss if they are present above a level of 0.1 % 0.1 % of the permeated sulfur species. Permeation devices shall be discarded when
the liquid content is reduced to less than 10 % 10 % of the initial volume. See Practice D3609 for further information on the proper
use of permeation devices.
NOTE 1—Warning: Sulfur compounds may be flammable and may be harmful if ingested or inhaled.
7.2.1 Warning—Sulfur compounds may be flammable and ma
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...