ASTM B825-19
(Test Method)Standard Test Method for Coulometric Reduction of Surface Films on Metallic Test Samples
Standard Test Method for Coulometric Reduction of Surface Films on Metallic Test Samples
SIGNIFICANCE AND USE
4.1 The present trend in environmental testing of materials with electrically conductive surfaces is to produce, under accelerated laboratory conditions, corrosion and film-forming reactions that are similar to those that cause failures in service environments. In many of these procedures the parts under test are exposed for days or weeks to controlled quantities of both water vapor and pollutant gases, which may be present in extremely dilute concentrations.
Note 2: Descriptions of such tests can be found in Practice B827.
4.2 Many of these environmental test methods require monitoring of the conditions within the chamber during the test in order to confirm that the intended environmentally related reactions are actually taking place. The most common type of monitor consists of copper, silver, or other thin metallic coupons of a few square centimeters that are placed within the test chamber and that react with the corrosive environment in much the same way as the significant surfaces of the parts under test.
4.3 In practice, a minimum number of control coupons are placed in each specified location (see Test Method B810) within the chamber for a specified exposure time, depending upon the severity of the test environment. At the end of this time interval, the metal samples are removed and analyzed by the coulometric reduction procedure.
4.4 Other corrosion film evaluation techniques for metallic coupons are also available. The most common of these is mass gain, which is nondestructive to the surface films, but is limited to the determination of the total amount of additional mass acquired by the metal as a result of the environmental attack. The most common is weighing using high performance microbalances or for purposes of real-time monitoring, quartz crystal microbalances (see Specification B808).
Note 3: Detailed instructions for conducting such weighings, as well as coupon cleaning and surface preparation procedures, are included as part of Test Met...
SCOPE
1.1 This test method covers procedures and equipment for determining the relative buildup of corrosion and tarnish films (including oxides) on metal surfaces by the constant-current coulometric technique, also known as the cathodic reduction method.
1.2 This test method is designed primarily to determine the relative quantities of tarnish films on control coupons that result from gaseous environmental tests, particularly when the latter are used for testing components or systems containing electrical contacts used in customer product environments.
1.3 This test method may also be used to evaluate test samples that have been exposed to indoor industrial locations or other specific application environments. (See 4.6 for limitations.)
1.4 This test method has been demonstrated to be applicable particularly to copper and silver test samples (see (1)).2 Other metals require further study to prove their applicability within the scope of this test method.
1.5 The values stated in SI units are the preferred units. The values provided in parentheses are for information only.
1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to become familiar with all hazards including those identified in the appropriate Material Safety Data Sheet (MSDS) for this product/material as provided by the manufacturer, to establish appropriate safety, health, and environmental practices, and determine the applicability of regulatory limitations prior to use.
1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Oct-2019
- Technical Committee
- B02 - Nonferrous Metals and Alloys
Relations
- Effective Date
- 01-Aug-2018
- Effective Date
- 01-Oct-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Oct-2011
- Effective Date
- 01-Oct-2010
- Refers
ASTM B827-05(2009)e1 - Standard Practice for Conducting Mixed Flowing Gas (MFG) Environmental Tests - Effective Date
- 01-Oct-2009
- Refers
ASTM B827-05(2009)e2 - Standard Practice for Conducting Mixed Flowing Gas (MFG) Environmental Tests - Effective Date
- 01-Oct-2009
- Effective Date
- 01-Aug-2008
- Effective Date
- 01-Dec-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-May-2005
- Effective Date
- 01-May-2005
- Effective Date
- 01-Nov-2003
- Refers
ASTM B827-97(2003) - Standard Practice for Conducting Mixed Flowing Gas (MFG) Environmental Tests - Effective Date
- 10-Jun-2003
- Effective Date
- 10-Feb-2003
Overview
ASTM B825-19: Standard Test Method for Coulometric Reduction of Surface Films on Metallic Test Samples is a widely recognized test method published by ASTM International. This standard provides procedures and necessary equipment for determining the buildup of corrosion and tarnish films, including oxides, on metal surfaces using the constant-current coulometric (cathodic reduction) technique. The method is particularly suitable for evaluating thin corrosion films formed during environmental testing, especially in accelerated laboratory conditions and real-world applications where metallic materials face exposure to moisture and pollutant gases.
ASTM B825-19 is most often used for copper and silver test samples, which serve as indicators during environmental and corrosion testing. The standard supports industry and research activities by enabling the quantitative assessment of surface film formation on control coupons, helping to ensure that lab-based corrosion tests accurately reflect real-world service conditions.
Key Topics
- Coulometric Reduction Technique: Uses a controlled electric current to reduce tarnish or corrosion films on metal samples, measuring the quantity of charge required to complete the reduction process. This technique offers high-resolution analysis of surface films, resolving complex mixtures into individual components.
- Control Coupons: Thin metal samples, typically copper or silver, are placed within environmental test chambers to mimic the behavior of actual product surfaces under controlled exposure to corrosive atmospheres.
- Test Environments: Applicable to both laboratory simulations (such as mixed flowing gas and humidity exposure) and real-world environments, including indoor industrial settings.
- Film Analysis Alternatives: Alternative methods such as mass gain (weighing) and quartz crystal microbalance are referenced, with distinctions drawn between destructive (coulometric) and non-destructive evaluation techniques.
- Measurement Accuracy: Emphasizes the importance of consistent sample preparation, current density control, and sample geometry to achieve reproducible and meaningful results.
- Film Identification: The technique's resolving power allows for fingerprinting environmental conditions and comparative evaluation of different test environments or chamber runs.
Applications
ASTM B825-19 is valuable for a range of industries and research fields focused on material reliability and product durability:
- Environmental Testing of Electrical Contacts and Components: Essential for industries such as electronics, automotive, and telecom, where electrical contact performance and longevity are influenced by corrosion and tarnish buildup.
- Corrosion Testing Laboratories: Widely applied in labs performing accelerated corrosion or mixed flowing gas (MFG) tests to certify the corrosion resistance of components and systems.
- Quality Assurance: Supports verification that environmental chambers are producing intended reactions by validating with control coupons.
- Material Development and Evaluation: Helps materials scientists assess the corrosion resistance of new alloys and surface treatments under controlled conditions.
- Real-World Exposure Studies: Supports evaluation of metallic samples after exposure to industrial or specific application environments, providing data for service life and maintenance planning.
Related Standards
ASTM B825-19 is intended to be used in conjunction with several other important standards that inform best practices in corrosion testing and coupon analysis:
- ASTM B810: Test Method for Calibration of Atmospheric Corrosion Test Chambers by Change in Mass of Copper Coupons
- ASTM B827: Practice for Conducting Mixed Flowing Gas (MFG) Environmental Tests
- ASTM B808: Test Method for Monitoring of Atmospheric Corrosion Chambers by Quartz Crystal Microbalances
- ASTM B809: Test Method for Porosity in Metallic Coatings by Humid Sulfur Vapor (Flowers-of-Sulfur)
- ASTM D1193: Specification for Reagent Water
Each of these standards supports or complements aspects of environmental testing and coupon evaluation, ensuring coordinated and accurate testing protocols.
Keywords: ASTM B825, coulometric reduction, surface films, metallic coupons, corrosion testing, cathodic reduction method, environmental test chambers, atmospheric corrosion, material reliability, control coupon analysis.
Buy Documents
ASTM B825-19 - Standard Test Method for Coulometric Reduction of Surface Films on Metallic Test Samples
Get Certified
Connect with accredited certification bodies for this standard

National Aerospace and Defense Contractors Accreditation Program (NADCAP)
Global cooperative program for special process quality in aerospace.

CARES (UK Certification Authority for Reinforcing Steels)
UK certification for reinforcing steels and construction.

DVS-ZERT GmbH
German welding certification society.
Sponsored listings
Frequently Asked Questions
ASTM B825-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Coulometric Reduction of Surface Films on Metallic Test Samples". This standard covers: SIGNIFICANCE AND USE 4.1 The present trend in environmental testing of materials with electrically conductive surfaces is to produce, under accelerated laboratory conditions, corrosion and film-forming reactions that are similar to those that cause failures in service environments. In many of these procedures the parts under test are exposed for days or weeks to controlled quantities of both water vapor and pollutant gases, which may be present in extremely dilute concentrations. Note 2: Descriptions of such tests can be found in Practice B827. 4.2 Many of these environmental test methods require monitoring of the conditions within the chamber during the test in order to confirm that the intended environmentally related reactions are actually taking place. The most common type of monitor consists of copper, silver, or other thin metallic coupons of a few square centimeters that are placed within the test chamber and that react with the corrosive environment in much the same way as the significant surfaces of the parts under test. 4.3 In practice, a minimum number of control coupons are placed in each specified location (see Test Method B810) within the chamber for a specified exposure time, depending upon the severity of the test environment. At the end of this time interval, the metal samples are removed and analyzed by the coulometric reduction procedure. 4.4 Other corrosion film evaluation techniques for metallic coupons are also available. The most common of these is mass gain, which is nondestructive to the surface films, but is limited to the determination of the total amount of additional mass acquired by the metal as a result of the environmental attack. The most common is weighing using high performance microbalances or for purposes of real-time monitoring, quartz crystal microbalances (see Specification B808). Note 3: Detailed instructions for conducting such weighings, as well as coupon cleaning and surface preparation procedures, are included as part of Test Met... SCOPE 1.1 This test method covers procedures and equipment for determining the relative buildup of corrosion and tarnish films (including oxides) on metal surfaces by the constant-current coulometric technique, also known as the cathodic reduction method. 1.2 This test method is designed primarily to determine the relative quantities of tarnish films on control coupons that result from gaseous environmental tests, particularly when the latter are used for testing components or systems containing electrical contacts used in customer product environments. 1.3 This test method may also be used to evaluate test samples that have been exposed to indoor industrial locations or other specific application environments. (See 4.6 for limitations.) 1.4 This test method has been demonstrated to be applicable particularly to copper and silver test samples (see (1)).2 Other metals require further study to prove their applicability within the scope of this test method. 1.5 The values stated in SI units are the preferred units. The values provided in parentheses are for information only. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to become familiar with all hazards including those identified in the appropriate Material Safety Data Sheet (MSDS) for this product/material as provided by the manufacturer, to establish appropriate safety, health, and environmental practices, and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The present trend in environmental testing of materials with electrically conductive surfaces is to produce, under accelerated laboratory conditions, corrosion and film-forming reactions that are similar to those that cause failures in service environments. In many of these procedures the parts under test are exposed for days or weeks to controlled quantities of both water vapor and pollutant gases, which may be present in extremely dilute concentrations. Note 2: Descriptions of such tests can be found in Practice B827. 4.2 Many of these environmental test methods require monitoring of the conditions within the chamber during the test in order to confirm that the intended environmentally related reactions are actually taking place. The most common type of monitor consists of copper, silver, or other thin metallic coupons of a few square centimeters that are placed within the test chamber and that react with the corrosive environment in much the same way as the significant surfaces of the parts under test. 4.3 In practice, a minimum number of control coupons are placed in each specified location (see Test Method B810) within the chamber for a specified exposure time, depending upon the severity of the test environment. At the end of this time interval, the metal samples are removed and analyzed by the coulometric reduction procedure. 4.4 Other corrosion film evaluation techniques for metallic coupons are also available. The most common of these is mass gain, which is nondestructive to the surface films, but is limited to the determination of the total amount of additional mass acquired by the metal as a result of the environmental attack. The most common is weighing using high performance microbalances or for purposes of real-time monitoring, quartz crystal microbalances (see Specification B808). Note 3: Detailed instructions for conducting such weighings, as well as coupon cleaning and surface preparation procedures, are included as part of Test Met... SCOPE 1.1 This test method covers procedures and equipment for determining the relative buildup of corrosion and tarnish films (including oxides) on metal surfaces by the constant-current coulometric technique, also known as the cathodic reduction method. 1.2 This test method is designed primarily to determine the relative quantities of tarnish films on control coupons that result from gaseous environmental tests, particularly when the latter are used for testing components or systems containing electrical contacts used in customer product environments. 1.3 This test method may also be used to evaluate test samples that have been exposed to indoor industrial locations or other specific application environments. (See 4.6 for limitations.) 1.4 This test method has been demonstrated to be applicable particularly to copper and silver test samples (see (1)).2 Other metals require further study to prove their applicability within the scope of this test method. 1.5 The values stated in SI units are the preferred units. The values provided in parentheses are for information only. 1.6 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to become familiar with all hazards including those identified in the appropriate Material Safety Data Sheet (MSDS) for this product/material as provided by the manufacturer, to establish appropriate safety, health, and environmental practices, and determine the applicability of regulatory limitations prior to use. 1.7 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM B825-19 is classified under the following ICS (International Classification for Standards) categories: 25.220.01 - Surface treatment and coating in general. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM B825-19 has the following relationships with other standards: It is inter standard links to ASTM B809-95(2018), ASTM B827-05(2014), ASTM B809-95(2013), ASTM B810-01a(2011), ASTM B808-10, ASTM B827-05(2009)e1, ASTM B827-05(2009)e2, ASTM B809-95(2008), ASTM B810-01a(2006), ASTM D1193-06, ASTM B827-05, ASTM B808-05, ASTM B808-97(2003), ASTM B827-97(2003), ASTM B809-95(2003). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM B825-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: B825 − 19
Standard Test Method for
Coulometric Reduction of Surface Films on Metallic Test
Samples
This standard is issued under the fixed designation B825; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope Development of International Standards, Guides and Recom-
mendations issued by the World Trade Organization Technical
1.1 This test method covers procedures and equipment for
Barriers to Trade (TBT) Committee.
determining the relative buildup of corrosion and tarnish films
(including oxides) on metal surfaces by the constant-current
2. Referenced Documents
coulometric technique, also known as the cathodic reduction
2.1 ASTM Standards:
method.
B808 Test Method for Monitoring of Atmospheric Corrosion
1.2 This test method is designed primarily to determine the
Chambers by Quartz Crystal Microbalances
relative quantities of tarnish films on control coupons that
B809 Test Method for Porosity in Metallic Coatings by
result from gaseous environmental tests, particularly when the
Humid Sulfur Vapor (“Flowers-of-Sulfur”)
latter are used for testing components or systems containing
B810 Test Method for Calibration of Atmospheric Corrosion
electrical contacts used in customer product environments.
Test Chambers by Change in Mass of Copper Coupons
1.3 This test method may also be used to evaluate test
B827 Practice for Conducting Mixed Flowing Gas (MFG)
samples that have been exposed to indoor industrial locations
Environmental Tests
or other specific application environments. (See 4.6 for limi-
D1193 Specification for Reagent Water
tations.)
3. Summary of Test Method
1.4 This test method has been demonstrated to be applicable
particularly to copper and silver test samples (see (1)). Other 3.1 In constant-current coulometry, a fixed reduction-
metals require further study to prove their applicability within current density is applied to the sample in an electrolytically
the scope of this test method. conductive solution, and the resulting variations in potential—
measured against a standard reference electrode in the same
1.5 The values stated in SI units are the preferred units. The
solution—are followed as a function of time. Typically, with
values provided in parentheses are for information only.
well-behaved surface films, the voltage-time plot should show
1.6 This standard does not purport to address all of the
a number of horizontal portions, or steps, each corresponding
safety concerns, if any, associated with its use. It is the
to a specific reduction potential or voltage (Fig. 1). The final
responsibility of the user of this standard to become familiar
potential step, which is always present with all substances,
with all hazards including those identified in the appropriate
corresponds to the reduction of hydrogen ions in the solution
Material Safety Data Sheet (MSDS) for this product/material
(to form hydrogen gas), and represents a limit beyond which no
as provided by the manufacturer, to establish appropriate
higher potential reduction process can occur.
safety, health, and environmental practices, and determine the
NOTE 1—As shown in Figs. 1 and 2, a differential circuit is recom-
applicability of regulatory limitations prior to use.
mended to help in resolving the individual voltage steps by pinpointing the
1.7 This international standard was developed in accor-
corresponding inflection points on the main reduction curve (see 6.2.3).
dance with internationally recognized principles on standard-
3.2 From the elapsed times at the various steps, conclusions
ization established in the Decision on Principles for the
can often be drawn regarding the corrosion processes that have
taken place to produce the surface films. Also, calculations can
This test method is under the jurisdiction of ASTM Committee B02 on
be made from the time at each voltage step in order to calculate
Nonferrous Metals and Alloys and is the direct responsibility of Subcommittee
the number of coulombs of electrical charge required to
B02.05 on Precious Metals and Electrical Contact Materials and Test Methods.
Current edition approved Nov. 1, 2019. Published November 2019. Originally
approved in 1997. Last previous edition approved in 2013 as B825 – 13 which was
withdrawn November 2018 and reinstated in November 2019. DOI: 10.1520/ For referenced ASTM standards, visit the ASTM website, www.astm.org, or
B0825-19. contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
The boldface numbers in parentheses refer to the list of references at the end of Standards volume information, refer to the standard’s Document Summary page on
this standard. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
B825 − 19
4.2 Many of these environmental test methods require
monitoring of the conditions within the chamber during the test
in order to confirm that the intended environmentally related
reactions are actually taking place. The most common type of
monitor consists of copper, silver, or other thin metallic
coupons of a few square centimeters that are placed within the
test chamber and that react with the corrosive environment in
much the same way as the significant surfaces of the parts
under test.
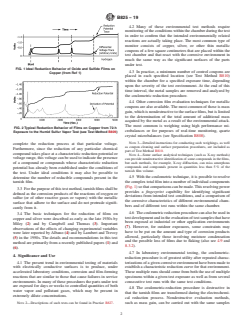
FIG. 1 Ideal Reduction Behavior of Oxide and Sulfide Films on
4.3 In practice, a minimum number of control coupons are
Copper (from Ref 1)
placed in each specified location (see Test Method B810)
within the chamber for a specified exposure time, depending
upon the severity of the test environment. At the end of this
time interval, the metal samples are removed and analyzed by
the coulometric reduction procedure.
4.4 Other corrosion film evaluation techniques for metallic
coupons are also available. The most common of these is mass
gain, which is nondestructive to the surface films, but is limited
to the determination of the total amount of additional mass
acquired by the metal as a result of the environmental attack.
The most common is weighing using high performance mi-
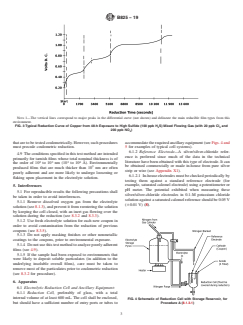
FIG. 2 Typical Reduction Behavior of Films on Copper from 72-h
crobalances or for purposes of real-time monitoring, quartz
Exposure to the Humid Sulfur Vapor Test (see Test Method B809)
crystal microbalances (see Specification B808).
NOTE 3—Detailed instructions for conducting such weighings, as well
complete the reduction process at that particular voltage.
as coupon cleaning and surface preparation procedures, are included as
Furthermore, since the reduction of any particular chemical
part of Test Method B810.
compound takes place at a characteristic reduction potential or
NOTE 4—Some surface analytical techniques (such as X-ray methods)
voltage range, this voltage can be used to indicate the presence
can provide nondestructive identification of some compounds in the films,
but such methods, for example, X-ray diffraction, can miss amorphous
of a compound or compounds whose characteristic reduction
compounds and compounds present in quantities less than 5 % of the
potential has already been established under the conditions of
tarnish film volume.
the test. Under ideal conditions it may also be possible to
determine the number of reducible compounds present in the 4.5 With the coulometric technique, it is possible to resolve
tarnish film. the complex total film into a number of individual components
(Fig. 1) so that comparisons can be made. This resolving power
3.3 For the purpose of this test method, tarnish films shall be
provides a fingerprint capability for identifying significant
defined as the corrosion products of the reactions of oxygen or
deviations from intended test conditions, and a comparison of
sulfur (or of other reactive gases or vapors) with the metallic
the corrosive characteristics of different environmental cham-
surface that adhere to the surface and do not protrude signifi-
bers and of different test runs within the same chamber.
cantly from it.
4.6 The coulometric reduction procedure can also be used in
3.4 The basic techniques for the reduction of films on
test development and in the evaluation of test samples that have
copper and silver were described as early as the late 1930s by
been exposed at industrial or other application environments
Miley (2) and by Campbell and Thomas (3). Important
(7). However, for outdoor exposures, some constraints may
observations of the effects of changing experimental variables
have to be put on the amount and type of corrosion products
were later reported by Albano (4) and by Lambert and Trevoy
allowed, particularly those involving moisture condensation
(5) in the 1950s. The details and recommendations in this test
and the possible loss of films due to flaking (also see 4.9 and
method are primarily from a recently published papers (1) and
8.3.2).
(6).
4.7 In laboratory environmental testing, the coulometric-
4. Significance and Use
reduction procedure is of greatest utility after repeated charac-
4.1 The present trend in environmental testing of materials terizations of a given corrosive environment have been made to
with electrically conductive surfaces is to produce, under establish a characteristic reduction curve for that environment.
accelerated laboratory conditions, corrosion and film-forming These multiple runs should come from both the use of multiple
reactions that are similar to those that cause failures in service specimens within a given test exposure as well as from several
environments. In many of these procedures the parts under test consecutive test runs with the same test conditions.
are exposed for days or weeks to controlled quantities of both
4.8 The coulometric-reduction procedure is destructive in
water vapor and pollutant gases, which may be present in
that the tarnish films are transformed during the electrochemi-
extremely dilute concentrations.
cal reduction process. Nondestructive evaluation methods,
NOTE 2—Descriptions of such tests can be found in Practice B827. such as mass gain, can be carried out with the same samples
B825 − 19
NOTE 1—The vertical lines correspond to major peaks in the differential curve (not shown) and delineate the main reducible film types from this
environment.
FIG. 3 Typical Reduction Curve of Copper from 48-h Exposure to High Sulfide (100 ppb H S) Mixed Flowing Gas (with 20 ppb Cl and
2 2
200 ppb NO )
that are to be tested coulometrically. However, such procedures accommodate the required ancillary equipment (see Figs. 4 and
must precede coulometric reduction. 5 for examples of typical cell systems).
6.1.2 Reference Electrode—A silver/silver-chloride refer-
4.9 The conditions specified in this test method are intended
ence is preferred since much of the data in the technical
primarily for tarnish films whose total nominal thickness is of
2 3 3 4
literature have been obtained with this type of electrode. It can
the order of 10 to 10 nm (10 to 10 Å). Environmentally
3 be obtained commercially or made in-house from pure silver
produced films that are much thicker than 10 nm are often
strip or wire (see Appendix X1).
poorly adherent and are more likely to undergo loosening or
6.1.2.1 In-house electrodes must be checked periodically by
flaking upon placement in the electrolyte solution.
testing them against a standard reference electrode (for
example, saturated calomel electrode) using a potentiometer or
5. Interferences
pH meter. The potential exhibited when measuring these
5.1 For reproducible results the following precautions shall
silver/silver-chloride electrodes in 0.1-M potassium chloride
be taken in order to avoid interferences.
solution against a saturated calomel reference should be 0.05 V
5.1.1 Remove dissolved oxygen gas from the electrolyte
(60.01 V) (8).
solution (see 8.1.3), and prevent it from reentering the solution
by keeping the cell closed, with an inert gas flowing over the
solution during the reduction (see 8.3.2 and 8.3.3).
5.1.2 Use fresh electrolyte solution for each new coupon in
order to avoid contamination from the reduction of previous
coupons (see 8.3.5).
5.1.3 Do not apply masking finishes or other nonmetallic
coatings to the coupons, prior to environmental exposure.
5.1.4 Do not use this test method to analyze poorly adherent
films (see 4.9).
5.1.5 If the sample had been exposed to environments that
were likely to deposit soluble particulates (in addition to the
underlying insoluble overall films), care must be taken to
remove most of the particulates prior to coulometric reduction
(see 8.3.2 for procedure).
6. Apparatus
6.1 Electrolytic Reduction Cell and Ancillary Equipment:
6.1.1 Reduction Cell, preferably of glass, with a total
internal volume of at least 600 mL. The cell shall be enclosed,
FIG. 4 Schematic of Reduction Cell with Storage Reservoir, for
but should have a sufficient number of entry ports or tubes to Procedure A (8.1.3.1)
B825 − 19
scale), though not essential, is helpful in data evaluation, and is
obtained easily with any 250-mm chart recorder. A digital
recording system, capable of data storage and graphic repre-
sentation can be used instead of, or in conjunction with, the
strip chart recorder system. Both systems shall have input
impedance of at least 10 Ω, preferably higher.
6.2.3 Differential Circuit, or Commercial Differential Volt-
age Output Apparatus—If a digital recording system is used in
conjunction with, or to replace, an analog recording system, the
following method can be used to create a differential curve.
After the reduction is recorded completely, each data point,
except for the first and last, must be analyzed. For a given
point, X, determine the slope to the previous point, X , and the
p
subsequent point, X . Knowing the time interval, T, between
s
each reading, the required slopes are as follows:
S 5 X 2 X /T S 5 X 2 X /T (1)
~ ! ~ !
p p s s
An approximation of the slope at X is then found by taking
the average of the slopes S and S as follows:
p s
S 5 ~S 1S !/2 (2)
p s
Each value of S is recorded with the concurrent value of X
for later analysis. Slopes at the first and last data points can be
assumed to be zero. A method for enhancing these digitally
FIG. 5 Schematic of Reduction Cell for Procedure B (8.1.3.2)
produced differential curves can be found in Appendix X2.
6.1.3 Inert-Gas Purging Tube—The end that is in the
7. Reagents
electrolyte should be fitted with fritted glass or drawn to a fine
7.1 The only reagents required for routine procedures are
tip (for example, 0.5-mm inner diameter or less).
ACS reagent-grade potassium chloride (for the electrolyte),
6.1.4 Counter-Electrodes—Pure platinum foil or wire shall
Pre-Purified-grade nitrogen or other inert gas, and a source of
be used. The number of counter-electrodes may vary from 2 to
distilled or deionized water (Type IV or better as specified in
4 and shall be positioned symmetrically around the sample.
Specification D1193).
The area of the counter-electrodes preferably should be equal
to or greater than the sample area.
8. Procedure
6.1.5 Wire Hook or Clip for Holding the Sample—The upper
8.1 Cell Preparation:
part of the hook or clip shall be attached to a wire (inserted into
8.1.1 Assemble the reduction cell in accordance with either
a glass or plastic tube) for ultimate connection to the negative
Fig. 4 or Fig. 5, making sure that all components are chemi-
output of the power supply. If the wire hook is to be immersed
cally clean. For each sample size or geometry, determine in
in the solution, it shall be made of the same metal as the
advance the level of liquid that is required to cover the
sample. If a clip is used, it shall be heavily gold plated (3 μm
specified sample surface. Mark this level on the outside of the
or more in thickness) and attached to a platinum wire hook for
cell. A minimum volume of 300-mL solution is recommended
electrical contact.
for each analysis.
6.2 Electronic Equipment—For producing the constant ca-
8.1.2 Attach the tubing system for the inert gas (assembled
thodic current and measuring the resulting voltages as a
in advance) to the regulator of the gas tank.
function of time comprises three basic functional modules
8.1.3 Potassium Chloride Solution 0.1 M—Deaerate 0.1-M
whose recommended characteristics (for routine tarnish-film
KCl solution, prepared in advance, with the inert gas (to
analysis) are listed as follows:
displace dissolved oxygen) using either Method A (8.1.3.1) or
6.2.1 Constant Current Power Supply, such as, a
Method B (8.1.3.2).
potentiostat/galvanostat, capable of supplying a constant direct
NOTE 5—If any oxygen gas (O ) is present in the working electrolyte,
current, and adjustable from 0.02 to 2 mA with a precision of
it will tend to interfere with the coulometric determinations, since O is
61 %. However, for certain limited applications (for example, 2
easily reduced in the same voltage range as many oxide or tarnish film
very large area samples), currents greater than 20 mA might
components. Dissolved oxygen is eliminated by deaerating the electrolyte
conceivably be required, see 8.2.1.
solution prior to use and by running the reduction in a closed cell under
6.2.2 Strip Chart or Digital Recorder, or Both—For a
an inert atmosphere.
strip-chart recorder, two pens are preferred, one pen for voltage
and the other for a voltage-time derivative curve. The chart
recorder shall have variable speed capability, from 10 mm/h to
Pre-Purified is a designation of Matheson Gas Co., East Rutherford, NJ, for a
100 mm/min, and full-scale voltage ranges from 0.5 to 2 V. A
specific
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...