ASTM F3295-18
(Guide)Standard Guide for Impingement Testing of Total Disc Prostheses
Standard Guide for Impingement Testing of Total Disc Prostheses
SIGNIFICANCE AND USE
5.1 This guide can be used to develop test parameters for evaluating fatigue and wear behavior of IVD prostheses under impingement loading. It must be recognized, however, that there are likely many possible impingement conditions for a given IVD prosthesis.
5.2 The user should attempt to determine the clinically relevant and geometrically possible impingement conditions and dictated by the design and impingement wear test parameters that may result in wear and fatigue damage for the IVD prosthesis. The user should also attempt to select the device size which will represent a worst case for the impingement conditions and parameters selected.
5.3 The user should reference and utilize existing sources of information to identify the impingement test parameters that produce the clinically relevant impingement wear and damage for their IVD prosthesis. Prior clinical experience with the device design may aid in the development of impingement test parameters through analysis of device retrievals and radiographs. However, prior clinical experience with the IVD being tested should not be considered as a prerequisite for performing impingement testing.
5.4 This guide details a three-step process for assessing device impingement under a selected set of conditions:
5.4.1 The user selects previously identified impingement conditions, one at a time, or clinically observed conditions.
5.4.2 The user selects the worst-case size of device to apply the selected conditions.
5.4.3 Solid modeling and the quasistatic test method should be employed to assess the impingement condition and determine the impingement test parameters – most importantly, the angular displacement limits to be used in the impingement wear test.
5.4.4 The impingement wear test is then conducted using the impingement test parameters.
5.5 This guide serves to evaluate devices with various designs, materials (i.e., metal-on-metal versus polymer-on-polymer), and stiffness in the impingement reg...
SCOPE
1.1 This standard is intended to provide guidance on the evaluation of wear and fatigue characteristics of total disc prostheses under cyclic impingement conditions.
1.2 This guide describes impingement testing of devices with articulating components. The user is cautioned that the methods described herein are intended to produce an impingement condition which may or may not be indicative of clinical performance and which may or may not be consistent with the intended use of the device, and that this should be considered when interpreting the data. Clinically, total disc prostheses should always be implanted per labeling and the manufacturer’s instructions for use.
1.3 Impingement has been observed in retrievals among several total disc prosthesis designs; however, impingement is not necessarily associated with device or clinical failure. It is the intent of this guide to investigate possible impingement-induced wear and mechanical failure modes associated with device design, as well as potential mechanical failure modes associated with clinical events such as subsidence, malpositioning, and improper implant sizing. Note that mechanical failure may or may not be associated with functional failure.
1.4 It is recommended that the user define the bearing and non-bearing features of the intervertebral disc (IVD) prosthesis and evaluate the performance of the IVD prosthesis under Mode 1 wear by using Guide F2423 or ISO 18192-1 prior to use of this guide. This standard is not intended to provide guidance on Mode I testing.
1.5 The goal of this guide is to evaluate impingement in IVD prostheses regardless of the intended region of the spine (cervical or lumbar), material or material combinations (ceramic, metal, polymer), and bearing type (fixed or mobile).
1.6 It is the intent of this guide to enable comparison of IVD prostheses with regard to wear and fatigue characteristics when tested under the specified cond...
General Information
- Status
- Published
- Publication Date
- 30-Jun-2018
- Technical Committee
- F04 - Medical and Surgical Materials and Devices
- Drafting Committee
- F04.25 - Spinal Devices
Relations
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Nov-2023
- Effective Date
- 01-Jan-2019
- Effective Date
- 01-Nov-2018
- Effective Date
- 01-Apr-2018
- Effective Date
- 01-Oct-2016
- Effective Date
- 01-Jun-2014
- Effective Date
- 01-Sep-2013
- Effective Date
- 01-Aug-2013
- Effective Date
- 15-Mar-2013
- Effective Date
- 01-Aug-2012
- Effective Date
- 01-Aug-2012
- Effective Date
- 01-Jul-2011
- Effective Date
- 01-Sep-2010
- Effective Date
- 01-Jun-2010
Overview
ASTM F3295-18: Standard Guide for Impingement Testing of Total Disc Prostheses provides a comprehensive framework for evaluating the wear and fatigue characteristics of total disc (intervertebral disc, IVD) prostheses under cyclic impingement conditions. Developed by ASTM International, this guide is a crucial resource for medical device manufacturers, researchers, and regulatory bodies aiming to assess the mechanical durability and performance risks of total disc replacements in the spine. By establishing recommended practices for impingement testing, the standard enhances consistency, reliability, and clinical relevance in orthopedic device evaluation.
Key Topics
- Purpose of Impingement Testing: The standard outlines methods for simulating and measuring fatigue and wear in IVD prostheses when subjected to impingement loading conditions that may occur in clinical scenarios such as malpositioning or improper implant sizing.
- Three-Step Testing Process: Defines a structured methodology:
- Identify clinically relevant impingement conditions based on device design, retrieval analyses, radiographs, or scientific literature.
- Select the worst-case device size and configuration to represent the most severe potential impingement scenario.
- Use solid modeling and quasistatic test methods to set impingement test parameters, focusing on angular displacement limits key to simulating wear and fatigue damage.
- Design and Device Considerations: Provides guidelines for evaluating devices with a variety of materials (metal, polymer, ceramic) and different bearing types (fixed or mobile), and applies to both cervical and lumbar total disc prostheses.
- Test Execution and Data Outputs: Details the procedures for quasistatic and wear testing, recommended sample sizes, and required use of non-biologic production-grade implants. Specifies outputs such as mass loss, volumetric loss, and dimensional change for comparative analysis.
- Data Interpretation: Underlines the importance of understanding that bench testing results may not always directly represent clinical performance, emphasizing the need for careful data interpretation in the context of intended device use.
Applications
- Medical Device Development: Enables manufacturers to design and refine IVD prostheses by providing a standardized approach to assess their resistance to wear and mechanical failure modes associated with impingement.
- Preclinical Regulatory Submission: Supports regulatory submissions by providing repeatable and clinically relevant wear and fatigue data under defined impingement conditions.
- Comparative Device Evaluation: Facilitates comparison of different prosthesis designs, materials, and configurations for performance under worst-case impingement scenarios, promoting device safety and efficacy.
- Post-Market Surveillance and Improvement: Helps analyze retrieval data and clinical outcomes to inform ongoing product improvements and risk assessments.
Related Standards
- ASTM F2423: Guide for Functional, Kinematic, and Wear Assessment of Total Disc Prostheses.
- ISO 18192-1: Implants for surgery-Wear of total intervertebral spinal disc prostheses-Loading and displacement parameters.
- ASTM F561: Practice for Retrieval and Analysis of Medical Devices, and Associated Tissues and Fluids.
- ASTM F1714: Guide for Gravimetric Wear Assessment of Prosthetic Hip Designs.
- ASTM E4: Practices for Force Verification of Testing Machines.
- ASTM E1488: Guide for Statistical Procedures to Use in Developing and Applying Test Methods.
By adhering to ASTM F3295-18, stakeholders in spinal implant development can optimize testing strategies to demonstrate device durability, benchmark new designs, and ultimately improve patient safety and clinical outcomes for total disc prostheses.
Buy Documents
ASTM F3295-18 - Standard Guide for Impingement Testing of Total Disc Prostheses
Get Certified
Connect with accredited certification bodies for this standard

BSI Group
BSI (British Standards Institution) is the business standards company that helps organizations make excellence a habit.

TÜV Rheinland
TÜV Rheinland is a leading international provider of technical services.

TÜV SÜD
TÜV SÜD is a trusted partner of choice for safety, security and sustainability solutions.
Sponsored listings
Frequently Asked Questions
ASTM F3295-18 is a guide published by ASTM International. Its full title is "Standard Guide for Impingement Testing of Total Disc Prostheses". This standard covers: SIGNIFICANCE AND USE 5.1 This guide can be used to develop test parameters for evaluating fatigue and wear behavior of IVD prostheses under impingement loading. It must be recognized, however, that there are likely many possible impingement conditions for a given IVD prosthesis. 5.2 The user should attempt to determine the clinically relevant and geometrically possible impingement conditions and dictated by the design and impingement wear test parameters that may result in wear and fatigue damage for the IVD prosthesis. The user should also attempt to select the device size which will represent a worst case for the impingement conditions and parameters selected. 5.3 The user should reference and utilize existing sources of information to identify the impingement test parameters that produce the clinically relevant impingement wear and damage for their IVD prosthesis. Prior clinical experience with the device design may aid in the development of impingement test parameters through analysis of device retrievals and radiographs. However, prior clinical experience with the IVD being tested should not be considered as a prerequisite for performing impingement testing. 5.4 This guide details a three-step process for assessing device impingement under a selected set of conditions: 5.4.1 The user selects previously identified impingement conditions, one at a time, or clinically observed conditions. 5.4.2 The user selects the worst-case size of device to apply the selected conditions. 5.4.3 Solid modeling and the quasistatic test method should be employed to assess the impingement condition and determine the impingement test parameters – most importantly, the angular displacement limits to be used in the impingement wear test. 5.4.4 The impingement wear test is then conducted using the impingement test parameters. 5.5 This guide serves to evaluate devices with various designs, materials (i.e., metal-on-metal versus polymer-on-polymer), and stiffness in the impingement reg... SCOPE 1.1 This standard is intended to provide guidance on the evaluation of wear and fatigue characteristics of total disc prostheses under cyclic impingement conditions. 1.2 This guide describes impingement testing of devices with articulating components. The user is cautioned that the methods described herein are intended to produce an impingement condition which may or may not be indicative of clinical performance and which may or may not be consistent with the intended use of the device, and that this should be considered when interpreting the data. Clinically, total disc prostheses should always be implanted per labeling and the manufacturer’s instructions for use. 1.3 Impingement has been observed in retrievals among several total disc prosthesis designs; however, impingement is not necessarily associated with device or clinical failure. It is the intent of this guide to investigate possible impingement-induced wear and mechanical failure modes associated with device design, as well as potential mechanical failure modes associated with clinical events such as subsidence, malpositioning, and improper implant sizing. Note that mechanical failure may or may not be associated with functional failure. 1.4 It is recommended that the user define the bearing and non-bearing features of the intervertebral disc (IVD) prosthesis and evaluate the performance of the IVD prosthesis under Mode 1 wear by using Guide F2423 or ISO 18192-1 prior to use of this guide. This standard is not intended to provide guidance on Mode I testing. 1.5 The goal of this guide is to evaluate impingement in IVD prostheses regardless of the intended region of the spine (cervical or lumbar), material or material combinations (ceramic, metal, polymer), and bearing type (fixed or mobile). 1.6 It is the intent of this guide to enable comparison of IVD prostheses with regard to wear and fatigue characteristics when tested under the specified cond...
SIGNIFICANCE AND USE 5.1 This guide can be used to develop test parameters for evaluating fatigue and wear behavior of IVD prostheses under impingement loading. It must be recognized, however, that there are likely many possible impingement conditions for a given IVD prosthesis. 5.2 The user should attempt to determine the clinically relevant and geometrically possible impingement conditions and dictated by the design and impingement wear test parameters that may result in wear and fatigue damage for the IVD prosthesis. The user should also attempt to select the device size which will represent a worst case for the impingement conditions and parameters selected. 5.3 The user should reference and utilize existing sources of information to identify the impingement test parameters that produce the clinically relevant impingement wear and damage for their IVD prosthesis. Prior clinical experience with the device design may aid in the development of impingement test parameters through analysis of device retrievals and radiographs. However, prior clinical experience with the IVD being tested should not be considered as a prerequisite for performing impingement testing. 5.4 This guide details a three-step process for assessing device impingement under a selected set of conditions: 5.4.1 The user selects previously identified impingement conditions, one at a time, or clinically observed conditions. 5.4.2 The user selects the worst-case size of device to apply the selected conditions. 5.4.3 Solid modeling and the quasistatic test method should be employed to assess the impingement condition and determine the impingement test parameters – most importantly, the angular displacement limits to be used in the impingement wear test. 5.4.4 The impingement wear test is then conducted using the impingement test parameters. 5.5 This guide serves to evaluate devices with various designs, materials (i.e., metal-on-metal versus polymer-on-polymer), and stiffness in the impingement reg... SCOPE 1.1 This standard is intended to provide guidance on the evaluation of wear and fatigue characteristics of total disc prostheses under cyclic impingement conditions. 1.2 This guide describes impingement testing of devices with articulating components. The user is cautioned that the methods described herein are intended to produce an impingement condition which may or may not be indicative of clinical performance and which may or may not be consistent with the intended use of the device, and that this should be considered when interpreting the data. Clinically, total disc prostheses should always be implanted per labeling and the manufacturer’s instructions for use. 1.3 Impingement has been observed in retrievals among several total disc prosthesis designs; however, impingement is not necessarily associated with device or clinical failure. It is the intent of this guide to investigate possible impingement-induced wear and mechanical failure modes associated with device design, as well as potential mechanical failure modes associated with clinical events such as subsidence, malpositioning, and improper implant sizing. Note that mechanical failure may or may not be associated with functional failure. 1.4 It is recommended that the user define the bearing and non-bearing features of the intervertebral disc (IVD) prosthesis and evaluate the performance of the IVD prosthesis under Mode 1 wear by using Guide F2423 or ISO 18192-1 prior to use of this guide. This standard is not intended to provide guidance on Mode I testing. 1.5 The goal of this guide is to evaluate impingement in IVD prostheses regardless of the intended region of the spine (cervical or lumbar), material or material combinations (ceramic, metal, polymer), and bearing type (fixed or mobile). 1.6 It is the intent of this guide to enable comparison of IVD prostheses with regard to wear and fatigue characteristics when tested under the specified cond...
ASTM F3295-18 is classified under the following ICS (International Classification for Standards) categories: 11.180.10 - Aids and adaptation for moving. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F3295-18 has the following relationships with other standards: It is inter standard links to ASTM E1488-23, ASTM E1402-13(2023), ASTM F561-19, ASTM E1402-13(2018), ASTM F1714-96(2018), ASTM F1877-16, ASTM E4-14, ASTM F561-13, ASTM E1402-13, ASTM F1714-96(2013), ASTM E1488-12e1, ASTM E1488-12, ASTM F2423-11, ASTM F561-05a(2010), ASTM F1877-05(2010). Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F3295-18 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F3295 − 18
Standard Guide for
Impingement Testing of Total Disc Prostheses
This standard is issued under the fixed designation F3295; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope 1.7 The values stated in SI units are to be regarded as the
standard with the exception of angular measurements which
1.1 This standard is intended to provide guidance on the
should be reported in degrees.
evaluation of wear and fatigue characteristics of total disc
1.8 The use of this standard may involve the operation of
prostheses under cyclic impingement conditions.
potentially hazardous equipment. This standard does not pur-
1.2 This guide describes impingement testing of devices
port to address all of the safety concerns, if any, associated
with articulating components. The user is cautioned that the
with its use. It is the responsibility of the user of this standard
methods described herein are intended to produce an impinge-
to establish appropriate safety, health, and environmental
ment condition which may or may not be indicative of clinical
practices and determine the applicability of regulatory limita-
performance and which may or may not be consistent with the
tions prior to use.
intended use of the device, and that this should be considered
1.9 This international standard was developed in accor-
when interpreting the data. Clinically, total disc prostheses
dance with internationally recognized principles on standard-
should always be implanted per labeling and the manufactur-
ization established in the Decision on Principles for the
er’s instructions for use.
Development of International Standards, Guides and Recom-
1.3 Impingement has been observed in retrievals among
mendations issued by the World Trade Organization Technical
several total disc prosthesis designs; however, impingement is
Barriers to Trade (TBT) Committee.
not necessarily associated with device or clinical failure. It is
2. Referenced Documents
the intent of this guide to investigate possible impingement-
induced wear and mechanical failure modes associated with
2.1 ASTM Standards:
device design, as well as potential mechanical failure modes
E4Practices for Force Verification of Testing Machines
associated with clinical events such as subsidence,
E1402Guide for Sampling Design
malpositioning, and improper implant sizing. Note that me-
E1488GuideforStatisticalProcedurestoUseinDeveloping
chanical failure may or may not be associated with functional
and Applying Test Methods
failure.
F561 Practice for Retrieval and Analysis of Medical
Devices, and Associated Tissues and Fluids
1.4 It is recommended that the user define the bearing and
F1714GuideforGravimetricWearAssessmentofProsthetic
non-bearingfeaturesoftheintervertebraldisc(IVD)prosthesis
Hip Designs in Simulator Devices
and evaluate the performance of the IVD prosthesis under
F1877Practice for Characterization of Particles
Mode 1 wear by using Guide F2423 or ISO 18192-1 prior to
F2423Guide for Functional, Kinematic, and Wear Assess-
use of this guide. This standard is not intended to provide
ment of Total Disc Prostheses
guidance on Mode I testing.
2.2 ISO Standard:
1.5 The goal of this guide is to evaluate impingement in
ISO 18192–1Implants for surgery—Wear of total interver-
IVD prostheses regardless of the intended region of the spine
tebral spinal disc prostheses—Part 1: Loading and dis-
(cervical or lumbar), material or material combinations
placement parameters for wear testing and corresponding
(ceramic, metal, polymer), and bearing type (fixed or mobile).
environmental
1.6 ItistheintentofthisguidetoenablecomparisonofIVD
3. Terminology
prostheseswithregardtowearandfatiguecharacteristicswhen
tested under the specified conditions.
3.1 Definitions of Terms Specific to This Standard:
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
This guide is under the jurisdiction ofASTM Committee F04 on Medical and contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Surgical Materials and Devices and is the direct responsibility of Subcommittee Standards volume information, refer to the standard’s Document Summary page on
F04.25 on Spinal Devices. the ASTM website.
Current edition approved July 1, 2018. Published August 2018. DOI: 10.1520/ Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
F3295–18. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F3295 − 18
3.1.1 axial force, n—theresultantforce F appliedto system,definedbytheuser.Deviceneutralpositionisoftenthe
axial force
the IVD prosthesis along the Z-axis that simulates the in vivo position when the device endplates are parallel to one another.
axial force. Based on a healthy disc, the primary component
3.1.3 device range of motion (ROM), n—the maximum
wouldbeanaxialcompressiveforce, F ,inthedirectionofthe
Z
amount of angular displacement that an IVD prosthesis can
negative global Z-axis, and it would pass through the center of
undergo from the device neutral position to the point at which
rotation of the IVD prosthesis.
initial impingement occurs around a defined global axis. For
3.1.2 coordinate system/axes, n—global XYZ orthogonal example, if a device impinges at 15° from the device neutral
axesaredefinedfollowingaright-handedCartesiancoordinate position in flexion and 20° from the device neutral position in
system in which the XY plane is to bisect the sagittal plane extension, the device range of motion can be defined as
angle between superior and inferior surfaces that are intended +15°/-20° in flexion-extension.
to simulate the adjacent vertebral end plates. The global axes
3.1.4 functional failure, n—permanent deformation or wear
arestationaryrelativetotheIVDprostheses’sinferiorendplate
that renders the IVD prosthesis assembly ineffective or unable
fixture, which in this standard guide is also considered to be
to resist force/motion or any secondary effects that result in a
stationarywithrespecttothetestmachine’sframe.Lowercase
substantial alteration of clinically relevant motions or the
letters, xyz, denote a local, moving orthogonal coordinate
motions intended by the design of the device.
system attached to the superior end plate-fixturing with direc-
3.1.5 impingement, n—contact between two components,
tions initially coincident with those of the global XYZ axes,
resulting in a restriction of motion (Fig. 1).
respectively.The 3D motion of the superior relative to inferior
3.1.5.1 impingement conditions, n—the angles determined
end plate-fixture is specified and is to be measured in terms of
to produce impingement in the device in flexion-extension,
sequential Eulerian angular rotations about the xyz axes,
lateral bending and axial rotation.
respectively (z, axial rotation; x, lateral bending; and y,
flexion-extension).
3.1.5.2 impingement test parameters, n—the test inputs for
rotations and forces which create the intended impingement
3.1.2.1 origin, n—center of the global coordinate system,
conditions and are used for impingement testing.
located at the initial position of the IVD’s instantaneous center
of rotation (COR). Note that some articulating devices do not
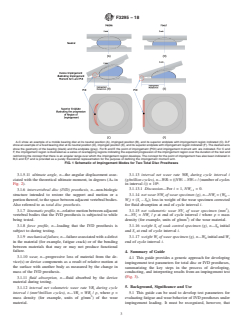
3.1.5.3 initial impingement angle (A in Fig. 2),n—the
haveafixedcenterofrotation,butinsteadhaveeitheramobile
angular displacement in a given plane, with respect to the
center of rotation or multiple distinct centers of rotation,
device neutral position, at which impingement initially occurs,
dependingonthedirectionofmovement.Inthiscasetheorigin
usually indicated by a sharp change in moment.
shouldbeexplicitlydefinedbytheuserwitharationaleforthat
3.1.5.4 impingement moment, n—the moment (N-m) mea-
determination.
sured or applied at the point of impingement (POI). It may be
3.1.2.2 X-axis, n—positive X-axis is a global fixed axis
determined as the product of the applied axial force and
relative to the testing machine’s stationary base and is to be
impingement moment arm.
directed anteriorly relative to the specimen’s initial unloaded
3.1.5.5 maximum impingement angular displacement,
position.
n—the greater of the two angular displacement test parameters
3.1.2.3 Y-axis, n—positive Y-axis is a global fixed axis
(farthest from the device neutral position); it is the ultimate
relative to the testing machine’s stationary base and is directed
angle plus 2.0°.
laterally relative to the specimen’s initial unloaded position.
3.1.5.6 minimum impingement angular displacement,
3.1.2.4 Z-axis, n—positive Z-axis is a global fixed axis
n—the lesser of the two angular displacement test parameters
relative to the testing machine’s stationary base and is to be
(that closer to the device neutral position). It is 2.0° less than
directed superiorly relative to the specimen’s initial unloaded
the initial impingement angle.
position.
3.1.5.7 impingement moment arm, mm(Fig.1bandFig.1e),
3.1.2.5 x-axis, n—positive x-axis is a fixed axis relative to
n—thedistanceinthe x-yplanefromthe z-axisofthedeviceto
the IVD prosthesis and a moving axis relative to the global
the POI.
coordinate system and is directed anteriorly relative to the
3.1.5.8 impingement region (Fig. 1C and Fig. 1F),
prosthesis.
n—physicalareaonthedevicecomponentswheretheimpinge-
3.1.2.6 y-axis, n—positive y-axis is a fixed axis relative to
ment wear scar develops as a result of repeated loading and
the IVD prosthesis and a moving axis relative to the global
motion cycles.
coordinate system and is directed laterally relative to the
3.1.5.9 point of impingement (POI) (Fig. 1C and Fig. 1F),
prosthesis.
n—the theoretical location on the IVD prosthesis’s x-y plane
3.1.2.7 z-axis, n—positive z-axis is a fixed axis relative to where impingement occurs with respect to the origin.
the IVD prosthesis and a moving axis relative to the global
3.1.5.10 theoretical ultimate moment (M in Fig. 2),n—the
t
coordinate system and is directed superiorly relative to the
mathematicalproductoftheaxialforcetobeappliedduringthe
prosthesis.
impingement wear test (Table 1) and the distance in the x-y
3.1.2.8 device neutral position, n—the device position plane from the z-axis of the device to the POI (mm); for
where the user considers the local xyz coordinate system example,foracervicalIVDprosthesiswithaPOI9.0mmfrom
initially parallel to those of the global XYZ axes coordinate the z-axis, M = (100 N) (9.0 mm) / (1000) = 0.9 Nm.
t
F3295 − 18
A-C show an example of a mobile bearing disc at its neutral position (A), impinged position (B), and its superior endplate with impingement region indicated (C). D-F
show an example of a fixed bearing disc at its neutral position (D), impinged position (E), and its superior endplate with impingement region indicated (F).The dashed arcs
show the geometry of the bearing (black) and the endplate (gray). For B and E the point of impingement (POI) and impingement moment arm are indicated. For C and
F, the impingement region is illustrated as a series of overlapping regions indicating the expected progression of the impingement region over the duration of the test and
reinforcing the concept that there is an angular range over which the impingement region develops. The concept for the point of impingement has also been indicated in
B,C and E,F and is provided as a purely theoretical representation for the purpose of defining the impingement moment arm.
FIG. 1 Schematic of Impingement Modes for Two Total Disc Prostheses
3.1.5.11 ultimate angle, n—the angular displacement asso- 3.1.13 interval net wear rate WR during cycle interval i
i
ciated with the theoretical ultimate moment, in degrees (A in (g/million cycles), n—WR =((NW – NW /(numberofcycles
u i i i-1
Fig. 2). in interval i))×10 .
3.1.13.1 Discussion—For i=1, NW =0.
i-1
3.1.6 intervertebral disc (IVD) prosthesis, n—non-biologic
structure intended to restore the support and motion or a 3.1.14 net wear NW of wear specimen (g), n—NW =(W –
i i 0
portionthereof,tothespacebetweenadjacentvertebralbodies. W)+(S –S ); loss in weight of the wear specimen corrected
i i 0
Also referred to as total disc prosthesis. for fluid absorption at end of cycle interval i.
3.1.7 kinematicprofile,n—relativemotionbetweenadjacent 3.1.15 net volumetric wear NV of wear specimen (mm ),
i
vertebral bodies that the IVD prosthesis is subjected to while n—NV = NW / ρ at end of cycle interval i where ρ = mass
i i
being tested. density (for example, units of g/mm ) of the wear material.
3.1.8 force profile, n—loading that the IVD prosthesis is 3.1.16 weight S of soak control specimen (g), n—S initial
i 0
subject to during testing. and S at end of cycle interval i.
i
3.1.9 mechanical failure, n—failureassociatedwithadefect 3.1.17 weight W of wear specimen (g), n—W initialand W
i 0 i
in the material (for example, fatigue crack) or of the bonding end of cycle interval i.
between materials that may or may not produce functional
failure.
4. Summary of Guide
3.1.10 wear, n—progressive loss of material from the de-
4.1 This guide provides a generic approach for developing
vice(s) or device components as a result of relative motion at
impingement test parameters for total disc or IVD prostheses,
the surface with another body as measured by the change in
summarizing the key steps in the process of developing,
mass of the IVD prosthesis.
conducting, and interpreting results from an impingement test
(Fig. 3).
3.1.11 fluid absorption, n—fluid absorbed by the device
material during testing.
5. Background, Significance and Use
3.1.12 interval net volumetric wear rate VR during cycle
i
interval i (mm /million cycles), n—VR = WR / ρ, where ρ = 5.1 This guide can be used to develop test parameters for
i i
mass density (for example, units of g/mm ) of the wear evaluating fatigue and wear behavior of IVD prostheses under
material. impingement loading. It must be recognized, however, that
F3295 − 18
In this plot, the horizontal blue line represents constant, low frictional torque expected as the device is rotated through its range of motion. The sloping dashed line OA
showsincreasingtorquethatmaybeanindicatorofCORmisalignment.PointArepresentstheinitialimpingementangle(A).Thearcbetweentheinitialimpingementangle
(A) and the ultimate angle (A ) and theoretical ultimate moment (M) illustrates the sharp upward torque experienced during impingement. Beyond the ultimate angle (A )
u t u
and theoretical ultimate moment (M) the curves for Quasistatic Test Method 1 (see 10.1) and Quasistatic Test Method 2 (see 10.2) diverge.
t
FIG. 2 Impingement Moment versus Angular Displacement Plot Showing Two Theoretical Curves for Impingement Under
Quasistatic Conditions
A
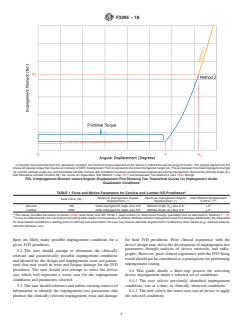
TABLE 1 Force and Motion Parameters for Cervical and Lumbar IVD Prostheses
Minimum Impingement Angular Maximum Impingement Angular Axial Rotation Displacement
Axial Force, (N)
B
Displacement, (°) Displacement, (°) Control, (°)
Cervical 100 Initial impingement angle, less 2.0° Ultimate Angle (A ) plus 2.0° ± 6°
u
Lumbar 1200 Initial impingement angle, less 2.0° Ultimate Angle (A ) plus 2.0° ± 2°
u
A
The values provided are based on Guide F2423 (axial force) and ISO 18192-1 (axial rotation) or determined through quasistatic test as described in Sections7–10.
B
Itmaybedeterminedbytheuserthatincorporatingaxialrotationisunnecessarytoachieveclinicallyrelevantimpingementwearanddamage.Additionally,themagnitude
for axial rotation provided is a starting point for defining test parameters; the user may choose alternate angular limits if justified by other means (e.g., retrieval analysis,
scientific literature, etc.).
there are likely many possible impingement conditions for a for their IVD prosthesis. Prior clinical experience with the
given IVD prosthesis. device design may aid in the development of impingement test
parameters through analysis of device retrievals and radio-
5.2 The user should attempt to determine the clinically
graphs. However, prior clinical experience with the IVD being
relevant and geometrically possible impingement conditions
testedshouldnotbeconsideredasaprerequisiteforperforming
and dictated by the design and impingement wear test param-
impingement testing.
eters that may result in wear and fatigue damage for the IVD
prosthesis. The user should also attempt to select the device
5.4 This guide details a three-step process for assessing
size which will represent a worst case for the impingement
device impingement under a selected set of conditions:
conditions and parameters selected.
5.4.1 The user selects previously identified impingement
conditions, one at a time, or clinically observed conditions.
5.3 Theusershouldreferenceandutilizeexistingsourcesof
information to identify the impingement test parameters that 5.4.2 The user selects the worst-case size of device to apply
produce the clinically relevant impingement wear and damage the selected conditions.
F3295 − 18
FIG. 3 Work Flow for Running the Impingement Test
5.4.3 Solid modeling and the quasistatic test method should offload the bearing surface for each cycle. Given that the
be employed to assess the impingement condition and deter- intended function of the devices is typically to articulate, it
mine the impingement test parameters – most importantly, the may be impossible to fully eliminate Mode 1 wear at the
angular displacement limits to be used in the impingement intended bearing interface.
wear test.
5.8 The point of impingement (POI) is a simplification for
5.4.4 The impingement wear test is then conducted using
the purpose of determining an impingement moment arm and
the impingement test parameters.
thus calculating the theoretical ultimate moment (M). M may
t t
5.5 This guide serves to evaluate devices with various be useful for comparing device designs.
designs, materials (i.e., metal-on-metal versus polymer-on-
5.9 The contribution of axial rotation to impingement dam-
polymer), and stiffness in the impingement region using the
age is still under-studied. However, retrieval analysis has
same axial force and angular displacement control.
provided evidence that it may contribute to impingement
5.5.1 In the case where the device has no limit in a given
damage. Many total disc replacements are unconstrained in
direction or does not allow motion in a given direction, a
axial rotation. Therefore, unlike flexion-extension or lateral
rationale for excluding that condition should be provided (e.g.,
bending where a moment versus angular displacement re-
intended design or function of the device).
sponse can be readily developed, axial rotation will have a
5.6 Impingementoccursoverarangebetweenaninitialand near-zero moment response. The axial rotation parameters
an ultimate angle rather than at a discrete angle and location provided in Section 15 are based on the Mode 1 wear test
because both design (e.g., mobile bearings) and material methods and should be assessed and altered if justification
combinations (e.g., inclusion of polymeric materials) may lead (e.g., wear patterns from retrievals, scientific literature, etc.)
to compliance, deformation and wear, which in turn may lead exists.
toachangeintheangulardisplacementatwhichcontactoccurs
over the course of the test.Arange of angular displacement is 6. Selection of Impingement Conditions
therefore prescribed to ensure that the impingement region is
6.1 Select the relevant impingement conditions in accor-
fully loaded during each impingement cycle.
dance with the guidance provided in this section.
5.7 The suggested test parameters in Table 1 have been 6.1.1 If the IVD prosthesis to be tested has been studied
provided with the objective of minimizing Mode I wear at the clinically and information on impinged devices is reported,
bearing surface while providing sufficient motion to fully these data may be utilized to develop clinically relevant
F3295 − 18
impingement test parameters. Specifically, retrieval and radio- guidanceisprovidedtoassistinselectingtheworst-casedevice
graphic analyses are the two main sources of information once a set of impingement test conditions has been selected:
obtained from the clinical study that may aid in the develop-
6.2.1 Devices are typically produced in a range of sizes or
ment of the test parameters. configurations. Therefore the user should consider which
6.1.1.1 If clinical information is available, radiographs may
device size or configuration would result in the greatest extent
be analyzed to select and justify the impingement test param- and magnitude of wear and damage. To determine the worst
eters. Radiographs from patients for whom devices show casewillrequiretheusertoconsidercontactstressandcontact
probable impingement may provide further insights into the area for the selected impingement conditions and parameters.
conditions that led to impingement. Motion modes that lead to
6.2.2 Select the design and size combination that allows the
impingementmaybenoted.Relativealignmentofcomponents least device ROM for the conditions selected.
(e.g., relative angulation or translation of endplates) may be
6.3 Once a set of impingement conditions and worst case
noted.
device(s) have been selected, the user should determine the
6.1.1.2 If retrievals are available, the objective of the
device range of motion using solid models. The user should
impingementstudyshouldbetoreplicateclinicalimpingement
also define the impingement moment arm and POI and
wear and damage. Clinical retrievals of the IVD prosthesis, if
associated M (see 3.1.5.10 for calculation).
t
available, should be analyzed for evidence of impingement.
6.3.1 Rotate and translate the device virtually using the
Retrievals that demonstrate evidence of impingement provide
models. Specifically, rotate the device from the device neutral
insights into the motion mode(s) most likely to lead to
position until impingement occurs.
impingement. Impingement wear scars on the retrieved device
maybeanalyzedinanattempttodetermineimpingementwear
7. Summary of Quasistatic Test Methods
and damage mechanisms.
7.1 The quasistatic bench test is performed to determine the
6.1.2 Existing clinical data should not be considered as a
responseofthedevicetoincreasingangulardisplacement.The
prerequisite to performing impingement testing. In such cases,
angular displacement limits for the impingement test are
a review of the scientific literature (e.g., [1-4] ), device
determined based on the results of quasistatic testing (Fig. 2).
modeling, and quasistatic testing may need to be more exten-
sive to determine the impingement conditions.
7.2 Two test methods are described in Section 9. The two
6.1.3 Impingement may occur in a variety of conditions
methods are expected to produce reasonably equivalent results
including, but not limited to, flexion, extension, lateral
but have not been studied for direct comparison.The user may
bending, axial rotation, and anterior, posterior or lateral
select either method for testing the quasistatic performance of
translation, shear, and combinations of the aforementioned
theirdevicebasedontheavailabilityoftherequiredequipment
motions.
for each method. Other methods which are reasonably ex-
6.1.3.1 Extension and flexion are most commonly associ-
pected to produce equivalent results are also acceptable.
atedwithimpingement;thusimpingementatboththeposterior
7.3 Bothmethodsareconductedusingloadframesequipped
and anterior aspects of the device should be explored and
with torsional actuators to apply angular rotation. Method 1
considered.
uses fixtures to constrain the device and a torsional actuator to
6.1.3.2 Lateralbendingimpingementhasalsobeenreported
apply angular displacement. Method 2 uses an axial actuator
in the literature. The user should attempt to determine if their
andfixturestoconstrainthedeviceandatorsionalactuatorthat
IVD prosthesis is more susceptible to lateral bending impinge-
isperpendiculartotheaxialactuatortoapplyangulardisplace-
ment and to consider testing in this motion mode if relevant.
ment. Other load frames and fixtures that are reasonably
6.1.3.3 Impingement may not occur in axial rotation for
expected to produce equivalent results are also acceptable.
devices unconstrained in this motion mode, but when coupled
with other motion modes, axial rotation may contribute to
8. Quasistatic Test Specimens
clinically relevant impingement wear and damage.
6.1.3.4 Anterior-posterior and/or lateral translation of com-
8.1 All components of the IVD prosthesis shall be previ-
ponentsshouldbeconsideredandpotentiallyincorporatedinto
ously unused parts only; no implants shall be retested. All
the test method if the device has a mobile core. In addition,
implantsshallbeproductionqualityparts.Anydeviationsfrom
clinicalimpingementmayresultfromtranslationsofthedevice
theintendedmarketedproductshallbenotedinthefinalreport.
endplates with respect to one another (e.g., anterior migration
8.2 It is permissible to exclude features that may either
of the inferior endplate). Such modes should be explored and
interfere with obtaining accurate results, may obstruct or
considered.
hinder proper support or clamping of the device, and/or may
6.1.3.5 Shearmayplayaroleintheimpingementmechanics
otherwise prevent the device from being tested, provided that
of IVDs [5]; thus the user should consider the role of shear in
exclusionofthefeaturedoesnotaltertheresultsofthetest.For
inducing impingement.
example, bone-implant interface features such as coatings or
6.2 The user should attempt to define the worst-case device
keels may be omitted, unless they are relevant to the investi-
size for the selected impingement conditions. The following
gation.
8.3 It is suggested that a sample size of three be used to
performthequasistatictest.Foradditionalguidanceonexperi-
Theboldfacenumbersinparenthesesrefertothelistofreferencesattheendof
this stndard. mental design, see Guides E1488 and E1402.
F3295 − 18
9. Quasistatic Test Apparatus the plane of interest to the point of impingement being
investigated. Passive force control is achieved by the radial
9.1 Test machines shall conform to the requirements of
constraint of the fixtures used in the method.
Practices E4. Specifically, the stiffness of the machine being
9.6.2 The IVD prosthesis is mounted in a torsional test
used for quasistatic testing and eventual impingement wear
frame such that the device COR is coaxial with the Z-axis of
testing should be the same or a justification for why they are
the testing machine’s actuator and force transducer. This
not the same. Ensuring the stiffness of the two machines is
ensures that the plane of interest is perpendicular to the Z-axis
comparable will ensure the quasitatic behavior is comparable
of the testing machine’s actuator and force transducer (Fig. 4).
to the behavior under impingement wear testing.
9.6.3 The device shall be mounted into the test system such
9.2 Eachendplateshallbeplacedinmetalorplasticfixtures.
thatthebearingsurfacesofthedeviceareincontactthroughout
The fixtures shall have surfaces that mate geometrically with
articulationofthedevicefromthedeviceneutralpositionupto
the endplate, fully support the endplate, and have a clamping
the initial impingement angle.
feature that secures the outer perimeter of the endplate to the
fixture.
NOTE 1—It is up to the user to determine the proper fit of the device in
thetestsystem.Inherently,somenominalforceonthe Z-axisofthedevice
9.3 Fixtures shall be designed such that load sharing by the
is necessary to ensure contact of the bearing surfaces.
fixture during impingement is minimized.
9.7 Quasistatic Test Method 2 –Controlled Axial Force:
9.4 The user should make a reasonable effort to ensure that
9.7.1 This setup is designed so that the applied angular
the implant/fixture assembly and its attachment to the test
displacementarticulateswiththedeviceaboutitsCORthrough
systemissufficientlystifftominimizeseparationofthefixtures
the plane of interest to the point of impingement being
from the implant as moment is applied to the impingement
investigated.
region of the device. This may require reinforcing the fixtures
9.7.2 The IVD prosthesis is mounted in an apparatus
in regions of high stress to reduce separation such as a support
equipped to apply a constant axial force perpendicular to a
ring guide and support ring (shown as items 5 and 6,
rotational axis. The rotational axis is controlled in angular
respectively, in Fig. 4).
displacement and monitors torque feedback through an inline
9.5 Quasistatic testing is intended to be conducted at room
torque transducer (Fig. 5).The rotational axis and device COR
temperatureandinair.Theusermaychoosetoconductthistest
are coaxial with the Z-axis of the testing machine’s actuator
undermorephysiologicconditions.Themethodsandexamples
and force transducer used to control the force through the
described below were conducted at room temperature and in
device.
air.
9.7.3 The device shall be mounted into the test system such
9.6 Quasistatic Test Method 1 – Passive Axial Force: thatthebearingsurfacesofthedeviceareincontactthroughout
9.6.1 This setup is designed so that the applied angular articulationofthedevicefromthedeviceneutralpositionupto
displacementarticulateswiththedeviceaboutitsCORthrough the initial impingement angle.
(a) Image of test setup; (b) Diagram of test setup; (c) Cut-section A of diagram shown in (b)
FIG. 4 Example of Test Fixtures and Positioning of Device for Quasistatic Test Method 1
F3295 − 18
(a) Image of test setup; (b) Diagram of test setup; (c) Cut-section A of diagram shown in (b)
FIG. 5 Example of Test Fixtures and Positioning of Device for Quasistatic Test Method 2
impingement fully occurs (A ), the force is shifted to the impingement
9.7.4 Axial force shall be applied in accordance with Table
u
region and the moment will therefore plateau. If there is a difference
1.
between the measured moment at the plateau and the theoretical moment
M, use the measured moment to determine A .
t u
10. Quasistatic Test Procedure
NOTE 4—Asteady increase or decrease of the measured moment from
10.1 Quasistatic Test Method 1 – Passively ControlledAxial thedeviceneutralpositionuptothePOImayindicatethatthedeviceCOR
is not properly aligned with the Z-axis of the test machine.
Force:
10.1.1 Ensure that the IVD prosthesis is in the device
10.3 Quasistatic Test Analyses:
neutralpositioninthe X-Yplane.Thismaybeaccomplishedby
10.3.1 Determine the initial impingement angle by identi-
ensuring that the mounting fixtures are coplanar. Thi
...


Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...