ASTM F2791-24
(Guide)Standard Guide for Assessment of Surface Texture of Non-Porous Biomaterials in Two Dimensions
Standard Guide for Assessment of Surface Texture of Non-Porous Biomaterials in Two Dimensions
SIGNIFICANCE AND USE
4.1 The term “surface texture” is used to describe the local deviations of a surface from an ideal shape. Surface texture usually consists of long wavelength repetitive features that occur as results of chatter, vibration, or heat treatments during the manufacture of implants. Short wavelength features superimposed on the long wavelength features of the surface, which may arise from polishing or etching of the implant, are referred to as roughness.
4.2 This guide provides an overview of techniques that are available for measuring the surface in terms of Cartesian coordinates and the parameters used to describe surface texture. It is important to appreciate that it is not possible to measure surface texture per se, but to derive values for parameters that can be used to describe it. ISO has published a series of standards on surface texture measurements that may be consulted for more information (ISO 3274, ISO 4287, ISO 4288, ISO 5436-2, ISO 10993-19, ISO 12179, ISO 13565-1, ISO 19606, ISO 21920-1, ISO 21920-2, ISO 21920-3, ISO 25178-1, ISO 25178-2, ISO 25178-3, ISO 25178-6, ISO 25178-70, ISO 25178-71, ISO 25178-72, ISO 25178-73, ISO 25178-600, ISO 25178-601, ISO 25178-602, ISO 25178-603, ISO 25178-604, ISO 25178-605, ISO 25178-606, ISO 25178-607, ISO 25178-700, ISO 25178-701).
SCOPE
1.1 This guide describes some of the more common methods that are available for measuring the topographical features of a surface and provides an overview of the parameters that are used to quantify them. Being able to reliably derive a set of parameters that describe the texture of biomaterial surfaces is a key aspect in the manufacture of safe and effective implantable medical devices that have the potential to trigger an adverse biological reaction in situ.
1.2 This guide is not intended to apply to porous structures with average pore dimensions in excess of approximately 50 nm (0.05 μm).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 14-Mar-2024
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.42 - Biomaterials and Biomolecules for TEMPs
Relations
- Effective Date
- 15-Mar-2024
- Effective Date
- 15-Mar-2024
- Effective Date
- 15-Mar-2024
- Effective Date
- 15-Mar-2024
Overview
ASTM F2791-24: Standard Guide for Assessment of Surface Texture of Non-Porous Biomaterials in Two Dimensions is a crucial reference for professionals involved in the design, manufacture, and quality assurance of medical implants. Developed by ASTM International, this guide provides recommendations and best practices to measure and describe the surface texture of non-porous biomaterials in two dimensions. The assessment of surface characteristics-such as roughness, waviness, and form-is essential, as these features can influence the biological response, implant performance, and overall device safety.
This standard applies specifically to non-porous biomaterials and is not intended for porous structures with average pore dimensions exceeding 50 nm. The guide emphasizes the importance of standardized measurement methods and clear reporting of parameters, promoting consistency and reliability across the medical device sector.
Key Topics
Surface Texture Terminology
- Surface Texture: Local deviations of a surface from its ideal form, typically composed of long-wavelength features (like waviness) and superimposed short-wavelength roughness.
- Parameters: Includes amplitude (Ra, Rq, Rp, Rv), spatial (RSm), and hybrid (RΔq) descriptors used to quantify roughness and other profile features.
Measurement Techniques
- Contact Methods: Stylus or mechanical profilometers, which trace the surface using a stylus. Best suited for harder surfaces; may not be appropriate for soft or delicate materials due to potential surface damage.
- Non-Contact Methods: Optical techniques such as confocal microscopy, white light interferometry, and structured light 3D scanners. These methods are preferred for softer or highly polished surfaces.
Filtering and Data Processing
- Use of filters (high-pass, low-pass, band-pass) to isolate specific features of interest in the surface profile.
- Importance of choosing appropriate evaluation lengths and sampling intervals based on ISO 4287 and ISO 4288.
- The process of removing form and waviness to focus on roughness for relevant medical device applications.
Biocompatibility Implications
- The guide highlights the link between surface texture, surface chemistry, and biocompatibility.
- Proper characterization helps in controlling cell adhesion, tissue integration, and minimizing adverse reactions.
Applications
Medical Device Manufacturing
- Implants: Orthopedic, dental, cardiovascular, and other implantable biomaterials where surface texture affects biological integration and device longevity.
- Quality Control: Ensuring consistent manufacturing processes through standardized surface assessment.
- Product Development: Facilitating compliance with regulatory requirements and industry best practices for new device introductions.
Materials Science and Research
- Surface Characterization: Academic and industrial research in biomaterials increasingly relies on standardized texture measurement to compare results across studies.
- Optimization: By understanding and controlling surface texture, manufacturers can optimize properties such as wear resistance or cell response.
Regulatory and Compliance
- Use in documentation and submissions to demonstrate conformance to industry-recognized best practices and risk management protocols for surface characterization.
Related Standards
- ISO 3274: Geometrical product specifications for surface texture profile methods.
- ISO 4287/4288: Definitions, parameters, and rules for assessment of surface texture.
- ISO 10993-19: Physico-chemical, morphological, and topographical characterization of medical materials.
- ISO 25178 Series: Comprehensive standard for areal and profile surface measurements.
- ASTM F2664: Guide for assessing cell attachment to biomaterial surfaces by physical methods.
- ASTM F2312: Terminology relating to tissue-engineered medical products.
Implementing ASTM F2791-24 ensures that manufacturers and researchers are using validated, internationally recognized methods to assess the critical feature of surface texture in non-porous biomaterials. This supports device performance, patient safety, and global harmonization of medical device standards.
Buy Documents
ASTM F2791-24 - Standard Guide for Assessment of Surface Texture of Non-Porous Biomaterials in Two Dimensions
REDLINE ASTM F2791-24 - Standard Guide for Assessment of Surface Texture of Non-Porous Biomaterials in Two Dimensions
Get Certified
Connect with accredited certification bodies for this standard

BSMI (Bureau of Standards, Metrology and Inspection)
Taiwan's standards and inspection authority.
Sponsored listings
Frequently Asked Questions
ASTM F2791-24 is a guide published by ASTM International. Its full title is "Standard Guide for Assessment of Surface Texture of Non-Porous Biomaterials in Two Dimensions". This standard covers: SIGNIFICANCE AND USE 4.1 The term “surface texture” is used to describe the local deviations of a surface from an ideal shape. Surface texture usually consists of long wavelength repetitive features that occur as results of chatter, vibration, or heat treatments during the manufacture of implants. Short wavelength features superimposed on the long wavelength features of the surface, which may arise from polishing or etching of the implant, are referred to as roughness. 4.2 This guide provides an overview of techniques that are available for measuring the surface in terms of Cartesian coordinates and the parameters used to describe surface texture. It is important to appreciate that it is not possible to measure surface texture per se, but to derive values for parameters that can be used to describe it. ISO has published a series of standards on surface texture measurements that may be consulted for more information (ISO 3274, ISO 4287, ISO 4288, ISO 5436-2, ISO 10993-19, ISO 12179, ISO 13565-1, ISO 19606, ISO 21920-1, ISO 21920-2, ISO 21920-3, ISO 25178-1, ISO 25178-2, ISO 25178-3, ISO 25178-6, ISO 25178-70, ISO 25178-71, ISO 25178-72, ISO 25178-73, ISO 25178-600, ISO 25178-601, ISO 25178-602, ISO 25178-603, ISO 25178-604, ISO 25178-605, ISO 25178-606, ISO 25178-607, ISO 25178-700, ISO 25178-701). SCOPE 1.1 This guide describes some of the more common methods that are available for measuring the topographical features of a surface and provides an overview of the parameters that are used to quantify them. Being able to reliably derive a set of parameters that describe the texture of biomaterial surfaces is a key aspect in the manufacture of safe and effective implantable medical devices that have the potential to trigger an adverse biological reaction in situ. 1.2 This guide is not intended to apply to porous structures with average pore dimensions in excess of approximately 50 nm (0.05 μm). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 The term “surface texture” is used to describe the local deviations of a surface from an ideal shape. Surface texture usually consists of long wavelength repetitive features that occur as results of chatter, vibration, or heat treatments during the manufacture of implants. Short wavelength features superimposed on the long wavelength features of the surface, which may arise from polishing or etching of the implant, are referred to as roughness. 4.2 This guide provides an overview of techniques that are available for measuring the surface in terms of Cartesian coordinates and the parameters used to describe surface texture. It is important to appreciate that it is not possible to measure surface texture per se, but to derive values for parameters that can be used to describe it. ISO has published a series of standards on surface texture measurements that may be consulted for more information (ISO 3274, ISO 4287, ISO 4288, ISO 5436-2, ISO 10993-19, ISO 12179, ISO 13565-1, ISO 19606, ISO 21920-1, ISO 21920-2, ISO 21920-3, ISO 25178-1, ISO 25178-2, ISO 25178-3, ISO 25178-6, ISO 25178-70, ISO 25178-71, ISO 25178-72, ISO 25178-73, ISO 25178-600, ISO 25178-601, ISO 25178-602, ISO 25178-603, ISO 25178-604, ISO 25178-605, ISO 25178-606, ISO 25178-607, ISO 25178-700, ISO 25178-701). SCOPE 1.1 This guide describes some of the more common methods that are available for measuring the topographical features of a surface and provides an overview of the parameters that are used to quantify them. Being able to reliably derive a set of parameters that describe the texture of biomaterial surfaces is a key aspect in the manufacture of safe and effective implantable medical devices that have the potential to trigger an adverse biological reaction in situ. 1.2 This guide is not intended to apply to porous structures with average pore dimensions in excess of approximately 50 nm (0.05 μm). 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2791-24 is classified under the following ICS (International Classification for Standards) categories: 17.040.20 - Properties of surfaces. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2791-24 has the following relationships with other standards: It is inter standard links to ASTM F2791-15, ASTM F3510-21, ASTM F2150-19, ASTM F2902-16e1. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2791-24 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2791 − 24
Standard Guide for
Assessment of Surface Texture of Non-Porous Biomaterials
in Two Dimensions
This standard is issued under the fixed designation F2791; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope F2312 Terminology Relating to Tissue Engineered Medical
Products
1.1 This guide describes some of the more common meth-
F2664 Guide for Assessing the Attachment of Cells to
ods that are available for measuring the topographical features
Biomaterial Surfaces by Physical Methods
of a surface and provides an overview of the parameters that
2.2 Other Standards:
are used to quantify them. Being able to reliably derive a set of
ISO 3274 Geometrical Product Specifications (GPS)—
parameters that describe the texture of biomaterial surfaces is
Surface Texture: Profile Method—Nominal Characteris-
a key aspect in the manufacture of safe and effective implant-
tics of Contact (Stylus) Instruments
able medical devices that have the potential to trigger an
ISO 4287 Geometrical Product Specifications (GPS)—
adverse biological reaction in situ.
Surface Texture: Profile Method—Terms, Definitions and
1.2 This guide is not intended to apply to porous structures
Surface Texture Parameters
with average pore dimensions in excess of approximately
ISO 4288 Geometrical Product Specifications (GPS)—
50 nm (0.05 μm).
Surface Texture: Profile Method—Rules and Procedures
1.3 The values stated in SI units are to be regarded as
for the Assessment of Surface Texture
standard. No other units of measurement are included in this
ISO 5436-2 Geometrical Product Specifications (GPS)—
standard.
Surface Texture: Profile Method; Measurement
Standards—Part 2: Software Measurement Standards
1.4 This standard does not purport to address all of the
ISO 10993-19 Biological Evaluation of Medical Devices—
safety concerns, if any, associated with its use. It is the
Part 19: Physico-chemical, Morphological and Topo-
responsibility of the user of this standard to establish appro-
graphical Characterization of Materials
priate safety, health, and environmental practices and deter-
ISO 12179 Geometrical Product Specifications (GPS)—
mine the applicability of regulatory limitations prior to use.
Surface Texture: Profile Method—Calibration of Contact
1.5 This international standard was developed in accor-
(Stylus) Instruments
dance with internationally recognized principles on standard-
ISO 13565-1 Geometrical Product Specifications (GPS)—
ization established in the Decision on Principles for the
Surface Texture: Profile Method—Surfaces Having Strati-
Development of International Standards, Guides and Recom-
fied Functional Properties; Filtering and General Measure-
mendations issued by the World Trade Organization Technical
ment Conditions
Barriers to Trade (TBT) Committee.
ISO 19606 Fine Ceramics (Advanced Ceramics, Advanced
Technical Ceramics)—Test Method for Surface Rough-
2. Referenced Documents
ness of Fine Ceramic Films by Atomic Force Microscopy
2.1 ASTM Standards:
ISO 21920-1 Geometrical Product Specifications (GPS)—
C813 Test Method for Hydrophobic Contamination on Glass
Surface Texture: Profile—Part 1: Indication of Surface
by Contact Angle Measurement
Texture
ISO 21920-2 Geometrical Product Specifications (GPS)—
Surface Texture: Profile—Part 2: Terms, Definitions and
This guide is under the jurisdiction of ASTM Committee F04 on Medical and
Surface Texture Parameters
Surgical Materials and Devices and is the direct responsibility of Subcommittee
ISO 21920-3 Geometrical Product Specifications (GPS)—
F04.42 on Biomaterials and Biomolecules for TEMPs.
Current edition approved March 15, 2024. Published March 2024. Originally Surface Texture: Profile—Part 3: Specification Operators
approved in 2009. Last previous edition approved in 2015 as F2791 – 15. DOI:
ISO 25178-1 Geometrical Product Specifications (GPS)—
10.1520/F2791-24.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available from American National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2791 − 24
Surface Texture: Areal—Part 1: Indication of Surface 3.1.1 biomaterial, n—any substance (other than a drug),
Texture synthetic or natural, that can be used as a system or part of a
system that treats, augments, or replaces any tissue, organ, or
ISO 25178-2 Geometrical Product Specifications (GPS)—
function of the body. F2312
Surface Texture: Areal—Part 2: Terms, Definitions and
Surface Texture Parameters
3.1.2 evaluation length, ln, n—length in the direction of the
ISO 25178-3 Geometrical Product Specifications (GPS)—
x-axis used for assessing the profile under evaluation.
Surface Texture: Areal—Part 3: Specification Operators
3.1.2.1 Discussion—The evaluation length may contain one
ISO 25178-6 Geometrical Product Specifications (GPS)—
or more sampling lengths. ISO 4287
Surface Texture: Areal—Part 6: Classification of Methods
3.1.3 hydrophilic, adj—having a strong affinity for water;
for Measuring Surface Texture
wettable.
ISO 25178-70 Geometrical Product Specification (GPS)—
3.1.3.1 Discussion—Hydrophilic surfaces exhibit zero con-
Surface Texture: Areal—Part 70: Material Measures
tact angles. C813
ISO 25178-71 Geometrical Product Specifications (GPS)—
3.1.4 hydrophobic, adj—having little affinity for water;
Surface Texture: Areal—Part 71: Software Measurement
nonwettable.
Standards
3.1.4.1 Discussion—Hydrophobic surfaces exhibit contact
ISO 25178-72 Geometrical Product Specifications (GPS)—
angles appreciably greater than zero: generally greater than 45°
Surface Texture: Areal—Part 72: XML File Format x3p
for the advancing angle. C813
ISO 25178-73 Geometrical Product Specifications (GPS)—
3.1.5 implant, n—in medicine, an object, structure, or device
Surface Texture: Areal—Part 73: Terms and Definitions
intended to reside within the body for diagnostic, prosthetic, or
for Surface Defects on Material Measures
other therapeutic purposes. F2664
ISO 25178-600 Geometrical Product Specifications (GPS)—
3.1.6 primary profile, n—total profile after application of the
Surface Texture: Areal—Part 600: Metrological Charac-
short wavelength filters. ISO 3274
teristics for Areal Topography Measuring Methods
ISO 25178-601 Geometrical Product Specifications (GPS)— 3.1.7 profile peak, n—an outwardly directed (from the
material to the surrounding medium) portion of the assessed
Surface Texture: Areal—Part 601: Nominal Characteris-
profile connecting two adjacent points of the intersection of the
tics of Contact (Stylus) Instruments
profile with the x-axis. ISO 4287
ISO 25178-602 Geometrical Product Specifications (GPS)—
Surface Texture: Areal—Part 602: Nominal Characteris-
3.1.8 profile valley, n—an inwardly directed (from surround-
tics of Non-Contact (Confocal Chromatic Probe) Instru-
ing medium to material) portion of the assessed profile
ments
connecting two adjacent points of the intersection of the
ISO 25178-603 Geometrical Product Specifications (GPS)— assessed profile with the x-axis. ISO 4287
Surface Texture: Areal—Part 603: Nominal Characteris-
3.1.9 real surface, n—of a workpiece, a set of features
tics of Non-Contact (Phase-Shifting Interferometric Mi-
which physically exist and separate the entire workpiece from
croscopy) Instruments
the surrounding medium. ISO 25178-600
ISO 25178-604 Geometrical Product Specifications (GPS)—
3.1.10 sampling length, lr, n—length in the direction of the
Surface Texture: Areal—Part 604: Nominal Characteris-
x-axis used for identifying the irregularities characterizing the
tics of Non-Contact (Coherence Scanning Interferometry)
profile under evaluation. ISO 4287
Instruments
3.1.11 scaffold, n—a support, delivery vehicle, or matrix for
ISO 25178-605 Geometrical Product Specifications (GPS)—
facilitating the migration, binding, or transport of cells or
Surface Texture: Areal—Part 605: Nominal Characteris-
bioactive molecules used to replace, repair, or regenerate
tics of Non-Contact (Point Autofocus Probe) Instruments
tissues. F2312
ISO 25178-606 Geometrical Product Specification (GPS)—
3.1.12 surface profile, n—profile that results from the inter-
Surface Texture: Areal—Part 606: Nominal Characteris-
section of the real surface by a specified plane. ISO 4287
tics of Non-Contact (Focus Variation) Instruments
3.1.12.1 Discussion—In practice, it is usual to choose a
ISO 25178-607 Geometrical Product Specifications (GPS)—
plane with a normal that nominally lies parallel to the real
Surface Texture: Areal—Part 607: Nominal Characteris-
surface and in a suitable direction.
tics of Non-Contact (Confocal Microscopy) Instruments
3.1.13 surface texture, n—irregularities on a surface (peaks
ISO 25178-700 Geometrical Product Specifications (GPS)—
and valleys) produced by the forming process.
Surface Texture: Areal—Part 700: Calibration, Adjust-
ment and Verification of Areal Topography Measuring
3.2 Definitions of Terms Specific to This Standard:
Instruments
3.2.1 lay, n—the direction of the predominant surface pat-
ISO 25178-701 Geometrical Product Specifications (GPS)—
tern.
Surface Texture: Areal—Part 701: Calibration and Mea-
surement Standards for Contact (Stylus) Instruments
4. Significance and Use
4.1 The term “surface texture” is used to describe the local
3. Terminology
deviations of a surface from an ideal shape. Surface texture
3.1 Definitions: usually consists of long wavelength repetitive features that
F2791 − 24
occur as results of chatter, vibration, or heat treatments during penetration depth is 10 nm or below (1)). The chemical
the manufacture of implants. Short wavelength features super- species present on the surface together with the surface
imposed on the long wavelength features of the surface, which topography determine how hydrophilic the surface is. Measur-
may arise from polishing or etching of the implant, are referred ing the contact angle between the surface and a fluid, usually
to as roughness. water, can assess the degree of hydrophilicity of a surface. Care
should be taken when comparing contact angle measurements
4.2 This guide provides an overview of techniques that are
made on different surfaces, as the relative contributions from
available for measuring the surface in terms of Cartesian
the surface chemistry and texture are unlikely to be the same.
coordinates and the parameters used to describe surface tex-
ture. It is important to appreciate that it is not possible to
6. Surfaces and Surface Profiles
measure surface texture per se, but to derive values for
6.1 Conventionally, surfaces are described in Cartesian
parameters that can be used to describe it. ISO has published a
coordinates where the x-axis is defined as being perpendicular
series of standards on surface texture measurements that may
to the lay direction. The y-axis is in-plane and is perpendicular
be consulted for more information (ISO 3274, ISO 4287,
to the x-axis direction. The z-axis is out of plane. The profile of
ISO 4288, ISO 5436-2, ISO 10993-19, ISO 12179,
a surface that has a uniform, non-directional texture can be
ISO 13565-1, ISO 19606, ISO 21920-1, ISO 21920-2, ISO
measured at any in-plane orientation (see Fig. 1(A)); however,
21920-3, ISO 25178-1, ISO 25178-2, ISO 25178-3, ISO
several profiles at different orientations should be measured to
25178-6, ISO 25178-70, ISO 25178-71, ISO 25178-72, ISO
find the maximum amplitude (see Fig. 1(A)). For patterned
25178-73, ISO 25178-600, ISO 25178-601, ISO 25178-602,
surfaces that have periodic features, a lay, the orientation of the
ISO 25178-603, ISO 25178-604, ISO 25178-605, ISO 25178-
profile is at right angles to it (see Fig. 1(B)).
606, ISO 25178-607, ISO 25178-700, ISO 25178-701).
6.2 The measured surface is composed of three components:
5. The Relationship Between Surface Texture, Surface
form, waviness, and roughness. The form corresponds to the
Chemistry, Surface Energy, and Biocompatibility
underlying shape and tilt of the surface with respect to the
measuring platform. The software packages used for surface
5.1 The biocompatibility of materials is influenced by many
texture analysis all have a methodology for removing the form
factors such as size, shape, material bulk, and surface chemical
from the surface. The “corrected” surface can then be used to
composition, surface energy, and surface topography (ISO
obtain a 2D profile that describes the surface texture. This
10993-19). Changing any one of these related characteristics of
profile after removal of form is defined according to ISO 3274
a biocompatible material can have a significant effect on cell
as the primary profile. The stages involved in the analysis of
behavior. The response of a cell to a biomaterial can be
the measured profile through primary profile to the roughness
assessed by measuring the adhesive strength between it and the
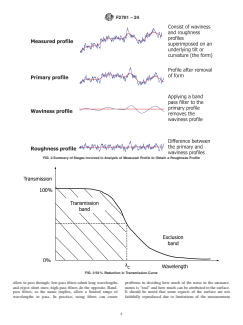
profile are shown in Fig. 2.
underlying surface, monitoring changes in its shape or in the
expression of biomarkers.
7. Filtering and the Cut-Off Wavelength
5.2 The chemical species present on a surface can be
7.1 Surface data can be filtered to remove unwanted noise or
mapped in detail using surface sensitive analysis techniques
to remove texture information at unwanted wavelengths. Fil-
(for example, X-ray photoelectron spectroscopy where the
ters are classified according to the spatial periodicity that they
The boldface numbers in parentheses refer to a list of references at the end of
this standard.
NOTE 1—The surface shown in (A) has no directionality or lay, therefore, profiles can be oriented at any angle. Profiles (dashed line arrow) are drawn
perpendicular to the lay (solid line arrow) in surfaces that have directionality (B).
FIG. 1 Profile Orientation and Surface Features
F2791 − 24
FIG. 2 Summary of Stages Involved in Analysis of Measured Profile to Obtain a Roughness Profile
FIG. 3 50 % Reduction in Transmission Curve
allow to pass through; low-pass filters admit long wavelengths problems in deciding how much of the noise in the measure-
and reject short ones; high-pass filters do the opposite. Band- ments is “real” and how much can be attributed to the surface.
pass filters, as the name implies, allow a limited range of It should be noted that some aspects of the surface are not
wavelengths to pass. In practice, using filters can create faithfully reproduced due to limitations of the measurement
F2791 − 24
method, for example, an inability to track the sides of steep 8.1.1 Amplitude parameters, which are measures of varia-
valleys that is in essence a form of filtering. This topic is tions in profile height. These parameters are split into two
further discussed in Section 11. subclasses: averaging parameters, and peak and valley param-
eters;
7.2 Filters used in surface texture measurements do not have
8.1.2 Spatial parameters, which describe in-plane variations
a sharp cut-off in spatial frequency above or below which
of surface texture; and
information is rejected. This gradual attenuation of high or low
spatial frequency data helps avoid distortion of the measure-
8.1.3 Hybrid parameters, which combine both amplitude
ments that can occur when strong features are close to the and spatial information (for example, mean slope).
filtration limits. The point on the transmission curve at which
8.2 Ra—The most widely used parameter to quantify sur-
the transmitted signal is reduced to 50 % is referred to as the
face texture is the arithmetical mean deviation of the absolute
cut-off wavelength, λc, of the filter (Fig. 3). For measurements
ordinate values, Z(x), of the profile from a center line (see
made using a stylus instrument (Section 11), the choice of λc
Table 2 and Fig. 5). Despite its common usage, Ra does not
depends on the sampling frequency and the speed at which the
provide a truly accurate representation of a surface profile since
stylus moves over the surface. For example, measurements
–1 any information regarding peak heights or valley depths can be
made at intervals of 0.01 mm from a device moving at 1 mms
lost in its derivation. This insensitivity to surface texture is
will generate data at a frequency of 100 Hz. Increasing the
apparent in Fig. 6, which shows that quite different profiles can
sampling interval to 0.1 mm will reduce the frequency at which
have the same Ra value. The statistical significance of Ra is
data are obtained to 10 Hz. A high-pass filter that suppresses all
improved by averaging the values obtained for each of the five
frequencies below 10 Hz effectively removes any surface
sampling lengths.
irregularities larger than 0.1 mm spacing from the data. Hence,
filters can be used to bias the experimental data towards
8.3 Rq—The root mean square value of all distances of the
detecting profile (surface texture after applying a low-pass to
measured profile away from the center line, Rq, although
filter the data), waviness (after applying a band-pass filter), and
similar in terms of its derivation to Ra, has a subtle but
roughness (after applying a high-pass filter). Measurement
significant difference. The deviations of the peak heights and
conditions are set for filters according to the respective values
valley depths from the midline appear as a squared term in Rq.
of the sampling interval, measurement speed, and filtration
That increases its sensitivity to high peaks or deep valleys. This
limits, according to ISO 3274.
sensitivity can be useful, but it should be noted that the
presence of a foreign body, for example, hair or a scratch in the
7.3 ISO 4287 specifies that 2D roughness parameters need
surface, can have a significant influence on the value of Rq.
to be determined over five sequential sampling lengths, lr,
unless otherwise specified. This grouping of five serial sam-
8.4 Rsk—Skewness, the distribution of peak heights and
pling lengths is referred to as the evaluation length, ln. The
valley depths, provides valuable information about surface
sampling length varies according to the length scale of the
texture. A surface that has a range of peak heights and valley
texture being assessed; larger features require a long sampling
depths will have a bell-shaped probability distribution centered
length. Guidance as to which sampling length to use for a given
on the mean. The dimensionless skewness parameter, Rsk, is
range of feature sizes is shown in Table 1.
used to quantify bias in the shape of this distribution. The
skewness of a perfectly random surface with a wide range of
TABLE 1 Guide to Choosing Sampling Lengths for the
peak heights and valley depths is zero. If the surface has more
A
Measurement of Periodic Profiles
valleys than peaks, then the distribution will skew away from
Mean profile element Sampling length,
the ideal distribution prod
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2791 − 15 F2791 − 24
Standard Guide for
Assessment of Surface Texture of Non-Porous Biomaterials
in Two Dimensions
This standard is issued under the fixed designation F2791; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This guide describes some of the more common methods that are available for measuring the topographical features of a
surface and provides an overview of the parameters that are used to quantify them. Being able to reliably derive a set of parameters
that describe the texture of biomaterial surfaces is a key aspect in the manufacture of safe and effective implantable medical devices
that have the potential to trigger an adverse biological reaction in situ.
1.2 This guide is not intended to apply to porous structures with average pore dimensions in excess of approximately 50 nm 50 nm
(0.05 μm).
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
C813 Test Method for Hydrophobic Contamination on Glass by Contact Angle Measurement
F2312 Terminology Relating to Tissue Engineered Medical Products
F2450 Guide for Assessing Microstructure of Polymeric Scaffolds for Use in Tissue-Engineered Medical Products
F2664 Guide for Assessing the Attachment of Cells to Biomaterial Surfaces by Physical Methods
2.2 Other Standards:
ISO 3274 Geometrical Product Specifications (GPS)—Surface Texture: Profile Method—Nominal Characteristics of Contact
(Stylus) Instruments
ISO 4287 Geometrical Product Specifications (GPS)—Surface Texture: Profile Method—Terms, Definitions and Surface
Texture Parameters
This guide is under the jurisdiction of ASTM Committee F04 on Medical and Surgical Materials and Devices and is the direct responsibility of Subcommittee F04.42
on Biomaterials and Biomolecules for TEMPs.
Current edition approved May 1, 2015March 15, 2024. Published June 2015March 2024. Originally approved in 2009. Last previous edition approved in 20142015 as
F2791F2791 – 15.– 14. DOI: 10.1520/F2791-15. DOI: 10.1520/F2791-24.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2791 − 24
ISO 4288 Geometrical Product Specifications (GPS)—Surface Texture: Profile Method—Rules and Procedures for the
Assessment of Surface Texture
ISO 5436-2 Geometrical Product Specifications (GPS)—Surface Texture: Profile Method; Measurement Standards—Part 2:
Software Measurement Standards
ISO 10993-19 Biological Evaluation of Medical Devices—Part 19: Physico-chemical, Morphological and Topographical
Characterization of Materials
ISO 12179 Geometrical Product Specifications (GPS)—Surface Texture: Profile Method—Calibration of Contact (Stylus)
Instruments
ISO 13565–1ISO 13565-1 Geometrical Product Specifications (GPS)—Surface Texture: Profile Method—Surfaces Having
Stratified Functional Properties; Filtering and General Measurement Conditions
ISO 19606 Fine Ceramics (Advanced Ceramics, Advanced Technical Ceramics)—Test Method for Surface Roughness of Fine
Ceramic Films by Atomic Force Microscopy
ISO 21920-1 Geometrical Product Specifications (GPS)—Surface Texture: Profile—Part 1: Indication of Surface Texture
ISO 21920-2 Geometrical Product Specifications (GPS)—Surface Texture: Profile—Part 2: Terms, Definitions and Surface
Texture Parameters
ISO 21920-3 Geometrical Product Specifications (GPS)—Surface Texture: Profile—Part 3: Specification Operators
ISO 25178-1 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 1: Indication of Surface Texture
ISO 25178-2 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 2: Terms, Definitions and Surface
Texture Parameters
ISO 25178-3 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 3: Specification Operators
ISO 25178-6 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 6: Classification of Methods for
Measuring Surface Texture
ISO 25178-70 Geometrical Product Specification (GPS)—Surface Texture: Areal—Part 70: Material Measures
ISO 25178-71 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 71: Software Measurement Standards
ISO 25178-72 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 72: XML File Format x3p
ISO 25178-73 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 73: Terms and Definitions for Surface
Defects on Material Measures
ISO 25178-600 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 600: Metrological Characteristics for
Areal Topography Measuring Methods
ISO 25178-601 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 601: Nominal Characteristics of
Contact (Stylus) Instruments
ISO 25178-602 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 602: Nominal Characteristics of
Non-Contact (Confocal Chromatic Probe) Instruments
ISO 25178-603 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 603: Nominal Characteristics of
Non-Contact (Phase-Shifting Interferometric Microscopy) Instruments
ISO 25178-604 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 604: Nominal Characteristics of
Non-Contact (Coherence Scanning Interferometry) Instruments
ISO 25178-605 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 605: Nominal Characteristics of
Non-Contact (Point Autofocus Probe) Instruments
ISO 25178-606 Geometrical Product Specification (GPS)—Surface Texture: Areal—Part 606: Nominal Characteristics of
Non-Contact (Focus Variation) Instruments
ISO 25178-607 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 607: Nominal Characteristics of
Non-Contact (Confocal Microscopy) Instruments
ISO 25178-700 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 700: Calibration, Adjustment and
Verification of Areal Topography Measuring Instruments
ISO 25178-701 Geometrical Product Specifications (GPS)—Surface Texture: Areal—Part 701: Calibration and Measurement
Standards for Contact (Stylus) Instruments
3. Terminology
3.1 Definitions of Terms Specific to This Standard:Definitions:
3.1.1 biomaterial, n—any substance (other than a drug), synthetic or natural, that can be used as a system or part of a system that
treats, augments, or replaces any tissue, organ, or function of the body. F2664F2312
3.1.2 evaluation length, ln, n—length in the direction of the x-axis used to assessfor assessing the profile under evaluation.
3.1.2.1 Discussion—
The evaluation length may contain one or more sampling lengths. ISO 4287
F2791 − 24
3.1.3 hydrophilic, adj—having a strong affinity for water; wettable.
3.1.3.1 Discussion—
Hydrophilic surfaces exhibit zero contact angles. C813
3.1.4 hydrophobic, adj—having little affinity for water; nonwettable.
3.1.4.1 Discussion—
Hydrophobic surfaces exhibit contact angles appreciably greater than zero: generally greater than 45° for the advancing angle.
C813
3.1.5 implant, n—a substancein medicine, or object that is put in the body as a prosthesis, or for treatment or diagnosis.an object,
structure, or device intended to reside within the body for diagnostic, prosthetic, or other therapeutic purposes. F2664
3.1.6 lay, n—the direction of the predominant surface pattern. ISO 13565–1
3.1.6 primary profile, n—thetotal profile after application of the short wavelength filters. ISO 3274
3.1.7 profile peak, n—an outwardly directed (from the material to the surrounding medium) portion of the assessed profile
connecting two adjacent points of the intersection of the profile with the x-axis. ISO 4287
3.1.8 profile valley, n—an inwardly directed (from surrounding medium to material) portion of the assessed profile connecting two
adjacent points of the intersection of the assessed profile with the x-axis. ISO 4287
3.1.9 real surface, n—surface limiting theof a workpiece, body and separating it a set of features which physically exist and
separate the entire workpiece from the surrounding medium. ISO 4287ISO 25178-600
3.1.10 sampling length, lr, n—length in the direction of the x-axis used for identifying the irregularities characterizing the profile
under evaluation. ISO 4287
3.1.11 scaffold, n—a support, delivery vehicle or metricvehicle, or matrix for facilitating the migration, binding, or transport of
cells or bioactive molecules used to replace, repair, or regenerate tissues. F2450F2312
3.1.12 surface profile, n—profile that results from the intersection of the real surface by a specified plane. ISO 4287
3.1.12.1 Discussion—
In practice, it is usual to choose a plane with a normal that nominally lies parallel to the real surface and in a suitable direction.
ISO 4287
NOTE 1—The surface shown in (A) has no directionality or lay, therefore, profiles can be oriented at any angle. Profiles (dashed line arrow) are drawn
perpendicular to the lay (solid line arrow) in surfaces that have directionality (B).
FIG. 1 Profile Orientation and Surface Features
F2791 − 24
3.1.13 surface texture, n—irregularities on a surface (peaks and valleys) produced by the forming process.
3.2 Definitions of Terms Specific to This Standard:
3.2.1 lay, n—the direction of the predominant surface pattern.
4. Significance and Use
4.1 The term “surface texture” is used to describe the local deviations of a surface from an ideal shape. Surface texture usually
consists of long wavelength repetitive features that occur as results of chatter, vibration, or heat treatments during the manufacture
of implants. Short wavelength features superimposed on the long wavelength features of the surface, which may arise from
polishing or etching of the implant, are referred to as roughness.
4.2 This guide provides an overview of techniques that are available for measuring the surface in terms of Cartesian coordinates
and the parameters used to describe surface texture. It is important to appreciate that it is not possible to measure surface texture
per se, but to derive values for parameters that can be used to describe it. ISO has published a series of standards on surface texture
measurements that may be consulted for more information (ISO 3274, ISO 4287, ISO 4288, ISO 5436-2, ISO 10993-19, ISO
12179, ISO 13565-1, ISO 19606, ISO 21920-1, ISO 21920-2, ISO 21920-3, ISO 25178-1, ISO 25178-2, ISO 25178-3, ISO
25178-6, ISO 25178-70, ISO 25178-71, ISO 25178-72, ISO 25178-73, ISO 25178-600, ISO 25178-601, ISO 25178-602, ISO
25178-603, ISO 25178-604, ISO 25178-605, ISO 25178-606, ISO 25178-607, ISO 25178-700, ISO 25178-701).
5. The Relationship Between Surface Texture, Surface Chemistry, Surface Energy, and Biocompatibility
5.1 The biocompatibility of materials is influenced by many factors such as size, shape, material bulk, and surface chemical
composition, surface energy, and surface topography. topography (ISO 10993-19). Changing any one of these related
characteristics of a biocompatible material can have a significant effect on cell behavior. The response of a cell to a biomaterial
can be assessed by measuring the adhesive strength between it and the underlying surface, monitoring changes in its shape or in
the expression of biomarkers.
5.2 The chemical species present on a surface can be mapped in detail using surface sensitive analysis techniques (for example,
X-ray photoelectron spectroscopy where the penetration depth is 10 nm or below (1)). The chemical species present on the surface
together with the surface topography determine how hydrophilic the surface is. Measuring the contact angle between the surface
and a fluid, usually water, can assess the degree of hydrophilicity of a surface. Care should be taken when comparing contact angle
measurements made on different surfaces, as the relative contributions from the surface chemistry and texture are unlikely to be
the same.
6. Surfaces and Surface Profiles
6.1 Conventionally, surfaces are described in Cartesian coordinates where the x-axis is defined as being perpendicular to the lay
direction. The y-axis is in-plane and is perpendicular to the x-axis direction. The z-axis is out of plane. The profile of a surface that
has a uniform, non-directional texture can be measured at any in-plane orientation (see Fig. 1(A)); however, several profiles at
different orientations should be measured to find the maximum amplitude (see Fig. 1(A)). For patterned surfaces that have periodic
features, a lay, the orientation of the profile is at right angles to it (see Fig. 1(B)).
6.2 The measured surface is composed of three components: form, waviness, and roughness. The form corresponds to the
underlying shape and tilt of the surface with respect to the measuring platform. The software packages used for surface texture
analysis all have a methodology for removing the form from the surface. The “corrected” surface can then be used to obtain a
2-D2D profile that describes the surface texture. This profile after removal of form is defined according to ISO 3274 as the primary
profile. The stages involved in the analysis of the measured profile through primary profile to the roughness profile are shown in
Fig. 2.
7. Filtering and the Cut-Off Wavelength
7.1 Surface data can be filtered to remove unwanted noise or to remove texture information at unwanted wavelengths. Filters are
The boldface numbers in parentheses refer to a list of references at the end of this standard.
F2791 − 24
FIG. 2 Summary of Stages Involved in Analysis of Measured Profile to Obtain a Roughness Profile
classified according to the spatial periodicity that they allow to pass through; low-pass filters admit long wavelengths and reject
short ones; high-pass filters do the opposite. Band-pass filters, as the name implies, allow a limited range of wavelengths to pass.
In practice, using filters can create problems in deciding how much of the noise in the measurements is “real” and how much can
be attributed to the surface. It should be noted that some aspects of the surface are not faithfully reproduced due to limitations of
the measurement method, for example, an inability to track the sides of steep valleys that is in essence a form of filtering. This
topic is further discussed in Section 11.
7.2 Filters used in surface texture measurements do not have a sharp cut-off in spatial frequency above or below which information
is rejected. This gradual attenuation of high or low spatial frequency data helps avoid distortion of the measurements that can occur
when strong features are close to the filtration limits. The point on the transmission curve at which the transmitted signal is reduced
to 50 % is referred to as the cut-off wavelength, λc, of the filter (Fig. 3). For measurements made using a stylus instrument (Section
11), the choice of λc depends on the sampling frequency and the speed at which the stylus moves over the surface. For example,
–1
measurements made at intervals of 0.01 mm from a device moving at 1 mms will generate data at a frequency of 100 Hz.
Increasing the sampling interval to 0.1 mm will reduce the frequency at which data are obtained to 10 Hz. A high-pass filter that
suppresses all frequencies below 10 Hz effectively removes any surface irregularities larger than 0.1 mm spacing from the data.
Hence, filters can be used to bias the experimental data towards detecting profile (surface texture after applying a low-pass to filter
the data), waviness (after applying a band-pass filter), and roughness (after applying a high-pass filter). Measurement conditions
are set for filters according to the respective values of the sampling interval, measurement speed, and filtration limits, according
to ISO 3274.
7.3 ISO 4287 specifies that 2D roughness parameters need to be determined over five sequential sampling lengths, lr, unless
otherwise specified. This grouping of five serial sampling lengths is referred to as the evaluation length, ln. The sampling length
varies according to the length scale of the texture being assessed; larger features require a long sampling length. Guidance as to
which sampling length to use for a given range of feature sizes is shown in Table 1.
ISO 4287 specifies that 2-D roughness parameters need to be determined over five sequential sampling lengths, lr, unless
otherwise specified. This grouping of five serial sampling lengths is referred to as the evaluation length, ln. The sampling length
varies according to the length scale of the texture being assessed; larger features require a long sampling length. Guidance as to
which sampling length to use for a given range of feature sizes is shown in Table 1. It may be necessary to perform one or more
F2791 − 24
FIG. 3 50 % Reduction in Transmission Curve
TABLE 1 Guide to Choosing Sampling Lengths for the
A
Measurement of Periodic Profiles
Mean profile element Sampling length,
width, RSm (μm) lr (μm)
13 < RSm # 40 80
40 < RSm # 130 250
130 < RSm # 400 800
400 < RSm # 1300 2500
1300 < RSm # 4000 8000
A
Based on ISO 4288. The evaluation length is usually taken to be five times the
sampling length.
iterations to identify the best value for lr. This can be achieved by calculating the mean width of a profile element, RSm (see Fig.
4), from a measured profile where the value for lr is based on a best guess. This initial iteration will enable a new value for RSm
to be determined and that leads to a potential revision of lr according to Table 1.
NOTE 1—The sum of each peak and adjacent valley, Xs is RSm.
i
FIG. 4 Mean Width of Profile Elements Over the Evaluation Length
F2791 − 24
TABLE 2 A Summary of Commonly Used Parameters for Quantifying 2-D2D Roughness Profiles
Type of
Parameter Definition
Parameter
Amplitude Ra Arithmetic mean deviation of the absolute ordinate value from a mean line.
(Average
Rq Root-mean-squared value of the deviation of ordinate values from a midline.
of cordinates) Rsk A measure of the skewness in the distribution of peaks and valleys across a surface.
Rku A measure of the similarity of the distribution of measured peaks and valleys to a Gaussian distribution (kurtosis).
Amplitude Rp Maximum profile height above a mean line.
(Peak and Rv Maximum profile depth below a mean line.
valley) Rz Difference between Rp and Rv.
Rc The mean value of the profile element widths within a sampling length.
Rt The total height of profile.
Spatial RSm The mean value of the profile element widths within a sampling length.
Hybrid RΔq The root mean square slope, dz/dx, over the length of the profile at a location y on the surface.
8. Quantification of Surface Profiles
8.1 Parameters that are used to characterize 2-D2D surface profiles are grouped as:
8.1.1 Amplitude parameters, which are measures of variations in profile height. These parameters are split into two subclasses:
averaging parameters, and peak and valley parameters;
8.1.2 Spatial
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...