ASTM G61-86(2018)
(Test Method)Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility of Iron-, Nickel-, or Cobalt-Based Alloys
Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility of Iron-, Nickel-, or Cobalt-Based Alloys
SIGNIFICANCE AND USE
3.1 An indication of the susceptibility to initiation of localized corrosion in this test method is given by the potential at which the anodic current increases rapidly. The more noble this potential, obtained at a fixed scan rate in this test, the less susceptible is the alloy to initiation of localized corrosion. The results of this test are not intended to correlate in a quantitative manner with the rate of propagation that one might observe in service when localized corrosion occurs.
3.2 In general, once initiated, localized corrosion can propagate at some potential more electropositive than that at which the hysteresis loop is completed. In this test method, the potential at which the hysteresis loop is completed is determined at a fixed scan rate. In these cases, the more electropositive the potential at which the hysteresis loop is completed the less likely it is that localized corrosion will occur.
3.3 If followed, this test method will provide cyclic potentiodynamic anodic polarization measurements that will reproduce data developed at other times in other laboratories using this test method for the two specified alloys discussed in 3.4. The procedure is used for iron-, nickel-, or cobalt-based alloys in a chloride environment.
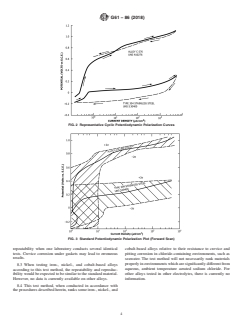
3.4 A standard potentiodynamic polarization plot is included. These reference data are based on the results from five different laboratories that followed the standard procedure, using specific alloys of Type 304 stainless steel, UNS S30400 and Alloy C-276, UNS N10276.3 Curves are included which have been constructed using statistical analysis to indicate the acceptable range of polarization curves.
3.5 The availability of a standard test method, standard material, and standard plots should make it easy for an investigator to check his techniques to evaluate susceptibility to localized corrosion.
SCOPE
1.1 This test method covers a procedure for conducting cyclic potentiodynamic polarization measurements to determine relative susceptibility to localized corrosion (pitting and crevice corrosion) for iron-, nickel-, or cobalt-based alloys in a chloride environment. This test method also describes an experimental procedure which can be used to check one's experimental technique and instrumentation.
1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2018
- Technical Committee
- G01 - Corrosion of Metals
- Drafting Committee
- G01.11 - Electrochemical Measurements in Corrosion Testing
Relations
- Effective Date
- 01-May-2018
- Effective Date
- 01-May-2019
- Effective Date
- 15-Dec-2014
- Effective Date
- 01-Nov-2014
- Effective Date
- 01-Dec-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 01-Feb-2013
- Effective Date
- 15-Nov-2012
- Effective Date
- 15-Nov-2011
- Effective Date
- 01-May-2010
- Effective Date
- 01-Mar-2006
- Effective Date
- 01-Nov-2004
- Effective Date
- 01-Nov-2004
- Effective Date
- 10-Aug-1999
Overview
ASTM G61-86(2018) is a key international standard from ASTM International that outlines the test method for conducting cyclic potentiodynamic polarization measurements to determine the localized corrosion susceptibility of iron-, nickel-, or cobalt-based alloys in a chloride environment. This method is widely recognized for evaluating corrosion resistance, particularly against pitting and crevice corrosion. Adoption of ASTM G61-86(2018) supports consistent, repeatable testing to assess materials’ behavior in environments such as seawater or other chloride-containing solutions.
Key Topics
- Cyclic Potentiodynamic Polarization: The procedure uses a controlled scan of electrode potential to provoke and measure corrosion behavior, providing insight into the initiation and propagation of localized corrosion on metallic alloys.
- Susceptibility Evaluation: The key results are the potentials at which rapid increases in anodic current occur, indicating corrosion susceptibility. A more noble (positive) potential means lower probability of corrosion initiation.
- Standardization and Precision: The method allows for reproducible data across different laboratories and equipment, assisting in quality control and research. Reference polarization plots and standard alloy samples are part of this standardization approach.
- Materials and Environments: The standard is relevant for iron-based alloys (like Type 304 stainless steel), nickel-based alloys, and cobalt-based alloys, especially for exposure to chloride ions.
- Quality Assurance: By following this method, laboratories and investigators can benchmark their corrosion testing techniques and data interpretation, ensuring credibility and comparability of results.
Applications
- Materials Selection: Used extensively in industries such as chemical processing, marine, and power generation to select alloys with superior resistance to pitting and crevice corrosion.
- Product Development: Supports engineers and researchers in developing new corrosion-resistant materials by providing a standardized way to gauge localized corrosion performance.
- Comparative Assessment: Enables manufacturers and end-users to compare different alloys and verify claims regarding corrosion resistance in chloride-containing environments.
- Quality Control: Acts as a validation tool for laboratories wishing to test their techniques and equipment by using standard reference materials and polarization plots.
- Regulatory Compliance: Facilitates adherence to recognized international test procedures, important for global trade and product certification.
Related Standards
- ASTM G3: Practice for conventions applicable to electrochemical measurements in corrosion testing, providing guidance on data interpretation and reporting.
- ASTM G5: Reference test method for making potentiodynamic anodic polarization measurements, closely related and often referenced for general polarization procedures.
- ASTM D1193: Specification for reagent water, detailing water purity requirements – essential for accuracy in electrochemical corrosion testing.
- ISO and NACE Standards: While ASTM G61 is widely referenced, users may also consult international standards from ISO or guidance from NACE International for related test methods and corrosion evaluation procedures.
Using ASTM G61-86(2018) provides significant practical value in the field of corrosion testing. The standard’s repeatable methodology and supporting resources, such as standard samples and polarization plots, help ensure that results from different laboratories are comparable and reliable. Laboratories, manufacturers, and engineers benefit from improved material selection, risk assessment, and compliance with best practices in localized corrosion testing. For any organization needing to evaluate or guarantee corrosion resistance of stainless steels and high-performance alloys in aggressive chloride environments, ASTM G61-86(2018) is an essential reference.
Buy Documents
ASTM G61-86(2018) - Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility of Iron-, Nickel-, or Cobalt-Based Alloys
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM G61-86(2018) is a standard published by ASTM International. Its full title is "Standard Test Method for Conducting Cyclic Potentiodynamic Polarization Measurements for Localized Corrosion Susceptibility of Iron-, Nickel-, or Cobalt-Based Alloys". This standard covers: SIGNIFICANCE AND USE 3.1 An indication of the susceptibility to initiation of localized corrosion in this test method is given by the potential at which the anodic current increases rapidly. The more noble this potential, obtained at a fixed scan rate in this test, the less susceptible is the alloy to initiation of localized corrosion. The results of this test are not intended to correlate in a quantitative manner with the rate of propagation that one might observe in service when localized corrosion occurs. 3.2 In general, once initiated, localized corrosion can propagate at some potential more electropositive than that at which the hysteresis loop is completed. In this test method, the potential at which the hysteresis loop is completed is determined at a fixed scan rate. In these cases, the more electropositive the potential at which the hysteresis loop is completed the less likely it is that localized corrosion will occur. 3.3 If followed, this test method will provide cyclic potentiodynamic anodic polarization measurements that will reproduce data developed at other times in other laboratories using this test method for the two specified alloys discussed in 3.4. The procedure is used for iron-, nickel-, or cobalt-based alloys in a chloride environment. 3.4 A standard potentiodynamic polarization plot is included. These reference data are based on the results from five different laboratories that followed the standard procedure, using specific alloys of Type 304 stainless steel, UNS S30400 and Alloy C-276, UNS N10276.3 Curves are included which have been constructed using statistical analysis to indicate the acceptable range of polarization curves. 3.5 The availability of a standard test method, standard material, and standard plots should make it easy for an investigator to check his techniques to evaluate susceptibility to localized corrosion. SCOPE 1.1 This test method covers a procedure for conducting cyclic potentiodynamic polarization measurements to determine relative susceptibility to localized corrosion (pitting and crevice corrosion) for iron-, nickel-, or cobalt-based alloys in a chloride environment. This test method also describes an experimental procedure which can be used to check one's experimental technique and instrumentation. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 An indication of the susceptibility to initiation of localized corrosion in this test method is given by the potential at which the anodic current increases rapidly. The more noble this potential, obtained at a fixed scan rate in this test, the less susceptible is the alloy to initiation of localized corrosion. The results of this test are not intended to correlate in a quantitative manner with the rate of propagation that one might observe in service when localized corrosion occurs. 3.2 In general, once initiated, localized corrosion can propagate at some potential more electropositive than that at which the hysteresis loop is completed. In this test method, the potential at which the hysteresis loop is completed is determined at a fixed scan rate. In these cases, the more electropositive the potential at which the hysteresis loop is completed the less likely it is that localized corrosion will occur. 3.3 If followed, this test method will provide cyclic potentiodynamic anodic polarization measurements that will reproduce data developed at other times in other laboratories using this test method for the two specified alloys discussed in 3.4. The procedure is used for iron-, nickel-, or cobalt-based alloys in a chloride environment. 3.4 A standard potentiodynamic polarization plot is included. These reference data are based on the results from five different laboratories that followed the standard procedure, using specific alloys of Type 304 stainless steel, UNS S30400 and Alloy C-276, UNS N10276.3 Curves are included which have been constructed using statistical analysis to indicate the acceptable range of polarization curves. 3.5 The availability of a standard test method, standard material, and standard plots should make it easy for an investigator to check his techniques to evaluate susceptibility to localized corrosion. SCOPE 1.1 This test method covers a procedure for conducting cyclic potentiodynamic polarization measurements to determine relative susceptibility to localized corrosion (pitting and crevice corrosion) for iron-, nickel-, or cobalt-based alloys in a chloride environment. This test method also describes an experimental procedure which can be used to check one's experimental technique and instrumentation. 1.2 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM G61-86(2018) is classified under the following ICS (International Classification for Standards) categories: 77.060 - Corrosion of metals. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM G61-86(2018) has the following relationships with other standards: It is inter standard links to ASTM G61-86(2014), ASTM G3-14(2019), ASTM G3-14, ASTM G5-14, ASTM G3-13, ASTM G5-13e1, ASTM G5-13, ASTM G5-13e2, ASTM G5-12, ASTM G5-94(2011)e1, ASTM G3-89(2010), ASTM D1193-06, ASTM G5-94(2004), ASTM G3-89(2004), ASTM G3-89(1999). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM G61-86(2018) is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: G61 − 86 (Reapproved 2018)
Standard Test Method for
Conducting Cyclic Potentiodynamic Polarization
Measurements for Localized Corrosion Susceptibility of
Iron-, Nickel-, or Cobalt-Based Alloys
ThisstandardisissuedunderthefixeddesignationG61;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoptionor,inthecaseofrevision,theyearoflastrevision.Anumberinparenthesesindicatestheyearoflastreapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 2.2 ASTM Adjuncts:
Standard Samples (set of two)
1.1 This test method covers a procedure for conducting
cyclic potentiodynamic polarization measurements to deter-
3. Significance and Use
mine relative susceptibility to localized corrosion (pitting and
3.1 An indication of the susceptibility to initiation of local-
crevicecorrosion)foriron-,nickel-,orcobalt-basedalloysina
chloride environment. This test method also describes an ized corrosion in this test method is given by the potential at
whichtheanodiccurrentincreasesrapidly.Themorenoblethis
experimental procedure which can be used to check one’s
experimental technique and instrumentation. potential, obtained at a fixed scan rate in this test, the less
susceptible is the alloy to initiation of localized corrosion.The
1.2 The values stated in SI units are to be regarded as
resultsofthistestarenotintendedtocorrelateinaquantitative
standard. No other units of measurement are included in this
manner with the rate of propagation that one might observe in
standard.
service when localized corrosion occurs.
1.3 This standard does not purport to address all of the
3.2 Ingeneral,onceinitiated,localizedcorrosioncanpropa-
safety concerns, if any, associated with its use. It is the
gate at some potential more electropositive than that at which
responsibility of the user of this standard to establish appro-
the hysteresis loop is completed. In this test method, the
priate safety, health, and environmental practices and deter-
potential at which the hysteresis loop is completed is deter-
mine the applicability of regulatory limitations prior to use.
mined at a fixed scan rate. In these cases, the more electro-
1.4 This international standard was developed in accor-
positive the potential at which the hysteresis loop is completed
dance with internationally recognized principles on standard-
the less likely it is that localized corrosion will occur.
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom- 3.3 If followed, this test method will provide cyclic poten-
mendations issued by the World Trade Organization Technical
tiodynamic anodic polarization measurements that will repro-
Barriers to Trade (TBT) Committee. duce data developed at other times in other laboratories using
this test method for the two specified alloys discussed in 3.4.
2. Referenced Documents The procedure is used for iron-, nickel-, or cobalt-based alloys
in a chloride environment.
2.1 ASTM Standards:
D1193Specification for Reagent Water 3.4 A standard potentiodynamic polarization plot is in-
G3Practice for Conventions Applicable to Electrochemical
cluded.These reference data are based on the results from five
Measurements in Corrosion Testing different laboratories that followed the standard procedure,
G5Reference Test Method for Making Potentiodynamic
using specific alloys of Type 304 stainless steel, UNS S30400
Anodic Polarization Measurements
and Alloy C-276, UNS N10276. Curves are included which
have been constructed using statistical analysis to indicate the
acceptable range of polarization curves.
This test method is under the jurisdiction of ASTM Committee G01 on
3.5 The availability of a standard test method, standard
Corrosion of Metals and is the direct responsibility of Subcommittee G01.11 on
material, and standard plots should make it easy for an
Electrochemical Measurements in Corrosion Testing.
investigator to check his techniques to evaluate susceptibility
Current edition approved May 1, 2018. Published June 2018. Originally
approvedin1986.Lastpreviouseditionapprovedin2014asG61–86(2014).DOI:
to localized corrosion.
10.1520/G0061-86R18.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available from ASTM International Headquarters. Order Adjunct No.
the ASTM website. ADJG0061. Original adjunct produced before 1995.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G61 − 86 (2018)
TABLE 1 Chemical Composition of Alloys Used in the Round
Robin, Weight %
Type 304
Alloy C-276
Element Stainless Steel
(UNS N10276)
(UNS S30400)
Carbon 0.003 0.060
Chromium 15.29 18.46
Cobalt 2.05 .
Columbium . 0.11
Copper . 0.17
Iron 5.78 balance
Manganese 0.48 1.43
Molybdenum 16.03 0.17
Nickel balance 8.74
Phosphorus 0.018 0.029
Silicon 0.05 0.60
Sulfur 0.006 0.014
Vanadium 0.20 .
Tungsten 3.62 .
4.4 Potential-Measuring Instruments (Note 1)—The
potential-measuring circuit should have a high input imped-
11 14
ance on the order of 10 to 10 Ω to minimize current drawn
from the system during measurements. Instruments should
have sufficient sensitivity and accuracy to detect a change in
FIG. 1 Schematic Diagram of Specimen Holder (see Footnotes 3
potential of 61 mV, usually included in commercial poten-
and 4)
tiostats. An output as a voltage is preferred for recording
purposes.
4. Apparatus
4.5 Current-Measuring Instruments (Note 1)—An instru-
mentthatiscapableofmeasuringacurrentaccuratelytowithin
4.1 The polarization cell should be similar to the one
1%oftheabsolutevalueoveracurrentrangebetween1.0and
described in Reference Test Method G5. Other polarization
10 µAshouldbeused.Manycommercialunitshaveabuild-in
cells may be equally suitable.
instrument with an output as a voltage, which is preferred for
4.1.1 The cell should have a capacity of about 1 L and
recording purposes. For the purpose of the present test a
should have suitable necks or seals to permit the introduction
logarithmic output is desirable.
of electrodes, gas inlet and outlet tubes, and a thermometer.
The Luggin probe-salt bridge separates the bulk solution from
4.6 Anodic Polarization Circuit—Ascanning potentiostat is
thesaturatedcalomelreferenceelectrode.Theprobetipshould
used for potentiodynamic measurements. Potential and current
beadjustablesothatitcanbebroughtintocloseproximitywith
are plotted continuously using an X-Y recorder and a logarith-
the working electrode.
mic converter (contained in the potentiostat or incorporated
into the circuit) for the current. Commercially available units
4.2 Specimen Holder:
are suitable.
4.2.1 Specimens should be mounted in a suitable holder
designed for flat strip, exposing 1 cm to the test solution (Fig.
4.7 Electrodes:
1). Such specimen holders have been described in the litera-
4.7.1 The standard Type 304 stainless steel (UNS S30400)
ture. It is important that the circular TFE-fluorocarbon gasket
andAlloy C-276 (UNS N10276) should be machined into flat
be drilled and machined flat in order to minimize crevices.
0.625-in. (14-mm) diameter disks. The chemical compositions
of the alloys used in the round robin are listed in Table 1.
4.3 Potentiostat (Note 1)—Apotentiostat that will maintain
4.7.2 Counter Electrodes—The counter electrodes may be
an electrode potential within 1 mV of a preset value over a
prepared as described in ReferenceTest Method G5 or may be
widerangeofappliedcurrentsshouldbeused.Forthetypeand
prepared from high-purity platinum flat stock and wire. A
size of standard specimen supplied, the potentiostat should
suitable method would be to seal the platinum wire in glass
have a potential range of−1.0 to+1.6 V and an anodic current
tubing and introduce the platinum electrode assembly through
output range of 1.0 to 10 µA. Most commercial potentiostats
a sliding seal. Counter electrodes should have an area at least
meet the specific requirements for these types of measure-
twice as large as the test electrode.
ments.
4.7.3 Reference Electrode —A saturated calomel electrode
NOTE 1—These instrumental requirements are based upon values
with a controlled rate of leakage (about 3 µL/h) is recom-
typical of the instruments in the five laboratories that have provided the
mended. This type of electrode is durable, reliable, and
data used in determining the standard
...



Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...