ASTM G36-94(2018)
(Practice)Standard Practice for Evaluating Stress-Corrosion-Cracking Resistance of Metals and Alloys in a Boiling Magnesium Chloride Solution
Standard Practice for Evaluating Stress-Corrosion-Cracking Resistance of Metals and Alloys in a Boiling Magnesium Chloride Solution
SIGNIFICANCE AND USE
5.1 For most applications, this environment provides an accelerated method of ranking the relative degree of stress-corrosion cracking susceptibility for stainless steels and related alloys in aqueous chloride-containing environments. Materials that normally provide acceptable resistance in hot chloride service may crack in this test. The test may not be relevant to stress-corrosion cracking in polythionic acid or caustic environments.
5.2 Resistance to stress-corrosion cracking in boiling magnesium chloride (155.0°C (311.0°F)) should, where possible, be correlated to resistance in service for the materials of interest. However, such correlations may not always be possible.
5.3 Boiling magnesium chloride may also cause pitting of many stainless alloys. This leads to the possibility of confusing stress-corrosion failures with mechanical failures induced by corrosion-reduced net cross sections. This danger is particularly great when small cross section samples, high applied stress levels, long exposure periods, stress-corrosion resistant alloys, or a combination thereof are being used. Careful examination is recommended for correct diagnosis of the cause of failure.
SCOPE
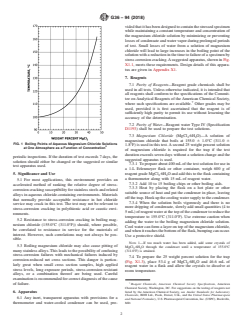
1.1 This practice describes a procedure for conducting stress-corrosion cracking tests in a boiling magnesium chloride solution. Although this test may be performed using various concentrations of magnesium chloride, this procedure covers a test solution held at a constant boiling temperature of 155.0 ± 1.0°C (311.0 ± 1.8°F). The boiling points of aqueous magnesium chloride solutions at one atmosphere pressure as a function of concentration are shown graphically in Fig. 1.2 A suggested test apparatus capable of maintaining solution concentration and temperature within the prescribed limits for extended periods of time is also described herein.3
FIG. 1 Boiling Points of Aqueous Magnesium Chloride Solutions at One Atmosphere as a Function of Concentration2
1.2 The boiling magnesium chloride test is applicable to wrought, cast, and welded stainless steels and related alloys. It is a method for detecting the effects of composition, heat treatment, surface finish, microstructure, and stress on the susceptibility of these materials to chloride stress corrosion cracking.4
1.3 This practice is concerned primarily with the test solution, which may be used with a variety of stress corrosion test specimens, surface finishes, and methods of applying stress.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 7 for specific safety precautions.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Sep-2018
- Technical Committee
- G01 - Corrosion of Metals
- Drafting Committee
- G01.06 - Environmentally Assisted Cracking
Relations
- Effective Date
- 01-Oct-2018
- Refers
ASTM G30-97(2015) - Standard Practice for Making and Using U-Bend Stress-Corrosion Test Specimens - Effective Date
- 01-Nov-2015
- Effective Date
- 01-Dec-2011
- Refers
ASTM G30-97(2009) - Standard Practice for Making and Using U-Bend Stress-Corrosion Test Specimens - Effective Date
- 01-May-2009
- Effective Date
- 01-Apr-2007
- Effective Date
- 01-Jul-2006
- Effective Date
- 01-Mar-2006
- Effective Date
- 15-Jun-2005
- Effective Date
- 01-Jan-2004
- Refers
ASTM G1-03 - Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens - Effective Date
- 01-Oct-2003
- Effective Date
- 10-Sep-2003
- Effective Date
- 10-Jun-2003
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM G36-94(2018): Standard Practice for Evaluating Stress-Corrosion-Cracking Resistance of Metals and Alloys in a Boiling Magnesium Chloride Solution provides a recognized, accelerated laboratory method for determining the susceptibility of stainless steels and related alloys to stress-corrosion cracking (SCC) in chloride-rich environments. By simulating aggressive conditions using a boiling magnesium chloride solution at controlled temperatures (155.0 ± 1.0°C / 311.0 ± 1.8°F), this practice offers a basis to compare the resistance of various metal compositions, heat treatments, and conditions to stress corrosion.
This standard is applicable to wrought, cast, and welded stainless steels and related alloys. It facilitates comparative analysis for industry professionals concerned with the integrity and durability of metallic materials exposed to chloride-containing aqueous environments.
Key Topics
- Test Environment: Utilizes a boiling magnesium chloride solution, maintained at a strict temperature range, to accelerate SCC and highlight potential vulnerabilities in materials.
- Test Applicability: Designed for stainless steel and related alloys in wrought, cast, or welded forms. Detects the effects of factors like composition, heat treatment, surface finish, microstructure, and applied stress.
- Specimen Handling and Stress Application: Compatible with various corrosion test specimen types and methods of stressing; details are outlined for maintaining solution integrity and correct specimen placement.
- Failure Mechanisms: Distinguishes between stress-corrosion cracking, pitting, and mechanical failure due to corrosion-reduced cross-sections, which is critical for accurate failure diagnosis.
- Limitations: Not intended to predict performance in polythionic acid or caustic environments, and results may not always correlate perfectly with field service conditions.
Applications
- Materials Selection: Provides manufacturers and engineers with a reliable tool to rank metals and alloys for use in environments subject to chloride-induced stress corrosion, supporting informed selection for process equipment, piping, and structural applications.
- Quality Assurance and R&D: Useful for new alloy development, optimization of heat treatment processes, and verification of production lots for SCC resistance.
- Failure Analysis: Serves as a comparative benchmark when investigating field failures suspected to result from chloride-induced SCC.
- Corrosion Testing Laboratories: Offers a standardized protocol for SCC testing enabling repeatable, comparable results across organizations and over time.
- Safety and Regulatory Compliance: Helps meet requirements for testing stainless steel materials to ensure suitability for aggressive service environments, supporting both product safety and regulatory compliance.
Related Standards
Practitioners implementing ASTM G36-94(2018) should also be familiar with standards that complement this practice and support comprehensive corrosion assessment:
- ASTM G1 – Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens
- ASTM D1193 – Specification for Reagent Water
- ASTM G30 – Practice for Making and Using U-Bend Stress-Corrosion Test Specimens
- ASTM G15 – Terminology Relating to Corrosion and Corrosion Testing (withdrawn, but historically referenced)
Practical Value
Using ASTM G36-94(2018), laboratories and engineering teams can:
- Identify metals and alloys with superior resistance to chloride stress-corrosion cracking
- Reduce the risk of in-service failure through appropriate material selection
- Streamline screening of materials prior to deployment in harsh, chloride-laden environments
- Support the safe operation of components in critical sectors such as chemical processing, oil and gas, power generation, desalination, and marine industries
Keywords: stress-corrosion cracking, boiling magnesium chloride, stainless steels, chloride environments, corrosion testing, SCC resistance, ASTM G36, accelerated test, material selection, laboratory standard.
Buy Documents
ASTM G36-94(2018) - Standard Practice for Evaluating Stress-Corrosion-Cracking Resistance of Metals and Alloys in a Boiling Magnesium Chloride Solution
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM G36-94(2018) is a standard published by ASTM International. Its full title is "Standard Practice for Evaluating Stress-Corrosion-Cracking Resistance of Metals and Alloys in a Boiling Magnesium Chloride Solution". This standard covers: SIGNIFICANCE AND USE 5.1 For most applications, this environment provides an accelerated method of ranking the relative degree of stress-corrosion cracking susceptibility for stainless steels and related alloys in aqueous chloride-containing environments. Materials that normally provide acceptable resistance in hot chloride service may crack in this test. The test may not be relevant to stress-corrosion cracking in polythionic acid or caustic environments. 5.2 Resistance to stress-corrosion cracking in boiling magnesium chloride (155.0°C (311.0°F)) should, where possible, be correlated to resistance in service for the materials of interest. However, such correlations may not always be possible. 5.3 Boiling magnesium chloride may also cause pitting of many stainless alloys. This leads to the possibility of confusing stress-corrosion failures with mechanical failures induced by corrosion-reduced net cross sections. This danger is particularly great when small cross section samples, high applied stress levels, long exposure periods, stress-corrosion resistant alloys, or a combination thereof are being used. Careful examination is recommended for correct diagnosis of the cause of failure. SCOPE 1.1 This practice describes a procedure for conducting stress-corrosion cracking tests in a boiling magnesium chloride solution. Although this test may be performed using various concentrations of magnesium chloride, this procedure covers a test solution held at a constant boiling temperature of 155.0 ± 1.0°C (311.0 ± 1.8°F). The boiling points of aqueous magnesium chloride solutions at one atmosphere pressure as a function of concentration are shown graphically in Fig. 1.2 A suggested test apparatus capable of maintaining solution concentration and temperature within the prescribed limits for extended periods of time is also described herein.3 FIG. 1 Boiling Points of Aqueous Magnesium Chloride Solutions at One Atmosphere as a Function of Concentration2 1.2 The boiling magnesium chloride test is applicable to wrought, cast, and welded stainless steels and related alloys. It is a method for detecting the effects of composition, heat treatment, surface finish, microstructure, and stress on the susceptibility of these materials to chloride stress corrosion cracking.4 1.3 This practice is concerned primarily with the test solution, which may be used with a variety of stress corrosion test specimens, surface finishes, and methods of applying stress. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 7 for specific safety precautions. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 5.1 For most applications, this environment provides an accelerated method of ranking the relative degree of stress-corrosion cracking susceptibility for stainless steels and related alloys in aqueous chloride-containing environments. Materials that normally provide acceptable resistance in hot chloride service may crack in this test. The test may not be relevant to stress-corrosion cracking in polythionic acid or caustic environments. 5.2 Resistance to stress-corrosion cracking in boiling magnesium chloride (155.0°C (311.0°F)) should, where possible, be correlated to resistance in service for the materials of interest. However, such correlations may not always be possible. 5.3 Boiling magnesium chloride may also cause pitting of many stainless alloys. This leads to the possibility of confusing stress-corrosion failures with mechanical failures induced by corrosion-reduced net cross sections. This danger is particularly great when small cross section samples, high applied stress levels, long exposure periods, stress-corrosion resistant alloys, or a combination thereof are being used. Careful examination is recommended for correct diagnosis of the cause of failure. SCOPE 1.1 This practice describes a procedure for conducting stress-corrosion cracking tests in a boiling magnesium chloride solution. Although this test may be performed using various concentrations of magnesium chloride, this procedure covers a test solution held at a constant boiling temperature of 155.0 ± 1.0°C (311.0 ± 1.8°F). The boiling points of aqueous magnesium chloride solutions at one atmosphere pressure as a function of concentration are shown graphically in Fig. 1.2 A suggested test apparatus capable of maintaining solution concentration and temperature within the prescribed limits for extended periods of time is also described herein.3 FIG. 1 Boiling Points of Aqueous Magnesium Chloride Solutions at One Atmosphere as a Function of Concentration2 1.2 The boiling magnesium chloride test is applicable to wrought, cast, and welded stainless steels and related alloys. It is a method for detecting the effects of composition, heat treatment, surface finish, microstructure, and stress on the susceptibility of these materials to chloride stress corrosion cracking.4 1.3 This practice is concerned primarily with the test solution, which may be used with a variety of stress corrosion test specimens, surface finishes, and methods of applying stress. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. See Section 7 for specific safety precautions. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM G36-94(2018) is classified under the following ICS (International Classification for Standards) categories: 77.060 - Corrosion of metals. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM G36-94(2018) has the following relationships with other standards: It is inter standard links to ASTM G36-94(2013), ASTM G30-97(2015), ASTM G1-03(2011), ASTM G30-97(2009), ASTM G15-07, ASTM G15-06, ASTM D1193-06, ASTM G15-05, ASTM G15-04, ASTM G1-03, ASTM G15-03a, ASTM G15-03, ASTM G15-02, ASTM D1193-99, ASTM D1193-99e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM G36-94(2018) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: G36 − 94 (Reapproved 2018)
Standard Practice for
Evaluating Stress-Corrosion-Cracking Resistance of Metals
and Alloys in a Boiling Magnesium Chloride Solution
ThisstandardisissuedunderthefixeddesignationG36;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoptionor,inthecaseofrevision,theyearoflastrevision.Anumberinparenthesesindicatestheyearoflastreapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope priate safety, health, and environmental practices and deter-
mine the applicability of regulatory limitations prior to use.
1.1 This practice describes a procedure for conducting
See Section 7 for specific safety precautions.
stress-corrosioncrackingtestsinaboilingmagnesiumchloride
1.5 This international standard was developed in accor-
solution. Although this test may be performed using various
dance with internationally recognized principles on standard-
concentrations of magnesium chloride, this procedure covers a
ization established in the Decision on Principles for the
test solution held at a constant boiling temperature of 155.0 6
Development of International Standards, Guides and Recom-
1.0°C (311.0 6 1.8°F). The boiling points of aqueous magne-
mendations issued by the World Trade Organization Technical
sium chloride solutions at one atmosphere pressure as a
2 Barriers to Trade (TBT) Committee.
function of concentration are shown graphically in Fig. 1. A
suggested test apparatus capable of maintaining solution con-
2. Referenced Documents
centration and temperature within the prescribed limits for
2.1 ASTM Standards:
extended periods of time is also described herein.
D1193Specification for Reagent Water
1.2 The boiling magnesium chloride test is applicable to
G1Practice for Preparing, Cleaning, and Evaluating Corro-
wrought, cast, and welded stainless steels and related alloys. It
sion Test Specimens
is a method for detecting the effects of composition, heat
G15Terminology Relating to Corrosion and CorrosionTest-
treatment, surface finish, microstructure, and stress on the
ing (Withdrawn 2010)
susceptibility of these materials to chloride stress corrosion
G30 Practice for Making and Using U-Bend Stress-
cracking.
Corrosion Test Specimens
1.3 This practice is concerned primarily with the test
solution, which may be used with a variety of stress corrosion
3. Terminology
test specimens, surface finishes, and methods of applying
3.1 Definitions—For definitions of terms used in this prac-
stress.
tice see Terminology G15.
1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
4. Summary of Practice
responsibility of the user of this standard to establish appro-
4.1 A predetermined quantity of reagent grade magnesium
chloride and some distilled water are added to a container.The
container and contents, with thermometer and condenser
This practice is under the jurisdiction ofASTM Committee G01 on Corrosion
of Metals and is the direct responsibility of Subcommittee G01.06 on Environmen-
affixed, are placed on a source of heat. When the magnesium
tally Assisted Cracking.
chloride solution boils, it is adjusted to maintain the desired
Current edition approved Oct. 1, 2018. Published November 2018. Originally
concentration and boiling point through the addition of small
approvedin1973.Lastpreviouseditionapprovedin2013asG36–94(2013).DOI:
10.1520/G0036-94R18. quantities of either water or salt.
Available data on the relationship of concentrations and boiling points of
4.2 After the solution has stabilized at the desired boiling
magnesium chloride solutions are critically reviewed and supplemented by I. B.
Casale in “Boiling Points of Magnesium Chloride Solutions—TheirApplication in pointforthetest,thestressedspecimensareadded.Depending
Stress Corrosion Studies,” Corrosion, Vol 23, 1967, pp. 314–17.
upon the intent of the test, the specimens should be given
The apparatus and test procedures for maintaining constant boiling tempera-
tures of magnesium chloride solutions for stress corrosion tests are described by M.
A. Streicher and A. J. Sweet in Corrosion, Vol 25, 1969, pp. 1–6.
4 5
The use of concentrated magnesium chloride solutions for determining the For referenced ASTM standards, visit the ASTM website, www.astm.org, or
susceptibility to stress corrosion cracking of austenitic and ferritic stainless steels contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
and related nickel-base alloys was first described by M. A. Scheil, Symposium on Standards volume information, refer to the standard’s Document Summary page on
Stress Corrosion Cracking of Metals, ASTM STP 64, ASTM, 1945, p. 395. the ASTM website.
(Although currently out of print, copies may be obtained from University Micro- The last approved version of this historical standard is referenced on
films, Inc., 300 North Zeeb Rd., Ann Arbor, MI 48106.) www.astm.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G36 − 94 (2018)
videdthatithasbeendesignedtocontainthestressedspecimen
while maintaining a constant temperature and concentration of
the magnesium chloride solution by minimizing or preventing
lossesofcondensateandwatervaporduringprolongedperiods
of test. Small losses of water from a solution of magnesium
chloride will lead to large increases in the boiling point of the
solutionwithareductioninthetimetofailureofaspecimenby
stresscorrosioncracking.Asuggestedapparatus,showninFig.
X1.1, meets these requirements. Design details of this appara-
tus are given in Appendix X1.
7. Reagents
7.1 Purity of Reagents—Reagent grade chemicals shall be
used in all tests. Unless otherwise indicated, it is intended that
all reagents shall conform to the specifications of the Commit-
tee onAnalytical Reagents of theAmerican Chemical Society,
where such specifications are available. Other grades may be
used, provided it is first ascertained that the reagent is of
sufficiently high purity to permit its use without lessening the
accuracy of the determination.
7.2 Purity of Water—Reagent water Type IV (Specification
D1193) shall be used to prepare the test solutions.
7.3 Magnesium Chloride (MgCl ·6H O)—A solution of
2 2
magnesium chloride that boils at 155.0 6 1.0°C (311.0 6
FIG. 1 Boiling Points of Aqueous Magnesium Chloride Solutions
1.8°F) is used in this test.Asecond 25 weight percent solution
at One Atmosphere as a Function of Concentration
of magnesium chloride is required for the trap if the test
duration exceeds seven days without a solution change and the
periodic inspections. If the duration of test exceeds 7 days, the
suggested apparatus is used.
solution should either be changed or the suggested or similar
7.3.1 Toprepareabout400mLofthetestsolutionforusein
test apparatus used.
a 1-L Erlenmeyer flask or other container, weigh 600 g of
5. Significance and Use reagentgradeMgCl ·6H Oandaddthistotheflaskcontaining
2 2
a thermometer along with 15 mL of reagent water.
5.1 For most applications, this environment provides an
7.3.2 Add 10 to 15 boiling chips or other boiling aids.
accelerated method of ranking the relative degree of stress-
7.3.3 Heat by placing the flask on a hot plate or other
corrosioncrackingsusceptibilityforstainlesssteelsandrelated
suitable source of heat and put the condenser in place, leaving
alloys in aqueous chloride-containing environments. Materials
offthetrap.Hookupthecoolingwatersupplytothecondenser.
that normally provide acceptable resistance in hot chloride
7.3.4 When the solution boils vigorously and there is no
service may crack in this test. The test may not be relevant to
more dripping of condensate, slowly add small quantities (4 to
stress-corrosion cracking in polythionic acid or caustic envi-
5mL)ofreagentwateratthetopofthecondensertoreducethe
ronments.
temperature to 155.0°C (311.0°F). Use extreme caution when
5.2 Resistance to stress-corrosion cracking in boiling mag-
adding the water to the boiling magnesium chloride solution.
nesium chloride (155.0°C (311.0°F)) should, where possible,
Coolwatercanformalayerontopofthemagnesiumchloride,
be correlated to resistance in service for the materials of
andwhenitreachesthebottomoftheflask,bumpingcanoccur.
interest. However, such correlations may not always be pos-
Use a protective shield.
sible.
NOTE 1—If too much water has been added, add some crystals of
5.3 Boiling magnesium chloride may also cause pitting of
MgCl ·6H O through the condenser until a temperature of 155.0°C
2 2
manystainlessalloys.Thisleadstothepossibilityofconfusing
(311.0°F) is attained.
stress-corrosion failures with mechanical failures induced by
7.4 To prepare the 25 weight percent solution for the trap
corrosion-reduced net cross sections. This danger is particu-
(Fig. X1.3), place 53.4 g of MgCl ·6H O and 46.6 mL of
2 2
larly great when small cross section samples, high applied
reagent water in a flask and allow the crystals to dissolve at
stress levels, long exposure periods, stress-corrosion resistant
room temperature.
alloys, or a combination thereof are being used. Careful
examinationisrecommendedforcorrectdiagnosisofthecause
of failure. Reagent Chemicals, American Chemical Society Specifications, American
Chemical Society, Washington, DC. For suggestions on the testing of reagents not
6. Apparatus listed by the American Chemical Society, see Analar Standards for Laboratory
Chemicals, BDH Ltd., Poole, Dorset, U.K., and the United States Pharmacopeia
6.1 Any inert, transparent apparatus with provisions for a
and National Formulary, U.S. Pharmacopeial Convention, Inc. (USPC), Rockville,
thermometer and water-cooled condenser can be used, pro- MD.
G36 − 94 (2018)
8. Safety Precautions 9.4 The test specimens must be kept from direct contact
withheatedsurfacesbyglasssupports.Metalspecimenholders
8.1 When cold, magnesium chloride can be handled with
used for stressing specimens should also be supported on glass
the minimum protective equipment of rubber gloves and
rods or tubes. The design for two types of test specimens that
goggles. Maximum protective measures should be taken to
can be used with the suggested apparatus can be found in
prevent boiling magnesium chloride from coming into contact
footnote 3.
with the skin. Severe burns can result as the hot magnesium
chloride adheres to the skin forming a crust which causes deep
10. Procedure
burns. The severity of the burns can be reduced by taking
10.1 Collecttheapparatusandtestspecimensinpreparation
proper and immediate first aid measures and by contacting a
for the test. If the suggested test apparatus is used, assemble as
physician.
outlined in Appendix X1.
8.1.1 Intheadventofaspilloraccident, the hot magnesium
10.2 Preparethetestsolutionbyaddingaknownquantityof
chloride should be quickly flushed from the skin with large
reagent grade MgCl ·6H O, reagent water,
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...