ASTM F2638-22
(Test Method)Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier
Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier
SIGNIFICANCE AND USE
6.1 This test method has been developed as a result of research performed by Air Dispersion Limited (Manchester, UK) and funded by the Barrier Test Consortium Limited. The results of this research have been published in a peer-reviewed journal.4 This research demonstrated that testing the barrier performance of porous packaging materials using microorganisms correlates with measuring the filtration efficiency of the materials.
6.2 This test method does not require the use of microbiological method; in addition, the test method can be conducted in a rapid and timely manner.
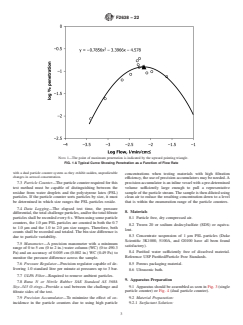
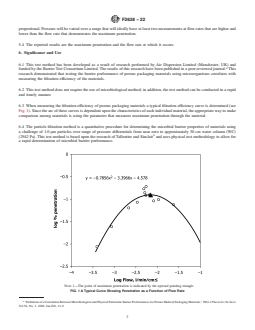
6.3 When measuring the filtration efficiency of porous packaging materials a typical filtration efficiency curve is determined (see Fig. 1). Since the arc of these curves is dependent upon the characteristics of each individual material, the appropriate way to make comparison among materials is using the parameter that measures maximum penetration through the material.
FIG. 1 A Typical Curve Showing Penetration as a Function of Flow Rate
Note 1: The point of maximum penetration is indicated by the upward pointing triangle.
6.4 The particle filtration method is a quantitative procedure for determining the microbial barrier properties of materials using a challenge of 1.0 µm particles over range of pressure differentials from near zero to approximately 30 cm water column (WC) (2942 Pa). This test method is based upon the research of Tallentire and Sinclair4 and uses physical test methodology to allow for a rapid determination of microbial barrier performance.
SCOPE
1.1 This test method measures the aerosol filtration performance of porous packaging materials by creating a defined aerosol of 1.0 μm particles and assessing the filtration efficiency of the material using either single or dual particle counters.
1.2 This test method is applicable to porous materials used to package terminally sterilized medical devices.
1.3 The intent of this test apparatus is to determine the flow rate through a material at which maximum penetration occurs.
1.4 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
General Information
- Status
- Published
- Publication Date
- 30-Apr-2022
- Technical Committee
- F02 - Primary Barrier Packaging
- Drafting Committee
- F02.15 - Chemical/Safety Properties
Relations
- Refers
ASTM E171/E171M-11(2020) - Standard Practice for Conditioning and Testing Flexible Barrier Packaging - Effective Date
- 01-May-2020
- Refers
ASTM E171/E171M-11(2015) - Standard Practice for Conditioning and Testing Flexible Barrier Packaging - Effective Date
- 01-Oct-2015
- Effective Date
- 01-May-2014
- Effective Date
- 01-May-2013
- Effective Date
- 01-May-2013
- Refers
ASTM E171/E171M-11 - Standard Practice for Conditioning and Testing Flexible Barrier Packaging - Effective Date
- 15-Nov-2011
- Effective Date
- 01-Nov-2011
- Effective Date
- 01-Oct-2010
- Effective Date
- 01-Oct-2008
- Effective Date
- 01-Oct-2008
- Refers
ASTM E177-06b - Standard Practice for Use of the Terms Precision and Bias in ASTM Test Methods - Effective Date
- 15-Nov-2006
- Refers
ASTM E177-06a - Standard Practice for Use of the Terms Precision and Bias in ASTM Test Methods - Effective Date
- 01-Nov-2006
- Effective Date
- 01-Nov-2005
- Effective Date
- 01-Nov-2004
- Effective Date
- 01-Nov-2004
Overview
ASTM F2638-22 is the Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier. Issued by ASTM International, this test method establishes a quantitative approach to evaluate the barrier properties of porous materials used in sterile medical device packaging. By utilizing 1.0 μm aerosol particles, it allows rapid, reproducible measurement of filtration efficiency without the need for microbiological methods, supporting efficient sterility assurance in healthcare packaging.
Key Topics
- Porous Packaging Materials: Focuses on materials used for packaging terminally sterilized medical devices, where barrier performance against microbes is critical for patient safety.
- Aerosol Filtration Testing: Employs a physical challenge using 1.0 μm polystyrene latex particles to simulate the barrier effectiveness against microbial ingress.
- Quantitative Analysis: The method uses either single or dual particle counters to determine maximum particle penetration and corresponding flow rates, enabling objective comparisons between materials.
- Test Procedure: Includes step-by-step guidance for apparatus setup, sample preparation, and sequential testing across a range of pressure differentials to generate filtration efficiency curves.
- Statistical Validity: Provides methodologies for consistent repeatability and reproducibility studies, supporting robust quality assurance programs.
Applications
- Sterile Barrier Packaging Validation: This standard is particularly beneficial for manufacturers and laboratories involved in developing or validating materials for sterile packaging of medical devices. It offers a rapid, non-microbiological assessment that correlates well with traditional microbial barrier testing.
- Material Comparison and Quality Control: Test results, specifically maximum penetration values at specified flow rates, serve as critical metrics for comparing different porous materials and confirming ongoing production quality.
- Regulatory Compliance: Use of ASTM F2638-22 supports documented proof of sterility barrier performance-meeting requirements for international and regional regulations around sterile medical devices.
- Research and Development: R&D teams can leverage the standard’s test method to screen new packaging materials and coatings, or to benchmark supplier materials before large-scale adoption.
Related Standards
- ASTM E171/E171M – Practice for Conditioning and Testing Flexible Barrier Packaging.
- ASTM E177 – Practice for Use of the Terms Precision and Bias in ASTM Test Methods.
- ASTM E691 – Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method.
- ISO 5636-3 – Paper and Board-Determination of Air Permeance (Medium Range)-Part 3: Bendtsen Method.
Practical Value
- Faster Results: By avoiding slow microbiological assessments, the method provides timely data to support production and release schedules.
- Standardized Measurements: Enables harmonized barrier performance testing globally, streamlining supplier qualification and regulatory submissions.
- Enhanced Patient Safety: Ensures reliable microbial barrier properties in packaging, directly supporting the sterility of medical devices.
Keywords: ASTM F2638-22, aerosol filtration, porous packaging materials, microbial barrier, sterile barrier packaging, filtration efficiency, medical packaging, particulate barrier, quality control, medical device packaging standards.
Buy Documents
ASTM F2638-22 - Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier
REDLINE ASTM F2638-22 - Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier
Get Certified
Connect with accredited certification bodies for this standard

BRCGS (Brand Reputation Compliance Global Standards)
Global food safety and quality standards owner.
Sponsored listings
Frequently Asked Questions
ASTM F2638-22 is a standard published by ASTM International. Its full title is "Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier". This standard covers: SIGNIFICANCE AND USE 6.1 This test method has been developed as a result of research performed by Air Dispersion Limited (Manchester, UK) and funded by the Barrier Test Consortium Limited. The results of this research have been published in a peer-reviewed journal.4 This research demonstrated that testing the barrier performance of porous packaging materials using microorganisms correlates with measuring the filtration efficiency of the materials. 6.2 This test method does not require the use of microbiological method; in addition, the test method can be conducted in a rapid and timely manner. 6.3 When measuring the filtration efficiency of porous packaging materials a typical filtration efficiency curve is determined (see Fig. 1). Since the arc of these curves is dependent upon the characteristics of each individual material, the appropriate way to make comparison among materials is using the parameter that measures maximum penetration through the material. FIG. 1 A Typical Curve Showing Penetration as a Function of Flow Rate Note 1: The point of maximum penetration is indicated by the upward pointing triangle. 6.4 The particle filtration method is a quantitative procedure for determining the microbial barrier properties of materials using a challenge of 1.0 µm particles over range of pressure differentials from near zero to approximately 30 cm water column (WC) (2942 Pa). This test method is based upon the research of Tallentire and Sinclair4 and uses physical test methodology to allow for a rapid determination of microbial barrier performance. SCOPE 1.1 This test method measures the aerosol filtration performance of porous packaging materials by creating a defined aerosol of 1.0 μm particles and assessing the filtration efficiency of the material using either single or dual particle counters. 1.2 This test method is applicable to porous materials used to package terminally sterilized medical devices. 1.3 The intent of this test apparatus is to determine the flow rate through a material at which maximum penetration occurs. 1.4 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
SIGNIFICANCE AND USE 6.1 This test method has been developed as a result of research performed by Air Dispersion Limited (Manchester, UK) and funded by the Barrier Test Consortium Limited. The results of this research have been published in a peer-reviewed journal.4 This research demonstrated that testing the barrier performance of porous packaging materials using microorganisms correlates with measuring the filtration efficiency of the materials. 6.2 This test method does not require the use of microbiological method; in addition, the test method can be conducted in a rapid and timely manner. 6.3 When measuring the filtration efficiency of porous packaging materials a typical filtration efficiency curve is determined (see Fig. 1). Since the arc of these curves is dependent upon the characteristics of each individual material, the appropriate way to make comparison among materials is using the parameter that measures maximum penetration through the material. FIG. 1 A Typical Curve Showing Penetration as a Function of Flow Rate Note 1: The point of maximum penetration is indicated by the upward pointing triangle. 6.4 The particle filtration method is a quantitative procedure for determining the microbial barrier properties of materials using a challenge of 1.0 µm particles over range of pressure differentials from near zero to approximately 30 cm water column (WC) (2942 Pa). This test method is based upon the research of Tallentire and Sinclair4 and uses physical test methodology to allow for a rapid determination of microbial barrier performance. SCOPE 1.1 This test method measures the aerosol filtration performance of porous packaging materials by creating a defined aerosol of 1.0 μm particles and assessing the filtration efficiency of the material using either single or dual particle counters. 1.2 This test method is applicable to porous materials used to package terminally sterilized medical devices. 1.3 The intent of this test apparatus is to determine the flow rate through a material at which maximum penetration occurs. 1.4 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only. 1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of regulatory limitations prior to use. 1.6 This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
ASTM F2638-22 is classified under the following ICS (International Classification for Standards) categories: 55.040 - Packaging materials and accessories. The ICS classification helps identify the subject area and facilitates finding related standards.
ASTM F2638-22 has the following relationships with other standards: It is inter standard links to ASTM E171/E171M-11(2020), ASTM E171/E171M-11(2015), ASTM E177-14, ASTM E691-13, ASTM E177-13, ASTM E171/E171M-11, ASTM E691-11, ASTM E177-10, ASTM E177-08, ASTM E691-08, ASTM E177-06b, ASTM E177-06a, ASTM E691-05, ASTM E177-06, ASTM E177-04. Understanding these relationships helps ensure you are using the most current and applicable version of the standard.
ASTM F2638-22 is available in PDF format for immediate download after purchase. The document can be added to your cart and obtained through the secure checkout process. Digital delivery ensures instant access to the complete standard document.
Standards Content (Sample)
This international standard was developed in accordance with internationally recognized principles on standardization established in the Decision on Principles for the
Development of International Standards, Guides and Recommendations issued by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
Designation: F2638 − 22
Standard Test Method for
Using Aerosol Filtration for Measuring the Performance of
Porous Packaging Materials as a Surrogate Microbial
Barrier
This standard is issued under the fixed designation F2638; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to
1.1 This test method measures the aerosol filtration perfor-
Determine the Precision of a Test Method
mance of porous packaging materials by creating a defined
2.2 ISO Standard:
aerosol of 1.0 µm particles and assessing the filtration effi-
ISO 5636–3 Paper and Board—Determination of Air Per-
ciency of the material using either single or dual particle
meance (Medium Range)—Part 3: Bendtsen Method
counters.
1.2 This test method is applicable to porous materials used
3. Terminology
to package terminally sterilized medical devices.
3.1 Definitions:
1.3 The intent of this test apparatus is to determine the flow
3.1.1 challenge aerosol—a sufficient quantity of aerosolized
rate through a material at which maximum penetration occurs.
1.0 µm particles that enable effective particle counting in the
1.4 The values stated in SI units are to be regarded as the
filtrate aerosol.
standard. The values given in parentheses are for information
3.1.2 filtrate aerosol—particlesthatremainaerosolizedafter
only.
passage through the test specimen.
1.5 This standard does not purport to address all of the
3.1.3 maximum penetration—the highest percent concentra-
safety concerns, if any, associated with its use. It is the
tionofparticlesinthefiltrateaerosolwhenaspecimenistested
responsibility of the user of this standard to establish appro-
over a range of pressure differentials or air flow rates.
priate safety, health, and environmental practices and deter-
3.2 Abbreviations and Symbols:
mine the applicability of regulatory limitations prior to use.
1.6 This international standard was developed in accor- Symbol Unit Description
C n Average particle count of the challenge aerosol
S
dance with internationally recognized principles on standard-
when using a single particle counter (Method A).
ization established in the Decision on Principles for the
C n Average particle count of the filtrate aerosol.
F
C n Average particle count of the challenge aerosol.
Development of International Standards, Guides and Recom- C
C N Average particle count of the filtrate aerosol prior to
LR
mendations issued by the World Trade Organization Technical
correction for dilution.
Barriers to Trade (TBT) Committee.
R % Percentage of particles from the challenge aerosol
that remain in the filtrate aerosol.
R % The calculated maximum of R.
M
2. Referenced Documents
P cm WC Pressure differential across a test specimen due to
2.1 ASTM Standards: the air flow required by the particle counter.
P cm WC Pressure differential across a test specimen.
E171/E171M Practice for Conditioning and Testing Flexible
F L/m/cm Air flow rate through the test specimen.
Barrier Packaging 2
F L/m/cm Air flow rate required by the particle counter when
E177 Practice for Use of the Terms Precision and Bias in measuring the filtrate aerosol.
F L/m/cm Air flow rate at which maximum penetration occurs.
M
1 4. Safety
This test method is under the jurisdiction ofASTM Committee F02 on Primary
Barrier Packaging and is the direct responsibility of Subcommittee F02.15 on
4.1 The waste and the vacuum venturi vents for the test
Chemical/Safety Properties.
equipment described in this test method emit an aerosol of
Current edition approved May 1, 2022. Published June 2022. Originally
approved in 2007. Last previous edition approved in 2018 as F2638 – 18. DOI: polystyrene particles and salt residues. These aerosols should
10.1520/F2638-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or
contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM
Standards volume information, refer to the standard’s Document Summary page on Available fromAmerican National Standards Institute (ANSI), 25 W. 43rd St.,
the ASTM website. 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2638 − 22
be exhausted from any enclosed environment or collected and journal. This research demonstrated that testing the barrier
filtered to remove all particles. performance of porous packaging materials using microorgan-
isms correlates with measuring the filtration efficiency of the
5. Summary of Test Method
materials.
5.1 A porous packaging material test specimen is placed in
6.2 This test method does not require the use of microbio-
a sample holder in such a way as to create a filter between the
logical method; in addition, the test method can be conducted
challenge and filtrate aerosols. On the challenge side of the
in a rapid and timely manner.
sample holder, an aerosol of particles is presented to the
6.3 When measuring the filtration efficiency of porous
surface of the test specimen. An air flow is generated through
packaging materials a typical filtration efficiency curve is
the test specimen. A laser particle counter is used to monitor
determined (see Fig. 1). Since the arc of these curves is
the particle concentrations in the challenge and filtrate aero-
dependent upon the characteristics of each individual material,
sols. Particle concentrations will be measured over a range of
the appropriate way to make comparison among materials is
flow rates in order to measure the percent penetration over the
using the parameter that measures maximum penetration
range of flow rates and determine the point of maximum
through the material.
penetration.
6.4 Theparticlefiltrationmethodisaquantitativeprocedure
5.2 This test uses an aerosol of polystyrene latex particles
for determining the microbial barrier properties of materials
(PSL) with a geometric mean particle diameter of 1.0 µm and
using a challenge of 1.0 µm particles over range of pressure
a standard deviation of less than 0.05 µm.
differentials from near zero to approximately 30 cm water
5.2.1 A single particle counter may be used to sequentially
column (WC) (2942 Pa). This test method is based upon the
measure the challenge and filtrate aerosols or two particle
research of Tallentire and Sinclair and uses physical test
counters may be used to measure them continuously. When
methodology to allow for a rapid determination of microbial
using a single particle counter the challenge and filtrate
barrier performance.
aerosols will be sequentially measured for each test flow rate.
The filtrate aerosol concentration is reported as the average
7. Apparatus
concentration of the filtrate aerosol over a time period of 45 to
7.1 Test Fixture—This consists of a base with associated
60 s, beginning no sooner than 1 min from the start of the
valves, tubing, sample holder and clamps necessary to perform
filtrate aerosol measurement. The challenge aerosol concentra-
the test. Dimensions of the sample holder (Fig. 2) and
tion is reported as the average concentration of the challenge
schematics of the single particle counter (Fig. 3) and dual
aerosol over a time period of not less than 45 s, beginning no
particlecounter(Fig.4)areshown.Thesignificantcomponents
soonerthan1minfromthestartofthechallengemeasurement.
of the text fixture include:
Challenge concentrations measured immediately before and
7.1.1 Sample Holder—This consists of two assemblies,
after each filtrate concentration measurement are averaged to
which form identical upper and lower manifolds and sample
determine the challenge concentration for a given flow rate.
cavities that deliver a uniform flow of the aerosol or sweep air
5.2.2 When using two particle counters, the challenge and
to the periphery of the test specimen while extracting it from
filtrate aerosols are counted continuously by dedicated particle
the center. To provide consistent sample material placement in
counters. The challenge and filtrate aerosol concentrations are
the sample holder, a meshed fixture is placed under the sample
reported as the average concentration of the challenge or
prior to the closing of the test fixture. Based on the fixture
filtrate aerosol over a time period of not less than 45 s,
dimensions in Fig. 2; the mesh shall have a diameter of less
beginning no sooner than 1 min after a change in flow rate.
than 100 mm and greater than 90 mm to fit into the flow cavity.
5.3 At the pressures used in this test, pressure differential
Amesh material of stainless steel T304; eight (8 by 8 per 2.54
across the sample and flow rate through the material are
cm) mesh T304 stainless steel 0.7112 mm wire diameter; 2.46
directly proportional. Pressure will be varied over a range that
mm opening size; 1.4224 mm overall thickness; weave type
will ideally have at least two measurements at flow rates that
PSW mill finish or equivalent is recommended. The same
are higher and lower than the flow rate that demonstrates the
fixture should be used, or the fixture noted, when comparing
maximum penetration.
data.
5.4 The reported results are the maximum penetration and 7.1.2 Normal Flow Range Needle Valve, 500 µm diameter
the flow rate at which it occurs. maximum orifice.
7.1.3 Low Flow Range Critical Orifice, 40 µm orifice.
6. Significance and Use
7.2 Aerosol Generator—Aconventional vertical style medi-
6.1 This test method has been developed as a result of
cal nebulizer is the preferred aerosol generator for use in a
research performed by Air Dispersion Limited (Manchester,
single counter system (Particle Measuring Systems PG100 or
UK) and funded by the Barrier Test Consortium Limited. The
equivalent).
results of this research have been published in a peer-reviewed NOTE 1—Atomizer style nebulizers are not recommended unless used
“Definition of a Correlation Between Microbiological and Physical Particulate
Barrier Performances for Porous Medical Packaging Materials,” PDA J Pharm Sci
Technol, Vol 56, No. 1, 2002, Jan-Feb, 11-9.
F2638 − 22
NOTE 1—The point of maximum penetration is indicated by the upward pointing triangle.
FIG. 1 A Typical Curve Showing Penetration as a Function of Flow Rate
with a dual particle counter system as they exhibit sudden, unpredictable
concentrations when testing materials with high filtration
changes in aerosol concentration.
efficiency, the use of precision accumulators may be needed.A
7.3 Particle Counter—The particle counter required for this precision accumulator is an inline vessel with a pre-determined
test method must be capable of distinguishing between the volume sufficiently large enough to pull a representative
residue from water droplets and the polystyrene latex (PSL) sample of the particle stream. The sample is then diluted using
particles. If the particle counter sorts particles by size, it must clean air to reduce the resulting concentration down to a level
be determined in which size ranges the PSL particles reside. that is within the enumeration range of the particle counters.
7.4 Data Logging—The elapsed test time, the pressure
8. Materials
differential, the total challenge particles, and/or the total filtrate
particles shall be recorded every 6 s.When using some particle
8.1 Particle free, dry compressed air.
counters, the 1.0 µm PSL particles are counted in both the 0.7
8.2 Tween 20 or sodium dodecylsulfate (SDS) or equiva-
to 1.0 µm and the 1.0 to 2.0 µm size ranges. Therefore, both
lent.
counts shall be recorded and totaled. The bin size difference is
8.3 Concentrate suspension of 1 µm PSL particles (Duke
due to particle variability.
Scientific 3K1000, 5100A, and G0100 have all been found
7.5 Manometer—A precision manometer with a minimum
satisfactory).
range of 0 to 5 cm (0 to 2 in.) water column (WC) (0 to 490.3
8.4 Purified water sufficiently free of dissolved material.
Pa) and an accuracy of 0.005 cm (0.002 in.) WC (0.49 Pa) to
Reference USP Purified/Particle Free Standards.
monitor the pressure difference across the sample.
8.5 Porous packaging material.
7.6 Pressure Regulator—Precision regulator capable of de-
livering 1.0 standard litre per minute at pressures up to 3 bar.
8.6 Ultrasonic bath.
7.7 ULPA Filter—Required to remove ambient particles.
9. Apparatus Preparation
7.8 Buna N or Nitrile Rubber SAE Standard AS 568A
9.1 Apparatus should be assembled as seen in Fig. 3 (single
Size–345 O-rings—Provide a seal between the challenge and
filtrate sides of the test. particle counter) or Fig. 4 (dual particle counter).
9.2 Material Preparation:
7.9 Precision Accumulator—To minimize the effect of co-
incidence in the particle counters due to using high particle 9.2.1 Surfactant Solution:
F2638 − 22
NOTE 1—Dimensions of the cavity in mm. The configuration of the top and bottom cavity is identical.
FIG. 2 Dimensions of the Sample Cavity
9.2.1.1 Prepare a 0.02 % v/v solution of surfactant (Tween 9.2.2.1 Prepare a suspension of 1 µm PSL particles in the
20, SDS, or equivalent) in purified water daily or as needed.
surfactant solution described above.
The solution should be sonicated before use to ensure the
NOTE 3— When making the suspension from a highly concentrated
elimination of aggregates of particles.
source (such as Duke Scientific 5100A) some of the particles will have
agglomerated into aggregates consisting of multiple particles. To ensure
NOTE 2—It is important to eliminate bubbling in the preparation as the
the aerosol consists of particles having only one PSL particle, place the
bubbles may interfere with the particle counts. If sonication results in
bottle containing the solution in an ultrasonic bath for 15 s. This will
excessive bubbling, mixing by other methods is acceptable.
disassociate the particles.
9.2.1.2 Aerosolize the surfactant solution and determine the
9.2.2.2 Check for particle concentration by monitoring
particle size distribution of this solution by measuring the
counts in particle counter for 1 min without any sample in
challenge aerosol. Ideally there should be no particles over 0.7
sample holder.The resulting challenge aerosol particle concen-
µm in diameter detected for the solution only. This does not
tration must be within the range of 200 to 8000 particles per cc
take into consideration the 1 µm challenge particles.The aim is
(e.g., this is equal to 600 to 24 000 counts per 6-s interval in
no more than 2 such particles detected within any 6-s period.
some particle counters).
Monitor surfactant solution for 1 min.
9.2.1.3 Table 1 is an example of the size distribution of 9.2.2.3 Check for instrument bias by measuring the chal-
surfactant solution suitable for use, each row being a 6-s lenge counts with a test specimen in place. Then remove the
counting interval. specimen and measure filtrate results. Check that the counts
9.2.2 Particle Suspension: differ by no more than 3 %.
F2638 − 22
FIG. 3 Equipment Configuration for a Single Particle Counter—Method A
NOTE 4—If concentrations higher than 8000 particles per cc are used,
the O-ring in the lower half of the sample holder. The sample
there will be significant errors due to coincidence (counting two particles
must cover the entire circumference of the seal O-ring. Before
as a single particle) in the particle counter detector.
the sample is placed in the holder, cover the flow cavity area
with a mesh material as described in 7.1.1. Critical dimensions
10. Sample Preparation
of the exposure chamber are shown in Fig. 2.
10.1 The handling of test materials and environmental
exposureimmediatelypriortotestingshouldbeconsideredand
11. Test Procedures
noted if necessary. If sample materials are pre-conditioned,
reference Practice E171/E171M.
11.1 Method A Single Particle Counter—Procedure when
using a single particle counter. Fig. 5 shows an example of the
10.2 Cut a sample of porous barrier material no less than
particle count results of a typical single measurement with
120 mm (the area of the sample exposed to the aerosol is 100
mm in diameter) in any dimension so that it completely covers readings every 6 s.
F2638 − 22
FIG. 4 Equipment Configuration for Dual Particle Counters—Method B
TABLE 1 Example of Particle Counts Generated from 0.02 %
Therefore, an estimate must be made of the challenge aerosol
Surfactant in Acceptably Purified Water
concentration at the time of the filtrate measurement.
0.1 0.2 0.3 0.4 0.5 0.7 1.0 2.0
11.1.2 Setupequipmentfor1particlecountermode,use0.7
µm µm µm µm µm µm µm µm
to 1.0 µm and 1.0 to 2.0 µm bin data, record manometer data
852 176 36 19 4000
every 6 s. Record pressure drop across sample during each 6-s
879 179 45 15 2100
808 155 38 12 1000 sample length while counting particles in filtrate stream.
802 176 37 14 2000
11.1.3 Testdistilledwater/surfactanttoensurewaterisclean
828 178 37 14 1000
as described in 9.2.1.
11.1.4 Prepare appropriate concentration (200 to 8000
particles/mL) of PSL suspension and confirm that the particle
counts are within 3 % as described in 9.2.2.3.
11.1.1 When only a single particle counter is in use, it must 11.1.5 Open sample holder, place mesh on flow cavity, then
be switched between the challenge and filtrate aerosol. place sample in the sample holder.
F2638 − 22
FIG. 5 Example of Data Produced from a Single Measurement Utilizing a Single Particle Counter
11.1.6 Select Low Flow range. Changing flow rates from particles. If maximum penetration value has not been reached,
lowtohighversushightolowavoidsthepoten
...
This document is not an ASTM standard and is intended only to provide the user of an ASTM standard an indication of what changes have been made to the previous version. Because
it may not be technically possible to adequately depict all changes accurately, ASTM recommends that users consult prior editions as appropriate. In all cases only the current version
of the standard as published by ASTM is to be considered the official document.
Designation: F2638 − 18 F2638 − 22
Standard Test Method for
Using Aerosol Filtration for Measuring the Performance of
Porous Packaging Materials as a Surrogate Microbial
Barrier
This standard is issued under the fixed designation F2638; the number immediately following the designation indicates the year of
original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A
superscript epsilon (´) indicates an editorial change since the last revision or reapproval.
1. Scope
1.1 This test method measures the aerosol filtration performance of porous packaging materials by creating a defined aerosol of
1.0 μm particles and assessing the filtration efficiency of the material using either single or dual particle counters.
1.2 This test method is applicable to porous materials used to package terminally sterilized medical devices.
1.3 The intent of this test apparatus is to determine the flow rate through a material at which maximum penetration occurs.
1.4 The values stated in SI units are to be regarded as the standard. The values given in parentheses are for information only.
1.5 This standard does not purport to address all of the safety concerns, if any, associated with its use. It is the responsibility
of the user of this standard to establish appropriate safety, health, and environmental practices and determine the applicability of
regulatory limitations prior to use.
1.6 This international standard was developed in accordance with internationally recognized principles on standardization
established in the Decision on Principles for the Development of International Standards, Guides and Recommendations issued
by the World Trade Organization Technical Barriers to Trade (TBT) Committee.
2. Referenced Documents
2.1 ASTM Standards:
E171/E171M Practice for Conditioning and Testing Flexible Barrier Packaging
E177 Practice for Use of the Terms Precision and Bias in ASTM Test Methods
E691 Practice for Conducting an Interlaboratory Study to Determine the Precision of a Test Method
2.2 ISO Standard:
ISO 5636–3 Paper and Board—Determination of Air Permeance (Medium Range)—Part 3: Bendtsen Method
3. Terminology
3.1 Definitions:
This test method is under the jurisdiction of ASTM Committee F02 on Primary Barrier Packaging and is the direct responsibility of Subcommittee F02.15 on
Chemical/Safety Properties.
Current edition approved Sept. 1, 2018May 1, 2022. Published September 2018June 2022. Originally approved in 2007. Last previous edition approved in 20122018 as
ɛ1
F2638 – 12F2638 – 18. . DOI: 10.1520/F2638-18.10.1520/F2638-22.
For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards
volume information, refer to the standard’s Document Summary page on the ASTM website.
Available from American National Standards Institute (ANSI), 25 W. 43rd St., 4th Floor, New York, NY 10036, http://www.ansi.org.
Copyright © ASTM International, 100 Barr Harbor Drive, PO Box C700, West Conshohocken, PA 19428-2959. United States
F2638 − 22
3.1.1 challenge aerosol—a sufficient quantity of aerosolized 1.0 μm particles that enable effective particle counting in the filtrate
aerosol.
3.1.2 filtrate aerosol—particles that remain aerosolized after passage through the test specimen.
3.1.3 maximum penetration—the highest percent concentration of particles in the filtrate aerosol when a specimen is tested over
a range of pressure differentials or air flow rates.
3.2 Abbreviations and Symbols:
Symbol Unit Description
C n Average particle count of the challenge aerosol
S
when using a single particle counter (Method A).
C n Average particle count of the filtrate aerosol.
F
C n Average particle count of the challenge aerosol.
C
C N Average particle count of the filtrate aerosol prior to
LR
correction for dilution.
R % Percentage of particles from the challenge aerosol
that remain in the filtrate aerosol.
R % The calculated maximum of R.
M
P cm WC Pressure differential across a test specimen due to
the air flow required by the particle counter.
P cm WC Pressure differential across a test specimen.
F L/m/cm Air flow rate through the test specimen.
F L/m/cm Air flow rate required by the particle counter when
measuring the filtrate aerosol.
F L/m/cm Air flow rate at which maximum penetration occurs.
M
4. Safety
4.1 The waste and the vacuum venturi vents for the test equipment described in this test method emit an aerosol of polystyrene
particles and salt residues. These aerosols should be exhausted from any enclosed environment or collected and filtered to remove
all particles.
5. Summary of Test Method
5.1 A porous packaging material test specimen is placed in a sample holder in such a way as to create a filter between the challenge
and filtrate aerosols. On the challenge side of the sample holder, an aerosol of particles is presented to the surface of the test
specimen. An air flow is generated through the test specimen. A laser particle counter is used to monitor the particle concentrations
in the challenge and filtrate aerosols. Particle concentrations will be measured over a range of flow rates in order to measure the
percent penetration over the range of flow rates and determine the point of maximum penetration.
5.2 This test uses an aerosol of polystyrene latex particles (PSL) with a geometric mean particle diameter of 1.0 μm and a standard
deviation of less than 0.05 μm.
5.2.1 A single particle counter may be used to sequentially measure the challenge and filtrate aerosols or two particle counters may
be used to measure them continuously. When using a single particle counter the challenge and filtrate aerosols will be sequentially
measured for each test flow rate. The filtrate aerosol concentration is reported as the average concentration of the filtrate aerosol
over a time period of 45 to 60 s, beginning no sooner than 1 min from the start of the filtrate aerosol measurement. The challenge
aerosol concentration is reported as the average concentration of the challenge aerosol over a time period of not less than 45 s,
beginning no sooner than 1 min from the start of the challenge measurement. Challenge concentrations measured immediately
before and after each filtrate concentration measurement are averaged to determine the challenge concentration for a given flow
rate.
5.2.2 When using two particle counters, the challenge and filtrate aerosols are counted continuously by dedicated particle counters.
The challenge and filtrate aerosol concentrations are reported as the average concentration of the challenge or filtrate aerosol over
a time period of not less than 45 s, beginning no sooner than 1 min after a change in flow rate.
5.3 At the pressures used in this test, pressure differential across the sample and flow rate through the material are directly
F2638 − 22
proportional. Pressure will be varied over a range that will ideally have at least two measurements at flow rates that are higher and
lower than the flow rate that demonstrates the maximum penetration.
5.4 The reported results are the maximum penetration and the flow rate at which it occurs.
6. Significance and Use
6.1 This test method has been developed as a result of research performed by Air Dispersion Limited (Manchester, UK) and
funded by the Barrier Test Consortium Limited. The results of this research have been published in a peer-reviewed journal. This
research demonstrated that testing the barrier performance of porous packaging materials using microorganisms correlates with
measuring the filtration efficiency of the materials.
6.2 This test method does not require the use of microbiological method; in addition, the test method can be conducted in a rapid
and timely manner.
6.3 When measuring the filtration efficiency of porous packaging materials a typical filtration efficiency curve is determined (see
Fig. 1). Since the arc of these curves is dependent upon the characteristics of each individual material, the appropriate way to make
comparison among materials is using the parameter that measures maximum penetration through the material.
6.4 The particle filtration method is a quantitative procedure for determining the microbial barrier properties of materials using
a challenge of 1.0 μm particles over range of pressure differentials from near zero to approximately 30 cm water column (WC)
(2942 Pa). This test method is based upon the research of Tallentire and Sinclair and uses physical test methodology to allow for
a rapid determination of microbial barrier performance.
NOTE 1—The point of maximum penetration is indicated by the upward pointing triangle.
FIG. 1 A Typical Curve Showing Penetration as a Function of Flow Rate
“Definition of a Correlation Between Microbiological and Physical Particulate Barrier Performances for Porous Medical Packaging Materials,” PDA J Pharm Sci Technol,
Vol 56, No. 1, 2002, Jan-Feb, 11-9.
F2638 − 22
7. Apparatus
7.1 Test Fixture—This consists of a base with associated valves, tubing, sample holder and clamps necessary to perform the test.
Dimensions of the sample holder (Fig. 2) and schematics of the single particle counter (Fig. 3) and dual particle counter (Fig. 4)
are shown. The significant components of the text fixture include:
7.1.1 Sample Holder—This consists of two assemblies, which form identical upper and lower manifolds and sample cavities that
deliver a uniform flow of the aerosol or sweep air to the periphery of the test specimen while extracting it from the center. To
provide consistent sample material placement in the sample holder, a meshed fixture is placed under the sample prior to the closing
of the test fixture. Based on the fixture dimensions in Fig. 2; the mesh shall have a diameter of less than 100 mm and greater than
90 mm to fit into the flow cavity. A mesh material of stainless steel T304; eight (8 by 8 per 2.54 cm) mesh T304 stainless steel
0.7112 mm wire diameter; 2.46 mm opening size; 1.4224 mm overall thickness; weave type PSW mill finish or equivalent is
recommended. The same fixture should be used, or the fixture noted, when comparing data.
7.1.2 Normal Flow Range Needle Valve, 500 μm diameter maximum orifice.
7.1.3 Low Flow Range Critical Orifice, 40 μm orifice.
NOTE 1—Dimensions of the cavity in mm. The configuration of the top and bottom cavity is identical.
FIG. 2 Dimensions of the Sample Cavity
F2638 − 22
FIG. 3 Equipment Configuration for a Single Particle Counter—Method A
7.2 Aerosol Generator—A conventional vertical style medical nebulizer is the preferred aerosol generator for use in a single
counter system (Particle Measuring Systems PG100 or equivalent).
NOTE 1—Atomizer style nebulizers are not recommended unless used with a dual particle counter system as they exhibit sudden, unpredictable changes
in aerosol concentration.
7.3 Particle Counter—The particle counter required for this test method must be capable of distinguishing between the residue
from water droplets and the polystyrene latex (PSL) particles. If the particle counter sorts particles by size, it must be determined
in which size ranges the PSL particles reside.
7.4 Data Logging—The elapsed test time, the pressure differential, the total challenge particles, and/or the total filtrate particles
F2638 − 22
FIG. 4 Equipment Configuration for Dual Particle Counters—Method B
shall be recorded every 6 s. When using some particle counters, the 1.0 μm PSL particles are counted in both the 0.7 to 1.0 μm
and the 1.0 to 2.0 μm size ranges. Therefore, both counts shall be recorded and totaled. The bin size difference is due to particle
variability.
7.5 Manometer—A precision manometer with a minimum range of 0 to 5 cm (0 to 2 in.) water column (WC) (0 to 490.3 Pa) and
an accuracy of 0.005 cm (0.002 in.) WC (0.49 Pa) to monitor the pressure difference across the sample.
7.6 Pressure Regulator—Precision regulator capable of delivering 1.0 standard litre per minute at pressures up to 3 bar.
7.7 ULPA Filter—Required to remove ambient particles.
F2638 − 22
7.8 Buna N or Nitrile Rubber SAE Standard AS 568A Size–345 O-rings—Provide a seal between the challenge and filtrate sides
of the test.
7.9 Precision Accumulator—To minimize the effect of coincidence in the particle counters due to using high particle
concentrations when testing materials with high filtration efficiency, the use of precision accumulators may be needed. A precision
accumulator is an inline vessel with a pre-determined volume sufficiently large enough to pull a representative sample of the
particle stream. The sample is then diluted using clean air to reduce the resulting concentration down to a level that is within the
enumeration range of the particle counters.
8. Materials
8.1 Particle free, dry compressed air.
8.2 Tween 20 or sodium dodecylsulfate (SDS) or equivalent.
8.3 Concentrate suspension of 1 μm PSL particles (Duke Scientific 3K1000, 5100A, and G0100 have all been found satisfactory).
8.4 Purified water sufficiently free of dissolved material. Reference USP Purified/Particle Free Standards.
8.5 Porous packaging material.
8.6 Ultrasonic bath.
9. Apparatus Preparation
9.1 Apparatus should be assembled as seen in Fig. 3 (single particle counter) or Fig. 4 (dual particle counter).
9.2 Material Preparation:
9.2.1 Surfactant Solution:
9.2.1.1 Prepare a 0.02 % v/v solution of surfactant (Tween 20, SDS, or equivalent) in purified water daily or as needed. The
solution should be sonicated before use to ensure the elimination of aggregates of particles.
NOTE 2—It is important to eliminate bubbling in the preparation as the bubbles may interfere with the particle counts. If sonication results in excessive
bubbling, mixing by other methods is acceptable.
9.2.1.2 Aerosolize the surfactant solution and determine the particle size distribution of this solution by measuring the challenge
aerosol. Ideally there should be no particles over 0.7 μm in diameter detected for the solution only. This does not take into
consideration the 1 μm challenge particles. The aim is no more than 2 such particles detected within any 6-s period. Monitor
surfactant solution for 1 min.
9.2.1.3 Table 1 is an example of the size distribution of surfactant solution suitable for use, each row being a 6-s counting interval.
9.2.2 Particle Suspension:
TABLE 1 Example of Particle Counts Generated from 0.02 %
Surfactant in Acceptably Purified Water
0.1 0.2 0.3 0.4 0.5 0.7 1.0 2.0
μm μm μm μm μm μm μm μm
852 176 36 19 4 0 0 0
879 179 45 15 2 1 0 0
808 155 38 12 1 0 0 0
802 176 37 14 2 0 0 0
828 178 37 14 1 0 0 0
F2638 − 22
9.2.2.1 Prepare a suspension of 1 μm PSL particles in the surfactant solution described above.
NOTE 3— When making the suspension from a highly concentrated source (such as Duke Scientific 5100A) some of the particles will have agglomerated
into aggregates consisting of multiple particles. To ensure the aerosol consists of particles having only one PSL particle, place the bottle containing the
solution in an ultrasonic bath for 15 s. This will disassociate the particles.
9.2.2.2 Check for particle concentration by monitoring counts in particle counter for 1 min without any sample in sample holder.
The resulting challenge aerosol particle concentration must be within the range of 200 to 8000 particles per cc (e.g., this is equal
to 600 to 24 000 counts per 6-s interval in some particle counters).
9.2.2.3 Check for instrument bias by measuring the challenge counts with a test specimen in place. Then remove the specimen
and measure filtrate results. Check that the counts differ by no more than 3 %.
NOTE 4—If concentrations higher than 8000 particles per cc are used, there will be significant errors due to coincidence (counting two particles as a single
particle) in the particle counter detector.
10. Sample Preparation
10.1 The handling of test materials and environmental exposure immediately prior to testing should be considered and noted if
necessary. If sample materials are pre-conditioned, reference Practice E171/E171M.
10.2 Cut a sample of porous barrier material no less than 120 mm (the area of the sample exposed to the aerosol is 100 mm in
diameter) in any dimension so that it completely covers the O-ring in the lower half of the sample holder. The sample must cover
the entire circumference of the seal O-ring. Before the sample is placed in the holder, cover the flow cavity area with a mesh
material as described in 7.1.1. Critical dimensions of the exposure chamber are shown in Fig. 2.
11. Test Procedures
11.1 Method A Single Particle Counter—Procedure when using a single particle counter. Fig. 5 shows an example of the particle
count results of a typical single measurement with readings every 6 s.
11.1.1 When only a single particle counter is in use, it must be switched between the challenge and filtrate aerosol. Therefore, an
estimate must be made of the challenge aerosol concentration at the time of the filtrate measurement.
11.1.2 Set up equipment for 1 particle counter mode, use 0.7 to 1.0 μm and 1.0 to 2.0 μm bin data, record manometer data every
6 s. Record pressure drop across sample during each 6-s sample length while counting particles in filtrate stream.
11.1.3 Test distilled water/surfactant to ensure water is clean as described in 9.2.1.
11.1.4 Prepare appropriate concentration (200 to 8000 particles/mL) of PSL suspension and confirm that the particle counts are
within 3 % as described in 9.2.2.3.
11.1.5 Open sample holder, place mesh on flow cavity, then place sample in the sample holder.
11.1.6 Select Low Flow range. Changing flow rates from low to high versus high to low avoids the potential for blocking and
blinding of sample materials, which can lead to misleading results.
11.1.7 Start aerosol flow, set particle counter to Count Challenge.
11.1.8 Close the venturi needle valve and increase inlet air pressure to 3 bar, open the needle valve until pressure differential across
the sample is 2 cm WC. Allow system to stabilize for at least 1 min before collecting challenge counts for no less than 45 s (45
to 60 s). Set the particle counter to Filtrate. Allow the system to stabilize for at least 2 min before collecting filtrate counts for no
less than 45 s (45 to 60 s) and record pressure. Set the particle counter back to Challenge, allow the system to stabilize for at least
1 min before collecting challenge counts again for no less than 45 s (45 to 60 s).
11.1.9 Adjust the venturi needle valve to increase the pressure differential across the sample by a factor of 2. If challenge particles
have not just been counted, collect data for no less than 45 s (45 to 60 s). Set the particle counter to Filtrate. Allow the system
F2638 − 22
FIG. 5 Example of Data Produced from a Single Measurement Utilizing a Single Particle Counter
to stabilize for at least 2 min before collecting filtrate counts for no less than 45 s (45 to 60 s) and record pressure. Set the particle
counter back to Challenge, allow the system to stabilize for at least 1 min before collecting challenge counts again for no less than
45 s (45 to 60 s). Continue to increase pressure differential by a factor of 2 until a maximum penetration value has been detected
or the venturi needle valve is closed. The pressure differential may be adjusted to the next value during the second count of
challenge particles. If maximum penetration va
...






Questions, Comments and Discussion
Ask us and Technical Secretary will try to provide an answer. You can facilitate discussion about the standard in here.
Loading comments...