ASTM D3942-19
(Test Method)Standard Test Method for Determination of the Unit Cell Dimension of a Faujasite-Type Zeolite
Standard Test Method for Determination of the Unit Cell Dimension of a Faujasite-Type Zeolite
SIGNIFICANCE AND USE
4.1 Zeolites Y and X, particularly for catalyst and adsorbent applications, are a major article of manufacture and commerce. Catalysts and adsorbents comprising these zeolites in various forms plus binder and other components have likewise become important. Y-based catalysts are used for fluid catalytic cracking (FCC) and hydrocracking of petroleum, while X-based adsorbents are used for desiccation, sulfur compound removal, and air separation.
4.2 The unit cell dimension of a freshly synthesized faujasite-type zeolite is a sensitive measure of composition which, among other uses, distinguishes between the two synthetic faujasite-type zeolites, X and Y. The presence of a matrix in a Y-containing catalyst precludes determination of the zeolite framework composition by direct elemental analysis.
4.3 Users of the test method should be aware that the correlation between framework composition and unit cell dimension is specific to a given cation form of the zeolite. Steam or thermal treatments, for example, may alter both composition and cation form. The user must therefore determine the correlation that pertains to his zeolite containing samples.3 In addition, one may use the test method solely to determine the unit cell dimension, in which case no correlation is needed.
4.4 Other crystalline components may be present in the sample whose diffraction pattern may cause interference with the selected faujasite-structure diffraction peaks. If there is reason to suspect the presence of such components, then a full diffractometer scan should be obtained and analyzed to select faujasite-structure peaks free of interference.
SCOPE
1.1 This test method covers the determination of the unit cell dimension of zeolites having the faujasite crystal structure, including synthetic Y and X zeolites, their modifications such as the various cation exchange forms, and the dealuminized, decationated, and ultra stable forms of Y. These zeolites have cubic symmetry with a unit cell parameter usually within the limits of 24.2 and 25.0 Å (2.42 and 2.50 nm).
1.2 The samples include zeolite preparation in the various forms, and catalysts and adsorbents containing these zeolites. The zeolite may be present in amounts as low as 5 %, such as in a cracking catalyst.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 31-Mar-2019
- Technical Committee
- D32 - Catalysts
- Drafting Committee
- D32.05 - Zeolites
Relations
- Effective Date
- 01-Apr-2019
- Effective Date
- 01-May-2013
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Nov-2005
- Effective Date
- 10-May-1999
- Effective Date
- 01-Apr-2019
Overview
ASTM D3942-19: Standard Test Method for Determination of the Unit Cell Dimension of a Faujasite-Type Zeolite sets the procedure for accurately measuring the unit cell dimension of zeolites with the faujasite crystal structure, including well-known synthetic variants X and Y. Zeolites of the faujasite type are critical as catalysts and adsorbents, widely used in the petrochemical industry and gas purification processes. This standard test method is applicable to zeolite materials in pure form or within catalyst and adsorbent matrixes, even at low concentrations (as low as 5%).
The unit cell dimension is a sensitive indicator of zeolite composition and can help distinguish between different forms and states (such as cation exchange or dealuminization). Accurate measurement supports quality control, product consistency, and proper application of zeolite-based catalysts and adsorbents.
Key Topics
- Scope: Covers both synthetic X and Y zeolites, including their variants and modified forms (cation exchange, dealuminized, decationated, and ultra-stable).
- Methodology: Utilizes X-ray diffraction (XRD) on samples blended with silicon powder as an internal standard to determine the unit cell dimension.
- Sample Types: Applicable to neat zeolite preparations, as well as zeolites incorporated in catalysts and adsorbents.
- Accuracy: Enables repeatability and reproducibility; results are considered reliable if repeated measurements differ by less than 0.02 Å and between-lab tests by less than 0.04 Å.
- Interferences: Advises careful selection of diffraction peaks to avoid interference from other crystalline phases in the sample.
Applications
ASTM D3942-19 is particularly valuable in industries and research domains where precise knowledge of the zeolite unit cell parameter is essential:
- Catalyst Manufacturing: Y-based zeolites are core components for fluid catalytic cracking (FCC) and hydrocracking catalysts in petroleum refining. This method helps discern composition changes due to processing or aging.
- Adsorbent Production: X-based zeolites are crucial for applications such as gas drying (desiccation), removal of sulfur compounds, and high-purity air separation.
- Quality Control: Regular measurement of the unit cell dimension assures batch-to-batch consistency and helps detect unwanted changes in crystal structure or composition.
- Research and Development: Enables identification, characterization, and differentiation of faujasite-type zeolites during synthesis, modification, or performance testing.
Related Standards
- ASTM E691: Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method - cited for evaluating repeatability and reproducibility.
- Other Zeolite Characterization Standards: Consider reference to industry guidance on X-ray diffraction methods and best practices for catalyst analysis.
- International Standardization Principles: Developed in alignment with WTO TBT Committee recommendations, ensuring its applicability and recognition in global trade and regulatory compliance.
Practical Value
This standard test method supports robust, standardized determination of the unit cell parameter for faujasite-type zeolites, providing:
- Reliable quality assurance for catalyst and adsorbent products
- Sensitive distinction between different zeolite types and treatments
- Compatibility with low-content samples, enabling comprehensive analysis of commercial materials
- Industry-recognized methodology that facilitates communication and trade across the zeolite value chain
Keywords: ASTM D3942-19, faujasite-type zeolite, unit cell dimension, X-ray diffraction, zeolite characterization, catalyst quality control, adsorbent analysis, synthetic zeolite, petrochemical catalyst testing.
Buy Documents
ASTM D3942-19 - Standard Test Method for Determination of the Unit Cell Dimension of a Faujasite-Type Zeolite
REDLINE ASTM D3942-19 - Standard Test Method for Determination of the Unit Cell Dimension of a Faujasite-Type Zeolite
Get Certified
Connect with accredited certification bodies for this standard

ECOCERT
Organic and sustainability certification.

Eurofins Food Testing Global
Global leader in food, environment, and pharmaceutical product testing.

Intertek Bangladesh
Intertek certification and testing services in Bangladesh.
Sponsored listings
Frequently Asked Questions
ASTM D3942-19 is a standard published by ASTM International. Its full title is "Standard Test Method for Determination of the Unit Cell Dimension of a Faujasite-Type Zeolite". This standard covers: SIGNIFICANCE AND USE 4.1 Zeolites Y and X, particularly for catalyst and adsorbent applications, are a major article of manufacture and commerce. Catalysts and adsorbents comprising these zeolites in various forms plus binder and other components have likewise become important. Y-based catalysts are used for fluid catalytic cracking (FCC) and hydrocracking of petroleum, while X-based adsorbents are used for desiccation, sulfur compound removal, and air separation. 4.2 The unit cell dimension of a freshly synthesized faujasite-type zeolite is a sensitive measure of composition which, among other uses, distinguishes between the two synthetic faujasite-type zeolites, X and Y. The presence of a matrix in a Y-containing catalyst precludes determination of the zeolite framework composition by direct elemental analysis. 4.3 Users of the test method should be aware that the correlation between framework composition and unit cell dimension is specific to a given cation form of the zeolite. Steam or thermal treatments, for example, may alter both composition and cation form. The user must therefore determine the correlation that pertains to his zeolite containing samples.3 In addition, one may use the test method solely to determine the unit cell dimension, in which case no correlation is needed. 4.4 Other crystalline components may be present in the sample whose diffraction pattern may cause interference with the selected faujasite-structure diffraction peaks. If there is reason to suspect the presence of such components, then a full diffractometer scan should be obtained and analyzed to select faujasite-structure peaks free of interference. SCOPE 1.1 This test method covers the determination of the unit cell dimension of zeolites having the faujasite crystal structure, including synthetic Y and X zeolites, their modifications such as the various cation exchange forms, and the dealuminized, decationated, and ultra stable forms of Y. These zeolites have cubic symmetry with a unit cell parameter usually within the limits of 24.2 and 25.0 Å (2.42 and 2.50 nm). 1.2 The samples include zeolite preparation in the various forms, and catalysts and adsorbents containing these zeolites. The zeolite may be present in amounts as low as 5 %, such as in a cracking catalyst. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 4.1 Zeolites Y and X, particularly for catalyst and adsorbent applications, are a major article of manufacture and commerce. Catalysts and adsorbents comprising these zeolites in various forms plus binder and other components have likewise become important. Y-based catalysts are used for fluid catalytic cracking (FCC) and hydrocracking of petroleum, while X-based adsorbents are used for desiccation, sulfur compound removal, and air separation. 4.2 The unit cell dimension of a freshly synthesized faujasite-type zeolite is a sensitive measure of composition which, among other uses, distinguishes between the two synthetic faujasite-type zeolites, X and Y. The presence of a matrix in a Y-containing catalyst precludes determination of the zeolite framework composition by direct elemental analysis. 4.3 Users of the test method should be aware that the correlation between framework composition and unit cell dimension is specific to a given cation form of the zeolite. Steam or thermal treatments, for example, may alter both composition and cation form. The user must therefore determine the correlation that pertains to his zeolite containing samples.3 In addition, one may use the test method solely to determine the unit cell dimension, in which case no correlation is needed. 4.4 Other crystalline components may be present in the sample whose diffraction pattern may cause interference with the selected faujasite-structure diffraction peaks. If there is reason to suspect the presence of such components, then a full diffractometer scan should be obtained and analyzed to select faujasite-structure peaks free of interference. SCOPE 1.1 This test method covers the determination of the unit cell dimension of zeolites having the faujasite crystal structure, including synthetic Y and X zeolites, their modifications such as the various cation exchange forms, and the dealuminized, decationated, and ultra stable forms of Y. These zeolites have cubic symmetry with a unit cell parameter usually within the limits of 24.2 and 25.0 Å (2.42 and 2.50 nm). 1.2 The samples include zeolite preparation in the various forms, and catalysts and adsorbents containing these zeolites. The zeolite may be present in amounts as low as 5 %, such as in a cracking catalyst. 1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.4 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM D3942-19 is classified under the following ICS (International Classification for Standards) categories: 71.100.40 - Surface active agents. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM D3942-19 has the following relationships with other standards: It is inter standard links to ASTM D3942-03(2013), ASTM E691-13, ASTM E691-11, ASTM E691-08, ASTM E691-05, ASTM E691-99, ASTM D4463/D4463M-19. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM D3942-19 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: D3942 − 19
Standard Test Method for
Determination of the Unit Cell Dimension of a Faujasite-
Type Zeolite
This standard is issued under the fixed designation D3942; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision.Anumber in parentheses indicates the year of last reapproval.A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope dimension is calculated from the X-ray diffraction pattern of
the mixture, using the silicon reflections as a reference.
1.1 This test method covers the determination of the unit
celldimensionofzeoliteshavingthefaujasitecrystalstructure,
4. Significance and Use
including synthetic Y and X zeolites, their modifications such
as the various cation exchange forms, and the dealuminized,
4.1 ZeolitesYandX,particularlyforcatalystandadsorbent
decationated, and ultra stable forms of Y. These zeolites have
applications,areamajorarticleofmanufactureandcommerce.
cubic symmetry with a unit cell parameter usually within the
Catalysts and adsorbents comprising these zeolites in various
limits of 24.2 and 25.0Å (2.42 and 2.50nm).
formsplusbinderandothercomponentshavelikewisebecome
1.2 The samples include zeolite preparation in the various important. Y-based catalysts are used for fluid catalytic crack-
forms, and catalysts and adsorbents containing these zeolites. ing (FCC) and hydrocracking of petroleum, while X-based
The zeolite may be present in amounts as low as 5%, such as adsorbents are used for desiccation, sulfur compound removal,
in a cracking catalyst.
and air separation.
1.3 This standard does not purport to address all of the
4.2 The unit cell dimension of a freshly synthesized
safety concerns, if any, associated with its use. It is the
faujasite-type zeolite is a sensitive measure of composition
responsibility of the user of this standard to establish appro-
which, among other uses, distinguishes between the two
priate safety, health, and environmental practices and deter-
synthetic faujasite-type zeolites, X and Y. The presence of a
mine the applicability of regulatory limitations prior to use.
matrixinaY-containingcatalystprecludesdeterminationofthe
1.4 This international standard was developed in accor-
zeolite framework composition by direct elemental analysis.
dance with internationally recognized principles on standard-
4.3 Users of the test method should be aware that the
ization established in the Decision on Principles for the
Development of International Standards, Guides and Recom- correlation between framework composition and unit cell
mendations issued by the World Trade Organization Technical dimension is specific to a given cation form of the zeolite.
Barriers to Trade (TBT) Committee. Steam or thermal treatments, for example, may alter both
composition and cation form. The user must therefore deter-
2. Referenced Documents
mine the correlation that pertains to his zeolite containing
samples. In addition, one may use the test method solely to
2.1 ASTM Standards:
determinetheunitcelldimension,inwhichcasenocorrelation
E691Practice for Conducting an Interlaboratory Study to
is needed.
Determine the Precision of a Test Method
4.4 Other crystalline components may be present in the
3. Summary of Test Method
sample whose diffraction pattern may cause interference with
3.1 A sample of the zeolite Y or X, or catalyst containing
the selected faujasite-structure diffraction peaks. If there is
zeolite is mixed with powdered silicon. The zeolite unit cell
reason to suspect the presence of such components, then a full
diffractometer scan should be obtained and analyzed to select
faujasite-structure peaks free of interference.
This test method is under the jurisdiction of ASTM Committee D32 on
Catalysts and is the direct responsibility of Subcommittee D32.05 on Zeolites.
Current edition approved April 1, 2019. Published May 2019. Originally
approved in 1980. Last previous edition approved in 2013 as D3942–03(2013). Three correlations have been published for pure synthetic faujasite-type
DOI: 10.1520/D3942-19. zeolites in the sodium or calcium form: Breck, D. W., and Flanigen, E. M.,
For referenced ASTM standards, visit the ASTM website, www.astm.org, or “Molecular Sieves,” Society of Chemical Industry, London, 1968, p. 47; WrightA.
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM C., Rupert, J. P., and Granquist W. T., Amer. Mineral., Vol 53, 1968, p. 1293; and
Standards volume information, refer to the standard’s Document Summary page on Dempsy,E.,Kuehl,G.H.,andOlson,D.H.,JournalofthePhysicalChemistry,Vol
the ASTM website. 73, 1968, p. 387.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3942 − 19
reflections,asforexamplewithequilibriumcatalystscontainingrareearth
5. Apparatus
elements, measure the strong zeolite reflections near 23.5°, 26.9°, and
5.1 X-Ray Diffractometer, able to scan at 0.25° 2θ/min. 2θ
31.2° and the silicon reflection at 28.5° 2θ (Cu Kα).
values in the following discussions were based on data
obtained with a copper tube, although other tubes such as
8. Calculation
molybdenum can be used.
8.1 Correctthemeasuredreflectionanglesforthezeoliteby
NOTE 1—A step-scanning accessory, to scan at a rate of 0.25° or less
adding to each the quantity (calculated minus measured angle
2θ/min, will increase the accuracy of the determination and will facilitate
ofthesiliconreflection).WhenthesiliconreflectionofCuKα
measurement in samples of low zeolite content.
NOTE 2—Modern analysis software might be used for the linear angle radiation is measured, the calculated angle is 56.123° 2θ; with
correction as well.
Cu Kα, the calculated angle is 56.173° 2θ.
5.2 Drying Oven, set at 110°C.
NOTE 8—The corresponding calculated angles when lower angle
5.3 Hydrator, maintained at 35% relative humidity by a reflectionsmustbeusedare28.443°2θ(CuKα )and28.467°2θ(CuKα).
saturated solution of salts such as CaCl ·6H O maintained at
2 2
8.2 Convert the corrected angles of reflection to d-spacing
23°C 6 3°C.
values using the equation:
6. Reagents and Materials
λ
d 5 (1)
hkl
2sinθ
6.1 Siliconpowder,finelygroundorball-milledtoaparticle
diameter less than 5 µm as determined by microscope. NIST
where:
offers a Standard Reference Material (silicon) as an X-ray
d = distance between reflecting planes having the Miller
hkl
internal standard (SMR 640) suitable for powder diffraction
indices
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: D3942 − 03 (Reapproved 2013) D3942 − 19
Standard Test Method for
Determination of the Unit Cell Dimension of a Faujasite-
Type Zeolite
This standard is issued under the fixed designation D3942; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method covers the determination of the unit cell dimension of zeolites having the faujasite crystal structure,
including synthetic Y and X zeolites, their modifications such as the various cation exchange forms, and the dealuminized,
decationated, and ultra stable forms of Y. These zeolites have cubic symmetry with a unit cell parameter usually within the limits
of 24.2 and 25.0 Å 25.0 Å (2.42 and 2.50 nm).2.50 nm).
1.2 The samples include zeolite preparation in the various forms, and catalysts and adsorbents containing these zeolites. The
zeolite may be present in amounts as low as 5 %, such as in a cracking catalyst.
1.3 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety safety, health, and healthenvironmental practices and determine the
applicability of regulatory limitations prior to use.
1.4 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
3. Summary of Test Method
3.1 A sample of the zeolite Y or X, or catalyst containing zeolite is mixed with powdered silicon. The zeolite unit cell dimension
is calculated from the X-ray diffraction pattern of the mixture, using the silicon reflections as a reference.
4. Significance and Use
4.1 Zeolites Y and X, particularly for catalyst and adsorbent applications, are a major article of manufacture and commerce.
Catalysts and adsorbents comprising these zeolites in various forms plus binder and other components have likewise become
important. Y-based catalysts are used for fluid catalytic cracking (FCC) and hydrocracking of petroleum, while X-based adsorbents
are used for desiccation, sulfur compound removal, and air separation.
4.2 The unit cell dimension of a freshly synthesized faujasite-type zeolite is a sensitive measure of composition which, among
other uses, distinguishes between the two synthetic faujasite-type zeolites, X and Y. The presence of a matrix in a Y-containing
catalyst precludes determination of the zeolite framework composition by direct elemental analysis.
4.3 Users of the test method should be aware that the correlation between framework composition and unit cell dimension is
specific to a given cation form of the zeolite. Steam or thermal treatments, for example, may alter both composition and cation
form. The user must therefore determine the correlation that pertains to his zeolite containing samples. In addition, one may use
the test method solely to determine the unit cell dimension, in which case no correlation is needed.
This test method is under the jurisdiction of ASTM Committee D32 on Catalysts and is the direct responsibility of Subcommittee D32.05 on Zeolites.
Current edition approved Dec. 1, 2013April 1, 2019. Published December 2013May 2019. Originally approved in 1980. Last previous edition approved in 20082013 as
D3942 – 03 (2008).(2013). DOI: 10.1520/D3942-03R13.10.1520/D3942-19.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Three correlations have been published for pure synthetic faujasite-type zeolites in the sodium or calcium form: Breck, D. W.W., and Flanigen, E. M. in “ Molecular
M., “Molecular Sieves,” Society of Chemical Industry, , London, 1968, p. 47,47; Wright A. C., Rupert, J. P.P., and Granquist W. T.T., Amer. Mineral., Vol 53, 1968, p. 1293;
and Dempsy, E., Kuehl, G. H., and Olson, D. H., Journal of the Physical Chemistry, Vol 73, 1968, p. 387.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
D3942 − 19
4.4 Other crystalline components may be present in the sample whose diffraction pattern may cause interference with the
selected faujasite-structure diffraction peaks. If there is reason to suspect the presence of such components, then a full
diffractometer scan should be obtained and analyzed to select faujasite-structure peaks free of interference.
5. Apparatus
5.1 X-Ray Diffractometer, able to scan at 0.25° 2θ/min. 2θ values in the following discussions were based on data obtained with
a copper tube, although other tubes such as molybdenum can be used.
NOTE 1—A step-scanning accessory, to scan at a rate of 0.25° or less 2θ/min, will increase the accuracy of the determination and will facilitate
measurement in samples of low zeolite content.
NOTE 2—Modern analysis software might be used for the linear angle correction as well.
5.2 Drying Oven, set at 110°C.110 °C.
5.3 Hydrator, maintained at 35 % relative humidity by a saturated solution of salts such as CaCl ·6H O maintained at
2 2
23°C23 °C 6 3°C.3 °C.
6. Reagents and Materials
6.1 Silicon powder, finely ground or ball-milled to a particle diameter less than 5 μm as determined by microscope. NIST offers
a Standard Reference Material (silicon) as an X-ray internal standard (SMR 640) suitable for powder diffraction measurements.
7. Procedure
7.1 Place about 1.5 g of powdered zeolite sample in the drying oven at 110°C110 °C for 1 h.
NOTE 3—The drying step eliminates excess water from the sample prior to equilibration at constant-humidity hydration. Most catalyst samples, when
received, will not contain excess water. Some sensitive samples may require a lower activation temperature.
7.2 Blend 1 g of powdered zeolite sample with about 0.05 g of silicon in a mortar and grind until intimately mixed. Place a thin
bed of the mixed sample in the hydrator for at least 16 h. Some samples may require a longer equilibration time.
7.3 Pack the hydrated sample in the diffractometer mount.
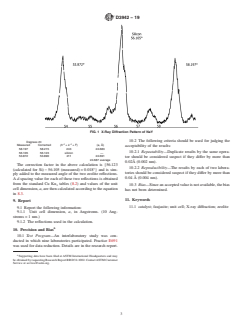
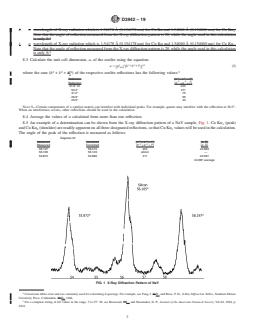
7.4 Determine the X-ray diffraction pattern across the range from 50 to 60° 2θ.
NOTE 4—Smaller slits are desirable for better peak resolution.
NOTE 5—In some catalyst samples, the zeolite reflections at about 53.4° and
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...