ASTM G28-22
(Test Method)Standard Test Methods for Detecting Susceptibility to Intergranular Corrosion in Wrought, Nickel-Rich, Chromium-Bearing Alloys
Standard Test Methods for Detecting Susceptibility to Intergranular Corrosion in Wrought, Nickel-Rich, Chromium-Bearing Alloys
SIGNIFICANCE AND USE
3.1 The boiling ferric sulfate-sulfuric acid test may be applied to the following alloys in the wrought condition:
Alloy
Testing Time, h
N06007
120
N06022
24
N06030
120
N06059
24
N06200
24
N06455
24
N06600
24
N06625
120
N06686
24
N06985
120
N08020
120
N08367
24
Alloy
Testing Time, h
N08800
120
N08825A
120
N10276
24(A) While the ferric sulfate-sulfuric acid test does detect susceptibility to inter- granular corrosion in Alloy N08825, the boiling 65 % nitric acid test, Practices A262, Practice C, for detecting susceptibility to intergranular corrosion in stainless steels is more sensitive and should be used if the intended service is nitric acid.
3.2 This test method may be used to evaluate as-received material and to evaluate the effects of subsequent heat treatments. In the case of nickel-rich, chromium-bearing alloys, the test method may be applied to wrought and weldments of products. The test method is not applicable to cast products.
SCOPE
1.1 These test methods cover two tests as follows:
1.1.1 Method A, Ferric Sulfate-Sulfuric Acid Test (Sections 3 – 10, inclusive)—This test method describes the procedure for conducting the boiling ferric sulfate—50 % sulfuric acid test which measures the susceptibility of certain nickel-rich, chromium-bearing alloys to intergranular corrosion (see Terminology G193), which may be encountered in certain service environments. The uniform corrosion rate obtained by this test method, which is a function of minor variations in alloy composition, may easily mask the intergranular corrosion components of the overall corrosion rate on alloys N10276, N06022, N06059, and N06455.
1.1.2 Method B, Mixed Acid-Oxidizing Salt Test (Sections 11 – 18, inclusive)—This test method describes the procedure for conducting a boiling 23 % sulfuric + 1.2 % hydrochloric + 1 % ferric chloride + 1 % cupric chloride test which measures the susceptibility of certain nickel-rich, chromium-bearing alloys to display a step function increase in corrosion rate when there are high levels of grain boundary precipitation.
1.2 The purpose of these two test methods is to detect susceptibility to intergranular corrosion as influenced by variations in processing or composition, or both. Materials shown to be susceptible may or may not be intergranularly corroded in other environments. This must be established independently by specific tests or by service experience.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Exception—Some desired corrosion rate units in 8.1.1 are given in inch-pound units.
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Warning statements are given in 5.1.1, 5.1.3, 5.1.9, 13.1.1, and 13.1.11.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Nov-2022
- Technical Committee
- G01 - Corrosion of Metals
- Drafting Committee
- G01.05 - Laboratory Corrosion Tests
Relations
- Effective Date
- 01-May-2013
- Effective Date
- 01-Apr-2010
- Effective Date
- 01-Mar-2008
- Effective Date
- 01-Mar-2006
- Effective Date
- 10-Nov-2002
- Effective Date
- 10-Nov-2002
- Effective Date
- 10-Nov-2002
- Effective Date
- 10-Nov-2002
- Effective Date
- 10-Jun-2002
- Effective Date
- 10-Feb-1999
- Effective Date
- 10-Feb-1999
Overview
ASTM G28-22: Standard Test Methods for Detecting Susceptibility to Intergranular Corrosion in Wrought, Nickel-Rich, Chromium-Bearing Alloys is an internationally recognized standard developed by ASTM International. This standard defines laboratory test methods for evaluating the susceptibility of nickel-rich, chromium-bearing wrought alloys to intergranular corrosion-a type of localized attack that occurs along grain boundaries and can critically impact component durability in corrosive environments.
ASTM G28-22 is primarily used to assess parts made from specific nickel-rich alloys, ensuring their reliability and structural integrity in service environments that may promote intergranular corrosion. The standard includes two methods:
- Method A: Ferric Sulfate-Sulfuric Acid Test
- Method B: Mixed Acid-Oxidizing Salt Test
Key Topics
Test Methods:
- Method A details a procedure using boiling ferric sulfate and 50% sulfuric acid to assess susceptibility to intergranular corrosion in various wrought alloys.
- Method B uses a boiling solution of sulfuric acid, hydrochloric acid, ferric chloride, and cupric chloride to reveal corrosion due to grain boundary precipitation in select alloys.
Applicability:
- Specifically covers wrought, nickel-rich, chromium-bearing alloys (e.g., N06007, N06022, N06030, N06600, N06625, N08020, N08800, N10276, among others).
- Not applicable to cast products; Method B is not intended for weldments.
Testing Times:
- Each alloy is subject to a prescribed exposure time (commonly 24 or 120 hours), depending on the material and method used.
Significance:
- The test methods detect intergranular corrosion as influenced by factors such as heat treatment, alloy composition, or fabrication process.
Reporting:
- Test results should include alloy designation, heat number, chemistry, heat treatment, test method, specimen details, and calculated corrosion rates.
Safety:
- Emphasizes adherence to safety and environmental protocols when handling acids and test solutions.
Applications
ASTM G28-22 is widely used in multiple industries where nickel-rich, chromium-bearing alloys are critical, including chemical processing, oil and gas, power generation, and marine engineering. Key applications include:
Material Qualification:
Ensures new or heat-treated materials meet specified corrosion resistance requirements before fabrication or installation.Process Evaluation:
Assesses the effects of heat treatment or fabrication methods on intergranular corrosion susceptibility of wrought alloys.Quality Control:
Applied in manufacturing environments for batch testing and quality assurance of critical components such as piping, tubing, heat exchangers, and pressure vessels.Failure Analysis:
Supports investigations when field components show signs of intergranular attack, helping to identify causes like improper material processing.
Related Standards
ASTM A262:
Practices for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels, especially useful when nitric acid environments are present.ASTM D1193:
Specification for Reagent Water, referenced for preparing test solutions.Terminology Standards (e.g., ASTM G193):
Definitions pertinent to corrosion and corrosion testing.
Using ASTM G28-22 supports robust material selection, enhances safety, and extends service life by helping engineers and quality professionals identify materials at risk of intergranular corrosion. This proactive approach is essential for compliance, safety, and performance in demanding industrial environments. For more information, refer to the official ASTM documentation and ensure all testing is performed by trained personnel in accordance with the standard’s requirements.
Buy Documents
ASTM G28-22 - Standard Test Methods for Detecting Susceptibility to Intergranular Corrosion in Wrought, Nickel-Rich, Chromium-Bearing Alloys
REDLINE ASTM G28-22 - Standard Test Methods for Detecting Susceptibility to Intergranular Corrosion in Wrought, Nickel-Rich, Chromium-Bearing Alloys
Get Certified
Connect with accredited certification bodies for this standard

Element Materials Technology
Materials testing and product certification.

Inštitut za kovinske materiale in tehnologije
Institute of Metals and Technology. Materials testing, metallurgical analysis, NDT.
Sponsored listings
Frequently Asked Questions
ASTM G28-22 is a standard published by ASTM International. Its full title is "Standard Test Methods for Detecting Susceptibility to Intergranular Corrosion in Wrought, Nickel-Rich, Chromium-Bearing Alloys". This standard covers: SIGNIFICANCE AND USE 3.1 The boiling ferric sulfate-sulfuric acid test may be applied to the following alloys in the wrought condition: Alloy Testing Time, h N06007 120 N06022 24 N06030 120 N06059 24 N06200 24 N06455 24 N06600 24 N06625 120 N06686 24 N06985 120 N08020 120 N08367 24 Alloy Testing Time, h N08800 120 N08825A 120 N10276 24(A) While the ferric sulfate-sulfuric acid test does detect susceptibility to inter- granular corrosion in Alloy N08825, the boiling 65 % nitric acid test, Practices A262, Practice C, for detecting susceptibility to intergranular corrosion in stainless steels is more sensitive and should be used if the intended service is nitric acid. 3.2 This test method may be used to evaluate as-received material and to evaluate the effects of subsequent heat treatments. In the case of nickel-rich, chromium-bearing alloys, the test method may be applied to wrought and weldments of products. The test method is not applicable to cast products. SCOPE 1.1 These test methods cover two tests as follows: 1.1.1 Method A, Ferric Sulfate-Sulfuric Acid Test (Sections 3 – 10, inclusive)—This test method describes the procedure for conducting the boiling ferric sulfate—50 % sulfuric acid test which measures the susceptibility of certain nickel-rich, chromium-bearing alloys to intergranular corrosion (see Terminology G193), which may be encountered in certain service environments. The uniform corrosion rate obtained by this test method, which is a function of minor variations in alloy composition, may easily mask the intergranular corrosion components of the overall corrosion rate on alloys N10276, N06022, N06059, and N06455. 1.1.2 Method B, Mixed Acid-Oxidizing Salt Test (Sections 11 – 18, inclusive)—This test method describes the procedure for conducting a boiling 23 % sulfuric + 1.2 % hydrochloric + 1 % ferric chloride + 1 % cupric chloride test which measures the susceptibility of certain nickel-rich, chromium-bearing alloys to display a step function increase in corrosion rate when there are high levels of grain boundary precipitation. 1.2 The purpose of these two test methods is to detect susceptibility to intergranular corrosion as influenced by variations in processing or composition, or both. Materials shown to be susceptible may or may not be intergranularly corroded in other environments. This must be established independently by specific tests or by service experience. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Exception—Some desired corrosion rate units in 8.1.1 are given in inch-pound units. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Warning statements are given in 5.1.1, 5.1.3, 5.1.9, 13.1.1, and 13.1.11. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 3.1 The boiling ferric sulfate-sulfuric acid test may be applied to the following alloys in the wrought condition: Alloy Testing Time, h N06007 120 N06022 24 N06030 120 N06059 24 N06200 24 N06455 24 N06600 24 N06625 120 N06686 24 N06985 120 N08020 120 N08367 24 Alloy Testing Time, h N08800 120 N08825A 120 N10276 24(A) While the ferric sulfate-sulfuric acid test does detect susceptibility to inter- granular corrosion in Alloy N08825, the boiling 65 % nitric acid test, Practices A262, Practice C, for detecting susceptibility to intergranular corrosion in stainless steels is more sensitive and should be used if the intended service is nitric acid. 3.2 This test method may be used to evaluate as-received material and to evaluate the effects of subsequent heat treatments. In the case of nickel-rich, chromium-bearing alloys, the test method may be applied to wrought and weldments of products. The test method is not applicable to cast products. SCOPE 1.1 These test methods cover two tests as follows: 1.1.1 Method A, Ferric Sulfate-Sulfuric Acid Test (Sections 3 – 10, inclusive)—This test method describes the procedure for conducting the boiling ferric sulfate—50 % sulfuric acid test which measures the susceptibility of certain nickel-rich, chromium-bearing alloys to intergranular corrosion (see Terminology G193), which may be encountered in certain service environments. The uniform corrosion rate obtained by this test method, which is a function of minor variations in alloy composition, may easily mask the intergranular corrosion components of the overall corrosion rate on alloys N10276, N06022, N06059, and N06455. 1.1.2 Method B, Mixed Acid-Oxidizing Salt Test (Sections 11 – 18, inclusive)—This test method describes the procedure for conducting a boiling 23 % sulfuric + 1.2 % hydrochloric + 1 % ferric chloride + 1 % cupric chloride test which measures the susceptibility of certain nickel-rich, chromium-bearing alloys to display a step function increase in corrosion rate when there are high levels of grain boundary precipitation. 1.2 The purpose of these two test methods is to detect susceptibility to intergranular corrosion as influenced by variations in processing or composition, or both. Materials shown to be susceptible may or may not be intergranularly corroded in other environments. This must be established independently by specific tests or by service experience. 1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard. 1.3.1 Exception—Some desired corrosion rate units in 8.1.1 are given in inch-pound units. 1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. Warning statements are given in 5.1.1, 5.1.3, 5.1.9, 13.1.1, and 13.1.11. 1.5 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM G28-22 is classified under the following ICS (International Classification for Standards) categories: 77.120.40 - Nickel, chromium and their alloys. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM G28-22 has the following relationships with other standards: It is inter standard links to ASTM A262-13, ASTM A262-10, ASTM A262-02a(2008), ASTM D1193-06, ASTM A262-02a, ASTM A262-02ae2, ASTM A262-02ae3, ASTM A262-02ae1, ASTM A262-02, ASTM D1193-99e1, ASTM D1193-99. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM G28-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: G28 − 22
Standard Test Methods for
Detecting Susceptibility to Intergranular Corrosion in
Wrought, Nickel-Rich, Chromium-Bearing Alloys
ThisstandardisissuedunderthefixeddesignationG28;thenumberimmediatelyfollowingthedesignationindicatestheyearoforiginal
adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.Asuperscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope 1.4 This standard does not purport to address all of the
safety concerns, if any, associated with its use. It is the
1.1 These test methods cover two tests as follows:
responsibility of the user of this standard to establish appro-
1.1.1 Method A, Ferric Sulfate-Sulfuric Acid Test (Sections
priate safety, health, and environmental practices and deter-
3–10, inclusive)—This test method describes the procedure
mine the applicability of regulatory limitations prior to use.
for conducting the boiling ferric sulfate—50 % sulfuric acid
Warning statements are given in 5.1.1, 5.1.3, 5.1.9, 13.1.1, and
test which measures the susceptibility of certain nickel-rich,
13.1.11.
chromium-bearing alloys to intergranular corrosion (see Ter-
1.5 This international standard was developed in accor-
minology G193), which may be encountered in certain service
dance with internationally recognized principles on standard-
environments. The uniform corrosion rate obtained by this test
ization established in the Decision on Principles for the
method, which is a function of minor variations in alloy
Development of International Standards, Guides and Recom-
composition, may easily mask the intergranular corrosion
mendations issued by the World Trade Organization Technical
components of the overall corrosion rate on alloys N10276,
Barriers to Trade (TBT) Committee.
N06022, N06059, and N06455.
1.1.2 Method B, Mixed Acid-Oxidizing Salt Test (Sections
2. Referenced Documents
11–18, inclusive)—This test method describes the procedure
2.1 ASTM Standards:
for conducting a boiling 23 % sulfuric + 1.2 % hydrochlo-
A262 Practices for Detecting Susceptibility to Intergranular
ric+1% ferric chloride+1% cupric chloride test which
Attack in Austenitic Stainless Steels
measures the susceptibility of certain nickel-rich, chromium-
D1193 Specification for Reagent Water
bearing alloys to display a step function increase in corrosion
G193 Terminology and Acronyms Relating to Corrosion
rate when there are high levels of grain boundary precipitation.
1.2 The purpose of these two test methods is to detect
METHOD A—Ferric Sulfate—Sulfuric Acid Test
susceptibility to intergranular corrosion as influenced by varia-
3. Significance and Use
tionsinprocessingorcomposition,orboth.Materialsshownto
be susceptible may or may not be intergranularly corroded in
3.1 The boiling ferric sulfate-sulfuric acid test may be
other environments.This must be established independently by
applied to the following alloys in the wrought condition:
specific tests or by service experience.
Alloy Testing Time, h
1.3 The values stated in SI units are to be regarded as
N06007 120
standard. No other units of measurement are included in this
N06022 24
N06030 120
standard.
N06059 24
1.3.1 Exception—Some desired corrosion rate units in 8.1.1
N06200 24
are given in inch-pound units.
N06455 24
N06600 24
N06625 120
N06686 24
N06985 120
N08020 120
N08367 24
These test methods are under the jurisdiction of ASTM Committee G01 on
Corrosion of Metals and are the direct responsibility of Subcommittee G01.05 on
Laboratory Corrosion Tests. For referenced ASTM standards, visit the ASTM website, www.astm.org, or
Current edition approved Dec. 1, 2022. Published January 2023. Originally contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
approved in 1971. Last previous edition approved in 2015 as G28–02 (2015). DOI: Standards volume information, refer to the standard’s Document Summary page on
10.1520/G0028-22. the ASTM website.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G28−22
5.1.3 Then measure 236 mL of reagent-grade sulfuric acid
Alloy Testing Time, h
(H SO ) of a concentration which must be in the range from
2 4
N08800 120
95.0 weight percent to 98.0 weight percent in a 250 mL
A
N08825 120
graduate.Add the acid slowly to the water in the flask to avoid
N10276 24
boiling by the heat evolved (Note 3). Externally cooling the
A
While the ferric sulfate-sulfuric acid test does detect susceptibility to inter-
flask with water during the mixing will also reduce overheat-
granular corrosion in Alloy N08825, the boiling 65 % nitric acid test, Practices
ing.
A262,PracticeC,fordetectingsusceptibilitytointergranularcorrosioninstainless
steels is more sensitive and should be used if the intended service is nitric acid.
NOTE 3—Loss of vapor results in concentration of the acid.
3.2 This test method may be used to evaluate as-received
5.1.4 Weigh 25 g of reagent grade ferric sulfate (contains
material and to evaluate the effects of subsequent heat treat-
about 75 % Fe (SO ) (Note 4)) and add to the H SO
2 4 3 2 4
ments. In the case of nickel-rich, chromium-bearing alloys, the
solution. A trip balance may be used.
test method may be applied to wrought and weldments of
products. The test method is not applicable to cast products.
NOTE 4—Ferritic sulfate is a specific additive that establishes and
controls the corrosion potential. Substitutions are not permitted.
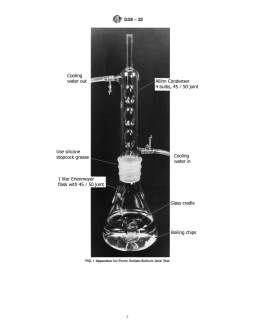
4. Apparatus
5.1.5 Add boiling chips.
4.1 The apparatus (Note 1) is illustrated in Fig. 1. 5.1.6 Lubricate the ground glass of the condenser joint with
4.1.1 Allihn or Soxhlet Condenser, 4-bulb, with a 45/50 silicone grease.
ground-glass joint, overall length about 330 mm, condensing
5.1.7 Cover the flask with the condenser and circulate
section about 240 mm.
cooling water.
4.1.2 Erlenmeyer Flask, 1 L, with a 45/50 ground-glass
5.1.8 Boil the solution until all ferric sulfate is dissolved.
joint. The ground-glass opening shall be 40 mm wide.
5.1.9 Warning—It has been reported that violent boiling
4.1.3 Glass Cradle (Fig. 2)—To pass through the ground-
can occur resulting in acid spills. It is important to ensure that
glass joint on the Erlenmeyer flask, the width of the cradle
theconcentrationofaciddoesnotincreaseandthatanadequate
should not exceed 40 mm and the front-to-back distance must
number of boiling chips (which are resistant to attack by the
be such that the cradle will fit the 40 mm diameter opening. It
test solution) are present.
should have three or four holes to increase circulation of the
test solution around the specimen (Note 2).
6. Test Specimens
NOTE 1—Substitution for this equipment may not be used. The 2
6.1 A specimen having a total surface area of 5 cm to
cold-finger type of standard Erlenmeyer flask may not be used.
20 cm is recommended.
NOTE 2—Other equivalent means of specimen support, such as glass
hooks or stirrups, may also be used.
6.2 The intent is to test a specimen representing as nearly as
4.1.4 Boiling Chips, or some other boiling aids must be
possible the material as used in service. The specimens should
used to prevent bumping.
becuttorepresentthegrainflowdirectionthatwillseeservice,
4.1.5 Silicone Grease, (for example, stopcock grease) is
for example, specimens should not contain cross-sectional
recommended for the ground-glass joint.
areas unless it is the intent of the test to evaluate these. Only
4.1.6 Electrically Heated Hot Plate, or equivalent to pro-
such surface finishing should be performed as is required to
vide heat for continuous boiling of the solution.
remove foreign material and obtain a standard, uniform finish
4.1.7 AnalyticalBalance,capableofweighingtothenearest
as specified in 6.4. For very heavy sections, specimens should
0.001 g.
be maintained to represent the appropriate surface while
maintaining reasonable specimen size for convenience in
5. Test Solution
testing. Ordinarily, removal of more material than necessary
will have little influence on the test results. However, in the
5.1 Prepare 600 mL of 50 % (49.4 % to 50.9 %) solution as
special case of surface decarburization or of carburization (the
follows:
latter is sometimes encountered in tubing when lubricants or
5.1.1 Warning—Protect the eyes and use rubber gloves for
binders containing carbonaceous materials are employed), it
handling acid. Place the test flask under a hood.
may be possible by heavy grinding or machining to remove the
5.1.2 First, measure 400 mL of Type IV reagent water
affected layer completely. Such treatment of test specimens is
(Specification D1193) in a 500 mL graduate and pour into the
not permissible, except in tests undertaken to demonstrate such
flask.
surface effects.
6.3 When specimens are cut by shearing, the deformed
3 material must be removed by machining or grinding to a depth
To avoid frequent chipping of the drip-tip of the condenser during handling, the
modified condenser described by Streicher, M.A., and Sweet,A. J., Corrosion,Vol
equal to the thickness of the specimen to remove cold worked
25, 1969, pp. 1, has been found suitable for this use.
metal.
The sole source of supply of the apparatus known to the committee at this time
is amphoteric alundum Hengar Boiling Granules, available from Hengar Company,
6.4 All surfaces of the specimen, including edges, should be
a division of Henry Troemner, LLC, 201 Wolf Drive, Thorofare, NJ 08086. If you
finished using wet No. 80 grit or dry No. 120 grit abrasive
are aware of alternative suppliers, please provide this information to ASTM
paper. If dry abrasive paper is used, polish slowly to avoid
International Headquarters. Your comments will receive careful consideration at a
meeting of the responsible technical committee, which you may attend. overheating. Sand blasting should not be used.
G28−22
FIG. 1 Apparatus for Ferric Sulfate-Sulfuric Acid Test
G28−22
FIG. 2 Glass Cradle
G28−22
6.5 Residual oxide scale has been observed to cause spuri- where:
ous specimen activation in the test solution. Therefore, the
K = a constant (see 8.1.1),
formation of oxide scale in stamped codes must be prevented,
T = time of exposure, h, to the nearest 0.01 h,
2 2
and all traces of oxide scale formed during heat treatment must A = area, cm , to the nearest 0.01 cm ,
be thoroughly removed prior to stamping identification codes. W = mass loss, g, to the nearest 0.001 g, and
D = density, g/cm (see 8.1.2).
6.6 Thespecimendimensionsshouldbemeasuredincluding
the edges and inner surfaces of any holes and the total exposed
8.1.1 Many different units are used to express corrosion
area calculated.
rates. Using the above units for T, A, W, and D, the corrosion
rate can be calculated in a variety of units with the following
6.7 The specimen should then be degreased using suitable
appropriate value of K:
nonchlorinated agents such as soap and acetone, dried, and
then weighed to the nearest 0.001 g. Constant K in Corrosion Rate
Corrosion Rate Units Desired
A
Equation
7. Procedure
mils per year (mpy) 3.45 × 10
inches per year (ipy) 3.45 × 10
7.1 Place the specimen in the glass cradle, remove the 2
inches per month (ipm) 2.87 × 10
condenser, immerse the cradle by means of a hook in the millimetres per year (mm/Y) 8.76 × 10
micrometres per year (µm/y) 8.76 × 10
actively boiling solution (Fig. 1), and immediately replace the
picometres per second (pm/s) 2.78 × 10
condenser. A fresh solution should be used for each test. 2 4 B
grams per square metre-hour (g/m h) 1.00 × 10 ×D
6 B
milligrams per square decimetre-day (mdd) 2.40 × 10 ×D
7.2 Mark the li
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: G28 − 02 (Reapproved 2015) G28 − 22
Standard Test Methods for
Detecting Susceptibility to Intergranular Corrosion in
Wrought, Nickel-Rich, Chromium-Bearing Alloys
This standard is issued under the fixed designation G28; the number immediately following the designation indicates the year of original
adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript
epsilon (´) indicates an editorial change since the last revision or reapproval.
This standard has been approved for use by agencies of the U.S. Department of Defense.
1. Scope
1.1 These test methods cover two tests as follows:
1.1.1 Method A, Ferric Sulfate-Sulfuric Acid Test (Sections 3 – 10, inclusive)—This test method describes the procedure for
conducting the boiling ferric sulfate—50 % sulfuric acid test which measures the susceptibility of certain nickel-rich,
chromium-bearing alloys to intergranular corrosion (see Terminology G15G193), which may be encountered in certain service
environments. The uniform corrosion rate obtained by this test method, which is a function of minor variations in alloy
composition, may easily mask the intergranular corrosion components of the overall corrosion rate on alloys N10276, N06022,
N06059, and N06455.
1.1.2 Method B, Mixed Acid-Oxidizing Salt Test (Sections 11 – 18, inclusive)—This test method describes the procedure for
conducting a boiling 23 % sulfuric + 1.2 % hydrochloric + 1 % ferric chloride + 1 % cupric chloride test which measures the
susceptibility of certain nickel-rich, chromium-bearing alloys to display a step function increase in corrosion rate when there are
high levels of grain boundary precipitation.
1.2 The purpose of these two test methods is to detect susceptibility to intergranular corrosion as influenced by variations in
processing or composition, or both. Materials shown to be susceptible may or may not be intergranularly corroded in other
environments. This must be established independently by specific tests or by service experience.
1.3 The values stated in SI units are to be regarded as standard. No other units of measurement are included in this standard.
1.3.1 Exception—Some desired corrosion rate units in 8.1.1 are given in inch-pound units.
These test methods are under the jurisdiction of ASTM Committee G01 on Corrosion of Metals and are the direct responsibility of Subcommittee G01.05 on Laboratory
Corrosion Tests.
Current edition approved Nov. 1, 2015Dec. 1, 2022. Published November 2015January 2023. Originally approved in 1971. Last previous edition approved in 20082015
as G28–02 (2008).(2015). DOI: 10.1520/G0028-02R15.10.1520/G0028-22.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
G28 − 22
1.4 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use. Warning statements are given in 5.1.1, 5.1.3, 5.1.9, 13.1.1, and 13.1.11.
1.5 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
A262 Practices for Detecting Susceptibility to Intergranular Attack in Austenitic Stainless Steels
D1193 Specification for Reagent Water
G15G193 Terminology and Acronyms Relating to Corrosion and Corrosion Testing (Withdrawn 2010)
METHOD A—Ferric Sulfate—Sulfuric Acid Test
3. Significance and Use
3.1 The boiling ferric sulfate-sulfuric acid test may be applied to the following alloys in the wrought condition:
Alloy Testing Time, h
N06007 120
N06007 120
N06022 24
N06030 120
N06059 24
N06200 24
N06455 24
N06600 24
N06625 120
N06686 24
N06985 120
N08020 120
N08367 24
Alloy Testing Time, h
N08800 120
A
N08825 120
N10276 24
A
While the ferric sulfate-sulfuric acid test does detect susceptibility to inter- granular corrosion in Alloy N08825, the boiling 65 % nitric acid test, Practices A262, Practice
C, for detecting susceptibility to intergranular corrosion in stainless steels is more sensitive and should be used if the intended service is nitric acid.
3.2 This test method may be used to evaluate as-received material and to evaluate the effects of subsequent heat treatments. In
the case of nickel-rich, chromium-bearing alloys, the test method may be applied to wrought and weldments of products. The test
method is not applicable to cast products.
4. Apparatus
4.1 The apparatus (Note 1) is illustrated in Fig. 1.
4.1.1 Allihn or Soxhlet Condenser, 4-bulb, with a 45/50 ground-glass joint, overall length about 330 mm, condensing section
about 240 mm.
4.1.2 Erlenmeyer Flask, 1-L, 1 L, with a 45/50 ground-glass joint. The ground-glass opening shall be 40 mm wide.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’sstandard’s Document Summary page on the ASTM website.
To avoid frequent chipping of the drip-tip of the condenser during handling, the modified condenser described by Streicher, M. A., and Sweet, A. J., Corrosion, Vol 25,
1969, pp. 1, has been found suitable for this use.
G28 − 22
FIG. 1 Apparatus for Ferric Sulfate-Sulfuric Acid Test
G28 − 22
FIG. 2 Glass Cradle
G28 − 22
4.1.3 Glass Cradle (Fig. 2)—To pass through the ground-glass joint on the Erlenmeyer flask, the width of the cradle should not
exceed 40 mm and the front-to-back distance must be such that the cradle will fit the 40-mm40 mm diameter opening. It should
have three or four holes to increase circulation of the test solution around the specimen (Note 2).
NOTE 1—Substitution for this equipment may not be used. The cold-finger type of standard Erlenmeyer flask may not be used.
NOTE 2—Other equivalent means of specimen support, such as glass hooks or stirrups, may also be used.
4.1.4 Boiling Chips, or or some other boiling aids must be used to prevent bumping.
4.1.5 Silicone Grease, (for example, stopcock grease) is recommended for the ground-glass joint.
4.1.6 Electrically Heated Hot Plate, or equivalent to provide heat for continuous boiling of the solution.
4.1.7 Analytical Balance, capable of weighing to the nearest 0.001 g.
5. Test Solution
5.1 Prepare 600 mL of 50 % (49.4(49.4 % to 50.9 %) solution as follows:
5.1.1 Warning—Protect the eyes and use rubber gloves for handling acid. Place the test flask under a hood.
5.1.2 First, measure 400 mL of Type IV reagent water (Specification D1193) in a 500-mL500 mL graduate and pour into the flask.
5.1.3 Then measure 236 mL of reagent-grade sulfuric acid (H SO ) of a concentration which must be in the range from 95.0
2 4
95.0 weight percent to 98.0 weight percent in a 250-mL250 mL graduate. Add the acid slowly to the water in the flask to avoid
boiling by the heat evolved (Note 3). Externally cooling the flask with water during the mixing will also reduce overheating.
NOTE 3—Loss of vapor results in concentration of the acid.
5.1.4 Weigh 25 g of reagent grade ferric sulfate (contains about 75 % Fe (SO ) (Note 4)) and add to the H SO solution. A trip
2 4 3 2 4
balance may be used.
NOTE 4—Ferritic sulfate is a specific additive that establishes and controls the corrosion potential. Substitutions are not permitted.
5.1.5 Add boiling chips.
5.1.6 Lubricate the ground glass of the condenser joint with silicone grease.
5.1.7 Cover the flask with the condenser and circulate cooling water.
5.1.8 Boil the solution until all ferric sulfate is dissolved.
5.1.9 Warning—It has been reported that violent boiling can occur resulting in acid spills. It is important to ensure that the
concentration of acid does not increase and that an adequate number of boiling chips (which are resistant to attack by the test
solution) are present.
6. Test Specimens
2 2
6.1 A specimen having a total surface area of 55 cm to 20 cm20 cm is recommended.
6.2 The intent is to test a specimen representing as nearly as possible the material as used in service. The specimens should be
The sole source of supply of the apparatus known to the committee at this time is amphoteric alundum Hengar Boiling Granules, available from Hengar Company, a
division of Henry Troemner, LLC, 201 Wolf Drive, Thorofare, NJ 08086. If you are aware of alternative suppliers, please provide this information to ASTM International
Headquarters. Your comments will receive careful consideration at a meeting of the responsible technical committee, which you may attend.
G28 − 22
cut to represent the grain flow direction that will see service, for example, specimens should not contain cross-sectional areas
unless it is the intent of the test to evaluate these. Only such surface finishing should be performed as is required to remove foreign
material and obtain a standard, uniform finish as specified in 6.4. For very heavy sections, specimens should be maintained to
represent the appropriate surface while maintaining reasonable specimen size for convenience in testing. Ordinarily, removal of
more material than necessary will have little influence on the test results. However, in the special case of surface decarburization
or of carburization (the latter is sometimes encountered in tubing when lubricants or binders containing carbonaceous materials
are employed), it may be possible by heavy grinding or machining to remove the affected layer completely. Such treatment of test
specimens is not permissible, except in tests undertaken to demonstrate such surface effects.
6.3 When specimens are cut by shearing, the deformed material must be removed by machining or grinding to a depth equal to
the thickness of the specimen to remove cold worked metal.
6.4 All surfaces of the specimen, including edges, should be finished using wet No. 80-grit80 grit or dry No. 120-grit120 grit
abrasive paper. If dry abrasive paper is used, polish slowly to avoid overheating. Sand blasting should not be used.
6.5 Residual oxide scale has been observed to cause spurious specimen activation in the test solution. Therefore, the formation
of oxide scale in stamped codes must be prevented, and all traces of oxide scale formed during heat treatment must be thoroughly
removed prior to stamping identification codes.
6.6 The specimen dimensions should be measured including the edges and inner surfaces of any holes and the total exposed area
calculated.
6.7 The specimen should then be degreased using suitable nonchlorinated agents such as soap and acetone, dried, and then
weighed to the nearest 0.001 g.0.001 g.
7. Procedure
7.1 Place the specimen in the glass cradle, remove the condenser, immerse the cradle by means of a hook in the actively boiling
solution (Fig. 1), and immediately replace the condenser. A fresh solution should be used for each test.
7.2 Mark the liquid level on the flask with wax crayon to provide a check on vapor loss which would result in concentration of
the acid. If there is an appreciable change in the level (a 0.5-cm0.5 cm or more drop), repeat the test with fresh solution and with
a fresh specimen or a reground specimen.
7.3 Continue immersion of the specimen for the length of time specified in Section 3, then remove the specimen, rinse in water
and acetone, and dry.
7.4 Weigh the specimen and subtract this mass from the original mass.
7.5 Intermediate weighing is not necessary, except as noted in 7.7. The tests can be run without interruption. However, if
preliminary results are desired, the specimen can be removed at any time for weighing.
7.6 Replacement of acid is not necessary during the test periods.
7.7 If the corrosion rate is extraordinarily high in Method A, as evidenced by a change in color (green) of the solution, additional
ferric sulfate must be added during the test. The amount of ferric sulfate that must be added, if the tot
...







Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...